Abstract
The mode of evolution of the biologically diverse forms of ascomycetes is not well understood, largely because the descent relationships remain unresolved. By using sequences of the nuclear gene RPB2, we have inferred with considerable resolution the phylogenetic relationships between major groups within the phylum Ascomycota. These relationships allow us to deduce a historical pattern of body plan evolution. Within Taphrinomycotina, the most basal group, two simple body plans exist: uncovered asci with unicellular growth, or rudimentary ascoma with hyphal growth. Ancestral ascomycetes were filamentous; hyphal growth was lost independently in the yeast forms of Taphrinomycotina and Saccharomycotina. Pezizomycotina, the sister group to Saccharomycotina, retained mycelial growth while elaborating two basic ontogenetic pathways for ascoma formation and centrum development. The RPB2 phylogeny shows with significant statistical support that taxa in Pezizomycotina with ascohymenial ontogeny (ascoma generally forms after nuclear pairing) are ancestral and paraphyletic, whereas ascolocular fungi with fissitunicate asci are a clade derived from them. Ascolocular lichens are polyphyletic, whereas ascohymenial lichens comprise a monophyletic group that includes the Lecanorales. Our data are not consistent with a derived origin of Eurotiomycetes including Aspergillus and Trichophyton from within a lichen-forming ancestral group. For these reasons, the results of this study are considerably at variance with the conclusion that major fungal lineages are derived from lichensymbiotic ancestors. Interpretation of our results in the context of early work suggests that ascoma ontogeny and centrum characters are not in conflict with the molecular data.
The Ascomycota comprise the largest phylum in Kingdom Fungi (1) and occupy a broad range of habitats. These fungi affect human life in many ways: as infectious agents in plant disease and human and animal mycoses, by producing mycotoxins and antibiotics, and through fermentative production of foodstuffs, chemicals, and pharmaceuticals.
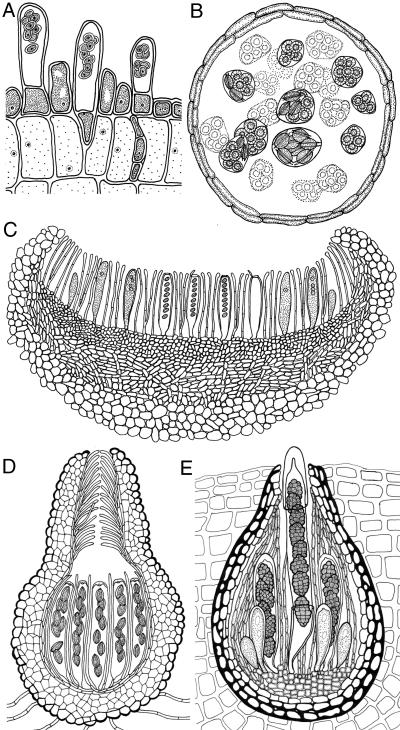
Essential to understanding the nature of these functionally diverse organisms is knowledge of the ascomycete body plan, the developmental program that specifies morphology at different stages of ontogeny. The defining feature of ascomycete fungi is the formation, after diploidization, of an ascus cell, composed of a rigid wall, and at maturity, the haploid products of meiosis (ascospores). For most ascomycetes, the ascus resides within an ascoma (fruiting body), which is a differentiated multicellular structure. Exceptions to this fact are the Saccharomycetes (budding yeasts) and many taxa in Taphrinomycotina (basal taxa), which have naked asci (Fig. 1A). In general, the ascoma structures of an ascomycete species is its most complex and characteristic developmental feature. The developmental morphologies of fungi largely represent a balance between genetic specifications and opportunistic environmental events. Body plan comparisons, in combination with a well resolved molecular phylogeny, can place major emergent characters on particular branches of the phylogenetic tree, and thus establish which features are ancestral and which are derived.
Fig. 1.
Diagrams of asci and ascomata. (A) Thick-walled naked asci of Taphrina deformans without ascoma. (B–D) Hymenoascomycetes (Ascohymeniales), as in Aspergillus sp. with a cleistothecial-closed ascoma and prototunicate asci (B), Peziza sp., with an apothecial-open ascoma and operculate unitunicate asci (C), and Neurospora sp., with a perithecial-closed ascoma having a pore at the top and inoperculate unitunicate asci (D). (E) Loculoascomycetes (Ascoloculars) produce ascostromata and fissitunicate asci as in Pleospora sp.
The ontogeny of ascomata, ascus structures, and centrum development have been used widely in ascomycete classification (2–6). The centrum consists of all of the structures within an ascoma and includes asci, sterile hyphae, and other tissues. A similar concept, a hamathecium, is the totality of the sterile cells and hyphae that are interspersed among asci or projecting into the locule or ostiole of an ascoma. The ascoma may originate in vegetative stromatic tissues or in hyphae. Ascomata with various shapes in Pezizomycotina (filamentous ascomycetes) have two basic developmental plans (refs. 3 and 7 and Fig. 1 B–E). For ascohymenials (Hymenoascomycetes), the formation of the ascoma follows nuclear pairing. The asci of these fungi are almost always unitunicate. For ascolocular fungi (Loculoascomycetes), on other the hand, the ascoma is initiated by the formation of a locule within a stroma before nuclear pairing in the dikaryon. The asci are bitunicate with fissitunicate dehiscence (3–5).
Previous efforts to infer an ascomycete phylogeny from molecular sequences have nearly all used rDNA sequence data sets. While these investigations have identified a number of monophyletic lineages, higher-order relationships, particularly in Pezizomycotina, could not be resolved with statistical significance (8–10). A formal outline of ascomycete classification for families and higher taxa based mainly on rDNA evidence was proposed by Eriksson and Winka (11, 12). In this proposed framework, while at least 11 classes in the Pezizomycotina could be discerned, their interrelationships remained unknown. The information needed to fill in this framework should, in principle, be obtainable from phylogenetic studies with slowly evolving genes that encode proteins.
Protein-encoding genes of the nucleus that are involved in the replication, transcription, and translation of genetic information have been singled out as appropriate for phylogenetic studies (13) because they have not been horizontally transferred in eukaryotes. Genes for the subunits of nuclear DNA-dependent RNA polymerases I, II, and III have coexisted since the initial eukaryotic ancestor. One of these genes is RPB2, encoding the second largest RNA polymerase subunit, which has the additional useful properties of being single copy in ascomycetes and having a relatively slow evolutionary rate (14). Moreover, the role that this RNA polymerase subunit plays in the cell is so general that it is little affected by major evolutionary adaptations in cell structure or physiology. In parallel studies on the phylogeny of basidiomycete fungi using RPB2, RPB1, and lsu rDNA sequences (15, 16), the two protein-coding genes resolve many internal branches that are unresolved in the rDNA tree. The basis for this difference appears to be a deficit in sites evolving at slow-to-moderate rates in rDNA. In this study, we analyzed the protein sequences of RPB2 in ascomycetes and outgroups by using both parsimony and Bayesian methods. We obtained from this a highly resolved ascomycete phylogeny, providing a framework on which to trace the evolution of body plan in Ascomycota.
Materials and Methods
Materials. Sixty-one fungal taxa were used in this study, including seven basidiomycetes from the major groups as outgroup, and 54 ascomycetes. Seven of the 11 classes of Ascomycota were sampled (17). The sources of fungal strains and GenBank accession numbers for RPB2 gene sequences are listed in Table 1, which is published as supporting information on the PNAS web site. The diverse morphologies of some representatives from the major ascomycete lineages are shown in Fig. 2.
Fig. 2.
Examples of ascomata of Ascomycota. (A) Taphrina deformans. (B) Neolecta vitellina.(C) Peziza sp. (D) Leotia viscose.(E) Microglossum viride.(F) Xanthoria polycarpa. (G) Peltigera membranacea. (H) Opegrapha varia. (I) Dermatocarpon reticulatum. Nonlichenized ascomycetes (A–E), lichens (F–I), hymenoascomycetes (C–G), and loculoascomycetes (H and I). Photographs are courtesy of Joe Ammirati for A, Raymond Boyer for B, Ben Woo for C, Taylor F. Lockwood for D, Mark Steinmetz for E, and Stephen Sharnoff for F–I.
Molecular Techniques and Phylogenetic Analyses. The methods for fungal culture, DNA isolation, PCR amplification, cloning, and DNA sequencing have been described (14). The set of general oligonucleotide primers for amplifying regions 3–11 of RPB2 genes were described (14).
The amino acid sequences of RPB2 translated from DNA sequences between regions 3 and 11 of 61 fungi were aligned by using clustal x (18), with subsequent visual adjustment, resulting in 928 aligned amino acid positions, including gaps. The regions that could not be aligned reliably were removed, leaving a total of 914 amino acid positions for phylogenetic analyses. Phylogenetic analyses were carried out by using maximum parsimony and Bayesian inference, based on RPB2 protein sequences. Parsimony analyses were conducted by using paup* 4.0B10 (19) with a weighted-step matrix converted from the JTT matrix (20, 21). The heuristic search using the random addition of taxon option was performed with 1,000 replicates to increase the chance of finding all of the most parsimonious trees. To evaluate the strength of the phylogenetic conclusions, 500 weighted parsimony bootstrap replicates were performed by using the heuristic search with the random addition of taxon option (10 times per replicate). Bayesian inference provides probabilistic measures of tree strength that use explicit models of sequence evolution to test phylogenetic hypotheses. Bayesian phylogenetic analyses with Markov chain Monte Carlo sampling were conducted by using mrbayes v. 2.01 (22). Six independent Markov chain Monte Carlo runs were carried out by using the JTT model for sequence evolution and uniform prior probabilities and tree topologies, including one run with 3 × 106 generations, two runs with 2 × 106 generations, and three runs with 1 × 106 generations, to ensure a sufficient number of generations and sampling of the same posterior probability landscape. Each run started with random trees for each of four simultaneous chains, resulted in concordant joint posterior probability distributions for the topology. The sampling was done every hundredth generation for each run. The samples before the convergence of the Markov chain were discarded for each run. The remaining samples from each run were combined into a single file with a total of 94,000 phylogenetic trees, which were then imported into paup* 4.0B10 to compute the 50% majority rule consensus tree. The percentages for the branches in the consensus tree represent the Bayesian posterior probabilities, which are the rough equivalent of a maximum likelihood search with bootstrapping (22).
Results
RPB2 Sequences and Their Signatures in Different Groups of Ascomycetes. We sequenced 2.7 kb of the RPB2 DNA-coding region, excluding sequences homologous to the first 600 and the last 360 base pairs of Saccharomyces cerevisiae RPB2 (14). When aligned, the predicted amino acid sequences of RPB2 reveal distinctive signatures in several regions that are diagnostic for Ascomycota, Sordariomycetes, Loculoascomycetes, and Hymenoascomycetes (Fig. 4, which is published as supporting information on the PNAS web site). Whereas phylogenetic analyses are quantitative measures of the molecular data, signature sequences for a particular group are diagnostic, and may give valuable hints on relationships between taxa. For example, the signature sequences and the highly resolved RPB2 phylogeny established the associations of some asexually reproduced taxa (anamorphs) with the sexually reproduced taxa (teleomorphs), such as relations of Cenococcum to the taxa in Melanommatales, Nattrassia to Botrosphaeria, and Thamnolia to Dibaeis (Fig. 3).
Fig. 3.
Protein sequence-based RPB2 phylogeny and biological characters associated with each taxon. Phylogeny of Ascomycota reconstructed by using (A) Maximum parsimony with bootstrap values >40% on the branches and (B) Bayesian inference with Bayesian posterior probabilities (percent) noted above individual branches. (C) Biological characters of each taxon. Filled circles indicate the presence of the character. The characters of imperfect ascomycetes with known teleomorphs are assigned based on the characters of their teleomorphs. Taxa with green shade are lichens.
Phylogenetic Relationships Between Major Lineages Based on RPB2 Protein Sequences. The single most parsimonious tree obtained by weighted analysis of RPB2 protein sequences is largely congruent with that from Bayesian analyses (Fig. 3). The only differences between these two trees are in the relationships among taxa of Taphrinomycotina and the position of Diploschistes. We attribute the sister relationship of Diploschistes to Xanthoria seen in parsimony analysis to long-branch attraction, because it conflicts both with the Bayesian tree and with morphology-based classification.
As Hillis and Bull (23) have pointed out, bootstrap support may be too conservative; in their simulations, a group appears on the true tree ≈95% of the time when P = 0.70. Therefore, a 70% bootstrap value is considered to be statistically significant support in this study. The Bayesian posterior probabilities of the nodes, given the assumptions concerning the model of DNA sequence evolution, are considered to be true probabilities, and therefore the nodes receiving >95% can be considered to have statistically significant support (24). The Bayesian method is especially apt at providing a measure of statistical support for a consistently resolved topology in analyses with short branches for which parsimony bootstrap values are low (25, 26).
The RPB2 phylogeny from Bayesian inference (Fig. 3B) is highly resolved except for two terminal branches and two internal branches within Taphrinomycotina that have <95% Bayesian posterior probability. Both methods of analysis find that Saccharomycotina are the sister group to Pezizomycotina. Basal to these branches are the Taphrinomycotina, a paraphyletic group with highly diverse morphologies. The node separating Taphrinomycotina from the Saccharomycotina and Pezizomycotina is strongly supported (90% bootstrap and 100% Bayesian, Fig. 3). Support for the nodes of monophyletic Saccharomycotina and monophyletic Pezizomycotina are remarkably strong (100% and 96% in parsimony, respectively, and 100% in Bayesian, Fig. 3).
In the Pezizomycotina, lineages A and B of Pezizales are at the base (Fig. 3). All other taxa fall either within clade C or clade D with 98% support in Bayesian analysis (Fig. 3). Regarding body plan evolution, lineages A, B, and C share the trait of ascohymenial ascoma development, indicating that this feature of their ontogeny is ancestral in Pezizomycotina. For the taxa in clade D, ascoma development is ascolocular (Fig. 3). Such support for monophyly of loculoascomycetes and for their early divergence from hymenoascomycetes has not been obtained in any of the phylogenetic studies based on rDNA sequences (8–10, 27).
Within the Loculoascomycete clade (D) are three robustly supported lineages: a Pleosporales clade (E), which includes members of the Pleosporales and Melanommatales; the Dothideales clade (F), and the well supported Chaetothyriales clade (G). Dothideales and Pleosporales are shown to be sister to one another with strong support (92% in parsimony analysis and 100% in Bayesian analysis, Fig. 3). The Hymenoascomycete clade (C) likewise includes three major lineages: Eurotiomycetes clade (K), an ascohymenial lichen clade (J), and a clade containing Helotiales and Sordariomycetes. This analysis, unlike those published previously (14, 28, 29), provides statistically significant support for a sister relationship between the Sordariomycete clade (H) and the Helotiales (clade L) (72% bootstrap and 100% Bayesian). Besides ascohymenial lichen clade J, ascolocular lichens occur at two positions in the Loculoascomycete clade D: the Dermatocarpon reticulatum and Opegrapha varia group within Chaetothyriales and Pleosporales, respectively (Fig. 3).
Discussion
Whereas the tree topology for Ascomycota differs between rDNA- and RPB2-based phylogenies, both identify three major lineages. These basal lineages are of Taphrinomycotina (a paraphyletic assemblage of basal taxa), and the two sister groups Saccharomycotina (true yeasts) and Pezizomycotina (mostly filamentous, ascoma-producing) (Fig. 3 and ref. 9). In addition, the RPB2 phylogeny resolved relationships among groups of Pezizomycotina that were uncertain, based on rDNA. Based on these data, we were able to infer body plan evolution regarding ascoma ontogeny, ascoma architecture, hymenium organization, and ascal form.
Ancestral Form of Ascomycetes. Taphrinomycotina are supported as a paraphyletic basal group of ascomycetes by both RPB2 and rDNA data (9, 14). This group is highly variable in morphological and biochemical characters, including saprobic and parasitic forms, represented here by Neolecta vitellina (Fig. 2B), the fission yeast Schizosaccharomyces pombe, the human pathogen Pneumocystis carinii, and the plant pathogen Taphrina deformans (Figs. 1A and 2A). Most members of the group other than Neolecta have a simple body plan, lacking differentiated cells and structures (9). Vegetatively, these organisms grow either unicellularly or with a sparse mycelium, whereas their sexual phase produces naked asci directly from the ascogenous cells. Neolecta is the only taxon in Taphrinomycotina that produces an ascoma. Its association with the Taphrinomycotina suggests an early occurrence of ascogenous hyphae and ascoma formation during the evolution of the Ascomycota (30). Placement of Neolecta in the basal lineage of the Ascomycota is supported by four genes, including 18S rDNA, 25S rDNA, RPB2, and β-tubulin (14, 30, 31).
Several lines of evidence suggest that the ascomycete common ancestor had mycelial growth. Zygomycetes, a clade shown by a combined RPB2 and RPB1 phylogeny to occupy a position immediately basal to the divergence of ascomycetes and basidiomycetes (data not shown), share their filamentous growth habit. Both basidiomycetes and ascomycetes, sister groups to each other, have septate hyphae and a dikaryotic stage. In addition, the basal members of Saccharomycotina have very dense hyphal growth (30). Thus, it appears that the yeast forms of Taphrinomycotina and Saccharomycotina result from independent loss of filamentous growth. In the two higher groups, which are sisters to one another, Saccharomycotina have simple mycelial and/or yeast dimorphic growth, whereas the euascomycete radiation evolved an array of different ascoma structures.
Evolution of Ascoma Ontogeny and Centrum Organization. Evolutionary innovations in Pezizomycotina, the most complex ascomycetes, include different types of ontogeny, ascoma architecture, organized centrum with diversified hamathecial tissues, ascogenous hyphae with crosiers, and specialized ascal structures associated with dehiscence. The crozier is a differentiated hook structure derived from an elongated and bent ascogenous hypha formed before ascus development. The binucleate crook cell is the ascus progenitor within which karyogamy and meiosis take place. Ascogenous hyphae and crozier branching patterns determine the arrangement of asci in the hymenium (a fertile layer of ascoma). Unlike taxa in Pezizomycotina, Neolecta forms a rudimentary ascoma. It has primitive characters, including an unorganized hymenium lacking paraphyses (upward growing basally attached sterile hyphae) among the asci, unitunicate asci with undifferentiated apices, and a lack of croziers before ascus development (33, 34). Molecular data indicate that the characters of forcibly discharged asci and ascoma formation evolved early in the Ascomycota, whereas the paraphyses and croziers of ascogenous hyphae are probably derived features of Pezizomycotina, although these features were lost secondarily in some taxa of Pezizomycotina.
Of the two basic types of ascoma ontogeny, ascohymenial and ascolocular, the RPB2 phylogeny shows that the Ascohymenial group is basal and paraphyletic (clades A, B, and C) and that ascolocular fungi are monophyletic and derived (clade D in Fig. 3). The RPB2 phylogeny shows that the apothecial taxa are basal and paraphyletic because apothecia are present in the lineages of the Pezizales, Helotiales, and lichen groups (clades A, B, L, and J in Fig. 3; Fig. 2 C–F). This result was also shown in some phylogenetic analyses based on the 18S rDNA data, but without statistical support (35). Apothecia with interspersed paraphyses and operculate unitunicate asci appear to be the ancestral conditions for Pezizomycotina, as evidenced by the members of Pezizales (Figs. 1C and 2C).
The RPB2 phylogeny shows that fungi with perithecia (most Sordariomycetes) and cleistothecia (Eurotiomycetes) each form a monophyletic lineage within the ascohymenial clade C (Fig. 3). The RPB2 phylogeny provides significant support for a sister relationship between perithecial and apothecial fungi (Sordariomycetes and Helotiales, respectively; Fig. 3). Perithecia and apothecia are distinct in appearance and are suitable characters for delineating the majority of Sordariomycete taxa and the Helotiales, respectively. Their sister relationship based on DNA mirrors the morphological relationships because both share the feature of ascohymenial ontogeny and an organized hymenium with interspersed paraphyses and inoperculate unitunicate asci. Phylogenies based on rDNA have variously put perithecial Sordariomycetes either in the position of sister group to apothecial Helotiales (28, 29) or sister to pseudothecial Pleosporales (10, 27). Our result to some degree supports Nannfeldt's hypothesis (5) that apothecial ascomata are the primitive state and perithecial and cleistothecial forms are derived.
Loculoascomycetes were first proposed by Luttrell (4) to include taxa with fissitunicate asci and ascostromata with ascolocular ontogeny. The RPB2 phylogeny supports monophyletic Loculoascomycetes, including the Pleosporales, Dothideales, Melanommatales, and Chaetothyriales (Fig. 3). The individual orders of loculoascomycetes are delimited mainly on the basis of centrum development (36). Members of Pleosporales and Melanommatales (Pleosporales complex) have downward-growing sterile hyphae that fuse with the base of the ascostromata (pseudoparaphyses) (Fig. 1E). The Dothideales have neither sterile cells nor sterile hyphae within the stroma. Members of the Chaetothyriales are typified by the presence of short apical sterile hyphae growing downwards in the locule but not reaching the base (periphysoids). In the RPB2 phylogeny, Pleosporales and Melanommatales (ascostroma with pseudoparaphyses) are closely related to Dothideales (ascostroma without pseudoparaphyses) (clades E and F in Fig. 3).
A major conclusion of our work is the phylogenetic placement of Chaetothyriales. It is a sister group to the Eurotiomycetes in most rDNA phylogenetic studies (10, 27, 29, 37). However, both the RPB2 phylogeny and RPB2 signature sequences support its sister relationship to a Pleosporales plus Dothideales clade (clade D in Fig. 3). The latter relationship was also weakly supported in one rDNA phylogenetic study (38). The inclusion of Chaetothyriales within the Loculoascomycetes based on DNA characters is further reinforced by the common presence of ascostromata, fissitunicate asci, and apical pseudoparaphyses. Eurotiomycetes are in the ascohymenial clade of the RPB2 phylogeny.
Ascus Evolution. The ascus is a sac-like cell generally containing eight haploid ascospores. Although ascus morphology is highly variable, as shown by light and electron microscope studies (2, 39), there are three basic ascus types based on the ascus wall structure and ascospore release mechanism: prototunicate, unitunicate, and bitunicate. Prototunicate asci have a thin, delicate wall, and passively discharge their ascospores (Fig. 1B). Unitunicate and bitunicate asci actively discharge their spores by forcible ejection. The wall of a unitunicate ascus has two layers that adhere closely together (Fig. 1 C and D). The two layers of the ascal wall in the bitunicate ascus separate from each other during dehiscence. A fissitunicate ascus is a specific type of bitunicate ascus present in loculoascomycetes wherein complete separation of the two wall layers by jack-in-the-box dehiscence (Fig. 1E and refs. 2 and 40). Lecanoralean asci with rostrate dehiscence have an inner wall layer thickened apically that breaks through the outer wall layer as spores discharge.
Unitunicate asci are the basal form in ascomycetes based on the RPB2 phylogeny (Fig. 3). Unitunicate asci with an undifferentiated apex appeared in Taphrinomycotina (Taphrina, Protomyces, and Neolecta), whereas operculate unitunicate asci are the basal form in Pezizomycotina (Pezizales), and inoperculate unitunicate forms occur in other ascohymenial taxa such as Sordariomycetes, Helotiales, and lichens. The RPB2 phylogeny shows that prototunicate asci evolved convergently and appear in polyphyletic taxa in Taphrinomycotina (Schizosaccharomyces, Saitoella, and Pneumocystis), Saccharomycotina, and Pezizomycotina (Eurotiomycetes). Therefore, prototunicate and unitunicate asci evolved early in the Ascomycota, and these features were gained or lost repeatedly during evolutionary divergence. Fissitunicate asci are monophyletic and are found only in loculoascomycetes and ascolocular lichens. Although the rostrate asci in Lecanorales sometimes have been referred to as bitunicate, due to the extrusion of the inner wall, none has the fissitunicate dehiscence of loculoascomycete asci (40, 41). The RPB2 phylogeny shows that rostrate asci are closely related to unitunicate asci of ascohymenial lichens, and are distantly related to fissitunicate asci, as in ascolocular lichens and nonlichenized loculoascomycetes.
Ascomycetous Lichen Evolution. Nearly half of all fungi are ascomycetes, and of those, half form associations with algae or cyanobacteria as lichens. Whether this large number of species also implies great phylogenetic diversification is an open question. All lichen-forming ascomycetes are in Pezizomycotina. Our sampling included the four major types of lichen-forming fungi, those with prototunicate asci, unitunicate asci with pore dehiscence, and lecanoralean asci with rostrate dehiscence and bitunicate asci with fissitunicate dehiscence. The RPB2 phylogeny shows that the first three of these lichens, all ascohymenial lichens, are a monophyletic group (clade J in Fig. 3). In this clade, lecanoralean lichens (Fig. 2 F and G) with rostrate dehiscence appear to be basal and paraphyletic, suggesting that prototunicate and unitunicate asci in lichens are derived from rostrate asci. Ascohymenial lichens (clade J) are closely related to the Sordariomycetes–Helotiales complex (clades H and L), a relationship supported by the shared common features of ascohymenial ontogeny and hymenium organization with interspersed paraphyses and asci.
The taxa Opegrapha (Fig. 2H) and Dermatocarpon (Fig. 2I) were analyzed as representatives of ascolocular lichens with fissitunicate asci. Opegrapha is in the pleosporales complex (clade E) and Dermatocarpon is clustered in Chaetothyriales clade (clade G) in the RPB2 phylogeny (Fig. 3). This result is supported by the presence of paraphysoids in Opegrapha, as is seen for Pleosporales and the presence of periphysoids in Dermatocarpon, as is found for Chaetothyriales.
The RPB2 gene tree is inconsistent with the primary conclusion of a previous phylogenetic study (27) based on the phylogenetic tree of rRNA genes, namely that major ascomycetous lineages are derived from lichen-symbiotic ancestors. Conceivably, this difference merely reflects differences in gene-specific evolutionary processes, in which case obtaining deeper insight must await more extensive genomic sequencing. Alternatively, the rDNA tree suggesting polyphyly of hymenial lichens may result from the inability of rDNA analysis to resolve internal branches with robust support.
In the phylogeny, we have determined (Fig. 3B) the internal branches have Bayesian posterior probability values between 98% and 100%. The major conclusions of this work that differ from these of Lutzoni et al. (27) are: (i) all ascohymenial lichen fungi belong to clade J, which has 99% Bayesian support; (ii) this clade is sister to the Sordariomycete and Eurotiomycete clades; and (iii) Loculoascomycetes are a monophyletic group, including ascolocular lichens. Thus, the RPB2 phylogeny suggests that lichens have arisen independently several time, if gain and loss of lichen association are equally weighted. More ascolocular lichen samples are needed to confirm whether Chaetothyriales are derived from lichens.
Conclusions
We have demonstrated that RPB2 provides a well resolved ascomycete phylogeny that serves as a basis for analyzing character evolution. Major conclusions about body plan evolution thus obtained are: (i) In Pezizomycotina, ascohymenial taxa are ancestral and paraphyletic, whereas ascolocular taxa with fissitunicate asci are monophyletic and have evolved within the ascohymenial taxa; (ii) in the former group, apothecia and operculate unitunicate asci are the ancestral characters as seen in Pezizales, the basal taxa of Pezizomycotina; perithecial Sordariomycetes and apothecial Helotiales are sister groups that share the characters of an organized hymenium with interspersed inoperculate asci and paraphyses; (iii) Loculoascomycetes, including Pleosporales-Melanommatales, Dothideales, and Chaetothyriales are monophyletic and share the characters of ascolocular ontogeny as well as fissitunicate asci; and (iv) Ascohymenial lichens are monophyletic and cluster with nonlichenized ascohymenial taxa. Ascolocular lichens are polyphyletic and cluster with nonlichenized ascolocular taxa having fissitunicate asci. Ascomycete lichens have arisen independently several times, if gain and loss of lichen association are equally weighted, based on the RPB2 phylogeny.
Supplementary Material
Acknowledgments
We thank Dr. Meredith Blackwell, Dr. Margaret E. Barr Bigelow, Dr. Joseph Ammirati, and Dr. John Stiller for comments on the manuscript; Ellen Duffield for assistance with fungal cultures; Suzanne Joneson and Katherine Glew for lichen specimens; Matthew Hodson for molecular cloning and sequencing some of the lichen RPB2 genes; Dr. Olen Yoder and Dr. Scott Baker for database searching of Syngenta genome sequences of Fusarium, Botrytis, and Cochliobolus; and Dr. Joseph Felsenstein and Dr. Sasa Stefanovic for advice on phylogenetic analyses.
Data deposition: The sequences reported in this paper have been deposited in the GenBank database (accession nos. AF107885–AF107798, AF107800–AF107810, AY485609 – AY485638, AY495590, AY533025, AY533830, D13337, and M15693).
References
- 1.Hawksworth, W. L., Kirk, P. M., Sutton, B. C. & Pegler, D. N. (1995) Ainsworth & Bisby's Dictionary of the Fungi (CAB International, Wallingford, Oxon, U.K.).
- 2.Eriksson, O. (1981) Opera Botanica 60, 1–220. [Google Scholar]
- 3.Luttrell, E. S. (1951) Univ. Mo. Stud. 3, 1–120. [Google Scholar]
- 4.Luttrell, E. S. (1955) Mycologia 47, 511–532. [Google Scholar]
- 5.Nannfeldt, J. A. (1932) Nova Acta Regiae Soc. Sci. Ups. 8, 1–368. [Google Scholar]
- 6.Reynolds, D. R. (1981) Ascomycete Systematics: The Luttrellian Concept (Springer, New York).
- 7.Barr, M. E. (2001) in The Mycota, eds. Esser, K. & Lemke, P. A. (Springer, Berlin), Vol. 7, pp. 161–177. [Google Scholar]
- 8.Berbee, M. L. (2001) Physiol. Mol. Plant Pathol. 59, 165–187. [Google Scholar]
- 9.Nishida, H. & Sugiyama, J. (1994) Mycoscience 35, 361–366. [Google Scholar]
- 10.Spatafora, J. W. (1995) Can. J. Bot. 73, S811–S815. [Google Scholar]
- 11.Eriksson, O. E. & Winka, K. (1997) Myconet 1, 1–16. [Google Scholar]
- 12.Eriksson, O. V. & Winka, K. (1998) Myconet 1, 17–24. [Google Scholar]
- 13.Sicheritz-Ponten, T. & Andersson, S. G. (2001) Nucleic Acids Res. 29, 545–552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu, Y., Whelen, S. & Hall, B. (1999) Mol. Biol. Evol. 16, 1799–1808. [DOI] [PubMed] [Google Scholar]
- 15.Matheny, P. B. (2003) in Biology (Univ. of Washington, Seattle), pp. 214.
- 16.Matheny, P. B., Liu, Y. J., Ammirati, J. F. & Hall, B. D. (2002) Am. J. Bot. 89, 688–698. [DOI] [PubMed] [Google Scholar]
- 17.Eriksson, O. E., Baral, H.-O., Currah, R. S., Hansen, K., Kurtzman, C. P., Rambold, G. & Laessøe, T. (2002) Outline of Fungi, Myconet Umeå, issue 8 (December 2, 2002), available at www.umu.se/myconet/Myconet.html.
- 18.Thompson, J. D., Plewniak, F. & Poch, O. (1999) Nucleic Acids Res. 27, 2682–2690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Swofford, D. L. (2003) paup*, Phylogenetic Analyses Using Parsimony (*and Other Methods), (Sinauer, Sunderland, MA), version 4.0b10.
- 20.Felsenstein, J. (1981) Biol. J. Linn. Soc. 16, 183–196. [Google Scholar]
- 21.Jones, D. T., Taylor, W. R. & Thornton, J. M. (1992) Comput. Appl. Biosci. 8, 275–282. [DOI] [PubMed] [Google Scholar]
- 22.Huelsenbeck, J. P. & Ronquist, J. P. (2001) Biometrics 17, 754–755. [DOI] [PubMed] [Google Scholar]
- 23.Hillis, D. M. & Bull, J. J. (1993) Syst. Biol. 42, 182–192. [Google Scholar]
- 24.Rannala, B. & Yang, Z. (1996) J. Mol. Evol. 43, 304–311. [DOI] [PubMed] [Google Scholar]
- 25.Murphy, W. J., Eizirik, E., O'Brien, S. J., Madsen, O., Scally, M., Douady, C. J., Teeling, E., Ryder, O. A., Stanhope, M. J., de Jong, W. W. & Springer, M. S. (2001) Science 294, 2348–2351. [DOI] [PubMed] [Google Scholar]
- 26.Karol, K. G., McCourt, R. M., Cimino, M. T. & Delwiche, C. F. (2001) Science 294, 2351–2353. [DOI] [PubMed] [Google Scholar]
- 27.Lutzoni, F., Pagel, M. & Reeb, V. (2001) Nature 411, 937–940. [DOI] [PubMed] [Google Scholar]
- 28.LoBuglio, K. F., Berbee, M. L. & Taylor, J. W. (1996) Mol. Phylogenet. Evol. 6, 287–294. [DOI] [PubMed] [Google Scholar]
- 29.Berbee, M. L. (1996) Mol. Biol. Evol. 13, 462–470. [DOI] [PubMed] [Google Scholar]
- 30.Landvik, S., Eriksson, O. E. & Berbee, M. (2001) Mycologia 93, 1151–1163. [Google Scholar]
- 31.O'Donnell, K. O., Cigelnik, E., Weber, N. S. & Trappe, J. M. (1997) Mycologia 89, 48–65. [Google Scholar]
- 32.Kurtzman, C. P. & Robnett, C. J. (1995) Can. J. Bot. 73, S824–S830. [Google Scholar]
- 33.Redhead, S. A. (1977) Can. J. Bot. 55, 301–306. [Google Scholar]
- 34.Korf, R. P. (1973) in The Fungi, eds. Ainsworth, G. C., Sparrow, F. K. & Sussman, A. S. (Academic, New York), Vol. 9 A, pp. 249–319. [Google Scholar]
- 35.Gargas, A. & Taylor, J. W. (1995) Exp. Mycol. 19, 7–15. [DOI] [PubMed] [Google Scholar]
- 36.Alexopoulos, C. J., Mims, C. W. & Blackwell, M. (1996) Introductory Mycology (Wiley, New York).
- 37.Haase, G., Sonntag, L., Peer, Y. V. D., Uijthof, J. M. J., Podbielski, A. & Melzer-Krick, B. (1995) Antonie Leeuwenhoek 68, 19–33. [DOI] [PubMed] [Google Scholar]
- 38.Winka, K., Eriksson, O. E. & Bång, A. (1998) Mycologia 90, 822–830. [Google Scholar]
- 39.Sherwood, M. A. (1981) Bot. J. Linn. Soc. 82, 15–34. [Google Scholar]
- 40.Hafellner, J. (1994) in Ascomycete Systematics: Problems and Perspectives in the Nineties, ed. Hawksworth., D. L. (Plenum, New York), pp. 315–320.
- 41.Hafellner, J. (1988) in CRC Handbook of Lichenology, ed. Galun, M. (CRC, Boca Raton, FL), Vol. 3, pp. 41–52. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.