Abstract
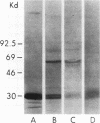
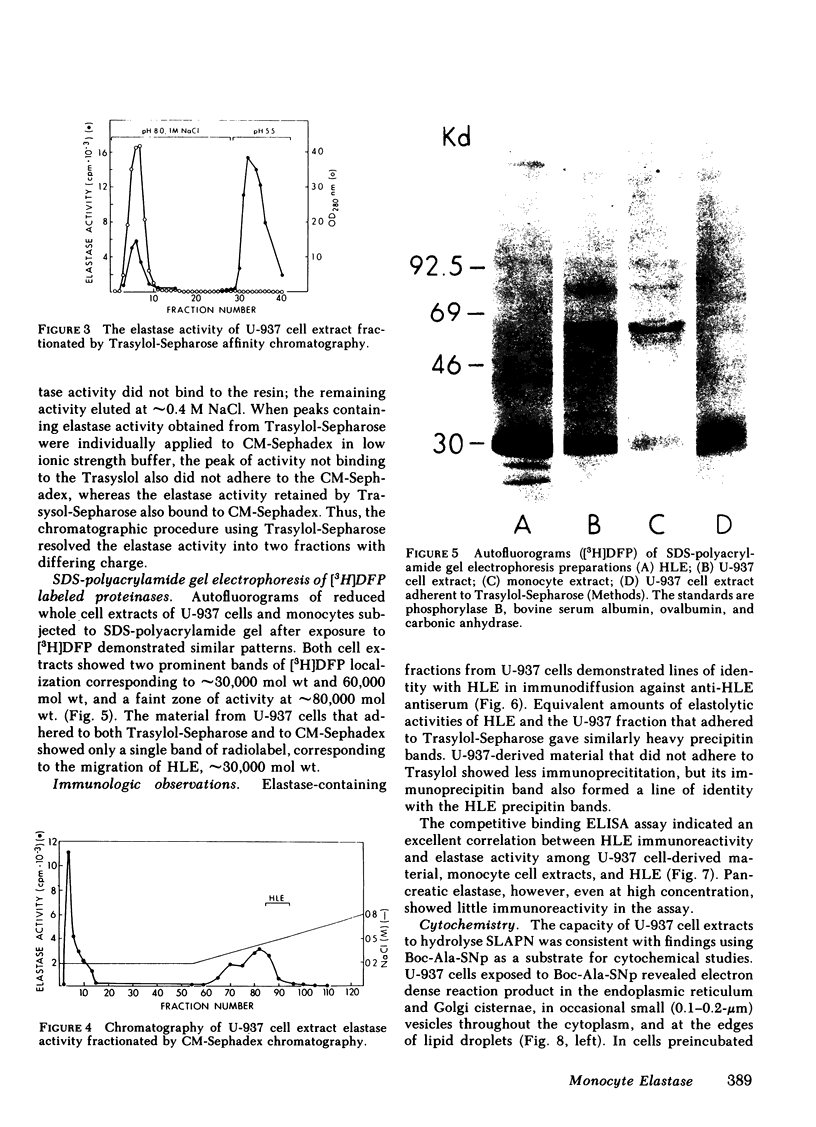
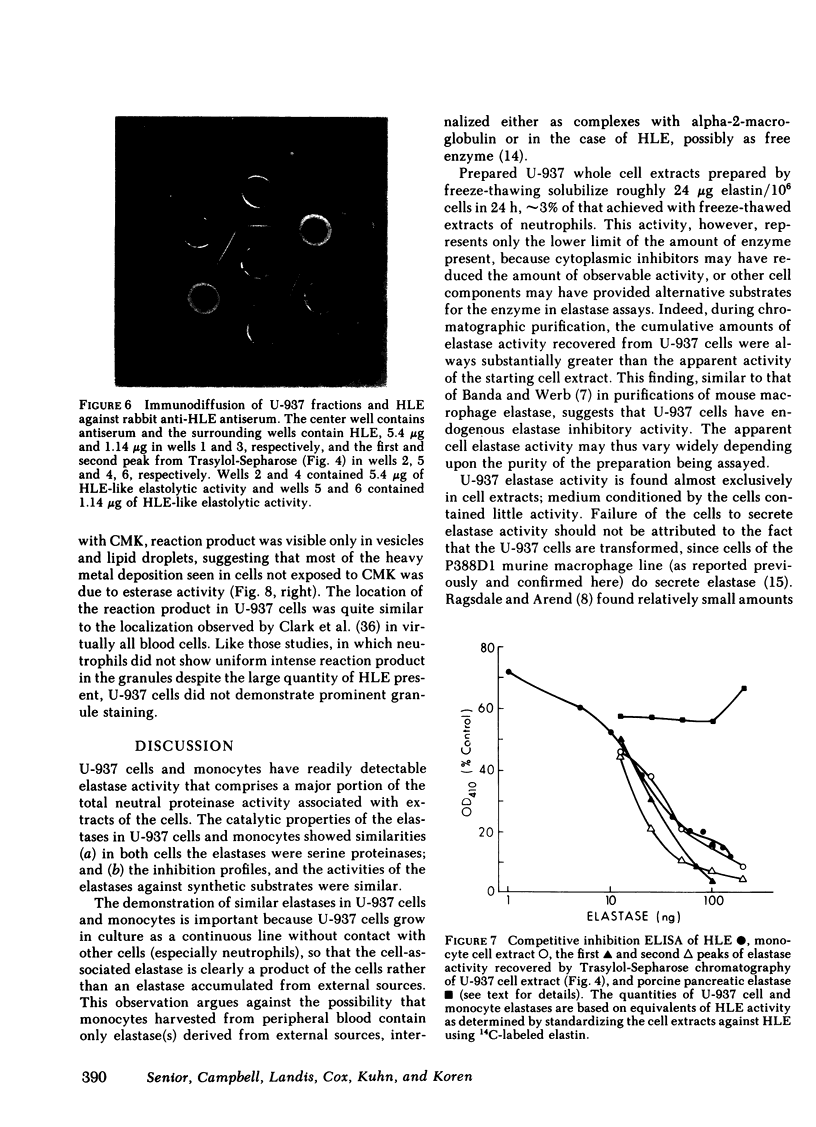
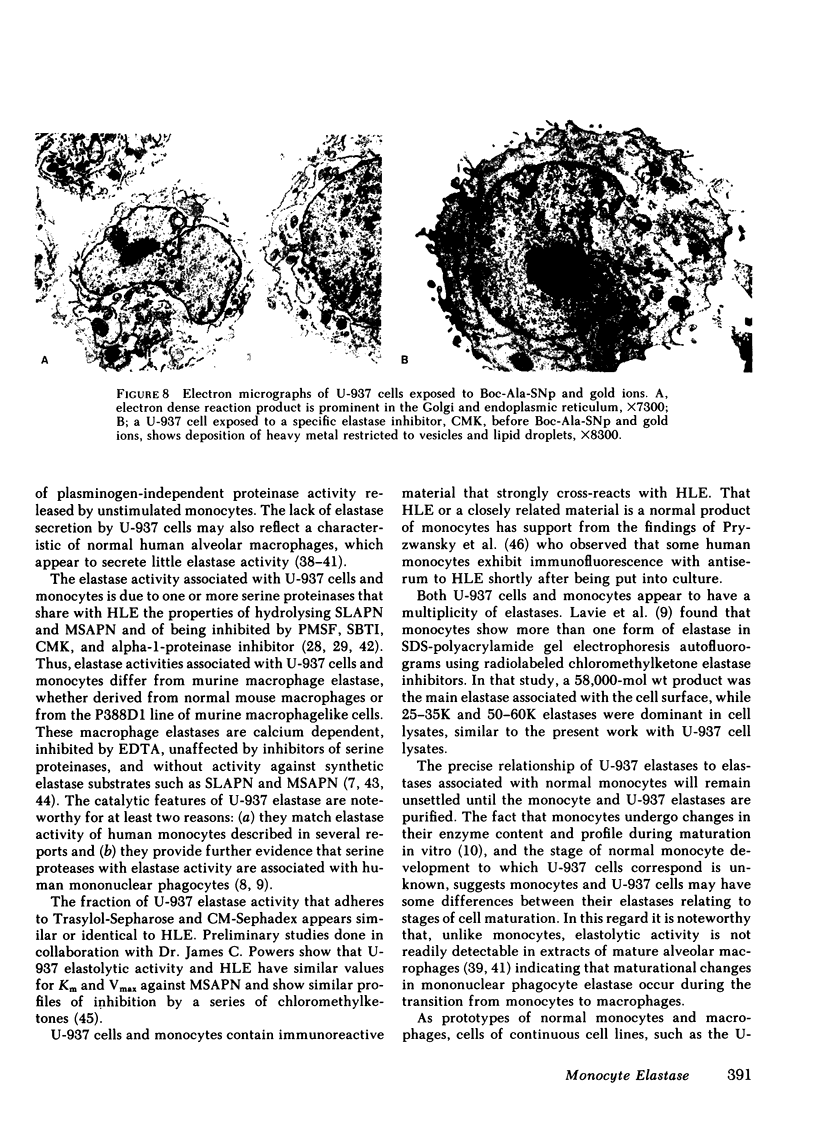
As an approach to facilitating the understanding of proteinases associated with monocytes we have studied U-937 monocytelike cells. Elastase activity was identified in U-937 cell extracts and compared to monocyte elastase activity, neutrophil elastase, and the elastase activity from a continuous line of murine macrophagelike cells (P388D1). Serine proteinase activity which solubilized 14C-labeled elastin accounted for >90% of the neutral proteinase activity of both U-937 cells and monocyte extracts. U-937 cell and monocyte elastase activities were similar catalytically, resembling neutrophil elastase. U-937 cells and monocytes showed other similarities: (a) both had activities reacting with [3H]diisopropylfluorophosphate that migrated in sodium dodecyl sulfate (SDS) polyacrylamide gels at ∼30,000 and 60,000 daltons and (b) both contained material that cross-reacted with antiserum raised to neutrophil elastase. Preliminary characterization of U-937 cell elastase activity by affinity chromatography and ion-exchange chromatography suggested the presence of at least two distinct elastases. Minimal elastase activity was found in U-937 cell-conditioned medium, indicating that the activity is not spontaneously released by the cells. In contrast to the elastase activity associated with U-937 cells and monocytes, the elastase activity associated with P388D1 cells was a metalloproteinase and was found principally in the culture medium. These results indicate (a) U-937 cells will be useful for further investigation of proteinases associated with normal monocytes; (b) monocytes and U-937 cells contain material with catalytic and immunologic similarities to neutrophil elastase; (c) monocyte elastase activity differs from elastase activity secreted by murine macrophages and murine macrophagelike cells of the P388D1 line.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Banda M. J., Werb Z. Mouse macrophage elastase. Purification and characterization as a metalloproteinase. Biochem J. 1981 Feb 1;193(2):589–605. doi: 10.1042/bj1930589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baugh R. J., Travis J. Human leukocyte granule elastase: rapid isolation and characterization. Biochemistry. 1976 Feb 24;15(4):836–841. doi: 10.1021/bi00649a017. [DOI] [PubMed] [Google Scholar]
- Bielefeld D. R., Senior R. M., Yu S. Y. A new method for determination of elastolytic activity using (14C) labeled elastin and its application to leukocytic elastase. Biochem Biophys Res Commun. 1975 Dec 15;67(4):1553–1559. doi: 10.1016/0006-291x(75)90203-x. [DOI] [PubMed] [Google Scholar]
- Bieth J., Spiess B., Wermuth C. G. The synthesis and analytical use of a highly sensitive and convenient substrate of elastase. Biochem Med. 1974 Dec;11(4):350–357. doi: 10.1016/0006-2944(74)90134-3. [DOI] [PubMed] [Google Scholar]
- Bodel P., Ralph P., Wenc K., Long J. C. Endogenous pyrogen production by Hodgkin's disease and human histiocytic lymphoma cell lines in vitro. J Clin Invest. 1980 Feb;65(2):514–518. doi: 10.1172/JCI109695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böyum A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand J Clin Lab Invest Suppl. 1968;97:77–89. [PubMed] [Google Scholar]
- Campbell E. J., White R. R., Senior R. M., Rodriguez R. J., Kuhn C. Receptor-mediated binding and internalization of leukocyte elastase by alveolar macrophages in vitro. J Clin Invest. 1979 Sep;64(3):824–833. doi: 10.1172/JCI109530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carp H., Janoff A. In vitro suppression of serum elastase-inhibitory capacity by reactive oxygen species generated by phagocytosing polymorphonuclear leukocytes. J Clin Invest. 1979 Apr;63(4):793–797. doi: 10.1172/JCI109364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castillo M. J., Nakajima K., Zimmerman M., Powers J. C. Sensitive substrates for human leukocyte and porcine pancreatic elastase: a study of the merits of various chromophoric and fluorogenic leaving groups in assays for serine proteases. Anal Biochem. 1979 Oct 15;99(1):53–64. doi: 10.1016/0003-2697(79)90043-5. [DOI] [PubMed] [Google Scholar]
- Clark J. M., Vaughan D. W., Aiken B. M., Kagan H. M. Elastase-like enzymes in human neutrophils localized by ultrastructural cytochemistry. J Cell Biol. 1980 Jan;84(1):102–119. doi: 10.1083/jcb.84.1.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danø K., Reich E. Serine enzymes released by cultured neoplastic cells. J Exp Med. 1978 Mar 1;147(3):745–757. doi: 10.1084/jem.147.3.745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Cremoux H., Hornebeck W., Jaurand M. C., Bignon J., Robert L. Partial characterisation of an elastase-like enzyme secreted by human and monkey alveolar macrophages. J Pathol. 1978 Aug;125(4):171–177. doi: 10.1002/path.1711250402. [DOI] [PubMed] [Google Scholar]
- Fischer D. G., Pike M. C., Koren H. S., Snyderman R. Chemotactically responsive and nonresposive forms of a continuous human monocyte cell line. J Immunol. 1980 Jul;125(1):463–465. [PubMed] [Google Scholar]
- Gadek J. E., Fells G. A., Crystal R. G. Cigarette smoking induces functional antiprotease deficiency in the lower respiratory tract of humans. Science. 1979 Dec 14;206(4424):1315–1316. doi: 10.1126/science.316188. [DOI] [PubMed] [Google Scholar]
- Gordon S., Todd J., Cohn Z. A. In vitro synthesis and secretion of lysozyme by mononuclear phagocytes. J Exp Med. 1974 May 1;139(5):1228–1248. doi: 10.1084/jem.139.5.1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green M. R., Lin J. S., Berman L. B., Osman M. M., Cerreta J. M., Mandl I., Turino G. M. Elastolytic activity of alveolar macrophages in normal dogs and human subjects. J Lab Clin Med. 1979 Oct;94(4):549–562. [PubMed] [Google Scholar]
- Hinman L. M., Stevens C. A., Matthay R. A., Gee J. B. Elastase and lysozyme activities in human alveolar macrophages. Effects of cigarette smoking. Am Rev Respir Dis. 1980 Feb;121(2):263–271. doi: 10.1164/arrd.1980.121.2.263. [DOI] [PubMed] [Google Scholar]
- Janoff A., Carp H., Lee D. K., Drew R. T. Cigarette smoke inhalation decreases alpha 1-antitrypsin activity in rat lung. Science. 1979 Dec 14;206(4424):1313–1314. doi: 10.1126/science.316187. [DOI] [PubMed] [Google Scholar]
- Janoff A. Human granulocyte elastase. Further delineation of its role in connective tissue damage. Am J Pathol. 1972 Sep;68(3):579–592. [PMC free article] [PubMed] [Google Scholar]
- Janoff A., Sloan B., Weinbaum G., Damiano V., Sandhaus R. A., Elias J., Kimbel P. Experimental emphysema induced with purified human neutrophil elastase: tissue localization of the instilled protease. Am Rev Respir Dis. 1977 Mar;115(3):461–478. doi: 10.1164/arrd.1977.115.3.461. [DOI] [PubMed] [Google Scholar]
- Jones P. A., Werb Z. Degradation of connective tissue matrices by macrophages. II. Influence of matrix composition on proteolysis of glycoproteins, elastin, and collagen by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1527–1536. doi: 10.1084/jem.152.6.1527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalb V. F., Jr, Bernlohr R. W. A new spectrophotometric assay for protein in cell extracts. Anal Biochem. 1977 Oct;82(2):362–371. doi: 10.1016/0003-2697(77)90173-7. [DOI] [PubMed] [Google Scholar]
- Kearney J. F., Radbruch A., Liesegang B., Rajewsky K. A new mouse myeloma cell line that has lost immunoglobulin expression but permits the construction of antibody-secreting hybrid cell lines. J Immunol. 1979 Oct;123(4):1548–1550. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lavie G., Zucker-Franklin D., Franklin E. C. Elastase-type proteases on the surface of human blood monocytes: possible role in amyloid formation. J Immunol. 1980 Jul;125(1):175–180. [PubMed] [Google Scholar]
- Matheson N. R., Wong P. S., Travis J. Enzymatic inactivation of human alpha-1-proteinase inhibitor by neutrophil myeloperoxidase. Biochem Biophys Res Commun. 1979 May 28;88(2):402–409. doi: 10.1016/0006-291x(79)92062-x. [DOI] [PubMed] [Google Scholar]
- McDonald J. A., Kelley D. G. Degradation of fibronectin by human leukocyte elastase. Release of biologically active fragments. J Biol Chem. 1980 Sep 25;255(18):8848–8858. [PubMed] [Google Scholar]
- Musson R. A., Shafran H., Henson P. M. Intracellular levels and stimulated release of lysosomal enzymes from human peripheral blood monocytes and monocyte-derived macrophages. J Reticuloendothel Soc. 1980 Sep;28(3):249–264. [PubMed] [Google Scholar]
- Newman S. L., Musson R. A., Henson P. M. Development of functional complement receptors during in vitro maturation of human monocytes into macrophages. J Immunol. 1980 Nov;125(5):2236–2244. [PubMed] [Google Scholar]
- Pike M. C., Fischer D. G., Koren H. S., Snyderman R. Development of specific receptors for N-formylated chemotactic peptides in a human monocyte cell line stimulated with lymphokines. J Exp Med. 1980 Jul 1;152(1):31–40. doi: 10.1084/jem.152.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powers J. C., Gupton B. F., Harley A. D., Nishino N., Whitley R. J. Specificity of porcine pancreatic elastase, human leukocyte elastase and cathepsin G. Inhibition with peptide chloromethyl ketones. Biochim Biophys Acta. 1977 Nov 23;485(1):156–166. doi: 10.1016/0005-2744(77)90203-0. [DOI] [PubMed] [Google Scholar]
- Pryzwansky K. B., Martin L. E., Spitznagel J. K. Immunocytochemical localization of myeloperoxidase, lactoferrin, lysozyme and neutral proteases in human monocytes and neutrophilic granulocytes. J Reticuloendothel Soc. 1978 Sep;24(3):295–310. [PubMed] [Google Scholar]
- Ragsdale C. G., Arend W. P. Neutral protease secretion by human monocytes. Effect of surface-bound immune complexes. J Exp Med. 1979 Apr 1;149(4):954–968. doi: 10.1084/jem.149.4.954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralph P., Moore M. A., Nilsson K. Lysozyme synthesis by established human and murine histiocytic lymphoma cell lines. J Exp Med. 1976 Jun 1;143(6):1528–1533. doi: 10.1084/jem.143.6.1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez R. J., White R. R., Senior R. M., Levine E. A. Elastase release from human alveolar macrophages: comparison between smokers and nonsmokers. Science. 1977 Oct 21;198(4314):313–314. doi: 10.1126/science.910131. [DOI] [PubMed] [Google Scholar]
- Senior R. M., Tegner H., Kuhn C., Ohlsson K., Starcher B. C., Pierce J. A. The induction of pulmonary emphysema with human leukocyte elastase. Am Rev Respir Dis. 1977 Sep;116(3):469–475. doi: 10.1164/arrd.1977.116.3.469. [DOI] [PubMed] [Google Scholar]
- Sundström C., Nilsson K. Establishment and characterization of a human histiocytic lymphoma cell line (U-937). Int J Cancer. 1976 May 15;17(5):565–577. doi: 10.1002/ijc.2910170504. [DOI] [PubMed] [Google Scholar]
- Varani J., Johnson K., Kaplan J. Development of a solid-phase assay for measurement of proteolytic enzyme activity. Anal Biochem. 1980 Sep 15;107(2):377–384. doi: 10.1016/0003-2697(80)90399-1. [DOI] [PubMed] [Google Scholar]
- WROBLEWSKI F., LADUE J. S. Lactic dehydrogenase activity in blood. Proc Soc Exp Biol Med. 1955 Oct;90(1):210–213. doi: 10.3181/00379727-90-21985. [DOI] [PubMed] [Google Scholar]
- Weiner R. S., Shah V. O. Purification of human monocytes: isolation and collection of large numbers of peripheral blood monocytes. J Immunol Methods. 1980;36(2):89–97. doi: 10.1016/0022-1759(80)90034-4. [DOI] [PubMed] [Google Scholar]
- Werb Z., Bainton D. F., Jones P. A. Degradation of connective tissue matrices by macrophages. III. Morphological and biochemical studies on extracellular, pericellular, and intracellular events in matrix proteolysis by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1537–1553. doi: 10.1084/jem.152.6.1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Banda M. J., Jones P. A. Degradation of connective tissue matrices by macrophages. I. Proteolysis of elastin, glycoproteins, and collagen by proteinases isolated from macrophages. J Exp Med. 1980 Nov 1;152(5):1340–1357. doi: 10.1084/jem.152.5.1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Foley R., Munck A. Glucocorticoid receptors and glucocorticoid-sensitive secretion of neutral proteinases in a macrophage line. J Immunol. 1978 Jul;121(1):115–121. [PubMed] [Google Scholar]
- Werb Z., Gordon S. Elastase secretion by stimulated macrophages. Characterization and regulation. J Exp Med. 1975 Aug 1;142(2):361–377. doi: 10.1084/jem.142.2.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White R., Lin H. S., Kuhn C., 3rd Elastase secretion by peritoneal exudative and alveolar macrophages. J Exp Med. 1977 Sep 1;146(3):802–808. doi: 10.1084/jem.146.3.802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wing E. J., Koren H. S., Fischer D. G., Kelley V. Stimulation of a human macrophage-like cell line (U-937) to inhibit multiplication of an intracellular pathogen. J Reticuloendothel Soc. 1981 Apr;29(4):321–328. [PubMed] [Google Scholar]
- Wong P. S., Travis J. Isolation and properties of oxidized alpha-1-proteinase inhibitor from human rheumatoid synovial fluid. Biochem Biophys Res Commun. 1980 Oct 16;96(3):1449–1454. doi: 10.1016/0006-291x(80)90113-8. [DOI] [PubMed] [Google Scholar]
- Zuckerman S. H., Ackerman S. K., Douglas S. D. Long-term human peripheral blood monocyte cultures: establishment, metabolism and morphology of primary human monocyte-macrophage cell cultures. Immunology. 1979 Oct;38(2):401–411. [PMC free article] [PubMed] [Google Scholar]