Abstract
Stage 21 to 22 chicken embryo limb bud cells were infected with a temperature-sensitive mutant of Rous sarcoma virus and were grown in culture. Although control, uninfected cells yielded definitive chondroblasts (by day 4) which initiated the synthesis of the cartilage-characteristic proteoglycan, the transformed cells grown at the permissive temperature failed to do so. These effects were fully reversible after a shift to the nonpermissive temperature. In addition, infected cells at the nonpermissive temperature expressed traits of terminal chondrogenic maturation 2 to 3 days earlier than parallel, uninfected cells. Thus, Rous sarcoma virus-induced transformation reversibly blocks terminal limb bud cell chondrogenesis in culture, at the nonpermissive temperature, viral infection may also induce intracellular or extracellular conditions which favor or accelerate the process of chondrogenic cell maturation.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams S. L., Boettiger D., Focht R. J., Holtzer H., Pacifici M. Regulation of the synthesis of extracellular matrix components in chondroblasts transformed by a temperature-sensitive mutant of Rous sarcoma virus. Cell. 1982 Sep;30(2):373–384. doi: 10.1016/0092-8674(82)90235-5. [DOI] [PubMed] [Google Scholar]
- Beug H., Palmieri S., Freudenstein C., Zentgraf H., Graf T. Hormone-dependent terminal differentiation in vitro of chicken erythroleukemia cells transformed by ts mutants of avian erythroblastosis virus. Cell. 1982 Apr;28(4):907–919. doi: 10.1016/0092-8674(82)90070-8. [DOI] [PubMed] [Google Scholar]
- Boettiger D., Durban E. M. Progenitor-cell populations can be infected by RNA tumor viruses, but transformation is dependent on the expression of specific differentiated functions. Cold Spring Harb Symp Quant Biol. 1980;44(Pt 2):1249–1254. doi: 10.1101/sqb.1980.044.01.135. [DOI] [PubMed] [Google Scholar]
- Boettiger D., Roby K., Brumbaugh J., Biehl J., Holtzer H. Transformation of chicken embryo retinal melanoblasts by a temperature-sensitive mutant of Rous sarcoma virus. Cell. 1977 Aug;11(4):881–890. doi: 10.1016/0092-8674(77)90299-9. [DOI] [PubMed] [Google Scholar]
- Caplan A. I. Effects of the nicotinamide-sensitive teratogen3-acetylpyridine on chick limb cells in culture. Exp Cell Res. 1970 Oct;62(2):341–355. doi: 10.1016/0014-4827(70)90564-1. [DOI] [PubMed] [Google Scholar]
- Chacko S., Abbott J., Holtzer S., Holtzer H. The loss of phenotypic traits by differentiated cells. VI. Behavior of the progeny of a single chondrocyte. J Exp Med. 1969 Aug 1;130(2):417–442. doi: 10.1084/jem.130.2.417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chevallier A., Kieny M., Mauger A. Limb-somite relationship: origin of the limb musculature. J Embryol Exp Morphol. 1977 Oct;41:245–258. [PubMed] [Google Scholar]
- Christ B., Jacob H. J., Jacob M. Experimental analysis of the origin of the wing musculature in avian embryos. Anat Embryol (Berl) 1977 Mar 30;150(2):171–186. doi: 10.1007/BF00316649. [DOI] [PubMed] [Google Scholar]
- De Luca S., Heinegård D., Hascall V. C., Kimura J. H., Caplan A. I. Chemical and physical changes in proteoglycans during development of chick limb bud chondrocytes grown in vitro. J Biol Chem. 1977 Oct 10;252(19):6600–6608. [PubMed] [Google Scholar]
- Dienstman S. R., Biehl J., Holtzer S., Holtzer H. Myogenic and chondrogenic lineages in developing limb buds grown in vitro. Dev Biol. 1974 Jul;39(1):83–95. doi: 10.1016/s0012-1606(74)80010-2. [DOI] [PubMed] [Google Scholar]
- Fiszman M. Y., Fuchs P. Temperature-sensitive expression of differentiation in transformed myoblasts. Nature. 1975 Apr 3;254(5499):429–431. doi: 10.1038/254429a0. [DOI] [PubMed] [Google Scholar]
- Gazzolo L., Samarut J., Bouabdelli M., Blanchet J. P. Early precursors in the erythroid lineage are the specific target cells of avian erythroblastosis virus in vitro. Cell. 1980 Dec;22(3):683–691. doi: 10.1016/0092-8674(80)90544-9. [DOI] [PubMed] [Google Scholar]
- Goetinck P. F., Pennypacker J. P., Royal P. D. Proteochondroitin sulfate synthesis and chondrogenic expression. Exp Cell Res. 1974 Aug;87(2):241–248. doi: 10.1016/0014-4827(74)90476-5. [DOI] [PubMed] [Google Scholar]
- Graf T., Ade N., Beug H. Temperature-sensitive mutant of avian erythroblastosis virus suggests a block of differentiation as mechanism of leukaemogenesis. Nature. 1978 Oct 12;275(5680):496–501. doi: 10.1038/275496a0. [DOI] [PubMed] [Google Scholar]
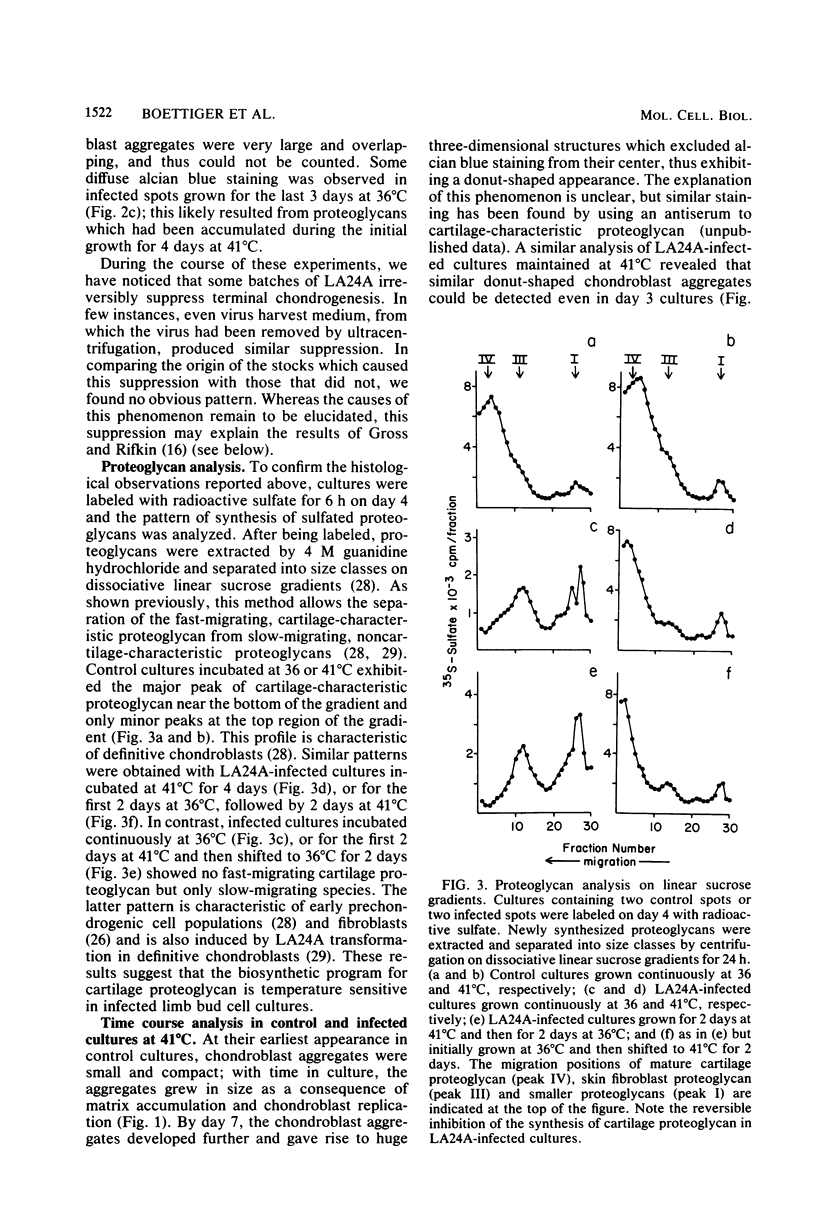
- Gross J. L., Rifkin D. B. The effect of avian retroviruses on limb bud chondrogenesis in vitro. Cell. 1979 Nov;18(3):707–718. doi: 10.1016/0092-8674(79)90125-9. [DOI] [PubMed] [Google Scholar]
- Hanafusa H. Rapid transformation of cells by Rous sarcoma virus. Proc Natl Acad Sci U S A. 1969 Jun;63(2):318–325. doi: 10.1073/pnas.63.2.318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holtzer H., Biehl J., Yeoh G., Meganathan R., Kaji A. Effect of oncogenic virus on muscle differentiation. Proc Natl Acad Sci U S A. 1975 Oct;72(10):4051–4055. doi: 10.1073/pnas.72.10.4051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz A. L., Dorfman A. The growth of cartilage cells in soft agar and liquid suspension. J Cell Biol. 1970 May;45(2):434–438. doi: 10.1083/jcb.45.2.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEV R., SPICER S. S. SPECIFIC STAINING OF SULPHATE GROUPS WITH ALCIAN BLUE AT LOW PH. J Histochem Cytochem. 1964 Apr;12:309–309. doi: 10.1177/12.4.309. [DOI] [PubMed] [Google Scholar]
- Linsenmayer T. F., Toole B. P., Trelstad R. L. Temporal and spatial transitions in collagen types during embryonic chick limb development. Dev Biol. 1973 Dec;35(2):232–239. doi: 10.1016/0012-1606(73)90020-1. [DOI] [PubMed] [Google Scholar]
- Lowe M. E., Pacifici M., Holtzer H. Effects of phorbol-12-myristate-13-acetate on the phenotypic program of cultured chondroblasts and fibroblasts. Cancer Res. 1978 Aug;38(8):2350–2356. [PubMed] [Google Scholar]
- Moss P. S., Honeycutt N., Pawson T., Martin G. S. Viral transformation of chick myogenic cells. The relationship between differentiation and the expression of the SRC gene. Exp Cell Res. 1979 Oct 1;123(1):95–105. doi: 10.1016/0014-4827(79)90425-7. [DOI] [PubMed] [Google Scholar]
- Okayama M., Pacifici M., Holtzer H. Differences among sulfated proteoglycans synthesized in nonchondrogenic cells, presumptive chondroblasts, and chondroblasts. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3224–3228. doi: 10.1073/pnas.73.9.3224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pacifici M., Boettiger D., Roby K., Holtzer H. Transformation of chondroblasts by Rous sarcoma virus and synthesis of the sulfated proteoglycan matrix. Cell. 1977 Aug;11(4):891–899. doi: 10.1016/0092-8674(77)90300-2. [DOI] [PubMed] [Google Scholar]
- Pacifici M., Cossu G., Molinaro M., Tato F. Vitamin A inhibits chondrogenesis but not myogenesis. Exp Cell Res. 1980 Oct;129(2):469–474. doi: 10.1016/0014-4827(80)90517-0. [DOI] [PubMed] [Google Scholar]
- Pacifici M., Fellini S. A., Holtzer H., De Luca S. Changes in the sulfated proteoglycans synthesized by "aging" chondrocytes. I. Dispersed cultured chondrocytes and in vivo cartilages. J Biol Chem. 1981 Jan 25;256(2):1029–1037. [PubMed] [Google Scholar]
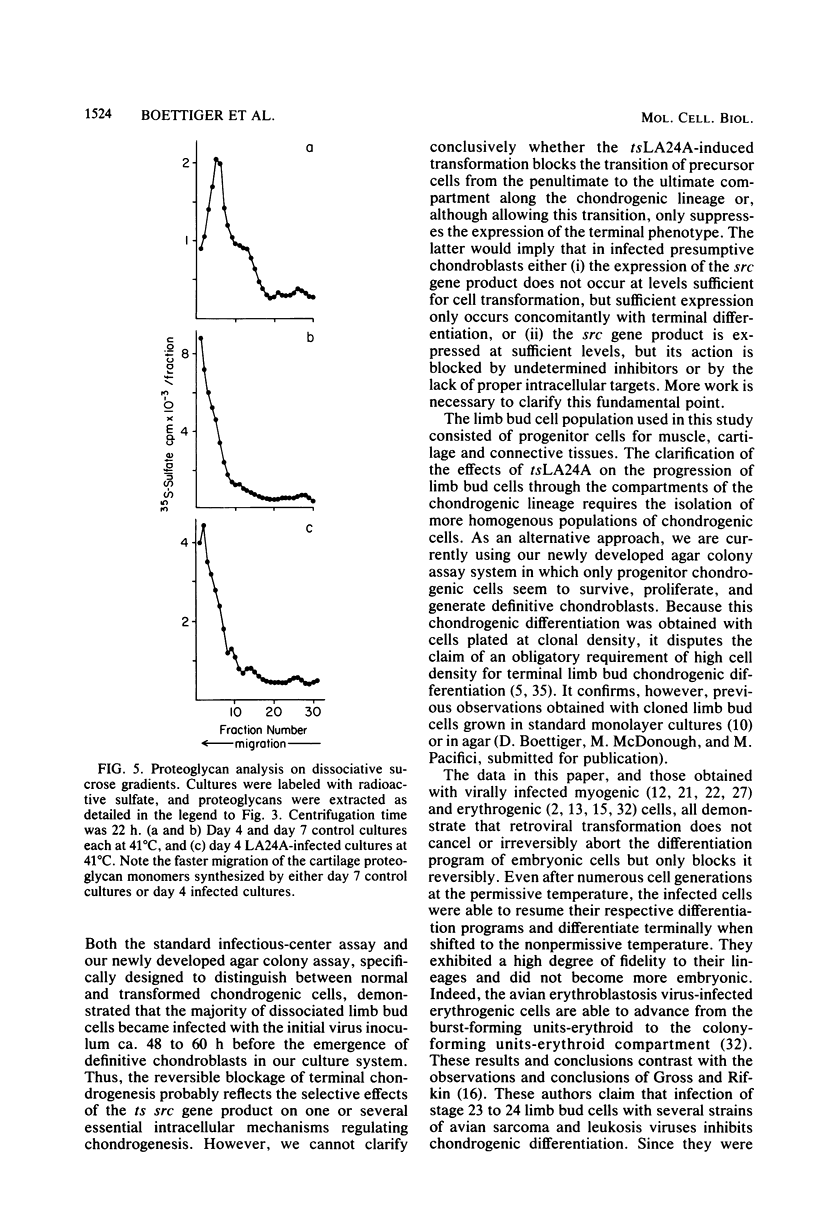
- Samarut J., Gazzolo L. Target cells infected by avian erythroblastosis virus differentiate and become transformed. Cell. 1982 Apr;28(4):921–929. doi: 10.1016/0092-8674(82)90071-x. [DOI] [PubMed] [Google Scholar]
- Sefton B. M., Hunter T., Beemon K. Temperature-sensitive transformation by Rous sarcoma virus and temperature-sensitive protein kinase activity. J Virol. 1980 Jan;33(1):220–229. doi: 10.1128/jvi.33.1.220-229.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanley D. J., Cossu G., Boettiger D., Holtzer H., Pacifici M. Transformation by Rous sarcoma virus induces similar patterns of glycosaminoglycan synthesis in chick embryo skin fibroblasts and vertebral chondroblasts. J Biol Chem. 1983 Jan 25;258(2):810–816. [PubMed] [Google Scholar]
- Umansky R. The effect of cell population density on the developmental fate of reaggregating mouse limb bud mesenchyme. Dev Biol. 1966 Feb;13(1):31–56. doi: 10.1016/0012-1606(66)90048-0. [DOI] [PubMed] [Google Scholar]
- Vasan N. S., Lash J. W. Monomeric and aggregate proteoglycans in the chondrogenic differentiation of embryonic chick limb buds. J Embryol Exp Morphol. 1979 Jan;49:47–59. [PubMed] [Google Scholar]