SUMMARY
At sites of inflammation, certain regulatory T cells (Treg cells) can undergo rapid reprogramming into helper-like cells, without loss of the transcription factor Foxp3. We show that reprogramming is controlled by down-regulation of the transcription factor Eos (Ikzf4), an obligate co-repressor for Foxp3. Reprogramming was restricted to a specific subset of “Eoslabile” Treg cells which were present in the thymus and identifiable by characteristic surface markers and DNA methylation. Mice made deficient in this subset became impaired in their ability to provide help for presentation of new antigens to naive T cells. Down-regulation of Eos required the pro-inflammatory cytokine IL-6, and mice lacking IL-6 had impaired development and function of the Eos-labile subset. Conversely, the immunoregulatory enzyme IDO blocked loss of Eos, and prevented the Eos-labile Treg cells from reprogramming. Thus, the Foxp3+ lineage contains a committed subset of Treg cells capable of rapid conversion into biologically important helper cells.
INTRODUCTION
Foxp3+ regulatory T cells (Treg cells) are essential to maintain self tolerance. However, some Treg cells also retain a degree of plasticity, and under certain conditions may change (“reprogram”) to adopt a pro-inflammatory phenotype (da Silva Martins and Piccirillo, 2012). This has been observed under conditions including lymphopenia (Komatsu et al., 2009; Yurchenko et al., 2012), autoimmunity (Zhou et al., 2009) and sepsis (Oldenhove et al., 2009). Because these are pathologic conditions, it has been somewhat controversial whether Treg reprogramming is a rare and abnormal occurrence, or a normal part of Treg cell biology. Recently, emerging studies suggest that destabilization of the Treg cell phenotype is a real phenomenon, and can have a major impact on the course of inflammatory responses in vivo (Laurence et al., 2012).
Under most circumstances the role of Foxp3+ Treg cells is clearly suppressive (Kim et al., 2007). However, in certain settings Treg cells may adopt a paradoxical “helper-like” role: this includes contexts as diverse as vaccination (Vendetti et al., 2010), tumors (Addey et al., 2011), graft rejection (Vokaer et al., 2010) and Th2 cell responses (Tsuji et al., 2009; Wang et al., 2010). In most of these studies the “helper-like” Treg cells appeared to have lost Foxp3. However, this finding would seem to be at odds with studies suggesting that Foxp3 expression is highly stable in Treg cells once established (Miyao et al., 2012; Rubtsov et al., 2010). Thus, there has been some controversy over whether fully-committed Foxp3-lineage Treg cells ever transform into helper cells.
In the current study, we asked whether inducible transformation into helper cells might in fact be a natural property of certain Foxp3+ cells – not as a loss of lineage fidelity, but as a normal role which some Treg cells inherently adopt under specific circumstances. This hypothesis arose from our previous observation that when Treg cells enter an inflammatory milieu, such as LNs draining a site of vaccination, some of them can rapidly convert into a pro-inflammatory phenotype, while still expressing Foxp3 (Sharma et al., 2010; Sharma et al., 2009). These reprogrammed Treg cells were found to play an important functional role in licensing DCs to present new antigens in naive hosts (Sharma et al., 2010).
However, these observations raised a paradox: because reprogrammed Treg cells continued to express Foxp3, how were they able to express pro-inflammatory genes that are normally repressed by Foxp3? In the current study we show that this phenotypic transformation is controlled by the co-repressor Eos (Ikzf4). Eos is a member of the Ikaros family of transcription factors that also includes Aiolos and Helios (Quintana et al., 2012; Thornton et al., 2010). Eos forms a complex with Foxp3, and is required in order for Foxp3 to inhibit its downstream target genes and maintain the suppressive Treg cell phenotype (Pan et al., 2009). We now show that downregulation of Eos, rather than loss of Foxp3, underlies rapid reprogramming of Treg cells in response to inflammation; and we identify a subset of Foxp3+ Treg cells that are constitutively committed to loss of Eos under specific inflammatory conditions.
RESULTS
Foxp3 is stable in reprogrammed Treg cells, but Eos is downregulated
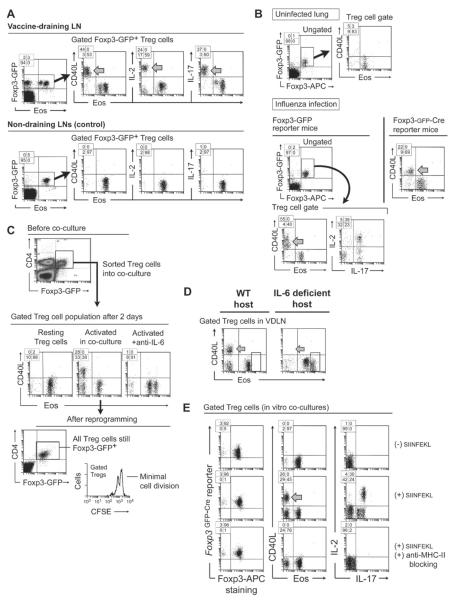
To create local inflammation, mice received CD8+ OT-I T cells specific for ovalbumin (OVA), followed by vaccination with whole OVA protein in incomplete Freund's adjuvant (IFA) plus the TLR9 ligand CpG. Four days later, Treg cells in vaccine-draining lymph nodes (VDLNs) were analyzed by flow cytometry for expression of Eos. Validation of intracellular Eos staining is shown in Figure S1A, available online. As markers of reprogramming, Treg cells were stained for IL-2, IL-17 and cell-surface CD40L. As controls, distant LNs, not draining the vaccination site, were taken from the same mouse. To identify Treg cells, host mice bore either a Foxp3-GFP reporter knock-in (Foxp3GFP mice) (Fontenot et al., 2005), or a BAC-transgenic GFP-Cre reporter under a Foxp3 promoter construct (Zhou et al., 2009). All experiments were confirmed in both reporter strains, and were further confirmed using intracellular Foxp3 staining in WT mice.
Figure 1A shows that in the control (distant) LNs virtually all Treg cells expressed Eos, and none expressed detectable CD40L, IL-2 or IL-17. In contrast, in the inflammatory VDLNs a population of Treg cells had lost Eos (arrows), and many of these had up-regulated IL-2, IL-17 and CD40L. In our system, surface CD40L correlated closely with the reprogrammed phenotype (Sharma et al., 2010), and was thus a useful marker for these cells. None of the reprogrammed Treg cells had lost Foxp3 (left-hand dot plots); rather, it was specifically the loss of Eos that correlated with the altered phenotype. Figure S1B shows that loss of Eos was rapid, starting within 6 hrs of vaccination.
Figure 1. Treg cells that undergo reprogramming continue to express Foxp3 but downregulate Eos.
(A) Foxp3GFP mice received OT-I cells plus vaccination with whole OVA protein + CpG adjuvant in IFA. After 4 days cells were harvested from VDLNs, or control non-draining LNs from the same mouse, and stained as shown. Inset shows the percentage of events in each quadrant.
(B) Foxp3GFP mice were infected intranasally with influenza virus strain X31. After 5 days, mononuclear cells were isolated from lung. Experiments were also replicated in Bactransgenic Foxp3GFP-Cre reporter mice (right-hand dot-plot).
(C) In vitro co-culture system: activated DCs were sorted from VDLNs two days after OT-I + vaccination. Resting splenic Treg cells were sorted from unimmunized Foxp3GFP mice. DCs and Treg cells were combined with additional OT-I cells plus SIINFEKL antigen in vitro, and co-cultured for 2 days. Where indicated, IL-6 was blocked with neutralizing antibody. Histogram shows Treg cell division from a similar experiment, using B6 Treg cells (CD4+CD25+) labeled with CFSE.
(D) IL-6 deficient (Il6−/−) mice or WT B6 controls received OT-I cells + OVA vaccine, then Treg cells (CD4+CD25+) from VDLNs were analyzed for reprogramming on day 4.
(E) Treg cells were sorted from BAC-transgenic Foxp3GFP-Cre mice and used in co-culture assays as in panel C, with or without siinfekl peptide and MHC-II blocking antibody (anti-IAb) as indicated.
Each panel is representative of between 3 and 9 experiments with similar results. See also Figure S1.
Eos was also downregulated in the setting of a mild, non-lethal influenza infection (Figure 1B). Many of these Eos-negative Treg cells upregulated CD40L (arrow) and co-expressed IL-2 and IL-17. Despite reprogramming, Treg cells continued to express Foxp3. The number of Eos-negative Treg cells during infection varied but they were not rare, showing that reprogramming could occur under conditions of mild physiologic inflammation.
Thus, under certain conditions, a population of Treg cells was able to downregulate expression of the co-repressor Eos and upregulate pro-inflammatory genes, without loss of Foxp3.
Down-regulation of Eos requires IL-6
To elucidate the pathways driving reprogramming, we used an in vitro co-culture model, as previously described (Sharma et al., 2010). Resting splenic Treg cells were co-cultured with activated DCs from VDLNs. To recapitulate the inflammatory milieu, co-cultures also received OT-I effector T cells plus cognate siinfekl antigen. Figure 1C shows that after 2 days in co-culture a substantial number of Treg cells had lost Eos, and many of these had upregulated CD40L (middle dot-plots), but all Treg cells continued to express the Foxp3GFP reporter (lower dot-plot). In experiments using CFSE-labeled Treg cells (bottom histogram), reprogramming could occur at a time when proliferation was still minimal.
In other systems, the pro-inflammatory cytokine IL-6 has been shown to drive Treg cell reprogramming (Yang et al., 2008). When we added neutralizing anti-IL-6 to co-cultures (Figure 1C, right-hand panel) this prevented loss of Eos and blocked functional reprogramming. Consistent with this, when mice lacking IL-6 (Il6−/− mice (Kopf et al., 1994)) were immunized in vivo, their Treg cells failed to undergo reprogramming (Figure 1D). Thus, IL-6 appeared to be an important driver for loss of Eos and reprogramming.
However, IL-6 did not act in isolation. Adding IL-6 alone to activated Treg cells did not cause reprogramming (Figure S1C). Rather, reprogramming required integrated signals from activated DCs, activated effector cells, and cognate MHC class II (Figure 1E). If antigen for the OT-I effector T cells was omitted (Figure 1E, top row) then Treg cell reprogramming could not occur. One key reason for this was that the activated effector cells were needed to trigger IL-6 expression by the DCs (Figure S1E). Further, it was necessary that the DCs be derived from an activated milieu (e.g., from LNs draining a vaccine site or tumor) rather than resting DCs (Figure S1E). Finally, reprogramming required cognate interaction between the Treg cells and activated DCs via MHC class II (Figure 1E, bottom row). Under all these various conditions, Foxp3 expression remained unchanged (Figure 1E, left-hand dot-plots; however, for technical caveats regarding intracellular staining of Foxp3 after reprogramming, see Supplemental Figure S1E.).
Taken together, these data showed that the combination of activated DCs, derived from an inflammatory milieu, plus antigen-activated effector T cells, together drove reprogramming of Treg cells. Under these conditions, IL-6 was a key cytokine mediating Eos downregulation.
Down-regulation of Eos is mechanistically required in order for Treg cells to undergo reprogramming
To test the mechanistic role of Eos, we forced over-expression of Eos in resting Treg cells by retroviral transduction and asked if this prevented reprogramming. For transduction studies T cells would normally be activated with mitogen, because retroviruses require host cells to be in cell-cycle; however, a nonspecific mitogen would have completely altered the physiologic response of resting Treg cells to reprogramming signals. Therefore we developed the transduction system shown in Figure S2, which took advantage of the fact that many resting Treg cells are already constitutively in cell cycle without the need for mitogen (O'Gorman et al., 2009).
Resting Treg cells were pre-incubated with bicistronic MSCV-based retroviral vector overnight in un-activated co-cultures (i.e., no SIINFEKL peptide). After 24 hrs the virus was washed off and SIINFEKL added to drive reprogramming. Under this protocol, typically 20–40% of Treg cells were transduced, with the transduced Treg cells being specifically those that were in cell-cycle (Ki67+, as shown in Figure S2). Essentially no other cells were transduced, since there was no mitogen to drive their proliferation at the time of virus exposure.
Figure 2A shows that Treg cells transduced with the control GFP vector and then exposed to reprogramming conditions underwent extensive loss of Eos; whereas Treg cells transduced with Eos vector remained >80% Eos+ under the same conditions, with many becoming EosHI. Figure 2B shows that Treg cells transduced with control vector displayed extensive phenotypic reprogramming (CD40L, IL-2, IL-17); while the same Treg cells transduced with Eos vector could not reprogram, and most retained high surface CTLA-4. In these cultures, Treg cells with forced over-expression of Eos created a dominantly suppressive milieu that affected all cells in co-culture, so the non-transduced population in those wells did not serve as a true, independent control. However, the vector-only co-cultures were true controls, and in those both transduced and non-transduced cells underwent reprogramming. Consistent with the role of IL-6 in driving Eos down-regulation, adding high levels of recombinant IL-6 to co-cultures could overcome transduction and force Eos downregulation and reprogramming (Figure 2C).
Figure 2. Forced over-expression of Eos in Treg cells prevents reprogramming.
(A, B, C) Resting B6 Treg cells (CD4+CD25+) were pre-incubated with retroviral vector (control GFP-only or Eos-GFP) in co-cultures as described in Supplemental Figure S2. After 24 hrs, co-cultures were washed free of vector and siinfekl added to trigger reprogramming. Cells were analyzed 2 days later. (A) expression of Eos in GFP-expressing (transduced) cells. (B) markers of reprogramming after transduction. (C) Addition of high-dose IL-6 (50 ng/ml) to co-cultures could overcome Eos transduction and force reprogramming.
(D) Rescue of CD40L-deficient hosts by reprogrammed Tregs. Cd40lg−/− hosts received adoptive transfer of CD40L-sufficient Foxp3GFP-Thy1.1 Treg cells or non-regulatory CD4+ cells. Mice then received CFSE-labeled OT-I cells plus OVA vaccine as in Figure 1A, and VDLNs analyzed on day 4.
(E) Similar to the preceding panel, but Thy1.1 Treg cells (without transgene, sorted CD4+CD25+) were first transduced in vitro with Eos-GFP vector or control GFP-only vector. Treg cells were recovered by CD4 bead-sorting, transferred into Cd40lg−/− hosts, and immunized. (Since the Eos-transduced Treg cells were dominantly suppressive, it was not necessary to separate them from the untransduced Treg cells.)
Each panel is representative of between 3 and 5 independent experiments with similar results. See also Figure S2.
Thus, loss of Eos was not simply a passive concomitant of Treg cell reprogramming, but rather was a required mechanism underlying the switch to a pro-inflammatory phenotype. This was consistent with the known role of Eos as a co-repressor, required in order for Foxp3 to silence pro-inflammatory target genes (Pan et al., 2009).
Forced overexpression of Eos abrogates the functional helper activity of reprogrammed Treg cells
To test the functional helper activity of reprogrammed Treg cells, we asked whether these cells could rescue the profound helper defect in mice lacking CD40L (Cd40lg−/− mice), as previously described (Sharma et al., 2010). CD40L is critical in order to “license” DCs, and Cd40lg−/− mice are unable to prime naive CD8+ cells to OVA protein (Grewal et al., 1996). Figure 2D shows that when CD40L-sufficient WT Treg cells were adoptively transferred into Cd40lg−/− hosts, many of them reprogrammed following vaccination and upregulated CD40L, and this rescued the CD8+ response to OVA vaccine (left panels). In contrast, non-regulatory CD4+ cells were unable to upregulate CD40L within the course of these assays, and could not rescue priming in Cd40lg−/− hosts (right panels).
Using this model, we next asked whether forced over-expression of Eos abrogated the ability of Treg cells to function as helper cells in vivo (Figure 2E). CD4+CD25+ Treg cells were transduced with Eos vector or control vector as above, then transferred into Cd40lg−/− hosts. Following immunization, Treg cells transduced with control vector were able to reprogram, express CD40L and provide help for OT-I priming. In contrast, Treg cells transduced with Eos could not upregulate CD40L and did not provide help for OT-I.
Thus, the phenotypic changes mediated by the downregulation of Eos reflected functional helper-cell activity in vivo. Specifically, the upregulation of CD40L by reprogrammed Treg cells allowed them to rescue the helper-cell defect in Cd40lg−/− hosts.
Identification of a specific subset of Treg cells with labile Eos expression
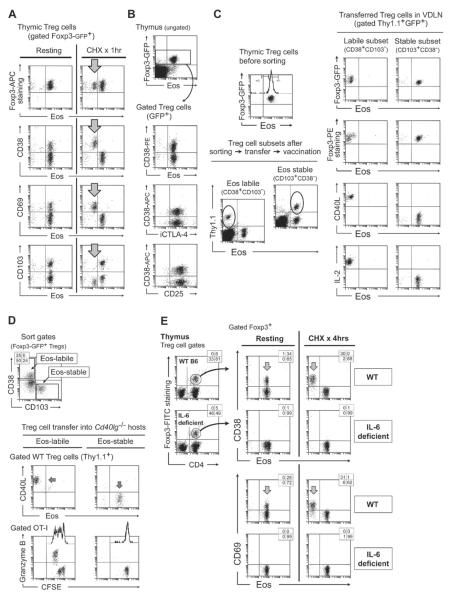
We noted that only a subset of Treg cells lost Eos and underwent reprogramming. We therefore asked if there might be a specific population of Treg cells in which Eos was inherently more unstable. As one marker for this instability, we examined the decay kinetics (half-life) of the Eos protein when new protein synthesis was blocked by the inhibitor cycloheximide (CHX). This was a purely biochemical test, and did not measure Treg cell reprogramming itself; rather, the goal was merely to ask whether Eos was less stable in certain Treg cells than others. (Likewise, any off-target effects of CHX (Hershko et al., 2004) were not problematic for the assay, because the goal was simply to disclose any Treg cells with unstable Eos.) For these studies we tested cells from thymus, where the Foxp3+ cells would be bona fide natural Treg cells. Figure 3A shows that thymic Treg cells displayed two distinct responses to CHX: most maintained stable Eos expression, but a subset showed rapid loss of Eos, with ~10-fold decrease within 1 hour. By screening a number of surface markers, we determined that this “Eos-labile” population was identified by expression of CD38, a surface antigen found on a subset of Treg cells (Fisson et al., 2003) and other lymphocytes (Figure S3A). The same population also co-expressed the activation marker CD69, while none of them expressed the maturation marker CD103. The Eos-labile subset of Treg cells expressed the same levels of Foxp3 as the Eos-stable subset (upper dot plots in Figure 3A), and expressed similar patterns of CD25 and intracellular CTLA-4 (Figure 3B). In the periphery, similar Eos-labile Treg cells were also found, with the same phenotype of CD38+CD103NEG, as shown in Figure S3B.
Figure 3. A specific subset of Treg cells with the ability to reprogram is identified by labile Eos expression at rest.
(A) Thymic cells from Foxp3GFP mice were disaggregated and incubated with cycloheximide (CHX) or vehicle control (“resting”) for 1 hr in vitro as described in Supplemental Procedures, then stained for Eos vs. cell-surface markers. Arrows indicate the Eos-labile subset. Representative of 15 similar experiments, using both Foxp3GFP and WT B6 mice.
(B) Resting thymic Treg cells showed similar expression of Foxp3, Eos, intracellular CTLA-4 and surface CD25 between CD38+ (Eos-labile) and CD38NEG (Eos-stable) subsets.
(C) Thymic cells from Foxp3GFP-Thy1.1 donors were incubated with CHX for 1 hr to improve separation of sorting markers, then Treg cells sorted into Eos-labile (CD38+CD103NEG) and Eos-stable (CD38NEGCD103+) cohorts and transferred into B6 mice. Mice were challenged with OT-I + vaccine and VDLNs analyzed on day 4. Representative of 4 experiments.
(D) Splenic Treg cell subsets were sorted as in the previous panel, and transferred into Cd40lg−/− hosts. Mice received CFSE-labeled OT-I + vaccine, and VDLNs analyzed on day 4. Representative of 5 experiments.
(E) Thymic cells from IL-6 deficient mice or WT B6 controls were tested in a 4 hr CHX assay, then stained for CD38 and CD69 as markers of the Eos-labile Treg cell subset. Examples of the Foxp3 gating for each strain are shown at left.
Each panel representative of at least 3 experiments, or as noted. See also Figure S3.
Taken together, these studies suggested that an identifiable population of Treg cells, present as early as the thymus and with a characteristic pattern of cell-surface markers, displayed an inherently more labile pattern of Eos expression in vitro. Based on this, we next asked whether these markers identified the cells that were committed to functional reprogramming.
The Eos-labile phenotype identifies the subset of Treg cells capable of functional reprogramming
For cell-sorting experiments, we selected the markers CD38+CD103NEG to enrich for Eos-labile Treg cells, and CD103+CD38NEG for Eos-stable Treg cells. (Of note, this pattern of markers applied to resting Treg cells, before any activation.) We excluded all double-positive and double-negative Treg cells, since these were ambiguous; further, the markers became optimally separated after 1 hr of CHX pre-treatment, so this was used where highest purity was desired. Thymic Treg cells were sorted into Eos-labile and Eos-stable cohorts and transferred into new B6 hosts. Recipients received OT-I and vaccine, then 4 days later the transferred Treg cells were harvested from VDLNs (Figure 3C). Following vaccination, the transferred Eos-labile Treg cells lost Eos and underwent reprogramming; whereas the Eos-stable Treg cells maintained Eos expression and showed no reprogramming. In both groups the endogenous host Treg cells underwent reprogramming as expected (not shown), so the host milieu for both populations was the same.
To test for functional helper activity, we used the Cd40lg−/− model described in Figure 2. Eos-labile and Eos-stable Foxp3+ populations were sorted from spleen, and each transferred into Cd40lg−/− hosts (Figure 3D). Following immunization, the Eos-labile Treg cells lost Eos, upregulated CD40L and functionally rescued OT-I priming; whereas the Eos-stable Treg cells maintained Eos expression, did not reprogram, and could not rescue OT-I.
Thus, in both thymus and periphery, a subset of resting Treg cells displayed a characteristic pattern of surface markers that identified them as susceptible to functional reprogramming. This suggested that the propensity to reprogram reflected a stable property, established during Treg differentiation.
The Eos-labile subset requires IL-6 for intra-thymic development
Based on Figure 1C, above, we knew that IL-6 was an important driver of functional reprogramming. IL-6 is also constitutively expressed in the thymus (Yarilin and Belyakov, 2004). Therefore, we asked if IL-6 might be involved in the development of the Eos-labile subset. Figure 3E compares thymic Treg cells from WT mice versus IL-6 deficient mice (Il6−/−). Thymus from Il6−/− mice displayed a normal number of Treg cells, but selectively failed to develop the specific CD38+CD69+ subset that normally marked the Eos-labile population. Consistent with this, none of the Treg cells in Il6−/− thymus showed labile Eos expression in the CHX assay (right-hand dot-plots). Further consistent, when mature splenic Treg cells from Il6−/− mice were tested in reprogramming assays (Figure S3C), none of them were able to undergo functional reprogramming. This appeared to reflect an intrinsic defect in Treg cells from Il6−/− mice, because all other cells in these co-cultures were from normal WT donors. Taken together, these data suggested that IL-6 was required for differentiation of the Eos-labile (but not the Eos-stable) subset of Treg cells. In the absence of IL-6, the Eos-labile subset selectively failed to develop in thymus and periphery.
Help from Eos-labile Treg cells is required during priming of naive mice to new antigen
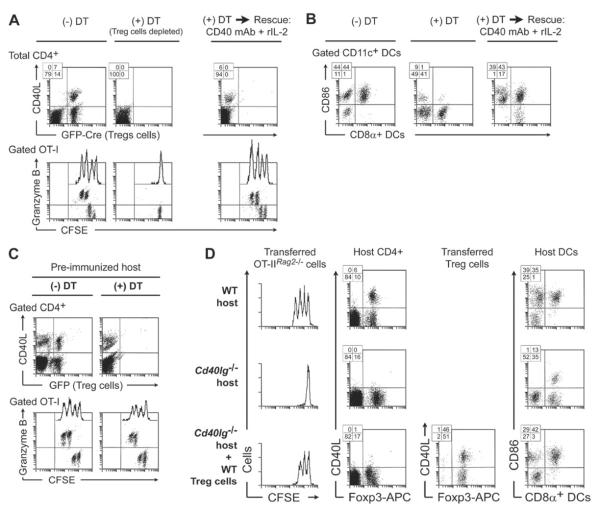
To further test whether help from reprogrammed Treg cells contributed to T cell priming, we used mice in which Treg cells could be inducibly ablated with diphtheria toxin (DT). This model allowed Treg cells to be selectively depleted from previously intact adult mice. However, it had the drawback that mice rapidly develop autoimmune inflammation when Treg cells are removed (Kim et al., 2007). Because of this limitation, we used only short-term readouts, and confirmed all results in the complementary Cd40lg−/− model. Foxp3GFP-Cre mice were crossed with a ROSA26lox-stop-loxDTR strain to produce mice expressing the DT receptor on all cells that had activated the Foxp3 promoter (Sharma et al., 2010). Without Treg cell depletion (Figure 4A, -DT group) OT-I cells activated normally in these mice, and virtually all of the CD40L-expressing cells in VDLNs were derived from reprogrammed Treg cells (marked by GFP-Cre). However, when Treg cells were depleted (+DT group) the expression of CD40L was markedly reduced during priming, and OT-I activation was compromised. Thus, consistent with the findings in the Cd40lg−/− model, help from reprogrammed Treg cells appeared important to support normal priming of naive CD8+ T cells. Further consistent with the Cd40lg−/− model, Figure S4A shows that adoptive transfer of the Eos-labile Treg cell subset (but not the Eos-stable subset) selectively restored the helper activity lost in DT-treated mice.
Figure 4. Help from reprogrammed Treg cells is required to support T cell priming in antigen-naive hosts.
(A, B) Foxp3GFP-Cre×ROSA26lox-stop-loxDTR mice received CFSE-labeled OT-I and OVA vaccine (day 0), with or without DT depletion on days −2, −1, +1 and +3. VDLNs were analyzed on day 4. Some groups treated with DT also received rescue with IL-2 (1 ug/dose every 12h on days 0–3) and agonist CD40 mAb (250 ug + 100 ug i.p. days 1 and 3). (A) T cell responses in VDLNs. (B) DC activation (CD86 expression) in VDLNs using the same treatment groups. Representative of 7 experiments, 2 with rescue.
(C) Prior to Treg cell depletion, Foxp3GFP-Cre×ROSA26lox-stop-loxDTR mice were pre-immunized with OVA vaccine (prime + boost × 2), then re-challenged with CFSE-labeled OT-I readout cells + OVA vaccine, with or without DT depletion. Response in VDLN is shown on day 4. One of 2 experiments.
(D) Cd40lg−/− or WT control mice received adoptive transfer of CFSE-labeled OT-IIRag2−/− CD4+ effector cells (similar to Figure S4C). Some Cd40lg−/− hosts were rescued by pre-transfer of CD40L-sufficient Tregs (Foxp3GFP-Thy1.1), as in Figure 2D. All mice were immunized with cognate peptide for OT-II cells in IFA+CpG, and responses measured in VDLNs. One of 2 experiments. See also Figure S4.
We next asked if the requirement for help from reprogrammed Treg cells could be replaced by adding back the specific molecular signals normally supplied by the depleted Treg cells. The right-hand panels in Figure 4A show that OT-I response could be fully rescued by treating Treg-depleted mice with a CD40-activating antibody plus recombinant IL-2 at the time of immunization. (This combination was empirically identified by screening key factors known to be supplied by helper T cells.)
One important biologic role of T-helper cells is to activate or “license” DCs (Schoenberger et al., 1998). We therefore tested the activation status of DCs after vaccination in the presence or absence of Treg cells. Figure 4B uses CD86 as a representative DC marker (similar effects were seen with CD80, CD40 and MHC class II, not shown). In mice with intact Treg cells, vaccination produced widespread activation of DCs in VDLNs as expected. However, when Treg cells were depleted, DC activation became significantly compromised. DC activation was fully restored by supplying the artificial rescue cocktail of CD40 agonist antibody + rIL-2. Thus, one important biologic effect of reprogrammed Treg cells was to help support activation of resting DCs.
Our system focused on initial priming to a new antigen, because this was when Treg cells were the most critical (see Figure S4B). We hypothesized that the priming step might be particularly dependent on help from Treg cells because conventional CD4+ cells were naive to the OVA antigen (naive CD4+ cells are slow to activate, and do not possess the pre-formed pool of CD40L found in memory T cells (Koguchi et al., 2011)). To test this hypothesis, we extensively pre-immunized Foxp3GFP-Cre×ROSA26lox-stop-loxDTR mice with OVA to create a pre-existing pool of OVA-specific memory CD4+ cells. Figure 4C shows that, in pre-immunized mice, help from Treg cells was no longer needed, and the memory (conventional) CD4+ cells could now fully support priming of new OT-I cells following DT depletion.
The preceding studies focused on cross-presentation to CD8+ T cells, which is known to be helper-dependent. However, the requirement for reprogrammed Treg cells in order to activate resting DCs (see Figure 4B, above) suggested that even CD4+ T cells might require support from reprogrammed Treg cells. Figure 4D tests this hypothesis using the Cd40lg−/− model. Priming of naive CD4+ effector cells (OT-IIRag2−/−) was markedly compromised in the absence of host CD40L, and activation of DCs was impaired. DC activation was restored and OT-IIRag2−/− proliferation rescued by adoptive transfer of WT (CD40L-sufficient) Treg cells (lower panels). The dependence of CD4+ OT-IIRag2−/− cells on help from Treg cells was further confirmed using DTR depletion (Figure S4C).
Taken together, the preceding studies showed that reprogrammed Treg cells supplied an important helper activity in the specialized setting of priming to a new antigen. Analogous help could be supplied by conventional memory CD4+ T cells, if available; but in the absence of memory cells, reprogrammed Treg cells supplied important early help to license DCs and support T cell priming.
Eos-labile cells are functionally suppressive Treg cells
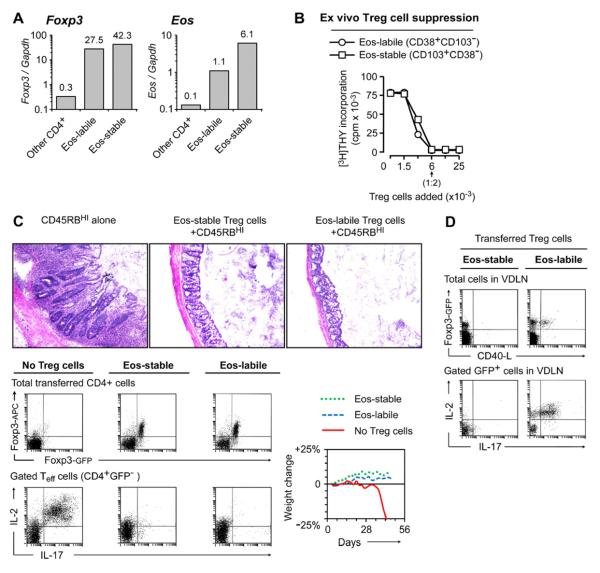
We next asked whether the Eos-labile subset were authentic Treg cells. Figure 5A shows that resting Eos-labile Treg cells constitutively expressed high levels of Foxp3 mRNA, similar to Eos-stable Treg cells and much higher than non-Treg cells (Figure 5A). Eos mRNA was also higher in both of the Treg cell subsets than in non-regulatory CD4+ cells (the Eos-labile Treg cells expressed somewhat less Eos mRNA than the Eos-stable cells, which may be consistent with their more labile Eos expression). Supplemental Figure S5A shows that expression of Foxp3 protein was stable in Eos-labile Treg cells, under both reprogramming and non-reprogramming conditions, and through multiple rounds of cell division.
Figure 5. The Eos-labile cells are functional Treg cells with stable Foxp3 expression.
(A) Eos-labile (CD38+CD103NEG) and Eos-stable (CD38NEGCD103+) Treg cell populations, and all other non-regulatory CD4+ cells (GFPNEG), were sorted from spleens of Foxp3GFP donors following 1 hr CHX treatment. mRNA was measured by quantitative RT-PCR and normalized to Gapdh.
(B) Functional suppression assay. Eos-labile and Eos-stable Treg cells were sorted and added to readout T cells + anti-CD3 mitogen, as described in Supplemental Procedures. Proliferation was measured by thymidine incorporation on day 3.
(C) Rag2−/− mice received CD4+CD45RBHI effector cells, alone or with sorted Foxp3GFP Treg cell subsets (Eos-labile or Eos-stable). When control mice reached 20% weight loss, all groups were analyzed for histology of colonic mucosa and for Treg cell phenotyping in mesenteric LNs.
(D) Rag2−/− mice received adoptive transfer as in panel C, then on day +42 the groups receiving Treg cells were challenged with OT-I + vaccine as shown, and VDLNs analyzed for reprogramming.
Panels A, B representative of at least 3–5 independent experiments; C, D two experiments each. See also Figure S5.
Figure 5B shows that Eos-labile Treg cells, sorted directly ex vivo, mediated functional suppressor activity comparable to Eos-stable Treg cells. The readout in these studies was a conventional in vitro suppression assay using anti-CD3 mitogen, so there were no vaccine-activated DCs or other conditions present to drive reprogramming.
One stringent test of Treg cell function is the ability to protect Rag-deficient hosts from autoimmune colitis caused by CD45RBHI effector cells. Figure 5C shows that sorted Eos-labile Treg cells were fully protective in a Rag2−/− colitis model. (The functional separation of the sorted Eos-labile and Eos-stable subsets is documented in Supplemental Figure S5B.) Despite the lymphopenic environment, Eos-labile Treg cells showed no loss of Foxp3, no down-regulation of Eos, and no reprogramming in the mesenteric LNs (Figure 5C dot-plots). Thus, the Eos-labile cells behaved as authentic Treg cells. However, even while protecting from colitis in the gut, Eos-labile Treg cells elsewhere in the same animal could reprogram when challenged with vaccine and OT-I (Figure 5D, right-hand plots). In contrast, under the same conditions the Eos-stable Treg cells showed no reprogramming in VDLNs (Figure 5D, left plots).
These data show that both Eos-labile and Eos-stable subsets were fully functional, suppressive Treg cells; but that each retained its characteristic propensity to reprogram or not, even after a prolonged period in Rag2−/− hosts.
Epigenetic analysis shows that Eos-labile Treg cells are distinct from Eos-stable Treg cells
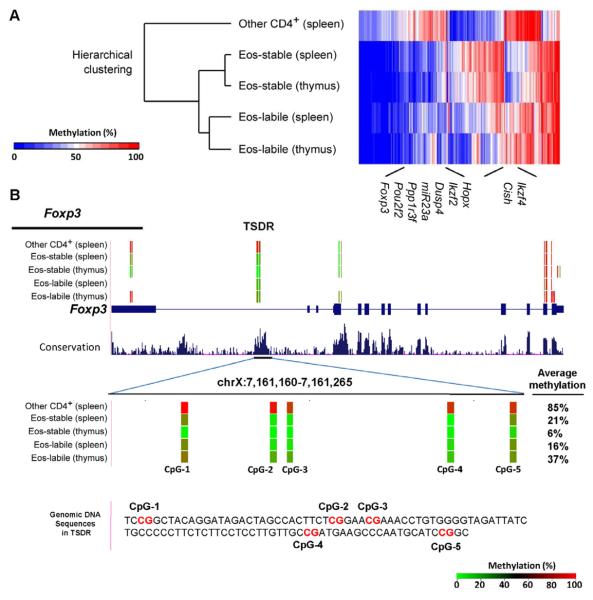
Eos-labile Treg cells appeared durably committed to their reprogrammable phenotype. Such commitment is often reflected in epigenetic changes. Therefore, we examined genome-wide DNA methylation patterns (Meissner et al., 2008) asking whether the Eos-labile Treg cells were identifiably distinct from their Eos-stable counterparts. Treg cell subsets were isolated from thymus (natural Treg cells) and spleen (mixture of peripheral Treg cells); controls were non-regulatory CD4+ cells from spleen. Figure 6A shows hierarchical clustering analysis of those regions with statistically significant differences in methylation between splenic Eos-labile Treg cells and control non-regulatory CD4+ cells. The dendrogram shows that all four Treg cell populations clustered together. The Eos-stable Treg cells from thymus and spleen were similar to each other. The Eos-labile Treg cells were distinct, but were closely related to the Eos-stable Treg cells, as would be expected given their uniform expression of Treg cell markers (Figure 3B, above) and functional suppressor activity (Figure 5). To illustrate this shared relationship, several informative loci are marked on the heat-map in Figure 6A, defined as genes that were similarly methylated in all the Treg cell populations, while all Treg cell populations were different from non-regulatory CD4+ cells. The methylation patterns for several such loci are shown in detail in Supplemental Figure S6.
Figure 6. Eos-labile Treg cells show a distinct pattern of DNA methylation.
Cells from thymus or spleen were incubated for 1 hr with CHX, then GFP+ Treg cells sorted into Eos-labile (CD38+CD103NEG) and Eos-stable (CD38NEGCD103+) populations. Control CD4+GFPNEG (other CD4+) cells were sorted from spleen. Genome-wide differential methylation analysis was performed using reduced representation bisulfite sequencing (RRBS), as described in Supplemental Procedures.
(A) Hierarchical clustering analysis and heat map. Rows represent the sample and columns represents the average methylation level (% of methylation) in each differentially methylated region.
(B) Analysis of DNA methylation for the Foxp3 locus in each population. Average methylation for the Treg-specific demethylation region (TSDR) region (CNS2) is shown as the superimposed percentage. Tracks (from top to bottom): DNA methylation at each CpG site derived from the bisulfite sequencing reads; RefSeq genes; and conservation score (UCSC genome browser). Red and green colors indicate methylated and unmethylated CpG sites, respectively. The average methylation values of CpG sites measured are shown next to the TSDR. See also Figure S6.
Figure 6B shows the lineage-specific TSDR (CNS2) region of the Foxp3 gene (Zheng et al., 2010). Eos-labile Treg cells were somewhat less highly demethylated at the TSDR than the Eos-stable Treg cells, but both showed greater demethylation than non-Treg cells. Functionally, TSDR demethylation is required in order to maintain stable Foxp3 gene expression during cell division (Zheng et al., 2010). Eos-labile Treg cells maintained stable Foxp3 protein levels throughout multiple rounds of cell division (Figure S5A, above), and their Foxp3 expression was stable in lymphopenic hosts (Figure 5C, above). Thus, the TSDR in Eos-labile Treg cells appeared functionally active, and able to maintain long-term Foxp3 expression.
Taken together, these data were consistent with the hypothesis that Eos-labile Treg cells formed a distinct developmental subset, related to, but not identical with, the Eos-stable subset, and that this distinction emerged as early as the thymus.
Down-regulation of Eos is prevented by tumor-induced IDO
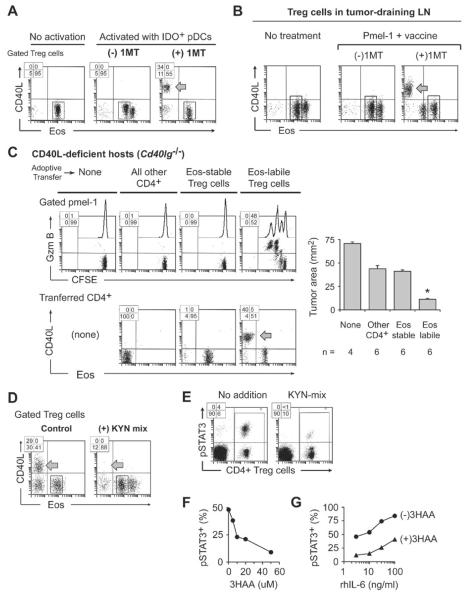
Because down-regulation of Eos was controlled by specific signals, we reasoned that there might be opposing signals that could prevent the loss of Eos. We had previously shown that the immunoregulatory enzyme indoleamine 2,3-dioxygenase (IDO) antagonized functional Treg cell reprogramming (Sharma et al., 2010; Sharma et al., 2009). Therefore, we isolated plasmacytoid DCs (pDCs) from tumor-draining LNs (TDLNs), where many pDCs express high amounts of IDO (Munn et al., 2004), and tested them in reprogramming assays with resting, naive Treg cells. To block activity of the IDO pathway, some co-cultures received the pharmacologic inhibitor 1-methyl-D-tryptophan (1MT). Figure 7A shows that when the IDO pathway was active (no 1MT) the Treg cells were unable to downregulate Eos and could not undergo reprogramming (middle dot-plot). When 1MT was added, the same Treg cells now lost Eos and were able to undergo reprogramming (right-hand dot-plot). The degree of reprogramming supported by TDLN pDCs was variable, with DCs from more advanced tumors being progressively more inhibitory for reprogramming. Consistent with a specific effect of IDO, when pDCs were isolated from TDLNs of Ido1−/− mice, these pDCs no longer blocked Treg cell reprogramming (Figure S7A).
Figure 7. IDO blocks Eos down-regulation.
(A) Plasmacytoid DCs (pDCs) with high IDO expression were obtained by sorting CD11c+ cells from TDLNs of mice with B16F10 tumors, and used in reprogramming co-cultures with resting Foxp3GFP Treg cells (from mice without tumors). Co-cultures received either no siinfekl (“no activation” group) or siinfekl (“activated” groups), with or without the IDO-pathway inhibitor 1-methyl-D-tryptophan (1MT).
(B) Foxp3GFP mice with established B16F10 tumors were treated with naive pmel-1 T cells (CD8+, CFSE-labeled) plus hgp100 peptide vaccine in IFA+CpG, with or without 1MT in drinking water. Representative of 5 experiments.
(C) Cd40lg−/− host mice received pre-adoptive transfer of sorted Eos-labile or Eos-stable Treg cells from Foxp3GFP mice; or all other non-regulatory CD4+ cells; or no transfer. Then all mice were implanted with B16F10 tumors. After 7 days, mice received pmel-1 + vaccine, with or without 1MT in drinking water. Four days after vaccination, TDLNs were analyzed and tumor size was measured at necropsy. Pooled data from 3 identical experiments; * p<.05 by ANOVA, bars show SD.
(D) DCs from VDLNs (prepared as in Figure 1C), which have low IDO expression, were used in reprogramming co-cultures, with or without added KYN-metabolite mixture as described in Supplemental Experimental Procedures.
(E–G) Treg cells were incubated for 18 hrs in reprogramming co-cultures as in panel D, then stained for intracellular phospho-STAT3 (pSTAT3). (E) effect of KYN metabolites. (F) dose-response for a representative KYN metabolite, 3-hydroxyanthranilic acid (3HAA); percentage of gated Treg cells staining positive for pSTAT3 is shown. (G) response of pSTAT3 to exogenous recombinant IL-6, with or without 50 uM 3HAA (gated Treg cells).
Each panel representative of at least 3 experiments, or as noted. See also Figure S7.
IDO suppressed Treg cell reprogramming in vivo (Figure 7B). Mice with established B16F10 tumors received tumor-specific CD8+ pmel-1 cells plus vaccination with cognate hgp100 peptide (Antony et al., 2005). Groups were treated with 1MT in drinking water, or vehicle control. When IDO was active (no 1MT) the Treg cells in TDLNs failed to downregulate Eos after vaccination and there was no reprogramming. However, when mice received 1MT then many Treg cells in TDLNs downregulated Eos and underwent reprogramming (expressed CD40L, as shown; and also IL-2 and L17, not shown).
To test whether Treg cell reprogramming was functionally important for anti-tumor T cell responses, we used helper-deficient Cd40lg−/− mice as tumor-bearing hosts, and asked whether adoptive transfer of WT Eos-labile Treg cells could rescue the activation of pmel-1 effector cells. Pmel-1 cells were informative in this regard because they are known to require CD4+ T cell help for optimal anti-tumor activity (Antony et al., 2005). In these studies, all mice received 1MT treatment so that Treg cells would be able to reprogram if they were capable of it. Figure 7C shows that only the Eos-labile Treg cells could undergo reprogramming, express CD40L and provide help for pmel-1 priming. Control mice, receiving Eos-stable Treg cells or non-regulatory CD4+ T cells, showed significantly impaired pmel-1 activation and anti-tumor activity (bar graph). Further, supplemental Figure S7B shows that helper activity from Eos-labile Treg cells was important to support effective cross-presentation of endogenous tumor antigens to a new population of anti-tumor T cells.
Thus, established tumors actively suppressed Treg cell reprogramming, and a major mechanism of this effect was attributable to the IDO pathway. Treg reprogramming and anti-tumor vaccine responses were impaired in tumor-bearing hosts unless IDO was blocked.
IDO activates a population of highly-suppressive EosHI Treg cells in TDLNs
In the course of the experiments in Figures 7A and 7B, we noted that when Treg cells were exposed to IDO, a sub-population increased their Eos expression by 5–10 fold (the basal, resting level is indicated by the box in each dot-plot). Cell-sorting studies in Figure S7C revealed that these new EosHI Treg cells were derived from the CD103+ (Eos-stable) population. Figures S7D and S7E shows that the EosHI Treg cells corresponded to the highly suppressive, IDO-activated Treg cells that we have previously described in TDLNs (Sharma et al., 2007). Thus, not only did IDO inhibit reprogramming of the Eos-labile subset, it also enhanced the suppressive function of the Eos-stable subset.
IDO blocks Eos down-regulation via KYN metabolites
One mechanism by which IDO can affect Treg cells is production of kynurenine-pathway (KYN) metabolites, which can signal through the aryl hydrocarbon receptor (AhR) (Mezrich et al., 2010). Figure 7D shows that the addition of KYN metabolites to co-cultures prevented Eos down-regulation and blocked reprogramming, even when the DCs were from VDLNs and did not express IDO. Pharmacologic-inhibitor studies indicated that the effect of KYN metabolites was mediated via the AhR (Figure S7F). STAT3 is a key component of signal transduction via the IL-6 receptor, and has been implicated as a driver of destabilization of the Treg cell phenotype during inflammation (Laurence et al., 2012). Figure 7E shows that Treg cells exposed to reprogramming conditions showed phosphorylation of STAT3, and this phosphorylation was strongly inhibited by the addition of KYN metabolites. Figure 7F shows a titration of a representative KYN metabolite, 3-hydroxyanthranilic acid (3HAA), demonstrating dose-dependent inhibition of STAT3 phosphorylation. Figure 7G shows that adding excess recombinant IL-6 to reprogramming assays progressively increased STAT3 phosphorylation, and this was antagonized by the presence of 3HAA (a shift in the ED50 for IL-6 of 1-2 orders of magnitude). Taken together, these studies supported the hypothesis that IDO blocked the down-regulation of Eos via KYN metabolites and the AhR, and that this effect was mediated at least in part by antagonism of IL-6-induced STAT3 activation.
DISCUSSION
In the current study we identify a committed subset of reprogrammable Treg cells characterized by rapid loss of the co-repressor Eos in response to specific inflammatory signals. Loss of Eos allowed a regulated transformation of these Treg cells into biologically important helper cells, without loss of Foxp3. Developmental commitment to the Eos-labile subset was evident as early as the thymus, and could be distinguished by characteristic cell-surface markers and DNA methylation. Using ablation, adoptive-transfer and gene depletion approaches we show that absence of the Eos-labile Treg cell subset selectively compromised the ability of naive mice to activate resting DCs and prime initial T cell responses to new antigen. Thus, the Eos-labile Treg cell subset supplied an important, natural helper activity in the immune system.
The Eos-labile cells were authentic Treg cells. Many of them arose in the thymus; they uniformly expressed Foxp3, CTLA-4 and CD25; and they were functionally suppressive in vitro and fully protective in the stringent Rag2−/− colitis model. In other systems, it has been shown that some conventional (non-regulatory) CD4+ cells may transiently express Foxp3 following activation, then later lose this aberrant Foxp3 (Miyao et al., 2012). However, Eos-labile Treg cells were not these cells, as shown by the fact that: (i) they were present even in thymus (natural Treg cells) rather than induced by activation; (ii) they constitutively expressed Foxp3 even at rest; and (iii) Foxp3 expression was stable, and was not lost even after reprogramming.
Because Foxp3 was never lost, our model differed from some other models in which Treg cells lose expression of Foxp3 (Komatsu et al., 2009; Laurence et al., 2012; Oldenhove et al., 2009; Zhou et al., 2009). These models often feature prolonged conditions of pathologic inflammation such as lymphopenia, sepsis, autoimmunity or GVHD. In contrast, our studies dealt with rapid events (hours to days) in the context of mild, natural immune activation such as vaccination. Under these conditions Foxp3 was not lost, and the transformation to a pro-inflammatory phenotype was instead controlled by Eos.
One key finding was that reprogrammed Treg cells provided help very rapidly. The Foxp3+ lineage is unique in that many Treg cells appear constitutively activated, and respond more rapidly than other cells to inflammation (O'Gorman et al., 2009). We speculate that this property of the Foxp3+ lineage allows Eos-labile Treg cells to act as “first responders”, delivering immediate help at a time when conventional, non-regulatory CD4+ cells have not yet had time to activate. If host mice were pre-immunized, and thus had conventional memory CD4+ T cells available, then help from reprogrammed Treg cells was no longer necessary; but in naive mice encountering a new antigen, help from reprogrammed Treg cells was critical. The antigen specificity of Eos-labile Treg cells is not yet known, but they were present in the thymus, and many thymic-derived Treg cells recognize self peptides. Thus, the unique attributes of Foxp3-lineage cells – hair-trigger responses and constant antigen exposure – may ideally position Eos-labile Treg cells to deliver rapid help.
However, this raises the paradox of why Treg cells that are able to reprogram do not lead to autoimmunity. We propose that autoimmunity is avoided by the strictly local, highly regulated nature of the reprogramming event. Thus, during vaccination, reprogramming was confined solely to the VDLNs. Likewise, in the Rag2−/− colitis model, some Eos-labile Treg cells could be suppressive in the gut even while others were reprogramming in VDLNs. Thus, Treg cells were allowed to become helper cells only in specific contexts where permissive pro-inflammatory signals already existed.
Similarly, the DC activating and licensing effect of reprogrammed Treg cells might appear paradoxical, because Treg cells normally suppress DCs and keep them in a quiescent state (Muth et al., 2012). However, this effect is exerted specifically on resting DCs; once DCs are activated via CD40 ligation, they become resistant to Treg-mediated suppression (Serra et al., 2003). Thus, we hypothesize that there exists a balance: systemic inhibition of DCs by suppressive Treg cells in the steady state; but local activation of DCs by reprogrammed Treg cells in an acute inflammatory milieu.
Finally, an additional important layer of regulation was provided by IDO, which blocked the loss of Eos and prevented Treg cell reprogramming. IDO is involved in immune suppression and tolerance, so it is logical that it would stabilize the suppressive Treg cell phenotype. We note, however, that in the pathological setting of tumors this effect became exaggerated, and it was necessary to pharmacologically inhibit IDO in order for the Eos-labile Treg cells to successfully reprogram and contribute their beneficial helper function.
In conclusion, we show that behavior of the co-repressor Eos defines two distinct, committed subsets of Foxp3+ Treg cells. In certain settings, the natural “rapid-response” role of Foxp3-lineage cells extends beyond their classical role of suppression, and also includes important helper activity mediated by the Eos-labile subset.
Experimental Procedures
Reagents and mice
Details of reagents and mouse strains are given in Supplemental Experimental Procedures. Animal studies were approved by the Institutional Animal Care and Use Committee of Georgia Health Sciences University.
Tumor, 1MT administration and influenza infection
Tumor studies were performed as previously described (Sharma et al., 2010). Tumor area was measured at necropsy on day 11 following dissection. Oral 1MT was administered at 2 mg/ml in drinking water as described (Sharma et al., 2009). Details of intranasal infection with HKx31 (X31, H3N2) influenza virus are in Supplemental Experimental Procedures.
Adoptive transfer and vaccination
OT-I or pmel-1 spleen cells were enriched using CD8 magnetic beads (Miltenyi Biotech) and 2×106 cells injected i.v. For Treg cell sorting, thymus or spleen from Foxp3GFP mice (Fontenot et al., 2005) were pre-incubated in vitro for 1 hr with CHX (20 ug/ml) then stained with CD103 vs CD38, and sorted by MoFlo cell-sorter with doublet discrimination. Treg cells (1×105 cells unless otherwise indicated) or non-regulatory CD4+ cells (CD4+GFP−, 1×106 cells) were injected i.v. Vaccines comprised whole OVA protein or hgp100 peptide plus CpG-1826 in IFA (see Supplemental Procedures) administered in the footpad, and popliteal LNs harvested after 4 days. For DTR experiments, mice received DT 1 ug/dose i.p.
FACS staining
Details of all staining protocols are in Supplemental Experimental Procedures, online.
Eos transduction
Mouse Eos was cloned into the pMIG plasmid to form a bicistronic MSCV-Eos-ires-GFP vector, and expressed in 293T cells using pCL-Eco (Imgenex) packaging plasmid. Control (GFP-only) virus used empty pMIG vector (Addgene). These viruses were used to transduce Treg cells in co-cultures, as described in Supplemental Experimental Procedures.
Methylation analysis
Methylation analysis of genomic DNA by reduced representation bisulfite sequencing (RRBS) is described in detail in Supplemental Procedures. The raw and analyzed sequencing data have been submitted to NCBI Gene Expression Omnibus (GEO) database, accession number GSE44380.
Statistical analysis
Multiple treatment groups were compared by ANOVA with Tukey's HSD test.
Supplementary Material
ACKNOWLEDGEMENTS
The authors thank Anita Sharma, Judy Gregory, Joyce Wilson and Jeanene Pihkala for expert technical assistance. Supported by NIH grants CA103320, CA096651, CA112431 (to DHM); HD41187, AI083005, AI063402 (to ALM); and CA72669, HL56067, AI34495 (to BRB); and an Immunotherapy Discovery Institute seed grant (to HS). HS is a Georgia Cancer Coalition Distinguished Cancer Scientist.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Addey C, White M, Dou L, Coe D, Dyson J, Chai JG. Functional plasticity of antigen-specific regulatory T cells in context of tumor. J Immunol. 2011;186:4557–4564. doi: 10.4049/jimmunol.1003797. [DOI] [PubMed] [Google Scholar]
- Antony PA, Piccirillo CA, Akpinarli A, Finkelstein SE, Speiss PJ, Surman DR, Palmer DC, Chan CC, Klebanoff CA, Overwijk WW, et al. CD8+ T cell immunity against a tumor/self-antigen is augmented by CD4+ T helper cells and hindered by naturally occurring T regulatory cells. J. Immunol. 2005;174:2591–2601. doi: 10.4049/jimmunol.174.5.2591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva Martins M, Piccirillo CA. Functional stability of Foxp3+ regulatory T cells. Trends Mol. Med. 2012;18:454–462. doi: 10.1016/j.molmed.2012.06.001. [DOI] [PubMed] [Google Scholar]
- Fisson S, Darrasse-Jeze G, Litvinova E, Septier F, Klatzmann D, Liblau R, Salomon BL. Continuous activation of autoreactive CD4+ CD25+ regulatory T cells in the steady state. J. Exp. Med. 2003;198:737–746. doi: 10.1084/jem.20030686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fontenot JD, Rasmussen JP, Williams LM, Dooley JL, Farr AG, Rudensky AY. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity. 2005;22:329–341. doi: 10.1016/j.immuni.2005.01.016. [DOI] [PubMed] [Google Scholar]
- Grewal IS, Foellmer HG, Grewal KD, Xu J, Hardardottir F, Baron JL, Janeway CA, Flavell RA. Requirement for CD40 ligand in costimulation induction, T cell activation, and experimental allergic encephalomyelitis. Science. 1996;273:1864–1867. doi: 10.1126/science.273.5283.1864. [DOI] [PubMed] [Google Scholar]
- Hershko DD, Robb BW, Wray CJ, Luo GJ, Hasselgren PO. Superinduction of IL-6 by cycloheximide is associated with mRNA stabilization and sustained activation of p38 map kinase and NF-kappaB in cultured caco-2 cells. J. Cell. Biochem. 2004;91:951–961. doi: 10.1002/jcb.20014. [DOI] [PubMed] [Google Scholar]
- Kim JM, Rasmussen JP, Rudensky AY. Regulatory T cells prevent catastrophic autoimmunity throughout the lifespan of mice. Nat Immunol. 2007;8:191–197. doi: 10.1038/ni1428. [DOI] [PubMed] [Google Scholar]
- Koguchi Y, Gardell JL, Thauland TJ, Parker DC. Cyclosporine-resistant, Rab27a-independent mobilization of intracellular preformed CD40 ligand mediates antigen-specific T cell help in vitro. J Immunol. 2011;187:626–634. doi: 10.4049/jimmunol.1004083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komatsu N, Mariotti-Ferrandiz ME, Wang Y, Malissen B, Waldmann H, Hori S. Heterogeneity of natural Foxp3+ T cells: a committed regulatory T-cell lineage and an uncommitted minor population retaining plasticity. Proc. Natl. Acad. Sci. USA. 2009;106:1903–1908. doi: 10.1073/pnas.0811556106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopf M, Baumann H, Freer G, Freudenberg M, Lamers M, Kishimoto T, Zinkernagel R, Bluethmann H, Kohler G. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature. 1994;368:339–342. doi: 10.1038/368339a0. [DOI] [PubMed] [Google Scholar]
- Laurence A, Amarnath S, Mariotti J, Kim YC, Foley J, Eckhaus M, O'Shea JJ, Fowler DH. STAT3 Transcription Factor Promotes Instability of nTreg Cells and Limits Generation of iTreg Cells during Acute Murine Graft-versus-Host Disease. Immunity. 2012;37:209–222. doi: 10.1016/j.immuni.2012.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meissner A, Mikkelsen TS, Gu H, Wernig M, Hanna J, Sivachenko A, Zhang X, Bernstein BE, Nusbaum C, Jaffe DB, et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature. 2008;454:766–770. doi: 10.1038/nature07107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mezrich JD, Fechner JH, Zhang X, Johnson BP, Burlingham WJ, Bradfield CA. An interaction between kynurenine and the aryl hydrocarbon receptor can generate regulatory T cells. J Immunol. 2010;185:3190–3198. doi: 10.4049/jimmunol.0903670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyao T, Floess S, Setoguchi R, Luche H, Fehling HJ, Waldmann H, Huehn J, Hori S. Plasticity of Foxp3(+) T cells reflects promiscuous Foxp3 expression in conventional T cells but not reprogramming of regulatory T cells. Immunity. 2012;36:262–275. doi: 10.1016/j.immuni.2011.12.012. [DOI] [PubMed] [Google Scholar]
- Munn DH, Sharma MD, Hou D, Baban B, Lee JR, Antonia SJ, Messina JL, Chandler P, Koni PA, Mellor A. Expression of indoleamine 2,3-dioxygenase by plasmacytoid dendritic cells in tumor-draining lymph nodes. J. Clin. Invest. 2004;114:280–290. doi: 10.1172/JCI21583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muth S, Schutze K, Schild H, Probst HC. Release of dendritic cells from cognate CD4+ T-cell recognition results in impaired peripheral tolerance and fatal cytotoxic T-cell mediated autoimmunity. Proc. Natl. Acad. Sci. USA. 2012;109:9059–9064. doi: 10.1073/pnas.1110620109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Gorman WE, Dooms H, Thorne SH, Kuswanto WF, Simonds EF, Krutzik PO, Nolan GP, Abbas AK. The initial phase of an immune response functions to activate regulatory T cells. J Immunol. 2009;183:332–339. doi: 10.4049/jimmunol.0900691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldenhove G, Bouladoux N, Wohlfert EA, Hall JA, Chou D, Dos Santos L, O'Brien S, Blank R, Lamb E, Natarajan S, et al. Decrease of Foxp3+ Treg cell number and acquisition of effector cell phenotype during lethal infection. Immunity. 2009;31:772–786. doi: 10.1016/j.immuni.2009.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan F, Yu H, Dang EV, Barbi J, Pan X, Grosso JF, Jinasena D, Sharma SM, McCadden EM, Getnet D, et al. Eos mediates Foxp3-dependent gene silencing in CD4+ regulatory T cells. Science. 2009;325:1142–1146. doi: 10.1126/science.1176077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quintana FJ, Jin H, Burns EJ, Nadeau M, Yeste A, Kumar D, Rangachari M, Zhu C, Xiao S, Seavitt J, et al. Aiolos promotes T(H)17 differentiation by directly silencing Il2 expression. Nat. Immunol. 2012 doi: 10.1038/ni.2363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubtsov YP, Niec RE, Josefowicz S, Li L, Darce J, Mathis D, Benoist C, Rudensky AY. Stability of the regulatory T cell lineage in vivo. Science. 2010;329:1667–1671. doi: 10.1126/science.1191996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenberger SP, Toes RE, van der Voort EI, Offringa R, Melief CJ. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature. 1998;393:480–483. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- Serra P, Amrani A, Yamanouchi J, Han B, Thiessen S, Utsugi T, J. Santamaria P. CD40 Ligation Releases Immature Dendritic Cells from the Control of Regulatory CD4(+)CD25(+) T Cells. Immunity. 2003;19:877–889. doi: 10.1016/s1074-7613(03)00327-3. [DOI] [PubMed] [Google Scholar]
- Sharma MD, Baban B, Chandler P, Hou DY, Singh N, Yagita H, Azuma M, Blazar BR, Mellor AL, Munn DH. Plasmacytoid dendritic cells from mouse tumor-draining lymph nodes directly activate mature Tregs via indoleamine 2,3-dioxygenase. J. Clin. Invest. 2007;117:2570–2582. doi: 10.1172/JCI31911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma MD, Hou DY, Baban B, Koni PA, He Y, Chandler PR, Blazar BR, Mellor AL, Munn DH. Reprogrammed Foxp3(+) Regulatory T Cells Provide Essential Help to Support Cross-presentation and CD8(+) T Cell Priming in Naive Mice. Immunity. 2010;33:942–954. doi: 10.1016/j.immuni.2010.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma MD, Hou DY, Liu Y, Koni PA, Metz R, Chandler P, Mellor AL, He Y, Munn DH. Indoleamine 2,3-dioxygenase controls conversion of Foxp3+ Tregs to TH17-like cells in tumor-draining lymph nodes. Blood. 2009;113:6102–6111. doi: 10.1182/blood-2008-12-195354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornton AM, Korty PE, Tran DQ, Wohlfert EA, Murray PE, Belkaid Y, Shevach EM. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol. 2010;184:3433–3441. doi: 10.4049/jimmunol.0904028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuji M, Komatsu N, Kawamoto S, Suzuki K, Kanagawa O, Honjo T, Hori S, Fagarasan S. Preferential generation of follicular B helper T cells from Foxp3+ T cells in gut Peyer's patches. Science. 2009;323:1488–1492. doi: 10.1126/science.1169152. [DOI] [PubMed] [Google Scholar]
- Vendetti S, Davidson TS, Veglia F, Riccomi A, Negri DR, Lindstedt R, Pasquali P, Shevach EM, De Magistris MT. Polyclonal Treg cells enhance the activity of a mucosal adjuvant. Immunol. Cell Biol. 2010;88:698–706. doi: 10.1038/icb.2010.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vokaer B, Van Rompaey N, Lemaitre PH, Lhomme F, Kubjak C, Benghiat FS, Iwakura Y, Petein M, Field KA, Goldman M, et al. Critical role of regulatory T cells in Th17-mediated minor antigen-disparate rejection. J Immunol. 2010;185:3417–3425. doi: 10.4049/jimmunol.0903961. [DOI] [PubMed] [Google Scholar]
- Wang Y, Souabni A, Flavell RA, Wan YY. An intrinsic mechanism predisposes Foxp3-expressing regulatory T cells to Th2 conversion in vivo. J Immunol. 2010;185:5983–5992. doi: 10.4049/jimmunol.1001255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang XO, Nurieva R, Martinez GJ, Kang HS, Chung Y, Pappu BP, Shah B, Chang SH, Schluns KS, Watowich SS, et al. Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity. 2008;29:44–56. doi: 10.1016/j.immuni.2008.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yarilin AA, Belyakov IM. Cytokines in the thymus: production and biological effects. Curr. Med. Chem. 2004;11:447–464. doi: 10.2174/0929867043455972. [DOI] [PubMed] [Google Scholar]
- Yurchenko E, Shio MT, Huang TC, Da Silva Martins M, Szyf M, Levings MK, Olivier M, Piccirillo CA. Inflammation-driven reprogramming of CD4+ Foxp3+ regulatory T cells into pathogenic Th1/Th17 T effectors is abrogated by mTOR inhibition in vivo. PLoS ONE. 2012;7:e35572. doi: 10.1371/journal.pone.0035572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y, Josefowicz S, Chaudhry A, Peng XP, Forbush K, Rudensky AY. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature. 2010;463:808–812. doi: 10.1038/nature08750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X, Bailey-Bucktrout SL, Jeker LT, Penaranda C, Martinez-Llordella M, Ashby M, Nakayama M, Rosenthal W, Bluestone JA. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat. Immunol. 2009;10:1000–1007. doi: 10.1038/ni.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.