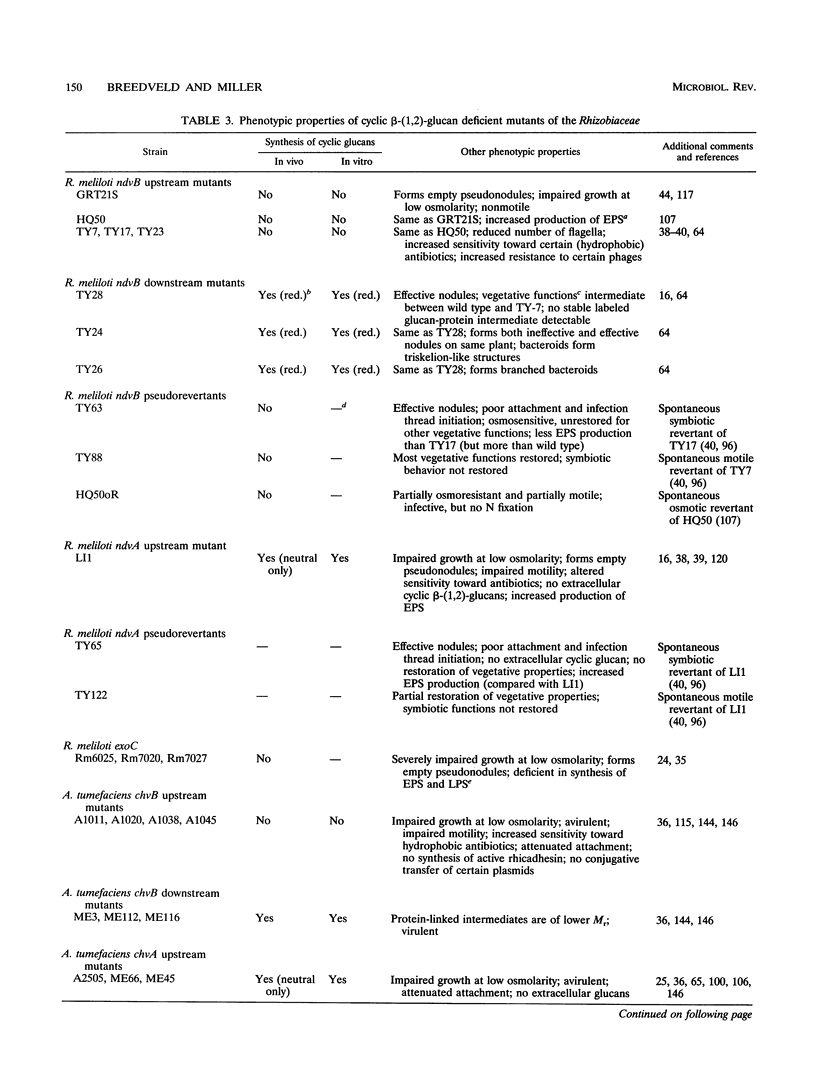
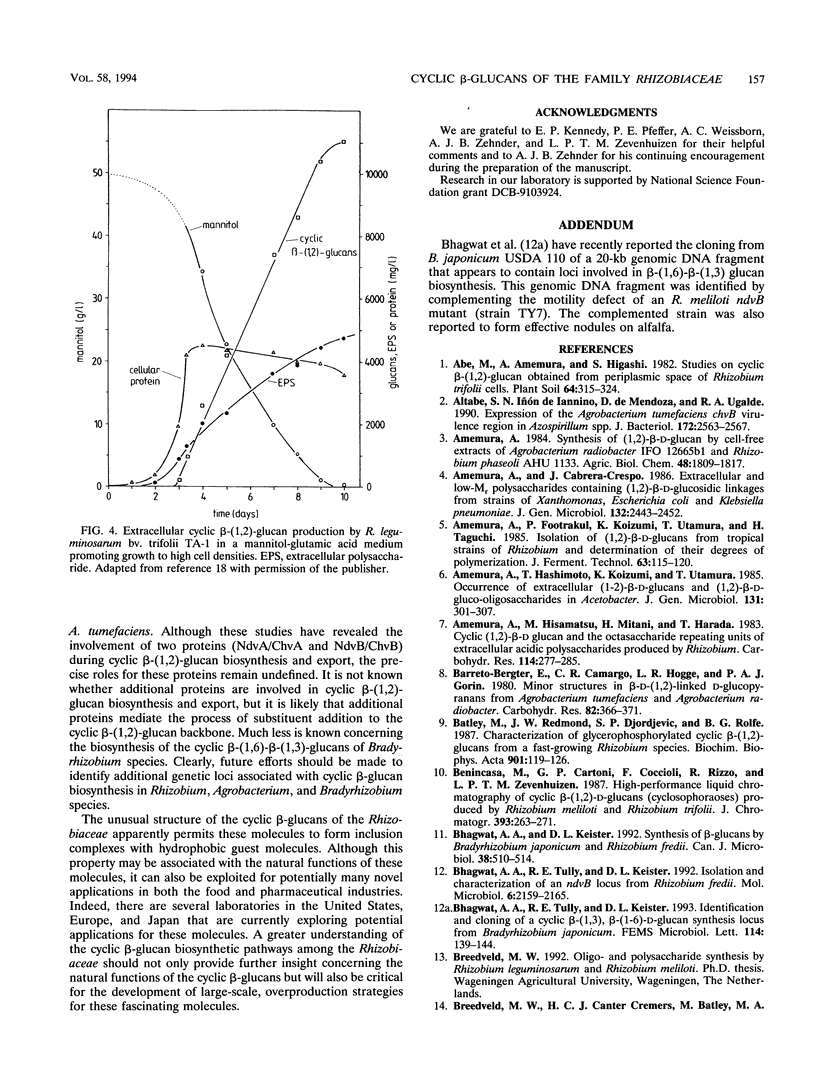
Abstract
Cyclic beta-glucans are low-molecular-weight cell surface carbohydrates that are found almost exclusively in bacteria of the Rhizobiaceae family. These glucans are major cellular constituents, and under certain culture conditions their levels may reach up to 20% of the total cellular dry weight. In Agrobacterium and Rhizobium species, these molecules contain between 17 and 40 glucose residues linked solely by beta-(1,2) glycosidic bonds. In Bradyrhizobium species, the cyclic beta-glucans are smaller (10 to 13 glucose residues) and contain glucose linked by both beta-(1,6) and beta-(1,3) glycosidic bonds. In some rhizobial strains, the cyclic beta-glucans are unsubstituted, whereas in other rhizobia these molecules may become highly substituted with moieties such as sn-1-phosphoglycerol. To date, two genetic loci specifically associated with cyclic beta-glucan biosynthesis have been identified in Rhizobium (ndvA and ndvB) and Agrobacterium (chvA and chvB) species. Mutants with mutations at these loci have been shown to be impaired in their ability to grow in hypoosmotic media, have numerous alterations in their cell surface properties, and are also impaired in their ability to infect plants. The present review will examine the structure and occurrence of the cyclic beta-glucans in a variety of species of the Rhizobiaceae. The possible functions of these unique molecules in the free-living bacteria as well as during plant infection will be discussed.
Full text
PDF













Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altabe S., Iñn de Iannino N., de Mendoza D., Ugalde R. A. Expression of the Agrobacterium tumefaciens chvB virulence region in Azospirillum spp. J Bacteriol. 1990 May;172(5):2563–2567. doi: 10.1128/jb.172.5.2563-2567.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amemura A., Cabrera-Crespo J. Extracellular oligosaccharides and low-Mr polysaccharides containing (1----2)-beta-D-glucosidic linkages from strains of Xanthomonas, Escherichia coli and Klebsiella pneumoniae. J Gen Microbiol. 1986 Sep;132(9):2443–2452. doi: 10.1099/00221287-132-9-2443. [DOI] [PubMed] [Google Scholar]
- Bhagwat A. A., Tully R. E., Keister D. L. Identification and cloning of a cyclic beta-(1-->3), beta-(1-->6)-D-glucan synthesis locus from Bradyrhizobium japonicum. FEMS Microbiol Lett. 1993 Dec 1;114(2):139–144. doi: 10.1111/j.1574-6968.1993.tb06564.x. [DOI] [PubMed] [Google Scholar]
- Bhagwat A. A., Tully R. E., Keister D. L. Isolation and characterization of an ndvB locus from Rhizobium fredii. Mol Microbiol. 1992 Aug;6(15):2159–2165. doi: 10.1111/j.1365-2958.1992.tb01389.x. [DOI] [PubMed] [Google Scholar]
- Breedveld M. W., Cremers H. C., Batley M., Posthumus M. A., Zevenhuizen L. P., Wijffelman C. A., Zehnder A. J. Polysaccharide synthesis in relation to nodulation behavior of Rhizobium leguminosarum. J Bacteriol. 1993 Feb;175(3):750–757. doi: 10.1128/jb.175.3.750-757.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breedveld M. W., Yoo J. S., Reinhold V. N., Miller K. J. Synthesis of glycerophosphorylated cyclic beta-(1,2)-glucans by Rhizobium meliloti ndv mutants. J Bacteriol. 1994 Feb;176(4):1047–1051. doi: 10.1128/jb.176.4.1047-1051.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breedveld M. W., Zevenhuizen L. P., Zehnder A. J. Excessive excretion of cyclic beta-(1,2)-glucan by Rhizobium trifolii TA-1. Appl Environ Microbiol. 1990 Jul;56(7):2080–2086. doi: 10.1128/aem.56.7.2080-2086.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breedveld M. W., Zevenhuizen L. P., Zehnder A. J. Synthesis of cyclic beta-(1,2)-glucans by Rhizobium leguminosarum biovar trifolii TA-1: factors influencing excretion. J Bacteriol. 1992 Oct;174(20):6336–6342. doi: 10.1128/jb.174.20.6336-6342.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bundle D. R., Cherwonogrodzky J. W., Perry M. B. Characterization of Brucella polysaccharide B. Infect Immun. 1988 May;56(5):1101–1106. doi: 10.1128/iai.56.5.1101-1106.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cangelosi G. A., Hung L., Puvanesarajah V., Stacey G., Ozga D. A., Leigh J. A., Nester E. W. Common loci for Agrobacterium tumefaciens and Rhizobium meliloti exopolysaccharide synthesis and their roles in plant interactions. J Bacteriol. 1987 May;169(5):2086–2091. doi: 10.1128/jb.169.5.2086-2091.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cangelosi G. A., Martinetti G., Leigh J. A., Lee C. C., Thienes C., Theines C., Nester E. W. Role for [corrected] Agrobacterium tumefaciens ChvA protein in export of beta-1,2-glucan. J Bacteriol. 1989 Mar;171(3):1609–1615. doi: 10.1128/jb.171.3.1609-1615.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cangelosi G. A., Martinetti G., Nester E. W. Osmosensitivity phenotypes of Agrobacterium tumefaciens mutants that lack periplasmic beta-1,2-glucan. J Bacteriol. 1990 Apr;172(4):2172–2174. doi: 10.1128/jb.172.4.2172-2174.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke H. R., Leigh J. A., Douglas C. J. Molecular signals in the interactions between plants and microbes. Cell. 1992 Oct 16;71(2):191–199. doi: 10.1016/0092-8674(92)90348-g. [DOI] [PubMed] [Google Scholar]
- Cohen J. L., Miller K. J. A novel membrane-bound glucosyltransferase from Bradyrhizobium japonicum. J Bacteriol. 1991 Jul;173(14):4271–4276. doi: 10.1128/jb.173.14.4271-4276.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Csonka L. N., Hanson A. D. Prokaryotic osmoregulation: genetics and physiology. Annu Rev Microbiol. 1991;45:569–606. doi: 10.1146/annurev.mi.45.100191.003033. [DOI] [PubMed] [Google Scholar]
- DEDONDER R. A., HASSID W. Z. THE ENZYMATIC SYNTHESIS OF A (BETA-I,2-)-LINKED GLUCAN BY AN EXTRACT OF RHIZOBIUM JAPONICUM. Biochim Biophys Acta. 1964 Aug 19;90:239–248. doi: 10.1016/0304-4165(64)90187-4. [DOI] [PubMed] [Google Scholar]
- Delcour A. H., Adler J., Kung C., Martinac B. Membrane-derived oligosaccharides (MDO's) promote closing of an E. coli porin channel. FEBS Lett. 1992 Jun 15;304(2-3):216–220. doi: 10.1016/0014-5793(92)80622-n. [DOI] [PubMed] [Google Scholar]
- Dickstein R., Bisseling T., Reinhold V. N., Ausubel F. M. Expression of nodule-specific genes in alfalfa root nodules blocked at an early stage of development. Genes Dev. 1988 Jun;2(6):677–687. doi: 10.1101/gad.2.6.677. [DOI] [PubMed] [Google Scholar]
- Douglas C. J., Staneloni R. J., Rubin R. A., Nester E. W. Identification and genetic analysis of an Agrobacterium tumefaciens chromosomal virulence region. J Bacteriol. 1985 Mar;161(3):850–860. doi: 10.1128/jb.161.3.850-860.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dylan T., Helinski D. R., Ditta G. S. Hypoosmotic adaptation in Rhizobium meliloti requires beta-(1----2)-glucan. J Bacteriol. 1990 Mar;172(3):1400–1408. doi: 10.1128/jb.172.3.1400-1408.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dylan T., Ielpi L., Stanfield S., Kashyap L., Douglas C., Yanofsky M., Nester E., Helinski D. R., Ditta G. Rhizobium meliloti genes required for nodule development are related to chromosomal virulence genes in Agrobacterium tumefaciens. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4403–4407. doi: 10.1073/pnas.83.12.4403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dylan T., Nagpal P., Helinski D. R., Ditta G. S. Symbiotic pseudorevertants of Rhizobium meliloti ndv mutants. J Bacteriol. 1990 Mar;172(3):1409–1417. doi: 10.1128/jb.172.3.1409-1417.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiedler W., Rotering H. Properties of Escherichia coli mutants lacking membrane-derived oligosaccharides. J Biol Chem. 1988 Oct 15;263(29):14684–14689. [PubMed] [Google Scholar]
- Geiger O., Russo F. D., Silhavy T. J., Kennedy E. P. Membrane-derived oligosaccharides affect porin osmoregulation only in media of low ionic strength. J Bacteriol. 1992 Feb;174(4):1410–1413. doi: 10.1128/jb.174.4.1410-1413.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geiger O., Weissborn A. C., Kennedy E. P. Biosynthesis and excretion of cyclic glucans by Rhizobium meliloti 1021. J Bacteriol. 1991 May;173(9):3021–3024. doi: 10.1128/jb.173.9.3021-3024.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geremia R. A., Cavaignac S., Zorreguieta A., Toro N., Olivares J., Ugalde R. A. A Rhizobium meliloti mutant that forms ineffective pseudonodules in alfalfa produces exopolysaccharide but fails to form beta-(1----2) glucan. J Bacteriol. 1987 Feb;169(2):880–884. doi: 10.1128/jb.169.2.880-884.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg D. E., Rumley M. K., Kennedy E. P. Biosynthesis of membrane-derived oligosaccharides: a periplasmic phosphoglyceroltransferase. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5513–5517. doi: 10.1073/pnas.78.9.5513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gore R. S., Miller K. J. Cell surface carbohydrates of microaerobic, nitrogenase-active, continuous cultures of Bradyrhizobium sp. strain 32H1. J Bacteriol. 1992 Dec;174(23):7838–7840. doi: 10.1128/jb.174.23.7838-7840.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gore R. S., Miller K. J. Cyclic [beta]-1,6 -1,3 Glucans Are Synthesized by Bradyrhizobium japonicum Bacteroids within Soybean (Glycine max) Root Nodules. Plant Physiol. 1993 May;102(1):191–194. doi: 10.1104/pp.102.1.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray J. X., Rolfe B. G. Exopolysaccharide production in Rhizobium and its role in invasion. Mol Microbiol. 1990 Sep;4(9):1425–1431. doi: 10.1111/j.1365-2958.1990.tb02052.x. [DOI] [PubMed] [Google Scholar]
- Göttfert M. Regulation and function of rhizobial nodulation genes. FEMS Microbiol Rev. 1993 Jan;10(1-2):39–63. doi: 10.1111/j.1574-6968.1993.tb05863.x. [DOI] [PubMed] [Google Scholar]
- Halverson L. J., Stacey G. Signal exchange in plant-microbe interactions. Microbiol Rev. 1986 Jun;50(2):193–225. doi: 10.1128/mr.50.2.193-225.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harada T. Special bacterial polysaccharides and polysaccharases. Biochem Soc Symp. 1983;48:97–116. [PubMed] [Google Scholar]
- Hisamatsu M. Cyclic (1----2)-beta-D-glucans (cyclosophorans) produced by Agrobacterium and Rhizobium species. Carbohydr Res. 1992 Jul 2;231:137–146. doi: 10.1016/0008-6215(92)84014-j. [DOI] [PubMed] [Google Scholar]
- Hoelzle I., Streeter J. G. Increased Accumulation of Trehalose in Rhizobia Cultured under 1% Oxygen. Appl Environ Microbiol. 1990 Oct;56(10):3213–3215. doi: 10.1128/aem.56.10.3213-3215.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland I. B., Kenny B., Blight M. Haemolysin secretion from E coli. Biochimie. 1990 Feb-Mar;72(2-3):131–141. doi: 10.1016/0300-9084(90)90138-7. [DOI] [PubMed] [Google Scholar]
- Höltje J. V., Fiedler W., Rotering H., Walderich B., van Duin J. Lysis induction of Escherichia coli by the cloned lysis protein of the phage MS2 depends on the presence of osmoregulatory membrane-derived oligosaccharides. J Biol Chem. 1988 Mar 15;263(8):3539–3541. [PubMed] [Google Scholar]
- Ielpi L., Dylan T., Ditta G. S., Helinski D. R., Stanfield S. W. The ndvB locus of Rhizobium meliloti encodes a 319-kDa protein involved in the production of beta-(1----2)-glucan. J Biol Chem. 1990 Feb 15;265(5):2843–2851. [PubMed] [Google Scholar]
- Jackson B. J., Kennedy E. P. The biosynthesis of membrane-derived oligosaccharides. A membrane-bound phosphoglycerol transferase. J Biol Chem. 1983 Feb 25;258(4):2394–2398. [PubMed] [Google Scholar]
- Kennedy E. P. Osmotic regulation and the biosynthesis of membrane-derived oligosaccharides in Escherichia coli. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1092–1095. doi: 10.1073/pnas.79.4.1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy E. P., Rumley M. K. Osmotic regulation of biosynthesis of membrane-derived oligosaccharides in Escherichia coli. J Bacteriol. 1988 Jun;170(6):2457–2461. doi: 10.1128/jb.170.6.2457-2461.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacroix J. M., Loubens I., Tempête M., Menichi B., Bohin J. P. The mdoA locus of Escherichia coli consists of an operon under osmotic control. Mol Microbiol. 1991 Jul;5(7):1745–1753. doi: 10.1111/j.1365-2958.1991.tb01924.x. [DOI] [PubMed] [Google Scholar]
- Leigh J. A., Coplin D. L. Exopolysaccharides in plant-bacterial interactions. Annu Rev Microbiol. 1992;46:307–346. doi: 10.1146/annurev.mi.46.100192.001515. [DOI] [PubMed] [Google Scholar]
- Long S. R. Rhizobium-legume nodulation: life together in the underground. Cell. 1989 Jan 27;56(2):203–214. doi: 10.1016/0092-8674(89)90893-3. [DOI] [PubMed] [Google Scholar]
- Lopez-Lara I. M., Orgambide G., Dazzo F. B., Olivares J., Toro N. Characterization and symbiotic importance of acidic extracellular polysaccharides of Rhizobium sp. strain GRH2 isolated from acacia nodules. J Bacteriol. 1993 May;175(10):2826–2832. doi: 10.1128/jb.175.10.2826-2832.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller K. J., Gore R. S., Benesi A. J. Phosphoglycerol substituents present on the cyclic beta-1,2-glucans of Rhizobium meliloti 1021 are derived from phosphatidylglycerol. J Bacteriol. 1988 Oct;170(10):4569–4575. doi: 10.1128/jb.170.10.4569-4575.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller K. J., Gore R. S., Johnson R., Benesi A. J., Reinhold V. N. Cell-associated oligosaccharides of Bradyrhizobium spp. J Bacteriol. 1990 Jan;172(1):136–142. doi: 10.1128/jb.172.1.136-142.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller K. J., Hadley J. A., Gustine D. L. Cyclic [beta]-1,6-1,3-Glucans of Bradyrhizobium japonicum USDA 110 Elicit Isoflavonoid Production in the Soybean (Glycine max) Host. Plant Physiol. 1994 Mar;104(3):917–923. doi: 10.1104/pp.104.3.917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller K. J., Kennedy E. P., Reinhold V. N. Osmotic adaptation by gram-negative bacteria: possible role for periplasmic oligosaccharides. Science. 1986 Jan 3;231(4733):48–51. doi: 10.1126/science.3941890. [DOI] [PubMed] [Google Scholar]
- Miller K. J., Kennedy E. P. Transfer of phosphoethanolamine residues from phosphatidylethanolamine to the membrane-derived oligosaccharides of Escherichia coli. J Bacteriol. 1987 Feb;169(2):682–686. doi: 10.1128/jb.169.2.682-686.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller K. J., Reinhold V. N., Weissborn A. C., Kennedy E. P. Cyclic glucans produced by Agrobacterium tumefaciens are substituted with sn-1-phosphoglycerol residues. Biochim Biophys Acta. 1987 Jul 10;901(1):112–118. doi: 10.1016/0005-2736(87)90262-8. [DOI] [PubMed] [Google Scholar]
- Moreno E., Stackebrandt E., Dorsch M., Wolters J., Busch M., Mayer H. Brucella abortus 16S rRNA and lipid A reveal a phylogenetic relationship with members of the alpha-2 subdivision of the class Proteobacteria. J Bacteriol. 1990 Jul;172(7):3569–3576. doi: 10.1128/jb.172.7.3569-3576.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
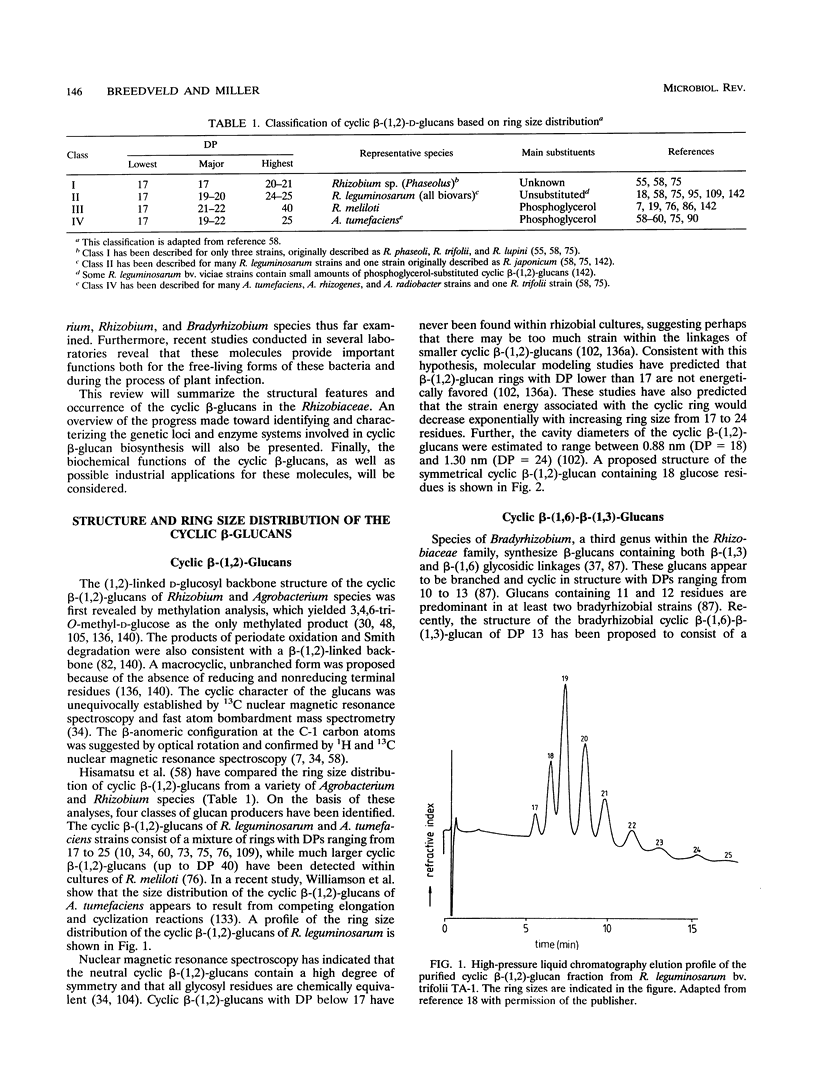
- Nagpal P., Khanuja S. P., Stanfield S. W. Suppression of the ndv mutant phenotype of Rhizobium meliloti by cloned exo genes. Mol Microbiol. 1992 Feb;6(4):479–488. doi: 10.1111/j.1365-2958.1992.tb01492.x. [DOI] [PubMed] [Google Scholar]
- Nap J. P., Bisseling T. Developmental biology of a plant-prokaryote symbiosis: the legume root nodule. Science. 1990 Nov 16;250(4983):948–954. doi: 10.1126/science.250.4983.948. [DOI] [PubMed] [Google Scholar]
- O'Connell K. P., Handelsman J. chvA locus may be involved in export of neutral cyclic beta-1,2-linked D-glucan from Agrobacterium tumefaciens. Mol Plant Microbe Interact. 1989 Jan-Feb;2(1):11–16. [PubMed] [Google Scholar]
- Okada Y., Horiyama S., Koizumi K. [Studies on inclusion complexes of non-steroidal anti-inflammatory agents with cyclosophoraose-A]. Yakugaku Zasshi. 1986 Mar;106(3):240–247. doi: 10.1248/yakushi1947.106.3_240. [DOI] [PubMed] [Google Scholar]
- Park K. G., Heys S. D., Harris C. I., Steele R. J., McNurlan M. A., Eremin O., Garlick P. J. Arginine metabolism in benign and malignant disease of breast and colon: evidence for possible inhibition of tumor-infiltrating macrophages. Nutrition. 1991 May-Jun;7(3):185–188. [PubMed] [Google Scholar]
- Planqué K., Kijne J. W. Binding of pea lectins to a glycan type polysaccharide in the cell walls of Rhizobium leguminosarum. FEBS Lett. 1977 Jan 15;73(1):64–66. doi: 10.1016/0014-5793(77)80016-1. [DOI] [PubMed] [Google Scholar]
- Poppe L., York W. S., van Halbeek H. Measurement of inter-glycosidic 13C-1H coupling constants in a cyclic beta(1-->2)-glucan by 13C-filtered 2D (1H,1H)ROESY. J Biomol NMR. 1993 Jan;3(1):81–89. doi: 10.1007/BF00242477. [DOI] [PubMed] [Google Scholar]
- Puvanesarajah V., Schell F. M., Stacey G., Douglas C. J., Nester E. W. Role for 2-linked-beta-D-glucan in the virulence of Agrobacterium tumefaciens. J Bacteriol. 1985 Oct;164(1):102–106. doi: 10.1128/jb.164.1.102-106.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- REESE E. T., PARRISH F. W., MANDELS M. beta-D-1, 2-Glucanases in fungi. Can J Microbiol. 1961 Jun;7:309–317. doi: 10.1139/m61-038. [DOI] [PubMed] [Google Scholar]
- Rolin D. B., Pfeffer P. E., Osman S. F., Szwergold B. S., Kappler F., Benesi A. J. Structural studies of a phosphocholine substituted beta-(1,3);(1,6) macrocyclic glucan from Bradyrhizobium japonicum USDA 110. Biochim Biophys Acta. 1992 Jun 12;1116(3):215–225. doi: 10.1016/0304-4165(92)90014-l. [DOI] [PubMed] [Google Scholar]
- Rumley M. K., Therisod H., Weissborn A. C., Kennedy E. P. Mechanisms of regulation of the biosynthesis of membrane-derived oligosaccharides in Escherichia coli. J Biol Chem. 1992 Jun 15;267(17):11806–11810. [PubMed] [Google Scholar]
- Sandermann H., Jr, Dekker R. F. Beta-1,2-glucosyl transfer by membrane preparations from Acetobacter xylinum. FEBS Lett. 1979 Nov 1;107(1):237–240. doi: 10.1016/0014-5793(79)80504-9. [DOI] [PubMed] [Google Scholar]
- Schneider J. E., Reinhold V., Rumley M. K., Kennedy E. P. Structural studies of the membrane-derived oligosaccharides of Escherichia coli. J Biol Chem. 1979 Oct 25;254(20):10135–10138. [PubMed] [Google Scholar]
- Schulman H., Kennedy E. P. Localization of membrane-derived oligosaccharides in the outer envelope of Escherichia coli and their occurrence in other Gram-negative bacteria. J Bacteriol. 1979 Jan;137(1):686–688. doi: 10.1128/jb.137.1.686-688.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smit G., Swart S., Lugtenberg B. J., Kijne J. W. Molecular mechanisms of attachment of Rhizobium bacteria to plant roots. Mol Microbiol. 1992 Oct;6(20):2897–2903. doi: 10.1111/j.1365-2958.1992.tb01748.x. [DOI] [PubMed] [Google Scholar]
- Spaink H. P. Rhizobial lipo-oligosaccharides: answers and questions. Plant Mol Biol. 1992 Dec;20(5):977–986. doi: 10.1007/BF00027167. [DOI] [PubMed] [Google Scholar]
- Stanfield S. W., Ielpi L., O'Brochta D., Helinski D. R., Ditta G. S. The ndvA gene product of Rhizobium meliloti is required for beta-(1----2)glucan production and has homology to the ATP-binding export protein HlyB. J Bacteriol. 1988 Aug;170(8):3523–3530. doi: 10.1128/jb.170.8.3523-3530.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stock J. B., Rauch B., Roseman S. Periplasmic space in Salmonella typhimurium and Escherichia coli. J Biol Chem. 1977 Nov 10;252(21):7850–7861. [PubMed] [Google Scholar]
- Streeter J. G. Accumulation of alpha,alpha-trehalose by Rhizobium bacteria and bacteroids. J Bacteriol. 1985 Oct;164(1):78–84. doi: 10.1128/jb.164.1.78-84.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutherland I. W. Biosynthesis and composition of gram-negative bacterial extracellular and wall polysaccharides. Annu Rev Microbiol. 1985;39:243–270. doi: 10.1146/annurev.mi.39.100185.001331. [DOI] [PubMed] [Google Scholar]
- Swart S., Smit G., Lugtenberg B. J., Kijne J. W. Restoration of attachment, virulence and nodulation of Agrobacterium tumefaciens chvB mutants by rhicadhesin. Mol Microbiol. 1993 Nov;10(3):597–605. doi: 10.1111/j.1365-2958.1993.tb00931.x. [DOI] [PubMed] [Google Scholar]
- Thomashow M. F., Karlinsey J. E., Marks J. R., Hurlbert R. E. Identification of a new virulence locus in Agrobacterium tumefaciens that affects polysaccharide composition and plant cell attachment. J Bacteriol. 1987 Jul;169(7):3209–3216. doi: 10.1128/jb.169.7.3209-3216.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tully R. E., Keister D. L., Gross K. C. Fractionation of the beta-Linked Glucans of Bradyrhizobium japonicum and Their Response to Osmotic Potential. Appl Environ Microbiol. 1990 Jun;56(6):1518–1522. doi: 10.1128/aem.56.6.1518-1522.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uttaro A. D., Cangelosi G. A., Geremia R. A., Nester E. W., Ugalde R. A. Biochemical characterization of avirulent exoC mutants of Agrobacterium tumefaciens. J Bacteriol. 1990 Mar;172(3):1640–1646. doi: 10.1128/jb.172.3.1640-1646.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R. C., Seror S. J., Blight M., Pratt J. M., Broome-Smith J. K., Holland I. B. Analysis of the membrane organization of an Escherichia coli protein translocator, HlyB, a member of a large family of prokaryote and eukaryote surface transport proteins. J Mol Biol. 1991 Feb 5;217(3):441–454. doi: 10.1016/0022-2836(91)90748-u. [DOI] [PubMed] [Google Scholar]
- Watanabe H., Igari D., Tanahashi Y., Harada K., Saito M. Measurements of size and weight of prostate by means of transrectal ultrasonotomography. Tohoku J Exp Med. 1974 Nov;114(3):277–285. doi: 10.1620/tjem.114.277. [DOI] [PubMed] [Google Scholar]
- Welsh D. T., Reed R. H., Herbert R. A. The role of trehalose in the osmoadaptation of Escherichia coli NCIB 9484: interaction of trehalose, K+ and glutamate during osmoadaptation in continuous culture. J Gen Microbiol. 1991 Apr;137(4):745–750. doi: 10.1099/00221287-137-4-745. [DOI] [PubMed] [Google Scholar]
- Willems A., Collins M. D. Phylogenetic analysis of rhizobia and agrobacteria based on 16S rRNA gene sequences. Int J Syst Bacteriol. 1993 Apr;43(2):305–313. doi: 10.1099/00207713-43-2-305. [DOI] [PubMed] [Google Scholar]
- Williamson G., Damani K., Devenney P., Faulds C. B., Morris V. J., Stevens B. J. Mechanism of action of cyclic beta-1,2-glucan synthetase from Agrobacterium tumefaciens: competition between cyclization and elongation reactions. J Bacteriol. 1992 Dec;174(24):7941–7947. doi: 10.1128/jb.174.24.7941-7947.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagi M., Yamasato K. Phylogenetic analysis of the family Rhizobiaceae and related bacteria by sequencing of 16S rRNA gene using PCR and DNA sequencer. FEMS Microbiol Lett. 1993 Feb 15;107(1):115–120. doi: 10.1111/j.1574-6968.1993.tb06014.x. [DOI] [PubMed] [Google Scholar]
- York W. S., McNeil M., Darvill A. G., Albersheim P. Beta-2-linked glucans secreted by fast-growing species of Rhizobium. J Bacteriol. 1980 Apr;142(1):243–248. doi: 10.1128/jb.142.1.243-248.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- York W. S., Thomsen J. U., Meyer B. The conformations of cyclic (1-->2)-beta-D-glucans: application of multidimensional clustering analysis to conformational data sets obtained by Metropolis Monte Carlo calculations. Carbohydr Res. 1993 Oct 4;248:55–80. doi: 10.1016/0008-6215(93)84116-n. [DOI] [PubMed] [Google Scholar]
- Zevenhuizen L. P. Cellular glycogen, beta-1,2,-glucan, poly beta-hydroxybutyric acid and extracellular polysaccharides in fast-growing species of Rhizobium. Antonie Van Leeuwenhoek. 1981;47(6):481–497. doi: 10.1007/BF00443236. [DOI] [PubMed] [Google Scholar]
- Zevenhuizen L. P., Scholten-Koerselman H. J. Surface carbohydrates of Rhizobium. I. Beta-1, 2-glucans. Antonie Van Leeuwenhoek. 1979;45(2):165–175. doi: 10.1007/BF00418581. [DOI] [PubMed] [Google Scholar]
- Zevenhuizen L. P., van Veldhuizen A., Fokkens R. H. Re-examination of cellular cyclic beta-1,2-glucans of Rhizobiaceae: distribution of ring sizes and degrees of glycerol-1-phosphate substitution. Antonie Van Leeuwenhoek. 1990 Apr;57(3):173–178. doi: 10.1007/BF00403952. [DOI] [PubMed] [Google Scholar]
- Zorreguieta A., Cavaignac S., Geremia R. A., Ugalde R. A. Osmotic regulation of beta(1-2) glucan synthesis in members of the family Rhizobiaceae. J Bacteriol. 1990 Aug;172(8):4701–4704. doi: 10.1128/jb.172.8.4701-4704.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zorreguieta A., Geremia R. A., Cavaignac S., Cangelosi G. A., Nester E. W., Ugalde R. A. Identification of the product of an Agrobacterium tumefaciens chromosomal virulence gene. Mol Plant Microbe Interact. 1988 Mar;1(3):121–127. doi: 10.1094/mpmi-1-121. [DOI] [PubMed] [Google Scholar]
- Zorreguieta A., Tolmasky M. E., Staneloni R. J. The enzymatic synthesis of beta 1-2 glucans. Arch Biochem Biophys. 1985 May 1;238(2):368–372. doi: 10.1016/0003-9861(85)90176-6. [DOI] [PubMed] [Google Scholar]
- Zorreguieta A., Ugalde R. A. Formation in Rhizobium and Agrobacterium spp. of a 235-kilodalton protein intermediate in beta-D(1-2) glucan synthesis. J Bacteriol. 1986 Sep;167(3):947–951. doi: 10.1128/jb.167.3.947-951.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zorreguieta A., Ugalde R. A., Leloir L. F. An intermediate in cyclic beta 1-2 glucan biosynthesis. Biochem Biophys Res Commun. 1985 Jan 16;126(1):352–357. doi: 10.1016/0006-291x(85)90613-8. [DOI] [PubMed] [Google Scholar]
- de Iannino N. I., Ugalde R. A. Biochemical characterization of avirulent Agrobacterium tumefaciens chvA mutants: synthesis and excretion of beta-(1-2)glucan. J Bacteriol. 1989 May;171(5):2842–2849. doi: 10.1128/jb.171.5.2842-2849.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Iannino N. I., Ugalde R. A. Biosynthesis of cyclic beta-(1-3),beta-(1-6) glucan in Bradyrhizobium spp. Arch Microbiol. 1993;159(1):30–38. doi: 10.1007/BF00244260. [DOI] [PubMed] [Google Scholar]


