Abstract
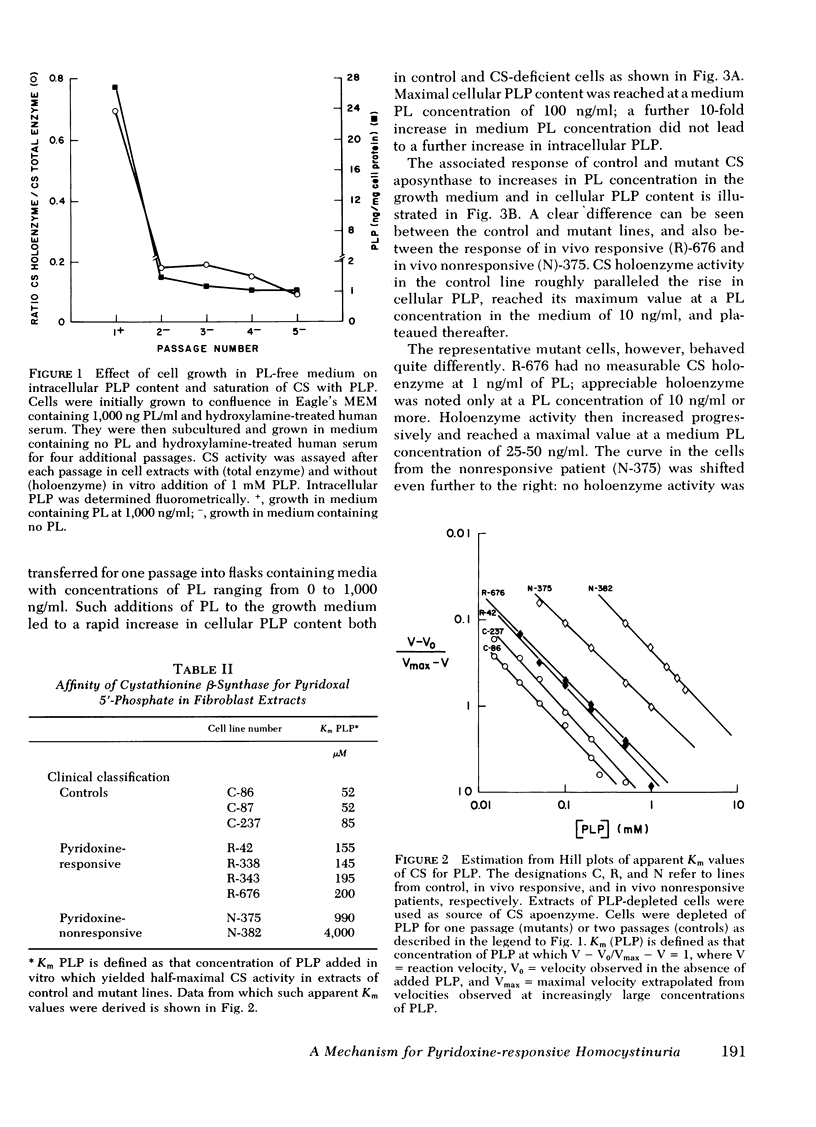
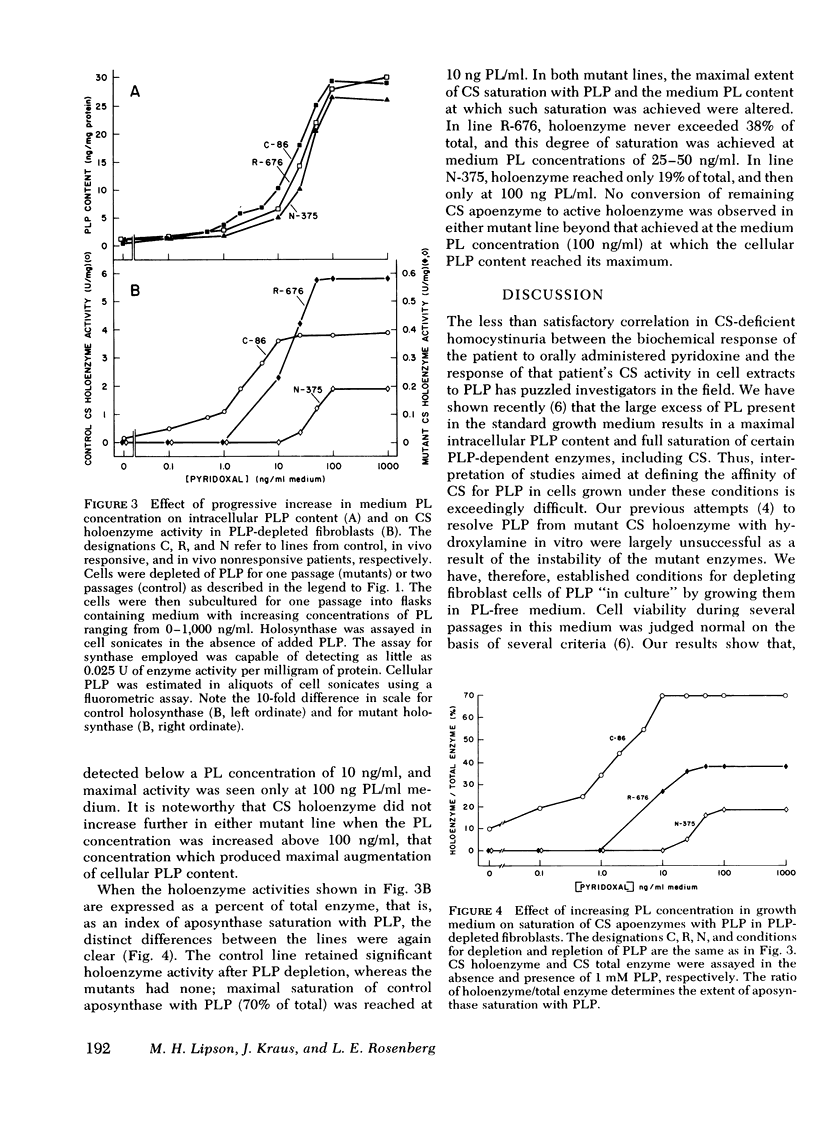
Previous attempts to correlate in vivo pyridoxine-responsiveness with in vitro assays of cystathionine β-synthase activity in synthase-deficient homocystinuric patients have been only partially successful. All such studies, however, have been conducted with extracts of cultured skin fibroblasts grown in medium containing a high concentration (1,000 ng/ml) of pyridoxal. Having recently shown that such growth conditions may obscure important aspects of enzyme-coenzyme interactions by saturating most synthase molecules with their cofactor, pyridoxal 5′-phosphate, we have established conditions for growth of cells in pyridoxal-free medium. Under these conditions, intracellular pyridoxal 5′-phosphate fell by >95%, and saturation of cystathionine β-synthase apoenzyme with pyridoxal 5′-phosphate decreased from a predepletion value of 70% to <10%. When such depleted cells were grown in media containing pyridoxal concentrations ranging from 0 to 1,000 ng/ml, cellular pyridoxal 5′-phosphate reached a maximum of 30 ng/mg cell protein at a medium pyridoxal concentration of 100 ng/ml. Maximal saturation of aposynthase with coenzyme in control cells was reached at a medium pyridoxal concentration of 10 ng/ml. In contrast, maximal saturation of residual aposynthase in cells from an in vivo responsive patient was achieved at a medium pyridoxal concentration of 25-50 ng/ml, whereas that from cells from an in vivo unresponsive patient was reached at 100 ng/ml. Estimates of the affinity of control and mutant cystathionine β-synthase for pyridoxal 5′-phosphate in cell extracts supported the differences observed in intact cells. The apparent Km of cystathionine β-synthase for pyridoxal 5′-phosphate in extracts of depleted cells from four in vivo-responsive patients was two to four times that of control. In contrast, the Km for pyridoxal 5′-phosphate in two lines from in vivo nonresponsive patients was 16- and 63-fold normal. These results suggest that cystathionine β-synthase activity in cells from patients containing a mutant enzyme with a moderately reduced affinity for pyridoxal 5′-phosphate can be increased by pyridoxine supplements in vivo, whereas that from patients whose enzyme has a more dramatically reduced affinity for the coenzyme cannot be so modulated because of limits on the capacity of such cells to accumulate and retain pyridoxal 5′-phosphate.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Fowler B., Kraus J., Packman S., Rosenberg L. E. Homocystinuria. Evidence for three distinct classes of cystathionine beta-synthase mutants in cultured fibroblasts. J Clin Invest. 1978 Mar;61(3):645–653. doi: 10.1172/JCI108976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grigo K. M., Von Redlich D., Glick D. Fluorometric determination of pyridoxal phosphate in microgram samples of tissue. Anal Biochem. 1972 Nov;50(1):28–34. doi: 10.1016/0003-2697(72)90480-0. [DOI] [PubMed] [Google Scholar]
- Kim Y. J., Rosenberg L. E. On the mechanism of pyridoxine responsive homocystinuria. II. Properties of normal and mutant cystathionine beta-synthase from cultured fibroblasts. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4821–4825. doi: 10.1073/pnas.71.12.4821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lumeng L., Li T. K. Characterization of the pyridoxal 5'-phosphate and pyridoxamine 5'-phosphate hydrolase activity in rat liver. Identity with alkaline phosphatase. J Biol Chem. 1975 Oct 25;250(20):8126–8131. [PubMed] [Google Scholar]
- Uhlendorf B. W., Conerly E. B., Mudd S. H. Homocystinuria: studies in tissue culture. Pediatr Res. 1973 Jul;7(7):645–658. doi: 10.1203/00006450-197307000-00008. [DOI] [PubMed] [Google Scholar]


