Abstract
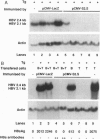
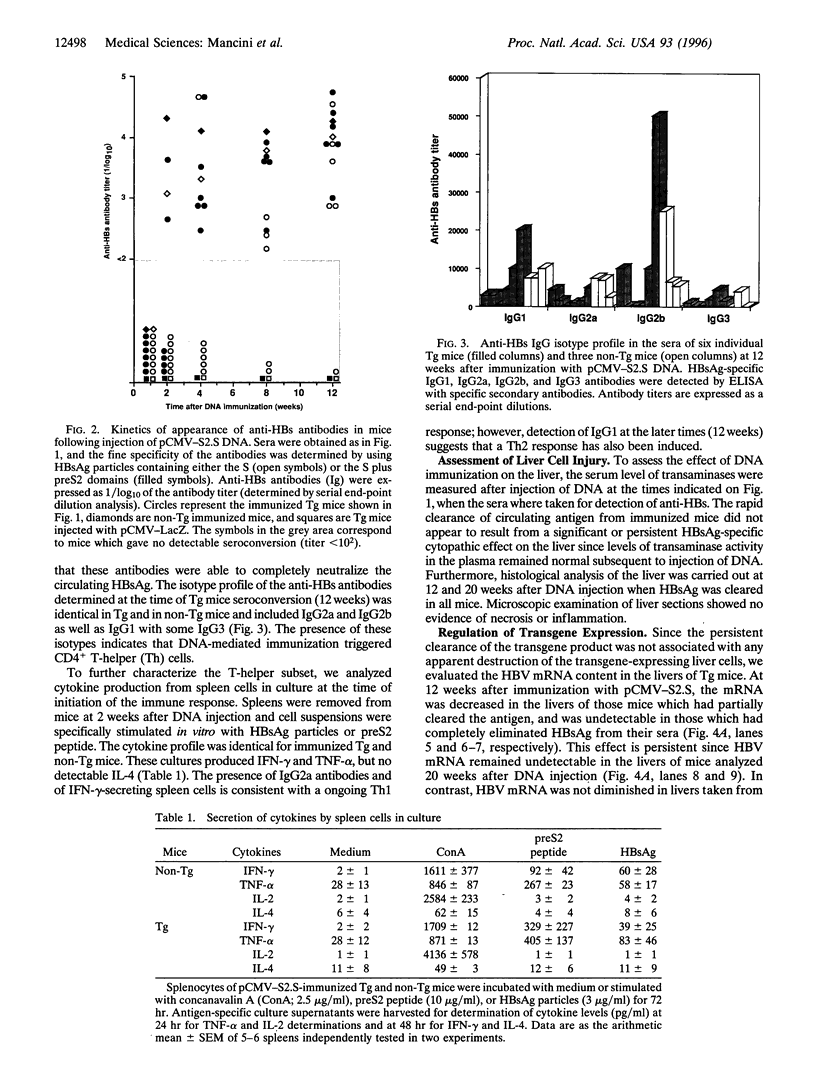
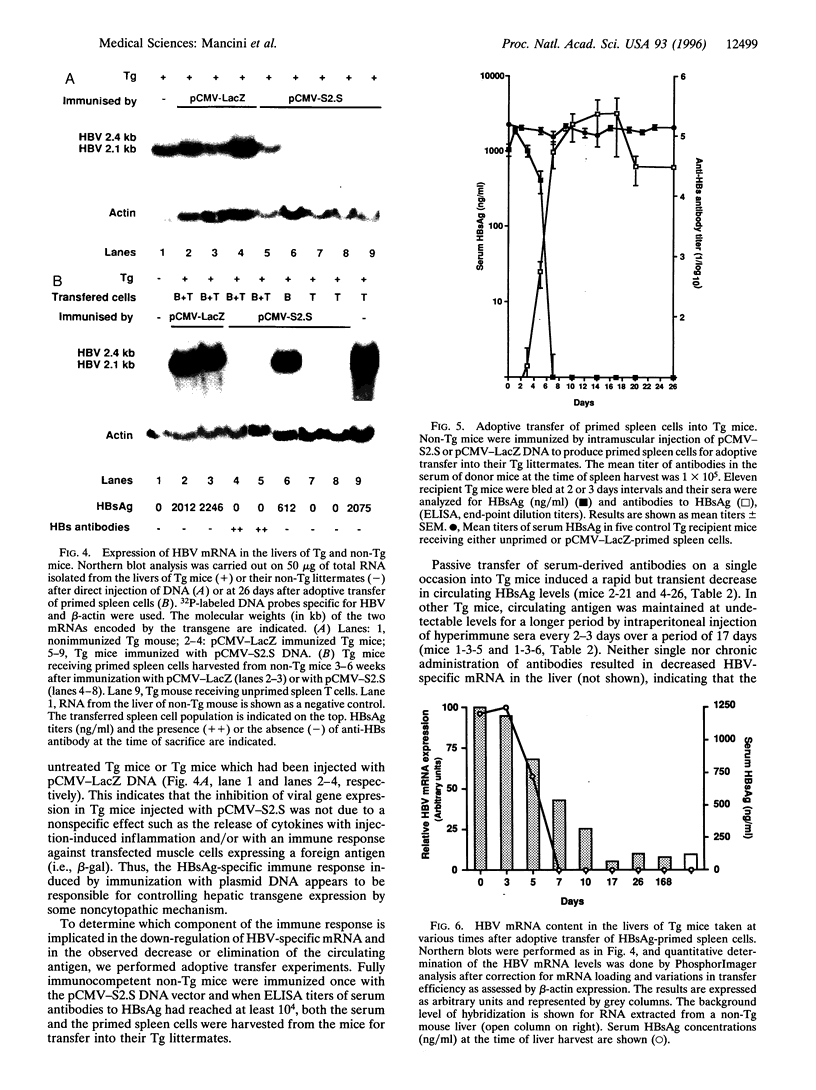
Transgenic mice expressing the sequences coding for the envelope proteins of the hepatitis B virus (HBV) in the liver have been used as a model of the HBV chronic carrier state. We evaluated the possibility of inducing a specific immune response to the viral envelope antigens and thus potentially controlling chronic HBV infection. Using HBV-specific DNA-mediated immunization in this transgenic model, we show that the immune response induced after a single intramuscular injection of DNA resulted in the complete clearance of circulating hepatitis B surface antigen and in the long-term control of transgene expression in hepatocytes. This response does not involve a detectable cytopathic effect in the liver. Adoptive transfer of fractionated primed spleen cells from DNA-immunized mice shows that T cells are responsible for the down-regulation of HBV mRNA in the liver of transgenic mice. To our knowledge, this is the first demonstration of a potential immunotherapeutic application of DNA-mediated immunization against an infectious disease and raises the possibility of designing more effective ways of treating HBV chronic carriers.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Babinet C., Farza H., Morello D., Hadchouel M., Pourcel C. Specific expression of hepatitis B surface antigen (HBsAg) in transgenic mice. Science. 1985 Dec 6;230(4730):1160–1163. doi: 10.1126/science.3865370. [DOI] [PubMed] [Google Scholar]
- Barnaba V., Franco A., Alberti A., Balsano C., Benvenuto R., Balsano F. Recognition of hepatitis B virus envelope proteins by liver-infiltrating T lymphocytes in chronic HBV infection. J Immunol. 1989 Oct 15;143(8):2650–2655. [PubMed] [Google Scholar]
- Beasley R. P. Hepatitis B virus. The major etiology of hepatocellular carcinoma. Cancer. 1988 May 15;61(10):1942–1956. doi: 10.1002/1097-0142(19880515)61:10<1942::aid-cncr2820611003>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- Bertoletti A., Ferrari C., Fiaccadori F., Penna A., Margolskee R., Schlicht H. J., Fowler P., Guilhot S., Chisari F. V. HLA class I-restricted human cytotoxic T cells recognize endogenously synthesized hepatitis B virus nucleocapsid antigen. Proc Natl Acad Sci U S A. 1991 Dec 1;88(23):10445–10449. doi: 10.1073/pnas.88.23.10445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Celis E., Zurawski V. R., Jr, Chang T. W. Regulation of T-cell function by antibodies: enhancement of the response of human T-cell clones to hepatitis B surface antigen by antigen-specific monoclonal antibodies. Proc Natl Acad Sci U S A. 1984 Nov;81(21):6846–6850. doi: 10.1073/pnas.81.21.6846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chisari F. V., Ferrari C. Hepatitis B virus immunopathogenesis. Annu Rev Immunol. 1995;13:29–60. doi: 10.1146/annurev.iy.13.040195.000333. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Demeneix B. A., Quantin B., Coulombe J., Whalen R. G. Plasmid DNA is superior to viral vectors for direct gene transfer into adult mouse skeletal muscle. Hum Gene Ther. 1993 Dec;4(6):733–740. doi: 10.1089/hum.1993.4.6-733. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Mancini M., Michel M. L., Whalen R. G. DNA-mediated immunization to hepatitis B surface antigen: longevity of primary response and effect of boost. Vaccine. 1996 Jun;14(9):910–915. doi: 10.1016/0264-410x(95)00255-y. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Michel M. L., Mancini M., Schleef M., Whalen R. G. Direct gene transfer in skeletal muscle: plasmid DNA-based immunization against the hepatitis B virus surface antigen. Vaccine. 1994 Dec;12(16):1503–1509. doi: 10.1016/0264-410x(94)90073-6. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Michel M. L., Whalen R. G. DNA-based immunization induces continuous secretion of hepatitis B surface antigen and high levels of circulating antibody. Hum Mol Genet. 1993 Nov;2(11):1847–1851. doi: 10.1093/hmg/2.11.1847. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Schirmbeck R., Reimann J., Whalen R. G. DNA-mediated immunization in mice induces a potent MHC class I-restricted cytotoxic T lymphocyte response to the hepatitis B envelope protein. Hum Gene Ther. 1995 Nov;6(11):1447–1456. doi: 10.1089/hum.1995.6.11-1447. [DOI] [PubMed] [Google Scholar]
- Davis H. L., Whalen R. G., Demeneix B. A. Direct gene transfer into skeletal muscle in vivo: factors affecting efficiency of transfer and stability of expression. Hum Gene Ther. 1993 Apr;4(2):151–159. doi: 10.1089/hum.1993.4.2-151. [DOI] [PubMed] [Google Scholar]
- Elson C. J., Barker R. N., Thompson S. J., Williams N. A. Immunologically ignorant autoreactive T cells, epitope spreading and repertoire limitation. Immunol Today. 1995 Feb;16(2):71–76. doi: 10.1016/0167-5699(95)80091-3. [DOI] [PubMed] [Google Scholar]
- Farza H., Salmon A. M., Hadchouel M., Moreau J. L., Babinet C., Tiollais P., Pourcel C. Hepatitis B surface antigen gene expression is regulated by sex steroids and glucocorticoids in transgenic mice. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1187–1191. doi: 10.1073/pnas.84.5.1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrari C., Bertoletti A., Penna A., Cavalli A., Valli A., Missale G., Pilli M., Fowler P., Giuberti T., Chisari F. V. Identification of immunodominant T cell epitopes of the hepatitis B virus nucleocapsid antigen. J Clin Invest. 1991 Jul;88(1):214–222. doi: 10.1172/JCI115280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrari C., Penna A., Bertoletti A., Valli A., Antoni A. D., Giuberti T., Cavalli A., Petit M. A., Fiaccadori F. Cellular immune response to hepatitis B virus-encoded antigens in acute and chronic hepatitis B virus infection. J Immunol. 1990 Nov 15;145(10):3442–3449. [PubMed] [Google Scholar]
- Guidotti L. G., Ando K., Hobbs M. V., Ishikawa T., Runkel L., Schreiber R. D., Chisari F. V. Cytotoxic T lymphocytes inhibit hepatitis B virus gene expression by a noncytolytic mechanism in transgenic mice. Proc Natl Acad Sci U S A. 1994 Apr 26;91(9):3764–3768. doi: 10.1073/pnas.91.9.3764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Julius M. H., Simpson E., Herzenberg L. A. A rapid method for the isolation of functional thymus-derived murine lymphocytes. Eur J Immunol. 1973 Oct;3(10):645–649. doi: 10.1002/eji.1830031011. [DOI] [PubMed] [Google Scholar]
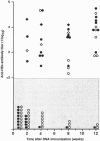
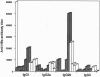
- Mancini M., Hadchouel M., Tiollais P., Pourcel C., Michel M. L. Induction of anti-hepatitis B surface antigen (HBsAg) antibodies in HBsAg producing transgenic mice: a possible way of circumventing "nonresponse" to HBsAg. J Med Virol. 1993 Jan;39(1):67–74. doi: 10.1002/jmv.1890390113. [DOI] [PubMed] [Google Scholar]
- Maruyama T., McLachlan A., Iino S., Koike K., Kurokawa K., Milich D. R. The serology of chronic hepatitis B infection revisited. J Clin Invest. 1993 Jun;91(6):2586–2595. doi: 10.1172/JCI116497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel M. L., Davis H. L., Schleef M., Mancini M., Tiollais P., Whalen R. G. DNA-mediated immunization to the hepatitis B surface antigen in mice: aspects of the humoral response mimic hepatitis B viral infection in humans. Proc Natl Acad Sci U S A. 1995 Jun 6;92(12):5307–5311. doi: 10.1073/pnas.92.12.5307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mor G., Klinman D. M., Shapiro S., Hagiwara E., Sedegah M., Norman J. A., Hoffman S. L., Steinberg A. D. Complexity of the cytokine and antibody response elicited by immunizing mice with Plasmodium yoelii circumsporozoite protein plasmid DNA. J Immunol. 1995 Aug 15;155(4):2039–2046. [PubMed] [Google Scholar]
- Nayersina R., Fowler P., Guilhot S., Missale G., Cerny A., Schlicht H. J., Vitiello A., Chesnut R., Person J. L., Redeker A. G. HLA A2 restricted cytotoxic T lymphocyte responses to multiple hepatitis B surface antigen epitopes during hepatitis B virus infection. J Immunol. 1993 May 15;150(10):4659–4671. [PubMed] [Google Scholar]
- Penna A., Chisari F. V., Bertoletti A., Missale G., Fowler P., Giuberti T., Fiaccadori F., Ferrari C. Cytotoxic T lymphocytes recognize an HLA-A2-restricted epitope within the hepatitis B virus nucleocapsid antigen. J Exp Med. 1991 Dec 1;174(6):1565–1570. doi: 10.1084/jem.174.6.1565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pennisi E. Teetering on the brink of danger. Science. 1996 Mar 22;271(5256):1665–1667. doi: 10.1126/science.271.5256.1665. [DOI] [PubMed] [Google Scholar]
- Pol S., Driss F., Michel M. L., Nalpas B., Berthelot P., Brechot C. Specific vaccine therapy in chronic hepatitis B infection. Lancet. 1994 Jul 30;344(8918):342–342. doi: 10.1016/s0140-6736(94)91384-6. [DOI] [PubMed] [Google Scholar]
- Schirmbeck R., Böhm W., Ando K., Chisari F. V., Reimann J. Nucleic acid vaccination primes hepatitis B virus surface antigen-specific cytotoxic T lymphocytes in nonresponder mice. J Virol. 1995 Oct;69(10):5929–5934. doi: 10.1128/jvi.69.10.5929-5934.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz R. H. A cell culture model for T lymphocyte clonal anergy. Science. 1990 Jun 15;248(4961):1349–1356. doi: 10.1126/science.2113314. [DOI] [PubMed] [Google Scholar]
- Whalen R. G., Davis H. L. DNA-mediated immunization and the energetic immune response to hepatitis B surface antigen. Clin Immunol Immunopathol. 1995 Apr;75(1):1–12. doi: 10.1006/clin.1995.1045. [DOI] [PubMed] [Google Scholar]
- Xiang Z. Q., Spitalnik S., Tran M., Wunner W. H., Cheng J., Ertl H. C. Vaccination with a plasmid vector carrying the rabies virus glycoprotein gene induces protective immunity against rabies virus. Virology. 1994 Feb 15;199(1):132–140. doi: 10.1006/viro.1994.1105. [DOI] [PubMed] [Google Scholar]