Abstract
The multisubunit enzyme aspartate transcarbamylase consists of six copies of two types of polypeptide chains, catalytic (C) and regulatory (R). A complex formed by the partial dissociation of this enzyme has been isolated. This species, which has the structure C6R4, is a likely intermediate in the stepwise dissociation of aspartate transcarbamylase induced by mercurials. The formation of the complex is the result of the release of a single regulatory dimer (R2) from the parent molecule.
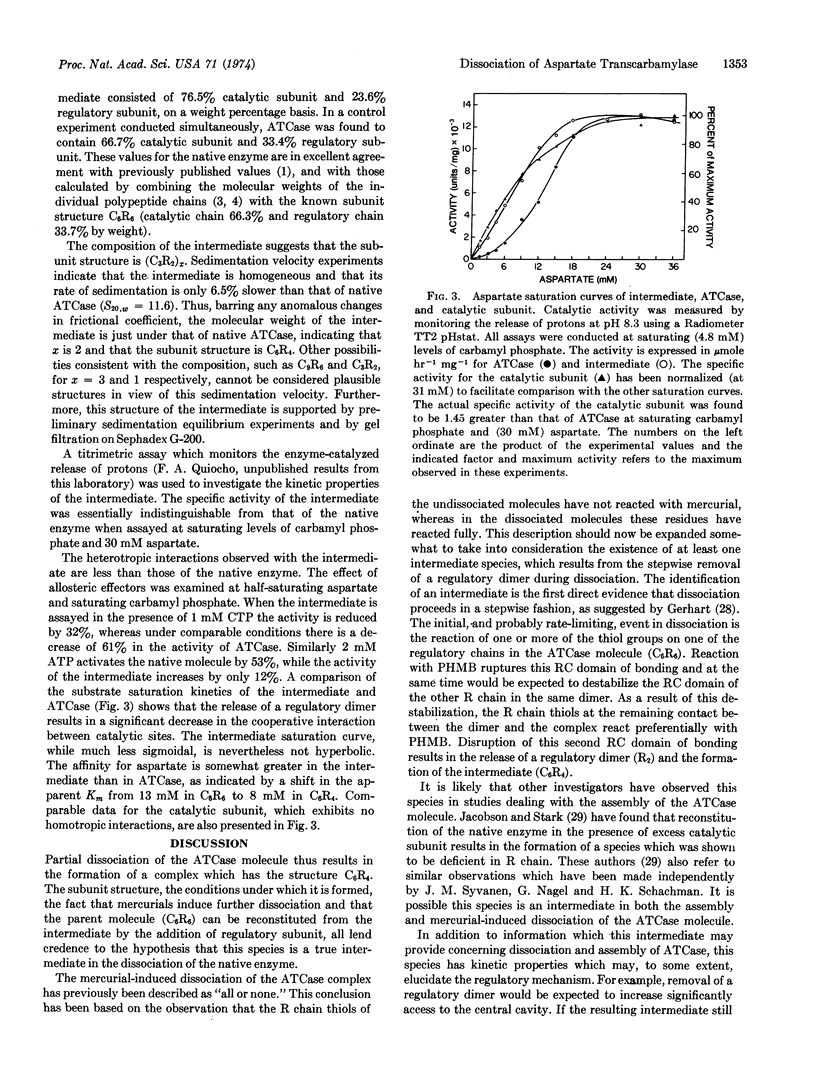
The specific activity of the intermediate is essentially the same as that of aspartate transcarbamylase. By contrast, both homotropic and heterotropic interactions are reduced, but not abolished. These observations suggest that the allosteric transitions involved in the control mechanisms do not require the intact structure C6R6.
Keywords: thiols, mercurials, dissociation, central cavity, intermediate
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buckman T. Spin-labeling studies of aspartate transcarbamylase. I. Effects of nucleotide binding and subunit separation. Biochemistry. 1970 Aug 4;9(16):3255–3265. doi: 10.1021/bi00818a020. [DOI] [PubMed] [Google Scholar]
- CORI C. F., MADSEN N. B. The interaction of muscle phosphorylase with p-chloromercuribenzoate. I. Inhibition of activity and effect on the molecular weight. J Biol Chem. 1956 Dec;223(2):1055–1065. [PubMed] [Google Scholar]
- Changeux J. P., Rubin M. M. Allosteric interactions in aspartate transcarbamylase. 3. Interpretation of experimental data in terms of the model of Monod, Wyman, and Changeux. Biochemistry. 1968 Feb;7(2):553–561. doi: 10.1021/bi00842a601. [DOI] [PubMed] [Google Scholar]
- Cohlberg J. A., Pigiet V. P., Jr, Schachman H. K. Structure and arrangement of the regulatory subunits in aspartate transcarbamylase. Biochemistry. 1972 Aug 29;11(18):3396–3411. doi: 10.1021/bi00768a013. [DOI] [PubMed] [Google Scholar]
- Cook R. A. Subunit interactions in aspartate transcarbamylase. Biochemistry. 1972 Sep 26;11(20):3792–3797. doi: 10.1021/bi00770a019. [DOI] [PubMed] [Google Scholar]
- Evans D. R., McMurray C. H., Lipscomb W. N. The thiol group in the catalytic chains of aspartate transcarbamoylase. Proc Natl Acad Sci U S A. 1972 Dec;69(12):3638–3642. doi: 10.1073/pnas.69.12.3638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. R., Warren S. G., Edwards B. F., McMurray C. H., Bethge P. H., Wiley D. C., Lipscomb W. N. Aqueous central cavity in aspartate transcarbamylase from Escherichia coli. Science. 1973 Feb 16;179(4074):683–685. doi: 10.1126/science.179.4074.683. [DOI] [PubMed] [Google Scholar]
- GERHART J. C., PARDEE A. B. ASPARTATE TRANSCARBAMYLASE, AN ENZYME DESIGNED FOR FEEDBACK INHIBITION. Fed Proc. 1964 May-Jun;23:727–735. [PubMed] [Google Scholar]
- GERHART J. C., PARDEE A. B. The enzymology of control by feedback inhibition. J Biol Chem. 1962 Mar;237:891–896. [PubMed] [Google Scholar]
- Gerhart J. C., Schachman H. K. Allosteric interactions in aspartate transcarbamylase. II. Evidence for different conformational states of the protein in the presence and absence of specific ligands. Biochemistry. 1968 Feb;7(2):538–552. doi: 10.1021/bi00842a600. [DOI] [PubMed] [Google Scholar]
- Gerhart J. C., Schachman H. K. Distinct subunits for the regulation and catalytic activity of aspartate transcarbamylase. Biochemistry. 1965 Jun;4(6):1054–1062. doi: 10.1021/bi00882a012. [DOI] [PubMed] [Google Scholar]
- Griffin J. H., Rosenbusch J. P., Blout E. R., Weber K. K. Conformational changes in aspartate transcarbamylase. II. Circular dichroism evidence for the involvement of metal ions in allosteric interactions. J Biol Chem. 1973 Jul 25;248(14):5057–5062. [PubMed] [Google Scholar]
- HIMES R. H., RABINOWITZ J. C. Formyltetrahydrofolate synthetase. II. Characteristics of the enzyme and the enzymic reaction. J Biol Chem. 1962 Sep;237:2903–2914. [PubMed] [Google Scholar]
- Jacobson G. R., Stark G. R. Aspartate transcarbamylase. A study of possible roles for the sulfhydryl group at the active site. J Biol Chem. 1973 Dec 10;248(23):8003–8014. [PubMed] [Google Scholar]
- Keresztes-Nagy S., Klapper M. H., Lazer L., Klotz I. M. Hybridization experiments: evidence of dissociation equilibrium in hemerythrin. Science. 1965 Oct 15;150(3694):357–359. doi: 10.1126/science.150.3694.357. [DOI] [PubMed] [Google Scholar]
- Markus G., McClintock D. K., Bussel J. B. Conformational changes in aspartate transcarbamylase. 3. A functional model for allosteric behavior. J Biol Chem. 1971 Feb 10;246(3):762–771. [PubMed] [Google Scholar]
- Matsumoto S., Hammes G. G. An equilibrium binding study of the interaction of aspartate transcarbamylase with cytidine 5'-triphosphate and adenosine 5'-triphosphate. Biochemistry. 1973 Mar 27;12(7):1388–1394. doi: 10.1021/bi00731a019. [DOI] [PubMed] [Google Scholar]
- Meighen E. A., Pigiet V., Schachman H. K. Hybridization of native and chemically modified enzymes. 3. The catalytic subunits of aspartate transcarbamylase. Proc Natl Acad Sci U S A. 1970 Jan;65(1):234–241. doi: 10.1073/pnas.65.1.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelbach M. E., Pigiet V. P., Jr, Gerhart J. C., Schachman H. K. A role for zinc in the quaternary structure of aspartate transcarbamylase from Escherichia coli. Biochemistry. 1972 Feb 1;11(3):315–327. doi: 10.1021/bi00753a002. [DOI] [PubMed] [Google Scholar]
- Richards K. E., Williams R. C. Electron microscopy of aspartate transcarbamylase and its catalytic subunit. Biochemistry. 1972 Aug 29;11(18):3393–3395. doi: 10.1021/bi00768a012. [DOI] [PubMed] [Google Scholar]
- Rosenbusch J. P., Weber K. Localization of the zinc binding site of aspartate transcarbamoylase in the regulatory subunit. Proc Natl Acad Sci U S A. 1971 May;68(5):1019–1023. doi: 10.1073/pnas.68.5.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenbusch J. P., Weber K. Subunit structure of aspartate transcarbamylase from Escherichia coli. J Biol Chem. 1971 Mar 25;246(6):1644–1657. [PubMed] [Google Scholar]
- Warren S. G., Edwards B. F., Evans D. R., Wiley D. C., Lipscomb W. N. Aspartate transcarbamoylase from Escherichia coli: electron density at 5.5 A resolution. Proc Natl Acad Sci U S A. 1973 Apr;70(4):1117–1121. doi: 10.1073/pnas.70.4.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K. Aspartate transcarbamylase from Escherichia coli. Characterization of the polypeptide chains by molecular weight, amino acid composition, and amino-terminal residues. J Biol Chem. 1968 Feb 10;243(3):543–546. [PubMed] [Google Scholar]
- Weber K. New structural model of E. coli aspartate transcarbamylase and the amino-acid sequence of the regulatory polypeptide chain. Nature. 1968 Jun 22;218(5147):1116–1119. doi: 10.1038/2181116a0. [DOI] [PubMed] [Google Scholar]
- Wiley D. C., Evans D. R., Warren S. G., McMurray C. H., Edwards B. F., Franks W. A., Lipscomb W. N. The 5.5 Angstrom resolution structure of the regulatory enzyme, asparate transcarbamylase. Cold Spring Harb Symp Quant Biol. 1972;36:285–290. doi: 10.1101/sqb.1972.036.01.038. [DOI] [PubMed] [Google Scholar]
- Wiley D. C., Lipscomb W. N. Crystallographic determination of symmetry of aspartate transcarbamylase. Nature. 1968 Jun 22;218(5147):1119–1121. doi: 10.1038/2181119a0. [DOI] [PubMed] [Google Scholar]
- Winlund C. C., Chamberlin M. J. Binding of cytidine triphosphate to aspartate transcarbamylase. Biochem Biophys Res Commun. 1970 Jul 13;40(1):43–49. doi: 10.1016/0006-291x(70)91043-0. [DOI] [PubMed] [Google Scholar]
- von Fellenberg R., Bethell M. R., Jones M. E., Levine L. Immunological studies of aspartate transcarbamylase. II. Effect of ligands on the conformation of the enzyme. Biochemistry. 1968 Dec;7(12):4322–4329. doi: 10.1021/bi00852a026. [DOI] [PubMed] [Google Scholar]