Abstract
Blood phagocytes of the dogfish Mustelus canis attack oocytes of the sea urchin Arbacia punctulata, first provoking a surrogate fertilization response and then killing the eggs. To test the hypothesis that secretion of lysosomal contents is critical in this model of phagocyte-mediated cell injury, we studied effects of agents that modify lysosomal enzyme secretion. Inhibitors of membrane transport (>0.1 mM) inhibited postphagocytic secretion of lysosomal β-glucuronidase from dogfish phagocytes: phloretin > ethacrynate > furosemide > 4-acetamido-4′-isothiocyanostilbene-2,2′-disulfonic acid >> pyridoxal phosphate > ouabain. The same order of activity was found for inhibition by these agents of killing of Arbacia eggs by phagocytes. Cell activation (fertilization response) and cytotoxicity were quantitated both morphologically and by measurements of enzyme (β-glucuronidase, catalase) release. The agents neither inhibited fertilization responses of eggs to calcium ionophore A23187 nor impaired their viability. Vital staining demonstrated that ethacrynate prevented phagocytes from degranulating upon contact with zymosan particles. The data not only suggest that agents primarily known for their capacity to inhibit membrane transport systems can inhibit lysosomal enzyme secretion from phagocytes but also support the hypothesis that secretion of lysosomal contents mediates activation and killing of target cells in phagocyte-mediated tissue injury.
Keywords: cytotoxicity, degranulation, ethacrynic acid, furosemide, phloretin
Full text
PDF

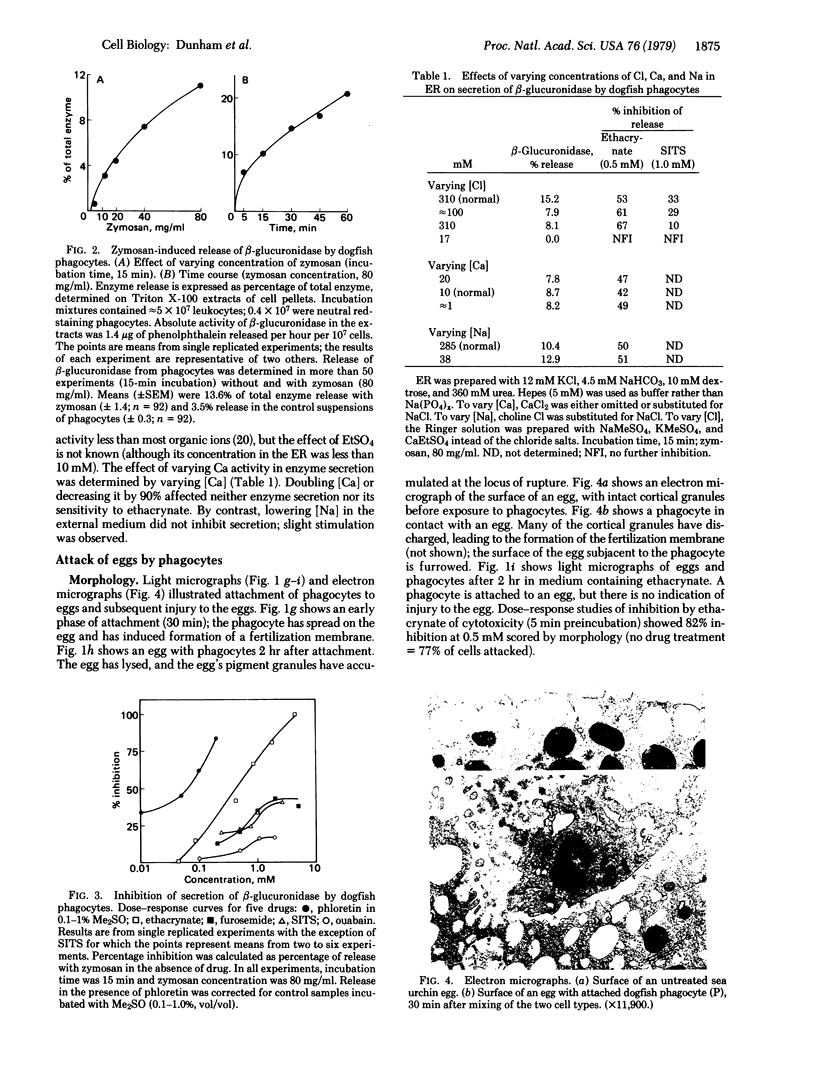


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersen O. S., Finkelstein A., Katz I., Cass A. Effect of phloretin on the permeability of thin lipid membranes. J Gen Physiol. 1976 Jun;67(6):749–771. doi: 10.1085/jgp.67.6.749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BEERS R. F., Jr, SIZER I. W. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem. 1952 Mar;195(1):133–140. [PubMed] [Google Scholar]
- Brazy P. C., Gunn R. B. Furosemide inhibition of chloride transport in human red blood cells. J Gen Physiol. 1976 Dec;68(6):583–599. doi: 10.1085/jgp.68.6.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown E. M., Pazoles C. J., Creutz C. E., Aurbach G. D., Pollard H. B. Role of anions in parathyroid hormone release from dispersed bovine parathyroid cells. Proc Natl Acad Sci U S A. 1978 Feb;75(2):876–880. doi: 10.1073/pnas.75.2.876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabantchik I. Z., Balshin M., Breuer W., Rothstein A. Pyridoxal phosphate. An anionic probe for protein amino groups exposed on the outer and inner surfaces of intact human red blood cells. J Biol Chem. 1975 Jul 10;250(13):5130–5136. [PubMed] [Google Scholar]
- Christoffersen C. R., Skibsted L. H. Calcium ion activity in physiological salt solutions: influence of anions substituted for chloride. Comp Biochem Physiol A Comp Physiol. 1975 Oct 1;52(2):317–322. doi: 10.1016/s0300-9629(75)80094-6. [DOI] [PubMed] [Google Scholar]
- Gale R. P., Zighelboim J. Polymorphonuclear leukocytes in antibody-dependent cellular cytotoxicity. J Immunol. 1975 Mar;114(3):1047–1051. [PubMed] [Google Scholar]
- Goldstein I. M., Hoffstein S. T., Weissmann G. Influence of divalent cations upon complement-mediated enzyme release from human polymorphonuclear leukocytes. J Immunol. 1975 Sep;115(3):665–670. [PubMed] [Google Scholar]
- Goldstein I. M., Kaplan H. B., Radin A., Frosch M. Independent effects of IgG and complement upon human polymorphonuclear leukocyte function. J Immunol. 1976 Oct;117(4):1282–1287. [PubMed] [Google Scholar]
- Henson P. M. Interaction of cells with immune complexes: adherence, release of constituents, and tissue injury. J Exp Med. 1971 Sep 1;134(3 Pt 2):114s–135s. [PubMed] [Google Scholar]
- Hoffstein S., Weissmann G. Mechanisms of lysosomal enzyme release from leukocytes. IV. Interaction of monosodium urate crystals with dogfish and human leukocytes. Arthritis Rheum. 1975 Mar-Apr;18(2):153–165. doi: 10.1002/art.1780180213. [DOI] [PubMed] [Google Scholar]
- Knauf P. A., Rothstein A. Chemical modification of membranes. II. Permeation paths for sulfhydryl agents. J Gen Physiol. 1971 Aug;58(2):211–223. doi: 10.1085/jgp.58.2.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korchak H. M., Weissmann G. Changes in membrane potential of human granulocytes antecede the metabolic responses to surface stimulation. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3818–3822. doi: 10.1073/pnas.75.8.3818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magro A. M. Ethacrynic acid inhibitable Ca2+ and Mg2+-activated membrane adenosine triphosphatase in rat mast cells. Clin Exp Immunol. 1977 Oct;30(1):160–167. [PMC free article] [PubMed] [Google Scholar]
- Motais R., Cousin J. L. Inhibitory effect of ethacrynic acid on chloride permeability. Am J Physiol. 1976 Nov;231(5 Pt 1):1485–1489. doi: 10.1152/ajplegacy.1976.231.5.1485. [DOI] [PubMed] [Google Scholar]
- Pazoles C. J., Pollard H. B. Evidence for stimulation of anion transport in ATP-evoked transmitter release from isolated secretory vesicles. J Biol Chem. 1978 Jun 10;253(11):3962–3969. [PubMed] [Google Scholar]
- Pollard H. B., Tack-Goldman K., Pazoles C. J., Creutz C. E., Shulman N. R. Evidence for control of serotonin secretion from human platelets by hydroxyl ion transport and osmotic lysis. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5295–5299. doi: 10.1073/pnas.74.12.5295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz A., Lindenmayer G. E., Allen J. C. The sodium-potassium adenosine triphosphatase: pharmacological, physiological and biochemical aspects. Pharmacol Rev. 1975 Mar;27(01):3–134. [PubMed] [Google Scholar]
- Showell H. J., Naccache P. H., Sha'afi R. I., Becker E. L. The effects of extracellular K+, Na+ and Ca++ on lysosomal enzyme secretion from polymorphonuclear leukocytes. J Immunol. 1977 Sep;119(3):804–811. [PubMed] [Google Scholar]
- Simchowitz L., Schur P. H. Lectin-dependent neutrophil-mediated cytotoxicity. II. Possible mechanisms. Immunology. 1976 Aug;31(2):313–322. [PMC free article] [PubMed] [Google Scholar]
- ULMER D. D., VALLEE B. L., WACKER W. E. Metalloenzymes and myocardial infarction. II. Malic and lactic dehydrogenase activities and zinc concentrations in serum. N Engl J Med. 1956 Sep 6;255(10):450–456. doi: 10.1056/NEJM195609062551001. [DOI] [PubMed] [Google Scholar]
- Vittoria A., Laghi Pasini F., De Gori V., Forconi S., Di Perri T. [Action of furosemide on vitro in platelet aggregation by ADP, adrenaline or collagen]. Boll Soc Ital Biol Sper. 1974 Sep 15;50(17):1391–1396. [PubMed] [Google Scholar]
- Weissmann G., Bloomgarden D., Kaplan R., Cohen C., Hoffstein S., Collins T., Gotlieb A., Nagle D. A general method for the introduction of enzymes, by means of immunoglobulin-coated liposomes, into lysosomes of deficient cells. Proc Natl Acad Sci U S A. 1975 Jan;72(1):88–92. doi: 10.1073/pnas.72.1.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissmann G., Finkelstein M. C., Csernansky J., Quigley J. P., Quinn R. S., Techner L., Troll W., Dunham P. B. Attack of sea urchin eggs by dogfish phagocytes: model of phagocyte-mediated cellular cytotoxicity. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1825–1829. doi: 10.1073/pnas.75.4.1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissmann G., Zurier R. B., Spieler P. J., Goldstein I. M. Mechanisms of lysosomal enzyme release from leukocytes exposed to immune complexes and other particles. J Exp Med. 1971 Sep 1;134(3 Pt 2):149s–165s. [PubMed] [Google Scholar]




