Abstract
Background and Aims
Plants display a wide range of traits that allow them to use animals for vital tasks. To attract and reward aggressive ants that protect developing leaves and flowers from consumers, many plants bear extrafloral nectaries (EFNs). EFNs are exceptionally diverse in morphology and locations on a plant. In this study the evolution of EFN diversity is explored by focusing on the legume genus Senna, in which EFNs underwent remarkable morphological diversification and occur in over 80 % of the approx. 350 species.
Methods
EFN diversity in location, morphology and plant ontogeny was characterized in wild and cultivated plants, using scanning electron microscopy and microtome sectioning. From these data EFN evolution was reconstructed in a phylogenetic framework comprising 83 Senna species.
Key Results
Two distinct kinds of EFNs exist in two unrelated clades within Senna. ‘Individualized’ EFNs (iEFNs), located on the compound leaves and sometimes at the base of pedicels, display a conspicuous, gland-like nectary structure, are highly diverse in shape and characterize the species-rich EFN clade. Previously overlooked ‘non-individualized’ EFNs (non-iEFNs) embedded within stipules, bracts, and sepals are cryptic and may represent a new synapomorphy for clade II. Leaves bear EFNs consistently throughout plant ontogeny. In one species, however, early seedlings develop iEFNs between the first pair of leaflets, but later leaves produce them at the leaf base. This ontogenetic shift reflects our inferred diversification history of iEFN location: ancestral leaves bore EFNs between the first pair of leaflets, while leaves derived from them bore EFNs either between multiple pairs of leaflets or at the leaf base.
Conclusions
EFNs are more diverse than previously thought. EFN-bearing plant parts provide different opportunities for EFN presentation (i.e. location) and individualization (i.e. morphology), with implications for EFN morphological evolution, EFN–ant protective mutualisms and the evolutionary role of EFNs in plant diversification.
Keywords: Ant–plant mutualism, ant protection, extrafloral nectaries, Senna, Fabaceae, functional morphology, homology, key innovation, morphological evolution, ontogeny, phylogeny
INTRODUCTION
Plants have evolved a myriad of traits that attract, reward and exploit animals for vital tasks, such as pollination, seed dispersal and herbivore defence (Schoonhoven et al., 2005; Bronstein et al., 2006). Both partners benefit from such mutualisms. In some cases, traits that mediate mutualisms in novel ways may ultimately play key roles in triggering the evolutionary diversification of plant lineages. As new species and clades originate, these traits are likely to persist in lineage evolution. They may diversify morphologically by keeping, modifying or losing their function, or they may be suppressed completely.
The location of these traits on the plant surface, and therefore where the interactions themselves take place, has often been overlooked. Reproductive and non-reproductive plant parts represent fundamentally different functional locations, as traits in these locations are expected to mediate interactions for fundamentally different vital tasks. For example, traits facilitating pollination involve reproductive plant parts and tissue. Flower spurs are a textbook example of a key innovation that have allowed certain angiosperms to undergo adaptive radiations with respect to their pollination biology (Hodges and Arnold, 1995; Hodges, 1997). Another example of key innovation is floral symmetry (Sargent, 2004; Bush and Zachgo, 2009). Indeed, plant–pollinator mutualisms are fundamentally linked to the evolutionary success and diversification of angiosperms (e.g. Lunau, 2004; Friis et al., 2006; van der Niet and Johnson, 2012).
In contrast, traits located on non-reproductive plant parts that facilitate seed dispersal or protection from natural enemies are less well studied (Janzen, 1984; Heil and McKey, 2003) and are likely to play a role in plant diversification in more indirect ways. Mutualisms with seed dispersers and ant protectors can increase plant survival and reproduction, leading to increased chances for plants to utilize emerging ecological opportunities and, ultimately, undergo speciation (Lengyel et al., 2009, 2010; Marazzi and Sanderson, 2010).
Extrafloral nectaries (EFNs), which offer a carbohydrate-rich liquid reward to ants in return for their protection from insect herbivores (Bentley, 1977; Koptur, 1992), appear to have played a key evolutionary role in the diversification of certain plant clades (Marazzi and Sanderson, 2010). EFNs are widespread in plants (Koptur, 1992; Keeler, 2011; Weber and Keeler, 2013). They have intrigued scientists for centuries, including Darwin (1876) and his son (Darwin, 1877). Yet, their impressive diversity remains underappreciated. This study aims to fill the gap in our understanding by exploring EFN diversity and diversification in a phylogenetic framework and by interpreting results in the context of EFN-mediated mutualisms with aggressive ants.
A wide range of plant parts can bear EFNs. They are found on several different vegetative plant parts (e.g. leaves), as well as on inflorescences (e.g. pedicels) and the outside of the outer floral organs not directly involved in pollination (e.g. sepals). Morphologically, EFNs represent a heterogeneous multitude of secretory structures, from simple glandular trichomes and cryptic secretory tissue embedded within EFN-bearing plant parts to conspicuous, complex vascularized glands, all of which produce and secrete nectar (Caspary, 1848; Delpino, 1868–1875; Zimmermann, 1932; Elias and Gelband, 1976; Fahn, 1979; Schmid, 1988; Vogel, 1997; Bernardello, 2007). EFN presence during plant ontogeny, from seed to reproductive adult, is a neglected but important temporal dimension of EFN diversity, as it is expected to correlate with where and when protection is needed by the plant over its life history.
EFN-bearing plants occur in varying proportions in many different kinds of vegetation (e.g. Keeler, 1980; Pemberton, 1988; Schupp and Feener, 1991; Blüthgen and Reifenrath, 2003; Díaz-Castelazo et al., 2004; Machado et al., 2008) and are especially well represented in the Leguminosae (=Fabaceae; Lewis et al., 2005). Legumes are particularly rich in their interactions with ants (McKey, 1989). Indeed, species from over 100 legume genera bear EFNs (Marazzi et al., 2012), including our study group, the caesalpinioid genus Senna. With approx. 350 species, Senna is one of the largest legume genera (Marazzi and Sanderson, 2010) and one of the angiosperm genera with the highest number of species bearing EFNs (Weber and Keeler, 2013).
The conspicuous EFNs on the leaves and sometimes the pedicels of Senna have long intrigued scientists. EFN histology has been well studied in some species (e.g. Bhattacharyya and Maheshwari, 1970; Pascal et al., 2000; Melo et al., 2010), and the diversity of EFN shapes and locations has been used for taxonomic purposes (e.g. Bentham, 1871; Irwin and Barneby, 1982; Randell, 1988, 1989; Marazzi et al., 2006b). EFNs were once considered a ‘primitive’ feature in Senna (Irwin and Barneby, 1982), but in fact they characterize a large clade well nested within the genus, which has been referred to as the ‘EFN clade’ (Marazzi et al. 2006a). The EFN clade diversified faster, becoming more species-rich (approx. 80 % of all extant species) and geographically more widespread than its closely related clades supposedly lacking EFNs, the non-EFN clades I–III (Marazzi and Sanderson 2010). For this reason, the evolution of EFNs has been considered a key innovation that drove diversification. To our knowledge, Senna is the only genus in which the evolutionary role of EFNs has been analysed using phylogenetic data (Marazzi and Sanderson, 2010). Thus, it represents a unique opportunity and an ideal model to reconstruct the evolution of EFN diversity and interpret results in the context of the mutualisms that the EFNs facilitate.
In this study, we focus on the two phenotypic components of EFN diversity in Senna: their morphology and their location on adult plants (as well as on various seedling stages). Using data from the literature, field observations, analysis of field-collected and cultivated individuals, ontogenetic studies, and the molecular phylogenetic trees from Marazzi and Sanderson (2010), we ask: (1) How is EFN diversity morphologically characterized and taxonomically distributed in Senna? In particular, we compare morphologies of EFNs on different locations on plants. (2) When do EFNs first develop during plant ontogeny? Are EFNs present from the cotyledon stage through to the adult flowering plant? (3) How did different kinds of Senna EFNs evolve? Ancestral EFNs were probably morphologically different from modern EFNs, but ancestral character state reconstruction may provide new insights into levels of homology of different kinds of EFNs as well as where EFNs first appeared on the plant. We use these data to discuss the implications of EFN diversity and evolution to illuminate our understanding of EFN–ant mutualisms and give insight into key innovations in this and perhaps other genera.
MATERIAL AND METHODS
Overview on Senna
Senna displays diverse habits (herbs, shrubs, trees and lianas) and has colonized a wide range of habitats at different latitudes and altitudes, from tropical rain forests and savannah-like habitats to deserts (Irwin and Barneby, 1982). Senna species are distributed worldwide in tropical and subtropical regions, especially in the Americas (74 % of the species), but also in Australia (13 %) and Africa and Madagascar (10 %). Only a few species occur in the Near East, Southeast Asia and the Pacific Islands (Marazzi and Sanderson, 2010). Molecular phylogenetic analyses of Senna (Marazzi et al., 2006a; Marazzi and Sanderson, 2010) recognized seven clades, I–VII, and supported as monophyletic several of the current taxonomic series by Irwin and Barneby (1982), including series Basiglandulosae (nested in clade VII) and Aphyllae (in clade VI), mentioned in this study.
Taxonomic sampling
We characterized the diversity of EFNs with respect to topology, morphology and plant ontogeny in a total of 83 Senna species (about one-quarter of the genus), reflecting the number of species represented in recent molecular phylogenetic analyses, 65 of which belonged to the EFN clade (Marazzi and Sanderson, 2010). We collected EFN-bearing material from 19 of these species from wild and cultivated individuals (one or two available accessions per species), representing different clades and the taxonomic distribution of diverse locations of EFNs on leaves according to previous work (Marazzi et al., 2006a; Marazzi and Sanderson, 2010). Only limited material of two species with EFNs at the base of pedicels was available in this study (S. cana, clade V; S. robiniifolia, clade VI). We compared the floral morphologies of three Senna species included in the EFN clade (S. obtusifolia of clade IV and S. pendula of clade VII both with EFNs between the first pair of leaflets; S. occidentalis of clade VII, with EFNs at the base of leaf stalks) and of three species included in clade II (S. didymobotrya, S. martiana, S. pleurocarpa). The latter species were included in the floral morphological study because field observations of ants around floral buds and discoloured stipules in S. martiana and its close relative S. alata suggested the presence of EFNs. No ants were observed around floral buds or leaves in the field in species of clade I (e.g. S. galeottiana, S. polyantha, S. silvestris) or clade III (e.g. S. wislizeni, S. spectabilis) and therefore we excluded these taxa from further analyses. Plants used for ontogenetic studies were cultivated from seeds available at the Desert Legume Program (DELEP; University of Arizona). They included five species representing different Senna clades and different locations of EFNs on the plant: S. aphylla, leafless in adult stage; S. costata, with EFNs on leaves between pairs of leaflets; S. hirsuta, EFNs at the base of leaf stalks; and S. pallida, EFNs on leaves between the first pair of leaflet, belong to the EFN clade; S. pleurocarpa, discoloured stipules, belongs to clade II. A list of the specimens studied and voucher information is given in Supplementary Data Appendix S1. Herbarium vouchers are deposited at ARIZ.
Morphological and ontogenetic studies
We documented EFN location on the plant, as well as EFN shape and colour, using in vivo observations and colour photographs for a total of 21 cultivated and field-collected Senna species (Supplementary Data Appendix S1). We complemented these data with information from the taxonomic literature (Irwin and Barneby, 1982). EFN-bearing plant parts for morphological studies were fixed and stored in 70 % ethanol. To assess levels of homology of EFNs, we investigated the general histological structure using serial microtome sectioning. We selected two mature EFNs per species and per analysis. Samples, prepared following standard procedures for scanning electron microscopy (SEM) at the University Spectroscopy and Imaging Facilities of the University of Arizona, were directly coated with palladium and analysed with a Hitachi S-3400 N Type II VPSEM (15 kV). Samples prepared for microtome sectioning were embedded either in Kulzer's Technovit 2-hydroethyl methacrylate (Igersheim, 1993) and sectioned with a Microm HM335 rotary microtome (Microm International GmbH, Walldorf, Germany) and conventional microtome knife (grade D) or in Polysciences's Gemcut Pink Sapphire Paraffin following routine embedding and sectioning techniques of the Histology Service Lab of the University of Arizona, using a Microm HM355S and disposable knife. Longitudinal and transverse section series of Technovit- and paraffin-embedded samples were cut at 5 or 7 µm, stained with ruthenium red and toluidine blue (protocols adapted from Weber and Igersheim, 1994), and mounted on glass slides. Sections from Technovit-embedded samples were mounted in Histomount (National Diagnostics, Atlanta, GA, USA). Paraffin was removed before staining the sections, which were mounted in a synthetic mounting medium (Leica Microsystems, Buffalo Grove, IL, USA). Section series were investigated with light microscopy (LM) and photographed with a Axiocam HRc digital camera (Carl Zeiss AG, Oberkochen, Germany). Ethanol-fixed EFN material and SEM samples mounted on stubs are preserved in the lab of M.M. at the University of Arizona and in the Institute of Systematic Botany of the University of Zurich (Z). All slides are deposited in Z.
For studies of EFN presence during plant ontogeny, seeds were cultivated in the DELEP's greenhouse. Three to seven successfully germinated individuals per species were observed during 2010–2011. In cases where EFN presence differed between seedling and adult plants, ethanol-fixed samples were analysed by SEM (see procedure above), and seedlings from other maternal plants at different sites were observed to confirm previous findings. We monitored plants biweekly during early plant ontogeny (cotyledons, early and late seedling stages) and monthly or every 2 months during later stages (young vegetative plant, flowering plant, fruit and seed setting). The various stages were documented with colour photographs, which are available upon request to B.M.
EFN evolution reconstruction
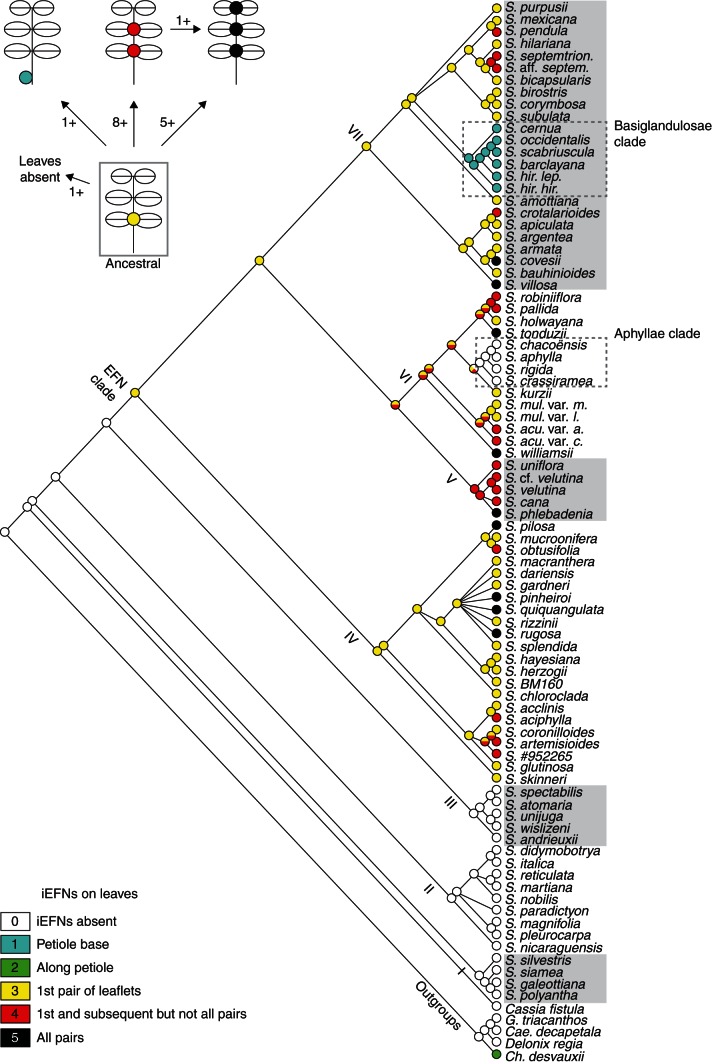
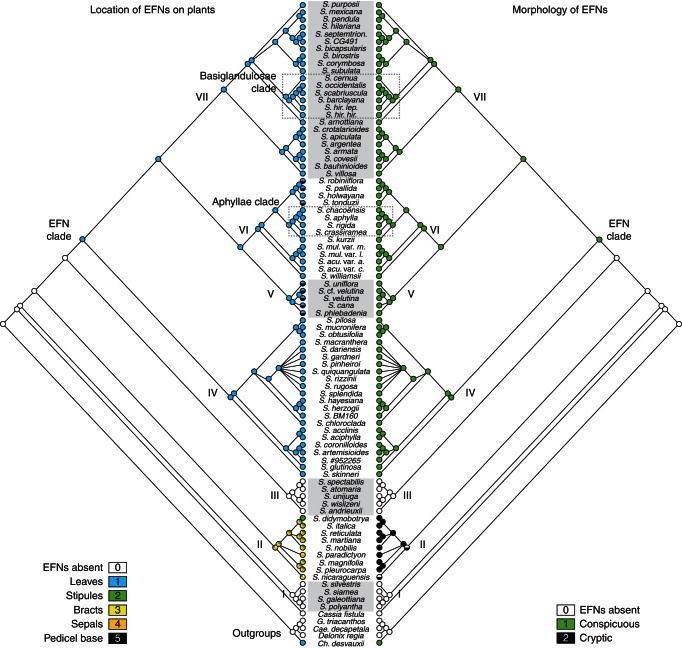
To explore the evolution of EFN morphology we analysed the phylogenetic distribution of EFN diversity by first tabulating our findings about three characters, (A) location of EFNs on a plant, (B) detailed location of EFNs on the leaves and (C) EFN morphology, on the molecular phylogenetic tree inferred from likelihood analyses by Marazzi and Sanderson (2010). We coded traits for ancestral character-state reconstruction, considering that EFNs on different functional locations (A) and of diverse morphologies (C) are similar in a broad sense (nectar-secreting structures), while EFNs on different parts of the compound Senna leaves (B) are homologous because of common ancestry (i.e. synapomorphy of the EFN clade; Marazzi et al., 2006a). Characters were coded with the following states (multi-state coding allowed): (A) 0 = EFNs absent, 1 = leaves, 2 = stipules, 3 = bracts, 4 = sepals, 5 = bract scar, 6 = base of pedicels; (B) 0 = no EFNs on leaves, 1 = EFNs at the base of the petiole (near or on the pulvinus), 2 = along the petiole (well above the base and below the first pair of leaflets), 3 = between the first (i.e. the most proximal) pair of leaflets only, 4 = between the first pair of leaflets and also the subsequent pair, 5 = between all pairs of leaflets; (C) 0 = EFNs absent, 1 = cryptic EFN morphology, 2 = conspicuous EFN morphology, 3 = trichomes. Missing information was coded with question marks as uncertain states. Because intraspecific variation made it difficult to assign EFN shapes and stalked versus sessile EFNs to discrete categories, we did not analyse these two traits. To reconstruct ancestral states we used Mesquite v. 2·75 (Maddison and Maddison, 2011), selecting the parsimony optimization criterion and the ‘unordered’ model of character evolution (i.e. same cost for all transitions) to trace the character history over the Senna maximum-likelihood (ML) tree and over 500 ML bootstrap trees to account for phylogenetic uncertainty (all trees from Marazzi and Sanderson, 2010, based on DNA sequences from three chloroplast markers). All most-parsimonious reconstructions presented by Mesquite were considered. We calculated tree length (number of steps), retention index (RI) and consistency index (CI) for each character.
RESULTS
How is EFN diversity characterized and distributed in Senna?
EFNs occur in Senna species belonging to clade II and to the ‘EFN clade’ (i.e. clades IV–VII) for a total of 257–312 species (approx. 89 % of the genus; clade size estimates from Marazzi and Sanderson, 2010). The relative phylogenetic position of these two clades within Senna and their respective number of species are illustrated in the simplified phylogenetic tree of Fig. 1.
Fig. 1.

Diversity of EFNs in Senna – clade II. The phylogenetic position and size of clade II with respect to the other clades is illustrated in the Senna cladogram at the bottom of the figure (adapted from Marazzi and Sanderson, 2010). (A–C) EFNs on lobes of partly modified, discoloured stipules (arrowheads). (D) Cross-section of a modified, stipule lobe (ventral side downward); the nectariferous tissue (n) is the darker, cell-denser area in between and around the vascular bundles (vb). (E, F) EFNs on various parts of the inflorescence: putative bract scar at the base of pedicels (arrow) and dorsal side of bracts and sepals (with and black arrowheads, respectively). (G, H) Ants suggesting presence of EFNs on sepals and bracts. (A) S. alata. (B, D) S. martiana (photo courtesy of L. Paganucci de Queiroz). (C, E–F) S. pleurocarpa. (G) S. italica (photo courtesy of A. Weaving/Ardea.com). (H) S. paradyctyon. Scale bars (indicative): (A–C, E–G) = 5 mm; (D) = 1 mm.
Clade II
In all species examined from this clade, EFNs occur on both stipules (Fig. 1A–D) and inflorescences (Fig. 1E–H) of the same plant, specifically along the inflorescence axis at the base of the pedicel, possibly as the scar of the bract that falls off (Fig. 1E), and on the dorsal side of bracts and sepals, especially of buds (Fig. 1E, F). In calyx EFNs of S. pleurocarpa, nectar accumulates primarily on the apical part, whereas droplets appear more randomly scattered on bracts (Fig. 1F).
Clade II exclusively exhibits cryptic EFNs that are integrated into partly modified, existing organs. The stipule blade may be glabrous or covered by trichomes and is strongly asymmetric and cordate, forming one lobe that is modified in colour and thickness (Fig. 1A–C) and includes the secretory tissue (Fig. 1D). Secretory trichomes appear to be absent from bracts and sepals of S. pleurocarpa (not shown). Our analyses of transverse sections of bracts and sepals were unable to identify the secretory units responsible for nectar production (not shown). No trichomes were observed on bracts and sepals, but it is possible that they fell off during SEM sample preparation. EFNs on bract scars, bracts and calyces are completely cryptic, and no external macroscopic features, except for the presence of nectar droplets, reveal their presence (see S. pleurocarpa, Fig. 1E, F). Ant attendance confirmed EFNs in S. alata (B. Marazzi, pers. observ.) and S. martiana (L. Paganucci de Queiroz, University of Feira de Santana, Brazil, pers. comm.): ants were repeatedly observed to ‘scrape’ the surface of stipules, bracts and calyx. EFNs are probably present in other species of clade II as well, for example in species in which ants have been observed to consistently visit stipules, bracts and sepals (S. italica, Fig. 1G; S. paradictyon, Fig. 1H).
EFN clade
EFNs present in species of this clade are always found on leaves (Fig. 2A–H), either (1) at the base of the petiole only (Basiglandulosae clade), (2) along the petiole, on the rachis (3) between the first pair of leaflets only (clades IV, V and VII), (4) between the first and subsequent pair of leaflets or (5) between all pairs of leaflets (clades IV, V and VII). In some species, EFNs are also present at the base of pedicels (clade V; many species of clade VI; Fig. 2I–L). EFNs never occur simultaneously at the base of the petiole and between pairs of leaflets.
Fig. 2.
Diversity of EFNs in Senna – EFN clade. The phylogenetic position and size of the EFN clade with respect to the other clades is illustrated in the Senna cladogram in Fig. 1. (A–H) iEFNs on leaves; species ordered by clade (clades IV–VII). (A–E, G, H) EFNs between leaflets (first only or also subsequent pairs); arrowheads indicate nectar drop. (F) EFNs at the base of petiole. (H) Median longitudinal section of an iEFN with four kinds of cell layers. (I–L) iEFNs at the base of pedicels (arrowheads). (A) S. artemisiodes (IVa). (B) S. pilifera (IVb). (C) S. cana var. calva (V). (D) S. aversiflora (VI). (E) S. occidentalis (VIIa). (F) S. purpusii (VIIa). (G) S. covesii (VIIb). (H) S. pendula (VIIb). (I, J) S. bracteosa (V; photo courtesy of D. Cardoso). (K) S. pallida (VI). (L) S. tonduzii (VI). Abbreviations: a, abscission of pedicel; b, bract; ep, epidermis; fl, flower developing; lf, first (proximal) pair of leaflets; np, nectary parenchyma; p, pedicel; pt, petiole; sp, subnectary parenchyma; uk, layers of unknown function; vb, vascular bundles. Scale bars (indicative): (A–G, I–L) = 5 mm; (H) = 1 mm.
All EFNs in species belonging to the EFN clade are highly conspicuous and gland-like. These EFNs are convex and sessile (e.g. Fig. 2A) or stipitate (e.g. Fig. 2F, G), i.e. the secretory portion of the organ is subtended by a non-secretory stalk. In species with hairy leaves, hairs may cover the EFN stalk but never the secretory head. EFNs on leaves display a range of forms and shapes, including globose, ovoid, pyriform, clavate and obclavate. Stipitate forms have globose, ovoid or pyriform heads. The longest EFNs may measure up to several millimetres.
EFNs at the base of pedicels are similar in form and shape to the EFNs on leaves in the same species. In studied species of clade VI, one EFN is present on the side of the pedicel base (Fig. 2K, L), whereas in studied species of clade V, two EFNs are present on each side of the pedicel (e.g. Fig. 2I).
In our histological analyses of EFNs in species of the EFN clade (e.g. Fig. 2H), the EFN consisted of the epidermis, the nectary parenchyma and the subnectary parenchyma (including the vascular bundles branching off from the main leaf vascular system). An additional fourth structure of one to two layers of cells can be observed between the nectary and the subnectary parenchyma in some of the species with a well-developed nectary parenchyma [S. occidentalis, S. pendula (Fig. 2H), S. obtusifolia].
When do EFNs first develop during plant ontogeny?
The presence of EFNs during plant ontogeny is summarized and illustrated in Fig. 3 and Supplementary Data Fig. S1. EFNs are absent from cotyledons in all studied species. The first photosynthetic leaf bears EFNs, except in S. aphylla (EFN clade), where gland-like EFNs are apparently absent from all seedling leaves (Supplementary Data Fig. S1D, and Fig. 1H), until leaves are replaced by scales in late seedling stages. Interestingly, the petiolules (i.e. leaflet stalks) of S. aphylla are bright orange during the early seedling stage (Supplementary Data Fig. S1H), but no exudate was observed.
Fig. 3.

EFN location during plant ontogeny in Senna hirsuta (EFN clade, clade VII). Leaves are numbered in order of development (1 = first leaf). (A) Mother plant with EFN visited by an ant. (B, C) Young seedling with cotyledons and leaf 1 (leaf developing in B, fully expanded in C). (C) Cotyledons and leaf base lacking EFNs. (D) EFN between the proximal pair of leaflets, with nectar droplet (arrowhead). (E, F) Older seedlings. (E) Base of petiole lacking EFNs. (F) Change in EFN location from leaf 8 onward, EFN at the base of the petiole. (G, H) SEM micrographs. (G) Pulvinus lacking EFN before shift in EFN location (leaf 7). (H) Proximal pair of leaflets lacking EFN after shift in EFN location (leaf 8). Arrowheads indicate EFNs. Abbreviations: lf, leaflet; pt, petiole; pu, pulvinus. Scale bars: (A, C, E) = approx. 5 mm; (G, H) = 0·5 mm. See also Supplementary Data Fig. S1 for plant ontogenies of S. aphylla (clade VI), S. costata (IV), S. pallida (VI) and S. pleurocarpa (II).
Leaves bear EFNs in the same location in seedlings and adult plants in all species except S. hirsuta (Fig. 3). In this species, the first seven leaves develop EFNs between the first pair of leaflets (Fig. 3B, E, G), whereas the 8th and all subsequent leaves (Fig. 3F, H) develop EFNs at the base of the petiole. We also observed this pattern both in plants grown in local nurseries in Tucson, Arizona, and in seedlings found in the wild.
How did different kinds of Senna EFNs evolve?
The character matrix used in our analyses is found in Supplementary Data Appendix S2. Ancestral character-state reconstructions, including tree values calculated for each character (i.e. number of parsimony steps, RI and CI) are presented in Figs 4 and 5. Character states reconstructed at each internal node for each of the trees (single optimal and 500 bootstrapped) are listed in Supplementary Data Appendix S3 and summarized in Supplementary Data Table S1. Inferences about the location on a plant (character A) suggest that EFNs evolved at least once on leaves (EFN clade), once on stipules (clade II), three times at or near the base of pedicels (clades II, V and VI), and once on bracts and sepals (clade II; Fig. 5). With regard to the location of EFNs on leaves (character B), the reconstructions strongly suggest that ancestral EFN-bearing species had EFNs on leaves between the first pair of leaflets only; later, multiple clades independently evolved EFNs on subsequent pairs of leaflets as well. In contrast, EFNs changed location on the plant body and evolved at the base of the petiole only in one clade, and were suppressed in another clade (Fig. 4). The reconstruction for the ancestral state at the node of clade V + VI is ambiguous: EFNs may have occurred between the first pair of leaflets only or between multiple pairs of leaflets. Finally, with regard to character C (i.e. EFN morphology) the results strongly suggest that conspicuous EFNs evolved exclusively in the EFN clade, and cryptic EFNs exclusively in clade II (Fig. 5).
Fig. 4.
Diversification history of EFN location on leaves in Senna. Parsimony reconstruction of ancestral states of character B (location of EFNs on leaves; 23 steps, RI = 0·64, CI = 0·22) on the ML tree by Marazzi and Sanderson (2010). Roman numbers indicate clades I–VII (Marazzi et al., 2006a). The graphic in the upper left corner summarizes results by illustrating the minimum number of transitions inferred among states. Results for each state at each node can be found in Supplementary Data Appendix S3 and summarized for selected nodes in Supplementary Data Table S1.
Fig. 5.
Diversification history of EFN location on plants and EFN morphology in Senna. Parsimony reconstruction of ancestral states of character A (location of EFNs on plants; 19 steps, RI = 0·93, CI = 0·95) and character C (EFN morphology; 3 steps, RI = 0·93, CI = 0·67) on the ML tree by Marazzi and Sanderson (2010). Roman numbers indicate clades I–VII (Marazzi et al., 2006a). Results for each state at each node can be found in Supplementary Data Appendix S3 and summarized for selected nodes in Supplementary Data Table S1.
DISCUSSION
The outstanding diversity of EFNs has been fascinating scientists for centuries (e.g. Caspary, 1848; Zimmermann, 1932; Vogel, 1997). Our study is the first to explore the diversity of both EFN location on the plant and EFN morphology during plant ontogeny, and to reconstruct the evolutionary history of the observed diversity on a phylogeny. Below, we address three questions regarding Senna's EFNs: how is EFN diversity characterized and distributed taxonomically, what life-history stages exhibit EFNs and how did the different types of EFNs evolve? In brief, we found that Senna EFNs are more diverse and widespread and their evolutionary history more complicated than previously thought (Irwin and Barneby, 1982; Marazzi and Sanderson, 2010). The conspicuousness of the gland-like EFNs on leaves in Senna has made them easy to find in dried herbarium specimens used for taxonomic work (e.g. Bentham, 1871; Irwin and Barneby, 1982), whereas only the presence of nectar droplets in greenhouse-cultivated specimens revealed the cryptic EFNs in species of clade II until now considered to lack EFNs.
How is EFN diversity characterized and distributed taxonomically in Senna?
Senna is one of the three richest plant genera in terms of the number of EFN-bearing species, surpassed only by Passiflora (329 spp., Passifloraceae) and more or less equalling the mimosoid legume Inga (294 spp.) (Weber and Keeler, 2013). EFNs in Senna display a remarkable diversity in location and morphology. They are found on five different plant parts (Figs 1 and 2): (1) leaves, (2) stipules, (3) along the inflorescence axis at the base of pedicel, and on the dorsal side of (4) bracts and (5) sepals. EFNs may occur on multiple locations of the same plant, but always as only (1), (1) and (3), or (2)–(5). In other words, EFNs are either on leaves or on stipules (but not both). If on leaves, they will also be found at the base of pedicels in some species. If on stipules, then they will also be found on the dorsal sides of bracts and sepals. Additionally, the leaves are even-pinnate with one to over 15 pairs of leaflets and bear EFNs at five locations in different species: (1) at the base of the petiole only, (2) along the petiole (rare); and on the rachis (3) between the first pair of leaflets only, (4) between the first and some but not all subsequent pairs of leaflets or (5) between all pairs of leaflets. EFNs on stipules, bracts and sepals are here reported for the first time in Senna. A tendency for EFN plants to allocate EFNs on or around leaves and flowers has long been noted in legumes and other taxa (Darwin, 1876; McKey, 1989; Koptur, 1992). EFNs in the inflorescence are not as widespread in plants as are EFNs on or around leaves. However, it is possible that they have previously been overlooked in legumes (Marazzi et al., 2012; and as shown in this study) as well as in and beyond the other 13 angiosperm families known to have EFNs on inflorescences (Bernardello, 2007).
Two distinct kinds of EFN morphologies exist in Senna, which we here interpret in relation to their degree of individualization, i.e. morphological differentiation and specialization, with respect to the organ that bears them. The newly discovered, cryptic EFNs are non-individualized EFNs (non-iEFNs), i.e. stipules, bracts and sepals that are EFN-bearing organs, in which specific zones differentiate into nectar-producing structures (Fig. 1). Such structures consist of nectariferous tissue embedded in the coloured stipule lobe (Fig. 1D) and probably of individual or small clusters of secretory cells in the dorsal side of bracts and sepals. Non-iEFNs occur exclusively in species of clade II, and may thus represent a new synapomorphy for this clade. While discoloration seems diagnostic for stipule EFNs, EFNs of bract scars, bracts and sepals are completely cryptic and non-structural. In general, non-structural nectaries are non-differentiated areas that sporadically secrete nectar, and are therefore hard to distinguish if there is no exudate (Zimmermann, 1932; Fahn, 1979; Bernardello, 2007). Thus, detailed anatomical studies are necessary to characterize them. Although no EFNs have been detected so far in Senna species of clades I and III, we will continue to look out for field reports of ‘potentially EFN-related ant activity’ on these species. Non-structural EFNs exist as both floral and extrafloral nectaries, and may in fact be much more common than currently known from eight angiosperm families (Bernardello, 2007).
In contrast, in the EFN clade, EFNs on leaves and at the base of pedicels are highly individualized (iEFNs). These are by definition easy to detect, being quite distinct from the organ that bears them, to the point where they appear to form a novel organ dedicated as an EFN. That is, these EFNs bear no obvious homology to other, pre-existing organs (see ‘true’ EFNs in Marazzi et al., 2012). iEFNs are structural nectaries and display the typical differentiation of nectar-producing organs into three histologically distinct areas (Bernardello, 2007; Nepi, 2007): epidermis, followed by the nectary parenchyma, and then the subnectary parenchyma with the vascular bundles branching off from the main vascular system of the EFN-bearing organ. In Senna, an additional structure of one or two layers of cells is present between the nectary and the subnectary parenchyma in species with a well-developed nectary parenchyma (this study, Fig. 2H; Pascal et al., 2000; Melo et al., 2010). The function of this additional structure is unclear, but it is perhaps related to the characteristic convex instead of concave structure of Senna iEFNs. Concave iEFNs occur on leaves in the closely related Chamaecrista (Pascal et al., 2000) as well as many mimosoid legumes. iEFNs appear to be absent from papilionoid legumes (McKey, 1989; Marazzi et al., 2012).
Finally, in Senna, EFNs occur separately in two of four clades (clade II and the large EFN clade, including clades VI –VII; Figs 4 and 5). Within these two clades, all plants studied have EFNs, but the nectaries vary greatly in number and location. Furthermore, according to a survey of the taxonomic literature on Senna and a set of diagnostic floral morphological traits, Marazzi and Sanderson (2010) concluded that all Senna species bearing gland-like iEFNs and not yet considered in published molecular phylogenetic analyses (approx. 170–220 spp.) would probably belong to the EFN clade. A more comprehensive (ideally complete) phylogeny of Senna would allow us to evaluate whether variation in EFN location and morphology and its phylogenetic distribution translates into taxonomically useful traits to formally describe infrageneric relationships within the genus.
Are EFNs present from the cotyledon stage to the adult flowering plant?
The presence of EFNs determines when during a plant's life cycle nectar can be secreted for attracting and rewarding ant defenders. In Senna, cotyledons lack EFNs, but EFNs are present (and secreting) from the first leaf or first pair of stipules onwards, with the exception of S. aphylla (Supplementary Data Fig. S1). Senna aphylla is one of a dozen species of the xerophytic Aphyllae clade (nested within the EFN clade VI) in which the presumed absence of EFNs has been thought to be related to the absence of normally developed leaves in adult plants (Bravo, 1978; Irwin and Barneby, 1982; Marazzi et al., 2006a; Marazzi and Sanderson, 2010). Yet, iEFNs appear to be absent from small leaves present in seedling stages as well, although they could be initiated in early leaf development but suppressed shortly thereafter. It remains unclear whether EFNs are present at the base of pedicels as in several of the other species of clade VI (e.g. Fig. 2K, 2L).
EFN location on leaves remains consistent throughout plant ontogeny, except in one species, S. hirsuta (EFN clade). Here, EFNs were expected at the base of the petiole as in adult plants (characteristic of the Basiglandulosae clade; see Fig. 4). However, seedlings instead developed EFNs between the first pair of leaflets during the first seven leaves in early plant ontogeny (Fig. 3). Only from the eighth leaf onward did the nectary develop at the base of the petiole. This phenomenon is reported here for the first time. Disentangling the processes that lead to developmental change requires an understanding of how particular ontogenies differ and, ultimately, how different developmental programmes are mediated (e.g. Gould, 1979; Raff and Kaufman, 1983; Hall, 1984). In S. hirsuta, morphological similarity of the EFNs before and after the ontogenetic shift (Fig. 3) suggests that the same EFN regulatory machinery is responsible for the development of EFNs in the two locations. Thus, a change in the timing of the developmental programme of the nectary, the leaf or both could perhaps suffice to produce a change in location. The genetic programmes underlying nectary development are, however, still poorly understood; only one gene needed for floral nectary development has thus far been clearly identified (Bowman and Smyth, 1999; Baum et al., 2001; Lee et al., 2005a, b).
How did different types of Senna EFNs evolve?
The two EFN morphologies in Senna are not homologous (by comparison to a phylogeny): they originated independently and underwent diversification histories leading to distinct morphological specialization (Figs 4 and 5). Non-iEFNs evolved exclusively in clade II as modified, discoloured stipule lobes and – on the same plant – also as non-structural and cryptic on bracts and sepals (and occasionally also bract scars). Non-iEFNs apparently underwent minimal morphological diversification, as extant species of clade II only differ in colour and size of the EFN-bearing stipule lobe. Specialized, gland-like iEFNs originated exclusively in the EFN clade and underwent an impressive diversification, including up to three levels of morphological evolution: (1) location on the same plant (on leaves and inflorescences at the base of pedicels), (2) location within the same EFN-bearing organ (petiole, between leaflets) and (3) individualized morphology (various shapes, colours, sessile versus stalked, etc.).
(1) Inflorescence iEFNs in species of clades V and VI (embedded in the EFN clade) are morphologically indistinguishable from the leaf iEFNs from the same plant (Fig. 2). However, they originate not on a leaf-homologue but on the axis itself at the base of pedicels, implying that they are associated with something that is missing, either bracts (early deciduous) or bracteoles (absent in Senna; Irwin and Barneby, 1982). Detailed knowledge of EFN development is necessary to verify whether iEFNs at the base of pedicels develop very early and persist during development of putative subtending bracts or bracteoles that, in contrast, are initiated, but then aborted.
-
(2) Senna iEFNs at different locations on the leaf – on the petiole or along the rachis between leaflets – all exhibit a similar morphology (Fig. 2), lending additional support to the hypothesis of evolutionary homology suggested by their single origin within the genus (Marazzi et al., 2006a). EFNs between leaflets occur in several clades, whereas iEFNs at the base of the petiole are a synapomorphy for a group in clade VII (Basiglandulosae clade; Marazzi and Sanderson, 2010). Leaves of species in the ancestral EFN clade probably bore iEFNs between the first pair of leaflets only (Fig. 4). Later, multiple lineages evolved leaves with iEFNs between subsequent pairs of leaflets, whereas in one lineage EFNs evolved at the base of the petiole but not between leaflets. In this latter lineage, the inferred sequence of evolution coincides with the change of iEFN location during early plant ontogeny observed at least in one extant member, S. hirsuta (see above). Curiously, before character evolution could be reconstructed using molecular phylogenies, it was common to interpret character states appearing early in organism ontogeny as ancestral (plesiomorphic) and those appearing later in ontogeny as derived (apomorphic) (e.g. see Kluge and Strauss, 1985, for a review). Our findings are consistent with this interpretation.
In other words, extant iEFN location on leaves may have evolved (a) by keeping the plesiomorphic state, (b) by shifting the location from between the first pair of leaflet to the base of the petiole and (c) by replicating the plesiomorphic location to the subsequent pairs of leaflets. As reflected in the different numbers of transitions between the plesiomorphic and the derived states (see Fig. 4 top left), it appears easier to replicate EFNs than shift their location to the petiole. This makes sense when considering that petiole and rachis represent two structurally different leaf parts and that the number of pairs of leaflets inserted along the rachis can vary considerably among and often also within Senna species (Irwin and Barneby, 1982). Loss of EFNs occurs only if the leaves are absent or highly modified (as in the Aphyllae clade).
(3) In terms of morphology, iEFNs in Senna are highly variable in their position relative to the leaf surface and in colour, with consequences for nectar presentation (Fig. 2). They can be sessile on the rachis, with the nectar presented near the leaf surface, or stalked, with the secretory head and the nectar presented up to several millimetres above the leaf surface. Non-stalked EFNs tend to be pale to dark-blackish green, while in stalked EFNs, the secretory head is often yellowish, orange or reddish. Elevating EFNs above the leaf level may play a role in ant attraction, especially if EFN colour serves as a visual cue for ants to detect nectar (Beattie, 1985) or if the leaf is hairy and nectar needs to be visible and accessible above the hairs. The coloured, secretory stipule lobe of non-iEFNs may also represent a visual cue for ants.
Implications for the EFN–ant mutualisms
Different locations and morphologies of EFNs can reflect differences in their function, which is to attract ants and mediate ant–plant protection mutualisms (e.g. Bentley, 1977; Koptur, 1992). EFNs are usually allocated on or around leaves and flowers and fruits, where they develop and start secreting nectar before leaf expansion and early during floral bud and seed development. This allows them to attract ants at a time when attacks by herbivores, florivores and seed predators would result in great damage (Beattie, 1985; McKey, 1989; Koptur, 1992; Rico-Gray and Oliveira, 2007). In Senna, iEFNs on leaves and non-iEFNs on stipules serve the same function of attracting ants around young developing leaves. However, leaves and stipules provide somewhat different opportunities for EFN allocation and nectar presentation to ants. Legume leaves are compound (with variable numbers of pairs of leaflets) and larger in size than stipules, thus possessing more opportunities for allocation of EFNs and timing of nectar presentation during leaf development. Furthermore, Senna leaves are long-lived, persisting throughout the year in many species. Secretory stipule lobes may attract ants early in leaf development, before leaf expansion. However, compared with leaves, stipules are short-lived organs, drying out and often falling off as leaves age. Therefore, the entire EFN is lost as stipules fall off. Similarly, iEFNs at the base of pedicels and non-iEFNs on bracts and calyx both serve to attract ants to floral buds and later developing fruits. While iEFNs are located at the base of pedicels and near the buds, and persist during inflorescence development, non-iEFNs on bracts and sepals attract ants directly on the developing bud, but bracts and sepals usually fall off before or when flowers open. iEFNs at the base of pedicels are unlikely to interfere with pollination of Senna flowers: flowers lack nectar, and pollen is concealed in tubular, lignified anthers accessible only to buzz-pollinating bees (Buchmann, 1974; Marazzi et al., 2007).
How and to what extent EFN diversity relates to, and/or influences the ecology of, ant–plant protective mutualisms is an open question. Comparative ecological studies are necessary to test whether different locations of EFNs (i.e. leaves, stipules, sepals, bracts, base of pedicels) differ in their effectiveness in attracting ants and, thus, potentially in their ability to protect developing leaves, flowers and fruits. Furthermore, comparative studies of nectar production would allow us to identify whether differences between iEFNs and non-iEFNs and among various iEFNs are associated with nectar secretion and presentation patterns. These in turn may be associated with the effectiveness of EFNs in mediating protective ant–plant mutualisms. Speculating further, if leaves with iEFNs are more effective than stipules in mediating protective ant–plant mutualisms, we would expect higher seedling survival rates and seed dispersal success in species with iEFNs on their leaves compared with those with non-iEFNs on stipules. Higher effectiveness of EFNs would contribute to support the key innovation role assumed by iEFNs in triggering the large-scale diversification of the EFN clade (Marazzi and Sanderson, 2010).
CONCLUSIONS
Our study of Senna shows that evolution of EFN diversity can be complex, resulting from multiple origins and diversification histories with different levels of morphological evolution involving diverse plant parts and EFN morphologies. Morphological development and differentiation of EFNs depend upon morphological development of the EFN-bearing plant part. Therefore, what plant part is involved plays an important role in EFN evolution. From the EFN perspective, this role is partly embodied in the concept of EFN individualization: increasing individualization correlates with increasing independence of the morphology and evolution of the EFN from the morphology and evolution of its bearing organ.
EFN diversity now needs to be investigated in a larger phylogenetic context. Indeed, iEFNs and non-iEFNs do not occur in Senna alone, but in several other lineages across the legume family. Recent analyses (Marazzi et al., 2012) suggest that the exceptional diversity of legume EFNs is best explained as having arisen from shared evolutionary (genetic or developmental) precursors rather than multiple ‘de novo’ origins and many recent losses. Disentangling the genetic programmes underlying EFN development and identifying possible ancestral morphological structures from which modern EFNs are derived are both crucial to understanding the origin of EFNs and the evolution of EFN diversity.
The present study is one of very few (Heil et al., 2004; Weber et al., 2012) to investigate non-reproductive traits mediating plant–animal interactions in a phylogenetic framework. Generalization from this work on the diversity and evolution of EFNs in Senna to other ecologically important and widespread traits involved in plant–animal mutualisms is conceivable. The location on plants of such traits corresponds to where the interactions themselves occur. In both reproductive and non-reproductive plant parts, trait location is determined by the plant organs that bear the traits and provide the structural material and support for the traits' morphological differentiation and presentation. Only by considering the diversity of trait-bearing organs and trait morphologies in ecological studies of mutualistic interactions can we fully appreciate and understand the evolution, ecology and evolutionary role of both the traits and the mutualisms in triggering lineage diversification.
SUPPLEMENTARY DATA
ACKNOWLEDGEMENTS
We thank M. Johnson and K. Coppola (greenhouse and plant care), A. Agellon (assistance with SEM), A. Grantham (assistance with paraffin microtome sections), H.-P. Linder (use of light microscope with digital camera), L. Paganucci de Queiroz (photos and samples of Senna martiana), D. Cardoso (photos of S. bracteosa), A. Weaving/Ardea.com (photos of S. italica), Peter Endress and the Bronstein and Conti labs (stimulating discussions), Jeff Karron and two anonymous reviewers (valuable comments on the manuscript), and the Arizona-Sonora Desert Museum, Campus Arboretum of the University of Arizona, Desert Botanical Garden and Desert Legume Program (cultivated specimens). This work was supported by the Swiss National Science Foundation (postdoctoral fellowship PA00P3-126248 to B.M.) and the National Geographic Research Program (8775-10 to B.M. with J.L.B., M.M.M. and M.J.S.). The Swiss Academy of Sciences supported early field-collecting trips (to B.M.).
LITERATURE CITED
- Baum SF, Eshed Y, Bowman JL. The Arabidopsis nectary is an ABC-independent floral structure. Development. 2001;128:4657–4667. doi: 10.1242/dev.128.22.4657. [DOI] [PubMed] [Google Scholar]
- Beattie AJ. The evolutionary ecology of ant–plant mutualisms. Cambridge: Cambridge University Press; 1985. [Google Scholar]
- Bentham G. Revision of the genus Cassia. Transactions of the Linnean Society of London. 1871;27:503–591. [Google Scholar]
- Bentley BL. Extrafloral nectaries and protection by pugnacious body-guards. Annual Review of Ecology and Systematics. 1977;8:407–427. [Google Scholar]
- Bernardello G. A systematic survey of floral nectaries. In: Nicolson SW, Nepi M, Pacini E, editors. Nectaries and nectar. Dordrecht: Springer: 2007. pp. 19–128. [Google Scholar]
- Bhattacharyya B, Maheshwari JK. Studies on extrafloral nectaries of the Leguminales. II. The genus Cassia Linn. (Caesalpiniaceae) Proceedings of the National Institute of Sciences of India. 1970;37(part b, 2):74–90. [Google Scholar]
- Blüthgen N, Reifenrath K. Extrafloral nectaries in an Australian rainforest: structure and distribution. Australian Journal of Botany. 2003;51:515–527. [Google Scholar]
- Bowman JL, Smyth DR. CRABS CLAW, a gene that regulates carpel and nectary development in Arabidopsis, encodes a novel protein with zinc finger and helix–loop–helix domains. Development. 1999;126:2387–2396. doi: 10.1242/dev.126.11.2387. [DOI] [PubMed] [Google Scholar]
- Bravo L. El género Cassia en la Argentina, I. Serie Aphyllae. Darwiniana. 1978;21:343–391. [Google Scholar]
- Bronstein JL, Alarcón R, Geber M. The evolution of plant–insect mutualisms. New Phytologist. 2006;172:412–428. doi: 10.1111/j.1469-8137.2006.01864.x. [DOI] [PubMed] [Google Scholar]
- Buchmann SL. Buzz pollination of Cassia quiedondilla (Leguminosae) by bees of the genera Centris and Melipona. Bulletin of the Southern California Academy of Science. 1974;73:171–173. [Google Scholar]
- Bush A, Zachgo S. Flower symmetry evolution: towards understanding the abominable mystery of angiosperm radiation. Bioessays. 2009;31:1181–1190. doi: 10.1002/bies.200900081. [DOI] [PubMed] [Google Scholar]
- Caspary R. De nectariis. 1848 Bonn: Nieuwenhuis-von Uxkiull-Giildenbandt (1907, pp. 203–206).]Adolphum Marcum. [Abstract in Bot. Zeitung 6: 628–630 and extensive summaries in Behrens (1879, ppp. 19–22) and Nieuwenhuis-von Uxkiull-Giildenbandt (1970, pp.203–206).] [Google Scholar]
- Darwin C. The effects of cross and self-fertilisation in the vegetable kingdom. London: John Murray; 1876. [Google Scholar]
- Darwin F. On the nectar glands of the common brakefern. Botanical Journal of the Linnean Society. 1877;15:407–409. [Google Scholar]
- Delpino F. Ulteriori osservazioni sulla dicogamia nel regno vegetale. 1868–1875 I & II. Atti della Società Italiana di Scienze Naturali, XI, XII. [Google Scholar]
- Díaz-Castelazo C, Rico-Gray V, Oliveira PS, Cuautle M. Extrafloral nectary-mediated ant–plant interactions in the coastal vegetation of Veracruz, Mexico: richness, occurrence, seasonality, and ant foraging patterns. Ecoscience. 2004;11:472–481. [Google Scholar]
- Elias TS, Gelband H. Morphology and anatomy of floral and extrafloral nectaries in Campsis (Bignoniaceae) American Journal of Botany. 1976;63:1349–1353. [Google Scholar]
- Fahn A. Secretory tissues in plants. London: Academic Press; 1979. [Google Scholar]
- Friis EM, Pedersen KR, Crane PR. Cretaceous angiosperm flowers: innovation and evolution in plant reproduction. Palaeogeography Palaeoclimtology Palaeoecolecology. 2006;232:251–293. [Google Scholar]
- Gould SJ. On the importance of heterochrony for evolutionary biology. Systematic Zoology. 1979;28:224–226. [Google Scholar]
- Hall BK. Developmental processes underlying heterochrony as an evolutionary mechanism. Canadian Journal of Zoology. 1984;62:1–7. [Google Scholar]
- Heil M, McKey D. Protective ant–plant interactions as a model system in ecological and evolutionary research. Annual Review of Ecology, Evolution and Systematics. 2003;34:425–453. [Google Scholar]
- Heil M, Greiner S, Meimberg H, et al. Evolutionary change from induced to constitutive expression of an indirect plant resistance. Nature. 2004;430:205–208. doi: 10.1038/nature02703. [DOI] [PubMed] [Google Scholar]
- Hodges SA. Floral nectar spurs and diversification. International Journal of Plant Sciences. 1997;158(Suppl.):S81–S88. [Google Scholar]
- Hodges SA, Arnold ML. Spurring plant diversification: are floral nectar spurs a key innovation? Proceedings of the Royal Society of London B. 1995;262:343–348. [Google Scholar]
- Igersheim A. The character states of the Caribbean monotypic endemic Strumpfia (Rubiaceae) Nordic Journal of Botany. 1993;13:545–559. [Google Scholar]
- Irwin HS, Barneby RC. The American Cassiinae. Memoirs of the New York Botanical Gardens. 1982;35:1–918. [Google Scholar]
- Keeler KH. Distribution of plants with extrafloral nectaries in temperate communities. American Midland Naturalist. 1980;104:274–280. [Google Scholar]
- Keeler KH. World list of plants with extrafloral nectaries. 2008 http://biosci-labs.unl.edu/Emeriti/keeler/extrafloral/Cover.htm . [Google Scholar]
- Kluge AG, Strauss RE. Ontogeny and systematics. Annual Review of Ecology and Systematics. 1985;16:247–268. [Google Scholar]
- Koptur S. Extrafloral nectary-mediated interactions between insects and plants. In: Bernays E, editor. Insect–plant interactions. IV. London: CRC Press; 1992. pp. 81–129. [Google Scholar]
- Janzen DH. Dispersal of small seeds by big herbivores: foliage is the fruit. American Naturalist. 1984;123:338–353. [Google Scholar]
- Lee JY, Baum SF, Alvarez J, Patel A, Chitwood DH, Bowman JL. Activation of CRABS CLAW in the nectaries and carpels of Arabidopsis. The Plant Cell. 2005a;17:25–36. doi: 10.1105/tpc.104.026666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JY, Baum SF, Oh SH, Jiang CZ, Chen JC, Bowman JL. Recruitment of CRABS CLAW to promote nectary development within the eudicot clade. Development. 2005b;132:5021–5032. doi: 10.1242/dev.02067. [DOI] [PubMed] [Google Scholar]
- Lengyel S, Gove AD, Latimer AM, Majer JD, Dunn RR. Ants sow the seeds of global diversification in flowering plants. PLoS ONE. 2009;4:e5480. doi: 10.1371/journal.pone.0005480. http://dx.doi.org/10.1371/journal.pone.0005480 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lengyel S, Gove AD, Latimer AM, Majer JD, Dunn RR. Convergent evolution of seed dispersal by ants, and phylogeny and biogeography in flowering plants: a global survey. Perspectives in Plant Ecology, Evolution and Systematics. 2010;12:43–55. [Google Scholar]
- Lewis G, Schrire B, Mackinder B, Lock M. The legumes of the world. Kew, London: Royal Botanic Gardens; 2005. [Google Scholar]
- Lunau K. Adaptive radiation and coevolution – pollination biology case studies. Organisms Diversity & Evolution. 2004;4:207–224. [Google Scholar]
- Machado SR, Morellato LPC, Sajo MG, Oliveira PS. Morphological patterns of extrafloral nectaries in woody plant species of the Brazilian cerrado. Plant Biology. 2008;10:660–673. doi: 10.1111/j.1438-8677.2008.00068.x. [DOI] [PubMed] [Google Scholar]
- Maddison WP, Maddison DR. Mesquite: a modular system for evolutionary analysis. Version 2.75. 2011 http://mesquiteproject.org . [Google Scholar]
- Marazzi B, Sanderson MJ. Large-scale patterns of diversification in the widespread legume genus Senna and the evolutionary role of extrafloral nectaries. Evolution. 2010;64:3570–3592. doi: 10.1111/j.1558-5646.2010.01086.x. [DOI] [PubMed] [Google Scholar]
- Marazzi B, Endress PK, Paganucci de Queiroz L, Conti E. Phylogenetic relationships within Senna (Leguminosae, Cassiinae) based on three chloroplast regions: patterns in the evolution of floral symmetry and extrafloral nectaries. American Journal of Botany. 2006a;93:288–303. doi: 10.3732/ajb.93.2.288. [DOI] [PubMed] [Google Scholar]
- Marazzi B, Fortunato R, Endress PK, Spichiger R. Senna (Cassiinae, Leguminosae) in Paraguay: synopsis, conservation, ecologic role and ethnobotany. Candollea. 2006a;61:315–329. [Google Scholar]
- Marazzi B, Conti E, Endress PK. Diversity of anthers and stigmas in the buzz-pollinated genus Senna (Leguminosae, Cassiinae) International Journal of Plant Science. 2007;168:371–391. [Google Scholar]
- Marazzi B, Ané C, Simon M, et al. Locating evolutionary precursors on a phylogenetic tree. Evolution. 2012;66:3918–3930. doi: 10.1111/j.1558-5646.2012.01720.x. [DOI] [PubMed] [Google Scholar]
- McKey D. Interactions between ants and leguminous plants. In: Stirton CH, Zarucchi JL, editors. Monographs in Systematic Botany from the Missouri Botanical Garden. Vol. 29. 1989. pp. 673–718. Advances in legume biology. [Google Scholar]
- Melo Y, Machado SR, Alves M. Anatomy of extrafloral nectaries in Fabaceae from dry-seasonal forest in Brazil. Botanical Journal of the Linnean Society. 2010;163:87–98. [Google Scholar]
- Nepi M. Nectary structure and ultrastructure. In: Nicolson SW, Nepi M, Pacini E, editors. Nectaries and nectar. Dordrecht: Springer: 2007. pp. 129–166. [Google Scholar]
- van der Niet T, Johnson SD. Phylogenetic evidence for pollinator-driven diversification of angiosperms. Trends in Ecology and Evolution. 2012;27:353–361. doi: 10.1016/j.tree.2012.02.002. [DOI] [PubMed] [Google Scholar]
- Pascal LM, Motte-Florac EF, McKey DB. Secretory structures on leaf rachis of Caesalpinieae and Mimosoideae (Leguminosae): implications for the evolution of nectary glands. American Journal of Botany. 2000;87:327–338. [PubMed] [Google Scholar]
- Pemberton RW. The abundance of plants bearing extrafloral nectaries in Colorado and Mojave Desert communities of southern California. Modroño. 1988;35:238–246. [Google Scholar]
- Raff RA, Kaufman TC. Embryos, genes, and evolution: The developmental-genetic basis of evolutionary change. New York: Macmillan; 1983. [Google Scholar]
- Randell BR. Revision of the Cassiinae in Australia. 1. Senna Miller Sect. Chamaefistula (Colladon) Irwin and Barneby. Journal of the Adelaide Botanic Gardens. 1988;11:19–49. [Google Scholar]
- Randell BR. Revision of the Cassiinae in Australia. 2. Senna Miller Sect. Psilorhegma (J. Vogel) Irwin and Barneby. Journal of the Adelaide Botanic Gardens. 1989;12:165–272. [Google Scholar]
- Rico-Gray V, Oliveira PS. The ecology and evolution of ant–plant interactions. Chicago: University Chicago Press; 2007. [Google Scholar]
- Sargent RD. Floral symmetry affects speciation rates in angiosperms. Proceedings of the Royal Society of London B. 2004;271:603–608. doi: 10.1098/rspb.2003.2644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid R. Reproductive versus extra-reproductive nectaries – historical perspective and terminological recommendations. Botanical Review. 1988;54:179–232. [Google Scholar]
- Schoonhoven LM, van Loon JJA, Dicke M. Insect–plant interactions, 2nd edn. New York: Oxford University Press; 2005. [Google Scholar]
- Schupp EW, Feener DH. Phylogeny, life form, and habitat dependence of ant-defended plants in a Panamanian forest. In: Huxley CR, Cutler DF, editors. Ant–plant interactions. Oxford: Oxford University Press; 1991. pp. 175–197. [Google Scholar]
- Vogel S. Remarkable nectaries: structure, ecology, organophyletic perspectives. I. Substitutive nectaries. Flora. 1997;192:305–333. [Google Scholar]
- Weber M, Igersheim A. Pollen buds in Ophiorrhiza (Rubiaceae) and their role in pollenkit release. Botanica Acta. 1994;107:257–262. [Google Scholar]
- Weber MG, Keeler KH. The phylogenetic distribution of extrafloral nectaries in plants. Annals of Botany. 2013;111:1251–1261. doi: 10.1093/aob/mcs225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber MG, Clement WL, Donoghue MJ, Agrawal AA. Phylogenetic and experimental tests of interactions among mutualistic plant defense traits in Viburnum (Adoxaceae) The American Naturalist. 2012;180:450–463. doi: 10.1086/667584. [DOI] [PubMed] [Google Scholar]
- Zimmermann JG. Über die extrafloralen Nektarien der Angiospermen. Beihefte zum Botanischen Zentralblatt. 1932;49:99–196. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.