Abstract
Purpose
To evaluate the efficacy of saracatinib (AZD0530), an oral Src inhibitor, in colorectal cancer (CRC) and to identify biomarkers that predict antitumor activity.
Experimental Design
Twenty-three CRC cell lines were exposed to saracatinib, and baseline gene expression profiles of three sensitive and eight resistant cell lines in vitro and in vivo were used to predict saracatinib sensitivity in an independent group of 10 human CRC explant tumors using the gene array K-Top Scoring Pairs (K-TSP) method. In addition, fluorescence in situ hybridization (FISH) and immunoblotting determined both Src gene copy number and activation of Src, respectively.
Results
Two of 10 explant tumors were determined to be sensitive to saracatinib. The K-TSP classifier (TOX>GLIS2, TSPAN7>BCAS4, and PARD6G>NXN) achieved 70% (7 of 10) accuracy on the test set. Evaluation of Src gene copy number by FISH showed a trend toward significance (P = 0.066) with respect to an increase in Src gene copy and resistance to saracatinib. Tumors sensitive to saracatinib showed an increase in the activation of Src and FAK when compared with resistant tumors.
Conclusions
Saracatinib significantly decreased tumor growth in a subset of CRC cell lines and explants. A K-TSP classifier (TOX>GLIS2, TSPAN7>BCAS4, and PARD6G>NXN) was predictive for sensitivity to saracatinib. In addition, increased activation of the Src pathway was associated with sensitivity to saracatinib. These results suggest that FISH, a K-TSP classifier, and activation of the Src pathway have potential in identifying CRC patients that would potentially benefit from treatment with saracatinib.
Src is a pleiotropic nonreceptor tyrosine kinase that plays a major role in the development and progression of many human cancers (1–4). It has been well established that one of the main functions of Src is regulating cellular proliferation through its interaction with many different growth factor receptors such as the epidermal growth factor receptor (EGFR), fibroblast growth factor, hepatocyte growth factor receptor, and insulin growth factor receptor (5–8). In addition to proliferation, elevated levels and activation of Src lead to the disruption of focal adhesions and adherens junctions resulting in an increase in motility and invasiveness of cancer cells (2, 9, 10). It is believed that Src affects growth during earlier stages of tumorigenesis, but alters other processes such as adhesion, invasion, and motility during later stages of tumor progression.
Many studies on Src have revealed the complex molecular mechanisms and interactions with many different substrates that are important in altering tumor cell behavior. In particular, Src binds and activates focal adhesion kinase (FAK), a kinase that facilitates changes in the cytoskeleton by disrupting cell-cell and cell-matrix interactions (9, 11). Both Src and FAK have been reported to be simultaneously increased in many human malignancies (12). In addition, Src activates Stat-3, resulting in a subsequent increase in the transcriptional activation of vascular endothelial growth factor, an important regulator of tumor angiogenesis (13, 14). Recently, Src has been shown to activate Trask in many human epithelial cancers and is believed to be activated during detachment and shedding of tumor cells (15). Several selective Src kinase inhibitors have been developed and have shown potential in inhibiting the growth of tumor cells as well as altering the metastatic phenotype evidence by a reduction in migration and invasiveness of cancer cells. A study by Huang et al. (16) tested dasatinib (Src/Abl inhibitor) against 23 breast cancer cell lines, and a genomic signature was developed that predicted dasatinib susceptibility. The 161-gene predictor was refined to a 6-gene predictor consisting of ANXA1, CAV1, CAV2, EPHA2, PTRF, and IGFBP2, and this was associated with a “triple-negative” pattern of estrogen receptor (ER), progesterone receptor (PR), and human EGFR 2 breast cancer when tested against archival breast cancer specimens.
The Src gene is located on chromosome 20q12–q13. It is widely accepted in the field that the Src gene is not amplified. However, a review of the Sanger database (http://www.sanger.ac.uk/) does suggest that there may be an increase in Src gene copy in some colorectal cancer (CRC) cell lines.
Previously, a human pancreas cancer explant model was developed whereby excess nondiagnostic tissue is obtained from the operating room and then placed directly into mice (17). The advantage of this model is that there is a continuous supply of human tumor tissue to develop and to validate novel correlative studies with new drugs and drug combinations. Most importantly, the tumor cells are not placed in culture and appear to retain most of the genetic features of the original tumor (17). This model has been used to identify predictive biomarkers of sensitivity to two different Src inhibitors in pancreatic cancer (18, 19). We have applied the same concept in CRC to identify predictive biomarkers of sensitivity and resistance to saracatinib.
CRC is one of the most common malignancies in Western countries, with an annual incidence of ~300,000 in the United States and Europe (20). About 50% of patients with locally advanced or metastatic disease will ultimately die of their disease. There is strong evidence that aberrant Src kinase activity is a critical component in all stages of CRC (21). The Src pathway is activated in 80% of all human colon tumors, and its activation has been suggested to play a central role in the induction and progression of tumors, especially in tumors with high malignant potential (22). When compared with the normal mucosa of the colon, Src activity is elevated in adenomatous polyps (23). In addition, Talamonti et al. (24) showed Src activity to be the greatest in liver metastatic lesions showing a role of Src in CRC progression. Finally, Src is an independent prognostic indicator at all stages of CRC (25). Taken together, increased levels of Src expression and activation have been shown to play a very important role in the sequential progression from premalignant lesions of the colon to metastasis.
There have been numerous developments in the CRC field over the past year, the most important being studies showing that patients with codon 12/13 KRAS mutations do not benefit from the EGFR inhibiting antibodies, cetuximab and panitumumab (26, 27). In addition, dual use of EGFR- and vascular endothelial growth factor–targeting antibodies has been found to harm patients (27, 28). These findings show that it is extremely important to have an understanding of the specific genetic/signaling defects of a particular tumor. To this end and in conjunction with the observations that Src may potentially be a significant contributor in CRC, we set out to identify possible mechanisms and potential predictive biomarkers of sensitivity and resistance to saracatinib (a Src kinase inhibitor currently in phase II clinical development). These studies show that saracatinib has activity in a CRC subset, which may be identified by the use of fluorescence in situ hybridization (FISH), a K-Top Scoring Pairs (K-TSP) classifier, and Src activation status.
Materials and Methods
Cell lines and culture
Human CRC cell lines were obtained from the American Type Culture Collection. Cells were cultured in RPMI supplemented with 10% fetal bovine serum, 1% nonessential amino acids, and 1% penicillin/streptomycin, and were maintained at 37°C under an atmosphere containing 5% CO2. The cells were routinely screened for the presence of Mycoplasma (MycoAlert; Cambrex BioScience) and were exposed to saracatinib (Astra Zeneca) when they reached ~70% confluence. All cell lines were tested and authenticated in the University of Colorado Cancer Center DNA Sequencing and Analysis Core. CRC cell line DNA was tested using the Profiler Plus kit (Applied Biosystems). The data obtained were compared with American Type Culture Collection data to ensure the cell lines have not changed. The cell lines were last tested in September 2009.
Evaluation of cytotoxicity
Cytotoxic effects were determined using the sulforhodamine B assay (13). Briefly, cells in logarithmic growth phase were transferred to 96-well flat-bottomed plates with lids. Cell suspensions (100 μL) containing 500 to 3,000 viable cells were plated into each well and incubated overnight before exposure to saracatinib for 72 hours. After drug treatment, medium was removed and cells were fixed with cold 10% TCA for 30 minutes at 4°C. Cells were then washed with water and stained with 0.4% sulforhodamine B (Fisher Scientific) for 30 minutes at room temperature. The plates were washed with 1% acetic acid followed by stain solubilization with 10 mmol/L Tris. The plate was then read on a plate reader (Biotek Synergy 2) set at an absorbance wavelength of 565 nm. Cell proliferation curves were derived from the raw absorbance data.
CRC cell line xenografts
SW620, SW480, LS174T, and LS180 were injected into the left and right flanks of 4- to 6-week-old female athymic (nu+/nu+) mice (Harlan Laboratories). Mice were randomized into the treatment group (saracatinib) or vehicle group when tumor volumes reached ~200 mm3. Mice were treated daily with saracatinib 50 mg/kg or vehicle by oral gavage. Mice were monitored daily for signs of toxicity, and tumor size was evaluated twice per week by caliper measurements using the following formula: tumor volume = (length × width2) × 0.52.
Flow cytometric analysis of cell cycle distribution
Cells (2 × 105 per well) were seeded in six-well plates and allowed to attach for 24 hours. The cells were then treated with saracatinib (1 μmol/L) for 24 and 48 hours, washed in PBS, and resuspended in Krishanaposs stain. The cells were stained for 24 hours at 4°C before analysis by flow cytometry at the University of Colorado Cancer Center Flow Cytometry Core Facility.
Induction of apoptosis
Cells (1,000/well) were plated in 96-well, white-walled plates and allowed to attach for 24 hours. The cells were then treated with 1 μmol/L saracatinib and apoptosis was determined by the measurement of caspase-3 and caspase-7 activity using a luminometric Caspase-Glo-3/7 assay (Promega) according to the manufacturer’s protocol and read using a 96-well plate reader (Biotek).
Sequencing of Src, KRAS, and BRAF
We performed sequencing to examine the mutational status of Src, KRAS, and BRAF in CRC cell lines and explants. DNA was isolated using the Qiagen DNA extraction kit (Qiagen). KRAS mutations were analyzed by the University of Colorado Cancer Center Pathology Core using a high-resolution melting temperature method with custom primers and the Roche LC480 real-time PCR machine. Briefly, template DNA was tested by high-resolution melting analysis using a Lightcycler 480 real-time PCR instrument (Roche Applied Science). Approximately 60 ng of template DNA, wild-type (WT) control DNA, and mutant control DNA were amplified on the Lightcycler 480 instrument using a high-resolution melting master mix (Roche Diagnostics), with the RASO1 and RASA2 primers and 1.75 mmol/L MgCl2 in 10 μL on a 96-well plate, using a two-step cycling program (95° melting, 72° annealing and extension) for 45 cycles. PCR products were then analyzed by high-resolution melting with 25 data acquisitions per degree of temperature increase, from 40°C to 90°C. The Lightcycler 480 Gene Scanning software using the known WT control samples for baseline calculation were used for these analyses. Src and BRAF mutations were analyzed by PCR amplification and direct sequencing of the products as previously described (29, 30).
Development of Src probe for FISH
A Src FISH probe was developed to evaluate Src gene copy number. The PAC clone RP4-681F2 (http://www.ensembl.org), which encompasses the SRC gene was selected for this study. The clone was obtained from CHORI and streaked on agar plates. Single-cell colonies were selected, and the PAC clone was validated using PCR (forward, 5′-AGTGTCTGACTTCGACAACGCCAA-3′; reverse, 5′-GAAGGAGACAAGGAAAGGAGGGAA-3′). After validation, the clone was expanded and purified using the Qiagen QIAamp DNA Mini kit. Repli-G kit (Qiagen) was used as per manufacturer’s instructions for whole genome amplification. The DNA inserts were then labeled by nick translation (Vysis Nick Translation kit) with SpectrumRed-conjugated dUTPs. Labeled DNA was ethanol precipitated with herring sperm and human Cot-1, and each DNA pellet was resuspended in 20 μL of c-DenHyb (Insitus Bio-technologies). The SRC probe was combined with the Aquarius LPE 20G probe (Cytocell) as a control for the centromere on chromosome 20.
Evaluation of Src gene copy in CRC cell lines and explants
Slides were incubated at 56°C for 4 hours, soaked in CitriSolv, and washed thrice for 10 minutes each. After drying, the slides were incubated in 2xSSC at 75°C for 20 minutes and then in 0.25 mg/mL proteinase K at 45°C for 20 to 25 minutes. After washing and dehydration, the specimens were sequentially placed in 70% ethanol, 85% ethanol, and 100% ethanol for 2 minutes, and allowed to air dry. The appropriate volume of the SRC/CEP20 probe was applied followed by DNA denaturation (85°C for 15 min) and hybridization (37°C for 36–48 h). Posthybridization washes were done in 2xSSC/0.3% NP40 at 74°C for 2 minutes and then in 2xSSC for 2 minutes each. After dehydration, 14 μL of 4′,6-diamidino-2-phenylindole/anti-fade (0.3 μg/mL in Vectashield mounting medium) was added to the slide. Analysis was done on an epifluorescence microscope using single interference filter sets for green (FITC), red (Texas red), and blue (4′,6-diamidino-2-phenylindole) as well as dual (red/green) and triple (blue, red, green) band pass filters. At least 20 metaphase spreads (cell lines) and 100 interphase nuclei (cell lines and explants) were analyzed for Src gene copy number and CEP 20 signals. Continuous variables for Src gene copy number were categorized, and a conservative approach was used for cell lines and explants in determining a gain in Src gene copy number. A Src copy number of ≥3 was considered a gain, and <3 was considered no gain.
CRC explant xenograft model
Patient-derived colorectal adenocarcinoma tumor specimens were obtained from consenting patients at the University of Colorado Hospital in accordance with protocols approved by the Colorado Multiple Institutional Review Board (COMIRB # 07-0570). Tumor material not required for histopathologic analysis was collected and placed in medium consisting of RPMI supplemented with 10 μmol/L HEPES, 4.5 g/L glucose, 1 μmol/L pyruvate sodium, 200 U/mL penicillin, and 200 μg/mL streptomycin. After washing several times in media, tumors were cut into 2- to 3-mm3 pieces in antibiotic-containing medium and coated in Matrigel. Four to 6-wk-old female athymic (nu+/nu+) mice were obtained from Harlan Laboratories under an approved research protocol by the Institutional Animal Care and Use Committee [IACUC # 51402007(09) 2E]. The tumor pieces were then implanted in mice, and expansion of the F1–F3 generations was carried out as previously described (17, 31). Both mutational analysis of common genes mutated in CRC (Ras, Raf, PI3K, PTEN, and Src) as well as gene expression patterns were evaluated in F0 and F3 generations to ensure genetic stability of tumors in subsequent generations. Tumors were expanded in the left and right flanks of five to six mice (10 evaluable tumors per group). Mice were randomized into the treatment group (saracatinib) or vehicle group when tumor volumes reached ~200 mm3. Mice were treated daily with saracatinib 50 mg/kg or vehicle by oral gavage for 28 days. Mice were monitored daily for signs of toxicity, and tumor size was evaluated twice per week by caliper measurements using the following formula: tumor volume = (length × width2) × 0.52.
Determination of sensitive and resistant molecular biomarkers
Total RNA (RNeasy kit) from all CRC explants and cell lines were profiled using the Affymetrix Human Gene 1.0 ST Array. This gene array has ~764,885 probes comprising ~28,869 genes. Sample preparation and processing procedure were done as described in the Affymetrix GeneChip Expression Analysis Manual (Affymetrix, Inc.). The gene expression levels were converted to a rank-based matrix and standardized (mean, 0; SD, 1) for each microarray. Using this preprocessing method, we can cluster the same explants from different data sets based on their gene expression profiles. Data analyses were done on this rank-based matrix.
Differentially expressed genes analysis
We used two different methods to identify the differentially expressed genes in this study. When comparing two phenotypes (e.g., saracatinib sensitive versus resistant), first, we used t statistics to compute the P values for each gene and only call differentially expressed genes when P < 0.0001 (DEG list 1). We also performed Statistically Analysis of Microarray (32) with stringent thresholds (fold change, >1.5; false discovery rate, <0.0001) to determine the differentially expressed genes (DEG list 2). Only genes found to be common in both lists were considered differentially expressed genes.
K-TSP classifier
We used the K-TSP algorithm (33) to construct a discriminative classifier in predicting tumors sensitive to saracatinib. In brief, the algorithm exploits the information contained in the rank-based matrix by focusing on “marker gene pairs” (i, j) for which there is a significant difference in the probability of the event (Ri < Rj) across the N samples from class Y = 1 (saracatinib sensitive) to Y = −1 (saracatinib resistant), in which the event (Ri < Rj) is equivalent to the rank of gene i is less than the rank of gene j if and only if gene i is expressed less then gene j (relative expression). Here, the quantities of interest are pij(m) = Prob(Ri < Rj | Y = m), m = (1, −1), i.e., the probabilities of observing Ri < Rj in each class. These probabilities were estimated by the relative frequencies of occurrences of Ri < Rj within profiles and over samples. Let Δij denote the “score” of gene pair (i, j), in which Δij = | pij(1) − pij(−1)|. A score Δij is computed for every pair of genes i, j ∈{1,…,P}, i ≠ j. Gene pairs with high scores were viewed as most informative for classification. Using an internal leave-one-out cross-validation, the final K-TSP classifier uses the k disjoint pairs of genes, which achieve the k best scores from the training set. In this study, maximum number of pairs (kmax) was fixed as 10.
Immunoblotting
Cells were seeded into six-well plates 24 hours before treatment with 500 nmol/L saracatinib. After treatment, 100 μL of 1× prechilled cell lysis buffer + 1 mmol/L phenylmethylsulfonyl fluoride (Cell Signaling Technology) were added to the well, and cells were scraped and placed in a microfuge tube. Tumor tissues (50–75 mg/mouse) were minced on ice and homogenized using a Dounce homogenizer, and centrifuged at 16,000 g at 4°C for 10 minutes. The total protein in samples was determined using the Bio-Rad Dc Protein Assay. Forty micrograms of sample were electrophoresed on 4% to 12% Bis-Tris pre-cast gels (Bio-Rad Laboratories, Inc.). After electrotransfer to Immobilon-P membranes (Millipore), membranes were blocked at room temperature with TBS [10 mmol/L Tris-HCl (pH 7.5), 0.5 mol/L NaCl, and 0.1% (v/v) Tween 20] containing 5% nonfat milk (Bio-Rad) for 1 hour. Primary antibodies for Src, p-Src family (Tyr416), FAK, Stat-3, p-Stat-3 (Tyr705) and Actin (Cell Signaling Technology), and p-FAK Tyr861 (BioSource International) were diluted at 1:1,000 in TBST containing 5% protease-free bovine serum albumin (Sigma-Aldrich), and the membranes were incubated with primary antibodies overnight at 4°C with rocking. After washing thrice with TBST, the membranes were incubated for 1 h at room temperature with anti-rabbit/mouse IgG horseradish peroxidase–conjugated antibody at a final dilution of 1:50,000 in TBST. After washing thrice with TBST, bound antibodies were detected by enhanced chemiluminescence (Millipore). Immunoblot experiments were done in triplicate for each antibody.
Immunohistochemistry
Tumor tissues from control and saracatinib-treated mice were fixed in formalin immediately after surgical excision. Sections were deparaffinized using standard histologic procedures to characterize a suite of immunohistochemical biomarkers across several biological effect areas. Immunohistochemical staining was scored by eye (from a minimum of 1,000 cells for the % markers) by pathology concordance panel (blinded).
Results
Saracatinib inhibits growth in a subset of colorectal cell lines in vitro and in vivo
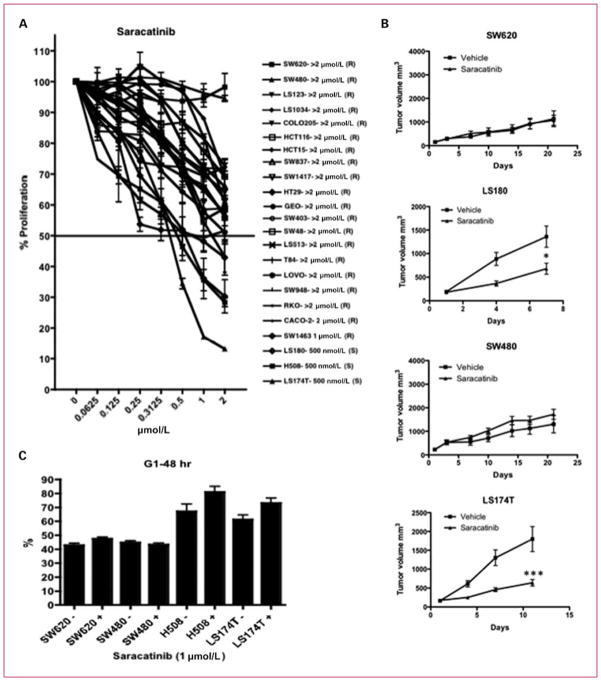
We tested the antiproliferative effects of saracatinib (dose range, 0.0625–2 μmol/L) on 23 CRC cell lines using an SRB assay. We considered an IC50 of <1 μmol/L as sensitive to saracatinib. As shown in Fig. 1A, LS180, H508, and LS174T were sensitive to saracatinib with an IC50 of 0.5 μmol/L. All other cell lines evaluated were shown to be resistant to saracatinib. To confirm the in vitro responses to saracatinib, we treated two of the most resistant cell lines (SW620 and SW480) and two sensitive cell lines (LS180 and LS174T) with saracatinib (50 mg/kg/d) in vivo. The two resistant cell lines proved resistant to saracatinib, whereas the two sensitive cell lines were shown to be sensitive to saracatinib in the xenograft model (Fig. 1B). This shows that the in vitro responses to saracatinib were the same in vivo with these cell lines.
Fig. 1.
The effects of saracatinib on proliferation of CRC cell lines. A, 23 CRC cell lines were treated with saracatinib (0.0625 μmol/L → 2 μmol/L) and assayed by SRB 72 h after exposure to drug. Cell lines (H508, LS180, and LS174T) with an IC50 of <1 μmol/L were considered sensitive to saracatinib. Cell lines are listed in order from the most resistant to the most sensitive; (R), resistant; (S), sensitive. B, two sensitive and two resistant cell lines (identified from the SRB assay) were injected in a xenograft model to confirm the antiproliferative effects of saracatinib in vivo. When tumor volumes reached ~200 mm3, mice were randomized and treated with saracatinib 50 mg/kg/d by oral gavage for 7 to 28 d. Tumor size was evaluated twice per week by caliper measurements using the formula: tumor volume = (length × width2) × 0.52. Treatment with saracatinib resulted in a significant decrease in growth in the LS180 (*, P < 0.05) and LS174T (***, P < 0.001). C, cell cycle was analyzed in two sensitive (H508 and LS174T) and two resistant (SW620 and SW480) cell lines by flow cytometry. Treatment with saracatinib resulted in a G1 cell cycle arrest (*, P < 0.05) in the sensitive cell lines. There were no effects on cell cycle in the resistant cell lines.
To investigate the mechanisms of inhibition of cellular proliferation, the two resistant cell lines (SW620 and SW480) and two sensitive cell lines (H508 and LS174T) were treated with 1 μmol/L of saracatinib for 48 hours, and cell cycle progression was determined by flow cytometry. H508 and LS174T cell lines treated with saracatinib showed an increase in G1 cell cycle arrest at 48 hours (Fig. 1C). In contrast, the resistant cell lines (SW620 and SW480) did not show any differences in cell cycle arrest after treatment. In addition, both resistant and sensitive cell lines did not show any increases in apoptosis evaluated by a caspase-3/7 assay (data not shown). These results suggest that the antiproliferative effects of saracatinib on sensitive cell lines are through cell cycle arrest and not apoptosis.
Heterogeneity in Src gene copy in CRC cell lines and explants
We developed a new FISH assay to assess Src gene copy number. Table 1 shows that a gain in Src gene copy number was common among CRC cell lines and explants. In addition, the use of a probe for centromere 20 shows that the gain in Src gene copy number is mainly from an increase in ploidy of chromosome 20, showing that the Src gene was not amplified. We next examined the relationship between Src gene copy and sensitivity or resistance to saracatinib in CRC cell lines and explants, and showed a (Fisher’s exact) trend toward significance (P = 0.066) with respect to an increase in Src gene copy number and resistance to saracatinib (Table 1). A Src gene copy number of ≥3 was considered a gain, and <3 was considered no gain. A representative figure shows that CRC007, a sensitive tumor, had normal Src gene copy number, whereas CRC027, a resistant tumor, has an increase in Src gene copy (Fig. 2A and B).
Table 1.
Src mutational status and Src gene copy (FISH) in CRC cell lines and explants
| Specimen ID | Saracatinib effects | Src (531) | SRC
|
Centromere 20
|
SRC/centromere 20 | SRC copy number gain | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||||||
| Cell lines | |||||||||
| SW620 | Resistant | WT | 3.23 | 0.85 | 3.03 | 0.39 | 1.07 | Yes | |
| SW480 | Resistant | WT | 4.40 | 1.21 | 4.40 | 1.21 | 1.00 | Yes | |
| LS123 | Resistant | WT | 4.09 | 0.70 | 4.04 | 0.74 | 1.01 | Yes | |
| LS1034 | Resistant | WT | 4.93 | 0.56 | 4.94 | 0.58 | 1.00 | Yes | |
| COLO205 | Resistant | WT | 5.10 | 1.38 | 4.95 | 0.96 | 1.03 | Yes | |
| HCT116 | Resistant | WT | 2.02 | 0.38 | 2.08 | 0.51 | 0.97 | No | |
| HCT15 | Resistant | WT | 2.10 | 0.48 | 2.02 | 0.48 | 1.04 | No | |
| SW837 | Resistant | WT | 2.99 | 0.98 | 1.64 | 0.69 | 1.82 | No | |
| SW1417 | Resistant | WT | 6.94 | 1.33 | 4.93 | 0.90 | 1.41 | Yes | |
| HT29 | Resistant | WT | 4.99 | 0.61 | 4.19 | 0.63 | 1.19 | Yes | |
| GEO | Resistant | WT | 4.00 | 0.64 | 2.95 | 0.56 | 1.36 | Yes | |
| SW403 | Resistant | WT | 4.76 | 0.65 | 4.76 | 0.65 | 1.00 | Yes | |
| SW48 | Resistant | WT | 2.02 | 0.14 | 2.00 | 0.14 | 1.01 | No | |
| LS513 | Resistant | WT | 2.00 | 0.25 | 2.02 | 0.20 | 0.99 | No | |
| T84 | Resistant | WT | 3.52 | 0.75 | 3.56 | 0.76 | 1.0 | Yes | |
| LoVo | Resistant | WT | 2.10 | 0.44 | 2.66 | 0.59 | 0.79 | No | |
| SW948 | Resistant | WT | 4.30 | 0.63 | 4.25 | 0.61 | 1.01 | Yes | |
| RKO | Resistant | WT | 3.40 | 1.03 | 3.22 | 0.97 | 1.06 | Yes | |
| CaCo2 | Resistant | WT | 7.76 | 1.75 | 7.56 | 1.70 | 1.03 | Yes | |
| SW1463 | Resistant | WT | 5.91 | 0.59 | 4.83 | 0.47 | 1.22 | Yes | |
| LS180 | Sensitive | WT | 1.99 | 0.27 | 1.98 | 0.32 | 1.01 | No | |
| NCI-H508 | Sensitive | WT | 6.19 | 1.02 | 3.91 | 0.90 | 1.58 | Yes | |
| LS174T | Sensitive | WT | 2.10 | 0.46 | 2.20 | 0.51 | 0.95 | No | |
| Explants | |||||||||
| CRC-001 | Resistant | WT | 2.24 | 1.02 | 2.12 | 0.8 | 1.06 | No | |
| CRC-006 | Resistant | WT | 2.44 | 0.97 | 2.24 | 0.8 | 1.09 | No | |
| CRC-007 | Sensitive | WT | 1.84 | 0.65 | 1.86 | 0.64 | 0.99 | No | |
| CRC-010 | Resistant | WT | 3.14 | 1.16 | 2.78 | 0.89 | 1.13 | Yes | |
| CRC-012 | Resistant | WT | 4.12 | 3.04 | 2.38 | 2.02 | 1.73 | Yes | |
| CRC-021 | Resistant | WT | 2.04 | 0.83 | 1.42 | 0.5 | 1.44 | No | |
| CRC-026 | Resistant | WT | 3.38 | 1.1 | 3.04 | 1.34 | 1.11 | Yes | |
| CRC-027 | Resistant | WT | 5.3 | 1.56 | 5.66 | 2.26 | 0.94 | Yes | |
| CRC-034 | Resistant | WT | 3.98 | 1.39 | 2.96 | 1.26 | 1.34 | Yes | |
| CRC-040 | Sensitive | WT | 2.44 | 0.86 | 2.4 | 0.83 | 1.02 | No | |
NOTE: A Src copy number of ≥3 was considered a gain, and <3 was considered no gain.
Fig. 2.
Evaluation of Src gene copy number by FISH. A, representative graphs of interphase nuclei showing normal copies (~2 genes per cell) of the Src gene (CRC007). B, gain (~5.3 gene copies per cell) in Src gene copy (CRC027).
Mutational analysis of Src
It has been suggested that Src (codon 531) has an activating mutation in 12% of patients with advanced CRC (29), although this finding has not been confirmed in other laboratories. Evaluation of this mutation in our CRC cell lines and explants did not reveal any mutations (Table 1).
Identification of sensitive and resistant biomarkers by K-TSP scoring in CRC
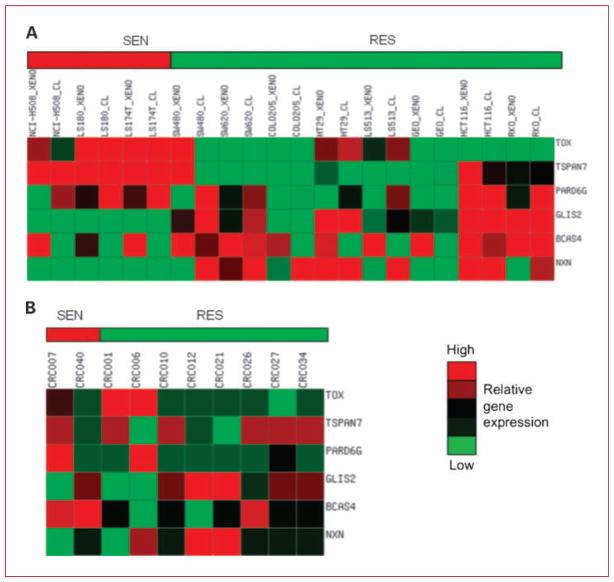
We next sought to develop a K-TSP gene classifier for predicting saracatinib sensitivity in CRC. Baseline gene expression profiles of three sensitive cell lines and a subset of 8 of 20 resistant cell lines in vitro (cell lines cultured) and in vivo (cell lines in xenograft model) were used as the training set. We used both in vitro and in vivo baseline gene array for a given cell line in the training set so that differences relating purely to the tumor microenvironment would be minimized. The K-TSP algorithm determined three gene pairs from the training set with an estimated leave-one-out cross-validation of 91% (95% confidence interval, 86.6–96.2%), 92% (95% confidence interval, 83.3–100%), and 91% (95% confidence interval, 85.3–97.3%) for accuracy, sensitivity, and specificity, respectively (34). The three gene pairs are thymocyte selection-associated high mobility group box (TOX), GLIS family zinc finger 2 (GLIS2), tetraspanin 7 (TSPAN7), breast carcinoma amplified sequence 4, (BCAS4), par-6 partitioning defective 6 homologue γ (PARD6G), and nucleoredoxin (NXN). The interpretation for the top gene pair is as follows: If the expression of TOX is higher than GLIS2, then predict sensitive to saracatinib, or else predict resistant to saracatinib. Similar interpretation applies to the second and third gene pairs. Figure 3A depicted the heat map of the K-TSP gene pair classifier.
Fig. 3.
Heat map of the K-TSP gene pairs for predicting saracatinib sensitivity. A, relative gene expression profiles in the training set of the three gene pairs in (three sensitive and eight resistant) CRC cell lines in vitro and in vivo. The three gene pairs that predict sensitivity to saracatinib include TOX > GLIS2, TSPAN7 > BCAS4, and PARD6G > NXN. High (red) and low (green) gene expression. B, heat map of expression in the validation set of the three gene pairs. CRC006 and CRC007 were predicted to be sensitive.
K-TSP achieves high accuracy in predicting saracatinib sensitivity in CRC explants
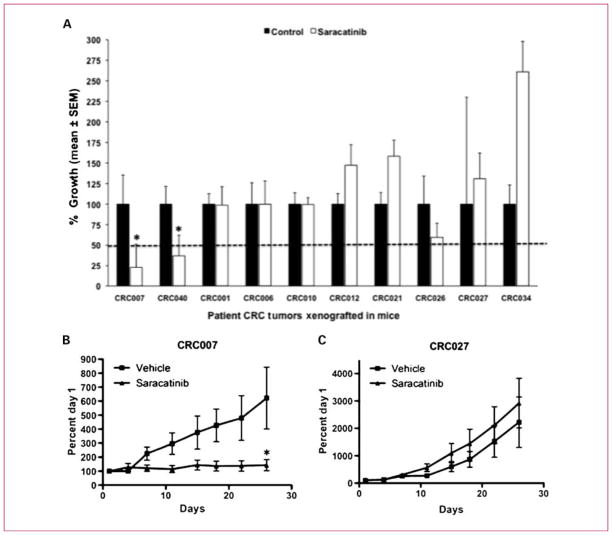
We next tested the predictive power of the K-TSP classifier from the CRC cell line training set in 10 patient-derived human CRC explants. Before treatment, baseline gene array was analyzed for the six markers. Patients CRC007, CRC001, and CRC006 (TOX>GLIS2, TSPAN7>BCAS4, and PARD6G>NXN) were predicted to be sensitive, and patients CRC010, CRC012, CRC021, CRC026, CRC027, CRC034, and CRC040 were predicted to be resistant (Fig. 3B). The 10 cases were then treated with saracatinib (50 mg/kg/d, oral gavage), and tumor growth inhibition (TGI) was evaluated 28 days after treatment. Recognizing that the explant model has a limited dynamic range, in this validation set, we considered a TGI of ≤50% as sensitive and a TGI of >50% as resistant. CRC007 (TGI of 23%) and CRC040 (TGI of 37%) were sensitive, whereas the other eight explants were resistant to saracatinib (Fig. 4). The gene classifier predicts 7 of 10 correct (70% accuracy), yielding 50% sensitivity (correctly predicting 1 of 2 in the sensitive cases) and 75% specificity (correctly predicting 6 of 8 in the resistant cases).
Fig. 4.
Three gene pairs identified by K-TSP predicts saracatinib sensitivity in an independent test set. A, prospective validation of efficacy in 10 xenografts predicted by the K-TSP classifier. The 10 xenografts predicted for saracatinib sensitivity were treated with saracatinib 50 mg/kg/d by oral gavage for 28 d. Tumor size was evaluated twice per week by caliper measurements using the formula: tumor volume = (length × width2) × 0.52. TGI was calculated by relative tumor growth of treated mice divided by relative tumor growth of control mice × 100. Cases with a TGI of <50% were considered sensitive, and TGI of >50% were considered resistant to saracatinib. Two xenografts (CRC006 and CRC040) were sensitive to saracatinib (TGI ≤ 50%), and eight xenografts were resistant to saracatinib (TGI > 50%). CRC001, CRC006 (predicted sensitive), and CRC040 (predicted resistant) by the K-TSP classifier were misclassified. Columns, mean (n = 8-0 tumors per group); bars, SEM. *, significance (P < 0.05) compared with vehicle-treated tumors. B, representative graph of a sensitive tumor (CRC007) and (C) resistant tumor (CRC027) treated with saracatinib.
Sensitive CRC cell lines and explants have increased levels and activity of Src and downstream mediators
Next, we assessed protein levels and activation of Src, FAK, and Stat-3 in two of the most resistant cell lines (SW620 and SW480), two of three sensitive cell lines (H508 and LS174T), and 10 CRC explants by Western blotting. We hypothesized that cell lines and explants that were more sensitive to saracatinib would have higher levels of Src pathway activity when compared with resistant cell lines and explants. As shown in Fig. 5A, both protein levels and activity of Src, FAK, and Stat-3 were significantly elevated in the sensitive cell lines when compared with the resistant cell lines. Treatment with saracatinib (0.5 μmol/L) for 1 hour resulted in a decrease in the phosphorylation of Src (Y416), FAK (Y861), and Stat-3 (Y705). Furthermore, the sensitive explants CRC007 and CRC040 had increased activation of Src, but not FAK and Stat-3 when compared with the resistant CRC explants (Fig. 5B). Evaluation of tumors from CRC007 and CRC027 28 days after saracatinib treatment showed a decrease in Src activation (Fig. 5C). Finally, immunohistochemistry shows that Src activity was increased in the sensitive tumor (CRC007) versus the resistant tumor (CRC027; Fig. 5D).
Fig. 5.
Saracatinib-sensitive CRC cell lines and explant have increased activation of Src and downstream mediators. A, the sensitive cell lines H508 and LS174T had greater activation of Src, FAK, and Stat-3 when compared with the resistant cell lines SW620 and SW480. Treatment with saracatinib (500 nmol/L) for 1 h resulted in a decrease in the phosphorylation of Src, FAK, and Stat-3 (n = 3). B, evaluation of Src, FAK, and Stat-3 activation in CRC explants by immunoblotting showed that CRC007 and CRC040, sensitive tumors, had the highest activation of Src and FAK but not Stat-3. C, representative graph of two tumors (CRC007 and CRC027) treated with saracatinib (50 mg/kg/d) showed a decrease in the activation of Src. D, immunohistochemical staining of p-Src (Y416) shows positive staining in the sensitive tumor (CRC007) and negative staining in the resistant tumor (CRC027).
Discussion
Significant advances in the molecular characterization of aberrant pathways in cancer have led to the development of targeted agents. However, there is considerable heterogeneity with respect to dysregulated pathways and mutations in genes involved in cell cycle control, proliferation, angiogenesis, and metastasis. Consequently, many targeted agents lack efficacy because they usually benefit a small subset of patients, and clinical trials are not sufficiently powered to detect these differences. Therefore, identifying this patient population through predictive biomarkers before treatment may lead to more effective therapy and ultimately better outcomes. In this study, we explored the efficacy of saracatinib (a Src inhibitor) in CRC and used a K-TSP classifier, FISH, and pathway activation to predict sensitivity to saracatinib in our patient-derived CRC explant model.
Saracatinib, a potent and selective inhibitor of Src/Abl kinase activity currently in phase II clinical development, has shown a range of antiproliferative effects in vitro in selected lung, breast, ovarian, prostate, pancreas, and colon cell lines (35). Whereas LoVo and SW403 CRC cell lines were particularly sensitive to in vitro growth inhibition by saracatinib, HT29 and HCT116 were not inhibited at pharmacologically relevant concentrations. Although saracatinib did not inhibit the growth of LoVo, Colo205, or HT29 tumors in vivo, Src kinase activity was inhibited as evidenced by reduced phosphorylation of Src substrates FAK and paxillin. Other Src inhibitors such as bosutinib and dasatinib have also shown antiproliferative effects in CRC (36, 37). In particular, dasatinib showed antiproliferative activity in the CRC cell lines DLD-1, HT29, WiDr, Caco-2, and SW48 (37). Additionally, bosutinib was shown to inhibit tumor growth in HT29, Colo205, HCT116, and DLD-1 in a mouse xenograft model (36). Although Src substrates such as FAK and paxillin may be suitable biomarkers of Src kinase activity, clearly there is a need for biomarkers that are predictive of tumor growth inhibition in vivo.
Here, we investigated the efficacy of saracatinib on 23 CRC cell lines and identified 20 resistant and 3 sensitive cell lines. Importantly, treatment of resistant and sensitive cell lines in a xenograft model confirmed the in vitro results; SW620 and SW480 were resistant, and LS180 and LS174T were sensitive to saracatinib. Moreover, we show in our human CRC explant model that 2 out 10 explants responded to saracatinib. Additionally, we show that the antiproliferative effects seen with saracatinib in cell lines are through G1 cell cycle arrest and not apoptosis. Taken together, these findings suggest that inhibition of Src with a selective inhibitor may prove to be beneficial in a subset of CRC patients. The identification of patient stratification markers for targeted therapies such as Src kinase inhibitors is a key challenge for drug development.
Whereas several Src kinase inhibitors have consistently inhibited Src-mediated events such as cell adhesion and cell motility in vitro, the phosphorylation of key substrates such as FAK and paxillin, or cell cycle perturbation (37–39), the pattern of gene expression in response to Src kinase inhibition seems to be pharmacophore related. Whereas saracatinib, dasatinib, and bosiutinib are all potent inhibitors of Src (and Src family kinases), their off-target kinase inhibition profiles are likely to differentially modulate gene expression. For example, K-TSP analysis of pancreatic tumors treated with bosutinib or saracatinib identified different pairs of genes indicative of sensitive tumors. We have previously described the development of K-TSP classifiers for Src kinase inhibitors in pancreas cancer. The K-TSP derived for bosutinib in pancreatic explant models included the gene pairs GPR116>LQK1, CEACAM7>SPFH2, and ALPK3>GUF1 (18), whereas saracatinib sensitivity was defined by LRRC19>IGFBP2 (19). Some pancreatic explant models showed an equivalent response to saracatinib or bosutinib, whereas other tumors were resistant to one agent but sensitive to the other. The kinase inhibitor profile, together with the pattern of pathway activation and tumor specific differences, are likely determinants of sensitivity and successful predictors of response.
The advantage of the K-TSP approach is that it is easy to interpret as the classifier involves only a few informative genes. In this study, we tested the LLRC19>IGFBP2 gene pair in our CRC model; however, this gene pair was not predictive, suggesting that the mechanisms of sensitivity and resistant are different between pancreatic and colon cancer. These results may also be related to enzyme inhibition profiles of the Src kinase inhibitors and the relative potency versus key enzymes in different tissues. The data suggest that a gene signature defining sensitivity to particular kinase inhibitors may not be universally applicable and will need to be established for each target tumor type.
Consequently, we evaluated other gene sets to predict sensitivity to saracatinib in CRC and identified three gene pairs that predict sensitivity: TOX>GLIS2, TSPAN7>BCAS4, and PARD6G>NXN. Four (TOX, TSPAN7, GLIS2, and NXN) of the K-TSP genes were among the top 100 differentially expressed genes. Using the training set (CRC cell lines in vitro and in vivo), the gene pairs were able to predict with 70% accuracy in the CRC explants.
Many of the genes identified by the K-TSP classifier have been implicated in cancer. Tetraspanins such as TSPAN7 are transmembrane proteins that form complexes with one another; in cancer, higher expression levels of tetraspanins are associated with metastasis (40). GLIS family zinc finger 2 is a transcriptional repressor that has been shown in conjunction with Src to enhance the nuclear translocation of p120 catenin, a protein important in facilitating cell-cell interactions (41). The BCAS4 gene was one of the most amplified genes in breast cancer cell lines (42); however, its function and role in cancer is unknown. TOX is a chromatin-associated protein that specifically binds to DNA. Genome-wide studies in breast cancer have revealed TOX as a susceptibility gene (43). NXN is a member of the thioredoxin family of proteins, which possess thioloxidoreductase activity. Recently, it has been shown to regulate the Wnt/β-catenin pathway, an important pathway regulating cellular differentiation and proliferation (44). Finally, PARD6G is a cell polarity component that plays a role in cell division (45). Because Src has hundreds of client proteins with which it interacts, it is not surprising that many proteins may contribute to drug sensitivity. Further functional studies are warranted to specifically address the importance and mechanism of these genes in altering sensitivity to this drug.
Studies have shown that the expression of Src is elevated in premalignant lesions and subsequently increases as tumors progress with some of the highest levels in metastatic lesions (24). It has also been shown that a gain in chromosome 20 commonly occurs in CRC (46). Given that Src levels are elevated in CRC, we investigated Src gene copy number by FISH. Here, we show that there is heterogeneity in Src gene copy number, with most of the CRC cell lines and explants having a gain in Src gene number. To our knowledge, this has not been reported previously. We have not seen Src gene amplification in either cell lines or human explants. Additionally, we show that cell lines and tumors that have a gain in Src gene copy number are associated with resistance to saracatinib. As Src mutations were not found in our study, these results indicate that an increase in Src gene copy rather than mutations are more prevalent in CRC and may possibly be a predictor of resistance to saracatinib. Characterization of Src gene copy number in annotated premalignant and malignant human colorectal tumor clinical samples, as well as clinical trial samples in patients treated with Src inhibitors, may elucidate the relevance of these findings.
Levels and activation of Src are elevated at all stages of CRC in which it is postulated to enhance tumor growth and progression by activating many downstream targets such as FAK and Stat-3. We found that the activation of Src, FAK, and Stat-3 are increased in sensitive cell lines when compared with resistant cell lines. Moreover, the activation of Src was also the highest in the sensitive CRC explants (CRC007 and CRC040) when compared with resistant explants. This suggests that Src pathway activation may be a marker of sensitivity to Src inhibitors such as saracatinib in CRC.
For this study, we developed a patient-derived human CRC explant model to evaluate efficacy and identify bio-markers of sensitivity to saracatinib. Although there are several advantages of this preclinical model compared with cell lines, there are also several weaknesses (47). First, it is unknown whether this model will be able to predict activity in the clinic. Future clinical trials where these biomarkers are prospectively evaluated in patients will likely resolve this issue. A second limitation of this model is that tumor cells in this xenograft model do not metastasize. Therefore, we could not evaluate the effects of saracatinib on metastasis, one of the main functions of Src in altering tumor cell behavior. A third challenge is the limited dynamic range of the model, in which small differences in TGI are difficult to interpret and may be irrelevant. A final issue with the model is the inability to assess immunologic mechanisms because the model uses immunocompromised mice to prevent graft rejection.
Like many targeted agents, the clinical activity of saracatinib as a monotherapy has been modest. There were no responses in a small phase II trial of 10 patients in third or fourth line CRC (48). It is likely that the future of this class of agents lies in proper selection of sensitive patient subsets, as well as combinational strategies. Initial results of a combination trial of FOLFOX, cetuximab, and dasatinib showed an impressive response rate with further results pending (49). It has been shown that oxaliplatin could lead to an increase in SRC expression and activition in HT29 CRC cell line (50). Furthermore, treatment with oxaliplatin and dasatinib resulted in significant reduction in tumor volume when compared with nontreated mice in a liver metastatic preclinical model, whereas there were no differences when single agent oxaliplatin or dasatinib were administered (50). Although the clinical implications of human explant models remains unknown, our results suggest that patient subsets sensitive to Src inhibitors may be identified. Follow-up biomarker-driven and/or combinatorial clinical trials may be warranted.
Translational Relevance.
In colorectal cancer, studies have shown that the expression of Src is elevated in premalignant lesions and subsequently increases as tumors progress to metastatic lesions. The objective of this study was to evaluate the efficacy of saracatinib, a Src inhibitor, and to identify predictive biomarker(s) of sensitivity or resistance to saracatinib in a colorectal cancer human explant preclinical model. These data show that saracatinib had antitumor activity in a small subset of tumors. Additionally, fluorescence in situ hybridization and a K-Top Scoring Pairs gene classifier derived from baseline gene array data had predictive power for saracatinib sensitivity. Because clinical trials of Src inhibitors with unselected patients have been disappointing thus far, these predictive biomarkers may prove to be valuable in the clinical setting by preselecting patients who may benefit from saracatinib therapy.
Footnotes
Disclosure of Potential Conflicts of Interest
J.J. Arcoli: advisory board for saracatinib, AstraZeneca. The other authors disclosed no potential conflicts of interest.
References
- 1.Bromann PA, Korkaya H, Courtneidge SA. The interplay between Src family kinases and receptor tyrosine kinases. Oncogene. 2004;23:7957–68. doi: 10.1038/sj.onc.1208079. [DOI] [PubMed] [Google Scholar]
- 2.Frame MC. Src in cancer: deregulation and consequences for cell behaviour. Biochim Biophys Acta. 2002;1602:114–30. doi: 10.1016/s0304-419x(02)00040-9. [DOI] [PubMed] [Google Scholar]
- 3.Schlessinger J. New roles for Src kinases in control of cell survival and angiogenesis. Cell. 2000;100:293–6. doi: 10.1016/s0092-8674(00)80664-9. [DOI] [PubMed] [Google Scholar]
- 4.Summy J, Gallick G. Src family kinases in tumor progression and metastasis. Cancer Metastasis Rev. 2003;22:337–58. doi: 10.1023/a:1023772912750. [DOI] [PubMed] [Google Scholar]
- 5.LaVallee T, Prudovsky I, McMahon G, Hu X, Maciag T. Activation of the MAP kinase pathway by FGF-1 correlates with cell proliferation induction while activation of the Src pathway correlates with migration. J Cell Biol. 1998;141:1647–58. doi: 10.1083/jcb.141.7.1647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peterson J, Kulik G, Jelinek T, Reuter C, Shannon J, Weber M. Src phosphorylates the insulin-like growth factor type I receptor on the autophosphorylation sites. Requirement for transformation by Src. J Biol Chem. 1996;271:31562–71. doi: 10.1074/jbc.271.49.31562. [DOI] [PubMed] [Google Scholar]
- 7.Rahimi N, Hung W, Tremblay E, Saulnier R, Elliott B. c-Src kinase activity is required for hepatocyte growth factor-induced motility and anchorage-independent growth of mammary carcinoma cells. J Biol Chem. 1998;273:33714–21. doi: 10.1074/jbc.273.50.33714. [DOI] [PubMed] [Google Scholar]
- 8.Tice D, Biscardi J, Nickles A, Parsons S. Mechanism of biological synergy between cellular Src and epidermal growth factor receptor. Proc Natl Acad Sci U S A. 1999;96:1415–20. doi: 10.1073/pnas.96.4.1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hauck C, Hsia D, Schlaepfer D. The focal adhesion kinase: a regulator of cell migration and invasion. IUBMB Life. 2002;53:115–9. doi: 10.1080/15216540211470. [DOI] [PubMed] [Google Scholar]
- 10.Jones RJ, Brunton VG, Frame MC. Adhesion-linked kinases in cancer;emphasis on src, focal adhesion kinase and PI 3-kinase. Eur J Cancer. 2000;36:1595–606. doi: 10.1016/s0959-8049(00)00153-2. [DOI] [PubMed] [Google Scholar]
- 11.Schaller M. Biochemical signals and biological responses elicited by the focal adhesion kinase. Biochimica et Biophysica Acta. 2001;1540:1–21. doi: 10.1016/s0167-4889(01)00123-9. [DOI] [PubMed] [Google Scholar]
- 12.Owens L, Xu L, Craven R, et al. Overexpression of the focal adhesion kinase (p125FAK) in invasive human tumors. Cancer Res. 1995;55:2752–5. [PubMed] [Google Scholar]
- 13.Niu G, Wright K, Huang M, et al. Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene. 2002;21:2000–8. doi: 10.1038/sj.onc.1205260. [DOI] [PubMed] [Google Scholar]
- 14.Yu CL, Meyer DJ, Campbell GS, et al. Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the Src oncoprotein. Science. 1995;269:81–3. doi: 10.1126/science.7541555. [DOI] [PubMed] [Google Scholar]
- 15.Wong CH, Baehner FL, Spassov DS, et al. Phosphorylation of the SRC epithelial substrate Trask is tightly regulated in normal epithelia but widespread in many human epithelial cancers. Clin Cancer Res. 2009;15:2311–22. doi: 10.1158/1078-0432.CCR-08-2533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang F, Reeves K, Han X, et al. Identification of candidate molecular markers predicting sensitivity in solid tumors to dasatinib: rationale for patient selection. Cancer Res. 2007;67:2226–38. doi: 10.1158/0008-5472.CAN-06-3633. [DOI] [PubMed] [Google Scholar]
- 17.Rubio-Viqueira B, Jimeno A, Cusatis G, et al. An in vivo platform for translational drug development in pancreatic cancer. Clin Cancer Res. 2006;12:4652–61. doi: 10.1158/1078-0432.CCR-06-0113. [DOI] [PubMed] [Google Scholar]
- 18.Messersmith WA, Rajeshkumar NV, Tan AC, et al. Efficacy and pharmacodynamic effects of bosutinib (SKI-606), a Src/Abl inhibitor, in freshly generated human pancreas cancer xenografts. Mol Cancer Ther. 2009;8:1484–93. doi: 10.1158/1535-7163.MCT-09-0075. [DOI] [PubMed] [Google Scholar]
- 19.Rajeshkumar NV, Tan AC, De Oliveira E, et al. Antitumor effects and biomarkers of activity of AZD0530, a Src inhibitor, in pancreatic cancer. Clin Cancer Res. 2009;15:4138–46. doi: 10.1158/1078-0432.CCR-08-3021. [DOI] [PubMed] [Google Scholar]
- 20.Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225–49. doi: 10.3322/caac.20006. [DOI] [PubMed] [Google Scholar]
- 21.Yeatman T. A renaissance for src. Nat Rev Cancer. 2004;4:470–80. doi: 10.1038/nrc1366. [DOI] [PubMed] [Google Scholar]
- 22.Malek RL, Irby RB, Guo QM, et al. Identification of Src transformation fingerprint in human colon cancer. Oncogene. 2002;21:7256–65. doi: 10.1038/sj.onc.1205900. [DOI] [PubMed] [Google Scholar]
- 23.Cartwright C, Meisler A, Eckhart W. Activation of the pp60c-src protein kinase is an early event in colonic carcinogenesis. Proc Natl Acad Sci U S A. 1990;87:558–62. doi: 10.1073/pnas.87.2.558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Talamonti MS, Roh MS, Curley SA, Gallick GE. Increase in activity and level of pp60c-src in progressive stages of human colorectal cancer. J Clin Invest. 1993;91:53–60. doi: 10.1172/JCI116200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Park J, Meisler A, Cartwright C. c-Yes tyrosine kinase activity in human colon carcinoma. Oncogene. 1993;8:2627–35. [PubMed] [Google Scholar]
- 26.Karapetis CS, Khambata-Ford S, Jonker DJ, et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med. 2008;359:1757–65. doi: 10.1056/NEJMoa0804385. [DOI] [PubMed] [Google Scholar]
- 27.Hecht JR, Mitchell E, Chidiac T, et al. A randomized phase IIIB trial of chemotherapy, bevacizumab, and panitumumab compared with chemotherapy and bevacizumab alone for metastatic colorectal cancer. J Clin Oncol. 2009;27:672–80. doi: 10.1200/JCO.2008.19.8135. [DOI] [PubMed] [Google Scholar]
- 28.Tol J, Koopman M, Cats A, et al. Chemotherapy, bevacizumab, and cetuximab in metastatic colorectal cancer. N Engl J Med. 2009;360:563–72. doi: 10.1056/NEJMoa0808268. [DOI] [PubMed] [Google Scholar]
- 29.Irby R, Mao W, Coppola D, et al. Activating SRC mutation in a subset of advanced human colon cancers. Nat Genet. 1999;21:187–90. doi: 10.1038/5971. [DOI] [PubMed] [Google Scholar]
- 30.Jhawer M, Goel S, Wilson AJ, et al. PIK3CA mutation/PTEN expression status predicts response of colon cancer cells to the epidermal growth factor receptor inhibitor cetuximab. Cancer Res. 2008;68:1953–61. doi: 10.1158/0008-5472.CAN-07-5659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dangles-Marie V, Pocard M, Richon S, et al. Establishment of human colon cancer cell lines from fresh tumors versus xenografts: comparison of success rate and cell line features. Cancer Res. 2007;67:398–407. doi: 10.1158/0008-5472.CAN-06-0594. [DOI] [PubMed] [Google Scholar]
- 32.Tusher VG, Tibshirani R, Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci U S A. 2001;98:5116–21. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tan AC, Naiman DQ, Xu L, Winslow RL, Geman D. Simple decision rules for classifying human cancers from gene expression profiles. Bioinformatics. 2005;21:3896–904. doi: 10.1093/bioinformatics/bti631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dreyfus GMG. Withdrawing an example from the training set: an analytic estimation of its effect on a non-linear parameterised model. Neurocomputing. 2000;35:195–201. [Google Scholar]
- 35.Green TP, Fennell M, Whittaker R, et al. Preclinical anticancer activity of the potent, oral Src inhibitor AZD0530. Mol Oncol. 2009;3:248–61. doi: 10.1016/j.molonc.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Golas JM, Lucas J, Etienne C, et al. SKI-606, a Src/Abl inhibitor with in vivo activity in colon tumor xenograft models. Cancer Res. 2005;65:5358–64. doi: 10.1158/0008-5472.CAN-04-2484. [DOI] [PubMed] [Google Scholar]
- 37.Serrels A, Macpherson IR, Evans TR, et al. Identification of potential biomarkers for measuring inhibition of Src kinase activity in colon cancer cells following treatment with dasatinib. Mol Cancer Ther. 2006;5:3014–22. doi: 10.1158/1535-7163.MCT-06-0382. [DOI] [PubMed] [Google Scholar]
- 38.Nam S, Kim D, Cheng JQ, et al. Action of the Src family kinase inhibitor, dasatinib (BMS-354825), on human prostate cancer cells. Cancer Res. 2005;65:9185–9. doi: 10.1158/0008-5472.CAN-05-1731. [DOI] [PubMed] [Google Scholar]
- 39.Chang YM, Bai L, Liu S, Yang JC, Kung HJ, Evans CP. Src family kinase oncogenic potential and pathways in prostate cancer as revealed by AZD0530. Oncogene. 2008;27:6365–75. doi: 10.1038/onc.2008.250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Le Naour F, Andre M, Greco C, et al. Profiling of the tetraspanin web of human colon cancer cells. Mol Cell Proteomics. 2006;5:845–57. doi: 10.1074/mcp.M500330-MCP200. [DOI] [PubMed] [Google Scholar]
- 41.Hosking CR, Ulloa F, Hogan C, et al. The transcriptional repressor Glis2 is a novel binding partner for p120 catenin. Mol Biol Cell. 2007;18:1918–27. doi: 10.1091/mbc.E06-10-0941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Barlund M, Monni O, Weaver JD, et al. Cloning of BCAS3 (17q23) and BCAS4 (20q13) genes that undergo amplification, overexpression, and fusion in breast cancer. Genes Chromosomes Cancer. 2002;35:311–7. doi: 10.1002/gcc.10121. [DOI] [PubMed] [Google Scholar]
- 43.Easton DF, Pooley KA, Dunning AM, et al. Genome-wide association study identifies novel breast cancer susceptibility loci. Nature. 2007;447:1087–93. doi: 10.1038/nature05887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Funato Y, Miki H. Nucleoredoxin, a novel thioredoxin family member involved in cell growth and differentiation. Antioxid Redox Signal. 2007;9:1035–57. doi: 10.1089/ars.2007.1550. [DOI] [PubMed] [Google Scholar]
- 45.Lee M, Vasioukhin V. Cell polarity and cancer-cell and tissue polarity as a non-canonical tumor suppressor. J Cell Sci. 2008;121:1141–50. doi: 10.1242/jcs.016634. [DOI] [PubMed] [Google Scholar]
- 46.Nanashima A, Yamaguchi H, Yasutake T, et al. Gain of chromosome 20 is a frequent aberration in liver metastasis of colorectal cancers. Dig Dis Sci. 1997;42:1388–93. doi: 10.1023/a:1018885803931. [DOI] [PubMed] [Google Scholar]
- 47.Jimeno AAS, Karikari C, Garrido-Laguna I, et al. A prospective validation of a direct tumor xenograft model in pancreatic ductal adenocarcinoma (PDA) J Clin Oncol. 2008;26:4500. [Google Scholar]
- 48.Fogelman DR, Kopetz S, Eng C. Emerging drugs for colorectal cancer. Expert Opin Emerg Drugs. 2008;13:629–42. doi: 10.1517/14728210802544575. [DOI] [PubMed] [Google Scholar]
- 49.Kopetz SRAW, Glover K, Henry L, et al. Phase I study of Src inhibition with dasatinib in combination with 5-fluoruracil, leucovorin, oxaliplatin (FOLFOX) and cetuximab in metastatic colorectal cancer. ASCO-2008 Gastrointestinal Cancers Symposium. 2008:325. [Google Scholar]
- 50.Kopetz S, Lesslie DP, Dallas NA, et al. Synergistic activity of the SRC family kinase inhibitor dasatinib and oxaliplatin in colon carcinoma cells is mediated by oxidative stress. Cancer Res. 2009;69:3842–9. doi: 10.1158/0008-5472.CAN-08-2246. [DOI] [PMC free article] [PubMed] [Google Scholar]