Abstract
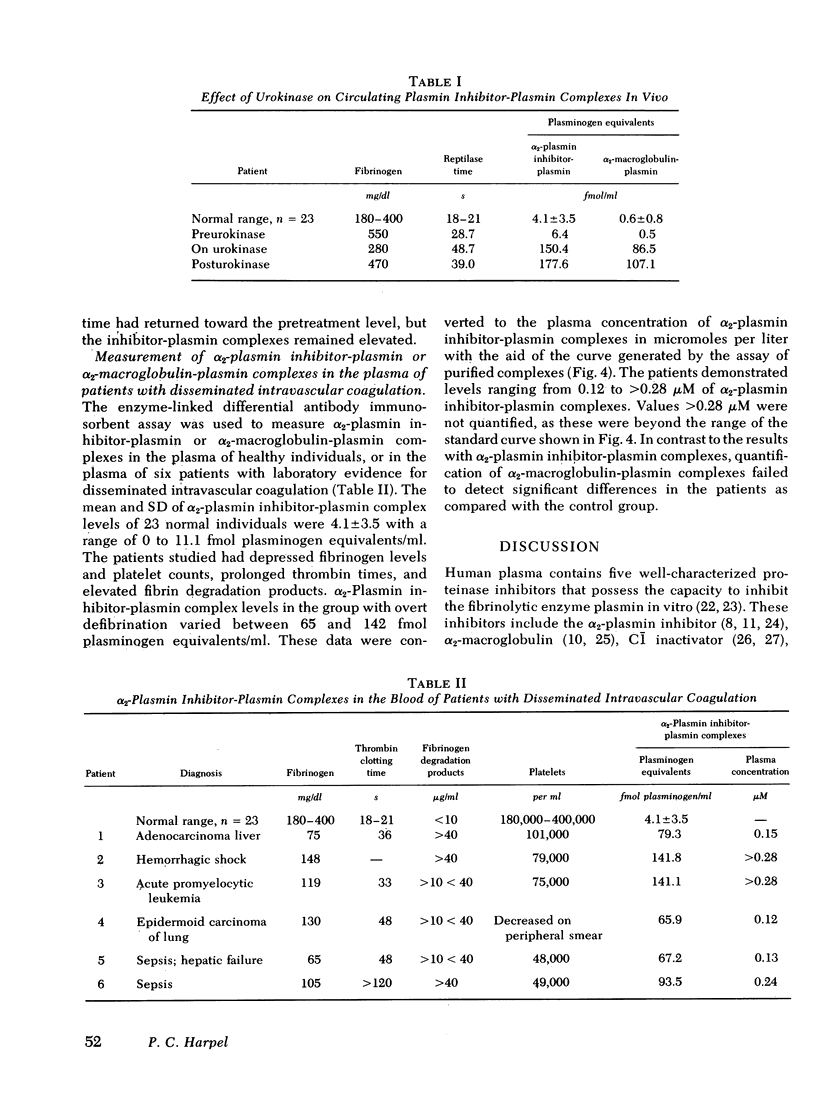
An enzyme-linked differential antibody immunosorbent assay has been developed for the quantification of alpha2-plasmin inhibitor-plasmin and alpha2-macroglobulin-plasmin complexes. In this method the inhibitor-plasmin complex is bound to a surface by an inhibitor-specific antibody, and the plasmin bound to the inhibitor is quantified by a second antibody, rabbit antiplasminogen F(ab')2, labeled with alkaline phosphatase. The hydrolysis of p-nitrophenyl phosphate by the alkaline phosphatase is expressed in femtomoles of plasminogen per milliliter, by reference to a standard plasminogen curve. Inhibitor-enzyme complexes were generated in plasma by the addition of plasmin or of urokinase. The concentration of plasmin added was well below the plasma concentration of alpha2-plasmin inhibitor (1 microM) or of alpha2-macroglobulin (3.5 microM), so that neither inhibitor would be fully saturated with enzyme. Under these conditions increasing amounts of plasmin generated an increase in both alpha2-plasmin inhibitor-plasmin and alpha2-macroglobulin-plasmin complexes. Varying amounts of plasmin were incubated with each of the purified inhibitors in the concentration found in plasma, and the complexes. Varying amounts of plasmin were incubated with each of the purified inhibitors in the concentration found in plasma, and the complexes that formed were quantified by immunoassay. These studies made it possible to quantify the distribution of plasmin between the two inhibitors in plasmin or urokinase-treated plasma. In plasmin-treated plasma, 10% or less of the plasmin bound to both inhibitors was in complex with alpha2-macroglobulin. In contrast, between 19 and 51% of the plasmin generated in urokinase-activated plasma was bound to alpha2-macroglobulin. Thus, major changes in the distribution of plasma were observed, according to whether plasmin was added to plasma or whether plasminogen was activated endogenously. The pattern of inhibitor plasmin complexes generated in vivo by the therapeutic infusion of urokinase was similar to that found for urokinase-activated plasma. 23 normal individuals had low levels of alpha2-plasmin inhibitor-plasmin complexes, whereas six patients with laboratory evidence for disseminated intravascular coagulation demonstrated a 16- to 35-fold increase in he concentration of these complexes. These data indicated that a useful new probe for the study of the fibrinolytic enzyme system had been developed.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aoki N., Moroi M., Matsuda M., Tachiya K. The behavior of alpha2-plasmin inhibitor in fibrinolytic states. J Clin Invest. 1977 Aug;60(2):361–369. doi: 10.1172/JCI108784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aoki N., Moroi M., Tachiya K. Effects of alpha2-plasmin inhibitor on fibrin clot lysis. Its comparison with alpha2-macroglobulin. Thromb Haemost. 1978 Feb 28;39(1):22–31. [PubMed] [Google Scholar]
- Aoki N., Saito H., Kamiya T., Koie K., Sakata Y., Kobakura M. Congenital deficiency of alpha 2-plasmin inhibitor associated with severe hemorrhagic tendency. J Clin Invest. 1979 May;63(5):877–884. doi: 10.1172/JCI109387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aoki N., Sakata Y., Matsuda M., Tateno K. Fibrinolytic states in a patient with congenital deficiency of alpha 2-plasmin inhibitor. Blood. 1980 Mar;55(3):483–488. [PubMed] [Google Scholar]
- Blatrix C., Amouch P., Drouet J., Steinbuch M. Study on the plasmatic elimination of the alpha2-macroglobulin-proteinase complexes. Pathol Biol (Paris) 1973 Nov;21(Suppl):11–14. [PubMed] [Google Scholar]
- Christensen U., Clemmensen I. Kinetic properties of the primary inhibitor of plasmin from human plasma. Biochem J. 1977 May 1;163(2):389–391. doi: 10.1042/bj1630389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collen D. Identification and some properties of a new fast-reacting plasmin inhibitor in human plasma. Eur J Biochem. 1976 Oct 1;69(1):209–216. doi: 10.1111/j.1432-1033.1976.tb10875.x. [DOI] [PubMed] [Google Scholar]
- Collen D., Wiman B. Turnover of antiplasmin, the fast-acting plasmin inhibitor of plasma. Blood. 1979 Feb;53(2):313–324. [PubMed] [Google Scholar]
- Collen D., de Cock F., Cambiaso C. L., Masson P. A latex agglutination test for rapid quantitative estimation of the plasmin-antipalsmin complex in human plasma. Eur J Clin Invest. 1977 Feb;7(1):21–26. doi: 10.1111/j.1365-2362.1977.tb01565.x. [DOI] [PubMed] [Google Scholar]
- Deutsch D. G., Mertz E. T. Plasminogen: purification from human plasma by affinity chromatography. Science. 1970 Dec 4;170(3962):1095–1096. doi: 10.1126/science.170.3962.1095. [DOI] [PubMed] [Google Scholar]
- Engvall E., Perlmann P. Enzyme-linked immunosorbent assay, Elisa. 3. Quantitation of specific antibodies by enzyme-labeled anti-immunoglobulin in antigen-coated tubes. J Immunol. 1972 Jul;109(1):129–135. [PubMed] [Google Scholar]
- Ganrot P. O. Inhibition of plasmin activity by alpha-2-macroglobulin. Clin Chim Acta. 1967 May;16(2):328–329. doi: 10.1016/0009-8981(67)90201-x. [DOI] [PubMed] [Google Scholar]
- Hall P. K., Roberts R. C. Physical and chemical properties of human plasma alpha2-macroglobulin. Biochem J. 1978 Jul 1;173(1):27–38. doi: 10.1042/bj1730027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harpel P. C., Cooper N. R. Studies on human plasma C1 inactivator-enzyme interactions. I. Mechanisms of interaction with C1s, plasmin, and trypsin. J Clin Invest. 1975 Mar;55(3):593–604. doi: 10.1172/JCI107967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harpel P. C., Hayes M. B., Hugli T. E. Heat-induced fragmentation of human alpha 2-macroglobulin. J Biol Chem. 1979 Sep 10;254(17):8669–8678. [PubMed] [Google Scholar]
- Harpel P. C., Hayes M. B. Immunoimmobilization of alpha 2-macroglobulin--beta-trypsin complexes: a novel approach for the biochemical characterization of modulator--protease interactions. Anal Biochem. 1980 Oct;108(1):166–175. doi: 10.1016/0003-2697(80)90708-3. [DOI] [PubMed] [Google Scholar]
- Harpel P. C. Human alpha2-macroglobulin. Methods Enzymol. 1976;45:639–652. doi: 10.1016/s0076-6879(76)45055-3. [DOI] [PubMed] [Google Scholar]
- Harpel P. C., Mosesson M. W. Degradation of human fibrinogen by plasms alpha2-macroglobulin-enzyme complexes. J Clin Invest. 1973 Sep;52(9):2175–2184. doi: 10.1172/JCI107402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harpel P. C. Plasmin inhibitor interactions. The effectiveness of alpha2-plasmin inhibitor in the presence of alpha2-macroglobulin. J Exp Med. 1977 Oct 1;146(4):1033–1040. doi: 10.1084/jem.146.4.1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harpel P. C., Rosenberg R. D. Alpha 2-macroglobulin and antithrombin-heparin cofactor: modulators of hemostatic and inflammatory reactions. Alpha 2-macroglobulin. Prog Hemost Thromb. 1976;3:145–189. [PubMed] [Google Scholar]
- Haupt H., Heimburger N. Humanserumproteine mit hoher Affinität zu Carboxymethylcellulose. I. Isolierung von Lysozym, C1q und bisher unbekannten -Globulinen. Hoppe Seylers Z Physiol Chem. 1972 Jul;353(7):1125–1132. [PubMed] [Google Scholar]
- Heimburger N., Haupt H., Kranz T., Baudner S. Humanserumproteine mit hoher Affinität zu Carboxymethylcellulose. II. Physikalisch-chemische und immunologische Charakterisierung eines histidinreichen 3,8S- 2 -Glykoproteins (CM-Protein I. Hoppe Seylers Z Physiol Chem. 1972 Jul;353(7):1133–1140. [PubMed] [Google Scholar]
- Highsmith R. F., Rosenberg R. D. The inhibition of human plasmin by human antithrombin-heparin cofactor. J Biol Chem. 1974 Jul 25;249(14):4335–4338. [PubMed] [Google Scholar]
- Johnson P. M., Faulk W. P. Rheumatoid factor: its nature, specificity, and production in rheumatoid arthritis. Clin Immunol Immunopathol. 1976 Nov;6(3):414–430. doi: 10.1016/0090-1229(76)90094-5. [DOI] [PubMed] [Google Scholar]
- Lijnen H. R., Hoylaerts M., Collen D. Isolation and characterization of a human plasma protein with affinity for the lysine binding sites in plasminogen. Role in the regulation of fibrinolysis and identification as histidine-rich glycoprotein. J Biol Chem. 1980 Nov 10;255(21):10214–10222. [PubMed] [Google Scholar]
- March S. C., Parikh I., Cuatrecasas P. A simplified method for cyanogen bromide activation of agarose for affinity chromatography. Anal Biochem. 1974 Jul;60(1):149–152. doi: 10.1016/0003-2697(74)90139-0. [DOI] [PubMed] [Google Scholar]
- Moroi M., Aoki N. Isolation and characterization of alpha2-plasmin inhibitor from human plasma. A novel proteinase inhibitor which inhibits activator-induced clot lysis. J Biol Chem. 1976 Oct 10;251(19):5956–5965. [PubMed] [Google Scholar]
- Müllertz S., Clemmensen I. The primary inhibitor of plasmin in human plasma. Biochem J. 1976 Dec 1;159(3):545–553. doi: 10.1042/bj1590545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NISONOFF A. ENZYMATIC DIGESTION OF RABBIT GAMMA GLOBULIN AND ANTIBODY AND CHROMATOGRAPHY OF DIGESTION PRODUCTS. Methods Med Res. 1964;10:134–141. [PubMed] [Google Scholar]
- Plow E. F., de Cock F., Collen D. Immunochemical characterization of the plasmin-antiplasmin system. Basis for the specific detection of the plasmin-antiplasmin complex by latex agglutination assays. J Lab Clin Med. 1979 Feb;93(2):199–209. [PubMed] [Google Scholar]
- Ratnoff O. D., Pensky J., Ogston D., Naff G. B. The inhibition of plasmin, plasma kallikrein, plasma permeability factor, and the C'1r subcomponent of the first component of complement by serum C'1 esterase inhibitor. J Exp Med. 1969 Feb 1;129(2):315–331. doi: 10.1084/jem.129.2.315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rimon A., Shamash Y., Shapiro B. The plasmin inhibitor of human plasma. IV. Its action on plasmin, trypsin, chymotrypsin, and thrombin. J Biol Chem. 1966 Nov 10;241(21):5102–5107. [PubMed] [Google Scholar]
- Shuman M. A., Majerus P. W. The measurement of thrombin in clotting blood by radioimmunoassay. J Clin Invest. 1976 Nov;58(5):1249–1258. doi: 10.1172/JCI108579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Travis J., Bowen J., Tewksbury D., Johnson D., Pannell R. Isolation of albumin from whole human plasma and fractionation of albumin-depleted plasma. Biochem J. 1976 Aug 1;157(2):301–306. doi: 10.1042/bj1570301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiman B., Collen D. On the kinetics of the reaction between human antiplasmin and plasmin. Eur J Biochem. 1978 Mar 15;84(2):573–578. doi: 10.1111/j.1432-1033.1978.tb12200.x. [DOI] [PubMed] [Google Scholar]
- Wiman B., Collen D. Purification and characterization of human antiplasmin, the fast-acting plasmin inhibitor in plasma. Eur J Biochem. 1977 Aug 15;78(1):19–26. doi: 10.1111/j.1432-1033.1977.tb11709.x. [DOI] [PubMed] [Google Scholar]


