SUMMARY
Eyes absent (Eya) genes regulate organogenesis in both vertebrates and invertebrates. Mutations in human EYA1 cause congenital Branchio-Oto-Renal (BOR) syndrome, while targeted inactivation of murine Eya1 impairs early developmental processes in multiple organs, including ear, kidney and skeletal system. We have now examined the role of Eya1 during the morphogenesis of organs derived from the pharyngeal region, including thymus, parathyroid and thyroid. The thymus and parathyroid are derived from 3rd pharyngeal pouches and their development is initiated via inductive interactions between neural crest-derived arch mesenchyme, pouch endoderm, and possibly the surface ectoderm of 3rd pharyngeal clefts. Eya1 is expressed in all three cell types during thymus and parathyroid development from E9.5 and the organ primordia for both of these structures failed to form in Eya1−/− embryos. These results indicate that Eya1 is required for the initiation of thymus and parathyroid gland formation. Eya1 is also expressed in the 4th pharyngeal region and ultimobranchial bodies. Eya1−/− mice show thyroid hypoplasia, with severe reduction in the number of parafollicular cells and the size of the thyroid lobes and lack of fusion between the ultimobranchial bodies and the thyroid lobe. These data indicate that Eya1 also regulates mature thyroid gland formation. Furthermore, we show that Six1 expression is markedly reduced in the arch mesenchyme, pouch endoderm and surface ectoderm in the pharyngeal region of Eya1−/− embryos, indicating that Six1 expression in those structures is Eya1 dependent. In addition, we show that in Eya1−/− embryos, the expression of Gcm2 in the 3rd pouch endoderm is undetectable at E10.5, however, the expression of Hox and Pax genes in the pouch endoderm is preserved at E9.5–10.5. Finally, we found that the surface ectoderm of the 3rd and 4th pharyngeal region show increased cell death at E10.5 in Eya1−/− embryos. Our results indicate that Eya1 controls critical early inductive events involved in the morphogenesis of thymus, parathyroid and thyroid.
Keywords: Eya1, Thymus, Parathyroid, Thyroid, Morphogenesis, Hox, Pax, Six1, Gcm2, Neural crest, Endoderm, Ectoderm, Apoptosis, Mouse
INTRODUCTION
The morphogenesis of mammalian organs derived from the pharyngeal region is complex (Hilfer and Brown, 1984). The thymus and parathyroids form as lateral evaginations of the 3rd pharyngeal pouches, which are derived from the foregut endoderm. The thyroid is formed by the fusion of two structures of separate embryonic origin: the thyroid diverticulum, derived from the endoepithelium in the floor of the pharynx; and the ultimobranchial bodies, formed as evaginations of the 4th pouches (Hilfer, 1968; Rogers, 1927; Rogers, 1971). For each of these endodermal evaginations, there is a corresponding ectodermal invagination or pharyngeal cleft. The ectodermal and endodermal epithelia, placed back to back, form the pharyngeal membrane (Cordier and Haumont, 1980). During their descent in the neck, these pharyngeal organs are surrounded by mesenchyme derived from the cranial neural crest (LeLievre and LeDouarin, 1975). The organ primordia for thymus, parathyroid and ultimobranchial body are formed through epithelial-mesenchymal interactions at approximately E10.5 (Smith, 1965; Moore and Owen, 1967; Owen and Ritter, 1969; LeDouarin and Joterean, 1975; Cordier and Haumont, 1980; LeDouarin et al., 1984; Manley, 2000; Owen et al., 2000). Chick-quail chimera experiments suggested that the endoderm provides the initiating signals for induction of the thymus, and is sufficient for the development of the major epithelial subtypes (LeDouarin and Joterean, 1975). All of these organs, the thyroid, thymus, parathyroid, and ultimobranchial body, develop concurrently and migrate from their sites of origin in the pharynx and pharyngeal pouches to their final positions along the ventral midline of the pharyngeal and upper thoracic region. During their migration, the organ primordia interact with one another to ensure their normal development (Manley and Capecchi, 1998). At present, the molecular basis for control of these events is largely unknown.
The cranial neural crest has been shown to play a critical role during the development of thymus, parathyroid and thyroid, initially by providing the mesenchymal cells that populate the pharyngeal region. Ablation of neural crest cells in chick results in aplasia or dysplasia of the thymus and abnormal development of other pharyngeal pouch derivatives, facial features, and the heart (Bockman and Kirby, 1984). It has been demonstrated that as development proceeds, the neural crest mesenchyme contributes connective tissue elements to organs developing in the pharyngeal region, including thymus and parathyroid, and forms the calcitonin-producing cells of thyroid gland (LeLievre and LeDouarin, 1975; LeDouarin, 1977; Noden, 1983; Noden, 1984). However, the precise role of neural crest cells in the development of organs derived from the pharyngeal region remains unclear.
The ectoderm has also been shown to play an important role in the development of thymus. In the chick embryos, ablation of the ectoderm of the 3rd and 4th clefts before it fuses with the endoderm of the 3rd and 4th pouches results in a rudimentary thymus, while the parathyroid and the ultimobranchial body develop normally (Hammond, 1954). It has been demonstrated that similarly to man, the entire thymus in the mouse is of a mixed ecto-endodermal origin and that the arrested development of the 3rd ectodermal cleft at E11.5 in nude mice results in thymic dysgenesis (Cordier and Haumond, 1980). Recently, it has been described that the Foxn1 gene, which encodes a transcription factor of the winged helix/forkhead class, is mutated in the nude mice (Nehls et al., 1994). However, the precise role of the ectoderm in thymus development is also not clear.
Several genes have been implicated in the development of the pharyngeal organs. Hoxa3, a member of the Hox gene family, which specifies positional identity in the developing embryo (Krumlauf, 1994), is expressed in both 3rd and 4th pharyngeal pouch endoderm and neural crest mesenchyme. Inactivation of the Hoxa3 gene in mice results in aparathyroid and athymia and persistent ultimobranchial bodies (Manley and Capecchi, 1995). Pax1 and Pax9 are closely related members of the paired-box gene family, which play critical roles in the development of multiple organs (Strachan and Read, 1994; Stuart et al., 1994; Neubuser et al., 1995; Dahl et al., 1997). Inactivation of Pax9 results in early failure of thymus, parathyroid and ultimobranchial body formation (Peters et al., 1998), while Pax1 mutants have hypoplastic parathyroid and thymus and disturbed thymocyte maturation (Wallin et al., 1996; Su et al., 2001). Recently, Gcm2, homologous to the Drosophila Glial cells missing gene encoding a transcription factor with a novel DNA binding domain, has been shown to play a key role specifically for the organogenesis of parathyroid glands (Akiyama et al., 1996; Kim et al., 1998; Gordon et al., 2001). In addition to these transcription factors, retinoid signaling has been shown to be essential for the formation of the 3rd and 4th pharyngeal arches (Dupe et al., 1999; Wendling et al., 2000). Although these studies have started to define specific genes controlling early pharyngeal organ development, the identity of the regulatory pathways has not been established.
Four mammalian Eya genes, homologues of the Drosophila eyes absent (eya) gene (Bonini et al., 1993), have recently been isolated (Xu et al., 1997a; Abdelhak et al., 1997; Duncan et al., 1997; Zimmerman et al., 1997; Borsani et al., 1999). The eya gene products contain a divergent N-terminal transactivation domain (Xu et al., 1997b) and a highly conserved 271 amino acid C-terminal Eya domain that participates in protein-protein interactions with So and Dac, the gene products encoded by the Drosophila sine oculis (so) and dachshund (dac) genes, respectively (Chen et al., 1997; Pignoni et al., 1997). In mice, Eya1 and Eya2 are widely expressed in the cranial sensory placodes and at the sites of inductive tissue interactions during organogenesis, while Eya3 and Eya4 are expressed in peri-placodal mesenchyme and dermamyotome, respectively (Xu et al., 1997a; Borsani et al., 1999). These features suggest major roles for Eya genes in the development of vertebrate organs and sensory systems, potentially by mediating the expression of inductive signals that act between tissue layers. In addition in many developing tissues, Eya genes are widely and strikingly co-expressed with Pax and Six genes, the latter representing the mammalian homologs of so (Oliver et al., 1995a; Oliver et al., 1995b; Xu et al., 1997a), suggesting possible interactions between their gene products. Besides the cranial placodes and sensory systems, Eya genes are also strongly expressed in the pharyngeal region and its derivatives from E9.5 during mouse development (Xu et al., 1997a). However, the function of Eya genes in pharyngeal development has not been previously described.
We have recently reported that Eya1-null animals lack ears and kidneys due to defective inductive tissue interactions and apoptotic regression of organ primordia (Xu et al., 1999a). Interestingly, numerous defects in a continuum of cranial structures that are dependent on neural- crest, beginning in the orbital region and extending through the face and neck into the heart, were observed in Eya1 mutants (Xu et al., 1999a) (P.-X. Xu, unpublished). We have now examined the development of organs derived from the pharyngeal region, including thymus, parathyroid and thyroid in Eya1−/− animals. In Eya1−/− embryos, the organ primordia for both thymus and parathyroids fail to form. A thyroid defect is also present, which results from a severe reduction in the number of parafollicular and follicular cells; these cells do not interact with each other owing to a lack of fusion between the ultimobranchial bodies and the thyroid lobe. Furthermore, we show that in Eya1−/− embryos, Six1 expression in pharyngeal arch mesenchyme, pouch endoderm and surface ectoderm was markedly reduced and that Gcm2 expression in the 3rd pouch endoderm was undetectable. Finally, we have shown that increased cell death was observed in the surface ectoderm of the 3rd and 4th pharyngeal region in Eya1−/− embryos of E10.5, indicating that Eya1 is required for the ectodermal cell survival. Our data indicate that Eya1 plays an essential role in the neural crest, endodermal and ectodermal cells that are required for the morphogenesis of mammalian thymus, parathyroid and thyroid.
MATERIALS AND METHODS
Animals and genotyping
Mice heterozygous for a targeted disruption of the Eya1 gene were intercrossed to produce embryos of all three possible genotypes. The mutant allele contained a neo cassette replacing most of the conserved Eya domain region (Xu et al., 1999a). This mutant allele is maintained in three different genetic backgrounds (129, BALB/c, C57BL/6J). Genotyping of mice and embryos was performed as described (Xu et al., 1999a).
The Six1 mutant allele was created by replacement of the endogenous start codon as well as the exon 1 with a promoterless E. coli ATG-lacZ-poly(A) cassette and the PGK-neo gene, and will be presented elsewhere (C. L. and P. M., unpublished). Mutant mice carrying Six1 mutant allele, Six1lacZ, were obtained using gene targeting technology. Eya1/Six1 double heterozygous mutant mice were generated by crossing mice carrying mutant alleles of Eya1 and Six1 (Six1lacZ). A set of lacZ primers are used to detect the mutant allele and a set of primers within the exon 1 of Six1 gene are used to detect Six1 wild-type allele.
Phenotype analyses and in situ hybridization
Embryos for histology and in situ hybridization were dissected out in PBS and fixed with 4% PFA at 4°C overnight. Embryonic membranes were saved in DNA isolation buffer for genotyping. Histology was performed as described previously (Xu et al., 1999a). To visualize Six1lacZ expression, mutant embryos were stained with X-gal and sectioned as described previously (Xu et al., 1999b).
For in situ hybridization, we used 3 wild-type or mutant embryos at each stage for each probe as described previously (Xu et al., 1997a).
Immunohistochemistry
Whole-mount immunostaining with Hoxb1-specific antibody (BabCO) and section staining with calcitonin-specific antibody (ICN) were performed as described previously (Manley and Capecchi, 1995). Both antibodies were detected using HRP-coupled secondary antisera (Vector Laboratories) and staining with diaminobenzidine (DAB). Immunostained sections were counterstained with diluted Ehrlich’s Hematoxylin, mounted in Permount and viewed using DIC.
TUNEL analysis
We performed TUNEL assay for detecting apoptotic cell death using the ApopTag detection kit (Intergen). We used 3 wild-type or mutant embryos for this assay.
RESULTS
Eya1 is required for normal development of the thymus and parathyroid
Eya1 is strongly expressed in the pharyngeal arches (a1–a4), the pouch endoderm (p2–p4) and the surface ectoderm including clefts (2nd, 3rd and 4th) in the pharyngeal region at E9.5–10.5 (Fig. 1A,B and data not shown) (Kalatzis et al., 1998). Subsequently, Eya1 expression was observed in the structures that are derived from the pharyngeal region, including thymus and parathyroid (Fig. 1C,D). To study whether Eya1 plays any role during the formation of thymus and parathyroid, serial histological sections of the neck and upper trunk region of Eya1−/− embryos from E14.5 to P0 were analyzed. The thymus and parathyroid, both derivatives of the 3rd pharyngeal pouches, are absent in Eya1−/− animals on both BALB/c and C57BL/6J backgrounds (Fig. 1E–H). These results suggest that Eya1 expression in the developing thymus and parathyroid glands is required for their normal development.
Fig. 1.
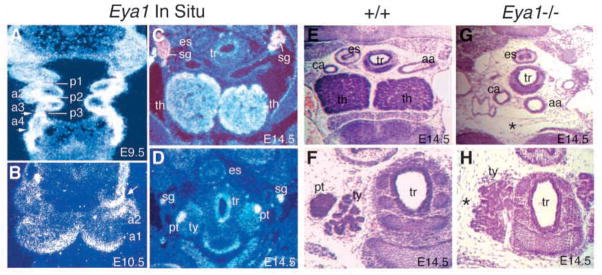
Eya1−/− embryos lack thymus and parathyroid glands. (A) A frontal section showing Eya1 expression in pharyngeal arch (a1–4) mesenchyme, pouch endoderm (p1–3) and pharyngeal clefts (arrows) at E9.5. Cranial is up. (B) A transverse section showing strong Eya1 expression in the distal part of the pharyngeal arches and the surface ectoderm of 2nd cleft (arrow) at E10.5. (C,D) Transverse sections showing Eya1 expression in the thymic lobes (th) and parathyroid glands (pt) at E14.5. In addition, Eya1 is also expressed in sympathetic ganglia (sg) and trachea (tr). Eya1 is not expressed in the thyroid lobe (ty). (E–H) H and E-stained transverse sections of the neck region at E14.5. (E,F) In wild-type embryos, two thymic lobes are present and the parathyroid glands are associated with the thyroid gland (ty). (G,H) In Eya1−/−embryos, no thymus and parathyroid formation (*) was found at the same level or in other regions of the neck and upper trunk. For B–H, dorsal is up. aa, aortic arches; ca, carotid artery; es, esophagus.
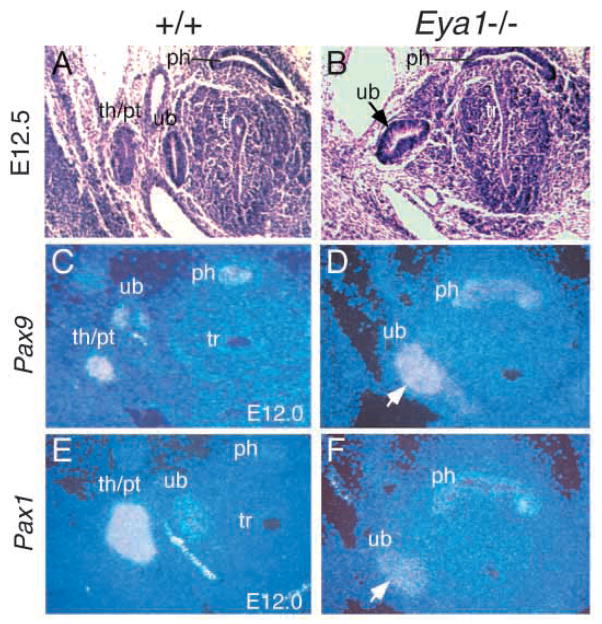
To determine the onset of phenotypic abnormalities during pharyngeal pouch development, we analyzed Eya1−/− embryos at E11.5–12.5. In wild-type embryos, the 3rd pouches evaginate and then separate as epithelial buds to form the primordia of thymus/parathyroid at approximately E12.0 (Fig. 2A). In Eya1−/− embryos, however the organ primordia of thymus/parathyroid were never seen (Fig. 2B; other data not shown). In contrast, the primordia of the ultimobranchial bodies derived from 4th pouches formed with slight indentations in both sides of the head in four out of six Eya1−/− BALB/c embryos (arrow, Fig. 2B).
Fig. 2.
The 3rd pharyngeal pouches do not separate as buds to form the primordia of thymus/parathyroid. (A,B) Transverse sections showing that in wild-type embryos (A), the 3rd pouches evaginate and then separate as buds to form the primordia of thymus/parathyroid (th/pt) at around E12.5, while the 4th pouches separate to form the rudiments of the ultimobranchial bodies (ub) at the same time; however in Eya1−/− embryos (B), the primordia of thymus/parathyroid failed to form, while the rudiments of ultimobranchial bodies (ub) were formed but with a slight indentation (arrow in B). (C–F) Transverse sections showing Pax1 and Pax9 expression in the rudiments of the thymus/parathyroid and ultimobranchial bodies at E12.0 of wild-type embryos (C,E). In Eya1−/− embryos, the expression of Pax1 and Pax9 was only detected in the rudiments of ultimobranchial bodies (arrows). The absence of Pax1 and Pax9 expression in the prospective region of the thymus/parathyroid rudiments confirms the absence of these structures in Eya1−/− embryos (D,F). Dorsal is up. ph, pharynx; tr, trachea.
Pax1 and Pax9 genes are expressed in pharyngeal pouch endoderm from E9.5 and in their derivatives (Fig. 2C,E) (Neubuser et al., 1995; Wallin et al., 1996). In Eya1−/−embryos, both Pax1 and Pax9 expression in the pouch endoderm is not detectably altered at E9.5–10.5, before the budding and outgrowth of thymus/parathyroid rudiments (Fig. 6C–F). However, at E11.5–12.5, Pax1 and Pax9 expression was only detected in the rudiments of the ultimobranchial bodies in Eya1−/− embryos (Fig. 2C–F). The absence of Pax1 and Pax9 expression in the prospective region of the thymus/parathyroid rudiments further confirms the absence of these structures in Eya1−/− embryos (Fig. 2C–F).
Fig. 6.
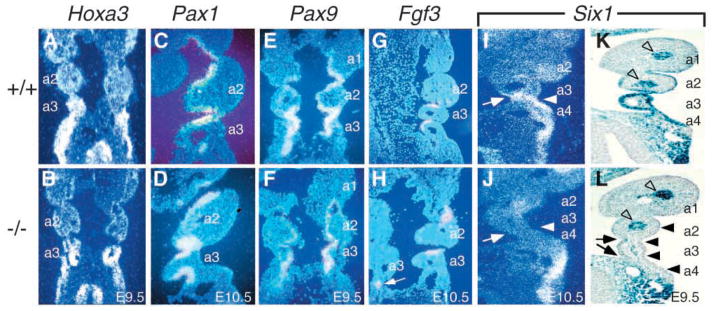
Expression of Six1 in the pharyngeal pouch endoderm and the ectoderm of pharyngeal clefts was markedly reduced in Eya1−/− embryos. (A,B) Coronal sections showing that Hoxa3 is normally expressed in the 3rd and 4th pharyngeal pouches and arches (A), and its expression was unaffected in Eya1−/− embryos at E9.5 (B). (C,D) Coronal sections showing that Pax1 is normally expressed in the pouch endoderm at E9.5–10.5 (C), and its expression is preserved in Eya1−/− embryos (D). (E,F) Coronal sections showing that Pax9 is also expressed in the pouch endoderm at E9.5–10.5 (E) and its expression is also preserved in Eya1−/− embryos (F). (G,H) Coronal sections showing that Fgf3 is normally expressed in the posterior half of the pouch endoderm at E10.5 (G) and its expression was unaffected in Eya1−/− embryos (H). (I,J) Coronal sections showing that Six1 is normally co-expressed with Eya1 in the 3rd pouch endoderm (arrow in I) and the surface ectoderm of 3rd clefts (arrowhead in I), and its expression was markedly reduced in both structures in Eya1−/− embryos (arrows and arrowheads in J). (K,L) Coronal sections showing strong Six1lacZ expression in wild type (K) in the pouch endoderm and surface ectoderm including 2nd, 3rd and 4th pharyngeal clefts; however, in Eya1−/− embryos, Six1lacZ expression was significantly reduced in the pouch endoderm (arrows) and surface ectoderm (arrowheads, L) in the 2nd, 3rd and 4th pharyngeal regions (a2–a4). In contrast, its expression surrounding the arteries of pharyngeal arches remains unaffected (open arrowheads).
To determine whether there are immunological deficits in Eya1 heterozygous mice, we have examined peripheral blood lymphocytes from Eya1+/− mice on 129, BALB/c and C57BL/6J inbred strains using flow immunocytofluorimetry. Monoclonal antibodies were used to assess the proportions of cells expressing antigens B220 (B cells), CD3 and CD4 (T cells), CD11 (macrophages), and NK1 (natural killer cells). Responsiveness to T cell and B cell mitogens in culture (4 days stimulation with conA and overnight stimulation with LPS) was evaluated by examining expression of the early activation antigen CD69 on Thy 1-positive T cells and B220-positive B cells. None of the three strains differed from wild-type mice (data not shown), suggesting that Eya1+/− mice are immunocompetent. In addition, the thymus in the three strains of Eya1+/− mice had normal morphology (data not shown).
Eya1 regulates mature thyroid formation
The thyroid gland is formed by the fusion of the thyroid diverticulum from the floor of the pharynx and the ultimobranchial body from 4th pharyngeal pouch (Hilfer, 1968; Pearse and Polak, 1971; Rogers, 1927; Rogers, 1971; LeLievre and LeDouarin, 1975; Fontaine, 1979). Eya1 is expressed in the pouch endoderm, the ectodermal clefts and the arch mesenchyme in the 4th pharyngeal region (Fig. 1A and data not shown). Although the 4th pouches are slightly indented in some Eya1−/− animals (Fig. 2B), they do separate as buds to form the rudiments of the ultimobranchial bodies and these rudiments express both Pax1 and Pax9 (Fig. 2B,D,F).
Next, we used Hoxb1 expression as an early marker of the 4th pouch endoderm (Frohman et al., 1990; Manley and Capecchi, 1995). Immunostaining revealed that at E10.5–11.5, the expression level of Hoxb1 in the 4th pouches (p4) was indistinguishable from that of wild type (brown stain in Fig. 3A,B, and data not shown). Therefore, our results indicate that the 4th pouches form the primordia of the ultimobranchial body in the absence of Eya1.
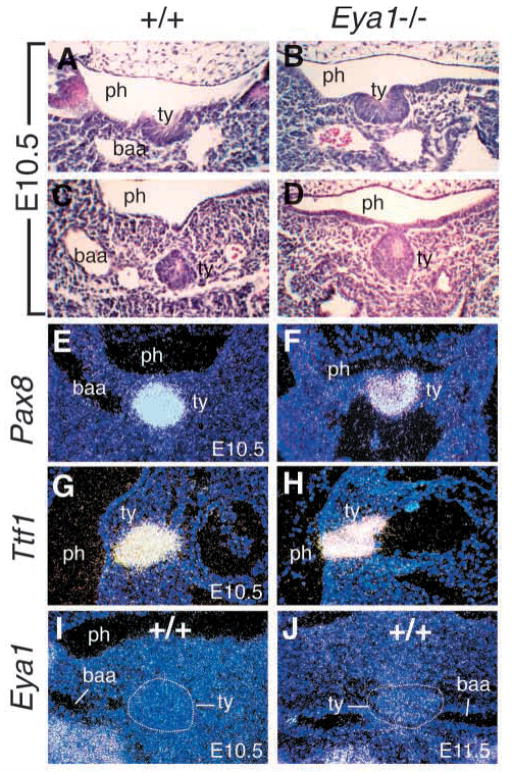
Fig. 3.
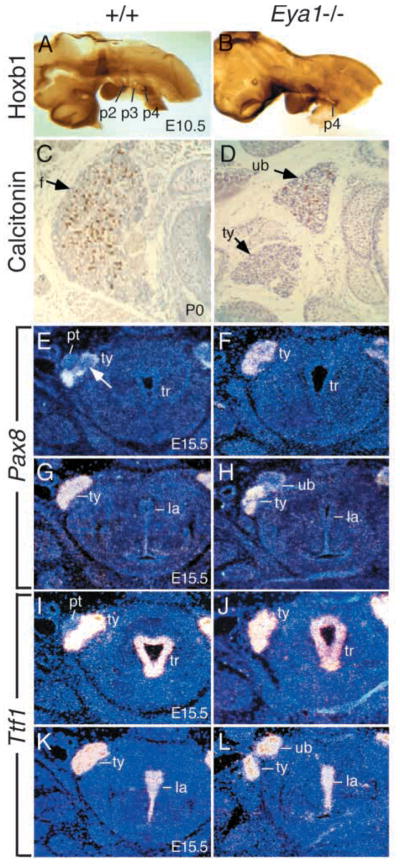
Ultimobranchial body and thyroid lobe defects in Eya1−/−mice. (A,B) The embryos of E10.5, after completion of neural crest migration, were stained with Hoxb1 antibody to label the 4th pharyngeal pouches (p4). The 4th pharyngeal pouches express Hoxb1 (brown stain) in both wild-type (A) and Eya1−/− embryos (B). No significant difference of Hoxb1 expression in the 4th pouch endoderm was observed in Eya1 mutants. (C,D) Transverse sections through newborns, stained with anti-calcitonin antibody (brown staining). (C) A wild-type thyroid with numerous calcitonin-positive cells and follicular cells (f, arrow) throughout the lobe. (D) An Eya1−/− animal with bilateral persistent ultimobranchial bodies and malformed thyroid lobes. Only a few follicles are formed in the main body of the thyroid (ty, arrow). Note that the dorsally placed vesicle which is strongly positive for calcitonin represents a persistent ultimobranchial body (ub) in Eya1-mutants, not fusing with the thyroid lobe. This ultimobranchial body also contains follicle-like structures (arrow). No isthmus was present in this animal. (E–H) Transverse sections at E15.5 showing Pax8 expression in the thyroid lobes in wild-type and Eya1−/− embryos. (E) The ultimobranchial body cell populations showing weak Pax8 expression (arrow) were visible within the thyroid lobes at this stage. Parathyroid (pt) was also visible. (F) No ultimobranchial body cell population was observed within the Eya1−/− thyroid lobes on the same level and parathyroid was also absent. However, the ultimobranchial bodies showing weak Pax8 expression were observed at the anterior end dorsal to the thyroid lobe in Eya1−/−embryos (H). No ultimobranchial bodies were found in wild-type embryos on the same level (G). (I–L) Transverse sections showing Ttf1 expression in the thyroid lobes in wild-type embryos at E15.5 (I,K) and in both the persistent ultimobranchial bodies and the thyroid lobes of Eya1−/− embryos (J,L). Similarly, the persistent ultimobranchial bodies were observed as separate structures located anterodorsally to the thyroid lobes in Eya1−/− animals (L). For C–L, dorsal is up. tr, trachea; la, larynx.
To confirm our observation, we used an antibody against calcitonin to identify parafollicular or C cells that are believed to be neural crest-derived in the ultimobranchial bodies on transverse sections of Eya1−/− and wild-type newborn animals. In wild-type mice, the thyroid is a bilobed structure, with two large lateral lobes joined across the ventral midline at their posterior aspects by the isthmus. Fig. 3C shows a transverse section through one lateral lobe of a control animal. Calcitonin-producing C cells (brown stain) were numerous and could be found throughout the lobe. In contrast, Eya1−/− mice had hypoplastic lobes and fewer calcitonin-producing cells, with 4 BALB/c animals showing a reduction of approximately 60% in the number of calcitonin-positive cells and all the calcitonin-positive cells are localized in an anterodorsally placed vesicular structure (Fig. 3D). These vesicles varied in size and were associated with, but had no visible connection to, the thyroid, which lacked calcitonin-positive cells. In addition, we have observed follicular structures in the calcitonin-positive vesicles (Fig. 3D). H and E stained sections of Eya1−/− mutants showed that these follicular structures did contain colloid, indicating that thyroglobulin is produced and secreted to the lumen of the follicles (data not shown). These calcitonin-positive vesicles probably present persistent ultimobranchial bodies that failed to fuse with the thyroid diverticulum, similar to that seen in Hoxa3 mutant mice (Manley and Capecchi, 1995) and in Echidna, in which ultimobranchial bodies never fuse with thyroid gland and also have colloid-containing follicles (Maurer, 1899).
We next examined the expression of the thyroid-specific genes Pax8 and Ttf1 in Eya1−/− thyroid morphogenesis. Pax8 is expressed in the developing thyroid gland from E9.0 and inactivation of Pax8 results in a total absence of the thyroid follicular cells (Lazzaro et al., 1991; Mansouri et al., 1998). The homeobox gene Ttf1 is expressed in both the ultimobranchial body and the developing thyroid from E9.0 and mice lacking Ttf1 lack both C cells and follicular cells (Lazzaro et al., 1991; Kimura et al., 1996; Mansouri et al., 1998). Both gene products have been shown to cooperate in transcriptional activation of the thyroglobulin gene promoter in follicular cells (Pasca di Magliano et al., 2000). In E15.5 wild-type embryos, although the ultimobranchial bodies are already embedded in the thyroid lobes, they are still visible as discrete cell populations, which show weak Pax8 expression within the thyroid (arrow, Fig. 3E). Parathyroid glands were visible on the sections in which the ultimobranchial body cell populations were evident (Fig. 3E). However, in Eya1−/− embryos, no ultimobranchial body cell population was observed within the thyroid lobes and Pax8 was strongly expressed throughout the lobe (Fig. 3F). The ultimobranchial bodies showing weak Pax8 expression were observed at the anterodorsal end of Eya1−/− thyroid lobes (Fig. 3H). The Ttf1 probe showed similar results with strong expression in both the ultimobranchial bodies and the thyroid lobes of Eya1−/− embryos (Fig. 3I–L) (Mansouri et al., 1998). The ultimobranchial bodies in Eya1−/− animals were detected as separate structures located at the anterodorsal end of the thyroid lobes (Fig. 3D,H,L). We examined 4 BALB/c animals at E15.5 with a Pax8 marker and 4 BALB/c newborn animals with calcitonin staining; 4 showed persistent ultimobranchial bodies on both sides and 4 showed persistent ultimobranchial body on one side only (Table 1).
Table 1.
Summary of thyroid and ultimobranchial body defects in Eya1−/− animals
| No. of animals analyzed | No. of ub defects | Types of ub defects | Thyroid isthmus defects |
|---|---|---|---|
| 4 at E15.5* | 4/4 | 2 persistent on both sides | |
| 2 persistent on one side and absent on the other side | |||
| 4 newborns† | 4/4 | 2 persistent on both sides | 3/4 absent |
| 2 persistent on one side and absent on the other side |
Animals at E15.5 were analyzed by Pax8 expression. Ultimobranchial bodies (ub) showing weak Pax8 expression and located as separate structures anterodorsal to the thyroid lobes were scored as persistent. Absent ubs were scored as absence of both a persistent ub and weak Pax8 expression within the thyroid lobe on a single side.
Animals at newborn stage were analyzed by calcitonin staining. ubs were scored as absent if there was no persistent ub and no calcitonin-positive cells within the thyroid lobe on a single side. Three animals lacked an isthmus.
The structure of the thyroid lobe itself was also affected in Eya1−/− newborn mice. In all cases examined the thyroid lobe was reduced by approximately 40–60% (Fig. 3D and data not shown) and three animals lacked an isthmus (Table 1). Follicles were formed, but the follicular cells were reduced in number. To determine whether Eya1 plays a direct role in the morphogenesis of thyroid gland, we analyzed the thyroid development in Eya1−/− embryos and Eya1 expression in the thyroid from earlier stages. The morphogenesis of thyroid glands begins at around E10.5 when a small region of the floor of the pharyngeal endoderm invaginates to form the thyroid bud (Fig. 4A). The thyroid bud migrates dorsocaudally to the ventral midline (Fig. 4C). In Eya1−/− embryos, the thyroid primordia form and express both Pax8 and Ttf1 (Fig. 4B,D,F,H). We next performed in situ hybridization on both transverse and sagittal sections of 6 wild-type embryos at each stage to determine whether Eya1 is expressed in the thyroid. We failed to detect convincing Eya1 expression in the thyroid at E9.5–14.5 (Fig. 4I,J and Fig. 1D and data not shown), indicating that Eya1 is not expressed in the thyroid gland at these stages. Therefore, it is possible that the defect detected in the thyroid lobe of Eya1−/− animals is due to the lack of fusion with the ultimobranchial body. Nonetheless, our results show that Eya1 is required for the formation of a mature thyroid gland.
Fig. 4.
Normal development of the thyroid primordia in Eya1−/−embryos. (A–D) Transverse sections showing the thyroid primordia (ty) in wild-type (A,C) and Eya1−/− (B,D) embryos at E10.5. The thyroid primordia were evident in Eya1−/− embryos. (E,F) Transverse sections showing Pax8 expression in the thyroid primordia of wild-type and Eya1−/− embryos at E10.5. (G,H) Sagittal sections showing Ttf1 expression in wild-type and Eya1−/− thyroid primordia at E10.5. (I,J) Transverse sections showing that Eya1 expression was not detectable in the thyroid primordia at E10.5–11.5 in wild-type embryos. ph, pharynx; baa, 2nd branchial arch artery. For transverse sections, dorsal is up; for sagittal sections, cranial is up and dorsal is to the left.
Eya1 is required for the expression of Six1 in pharyngeal arch mesenchyme
The formation of the thymus, parathyroid and ultimobranchial body involves interactions between several embryonic cell types (Auerbach, 1960; Rogers, 1971; Bockman and Kirby, 1984). These morphogenetic events require the properly timed interactions of neural crest-derived arch mesenchyme, pouch endoderm, and possibly surface ectoderm from pharyngeal clefts (Auerbach, 1960; Cordier and Haumont, 1980; Bockman and Kirby, 1984; Kuratani and Bockman, 1990). Eya1 is expressed in all three cell types from early embryonic stages (Fig. 1A,B). To determine the earliest stage at which a defect in thymus, parathyroid and thyroid development could be detected, we examined the expression of genes that are known to be expressed in the neural crest, endodermal or ectodermal cells as molecular markers. Embryos were initially analyzed at E9.5 and 10.5, after neural crest migration is complete (E10.5), but before overt thymus and parathyroid development is detectable.
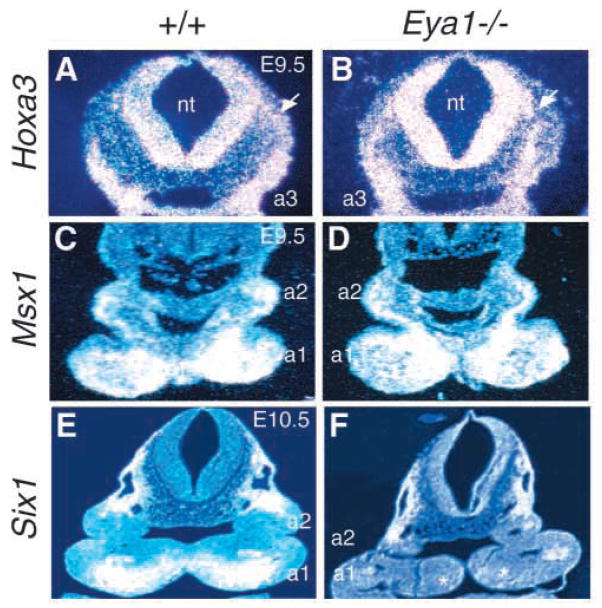
We first examined the expression of Hoxa3, Msx1 and Six1 in the neural crest cells in Eya1−/− embryos. Hoxa3 is expressed in premigratory neural crest cells in hindbrain rhombomeres 5, 6 and migratory neural crest cells that populate the 3rd and 4th pharyngeal arches (Manley and Capecchi, 1995). No significant difference of Hoxa3 expression in the neural tube, migratory neural crest cells or 3rd and 4th pharyngeal arches was observed in Eya1−/− embryos at E9.5–10.5 (Fig. 5A,B and data not shown). Msx1, another homeobox gene, is normally expressed in the distal pharyngeal arch and below the epithelial layer at E10.5 (Brown et al., 1993), which overlaps with the expression domain of Eya1. No significant difference of Msx1 expression in the pharyngeal arches was observed between wild-type and Eya1−/− embryos at E9.5 (Fig. 5C,D). At E10.5, after completion of neural crest migration, the Msx1 expression domain became smaller in Eya1−/− embryos (data not shown). Since Eya1−/− embryos show hypoplasia of the pharyngeal arches, determined by H and E staining of 12 E10.5 Eya1−/− embryos on both transverse and sagittal sections (data not shown), this indicates that the reduction of the Msx1 expression domain could be a result of the size reduction of Eya1−/− pharyngeal arches. Nonetheless, our results indicate that Eya1 is not required for the expression of Msx1 in the pharyngeal arch mesenchyme.
Fig. 5.
Neural crest defects in the pharyngeal arches of Eya1−/−embryos. (A,B) Transverse sections of wild-type and Eya1−/−embryos at E9.5 showing Hoxa3 expression in the hindbrain neural tube, migrating neural crest cells (arrow) and 3rd pharyngeal arches (a3). No significant change was found in Eya1 mutants. (C,D) Transverse sections showing Msx1 expression in the neural crest cells in the pharyngeal arches of wild-type and Eya1−/− embryos at E9.5. No significant difference of Msx1 expression was observed at E9.5 in Eya1−/− embryos. (E,F) Transverse sections showing Six1 expression in the distal edge of arch mesenchyme in wild-type and Eya1−/− embryos at E10.5. Six1 expression was not detectable in Eya1−/− embryos (asterisks in F). Dorsal is up.
Six1, a member of the Six gene family, is normally co-expressed with Eya1 in the pharyngeal arch mesenchyme (Fig. 5E), and its expression depends on Eya1 function during otic development (Xu et al., 1999a). Similar to otic development, Six1 expression was markedly reduced in Eya1−/− pharyngeal arches at E9.5 and 10.5 (Fig. 5F and data not shown). Therefore, our results show that Eya1 is required for the normal expression of Six1 in the neural crest of pharyngeal arches.
Eya1 is required for the expression of Six1 in the pouch endoderm and the surface ectoderm
We next examined the expression of Hoxa3, Pax1, Pax9, Fgf3 and Six1 in the pharyngeal pouch endoderm at E9.5–10.5. Hoxa3 is also expressed in 3rd and 4th pouch endoderm and neural crest mesenchyme, and absence of Hoxa3 results in aparathyroid, athymia and thyroid hypoplasia (Manley and Capecchi, 1995). Similar to that seen in neural crest cells, Hoxa3 expression is not affected in the pouch endoderm in Eya1−/− embryos (Fig. 6A,B). Pax1 and Pax9 are normally expressed in pouch endoderm and deletion of Pax1 results in parathyroid and thymus hypoplasia, while deletion of Pax9 results in absence of all derivatives from 3rd and 4th pharyngeal pouches (Dietrich and Gruss, 1995; Neubuser et al., 1995; Wallin et al., 1996; Peters et al., 1998). At E9.5–10.5, both Pax1 and Pax9 expression in pouch endoderm is not detectably altered in Eya1−/− embryos (Fig. 6C–F and data not shown). Fgf3 is expressed in the posterior half of the pouch endoderm (Fig. 6G) (Wilkinson et al., 1989), and its expression depends on Eya1 function during otic development (Xu et al., 1999a). However, no significant difference in Fgf3 expression was detected in the pharyngeal pouches between control and mutant embryos (Fig. 6G,H). Taken together, these results indicate that Hoxa3, Pax1, Pax9 and Fgf3 function either upstream or independently of Eya1.
In contrast, the expression of Six1, also co-expressed with Eya1 in pharyngeal pouches at E9.5–10.5, was significantly reduced in the pouch endoderm of Eya1−/− embryos (arrows in Fig. 6I,J and data not shown). In addition, the expression of Six1 in the ectodermal clefts was also markedly reduced (arrowheads, Fig. 6I,J). To further confirm our observation, we then crossed the Six1lacZ allele into Eya1+/− mice and generated Eya1−/−Six1lacZ/+ embryos to determine the expression of Six1 in Eya1−/− pouch endoderm and surface ectoderm using X-gal staining for Six1lacZ. As shown in Fig. 6K, Six1lacZ was strongly expressed in the 2nd, 3rd and 4th pouch endoderm and the surface ectoderm including 2nd, 3rd and 4th pharyngeal clefts. However, its expression in Eya1−/− embryos was markedly reduced in both the pouch endoderm and the surface ectoderm (arrows and arrowheads, Fig. 6L). The expression of Six1lacZ surrounding the arteries of pharyngeal arches remained unaffected in Eya1−/− embryos (open arrowheads, Fig. 6K,L). These results indicate that the expression of Six1 also requires Eya1 function in both the pouch endoderm and the surface ectoderm.
Gcm2 expression in the 3rd pharyngeal pouch endoderm is also Eya1-dependent
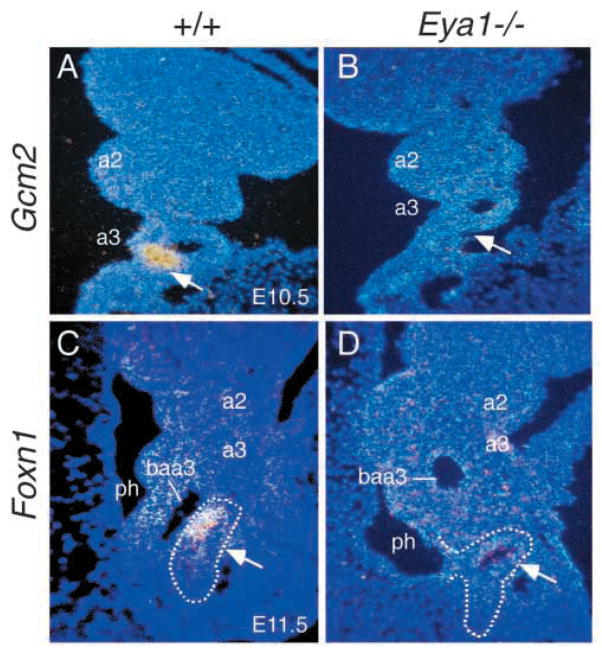
To further investigate the molecular defects in the formation of thymus and parathyroid primordia in Eya1−/− embryos, we examined the expression of the parathyroid-specific Gcm2 and thymus-specific Foxn1 genes. Mice lacking Gcm2 have no identifiable parathyroid glands, but have normal thymus (Gunther et al., 2000). Gcm2 is expressed in the 3rd pouch endoderm from E9.5 and its expression becomes restricted to a small domain within the 3rd pouch endoderm by E10.5 (Fig. 7A and data not shown) (Gordon et al., 2001). However, Gcm2 expression was not detectable in the 3rd pouches of Eya1−/− embryos at E9.5–11.5 (Fig. 7B and data not shown), indicating that Gcm2 expression in the 3rd pouch endoderm is also Eya1 dependent.
Fig. 7.
Gcm2 and Foxn1 expression was not detectable in Eya1−/−embryos. (A,B) Coronal sections through the 3rd pharyngeal pouch of E10.5 wild-type and Eya1−/− embryos showing that Gcm2 is strongly expressed in the 3rd pouch endoderm in wild-type embryos (arrow, A) and its expression was not detectable in Eya1−/− embryos (arrow, B). (C,D) Sagittal sections through the 3rd pharyngeal region showing that Foxn1 is expressed in the ventral region of the thymus/parathyroid primordia at E11.5 of wild-type embryos (arrow, C), but its expression was not detectable in Eya1−/− embryos (arrow, D). Note that the 3rd pouch is attached to the pharyngeal endoderm and does not separate as buds to form the primordia of thymus/parathyroid in Eya1−/− embryos. Cranial is up (A–D) and dorsal is to the left (C,D). a, pharyngeal arches; baa3, 3rd branchial arch arteries; ph, pharynx.
Foxn1 is required cell-autonomously for thymic epithelial cell differentiation (Nehls et al., 1994; Blackburn et al., 1996), but is not required for the initiation of thymus organogenesis (Nehls et al., 1996). Foxn1 is first expressed in the common primordia of thymus/parathyroid after it forms at around E11.5 and its expression marks the presumptive thymus-specific region in a complementary pattern to the parathyroid marker Gcm2 (Fig. 7C) (Gordon et al., 2001). In Eya1−/− embryos, the 3rd pouches do not separate from the pharyngeal endoderm as epithelial buds and no Foxn1 expression was detected in the 3rd pharyngeal region (Fig. 7D and data not shown). The absence of Foxn1 expression in the prospective region of the thymus/parathyroid primordia in Eya1−/− embryos further suggests that the formation of the primordia is not initiated in Eya1−/− embryos.
Abnormal induction of apoptosis of the pharyngeal surface ectoderm
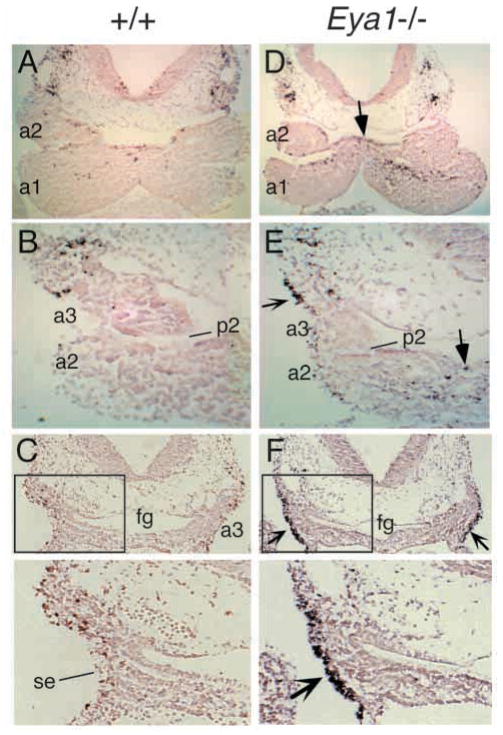
Since we have previously observed that Eya1 is required for cell survival in the otic epithelium and metanephric mesenchyme, we analyzed whether the hypoplasia of the pharyngeal arches and the defective formation of pharyngeal organs in Eya1−/− embryos could partly result from programmed cell death (apoptosis). Apoptotic cells were detected in the pouch endoderm and arch mesenchyme in wild-type embryos at E10.5–11.5 (Fig. 8A–C and data not shown). In Eya1−/− embryos, the number of apoptotic cells detected by TUNEL assay was slightly increased in the proximal region of the 1st and 2nd pharyngeal arches (arrows, Fig. 8D,E). However, no significant changes of apoptotic cells were found in the region where Eya1 is strongly expressed in the 1st and 2nd pharyngeal arches of Eya1−/−embryos (Fig. 8A,B,D,E). We have also not detected significant changes of apoptotic cells in the 3rd and 4th pharyngeal arch mesenchyme and pouch endoderm (Fig. 8B,C,E,F and data not shown), indicating that apoptosis was not altered in the 3rd and 4th pharyngeal arches and pouches in the absence of Eya1.
Fig. 8.
TUNEL analysis of transverse sections through the pharyngeal region in wild-type and Eya1−/− embryos at E10.5. (A–C) Apoptotic cells were observed in the pharyngeal arches (a) and pouches (p) in wild-type embryos. (D–F) In Eya1−/− embryos, there were more apoptotic cells in the proximal region of the 1st and 2nd arches (arrows in D,E, other data not shown). No significant changes of apoptotic cells were observed either in the neural crest mesenchyme or the pouch endoderm in the 3rd and 4th pharyngeal regions (E,F and other data not shown). Note that numerous apoptotic cells were detected in the surface ectoderm (se) of the 3rd (reflexed arrows in E,F) and 4th pharyngeal regions (data not shown). The panels below C and F are higher magnification of the boxed areas. Dorsal or cranial is up. fg, pharyngeal region of foregut.
In contrast, abnormal apoptosis was observed in the surface ectoderm including 3rd and 4th clefts in Eya1−/− embryos (reflexed arrows, Fig. 8E,F and data not shown). This indicates that Eya1 is required for the pharyngeal ectodermal cell survival.
DISCUSSION
Development of the thymus, parathyroid and thyroid normally occurs between E10.5 and E15.5 of mouse embryogenesis. In the absence of Eya1, the failure of thymus and parathyroid development occurs coincidently with the initial induction of organogenesis. Our data indicate that Eya1 is expressed and plays a role in the neural crest, endodermal and ectodermal cells that are required for the induction of thymus and parathyroid organogenesis.
The role of Eya1 in the development of neural crest-derived pharyngeal arch mesenchyme
Since the neural crest mesenchymal structures derived from the 3rd and 4th pharyngeal arches and pouches are affected similarly by neural crest ablation in chick, and by loss of Eya1, we conclude that Eya1 is required for normal neural crest development in pharyngeal arches (Bockman and Kirby, 1984; Bockman et al., 1989; Kuratani and Bockman, 1990). Eya1 is not expressed in premigratory neural crest cells and our marker analysis of Hoxa3 shows that there is no significant change of Hoxa3 expression in premigratory, migratory neural crest cells or in the 3rd and 4th pharyngeal arches in Eya1−/− embryos. Thus, the neural crest defects seen in Eya1 mutants must occur at the postmigratory stage. Interestingly, our analysis has shown that Six1, which is normally co-expressed with Eya1 in pharyngeal arch mesenchyme, was markedly reduced in Eya1−/− embryos. Besides the defects detected in neural crest mesenchyme of pharyngeal arches, Eya1−/− mice show multiple defects in cranial structures which are dependent on neural crest, including lack of multiple cranial ganglia and nerves, malformation of the skull, abnormal development of eyelids, middle ear ossicles, multiple skeleton components and other derivatives from the pharyngeal arches and pouches (including the parafollicular cells), cleft secondary palate formation, malformation of the tongue and heart (P-X. Xu, unpublished). Thus, Eya1 probably plays a critical role in neural crest development. The neural crest defects seen in Eya1−/− embryos may be caused by inappropriate cell proliferation. Consistent with this view, no abnormal cell death in both 3rd and 4th pharyngeal arches was observed. Nonetheless, the neural crest defect detected in the pharyngeal arches of Eya1−/− embryos could contribute to the defective formation of thymus and parathyroid.
A Pax-Eya-Six regulatory hierarchy in pharyngeal pouch endoderm
Since Eya1 is expressed in the 3rd pouch endoderm before the initiation of thymus and parathyroid formation and our marker gene analyses show that the expression of Six1 was undetectable in the Eya1−/− pouch endoderm, indicating that Eya1 also plays a role in the pouch endoderm. Our data also suggest that similar to the otic and kidney induction (Xu et al., 1999a), Eya1 functions in an evolutionarily conserved cassette downstream of Pax genes and upstream of Six genes during the development of pharyngeal pouch endoderm. It has been shown that deletion of Pax9 results in lack of 3rd and 4th pouches, while deletion of Pax1 results in parathyroid and thymus abnormalities at later stages (Dietrich and Gruss, 1995; Wallin et al., 1996; Peters et al., 1998; Su et al., 2001). In Eya1−/− pouch endoderm, Pax1 and Pax9 expression was not affected. We have also assayed Eya1 expression in Pax9 mutant embryos and found no significant changes of Eya1 expression in the Pax9-deficient pharyngeal pouch endoderm at E9.5 (data not shown). Although Pax1 apparently does not compensate for Pax9-deficiency during embryonic parathyroid and thymus formation, it is possible that Pax1 and Pax9 function redundantly in the pharyngeal pouches and that a critical threshold of Pax1/Pax9 protein expression in pharyngeal endoderm regulates Eya1 expression. Such compensatory up-regulation has been noted for Pax1 and Pax9 in the formation of other organs (Peters et al., 1999). Consistent with this genetic relationship between Pax1, Pax9 and Eya1, our data suggest that Eya1 also functions downstream of Hoxa3 in the 3rd and 4th pouch endoderm. Hoxa3 expression is unaffected in the 3rd and 4th pouch endoderm of Eya1 mutant embryos, and Pax1 and Pax9 expression is down-regulated in Hoxa3 mutants, which also lack parathyroid and thymus development (Chisaka and Capecchi, 1991; Manley and Capecchi, 1995). During parathyroid organogenesis, recent studies have shown that Hoxa3 is required for the initiation of Gcm2 expression in the 3rd pouch endoderm and both Hoxa3 and Pax1 are required for the maintenance of Gcm2 expression (Su et al., 2001) (S. Ellis, J. Koushik, J. Gordon and N. R. Manley, unpublished). Our data indicate that Gcm2 requires Eya1 function for its expression in the 3rd pouch endoderm before the formation of thymus/parathyroid primordia. Therefore, it is possible that Gcm2 expression is regulated by the Hoxa3-Pax1/9-Eya1 pathway during parathyroid organogenesis. Gcm2 expression is initiated normally at E9.5–10.5 in Pax1 single mutants (Su et al., 2001). This could be the result of Eya1 expression being correctly initiated in the 3rd pouch endoderm in Pax1 single mutants, similar to that seen in Pax9 single mutants (data not shown). The genetic relationship between Eya and Pax genes may be pertinent in other developmental contexts, since both Eya1 and Pax9 mutants exhibit related palate defects (Peters et al., 1998), and Pax9 expression was unaffected in Eya1 mutant palatal shelves (P.-X. Xu, unpublished). We are in the process of further clarifying the genetic relationship between Pax and Eya1 genes by examining the Eya1 expression in pharyngeal pouches and palatal shelves of Pax1/Pax9 double mutants, and in the otic epithelium and kidney of Pax2/Pax8 double mutants.
The role of Eya1 in the development of pharyngeal surface ectoderm
Since Eya1 is also expressed in the surface ectoderm, including that of the 2nd, 3rd and 4th clefts, and in the absence of Eya1 the surface ectoderm undergoes abnormal apoptosis, it would apppear that Eya1 also plays a role in the development of pharyngeal surface ectoderm.
Previous studies have demonstrated the importance of the ectoderm in the thymus development. It has been suggested that most of the ectoderm is formed by the proliferation of the 3rd cleft, which covers the endodermal part at around E11.0 (Cordier and Haumont, 1980). In addition, the cervical vesicle, formed by an invagination of the dorsal segment of the ectoderm between the 3rd and 4th pharyngeal clefts, also contributes to the formation of the thymus (Cordier and Haumont, 1980). In the chick embryo, after removal of the ectoderm of the 3rd and 4th clefts before it fuses with the pouch endoderm, the endoderm produces only a rudimentary thymus, while the parathyroid and the ultimobranchial body develop normally (Hammond, 1954). In Foxn1 mutant nude mice, the arrested development of the 3rd ectodermal cleft resulted in thymic dysgenesis, while the parathyroid develops normally (Cordier and Haumond, 1980). It has been proposed that the thymic dysgenesis in the nude mice is caused by the fact that the endoderm is no longer covered by the ectoderm and is deprived of its normal inducing agent (Cordier and Haumont, 1980). In Eya1−/− embryos, we found that Six1 expression in the surface ectoderm, including the 3rd cleft, was significantly reduced compared to wild-type embryos. This indicates that Eya1 may function along with Six1 in a molecular pathway within the ectoderm, which may provide inducing signals for the development of pouch endoderm. In addition, our data indicate that Eya1 is required for ectodermal cell survival. Thus, the thymic agenesis detected in Eya1−/− animals may partly result from the defects in the ectoderm.
Thyroid phenotype
The thyroid phenotype observed in Eya1 mutants does not occur as early as the defects in the thymus and parathyroid development. Although Eya1 mutant mice show reduction in the number of C cells in the thyroid, Eya1 is not required for the differentiation of either C or follicular cells. Interestingly, C cells are only observed in the persistent ultimobranchial bodies, while follicular cells are found in both the persistent ultimobranchial bodies and the thyroid lobes, similar to the phenotype observed in Hoxa3 mutant mice (Manley and Capecchi, 1995; Manley and Capecchi, 1998).
Previous studies have shown that Pax8 is required for the differentiation of thyroid follicular cells (Plachov et al., 1990; Mansouri et al., 1998). In Pax8−/− mice, the ultimobranchial bodies form and calcitonin-positive C cells are identified, however, both thyroglobulin and thyroperoxidase expression was not detected in Pax8−/− ultimobranchial bodies (Mansouri et al., 1998), indicating that there are no follicular cells. Previous studies have also shown that Pax8 and Ttf1 gene products cooperate to activate the expression of thyroglobulin gene in the follicular cells (Pasca di Magoliano et al., 2000). Interestingly, we have found that Pax8 expression is ectopically turned on in Eya1−/− ultimobranchial bodies at E14.5–15.5, when the thyroglobulin gene begins to be expressed (Lazzaro et al., 1991). This suggests that the ectopic expression of Pax8 may be required for the expression of thyroglobulin in the follicular cells of Eya1−/−ultimobranchial bodies. Consistent with this, H&E stained sections of Eya1−/− persistent ultimobranchial bodies showed the presence of colloid-containing follicles, indicating that thyroglobulin is produced and secreted into the lumen of the follicles (data not shown). Since Eya1 is not expressed in the thyroid primordia at E10.5–14.5, it is possible that the defect detected in the thyroid lobe is due to the lack of fusion with the ultimobranchial body.
In summary, our results suggest that the Eya1 protein level or activity critically affects the development of the thymus, parathyroid and thyroid and that Eya1 controls critical early inductive events that are required for the morphogenesis of mammalian thymus, parathyroid and thyroid.
Acknowledgments
We thank G. Carlson, K. Chiotti, and J. Gilchrist at McLaughlin Research Institute for help with the immunological analysis of Eya1 heterozygous mice and N. Manley for providing Gcm2 and Foxn1 probes. This work was partly supported by NSF IBN-0078246 and NIH P20RR 12345-02 (P-X. X.).
References
- Abdelhak S, Kalatzis V, Heilig R, Compain S, Samson D, Vincent C, et al. A human homologue of the Drosophila eyes absent gene underlies Branchio-Oto-Renal (BOR) syndrome and identifies a novel gene family. Nature Genet. 1997;15:157–164. doi: 10.1038/ng0297-157. [DOI] [PubMed] [Google Scholar]
- Akiyama Y, Hosoya T, Poole AM, Hotta Y. The gcm-motif: a novel DNA-binding motif conserved in Drosophila and mammals. Proc Natl Acad Sci USA. 1996;93:14912–14916. doi: 10.1073/pnas.93.25.14912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auerbach R. Morphogenetic interactions in the development of the mouse thymus gland. Dev Biol. 1960;2:271–284. doi: 10.1016/0012-1606(60)90009-9. [DOI] [PubMed] [Google Scholar]
- Blackburn CC, Augustine CL, Li R, Harvey RP, Malin MA, Boyd RL, Miller JF, Morahan G. The nu gene acts cell-autonomously and is required for differentiation of thymic epithelial progenitors. Proc Natl Acad Sci USA. 1996;93:5742–5746. doi: 10.1073/pnas.93.12.5742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bockman DE, Kirby ML. Dependence of thymus development on derivatives of the neural crest. Science. 1984;223:498–500. doi: 10.1126/science.6606851. [DOI] [PubMed] [Google Scholar]
- Bockman DE, Redmond ME, Kirby ML. Alteration of early vascular development after ablation of cranial neural crest. Anat Rec. 1989;225:209–217. doi: 10.1002/ar.1092250306. [DOI] [PubMed] [Google Scholar]
- Bonini NM, Leiserson WM, Benzer S. The eyes absent gene: genetic control of cell survival and differentiation in the developing Drosophila eye. Cell. 1993;72:379–395. doi: 10.1016/0092-8674(93)90115-7. [DOI] [PubMed] [Google Scholar]
- Borsani G, DeGrandi A, Ballabio A, Bulfone A, Bernard L, Banfi S, et al. Identification and characterization of EYA4, a novel mammalian homolog of the Drosophila melanogaster eyes absent gene. Hum Mol Genet. 1999;8:11–23. doi: 10.1093/hmg/8.1.11. [DOI] [PubMed] [Google Scholar]
- Brown JM, Wedden SE, Millburn GH, Robson LG, Hill RE, Davidson DR, Tickle C. Experimental analysis of the control of expression of the homeobox-gene Msx-1 in the developing limb and face. Development. 1993;119:41–48. doi: 10.1242/dev.119.Supplement.41. [DOI] [PubMed] [Google Scholar]
- Chen R, Amoui M, Zhang Z, Mardon G. Dachshund and eyes absent proteins form a complex and function synergistically to induce ectopic eye formation in Drosophila. Cell. 1997;91:893–903. doi: 10.1016/s0092-8674(00)80481-x. [DOI] [PubMed] [Google Scholar]
- Chisaka O, Capecchi MR. Regionally restricted developmental defects resulting from targeted disruption of the mouse homeobox gene hox-1.5. Nature. 1991;350:473–479. doi: 10.1038/350473a0. [DOI] [PubMed] [Google Scholar]
- Cordier AC, Haumont SM. Development of thymus, parathyroids, and ultimobranchial bodies in NMRI and nude mice. Am J Anat. 1980;157:227–263. doi: 10.1002/aja.1001570303. [DOI] [PubMed] [Google Scholar]
- Dahl E, Koseki H, Balling R. Pax genes and organogenesis. BioEssays. 1997;19:755–765. doi: 10.1002/bies.950190905. [DOI] [PubMed] [Google Scholar]
- Dietrich S, Gruss P. Undulated phenotypes suggest a role of Pax-1 for the development of vertebral and extravertebral structures. Dev Biol. 1995;167:529–548. doi: 10.1006/dbio.1995.1047. [DOI] [PubMed] [Google Scholar]
- Duncan MK, Kos L, Jenkins NA, Gilbert DJ, Copeland NG, Tomarev SI. Eyes absent: a gene family found in several metazoan phyla. Mamm Genome. 1997;8:479–485. doi: 10.1007/s003359900480. [DOI] [PubMed] [Google Scholar]
- Dupe V, Ghyselinck NB, Wendling O, Chambon P, Mark M. Key roles of retinoic acid receptors alpha and beta in the patterning of the caudal hindbrain, pharyngeal arches and otocyst in the mouse. Development. 1999;126:5051–5059. doi: 10.1242/dev.126.22.5051. [DOI] [PubMed] [Google Scholar]
- Fontaine J. Multiple migration of calcitonin cell precursors during ontogeny of the mouse pharynx. Gen Comp Endocrinol. 1979;37:482–490. doi: 10.1016/0016-6480(79)90049-2. [DOI] [PubMed] [Google Scholar]
- Frohman MA, Boyle M, Martin GR. Isolation of the mouse Hox-2.9 gene; analysis of embryonic expression suggests that positional information along the anterior-posterior axis is specified by mesoderm. Development. 1990;110:589–607. doi: 10.1242/dev.110.2.589. [DOI] [PubMed] [Google Scholar]
- Gordon J, Bennett AR, Blackburn CC, Manley NR. Gcm2 and Foxn1 mark early parathyroid- and thymus-specific domains in the developing third pharyngeal pouch. Mech Dev. 2001;103:141–143. doi: 10.1016/s0925-4773(01)00333-1. [DOI] [PubMed] [Google Scholar]
- Gunther T, Chen ZF, Kim J, Priemel M, Rueger JM, Amling M, Moseley JM, Martin TJ, Anderson DJ, Karsenty G. Genetic ablation of parathyroid glands reveals another source of parathyroid hormone. Nature. 2000;406:199–203. doi: 10.1038/35018111. [DOI] [PubMed] [Google Scholar]
- Hammond WS. Origin of thymus in the chick embryos. J Morphol. 1954;95:501–521. [Google Scholar]
- Hilfer SR. Cellular interactions in morphogenesis of the thyroid gland. Am Zool. 1968;8:273–284. doi: 10.1093/icb/8.2.273. [DOI] [PubMed] [Google Scholar]
- Hilfer SR, Brown JW. The Development of pharyngeal endocrine organs in mouse and chick embryos. Scan Electron Microsc. 1984;(Pt 4):2009–2022. [PubMed] [Google Scholar]
- Kalatzis V, Sahly I, El-Amraoui A, Petit C. Eya1 expression in the developing ear and kidney: Towards the understanding of the pathogenesis of Branchio-Oto-Renal (BOR) syndrome. Dev Dyn. 1998;213:486–499. doi: 10.1002/(SICI)1097-0177(199812)213:4<486::AID-AJA13>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Kim J, Jones BW, Zock C, Chen Z, Wang H, Goodman CS, Anderson DJ. Isolation and characterization of mammalian homologs of the Drosophila gene glial cells missing. Proc Natl Acad Sci USA. 1998;95:12364–12369. doi: 10.1073/pnas.95.21.12364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura S, Hara Y, Pineau T, Fernandez-Salguero P, Fox CH, Ward JM, Gonzalez FJ. The T/ebp null mouse: thyroid-specific enhancer-binding protein is essential for the organogenesis of the thyroid, lung, ventral forebrain, and pituitary. Genes Dev. 1996;10:60–69. doi: 10.1101/gad.10.1.60. [DOI] [PubMed] [Google Scholar]
- Krumlauf R. Hox genes in vertebrate development. Cell. 1994;78:191–201. doi: 10.1016/0092-8674(94)90290-9. [DOI] [PubMed] [Google Scholar]
- Kuratani S, Bockman DE. The participation of neural crest derived mesenchymal cells in development of the epithelial primordium of the thymus. Arch Histol Cytol. 1990;53:267–273. doi: 10.1679/aohc.53.267. [DOI] [PubMed] [Google Scholar]
- Lazzaro D, Price M, de Felice M, Di Lauro R. The transcription factor TTF-1 is expressed at the onset of thyroid and lung morphogenesis and in restricted regions of the foetal brain. Development. 1991;113:1093–1104. doi: 10.1242/dev.113.4.1093. [DOI] [PubMed] [Google Scholar]
- LeDouarin N. Thymus Ontogeny Studied in Interspecific Chimeras. In: Cooper M, Dayton D, editors. Development of Host Defenses. New York: Raven Press; 1977. pp. 107–114. [Google Scholar]
- LeDouarin NM, Joterean FV. Tracing of cells of the avian thymus through embryonic life in interspecific chimeras. J Exp Med. 1975;142:17–40. doi: 10.1084/jem.142.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeDouarin NM, Dieterlen-Lievre F, Oliver PD. Ontogeny of primary lymphoid organs and lymphoid stem cells. Am J Anat. 1984;170:261–299. doi: 10.1002/aja.1001700305. [DOI] [PubMed] [Google Scholar]
- LeLievre CS, LeDouarin NM. Mesenchymal derivatives of the neural crest: analysis of chimeric quail and chick embryos. J Embryol Exp Morphol. 1975;34:125–154. [PubMed] [Google Scholar]
- Manley N. Thymus organogenesis and molecular mechanisms of thymic epithelial cell differentiation. Semin Immunol. 2000;12:421–428. doi: 10.1006/smim.2000.0263. [DOI] [PubMed] [Google Scholar]
- Manley N, Capecchi MR. The role of HoxA3 in mouse thymus and thyroid development. Development. 1995;121:1989–2003. doi: 10.1242/dev.121.7.1989. [DOI] [PubMed] [Google Scholar]
- Manley N, Capecchi MR. Hox group 3 paralogs regulate the development and migration of the thymus, thyroid, and parathyroid glands. Dev Biol. 1998;195:1–15. doi: 10.1006/dbio.1997.8827. [DOI] [PubMed] [Google Scholar]
- Mansouri A, Chowdhury K, Gruss P. Follicular cells of the thyroid gland require Pax8 gene function. Nat Genet. 1998;19:87–90. doi: 10.1038/ng0598-87. [DOI] [PubMed] [Google Scholar]
- Maurer F. Die Schlundspalten-derivate von Echidda. Anat Anz Ergazungshefte zu bd. 1899;16:S. 88–101. [Google Scholar]
- Moore MA, Owen JJ. Experimental studies on the development of the thymus. J Exp Med. 1967;126:715–726. doi: 10.1084/jem.126.4.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moseley JM, Matthews EW, Breed RH, Galante L, Tse A, MacIntyre I. The ultimobranchial origin of calcitonin. Lancet. 1968;1:108–110. doi: 10.1016/s0140-6736(68)92720-7. [DOI] [PubMed] [Google Scholar]
- Neubuser A, Koseki H, Balling R. Characterization and developmental expression of Pax9, a paired-box-containing gene related to Pax1. Dev Biol. 1995;170:701–716. doi: 10.1006/dbio.1995.1248. [DOI] [PubMed] [Google Scholar]
- Noden DM. The embryonic origins of avian cephalic and cervical muscles and associated connective tissues. Am J Anat. 1983;168:257–276. doi: 10.1002/aja.1001680302. [DOI] [PubMed] [Google Scholar]
- Noden DM. Craniofacial development: new views on old problems. Anat Rec. 1984;208:1–13. doi: 10.1002/ar.1092080103. [DOI] [PubMed] [Google Scholar]
- Oliver G, Wehr R, Jenkins NA, Copeland NG, Cheyette BNR, Hartenstein V, Zipersky SL, Gruss P. Homeobox genes and connective tissue patterning. Development. 1995a;121:693–705. doi: 10.1242/dev.121.3.693. [DOI] [PubMed] [Google Scholar]
- Oliver G, Mailhos A, Wehr R, Copeland NG, Jenkins NA, Gruss P. Six3, a murine homologue of the sine oculis gene, demarcates the most anterior border of the developing neural plate and is expressed during eye development. Development. 1995b;121:4045–4055. doi: 10.1242/dev.121.12.4045. [DOI] [PubMed] [Google Scholar]
- Owen JJ, Ritter MA. Tissue interaction in the development of thymus lymphocytes. J Exp Med. 1969;129:431–436. doi: 10.1084/jem.129.2.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owen JJ, McLoughlin DE, Suniara RK, Jenkinson EJ. The role of mesenchyme in thymus development. Curr Top Microbiol Immunol. 2000;251:33–37. doi: 10.1007/978-3-642-57276-0_17. [DOI] [PubMed] [Google Scholar]
- Pasca di Magliano M, Di Lauro R, Zannini M. Pax8 has a key role in thyroid cell differentiation. Proc Natl Acad Sci. 2000;97:13144–13149. doi: 10.1073/pnas.240336397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearse AG, Carvalheira AF. Cytochemical evidence for an ultimobranchial origin of rodent thyroid C cells. Nature. 1967;214:929–930. doi: 10.1038/214929a0. [DOI] [PubMed] [Google Scholar]
- Pearse AG, Polak JM. Cytochemical evidence for the neural crest origin of mammalian ultimobranchial C cells. Histochemie. 1971;27:96–102. doi: 10.1007/BF00284951. [DOI] [PubMed] [Google Scholar]
- Peters H, Neubüser A, Kratochwil K, Balling R. Pax9-deficient mice lack pharyngeal pouch derivatives and teeth and exhibit craniofacial and limb abnormalities. Genes Dev. 1998;12:2735–2747. doi: 10.1101/gad.12.17.2735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters H, Wilm B, Sakai N, Imai K, Maas R, Balling R. Pax1 and Pax9 synergistically regulate vertebral column development. Development. 1999;126:5399–5408. doi: 10.1242/dev.126.23.5399. [DOI] [PubMed] [Google Scholar]
- Pignoni F, Hu B, Zavitz KH, Xiao J, Garrity PA, Zipursky SL. The eye-specification proteins So and Eya form a complex and regulate multiple steps in Drosophila eye development. Cell. 1997;91:881–891. doi: 10.1016/s0092-8674(00)80480-8. [DOI] [PubMed] [Google Scholar]
- Plachov D, Chowdhury K, Walther C, Simon D, Guenet JL, Gruss P. Pax8, a murine paired box gene expressed in the developing excretory system and thyroid gland. Development. 1990;110:643–651. doi: 10.1242/dev.110.2.643. [DOI] [PubMed] [Google Scholar]
- Rogers WM. The fate of ultimobranchial body in the white rat. Am J Anat. 1927;38:349–477. [Google Scholar]
- Rogers WM. Normal and anomalous development of the thyroid. In: Werner SC, Ingbar SH, editors. The Thyroid. New York: Harper and Row; 1971. pp. 303–317. [Google Scholar]
- Smith C. Studies on the thymus of the mammal XIV: Histology and histochemistry of embryonic and early postnatal thymuses of C57BL/6 and AKR strain mice. Am J Anat. 1965;116:611–630. doi: 10.1002/aja.1001160307. [DOI] [PubMed] [Google Scholar]
- Strachan T, Read AP. PAX genes. Curr Opin Gen Dev. 1994;4:427–438. doi: 10.1016/0959-437x(94)90032-9. [DOI] [PubMed] [Google Scholar]
- Stuart ET, Kioussi C, Gruss P. Mammalian Pax genes. Ann Rev Genet. 1994;28:219–236. doi: 10.1146/annurev.ge.28.120194.001251. [DOI] [PubMed] [Google Scholar]
- Su D, Ellis S, Napier A, Lee K, Manley NR. Hoxa3 and Pax1 regulate epithelial cell death and proliferation during thymus and parathyroid organogenesis. Dev Biol. 2001;236:316–329. doi: 10.1006/dbio.2001.0342. [DOI] [PubMed] [Google Scholar]
- Wallin J, Eibel H, Neubuser A, Wilting J, Koseki H, Balling R. Pax1 is expressed during development of the thymus epithelium and is required for normal T-cell maturation. Development. 1996;122:23–30. doi: 10.1242/dev.122.1.23. [DOI] [PubMed] [Google Scholar]
- Wang Y, Sassoon D. Ectoderm-mesenchyme and mesenchyme-mesenchyme interactions regulate Msx-1 expression and cellular differentiation in the murine limb bud. Dev Biol. 1995;168:374–382. doi: 10.1006/dbio.1995.1087. [DOI] [PubMed] [Google Scholar]
- Wendling O, Dennefeld C, Chambon P, Mark M. Retinoid signaling is essential for patterning the endoderm of the third and fourth pharyngeal arches. Development. 2000;127:1553–1562. doi: 10.1242/dev.127.8.1553. [DOI] [PubMed] [Google Scholar]
- Wilkinson DG, Bhatt S, McMahon AP. Expression pattern of the fgf-related proto-oncogene int-2 suggests multiple roles in fetal development. Development. 1989;105:131–136. doi: 10.1242/dev.105.1.131. [DOI] [PubMed] [Google Scholar]
- Xu PX, Woo I, Her H, Beier D, Maas RL. Mouse homologues of the Drosophila eyes absent gene require Pax6 for expression in lens and nasal placodes. Development. 1997a;124:219–231. doi: 10.1242/dev.124.1.219. [DOI] [PubMed] [Google Scholar]
- Xu PX, Cheng J, Epstein JA, Maas R. Mouse Eya genes are expressed during limb tendon development and encode a transcriptional activation function. Proc Natl Acad Sci USA. 1997b;94:11974–11979. doi: 10.1073/pnas.94.22.11974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu PX, Adams J, Peters H, Brown MC, Heaney S, Maas R. Eya1-deficient mice lack ears and kidneys and show abnormal apoptosis of organ primordia. Nat Genet. 1999a;23:113–117. doi: 10.1038/12722. [DOI] [PubMed] [Google Scholar]
- Xu PX, Zhang X, Heaney S, Yoon A, Michelson A, Maas RL. Regulation of Pax6 expression is conserved between mice and flies. Development. 1999b;126:383–395. doi: 10.1242/dev.126.2.383. [DOI] [PubMed] [Google Scholar]
- Zimmerman JE, Bui QT, Steingrimsson E, Nagle DL, Fu W, Genin A, Spinner NB, Copeland NG, Jenkins NA, Bucan M, Bonini NM. Cloning and characterization of two vertebrate homologs of the Drosophila eyes absent gene. Genome Res. 1997;7:128–141. doi: 10.1101/gr.7.2.128. [DOI] [PubMed] [Google Scholar]