Abstract
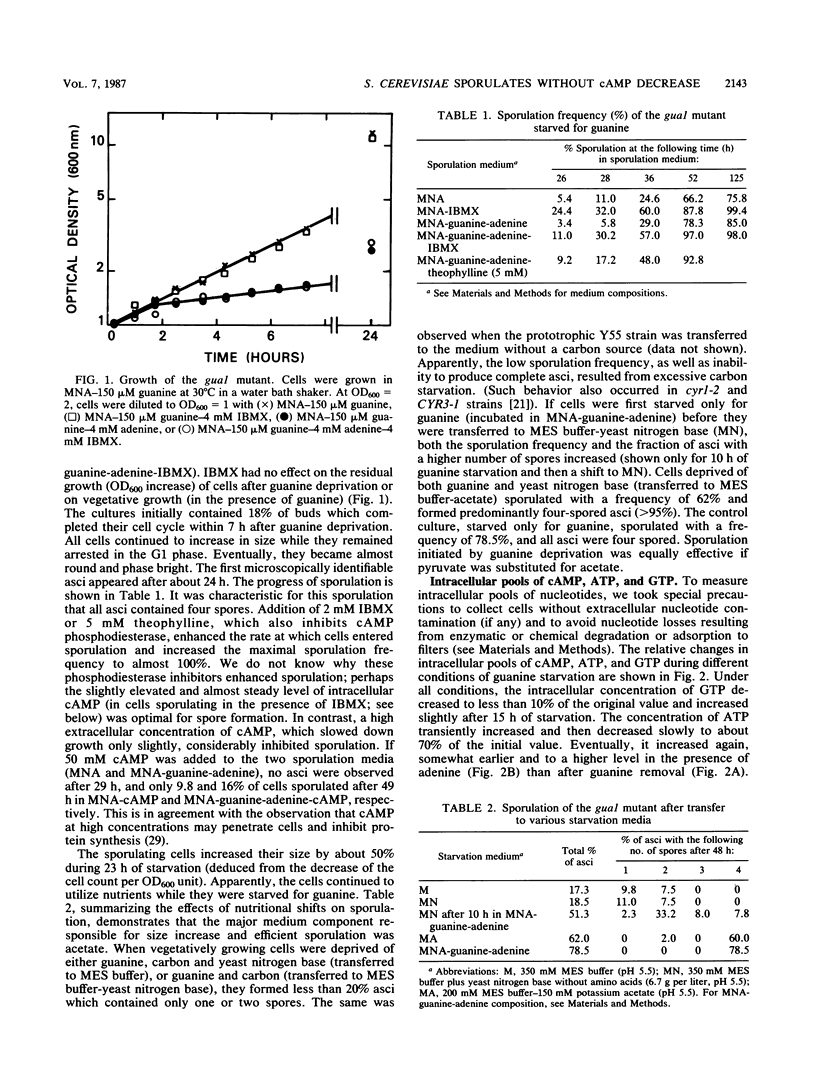
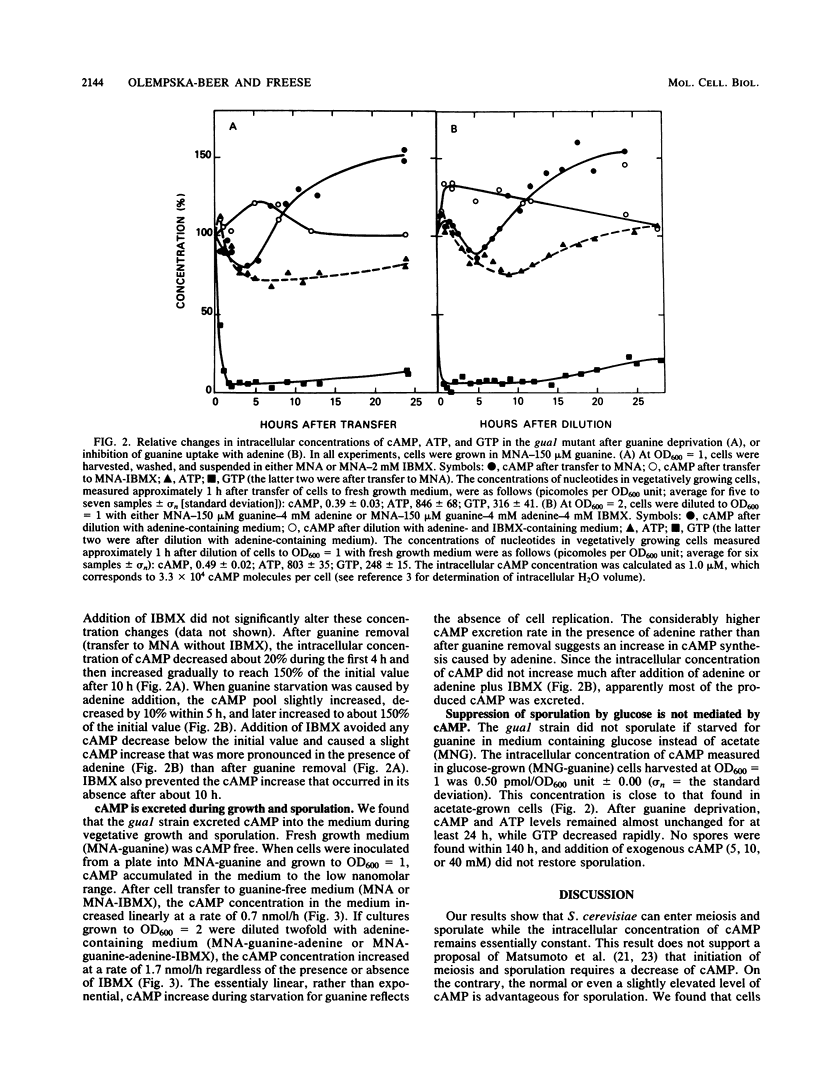
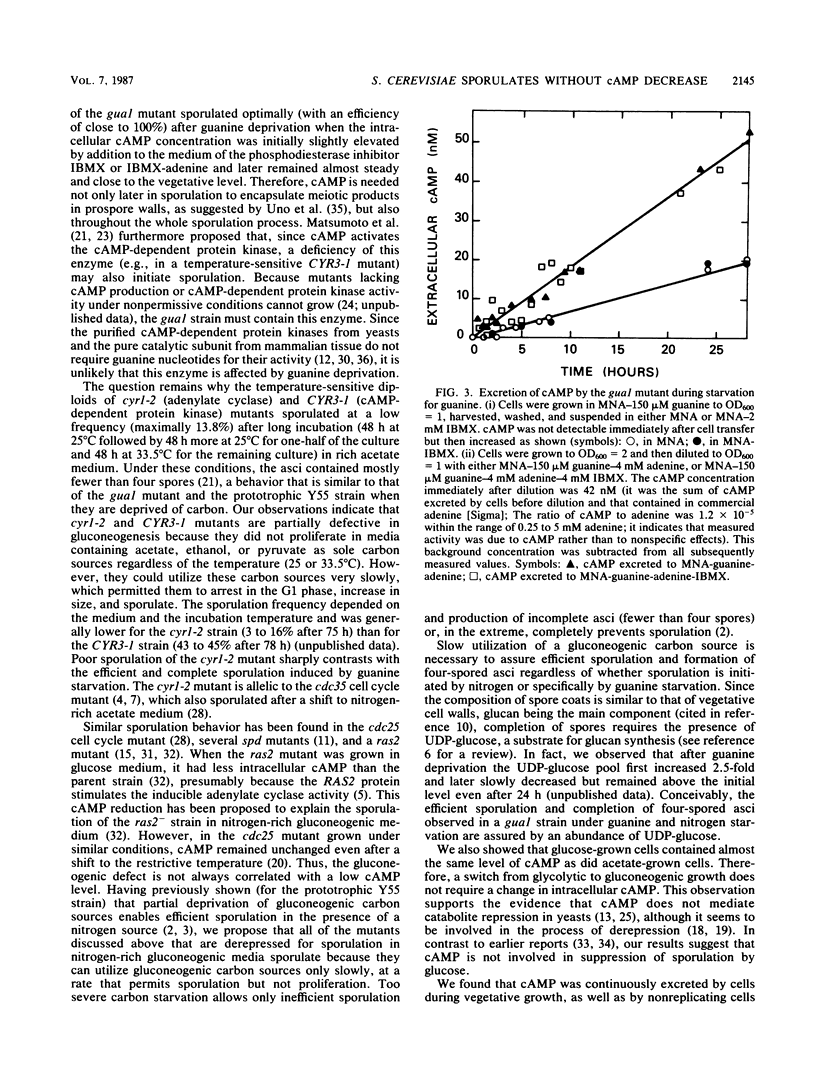
Meiosis and sporulation of Saccharomyces cerevisiae are initiated in a guanine auxotroph by guanine deprivation (E. Bautz Freese, Z. Olempska-Beer, A. Hartig, and E. Freese, Dev. Biol. 102:438-451, 1984). We used this condition to examine a hypothesis (K. Matsumoto, I. Uno, and T. Ishikawa, Cell 32:417-423, 1983) that initiation of meiosis requires a low level of cAMP. We found that, after guanine deprivation, the intracellular concentration of cAMP transiently decreased not more than 20% and not at all if the cAMP phosphodiesterase inhibitor 3-isobutyl-1-methylxanthine (IBMX) was added to the medium. Under these conditions, at least 76% of the cells sporulated in the absence of IBMX, and almost 100% sporulated in its presence. The sporulating cells continually excreted cAMP and utilized the gluconeogenic carbon source. The cells failed to sporulate efficiently and to form four-spored asci if simultaneously deprived of guanine and carbon. After guanine deprivation in glucose medium, sporulation remained suppressed and intracellular cAMP was unchanged. We conclude that, under conditions of guanine starvation, cAMP deficiency is not required for initiation of meiosis and sporulation, cAMP is produced in excess and excreted to the medium, the cells sporulate better if the cAMP concentration is increased by addition of IBMX, the cells require a gluconeogenic carbon source for complete and efficient sporulation, and suppression of sporulation by glucose is not mediated by cAMP.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boutelet F., Petitjean A., Hilger F. Yeast cdc35 mutants are defective in adenylate cyclase and are allelic with cyr1 mutants while CAS1, a new gene, is involved in the regulation of adenylate cyclase. EMBO J. 1985 Oct;4(10):2635–2641. doi: 10.1002/j.1460-2075.1985.tb03981.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broek D., Samiy N., Fasano O., Fujiyama A., Tamanoi F., Northup J., Wigler M. Differential activation of yeast adenylate cyclase by wild-type and mutant RAS proteins. Cell. 1985 Jul;41(3):763–769. doi: 10.1016/s0092-8674(85)80057-x. [DOI] [PubMed] [Google Scholar]
- Cabib E., Roberts R., Bowers B. Synthesis of the yeast cell wall and its regulation. Annu Rev Biochem. 1982;51:763–793. doi: 10.1146/annurev.bi.51.070182.003555. [DOI] [PubMed] [Google Scholar]
- Casperson G. F., Walker N., Bourne H. R. Isolation of the gene encoding adenylate cyclase in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1985 Aug;82(15):5060–5063. doi: 10.1073/pnas.82.15.5060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casperson G. F., Walker N., Brasier A. R., Bourne H. R. A guanine nucleotide-sensitive adenylate cyclase in the yeast Saccharomyces cerevisiae. J Biol Chem. 1983 Jul 10;258(13):7911–7914. [PubMed] [Google Scholar]
- Dawes I. W., Calvert G. R. Initiation of sporulation in Saccharomyces cerevisiae. Mutations causing derepressed sporulation and G1 arrest in the cell division cycle. J Gen Microbiol. 1984 Mar;130(3):605–613. doi: 10.1099/00221287-130-3-605. [DOI] [PubMed] [Google Scholar]
- Eraso P., Gancedo J. M. Catabolite repression in yeasts is not associated with low levels of cAMP. Eur J Biochem. 1984 May 15;141(1):195–198. doi: 10.1111/j.1432-1033.1984.tb08174.x. [DOI] [PubMed] [Google Scholar]
- Fraenkel D. G. On ras gene function in yeast. Proc Natl Acad Sci U S A. 1985 Jul;82(14):4740–4744. doi: 10.1073/pnas.82.14.4740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freese E. B., Chu M. I., Freese E. Initiation of yeast sporulation of partial carbon, nitrogen, or phosphate deprivation. J Bacteriol. 1982 Mar;149(3):840–851. doi: 10.1128/jb.149.3.840-851.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freese E. B., Olempska-Beer Z., Hartig A., Freese E. Initiation of meiosis and sporulation of Saccharomyces cerevisiae by sulfur or guanine deprivation. Dev Biol. 1984 Apr;102(2):438–451. doi: 10.1016/0012-1606(84)90209-4. [DOI] [PubMed] [Google Scholar]
- Gopalan G., Rajamanickam C. Heme-mediated effect of cAMP on mitochondriogenesis during glucose repression-derepression in Saccharomyces cerevisiae. Indian J Biochem Biophys. 1985 Aug;22(4):214–217. [PubMed] [Google Scholar]
- Mahler H. R., Lin C. C. Exogenous adenosine 3': 5'-monophosphate can release yeast from catabolite repression. Biochem Biophys Res Commun. 1978 Aug 14;83(3):1039–1047. doi: 10.1016/0006-291x(78)91500-0. [DOI] [PubMed] [Google Scholar]
- Martegani E., Baroni M., Wanoni M. Interaction of cAMP with the CDC25-mediated step in the cell cycle of budding yeast. Exp Cell Res. 1986 Feb;162(2):544–548. doi: 10.1016/0014-4827(86)90358-7. [DOI] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Ishikawa T. Genetic analysis of the role of cAMP in yeast. Yeast. 1985 Sep;1(1):15–24. doi: 10.1002/yea.320010103. [DOI] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Ishikawa T. Identification of the structural gene and nonsense alleles for adenylate cyclase in Saccharomyces cerevisiae. J Bacteriol. 1984 Jan;157(1):277–282. doi: 10.1128/jb.157.1.277-282.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Ishikawa T. Initiation of meiosis in yeast mutants defective in adenylate cyclase and cyclic AMP-dependent protein kinase. Cell. 1983 Feb;32(2):417–423. doi: 10.1016/0092-8674(83)90461-0. [DOI] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Oshima Y., Ishikawa T. Isolation and characterization of yeast mutants deficient in adenylate cyclase and cAMP-dependent protein kinase. Proc Natl Acad Sci U S A. 1982 Apr;79(7):2355–2359. doi: 10.1073/pnas.79.7.2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Toh-E A., Ishikawa T., Oshima Y. Cyclic AMP may not be involved in catabolite repression in Saccharomyes cerevisiae: evidence from mutants capable of utilizing it as an adenine source. J Bacteriol. 1982 Apr;150(1):277–285. doi: 10.1128/jb.150.1.277-285.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olempska-Beer Z., Freese E. B. Optimal extraction conditions for high-performance liquid chromatographic determination of nucleotides in yeast. Anal Biochem. 1984 Jul;140(1):236–245. doi: 10.1016/0003-2697(84)90159-3. [DOI] [PubMed] [Google Scholar]
- Reichert U., Winter M. Uptake and accumulation of purine bases by stationary yeast cells pretreated with glucose. Biochim Biophys Acta. 1974 Jul 12;356(1):108–116. doi: 10.1016/0005-2736(74)90298-3. [DOI] [PubMed] [Google Scholar]
- Shilo V., Simchen G., Shilo B. Initiation of meiosis in cell cycle initiation mutants of Saccharomyces cerevisiae. Exp Cell Res. 1978 Mar 15;112(2):241–248. doi: 10.1016/0014-4827(78)90206-9. [DOI] [PubMed] [Google Scholar]
- Singh B., Guptaroy B., Hasan G., Datta A. Inhibitory effect of glucose and adenosine 3',5'-monophosphate on the synthesis of inducible N-acetylglucosamine catabolic enzymes in yeast. Biochim Biophys Acta. 1980 Oct 15;632(3):345–353. doi: 10.1016/0304-4165(80)90230-5. [DOI] [PubMed] [Google Scholar]
- Sy J., Roselle M. Cyclic AMP-dependent protein kinase of yeast. FEBS Lett. 1981 Nov 30;135(1):93–96. doi: 10.1016/0014-5793(81)80951-9. [DOI] [PubMed] [Google Scholar]
- Tatchell K., Robinson L. C., Breitenbach M. RAS2 of Saccharomyces cerevisiae is required for gluconeogenic growth and proper response to nutrient limitation. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3785–3789. doi: 10.1073/pnas.82.11.3785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toda T., Uno I., Ishikawa T., Powers S., Kataoka T., Broek D., Cameron S., Broach J., Matsumoto K., Wigler M. In yeast, RAS proteins are controlling elements of adenylate cyclase. Cell. 1985 Jan;40(1):27–36. doi: 10.1016/0092-8674(85)90305-8. [DOI] [PubMed] [Google Scholar]
- Tsuboi M., Yanagishima N. Comparative studies on sporulation-promotive actions on cyclic AMP, theophylline and caffeine in Saccharomyces cerevisiae. Arch Microbiol. 1975 Oct 27;105(2):83–86. doi: 10.1007/BF00447118. [DOI] [PubMed] [Google Scholar]
- Tsuboi M., Yanagishima N. Effect of cyclic AMP, theophylline and caffeine on the glucose repression of sporulation in Saccharomyces cerevisiae. Arch Mikrobiol. 1973 Oct 4;93(1):1–12. doi: 10.1007/BF00666076. [DOI] [PubMed] [Google Scholar]
- Uno I., Matsumoto K., Hirata A., Ishikawa T. Outer plaque assembly and spore encapsulation are defective during sporulation of adenylate cyclase-deficient mutants of Saccharomyces cerevisiae. J Cell Biol. 1985 Jun;100(6):1854–1862. doi: 10.1083/jcb.100.6.1854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uno I., Matsumoto K., Ishikawa T. Characterization of cyclic AMP-requiring yeast mutants altered in the regulatory subunit of protein kinase. J Biol Chem. 1982 Dec 10;257(23):14110–14115. [PubMed] [Google Scholar]
- de la Houssaye B. A., Masaracchia R. A. Standardization of the assay for the catalytic subunit of cyclic AMP-dependent protein kinase using a synthetic peptide substrate. Anal Biochem. 1983 Jan;128(1):54–59. doi: 10.1016/0003-2697(83)90343-3. [DOI] [PubMed] [Google Scholar]



