Abstract
Autophagy is critical to the process of development as mouse models have shown that lack of autophagy leads to developmental arrest during the pre-implantation stage of embryogenesis. The process of autophagy is regulated through signaling pathways, which respond to the cellular environment. Therefore, any alteration in environment may lead to the dysregulation of the autophagic process potentially resulting in cell death. Using both in vitro and in vivo models to study autophagy in the pre-implantation murine embryo, we observed that the cells responds to environmental stressors (i.e. hyperglycemic environment) by increasing activation of autophagy in a differential pattern within the embryo. This upregulation is accompanied by an increase in apoptosis, which appears to plateau at high concentrations of glucose. Activation of the autophagic pathway was further confirmed by an increase in GAPDH activity in both in vivo and in vitro hyperglycemic models, which has been linked to autophagy through activation of the Atg12 gene. Furthermore, this increase in autophagy in response to a hyperglycemic environment was observed as early as the oocyte stage. In conclusion, in this paper we provided evidence for a differential response of elevated activation of autophagy in embryos and oocytes exposed to a hyperglycemic environment.
Keywords: Autophagy, diabetes, blastocyst, oocyte, hyperglycemia
Introduction
Autophagy is a programmed method of protein degradation and recycling of the necessary cellular building blocks of glucose, amino acids and fatty acids. This process has been shown to be critical to cell survival during periods of nutrient and specifically glucose deprivation, as well as during development and differentiation (Aki et al. 2003; Mizushima et al. 2004; Scott et al. 2004; Singh et al. 2009). During the developmental progression from oocyte to cleavage stage embryo, maternal proteins and RNAs are degraded and quickly replaced by newly synthesized embryonic counterparts. Mizushima et al. recently elucidated a physiological role of autophagy to aid in this process (Tsukamoto et al. 2008a; Tsukamoto et al. 2008b). By creating an oocyte-specific Atg5 (autophagy-related 5, a critical component to the formation of autophagosomes) knockout mouse, the authors determined that fertilization of Atg5 null oocytes with Atg5 null sperm resulted in developmental arrest between the 4-cell and 8-cell stage. Fertilization of the Atg5 null oocyte with wild-type sperm, however, resulted in normal embryo development. They also demonstrated that protein-recycling rates were abnormal in the embryos entirely devoid of ATG5, and concluded that proper autophagic degradation within early embryos is essential for pre-implantation development. Results suggesting a role for autophagy during embryoid cavitation were also recently published (Qu et al. 2007). This group reported that loss of either Beclin-1 (required for the initiation of autophagosome formation) or Atg5, both essential autophagic genes, leads to failure of cavitation due to the persistence of cellular corpses in murine ESCs. These cells fail to display signals to indicate removal. This dysregulation is also associated with low levels of ATP. Overall, the lethality associated with removal of this pathway, indicates the critical roles that this pathway plays in embryo development, many of which may not be elucidated yet.
The activation of autophagy has been characterized as a survival mechanism in the prevention of cell death (Boya et al. 2005). This role for autophagy has been reviewed elsewhere (Maiuri et al. 2007; Thorburn 2008). Briefly, the interplay between survival and autophagy is demonstrated by studies showing that lack of Beclin 1 expression, leads to excessive apoptotic cell death with embryo lethality at a peri-implantation stage (Yue, Jin et al. 2003). While the role for autophagy as a pro-survival mechanism is well accepted, the idea that excess autophagy with elevated levels of autophagosomes leads to cell death is still in debate. However, it is accepted that excessive autophagy leads to cell death during ischemia (Akazawa et al. 2004). Therefore, there may be certain circumstances in which this interrelationship exists. And while the mechanisms may not be entirely clear, recent studies suggest that oxidative stress plays a role in this switch from survival to death (Grune et al. 2003; Kiffin et al. 2006; Rodriguez-Enriquez et al. 2004; Sakaida et al. 1990).
In diabetes models it has been postulated that, autophagy is activated in certain tissues as a mechanism of protection against cellular damage resulting from oxidative stress. This activation is evident in the pancreatic beta cells of diabetic animal models and humans (Fujitani et al. 2009; Masini et al. 2009). Other tissues that exhibit an increased level of autophagy in diabetic models include the muscle, neuronal tissue, and liver (Amherdt et al. 1974; Towns et al. 2005). However, a decrease in autophagy mediated protein turnover is observed in the renal cortex of the diabetic kidney (Sooparb et al. 2004). Therefore, it appears that tissues may regulate autophagy differently in response to a diabetic environment, and thus, investigation of this pathway is important as a potential therapeutic target for problems associated with diabetes.
The uterine milieu plays an important role in the development and implantation of an embryo. Previously, we have determined that a maternal diabetic state in mice results in an increase in number of fetal resorptions and an increase in congenital malformations of the fetus. These deleterious effects resulted with only a 96-hour exposure to the diabetic environment, as blastocyst stage embryos transferred from diabetic mothers into non-diabetic control mothers also displayed these phenotypes (Wyman et al. 2008). In addition, we have shown that the hyperglycemic environment of the uterus, by treatment with high glucose leads to an increase in apoptosis in the embryo (Moley et al. 1998a). Therefore, we wanted to investigate the autophagic pathway in a diabetic milieu.
In this paper we present evidence that the pre-implantation embryo alters autophagy in response to the external environment. The embryos respond to stressors including a known activator of autophagy and a hyperglycemic environment by increasing the activation of autophagy, identifying autophagy as a pathway for survival in the blastocyst. Furthermore, the embryonic response is differentially activated in what appears to be specific cell niches within the embryos.
Material and Methods
Oocyte and embryo retrieval
To collect ovulated oocytes, control and diabetic B6SJL mice (Jackson Laboratory, Bar Harbor, Maine) received an injection of 10 IU human Chorionic Gonadotropin (hCG) 2 days after PMSG priming (PSMG 2000I.U., National Hormone & Peptide Program, Torrance, California; hCG, Sigma, St. Louis, Missouri). Oocytes were recovered from oviductal ampullae 13.5 h post-hCG, and cumulus cells were removed by incubating briefly in 1 mg/ml hyaluronidase. For embryo retrieval, the mice received the hormonal stimulation mentioned above and then were mated overnight with males of proven fertility. Mating was confirmed by the presence of a vaginal plug. Embryos for the in vivo blastocyst study (Figure 3A) were obtained by flushing the uterine horns 96 h post-hCG. For the in vitro hyperglycemia and autophagy activation studies (Figures 1A-E, 2A-J and 3B), embryos were recovered from control mice 76 h post-hCG and mating, and were cultured for 30 h in KSOM/0.25%BSA(v/v) (KSOM, Millipore Specialty Media, Massachusetts) and the indicated culture conditions at 37 °C in 5% CO2.
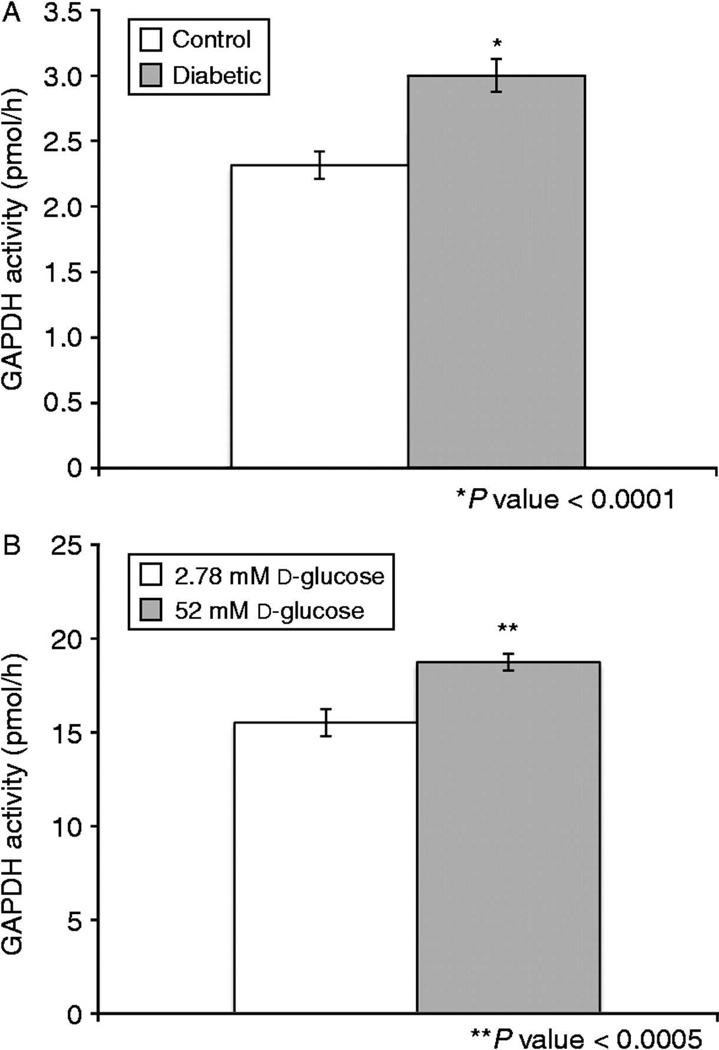
Figure 3. GAPDH activity and autophagy are increased in embryos exposed to diabetic environments both in vivo and in vitro.
GAPDH activity (in vivo): embryos from control versus streptozocin-induced diabetic mothers (A) *p-value<0.0001. GAPDH activity (in vitro): embryos exposed to 2.78mM versus 52mM D-glucose (B), *p-value<0.0005 (n= at least 50 for each group).
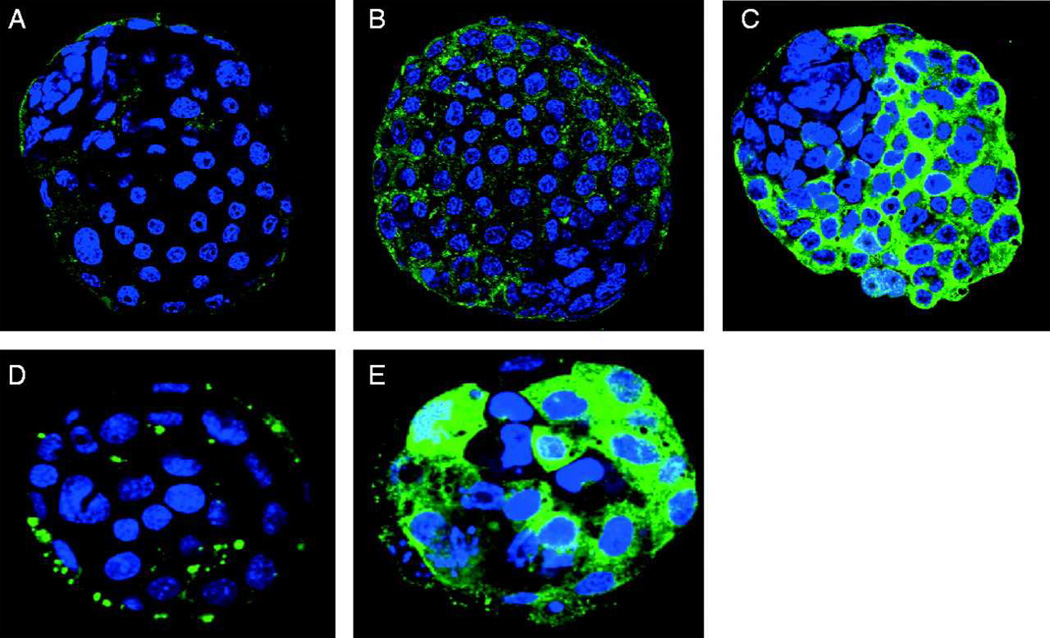
Figure 1. Beclin-1 and LC3 expression in rapamycin treated embryos.
Ten mouse control blastocysts were incubated for 30 hours in each group: DMSO control (A), 1µM (B), or 5µM (C) rapamycin. This experiment was repeated 3 times. Beclin-1 expression is increased in a variable pattern, with the most expression observed in the trophectoderm cells. LC3 expression in mouse control blastocysts incubated in DMSO (D, n=10) or 5µM rapamycin (E, n=10). This experiment was conducted 3 times.
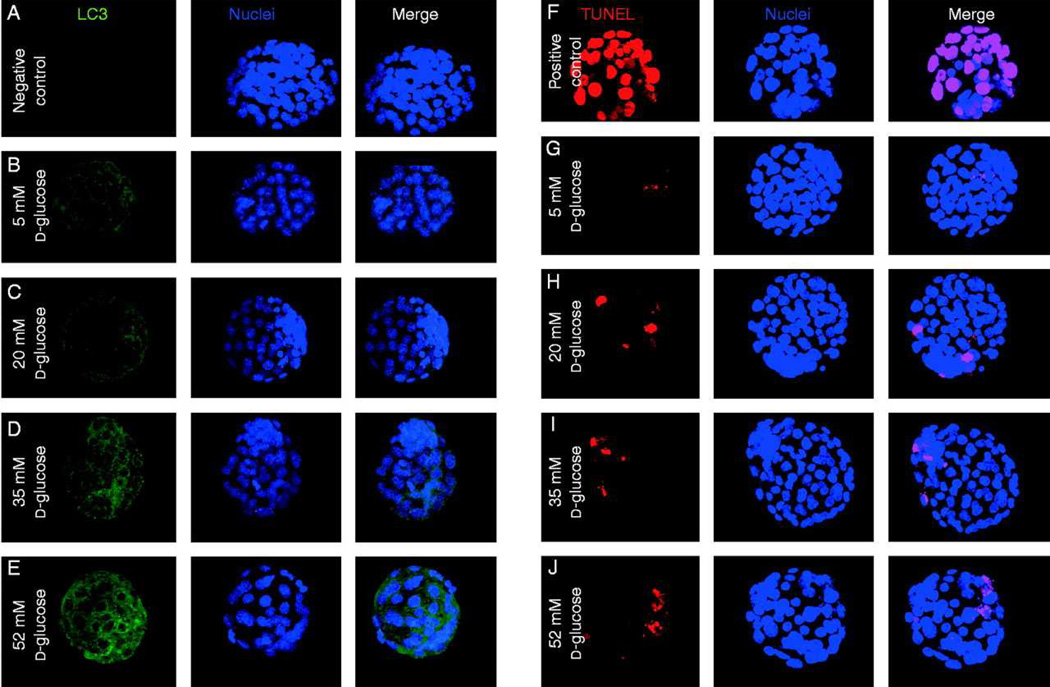
Figure 2. Autophagic and apoptoic responses to varying levels of excess glucose.
LC3 expression in embryos. Negative control (A), physiologic 5mM (B), 20mM (C), 35mM (D) and 52mM (E) glucose. Cell death detection assay for apoptosis. Positive control of DNase treated embryos (F), physiologic 5mM (G), 20mM (H), 35mM (I), 52mM (J) glucose. Each group consisted of at least 30 embryos and the experiment was repeated 3 times.
Immunofluorescence staining
Embryos were collected and fixed in 3% Paraformaldehyde in PBS/2%BSA(v/v) for 20 minutes at room temperature. The embryos were permeabilized in 0.5% TritonX-100 in PBS/2%BSA for 30 minutes. To block, the embryos were placed in 5% normal goat serum in PBS/2%BSA for one hour. Then the embryos were incubated in the primary antibody (1:250) in PBS/2%BSA overnight at 4°C, (LC3:Novus Biologicals, Littleton, Colorado; Beclin-1, Cell Signaling Technology, Massachusetts). The embryos were washed three times in PBS/2%BSA and then incubated in secondary antibody (Alexa Fluor goat-anti-rabbit IgG 488, Molecular Probes, Eugene, Oregon) for 45 minutes at room temperature. The embryos were rinsed three times again and placed in To-pro®-3-iodide (Molecular Probes, Eugene, Oregon) (1:500) in PBS for 15 minutes. After three more rinses in PBS, the embryos were mounted on slides using Vectashield (Vecta, Burlingame, California) and visualize by confocal microscopy. All experimental groups (i.e. Figure 1A-C or D and E; Figure 2) were conducted on the same day in order to use the same prepared secondary antibody preparation. Relative fluorescence was quantified by a blinded observer as described previously (Jungheim et al. 2009).
Cell death assay
Apoptosis was detected using the In Situ Cell Death Detection Kit, TMR red (Roche, Mannheim, Germany). The embryos were fixed and permeabilized as indicated in the immunofluorescence staining method. A positive control was obtained by treating embryos with DNase (0.5µL/mL in PBS) for 20 minutes at 37°C. Then the embryos were incubated in the reaction mix, as per manufacturer’s specifications for 1 hour at 37°C in the dark. All subsequent reactions were also carried out in the dark. After three rinses in PBS/2%BSA, the embryos were counterstained with To-pro®-3-iodide as mentioned before, rinsed and then mounted on slides. The slides were viewed on a confocal microscope.
GAPDH activity assay
This assay has been previously described for individual cell activity measurements (Passonneau & Lowry, 1993). Individual mouse blastocysts were extracted in 20mM Phosphate buffer (pH 7.4), 0.02% BSA, 0.5mM EDTA (pH 7.0), 5mM B-Me, 0.25% glycerol and 0.5% Triton X-100(v/v) at room temperature for 120 min under oil and stored at −75°C. A 0.1µL aliquot was added to 1µL GAPDH reagent containing 50mM Imidazole HCL (pH 7.0), 0.05%BSA, 1mM EDTA (pH 7.0), 1mM B-Me, 1mM Na2HASO4, 100µM NAD, and 100µM Glyceraldehyde 3-phosphate at room temperature for 1 hour. NADH standards in GAPDH reagent were added in this step. The reaction was stopped by the addition of 1µL of 0.12N NaOH and heated to 80°C for 25 min. To amplify, a 0.5µL aliquot was taken out and added to 10µL NAD cycling reagent at room temperature overnight under oil for 15,000 fold amplification, as performed previously by our group (Chi et al. 2002). The reaction was stopped by the addition of 1µL 1N NaOH and heated at 80°C for 25 min. To read the samples, a 10µL aliquot was taken out and added to 1mL Malate indicator reagent, as performed previously to obtain the NADH final reading in an A-1 Filter Fluorometer (Farrand Optical Components and Instruments, Valhalla, New York) (Chi et al. 2002). All calculations were based on the internal NADH standards.
Generation of diabetic mice
To generate an in vivo diabetic model, 3-week old female B6SJLF1 mice received a single injection of streptozotocin at a dose of 190 mg/kg. Four days after injection, a tail-blood sample was measured for glucose concentrations via a Contour TS One Touch Glucometer (Bayer, Mishawaka, Indiana). Glucose levels greater than 300 mg/dl, were considered diabetic. Age-matched controls injected with PBS were used.
Transmission electron microscopy
For ultrastructural analysis of autophagy, 100 oocytes of each group were processed for transmission electron microscopy as described previously in the Molecular Microbiology Imaging Facility at Washington University in St. Louis (Gualtieri et al. 2009). The presence of autophagosomes were determined from electron micrographs at 10,000X magnification. To quantify the autophagosomes, the number of autophagosomes and the area in 10 random sections of each group were recorded using ImageJ (National Institutes of Health, Bethesda, Maryland). The following criteria for identifying autophagosomes were used: vesicles with a double membrane, between 0.3 and 2 um, with clearly recognizable cytoplasmic contents and are not multilamellar bodies.
Statistical analysis
All experiments were completed in triplicate with at least 10 embryos or oocytes per group in each experiment. The images in the figures are representative of the group of embryos in each group. For the GAPDH assay, a student’s t-test was performed. Significance was defined as p<0.05.
Results
Embryonic autophagy is influenced by environment
Our lab and others have observed a basal level of autophagic activation during all stages of pre-implantation embryonic development in mice (Cooper et al., 2008; Tsukamoto et al. 2008a; Tsukamoto et al. 2008b). However, an autophagic response due to environmental stress or a known inhibitor has yet to be studied during this crucial developmental time period. It has been well established that mTOR is an upstream repressor of autophagy. Therefore, the mTOR inhibitor rapamycin is widely used to activate autophagy within in vitro systems. In order to investigate the embryonic response in the presence of an autophagic activator, we exposed morula stage embryos (76 h post-hCG) to culture media containing either 1µM or 5µM rapamycin for 30 hours and observed the levels of Beclin-1 and LC-3 expression by immunofluorescence compared to embryos treated with DMSO (vehicle control) (Figure 1A-C). We observed autophagic activation as an increase in the levels of Beclin-1 protein expression in the treated groups. This activation was confirmed by LC3 protein expression, a major component of the autophagosomal membrane (Figure 1D-E). While the increase in autophagic response was expected, there was an unusual variable patterning of response between the single cells within the blastocyst. Beclin-1 protein expression was limited to the trophectoderm of the blastocyst, with the ICM seemingly unaffected (Figure 1C). In addition, we examined the expression of LC3, a well know marker for autophagy, as it is localized to the autophagosomal membrane throughout the bulk of the autophagic process. The expression of LC3 was also detected in a sporadic pattern throughout the embryo, however, this pattern was different than that of Beclin-1 (Figure 1E). This pattern may represent a differential autophagic response in certain cell niches.
Additionally, a cell death assay was used to determine whether the treatment affected cell quality. We found no significant difference in cell death, the percentage of apoptotic cells, between the rapamycin treated and control groups; however there was a difference in total cell number (Supplemental Figure 1A and B). This difference may be due to either a decrease in proliferation or non-apoptotic cell death.
The amount of glucose in the environment is critical to baseline autophagy levels
Due to the importance of autophagy in energy homeostasis in other tissues and the alterations of this pathway in diabetic models, we investigated autophagy in the developing embryo exposed to different glucose levels in vitro.
As an embryo transitions from the morula to blastocyst stage, the amount of environmental glucose is critical as the embryos switches from using pyruvate and lactate as energy to glucose (Brown & Whittingham 1991; Leese 1991). In previous papers, we have determined that the hyperglycemic conditions present in the milieu of diabetic mothers leads to a decrease in glucose transporter expression, deceased intracellular glucose and an increase in apoptosis in murine blastocysts (Moley et al. 1998a; Moley et al. 1998b). Additionally, when embryos were exposed to a hyperglycemic environment for 72 hours and then transplanted to a control pseudo-pregnant foster mouse, an increased number of resorptions and malformations in the fetuses were observed (Wyman et al. 2008). Therefore, the maternal environment and in particular the amount of glucose present is critical to fetal outcome.
To investigate whether the amount of glucose present in the environment alters the basal level of autophagy present in embryos, we exposed the developing embryos to varying concentrations of glucose in vitro during morula to blastocyst transition for 30 hours. We have previously determined that a level of 52mM D-glucose closely mimics the phenotypic effects we observe in the embryos exposed to an in vivo diabetic environment, with similar apoptosis, glucose transporter downregulation and decreased intracellular glucose (Moley et al. 1998a). Following this exposure to high glucose, we then attempted to detect LC3 by immunofluorescence, as a marker for autophagosomes. At more physiological levels (5mM glucose) a minimal level autophagy is observed. However, the levels of LC3 appear to increase as the embryos are cultured in higher concentrations of glucose (Figure 2A-E). We anticipate that the drop in intracellular glucose triggers an autophagic response in a rescue attempt to recycle cellular substrates and generate alternative energy substrates.
To investigate the interchange between autophagy and apoptosis in the developing blastocyst, we repeated the experiment and used a cell death detection assay to determine the level of cell death occurring in each of the conditions. Previously, we have published results indicating an increase in apoptosis in embryos exposed to a hyperglycemic environment in vivo or in vitro (Moley et al. 1998a) . While we observed a similar result, the levels of apoptosis appear to plateau above 20mM glucose concentrations (Figure 2F-J).
Therefore, we conclude that higher levels of cell stress induced by a diabetic milieu result in a dose dependent increase of autophagy, as measured by the autophagosomal marker LC3. Interestingly, the embryos appear to display an internal rheostat that controls the level of apoptosis that occurs. We speculate that this persistence of the cells within the blastocyst that do not undergo apoptosis but display a compensatory increase in autophagy may result in significant changes in these specific cells thus permanently altering the lineages derived from this cell of the blastocyst. These cell specific lineages changes could be responsible for the occurrence of malformations and/or miscarriages of these fetuses.
Changes in GAPDH activity reflect autophagic activation and inhibition
While it is well established that increased GAPDH activity triggers flux through the glycolytic pathway in general and triose metabolism specifically, a recent study has demonstrated a previously unknown role for GAPDH in signaling the activation of autophagy (Colell et al. 2007). By overexpressing GAPDH the investigators were able to protect the cells from caspase-independent cell death and even promote cell survival. Additionally, they showed that increased GAPDH activity leads to an elevation in intracellular ATP as well as upregulation of Atg12, an autophagic protein involved in formation of the autophagosome. We chose GAPDH activity as a metabolic marker of the autophagic response in these embryos.
To test whether GAPDH activity could be used as a marker of autophagy in blastocysts, we measured the activity using microanalytic enzymatic assays (Passonneau & Lowry, 1993) in individual blastocysts exposed to either DMSO as vehicle control or 5µM rapamycin for 30 hours to activate autophagy (Suppl Figure 2A). Activity increased significantly over vehicle. Conversely, blastocysts were exposed to control media vs. media with added 4mM vs. 8mM 3-methyladenine, a known inhibitor of autophagy for 30 hours. Activity of GAPDH was significantly decreased in a dose dependent manner by 3-MA (Suppl Figure 2B). After this confirmation that GAPDH activity could serve as an accurate measure of autophagic activation in the embryos, blastocysts were collected from both in vivo and in vitro diabetic conditions and GAPDH activity was measured in each group (Figure 3A & B). Our results indicate that these diabetic conditions result in an increase in GAPDH activity and we believe this increase signifies an upregulation of autophagy in an attempt to protect the cell from damage as our other results suggest.
Autophagosomes are more abundant in oocytes of diabetic mice
Previously, our lab has published results indicating that in vivo maternal diabetes leads to an increase of apoptosis in the ovarian follicles of mice (Chang et al. 2005). Recently, our lab has also concluded that the mitochondria of diabetic oocytes display an abnormal morphology and distribution leading to a decrease in the metabolites. Furthermore, these oocytes display meiotic spindles defects and chromatin misalignment (Wang et al. 2009). Using GFP-LC3 mice, Mizushima et al. observed a basal level of autophagy occurring at the unfertilized oocyte stage, however, much lower than that of the fertilized oocyte (Tsukamoto et al. 2008a). Therefore, we hypothesize; the metabolic milieu of diabetes may lead to a dysregulation of autophagy in the oocytes of these mice.
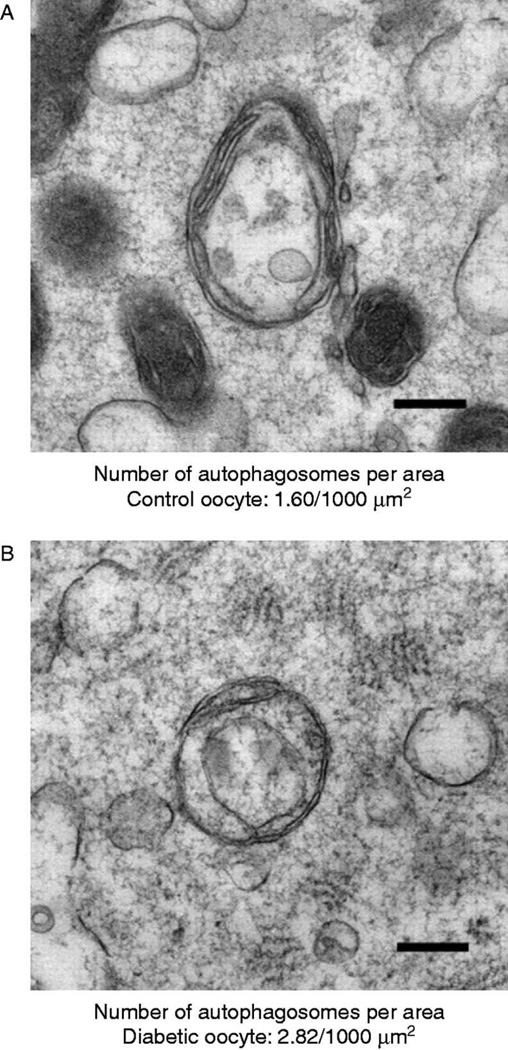
By using transmission electron microscopy we were able to obtain a more accurate estimation of the prevalence of autophagy during this stage (Figure 4). Our results indicate the presence of a basal level autophagosomes in control oocytes. The numbers of autophagosomes per area increase in oocytes obtained from diabetic mice. This increase suggests an increased activation of the autophagic pathway.
Figure 4. A representative autophagosome in a control (left) and diabetic (right) oocyte.
There is a 43% increase in number of autophagosomes in the diabetic oocyte per area (sample size=100, 10 sections from each group quantitated).
Discussion
While it has been determined that genetic removal of key players in the autophagic pathway leads to developmental arrest and embryonic malformations (Cecconi et al. 2008; Fimia et al. 2007; Zheng et al. 2006), the dysregulation of this pathway in response to embryonic environmental stressors has yet to be elucidated. This study suggests that an autophagic response to the environmental stress of a hyperglycemic milieu occurs during the oocyte and blastocyst stages of development and that this event is heterogeneously triggered at the blastocyst stage with some cells undergoing cell death and others surviving, but significantly and perhaps permanently altered by the autophagic process. We speculate that these remaining cells may alter their fate, thus changing the originally assigned cell lineages and possibly having developmental consequences.
By exposing the embryos to an autophagic activator, rapamycin, we were able to observe an increase in autophagic proteins, Beclin-1 and LC3 as indicators of an increased activation of autophagy. Interestingly, the embryos display a heterogeneous autophagic response to culture conditions. This differential display of activation of a molecular pathway in embryos has been detailed before, however, this was in regards to apoptosis (Pampfer 2000). In those studies, it was suggested that the inner cell mass (ICM), which will develop into the embryo, and the trophectoderm (TE), which develops into the extraembryonic tissues including the placenta, consist of separate “micro-environments.” This internal separation establishes differential gene expression, different levels of ion and glucose transport and a varying response to cytotoxic agents (Pampfer 2000). Furthermore, cells within each of these compartments display a disparity. It has been reported, using cell lines derived from either the ICM or the trophectoderm, that the trophectoderm is more viable in a hyperglycemic environment, whereas, the ICM is more susceptible to hyperglycemic environments, leading to an increase incidence in apoptosis (Pampfer 2000). This is recapitulated in vivo as the number of cells in the ICM of embryos exposed to maternal hyperglycemia is reduced compared to controls (Lea et al. 1996; Pampfer et al. 1997).
In this study we show a different pattern of expression than apoptosis. Although, the expression of Beclin-1 appear exclusively in the trophectoderm, the autophagic activation by LC3 is seen in random individual cells of the blastocyst. Murine embryos inherit a pool of maternal transcripts that are progressively degraded and replaced by the products of embryonic transcription. This embryonic genomic activation is triggered when the blastomeres still exhibit developmental plasticity and can change their cell fate (Zernicka-Goetz et al. 2009). It is possible that this differential autophagic response in single cells in the embryo during pre-implantation development affects the subsequent lineage allocation in that cell and its resulting daughter cells. We postulate that this early change in lineage patterning may result not only in the growth abnormalities of infants and placentas, but also in malformations commonly seen in fetuses from diabetic mothers.
While the effect of a maternal hyperglycemic environment on embryo development has been studied, much is still left unanswered. Our lab has previously published data establishing a result of increased apoptosis in murine blastocysts exposed to a hyperglycemic diabetic environment, in vivo or in vitro (Moley et al. 1998a). Furthermore, when transferred into pseudo-pregnant females, these exposed blastocysts display an increase rate of resorptions and malformations (Wyman et al. 2008). Surprisingly, one cell zygotes when transferred from diabetic to nondiabetic mice also demonstrated a significantly higher rate of malformations and growth retardation, suggesting an even earlier period of vulnerability of the zygote to hyperglycemia. More recent studies by our lab have confirmed this hypothesis. Recently, we determined that GV and MII oocytes from diabetic mice have biochemical and meiotic abnormalities which could predispose them to developmental problems post fertilization and perhaps implantation (Ratchford et al. 2007; Wang et al., 2009). We established that oocytes from diabetic mice have significantly lower ATP levels at both the GV and MII stages (Ratchford et al. 2007). In addition, we demonstrate a significantly higher number of autophagosomes in GV stage oocytes by electron microscopy (Wang et al. 2009). Other groups have shown that the autophagic protein LC3 is not detected until after fertilization (Tsukamoto et al. 2008a), however, we show a significant difference in activation in the unfertilized oocyte from diabetic mice. This raises the possibility that maternal proteins at this very early stage may be prematurely degraded by autophagy, resulting in oocytes deficient in protein and perhaps predisposed to increased autophagy and other abnormalities, as we see in the blastocysts from diabetic mice. Future studies will be designed to test this hypothesis.
Recent studies have implicated GAPDH as a possible mediator and biochemical indicator of autophagic activation. Although the mechanism is not entirely elucidated, those studies demonstrated that overexpressing GAPDH in cells induced to undergo autophagy, prevented this process. The conclusions were that GAPDH activity not only triggered metabolism of trioses with in the glycolytic pathway but also, directly or indirectly, induced an increase in the transcription of Atg12, a key autophagic protein. Our study demonstrated a significant increase in GAPDH activity within the whole blastocyst in response to maternal diabetes or in vitro high glucose. In addition, we confirmed that changes in this enzyme activity are predictive of either activation of autophagy by rapamycin or inhibition of autophagy by 3-methyladenine.
In conclusion, we show in this paper that the levels of autophagy are altered during oocyte and pre-implantation development in response to environmental stressors (Figure 5). With the addition of the autophagy activator rapamycin, levels of autophagy are increased in a cell specific manner. Additionally, embryos cultured in a hyperglycemic environment, mimicking maternal diabetes result in the elevation of the autophagic pathway. This elevation is also seen as an increase in GAPDH activity, which has recently been suggested as an activator of the autophagic pathway. Furthermore, it appears that the autophagic pathway may be altered as early as the oocytes as a result of maternal diabetes. This early alteration in autophagy may result in changes in quality of the embryo after fertilization. However, further studies will need to be conducted to validate this hypothesis. Overall, our data suggests that the hyperglycemic environment leads to an increased activation of autophagy during the oocyte growth phase and pre-implantation development resulting in a differential response by the individual cells within the embryo.
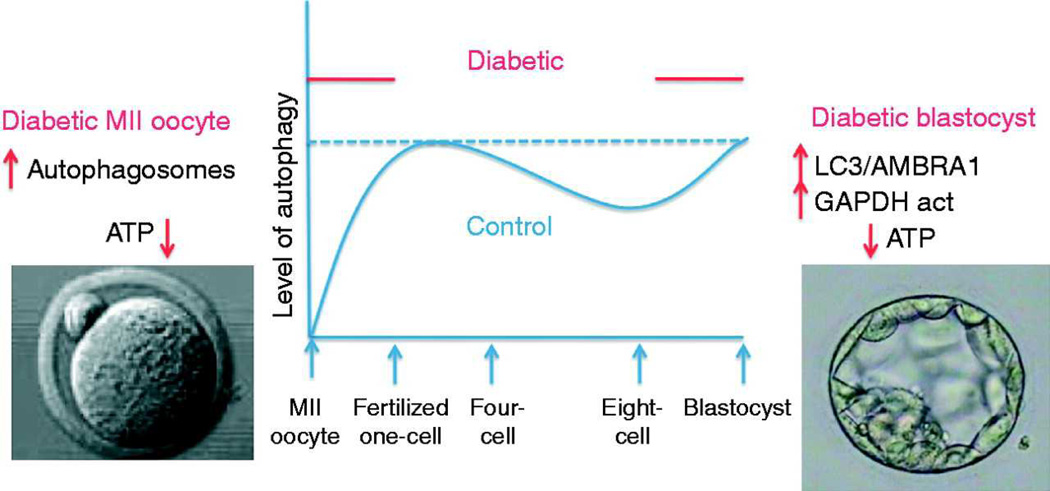
Figure 5. Cartoon of the level of autophagy thought to exist in control vs. diabetic oocytes and embryos.
Whereas, no autophagy was detected in MII oocytes from control mice (Tsukamoto et al, 2008), we determined increased autophagosomes (in this work) and decreased ATP in MII oocytes from diabetic mice (Ratchford et al, 2007). In addition, although some autophagic proteins have been detected in control blastocysts, diabetic blastocysts demonstrated increased LC3 and Beclin-1 protein (this study) as well as decreased ATP (Chi et al, 2002). The sinusoidal pattern of autophagy is an estimation based on prior work as well as this study. Furthermore, we speculate that increased autophagy at the oocyte stage may prematurely degrade maternal proteins leading to developmental arrest and increased autophagy at the blastocyst stage may lead to abnormal degradation of embryonic proteins essential for proper development.
Supplementary Material
Acknowledgements
The authors would like to acknowledge Wandy Beatty in the Molecular Microbiology Imaging Facility at Washington University in St. Louis for the transmission electron microscopy imaging and The Bakewell Neuroimaging Laboratory for the use of the confocal microscopy for imaging.
Grant support: KLA: National Institute of Health (T32 GM07067), KHM: National Institute of Health (R01 HD40390).
Footnotes
Disclosure summary: The authors of this manuscript have nothing to disclose.
References
- Akazawa H, Komazaki S, Shimomura H, Terasaki F, Zou Y, Takano H, Nagai T, Komuro I. Diphtheria toxin-induced autophagic cardiomyocyte death plays a pathogenic role in mouse model of heart failure. J Biol Chem. 2004;279:41095–41103. doi: 10.1074/jbc.M313084200. [DOI] [PubMed] [Google Scholar]
- Aki T, Yamaguchi K, Fujimiya T, Mizukami Y. Phosphoinositide 3-kinase accelerates autophagic cell death during glucose deprivation in the rat cardiomyocyte-derived cell line H9c2. Oncogene. 2003;22:8529–8535. doi: 10.1038/sj.onc.1207197. [DOI] [PubMed] [Google Scholar]
- Amherdt M, Harris V, Renold AE, Orci L, Unger RH. Hepatic autography in uncontrolled experimental diabetes and its relationships to insulin and glucagon. J Clin Invest. 1974;54:188–193. doi: 10.1172/JCI107742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boya P, Gonzalez-Polo RA, et al. Inhibition of macroautophagy triggers apoptosis. Mol Cell Biol. 2005;25:1025–1040. doi: 10.1128/MCB.25.3.1025-1040.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown JJ, Whittingham DG. The roles of pyruvate, lactate and glucose during preimplantation development of embryos from F1 hybrid mice in vitro. Development. 1991;112:99–105. doi: 10.1242/dev.112.1.99. [DOI] [PubMed] [Google Scholar]
- Cecconi F, Piacentini M, Fimia GM. The involvement of cell death and survival in neural tube defects: a distinct role for apoptosis and autophagy? Cell Death Differ. 2008;15:1170–1177. doi: 10.1038/cdd.2008.64. [DOI] [PubMed] [Google Scholar]
- Chang AS, Dale AN, Moley KH. Maternal diabetes adversely affects preovulatory oocyte maturation, development, and granulosa cell apoptosis. Endocrinology. 2005;146:2445–2453. doi: 10.1210/en.2004-1472. [DOI] [PubMed] [Google Scholar]
- Chi MM, Hoehn A, Moley KH. Metabolic changes in the glucose-induced apoptotic blastocyst suggest alterations in mitochondrial physiology. Am J Physiol Endocrinol Metab. 2002;283:E226–E232. doi: 10.1152/ajpendo.00046.2002. [DOI] [PubMed] [Google Scholar]
- Colell A, Ricci JE, et al. GAPDH and autophagy preserve survival after apoptotic cytochrome c release in the absence of caspase activation. Cell. 2007;129:983–997. doi: 10.1016/j.cell.2007.03.045. [DOI] [PubMed] [Google Scholar]
- Cooper ASR, Boehle K, Riley J, Moley K. Autophagy is a physiologic process regulated by glucose availability in the murine preimplantation blastocyst. Reproductive Sciences (Supplement) 2008:73A. [Google Scholar]
- Fimia GM, Stoykova A, et al. Ambra1 regulates autophagy and development of the nervous system. Nature. 2007;447:1121–1125. doi: 10.1038/nature05925. [DOI] [PubMed] [Google Scholar]
- Fujitani Y, Kawamori R, Watada H. The role of autophagy in pancreatic beta-cell and diabetes. Autophagy. 2009;5:280–282. doi: 10.4161/auto.5.2.7656. [DOI] [PubMed] [Google Scholar]
- Grune T, Merker K, Sandig G, Davies KJ. Selective degradation of oxidatively modified protein substrates by the proteasome. Biochem Biophys Res Commun. 2003;305:709–718. doi: 10.1016/s0006-291x(03)00809-x. [DOI] [PubMed] [Google Scholar]
- Gualtieri R, Iaccarino M, Mollo V, Prisco M, Iaccarino S, Talevi R. Slow cooling of human oocytes: ultrastructural injuries and apoptotic status. Fertil Steril. 2009;91:1023–1034. doi: 10.1016/j.fertnstert.2008.01.076. [DOI] [PubMed] [Google Scholar]
- Jungheim ES, Schoeller EL, Marquard KL, Louden ED, Schaffer JE, Moley KH. Diet-induced obesity model: abnormal oocytes and persistent growth abnormalities in the offspring. Endocrinology. 151:4039–4046. doi: 10.1210/en.2010-0098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiffin R, Bandyopadhyay U, Cuervo AM. Oxidative stress and autophagy. Antioxid Redox Signal. 2006;8:152–162. doi: 10.1089/ars.2006.8.152. [DOI] [PubMed] [Google Scholar]
- Lea RG, McCracken JE, McIntyre SS, Smith W, Baird JD. Disturbed development of the preimplantation embryo in the insulin-dependent diabetic BB/E rat. Diabetes. 1996;45:1463–1470. doi: 10.2337/diab.45.11.1463. [DOI] [PubMed] [Google Scholar]
- Leese HJ. Metabolism of the preimplantation mammalian embryo. Oxf Rev Reprod Biol. 1991;13:35–72. [PubMed] [Google Scholar]
- Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol. 2007;8:741–752. doi: 10.1038/nrm2239. [DOI] [PubMed] [Google Scholar]
- Masini M, Bugliani M, Lupi R, del Guerra S, Boggi U, Filipponi F, Marselli L, Masiello P, Marchetti P. Autophagy in human type 2 diabetes pancreatic beta cells. Diabetologia. 2009;52:1083–1086. doi: 10.1007/s00125-009-1347-2. [DOI] [PubMed] [Google Scholar]
- Mizushima N, Yamamoto A, Matsui M, Yoshimori T, Ohsumi Y. In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker. Mol Biol Cell. 2004;15:1101–1111. doi: 10.1091/mbc.E03-09-0704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moley KH, Chi MM, Knudson CM, Korsmeyer SJ, Mueckler MM. Hyperglycemia induces apoptosis in pre-implantation embryos through cell death effector pathways. Nat Med. 1998a;4:1421–1424. doi: 10.1038/4013. [DOI] [PubMed] [Google Scholar]
- Moley KH, Chi MM, Mueckler MM. Maternal hyperglycemia alters glucose transport and utilization in mouse preimplantation embryos. Am J Physiol. 1998b;275:E38–E47. doi: 10.1152/ajpendo.1998.275.1.E38. [DOI] [PubMed] [Google Scholar]
- Pampfer S. Apoptosis in rodent peri-implantation embryos: differential susceptibility of inner cell mass and trophectoderm cell lineages--a review. Placenta. 2000;21 Suppl A:S3–S10. doi: 10.1053/plac.1999.0519. [DOI] [PubMed] [Google Scholar]
- Pampfer S, Vanderheyden I, McCracken JE, Vesela J, De Hertogh R. Increased cell death in rat blastocysts exposed to maternal diabetes in utero and to high glucose or tumor necrosis factor-alpha in vitro. Development. 1997;124:4827–4836. doi: 10.1242/dev.124.23.4827. [DOI] [PubMed] [Google Scholar]
- Passonneau JL, OH . Enzymatic Analysis. Totowa, NJ: Humana Press; 1993. [Google Scholar]
- Qu X, Zou Z, Sun Q, Luby-Phelps K, Cheng P, Hogan RN, Gilpin C, Levine B. Autophagy gene-dependent clearance of apoptotic cells during embryonic development. Cell. 2007;128:931–946. doi: 10.1016/j.cell.2006.12.044. [DOI] [PubMed] [Google Scholar]
- Ratchford AM, Chang AS, Chi MM, Sheridan R, Moley KH. Maternal diabetes adversely affects AMP-activated protein kinase activity and cellular metabolism in murine oocytes. Am J Physiol Endocrinol Metab. 2007;293:E1198–E1206. doi: 10.1152/ajpendo.00097.2007. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Enriquez S, He L, Lemasters JJ. Role of mitochondrial permeability transition pores in mitochondrial autophagy. Int J Biochem Cell Biol. 2004;36:2463–2472. doi: 10.1016/j.biocel.2004.04.009. [DOI] [PubMed] [Google Scholar]
- Sakaida I, Kyle ME, Farber JL. Autophagic degradation of protein generates a pool of ferric iron required for the killing of cultured hepatocytes by an oxidative stress. Mol Pharmacol. 1990;37:435–442. [PubMed] [Google Scholar]
- Scott RC, Schuldiner O, Neufeld TP. Role and regulation of starvation-induced autophagy in the Drosophila fat body. Dev Cell. 2004;7:167–178. doi: 10.1016/j.devcel.2004.07.009. [DOI] [PubMed] [Google Scholar]
- Singh R, Xiang Y, et al. Autophagy regulates adipose mass and differentiation in mice. J Clin Invest. 2009;119:3329–3339. doi: 10.1172/JCI39228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sooparb S, Price SR, Shaoguang J, Franch HA. Suppression of chaperone-mediated autophagy in the renal cortex during acute diabetes mellitus. Kidney Int. 2004;65:2135–2144. doi: 10.1111/j.1523-1755.2004.00639.x. [DOI] [PubMed] [Google Scholar]
- Thorburn A. Apoptosis and autophagy: regulatory connections between two supposedly different processes. Apoptosis. 2008;13:1–9. doi: 10.1007/s10495-007-0154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towns R, Kabeya Y, Yoshimori T, Guo C, Shangguan Y, Hong S, Kaplan M, Klionsky DJ, Wiley JW. Sera from patients with type 2 diabetes and neuropathy induce autophagy and colocalization with mitochondria in SY5Y cells. Autophagy. 2005;1:163–170. doi: 10.4161/auto.1.3.2068. [DOI] [PubMed] [Google Scholar]
- Tsukamoto S, Kuma A, Mizushima N. The role of autophagy during the oocyte-to-embryo transition. Autophagy. 2008a;4:1076–1078. doi: 10.4161/auto.7065. [DOI] [PubMed] [Google Scholar]
- Tsukamoto S, Kuma A, Murakami M, Kishi C, Yamamoto A, Mizushima N. Autophagy is essential for preimplantation development of mouse embryos. Science. 2008b;321:117–120. doi: 10.1126/science.1154822. [DOI] [PubMed] [Google Scholar]
- Wang Q, Ratchford AM, Chi MM, Schoeller E, Frolova A, Schedl T, Moley KH. Maternal diabetes causes mitochondrial dysfunction and meiotic defects in murine oocytes. Mol Endocrinol. 2009;23:1603–1612. doi: 10.1210/me.2009-0033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyman A, Pinto AB, Sheridan R, Moley KH. One-cell zygote transfer from diabetic to nondiabetic mouse results in congenital malformations and growth retardation in offspring. Endocrinology. 2008;149:466–469. doi: 10.1210/en.2007-1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yue Z, Jin S, Yang C, Levine AJ, Heintz N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci U S A. 2003;100:15077–15082. doi: 10.1073/pnas.2436255100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zernicka-Goetz M, Morris SA, Bruce AW. Making a firm decision: multifaceted regulation of cell fate in the early mouse embryo. Nat Rev Genet. 2009;10:467–477. doi: 10.1038/nrg2564. [DOI] [PubMed] [Google Scholar]
- Zheng B, Tang T, Tang N, Kudlicka K, Ohtsubo K, Ma P, Marth JD, Farquhar MG, Lehtonen E. Essential role of RGS-PX1/sorting nexin 13 in mouse development and regulation of endocytosis dynamics. Proc Natl Acad Sci U S A. 2006;103:16776–16781. doi: 10.1073/pnas.0607974103. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.