Abstract
The mod5-1 mutation is a nuclear mutation in Saccharomyces cerevisiae that reduces the biosynthesis of N6-(delta 2-isopentenyl)adenosine in both cytoplasmic and mitochondrial tRNAs to less than 1.5% of wild-type levels. The tRNA modification enzyme, delta 2-isopentenyl pyrophosphate:tRNA isopentenyl transferase, cannot be detected in vitro with extracts from mod5-1 cells. A characterization of the MOD5 gene would help to determine how the same enzyme activity in different cellular compartments can be abolished by a single nuclear mutation. To that end we have cloned the MOD5 gene and shown that it restores delta 2-isopentenyl pyrophosphate:tRNA isopentenyl transferase activity and N6-(delta 2-isopentenyl)adenosine to tRNA in both the mitochondria and the nucleus/cytoplasm compartments of mod5-1 yeast cells. That MOD5 sequences are expressed in Escherichia coli and can complement an N6-(delta 2-isopentenyl)-2-methylthioadenosine-deficient E. coli mutant leads us to conclude that MOD5 is the structural gene for delta 2-isopentenyl pyrophosphate:tRNA isopentenyl transferase.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beltzer J. P., Chang L. F., Hinkkanen A. E., Kohlhaw G. B. Structure of yeast LEU4. The 5' flanking region contains features that predict two modes of control and two productive translation starts. J Biol Chem. 1986 Apr 15;261(11):5160–5167. [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlson M., Botstein D. Two differentially regulated mRNAs with different 5' ends encode secreted with intracellular forms of yeast invertase. Cell. 1982 Jan;28(1):145–154. doi: 10.1016/0092-8674(82)90384-1. [DOI] [PubMed] [Google Scholar]
- Chiu A. O., Suyama Y. Immunologic studies on intracellular isoenzymes; the mitochondrial and cytoplasmic leucyl-tRNA synthetases. Biochim Biophys Acta. 1973 Apr 11;299(4):557–563. doi: 10.1016/0005-2787(73)90227-x. [DOI] [PubMed] [Google Scholar]
- Chiu A. O., Suyama Y. The absence of structural relationship between mitochondrial and mitochondrial and cytoplasmic leucyl-tRNA synthetases from Tetrahymena pyriformis. Arch Biochem Biophys. 1975 Nov;171(1):43–54. doi: 10.1016/0003-9861(75)90005-3. [DOI] [PubMed] [Google Scholar]
- Davis R. W., Thomas M., Cameron J., St John T. P., Scherer S., Padgett R. A. Rapid DNA isolations for enzymatic and hybridization analysis. Methods Enzymol. 1980;65(1):404–411. doi: 10.1016/s0076-6879(80)65051-4. [DOI] [PubMed] [Google Scholar]
- Doonan S., Barra D., Bossa F. Structural and genetic relationships between cytosolic and mitochondrial isoenzymes. Int J Biochem. 1984;16(12):1193–1199. doi: 10.1016/0020-711x(84)90216-7. [DOI] [PubMed] [Google Scholar]
- Eisenberg S. P., Yarus M., Soll L. The effect of an Escherichia coli regulatory mutation on transfer RNA structure. J Mol Biol. 1979 Nov 25;135(1):111–126. doi: 10.1016/0022-2836(79)90343-7. [DOI] [PubMed] [Google Scholar]
- Ellis S. R., Morales M. J., Li J. M., Hopper A. K., Martin N. C. Isolation and characterization of the TRM1 locus, a gene essential for the N2,N2-dimethylguanosine modification of both mitochondrial and cytoplasmic tRNA in Saccharomyces cerevisiae. J Biol Chem. 1986 Jul 25;261(21):9703–9709. [PubMed] [Google Scholar]
- Faye G., Sor F. Analysis of mitochondrial ribosomal proteins of Saccharomyces cerevisiae by two dimensional polyacrylamide gel electrophoresis. Mol Gen Genet. 1977 Sep 21;155(1):27–34. doi: 10.1007/BF00268557. [DOI] [PubMed] [Google Scholar]
- Fittler F., Kline L. K., Hall R. H. N6-(Delta 2-isopentenyl)adenosine: biosynthesis in vitro by an enzyme extract from yeast and rat liver. Biochem Biophys Res Commun. 1968 May 23;31(4):571–576. doi: 10.1016/0006-291x(68)90516-0. [DOI] [PubMed] [Google Scholar]
- Geuskens M., Hardt N., Pedrali-Noy G., Spadari S. An autoradiographic demonstration of nuclear DNA replication by DNA polymerase alpha and of mitochondrial DNA synthesis by DNA polymerase gamma. Nucleic Acids Res. 1981 Apr 10;9(7):1599–1613. doi: 10.1093/nar/9.7.1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinnen A., Hicks J. B., Fink G. R. Transformation of yeast. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1929–1933. doi: 10.1073/pnas.75.4.1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holloway P. W., Popják G. The purification of 3,3-dimethylallyl- and geranyl-transferase and of isopentenyl pyrophosphate isomerase from pig liver. Biochem J. 1967 Jul;104(1):57–70. doi: 10.1042/bj1040057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes D. S., Quigley M. A rapid boiling method for the preparation of bacterial plasmids. Anal Biochem. 1981 Jun;114(1):193–197. doi: 10.1016/0003-2697(81)90473-5. [DOI] [PubMed] [Google Scholar]
- Hopper A. K., Furukawa A. H., Pham H. D., Martin N. C. Defects in modification of cytoplasmic and mitochondrial transfer RNAs are caused by single nuclear mutations. Cell. 1982 Mar;28(3):543–550. doi: 10.1016/0092-8674(82)90209-4. [DOI] [PubMed] [Google Scholar]
- Hopper A. K., Schultz L. D., Shapiro R. A. Processing of intervening sequences: a new yeast mutant which fails to excise intervening sequences from precursor tRNAs. Cell. 1980 Mar;19(3):741–751. doi: 10.1016/s0092-8674(80)80050-x. [DOI] [PubMed] [Google Scholar]
- Ito H., Fukuda Y., Murata K., Kimura A. Transformation of intact yeast cells treated with alkali cations. J Bacteriol. 1983 Jan;153(1):163–168. doi: 10.1128/jb.153.1.163-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobsen H., Epstein D. A., Friedmann R. M., Safer B., Torrence P. F. Double-stranded RNA-dependent phosphorylation of protein P1 and eukaryotic initiation factor 2 alpha does not correlate with protein synthesis inhibition in a cell-free system from interferon-treated mouse L cells. Proc Natl Acad Sci U S A. 1983 Jan;80(1):41–45. doi: 10.1073/pnas.80.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston S. A., Hopper J. E. Isolation of the yeast regulatory gene GAL4 and analysis of its dosage effects on the galactose/melibiose regulon. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6971–6975. doi: 10.1073/pnas.79.22.6971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laten H. M., Timmons R. M., Suid S. An antisuppressor mutant of Saccharomyces cerevisiae deficient in isopentenylated tRNA has reduced delta 2-isopentenylpyrophosphate: tRNA-delta 2-isopentenyl transferase activity. FEBS Lett. 1985 Jan 7;179(2):307–310. doi: 10.1016/0014-5793(85)80540-8. [DOI] [PubMed] [Google Scholar]
- Laten H., Gorman J., Bock R. M. Isopentenyladenosine deficient tRNA from an antisuppressor mutant of Saccharomyces cerevisiae. Nucleic Acids Res. 1978 Nov;5(11):4329–4342. doi: 10.1093/nar/5.11.4329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maniatis T., Jeffrey A., Kleid D. G. Nucleotide sequence of the rightward operator of phage lambda. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1184–1188. doi: 10.1073/pnas.72.3.1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin N. C., Hopper A. K. Isopentenylation of both cytoplasmic and mitochondrial tRNA is affected by a single nuclear mutation. J Biol Chem. 1982 Sep 25;257(18):10562–10565. [PubMed] [Google Scholar]
- McDonell M. W., Simon M. N., Studier F. W. Analysis of restriction fragments of T7 DNA and determination of molecular weights by electrophoresis in neutral and alkaline gels. J Mol Biol. 1977 Feb 15;110(1):119–146. doi: 10.1016/s0022-2836(77)80102-2. [DOI] [PubMed] [Google Scholar]
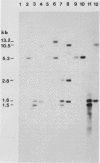
- Najarian D., Dihanich M. E., Martin N. C., Hopper A. K. DNA sequence and transcript mapping of MOD5: features of the 5' region which suggest two translational starts. Mol Cell Biol. 1987 Jan;7(1):185–191. doi: 10.1128/mcb.7.1.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasmyth K. A., Tatchell K. The structure of transposable yeast mating type loci. Cell. 1980 Mar;19(3):753–764. doi: 10.1016/s0092-8674(80)80051-1. [DOI] [PubMed] [Google Scholar]
- Natsoulis G., Hilger F., Fink G. R. The HTS1 gene encodes both the cytoplasmic and mitochondrial histidine tRNA synthetases of S. cerevisiae. Cell. 1986 Jul 18;46(2):235–243. doi: 10.1016/0092-8674(86)90740-3. [DOI] [PubMed] [Google Scholar]
- Orr-Weaver T. L., Szostak J. W., Rothstein R. J. Yeast transformation: a model system for the study of recombination. Proc Natl Acad Sci U S A. 1981 Oct;78(10):6354–6358. doi: 10.1073/pnas.78.10.6354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson R. L., Weiss J. F., Kelmers A. D. Improved separation of transfer RNA's on polychlorotrifuoroethylene-supported reversed-phase chromatography columns. Biochim Biophys Acta. 1971 Feb 11;228(3):770–774. doi: 10.1016/0005-2787(71)90748-9. [DOI] [PubMed] [Google Scholar]
- Schatz G., Butow R. A. How are proteins imported into mitochondria? Cell. 1983 Feb;32(2):316–318. doi: 10.1016/0092-8674(83)90450-6. [DOI] [PubMed] [Google Scholar]
- Schatz G. How mitochondria import proteins from the cytoplasm. FEBS Lett. 1979 Jul 15;103(2):203–211. doi: 10.1016/0014-5793(79)81328-9. [DOI] [PubMed] [Google Scholar]
- Schneller J. M., Schneller C., Martin R., Stahl A. J. Nuclear origin of specific yeast mitochondrial aminoacyl-tRNA synthetases. Nucleic Acids Res. 1976 May;3(5):1151–1165. doi: 10.1093/nar/3.5.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Senapathy P., Jacob M. T. Identification and purification of tRNAs containing N6-(delta 2-isopentenyl) adenosine using antibodies specific for N6-(delta-isopentenyl) adenosine. J Biol Chem. 1981 Nov 25;256(22):11580–11584. [PubMed] [Google Scholar]
- Smolar N., Svensson I. Transfer RNA methylating activity of yeast mitochondria. Nucleic Acids Res. 1974 May;1(5):707–718. doi: 10.1093/nar/1.5.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villa-Komaroff L., Efstratiadis A., Broome S., Lomedico P., Tizard R., Naber S. P., Chick W. L., Gilbert W. A bacterial clone synthesizing proinsulin. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3727–3731. doi: 10.1073/pnas.75.8.3727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakian V. A., Scott J. F. Construction, replication, and chromatin structure of TRP1 RI circle, a multiple-copy synthetic plasmid derived from Saccharomyces cerevisiae chromosomal DNA. Mol Cell Biol. 1982 Mar;2(3):221–232. doi: 10.1128/mcb.2.3.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Loon A. P., Young E. T. Intracellular sorting of alcohol dehydrogenase isoenzymes in yeast: a cytosolic location reflects absence of an amino-terminal targeting sequence for the mitochondrion. EMBO J. 1986 Jan;5(1):161–165. doi: 10.1002/j.1460-2075.1986.tb04191.x. [DOI] [PMC free article] [PubMed] [Google Scholar]