Abstract
ADP-ribosylation of proteins was analyzed by in vivo labeling of cells with [3H]adenosine, followed by separation of their protein components by two-dimensional isoelectric focusing/NaDodSO4 polyacrylamide gel electrophoresis. We show here that in several cell types of avian and mammalian origin the major [34H]adenosine acceptor in vivo is a polypeptide with a Mr of 83,000 and isoelectric point of approximately equal to 5.3. This polypeptide is identical to one of the stress-inducible and glucose-regulated proteins (here called SP83) previously described in avian and mammalian cells. Snake venom phosphodiesterase digestion of purified 3H-labeled SP83 releases 5'-AMP and a minor fraction of 2'-(5"-phosphoribosyl)-5-AMP. In vitro labeling with [32P]NAD+ of total cell lysates made in the presence of non-ionic detergents also results in incorporation of radioactivity into SP83. Both of these results strongly suggest that the modification is an ADP-ribosylation. Heat shock and glucose starvation of cells induce a rapid and extensive decrease in the incorporation of ADP-ribose into SP83, suggesting that ADP-ribosylation may be important for the regulation of the function of this protein.
Full text
PDF

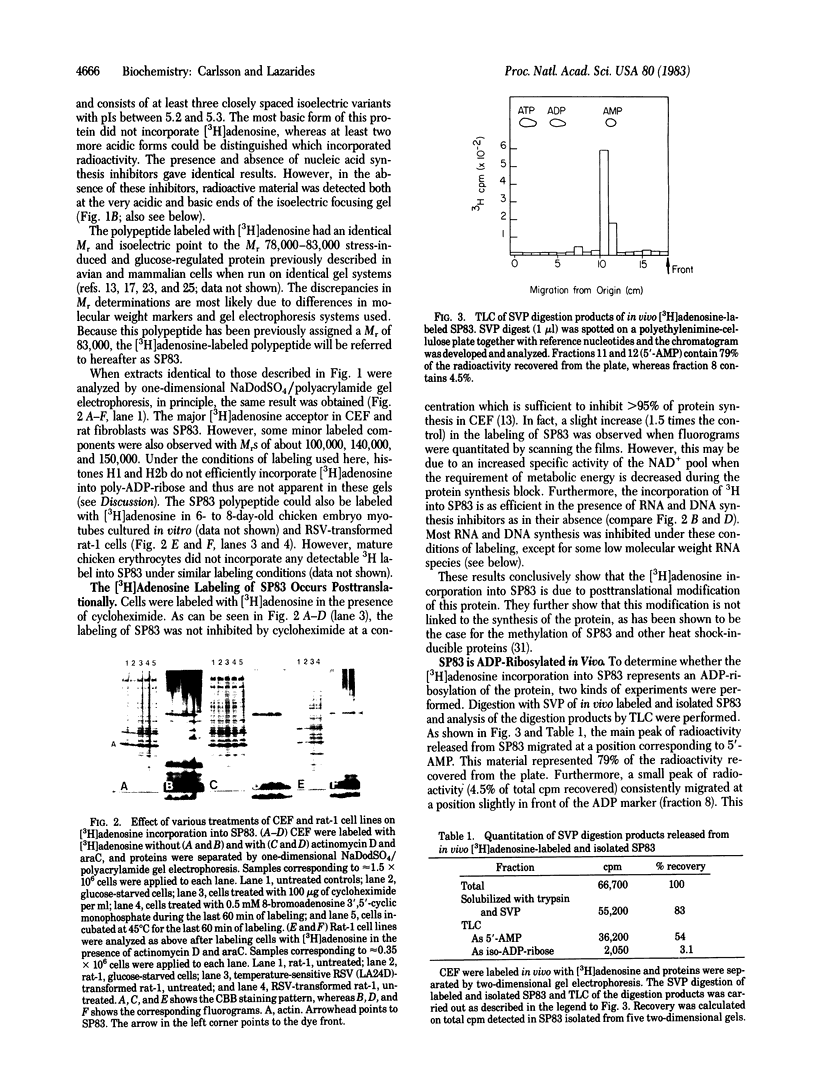


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abood M. E., Hurley J. B., Pappone M. C., Bourne H. R., Stryer L. Functional homology between signal-coupling proteins. Cholera toxin inactivates the GTPase activity of transducin. J Biol Chem. 1982 Sep 25;257(18):10540–10543. [PubMed] [Google Scholar]
- Amir-Zaltsman Y., Ezra E., Scherson T., Zutra A., Littauer U. Z., Salomon Y. ADP-ribosylation of microtubule proteins as catalyzed by cholera toxin. EMBO J. 1982;1(2):181–186. doi: 10.1002/j.1460-2075.1982.tb01144.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashburner M., Bonner J. J. The induction of gene activity in drosophilia by heat shock. Cell. 1979 Jun;17(2):241–254. doi: 10.1016/0092-8674(79)90150-8. [DOI] [PubMed] [Google Scholar]
- Bochner B. R., Ames B. N. Complete analysis of cellular nucleotides by two-dimensional thin layer chromatography. J Biol Chem. 1982 Aug 25;257(16):9759–9769. [PubMed] [Google Scholar]
- Cassel D., Pfeuffer T. Mechanism of cholera toxin action: covalent modification of the guanyl nucleotide-binding protein of the adenylate cyclase system. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2669–2673. doi: 10.1073/pnas.75.6.2669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill D. M., Meren R. ADP-ribosylation of membrane proteins catalyzed by cholera toxin: basis of the activation of adenylate cyclase. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3050–3054. doi: 10.1073/pnas.75.7.3050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goff C. G. Chemical structure of a modification of the Escherichia coli ribonucleic acid polymerase alpha polypeptides induced by bacteriophage T4 infection. J Biol Chem. 1974 Oct 10;249(19):6181–6190. [PubMed] [Google Scholar]
- Goldman N., Brown M., Khoury G. Modification of SV40 T antigen by poly ADP-ribosylation. Cell. 1981 May;24(2):567–572. doi: 10.1016/0092-8674(81)90347-0. [DOI] [PubMed] [Google Scholar]
- Hightower L. E., White F. P. Cellular responses to stress: comparison of a family of 71--73-kilodalton proteins rapidly synthesized in rat tissue slices and canavanine-treated cells in culture. J Cell Physiol. 1981 Aug;108(2):261–275. doi: 10.1002/jcp.1041080216. [DOI] [PubMed] [Google Scholar]
- Honjo T., Nishizuka Y., Hayaishi O. Diphtheria toxin-dependent adenosine diphosphate ribosylation of aminoacyl transferase II and inhibition of protein synthesis. J Biol Chem. 1968 Jun 25;243(12):3553–3555. [PubMed] [Google Scholar]
- Hubbard B. D., Lazarides E. Copurification of actin and desmin from chicken smooth muscle and their copolymerization in vitro to intermediate filaments. J Cell Biol. 1979 Jan;80(1):166–182. doi: 10.1083/jcb.80.1.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaslow H. R., Groppi V. E., Abood M. E., Bourne H. R. Cholera toxin can catalyze ADP-ribosylation of cytoskeletal proteins. J Cell Biol. 1981 Nov;91(2 Pt 1):410–413. doi: 10.1083/jcb.91.2.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelley P. M., Schlesinger M. J. Antibodies to two major chicken heat shock proteins cross-react with similar proteins in widely divergent species. Mol Cell Biol. 1982 Mar;2(3):267–274. doi: 10.1128/mcb.2.3.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelley P. M., Schlesinger M. J. The effect of amino acid analogues and heat shock on gene expression in chicken embryo fibroblasts. Cell. 1978 Dec;15(4):1277–1286. doi: 10.1016/0092-8674(78)90053-3. [DOI] [PubMed] [Google Scholar]
- Khandjian E. W., Türler H. Simian virus 40 and polyoma virus induce synthesis of heat shock proteins in permissive cells. Mol Cell Biol. 1983 Jan;3(1):1–8. doi: 10.1128/mcb.3.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konigsberg I. R. Skeletal myoblasts in culture. Methods Enzymol. 1979;58:511–527. doi: 10.1016/s0076-6879(79)58166-x. [DOI] [PubMed] [Google Scholar]
- Kun E., Zimber P. H., Chang A. C., Puschendorf B., Grunicke H. Macromolecular enzymatic product of NAD+ in liver mitochondria. Proc Natl Acad Sci U S A. 1975 Apr;72(4):1436–1440. doi: 10.1073/pnas.72.4.1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee A. S., Delegeane A. M., Baker V., Chow P. C. Transcriptional regulation of two genes specifically induced by glucose starvation in a hamster mutant fibroblast cell line. J Biol Chem. 1983 Jan 10;258(1):597–603. [PubMed] [Google Scholar]
- Lee A. S., Delegeane A., Scharff D. Highly conserved glucose-regulated protein in hamster and chicken cells: preliminary characterization of its cDNA clone. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4922–4925. doi: 10.1073/pnas.78.8.4922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee A. S. The accumulation of three specific proteins related to glucose-regulated proteins in a temperature-sensitive hamster mutant cell line K12. J Cell Physiol. 1981 Jan;106(1):119–125. doi: 10.1002/jcp.1041060113. [DOI] [PubMed] [Google Scholar]
- Melero J. A. Identification of the glucose/glycosylation-regulated proteins as those which accumulate in the temperature-sensitive cell line K12. J Cell Physiol. 1981 Oct;109(1):59–67. doi: 10.1002/jcp.1041090108. [DOI] [PubMed] [Google Scholar]
- Moss J., Stanley S. J., Watkins P. A. Isolation and properties of an NAD- and guanidine-dependent ADP-ribosyltransferase from turkey erythrocytes. J Biol Chem. 1980 Jun 25;255(12):5838–5840. [PubMed] [Google Scholar]
- Nevins J. R. Induction of the synthesis of a 70,000 dalton mammalian heat shock protein by the adenovirus E1A gene product. Cell. 1982 Jul;29(3):913–919. doi: 10.1016/0092-8674(82)90453-6. [DOI] [PubMed] [Google Scholar]
- Okayama H., Ueda K., Hayaishi O. Purification of ADP-ribosylated nuclear proteins by covalent chromatography on dihydroxyboryl polyacrylamide beads and their characterization. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1111–1115. doi: 10.1073/pnas.75.3.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raeburn S., Collins J. F., Moon H. M., Maxwell E. S. Aminoacyltransferase II from rat liver. I. Purification and enzymatic properties. J Biol Chem. 1971 Feb 25;246(4):1041–1048. [PubMed] [Google Scholar]
- Rein A., Rubin H. Effects of local cell concentrations upon the growth of chick embryo cells in tissue culture. Exp Cell Res. 1968 Mar;49(3):666–678. doi: 10.1016/0014-4827(68)90213-9. [DOI] [PubMed] [Google Scholar]
- Roberts J. H., Stard P., Giri C. P., Smulson M. Cytoplasmic poly(ADP-ribose) polymerase during the HeLa cell cycle. Arch Biochem Biophys. 1975 Nov;171(1):305–315. doi: 10.1016/0003-9861(75)90037-5. [DOI] [PubMed] [Google Scholar]
- Steimer K. S., Klagsbrun M. Serum-free growth of normal and transformed fibroblasts in milk: differential requirements for fibronectin. J Cell Biol. 1981 Feb;88(2):294–300. doi: 10.1083/jcb.88.2.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueda K., Omachi A., Kawaichi M., Hayaishi O. Natural occurrence of poly(ADP-ribosyl) histones in rat liver. Proc Natl Acad Sci U S A. 1975 Jan;72(1):205–209. doi: 10.1073/pnas.72.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., Asai D. J., Lazarides E. The 68,000-dalton neurofilament-associated polypeptide is a component of nonneuronal cells and of skeletal myofibrils. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1541–1545. doi: 10.1073/pnas.77.3.1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., Gomer R. H., Lazarides E. Heat shock proteins are methylated in avian and mammalian cells. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3531–3535. doi: 10.1073/pnas.78.6.3531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., Lazarides E., O'Connor C. M., Clarke S. Methylation of chicken fibroblast heat shock proteins at lysyl and arginyl residues. J Biol Chem. 1982 Jul 25;257(14):8356–8362. [PubMed] [Google Scholar]
- Welch W. J., Feramisco J. R. Purification of the major mammalian heat shock proteins. J Biol Chem. 1982 Dec 25;257(24):14949–14959. [PubMed] [Google Scholar]





