Abstract
This conference commemorates, almost to the day, the 45th anniversary of the discovery of the Na+/Ca2+ exchanger (NCX). The discovery was serendipitous, as is so often the case with scientific breakthroughs. Indeed, that is what is so fascinating and romantic about scientific research. I will describe the discovery of NCX, but will begin by explaining how I got there, and will then discuss how the discovery altered my career path.
“Sit down before fact as a little child, be prepared to give up every conceived notion, follow humbly wherever, whatever abysses nature leads, or you will learn nothing”
Thomas H. Huxley, letter to C. Kingsley, September 20, 1863
For the love of physiology
I was introduced to cell physiology by Howard Schneiderman, a distinguished insect physiologist and developmental biologist, during my undergraduate days at Cornell University. I was interested in neurophysiology and the mind-brain problem, but came under the spell of Daniel Tosteson when I was a medical student at Washington University in St. Louis. Dan convinced me to work on the Na+,K+-ATPase (‘sodium pump’), which had just been discovered (Skou, 1957). I spent a year-and-a-half in Dan’s lab studying the red blood cell cardiotonic steroid-sensitive Na+ pump. In 1963, after completing medical school and an internship at Boston City Hospital, I was offered a naval commission to work at the US Naval Medical Research Institute in Bethesda, MD (much better than a tour in Vietnam!). Thus, I returned to neurophysiology and, under David Goldman (of the Goldman-Hodgkin Katz equation), I studied the effects of divalent cations and anesthetics on lobster nerve conduction (Blaustein and Goldman, 1966; Blaustein, 1968). I also was fortunate to spend a few weeks at Woods Hole with John Moore and Toshio Narahashi working on tetrodotoxin’s action on squid axons (Moore et al., 1967).
I was planning to continue my career in cellular neurophysiology, and arranged for a position in Alan Hodgkin’s laboratory in Cambridge, England, with a Special Fellowship from the NIH. My family and I arrived in Cambridge in late August of 1966. After a family trip to Vienna for the International Congress of Biophysics, I left my wife Ellen and our two children (ages 3 and 5) in Cambridge, and headed off to the Laboratory of the Marine Biological Association in Plymouth, England, for the Fall squid season.
“All hands to the pump”
My expectation was to study squid axon electrophysiology, but Peter Baker, Alan’s junior associate, a lecturer at Emmanuel College, Cambridge, took a mini-sabbatical that Fall, and he wanted all Plymouth squid researchers to work on the Na+ pump. I was paired up with Rick Steinhardt, Richard Keynes’ postdoctoral fellow, and we were tasked with studying the activation of the Na+ pump by external cations. Richard came to Plymouth at the end of September; he showed us how to dissect squid axons (not knowing of my prior experience) and how to measure 22Na+ efflux after injecting the giant axons (0.8-1.2 mm diameter) with a microsyringe that he and Alan designed (Hodgkin and Keynes, 1956). Richard then went off to Homburg (Saar), Germany, to teach in a course on membrane biophysics organized by Hermann Passow and Robert Stampfli (more about this later).
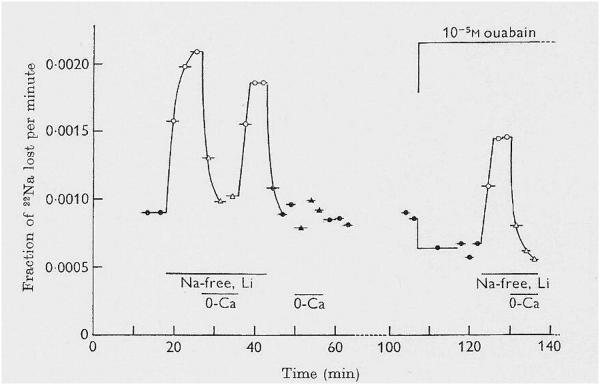
Rick and I began our 22Na+ efflux experiments on squid axons, and we rapidly identified a component that depended on external K+, and was blocked by ouabain, i.e., the Na+ pump component. When we removed external Na+ and K+ (Nao and Ko, respectively), preparatory to adding back one monovalent cation at a time, we exposed a very large 22Na+ efflux that did not depend upon Ko, and was not blocked by ouabain (Figure 1). This efflux persisted when the external NaCl and KCl were replaced by sucrose, LiCl or choline Cl−. We consulted with Peter Baker, who agreed that internal Na+ could be exiting with an anion, or exchanging for a cation. It was easiest to remove the other external cations, Ca2+ and Mg2+, so we first removed the Ca2+ - et, Voila! The large Na+ efflux was reversibly abolished; i.e., the Na+ efflux was external Ca2+-dependent (Figure 1). Removal of external Mg2+ had negligible effect on the Na+ efflux, so we had our answer: Na/Ca exchange! This was just the first month of my fellowship!
Figure 1.
Reduction of [Na+]o activates a large Cao-dependent, ouabain-resistant 22Na+ efflux in squid axons with high [Na+]i. Replacement of 460 mM NaCl in the artificial sea water (ASW) by LiCl greatly increased 22Na+ efflux, measured as the fraction of 22Na+ lost per minute. The increment was abolished by removal of the 11 mM CaCl2 in the ASW (MgCl2 was increased from 55 to 66 mM). This is evidence of “Nao-Cao antagonism”, and indirect evidence for Na+/Ca2+ exchange. Ouabain (10−5 M) reduced the 22Na+ efflux in control Na ASW (i.e., it inhibited the Na+ pump), but it had no effect on the large Cao-dependent 22Na+ efflux in Li ASW; right). Reprinted from Baker et al., 1969, with permission.
Peter recalled reading Ralph Niedergerke’s articles on Na+-Ca2+ interactions in frog cardiac muscle, and he suggested that they might be of interest. There was no time to read, however: we were working 14-16 hr days during October because I feared that a gale would interrupt our daily squid supply; the large squid did not survive in the relatively small holding tanks. In fact, the squid usually were injured in the nets and would die before the collection boat docked. Therefore, as soon as squid were caught, the fishermen removed the head and internal organs, and placed the mantle (containing the giant axons) in a thermos of iced sea water. With the Na+ pumps thus turned off, the axons slowly gained Na+; the axons didn’t last overnight. As we shall see, this rise in the intracellular Na+ concentration, [Na+]i, was fortuitous for the Ca2+ influx experiments.
In the meantime, we ordered some 45Ca2+ to test the Na+/Ca2+ exchange idea directly. The 45Ca2+ arrived the first week in November, but just before that, we had a gale. Finally, I had a chance to catch my breath. It was a miserable, stormy afternoon, and the laboratory building was deserted; I completed my data analysis for the last experiments, and sauntered down the hall to the library. As soon as I started to read the description (Luttgau and Niedergerke, 1958) of extracellular Na+-Ca2+ ([Na+]o-[Ca2+]o) antagonism and its influence on frog cardiac contraction (reduced [Na+]o induces cardiac contraction), I got very excited. I immediately recognized that NCX must be widely distributed in both tissues and species, including vertebrate heart. Therefore, since NCX apparently functions in the heart, it is the missing link to the puzzle that had stumped me ever since my first studies on the Na+ pump and, as an intern, my use of digitalis to treat patients with heart failure: How does Na+ pump inhibition by cardiotonic steroids increase the force of contraction of the heart? Because of both my clinical and research experiences, I frequently thought about this enigma. Here was the answer: raising [Na+]i promotes net Ca2+ gain by NCX, and thereby enhances cardiac contraction. That ‘Eureka! moment’ was even more thrilling than the discovery of NCX itself. I was, for a brief time, the only one in the world who understood how cardiotonic steroids enhance cardiac contraction! I was so exhilarated that I went off, alone, to the nearby Green Lantern restaurant, for a fine celebratory dinner with a bottle of claret. Then, slightly inebriated, I returned to the lab to re-read Luttgau-Niedergerke, to be sure I wasn’t delusional. It was a great day!
45Ca2+ flux studies: verification of Na+/Ca2+ exchange
The following Monday afternoon, Alan Hodgkin came down to Plymouth to see how I was getting on. After we dissected a few axons for the evening’s 22Na+ efflux experiments, he and I went to dinner with Trevor Shaw, another Plymouth squidder. We talked about the NCX, including my explanation of how Na+ pump inhibitors exert their cardiotonic effect. Alan asked a few questions but was, otherwise, impassive. I was crestfallen. How could he fail to be enthused by the story? Two days later, however, Alan asked me if I would mind if he remained in Plymouth to perform the 45Ca2+ flux experiments with me. Would I mind? I was ecstatic! We had won him over.
Alan and I performed the first influx experiments the next Monday. Axons were incubated for 1 hr in artificial sea water (ASW, the external fluid) containing either NaCl or LiCl as the predominant salt, and labeled with 45Ca2+. The axons were then washed in tracer-free solution, and the axoplasm was extruded and weighed, and 45Ca activity was measured in an old Panax counter with a Nixie tube display. After the first sample from NaCl ASW, with a low count, axoplasm from a LiCl ASW axon was counted: the Nixie tubes lit up with a very high count. Because [Na+]i was high in these axons from refrigerated mantles, the differences between the Ca2+ influx from Na+ ASW and Li+ ASW were large, and easy to detect. Alan’s eyes twinkled as he pulled his pipe from his mouth and broke into a broad grin. Reduction of [Na+]o increased Ca2+ influx (Table 1). Na+/Ca2+ exchange was confirmed! At 2 am, after measuring several replicate samples, we celebrated with a little “medicinal”Scotch before heading off to bed. Another great day!
Table 1.
Effects of external cations and of ouabain on 45Ca2+ influx in squid axons. Reduction of [Na+]o increased 45Ca2+ influx. This is evidence of “Nao-Cao antagonism”. The increase in Ca2+ influx was much greater in the axons from refrigerated squid mantles (with a high [Na+]i) than in axons from live squid (with a lower [Na+]i). This is direct evidence of Na+/Ca2+ exchange. Data from Baker et al., 1969.
| External solution |
|
n | |
| A. Axons from refrigerated mantles |
| Collected results*: |
| Na sea water, 0 and 10-K, ± ouabain | 0.15 ± 0.02 | 0.04 − 0.57 | 30 |
| Li sea water, 0 and 10-K, ± ouabain | 4.33 ± 0.43 | 0.90 − 9.50 | 29 |
| Dextrose sea water, 0 and 10-K, ± ouabain | 2.46 ± 0.29 | 0.80 − 5.30 | 15 |
| B. Axons from live squid |
| 10-K, Na sea water | 0.23 | – | 1 |
| 10-K, Li sea water | 0.66 ± 0.48 | 0.16 − 1.60 | 3 |
n is the number of axons in each group.,
Li sea water contained 460 mM LiCl in place of NaCl; dextrose sea water contained 720 mM dextrose in place of 460 mM NaCl. In K-free (“0K”) sea waters, the 10 mM KCl was replaced by 10 mM NaCl or LiCl, or 20 mM dextrose. Because neither replacement of external K+ nor addition of 10−5 M ouabain affected the Ca2+ influx, the 0K and ouabain treatment data were not separated.
When I wrote up the results for publication, Alan was reluctant to include the proposed explanation for the cardiotonic action of cardiac glycosides. Therefore, while Alan was on a lecture tour in Eastern Europe, Peter Baker and I contrived to submit the manuscript, including the cardiotonic steroid hypothesis (Baker et al., 1969). [Note: J Physiol authorship was alphabetical in those days; there was no jockeying for “first” or “senior” authorship.] Our explanation for the cardiotonic steroid effect was later verified with Ca2+ measurements (Wier and Hess, 1984; Altamirano et al., 2006), NCX-knockout mice (Reuter et al., 2002) and NCX blockers (Tanaka et al., 2007).
NCX on the Continent
At the very same time that we were performing these experiments, the Fall of 1966, Harald Reuter, from Mainz, Germany, was attending the Membrane Biophysics Lab Techniques course in Homburg. There, he learned about Na+/Na+ exchange, using tracer 22Na+, from Peter Caldwell and Richard Keynes (who did not yet know of the results in Plymouth). After completing the course, Harald, a cardiac pharmacologist who was familiar with the Luttgau-Niedergerke articles, immediately set out to look for NCX in cardiac muscle. And, of course, he found it (Reuter and Seitz, 1968). He knew what he was looking for; we were simply lucky, albeit prepared to recognize it when we saw it! As Louis Pasteur put it, “le hazard ne favorise que les esprits prepares.”
Harald and I first met at the International Physiology Congress in Washington, DC, in August, 1968. I had just returned from England, and was then heading to St. Louis to take a faculty position in the Department of Physiology and Biophysics at Washington University. It was a very amicable meeting that soon led to extremely beneficial consequences. Before describing those consequences, however, I should mention some other fallout from our Cambridge years.
Cambridge Collateral: Ellen’s contributions
While I was off squidding in Plymouth, my ‘squidow’ (the local name for squidder spouses), Ellen, when she wasn’t antiquing or rubbing church brasses, met several Cambridge sabbatical spouses. Upon my return to Cambridge in mid-December, after the 1966 squid season, Ellen invited Marcella and Len Ross for dinner. Len, an anatomist from Philadelphia, told me of his work with Victor Whittaker on the structure of isolated nerve endings, “synaptosomes”. I was fascinated, and hypothesized (to myself) that the terminals might reseal and become functionally competent; I planned to test this idea when I returned to the States. In fact, my colleagues and I subsequently showed that synaptosomes do reseal, and that they function like intact terminals. They have functional ion transport mechanisms, including Na+ pumps and NCX, and they generate ion gradients and membrane potentials (Blaustein and Wiesmann, 1970; Blaustein and Goldring, 1975; Fontana et al., 1995). Synaptosomes can be triggered to release neurotransmitters and to recycle synaptic vesicle membranes (Fried and Blaustein, 1976; Drapeau and Blaustein, 1983), and they sequester Ca2+ in the endoplasmic reticulum (Kendrick et al., 1977). We discovered voltage-gated Ca2+ channels in synaptosomes that were resistant to dihydropyridines (Nachshen and Blaustein, 1979); this was the first evidence for N, P and Q type Ca2+ channels (Catterall, 1998; Cao and Tsien, 2010) that play such an important role in brain physiology. We also used synaptosomes as an assay for the identification of a number of novel Na+ and K+ channel inhibitors (Krueger et al., 1980; Blaustein et al., 1991). In all, we published more than 65 articles on synaptosomes, including 6 in Nature or PNAS. In an extension of the synaptosome studies, we are currently testing the NCX-mediated effects of nanomolar ouabain on Ca2+ signaling in cultured neurons and astrocytes. Thus, Ellen’s friendship with Marcella Ross paid off in a line of research that has continued to the present day.
Smooth muscle is not such smooth sailing
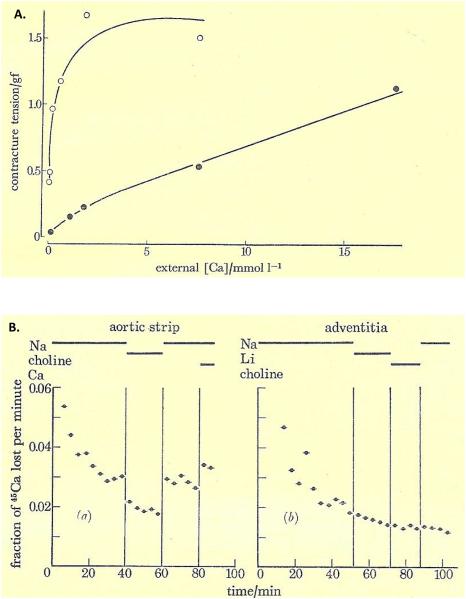
During our second year in Cambridge, again thanks to Ellen’s initial contact while I was in Plymouth, she and I became very friendly with our next-door neighbors, Joy and Mani Matter, from Bern, Switzerland. Mani was the Bern Town Counsel, and was spending a year studying international law in Cambridge; Joy was a teacher who later became Minister of Education for the Canton of Bern. We maintained contact when we returned to our respective home countries, and I visited the Matters in 1970, after presenting my first paper on NCX in synaptosomes at a meeting in Sweden. Ellen, too, was anxious to see the Matters again, so I arranged for a NATO Fellowship-funded mini-sabbatical with Harald Reuter, who had just become chairman of the Department of Pharmacology in Bern (what a fortunate coincidence!). And off we went to Bern, from May to September of 1971. On Harald’s suggestion, I agreed to look for NCX in vascular smooth muscle. The experiments were affirmative from the very beginning. We demonstrated antagonism between Nao and Cao (reduced [Na+]o promoted Ca2+ entry and arterial contraction; (Figure 2A) as well as Nao-dependent 45Ca2+ extrusion from guinea pig aortic smooth muscle (Figure 2B).
Figure 2.
(A) Effect of the external Ca2+ concentration, [Ca2+]o, on contraction of rabbit aorta. Contractile tension (gf) is increased by reduction of [Na+]o. Either 80% (filled circles) or 100% (open circles) of the NaCl in the standard medium (137 mM) was replaced by equiosmotic sucrose. The greater effectiveness of low [Ca2+]o in inducing contraction when [Na+]o was lowered is evidence of “Nao-Cao antagonism”. (B) Reduction of [Na+]o decreases 45Ca2+ efflux (fraction of 45Ca2+ lost per min) in a rabbit aortic strip (left), but not in the isolated adventitia (right). The NaCl in the standard medium (137 mM) was replaced by equimolar LiCl or choline-Cl during the periods indicated by the bars at the top. To avoid influx of Ca2+ and dilution of intracellular tracer 45Ca2+, the efflux (extracellular) solutions were Ca-free and contained 0.5 mM EGTA, except during the period indicated by the “Ca” bar at the end of the experiment on the left. The Nao-dependent 45Ca2+ efflux in intact intact aortic strip is evidence of Na+/Ca2+ exchange. Reprinted from Reuter et al., 1973, with permission.
In a manuscript describing these results, we suggested that NCX helps to regulate vascular tone. We also postulated a close approximation between the plasma membrane (PM) NCX and the sarcoplasmic reticulum; this idea foreshadowed the “buffer-barrier” hypothesis (van Breemen et al., 1986; van Breemen et al., 1995) and the “PLasmERosome” model (Blaustein et al., 1998; Arnon et al., 2000b) of local Ca2+ control. The manuscript (Blaustein and Reuter) was rejected by Nature, Science, and Experientia. It was published in a non-refereed journal, Phil Trans Roy Soc Lond, because Harald was invited to present the results at a Royal Society conference on smooth muscle (Reuter et al., 1973); the article has been cited ~300 times.
This novel idea that NCX is functionally important in vascular smooth muscle was, at first, ignored, or even disparaged (Somlyo et al., 1986; Murphy, 1988). Views about this concept slowly began to change more than two decades after its discovery (Murphy, 1993; Somlyo and Somlyo, 1994), once the NCX was cloned (Nicoll et al., 1990) and could be readily identified by immunocytochemistry and immunoblotting (Vemuri et al., 1990; Blaustein et al., 1992; Juhaszova et al., 1994).
Tragically, Mani Matter was killed in an auto accident in 1972. Therefore, we were most fortunate to have spent time in Bern in 1971. Mani had become a Swiss folk-hero because, as an avocation, he founded the Bernese Troubadors, who wrote and sang humorous and satirical songs in Swiss-German dialect that are still popular today.
PLasmERosomes: a structural basis for functional coupling of transport proteins
The NCX, like many other Na+-coupled transporters, is driven by the Na+ electrochemical gradient across the PM (Blaustein and Lederer, 1999). Nevertheless, we did not anticipate that the NCX would be co-localized with certain other transporters in PM microdomains adjacent to ‘junctional’ sarcoplasmic or endoplasmic reticulum (jS/ER) in a variety of cell types including neurons, glia and arterial myocytes (Juhaszova and Blaustein, 1997b). In fact, we were somewhat astonished (but shouldn’t have been) that the NCX was confined to the PM microdomains that also contained the Na+ pumps with a high ouabain affinity catalytic subunit (α2 or α3, depending upon cell type) (Juhaszova and Blaustein, 1997a). Moreover, both immunocytochemical and functional studies indicated that the much more prevalent Na+ pumps with an α1 catalytic subunit that, in rodents, has low ouabain affinity, were apparently excluded from these microdomains (Lee et al., 2006; Song et al., 2006). Another mechanism that extrudes Ca2+ from most types of cells, the PM Ca2+-ATPase (Ca2+ pump) may be excluded from these PM microdomains (Lencesova et al., 2004).
Some other Na+ and Ca2+ transporters also co-localize to these microdomains, notably TRPC proteins that are components of receptor- and store-operated channels (ROCs and SOCs, respectively) (Golovina, 2005; Lee et al., 2006; Zulian et al., 2010a). Many ROCs and SOCs are relatively non-selective cation channels, permeable to both Na+ and Ca2+ (Arnon et al., 2000a; Owsianik et al., 2006).
These findings provided a structural basis for the observations that the high ouabain affinity Na+ pumps, NCX, ROCs and SOCs, and the adjacent jS/ER function cooperatively to regulate Ca2+ signaling in a variety of cell types (Arnon et al., 2000a, b; Eder et al., 2005; Poburko et al., 2007). I called this PM microdomain-jS/ER complex, the “PLasmERosome” (Figure 3). PLasmERosomes appear to be widely distributed, fundamental units that play a key role in cell Ca2+ signaling. They can modulate Ca2+ signaling differently in different types of cells, depending upon: 1. The specific complement of TRPC proteins, and perhaps other channel proteins (e.g., voltage-gated Ca2+ channels, TRPM proteins), and 2. Various regulatory molecules that modulate the NCX and other transporters in the PLasmERosomes such as protein kinases, PIP2 (phosphatidylinositol bis-phosphate) and calmodulin. Not surprisingly, then, these mechanisms may also play prominent roles in pathophysiological processes (Blaustein, 1977), as discussed in the example below.
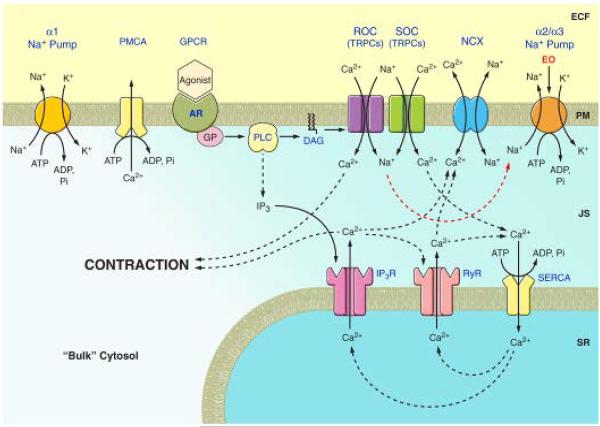
Figure 3.
Diagram of ouabain/EO-regulated α2 Na+ pump-modulated Ca2+ signaling at the plasma membrane-sarco/endoplasmic reticulum junction (PLasmERosome) in arterial smooth muscle. Endogenous ouabain (EO) reduces Na+ extrusion by the α2 Na+ pump, thereby increasing local [Na+] in the junctional space (JS) and reducing Ca2+ extrusion by the Na/Ca exchanger (NCX). This enhances Ca2+ signaling and, in arterial smooth muscle, contraction. ADP, adenosine diphosphate; AR, agonist receptor; ATP, adenosine triphosphate; DAG, diacylglycerol; ECF, extracellular fluid; GP, G-protein; GPCR, G-protein coupled receptor; IP3, inositol trisphosphate; IP3R, IP3, receptor; Pi, inorganic phosphate; PLC, phospholipase C; PMCA, plasma membrane Ca2+ pump; SERCA, sarco-/endoplasmic reticulum Ca2+ pump; SR, sarcoplasmic reticulum; SOC store-operated channel, and ROC, receptor-operated channel (both composed of TRPC proteins). Reprinted from Blaustein et al., 2011, with permission.
Linkage to hypertension and other heretical concepts
I learned from Alan Hodgkin that, if you perform careful, controlled experiments, you should be very confident about your own data. So, ignoring the naysayers, I continued to ponder the role of NCX in vascular smooth muscle, and even began to think that it might play a role in hypertension (Blaustein, 1974). This led me to propose a naïve, but useful hypothesis about the roles of an hypothetical endogenous cardiotonic steroid, its Na+ pump receptor, and NCX, in the pathogenesis of salt-dependent hypertension (Blaustein, 1977). That was before NCX and the Na+ pump were cloned and their isoforms discovered; and, of course, the endogenous cardiotonic steroid was then just a figment of our imagination.
Despite the previously mentioned skepticism of vascular biologists, however, this hypertension hypothesis aroused great interest in the idea of an endogenous ligand for the Na+ pump ouabain binding site. In particular, it excited one young man, John Hamlyn, who was a pre-teenager in Plymouth, England, when we discovered the NCX there. John came to my laboratory in 1980, as a post-doctoral fellow, with the idea of trying to purify this hypothetical agent. Ignoring the enormous difficulties, the fierce competition, and the repeated funding rejections from NIH, our team (John and our colleagues at the Upjohn Company) plunged ahead. In 1991, we purified from two tons of human plasma, and identified, by mass spectroscopy, an endogenous compound that, astonishingly, is indistinguishable from plant ouabain, i.e., “endogenous ouabain” or EO (Hamlyn et al., 1991). That paper, too, was rejected by Nature: one reviewer had a “gut feeling” that our result was an artifact, although another reviewer offered to write a “News and Views” article about the discovery. The Nature editor, John Maddox, had just taken the heat for publishing articles on “cold fusion” and “infinite dilution”; he apparently didn’t want to take a chance on being burned once more. Fortunately, our friend, Joseph Hoffman, a Na+ pump expert, agreed to communicate the manuscript to PNAS.
Again, many colleagues were skeptical (and some still are), even though the identification of EO was replicated in bovine adrenal and hypothalamic samples by three distinguished groups (Tamura et al., 1994; Schneider et al., 1998; Kawamura et al., 1999). Nevertheless, our Upjohn co-workers, who performed the original mass spectrometry, convinced us of the correctness of the result. Although the EO biosynthetic pathway has not yet been elucidated (funding bodies are not interested in such mundane studies), John Hamlyn and colleagues demonstrated net synthesis of EO by primary cultured bovine adrenocortical cells (Laredo et al., 1994). Furthermore, plasma EO levels are very low in adrenalectomized rodents (Hamlyn et al., 1991) and in humans with adrenocortical insufficiency (Sophocleous et al., 2003).
Ideas about the involvement of EO in the pathogenesis of some forms of hypertension have come a long way since my first, rather simplistic (but perhaps not so far-fetched) thoughts on the subject (Blaustein, 1977). Recent work is summarized to two reviews (Blaustein and Hamlyn, 2010; Blaustein et al., 2011).
NCX: a key player in the pathogenesis of hypertension?
I have already alluded to the central role of NCX in Ca2+ signaling that results from its co-localization with certain (ouabain-sensitive) Na+ pumps and ROCs and SOCs in PM microdomains at PM-S/ER junctions (PLasmERosomes). This fit with the simplistic notion that, if circulating EO levels are elevated in patients with hypertension (Rossi et al., 1995), we would expect the PLasmERosome mechanisms to amplify the Ca2+ signals. In fact, this is precisely what we observed in isolated, pressurized rodent small arteries: Ca2+ signals and myogenic constriction were augmented by ouabain with an EC50 of ~1 nM (Zhang et al., 2005). Could this account for the increased peripheral vascular resistance and elevation of blood pressure, as I initially proposed (Blaustein, 1977)?
Alas, life is not so simple! (A good thing, because that’s what keeps us scientists in business.) As my colleague, Vera Golovina discovered, the situation in hypertension is much more complex. She and her associates were studying arterial smooth muscle acutely isolated from rats in which hypertension was induced by prolonged subcutaneous infusion of ouabain (“ouabain hypertension”). They found that NCX1 (the vascular isoform) and TRPC6 protein (a component of ROCs) were greatly over-expressed in the myocytes, and that this was reflected in augmented Ca2+ signaling (Pulina et al., 2010a). The effects were not due to the elevation of blood pressure: both the protein up-regulation and the augmented Ca2+ signaling were also induced in primary cultured normal rat arterial myocytes incubated with 100 nM ouabain for 72-96 hr. [In an important twist, digoxin did not up-regulate NCX1 or TRPC6 expression (Zulian et al., 2010b) and, unlike ouabain, it does not induce hypertension in rats (Manunta et al., 2000) – but that’s another, as yet incomplete, story.]
Vera and her colleagues also found that NCX1 and TRPC6 were both markedly up-regulated in arterial myocytes from Dahl salt-sensitive rats fed a high salt diet to induce hypertension (Table 2), and in myocytes from Milan hypertensive rats, but not the normotensive strain (Zulian et al., 2010a). In fact, arterial myocyte NCX1 and either TRPC6 or TRPC3 (another component of ROCs) are up-regulated in all models of hypertension that have been tested to date (Table 2). An editorial commentary (Giachini and Tostes, 2010) on the articles from Vera’s laboratory stated that, “Given the… interrelationship among TRPC6, Na+ influx, NCX, and Ca2+ influx, it seems that this is… the key to better understand the role of Na+ in hypertension, vascular reactivity, and blood pressure regulation”. Vindication! This is the diametric opposite of the view that, “The Na/Ca exchanger does not play an essential role in the regulation of cytoplasmic Ca2+ in smooth muscle” (Somlyo et al., 1986). The up-regulation of NCX1 and TRPC6 expression may, in fact, be a key mechanism underlying the phenomenon of “whole body autoregulation” (Guyton, 1989) that sustains the increased peripheral vascular resistance and elevated BP in hypertension.
Table 2.
Expression of Na/Ca exchanger-1 (NCX1) and some TRPC protein components of receptor-operated channels (ROCs) is increased in several hypertensive animal models and in human primary pulmonary hypertension. Modified from Blaustein et al, 2011).
| Hypertension | Artery Smooth Muscle | Reference | |||
|---|---|---|---|---|---|
| NCX1 | ROC | ||||
| TRPC3 | TRPC6 | ||||
| 1 | Ouabain* (vs vehicle & digoxin) | ↑ | ↑ | (Pulina et al., 2010b; Zulian et al., 2010b) |
|
| 2 | DOCA-salt* | ND** | ↑ | (Bae et al., 2007) | |
| 3 | Milan hypertension* (vs Milan NT)** | ↑ | ↑ | (Zulian et al., 2010a) | |
| 4 | SHR (vs WKY)** | ↑ | ↑ | (Taniguchi et al., 2004; Liu et al., 2009) | |
| 5 | Dahl salt-sensitive/high (vs low) salt* | ↑ | ↑ | VA Golovina (unpublished) | |
| 6 | NCX1SM.Tg/Tg ** | ↑ | ↑ | (Iwamoto et al., 2004); J Zhang & MP Blaustein (unpublished) |
|
| 7 | Angiotensin II | ↑ | ND** | L Chen, M Li & MP Blaustein (unpublished) |
|
| 8 | Human primary pulmonary hypertension | ↑ | ↑ | ↑ | (Yu et al., 2004; Zhang et al., 2007a; Zhang et al., 2007b) |
Hypertension associated with elevated plasma ouabain levels.
Not determined.
Abbreviations: NT, normotensive control; SHR, spontaneously hypertensive rat; WKY, Wistar-Kyoto normotensive control for SHR; NCX1SMTg/Tg, smooth muscle-specific NCX1 knockout mouse (Zhang et al., 2010).
This, of course, is not the end of the story, but just the beginning of the next chapter. The fun in research is that every new discovery leads to new questions. In this case, we must now ask: “What is the mechanism by which ouabain up-regulates NCX1 and TRPC6 expression?” The up-regulation appears to be mediated by a novel activity of Na+ pumps first elucidated by Zi-Jian Xie, namely ouabain-induced activation of protein kinase signaling cascades (Xie and Askari, 2002; Liu et al., 2007; Liu and Xie, 2010; Liu et al., 2011). [Zi-Jian’s new idea, that Na+ pumps could do something other than pump Na+ and K+, was greeted with profound skepticism – and I was originally one of the skeptics – but the data speak for themselves.] It is the anticipation of learning something new at any moment that keeps me young and keeps me going. That is the lesson I most enjoy conveying to students; it is the essence (and the excitement) of scientific research. The fact that such moments are not every-day occurrences makes them so special.
In closing:
I have been most fortunate to have had the opportunity to engage in this exciting adventure. I was well-prepared by my teachers, but I could not have had such success without wonderful students, colleagues and collaborators. My wife, Ellen, in addition to providing extraordinary support, also played a critical part (mentioned above) that neither she nor I could have imagined a priori. And, of course, none of this would have been possible without the many years of financial support from the National Institutes of Health and the American Heart Association.
“After all, in spite of opinion, prejudice, or error, Time will fix the real value upon this discovery, and determine whether I have imposed upon myself and others, or contributed to the benefit of science and mankind.”
William Withering, “An Account of the Foxglove and Some of its Medical Uses” Birmingham, England, July 1, 1785.
Acknowledgements
I thank Suzanne Ventura for assistance with the manuscript. Supported by NIH/NHLBI grants HL-045215, HL-078870 and HL-107555.
Footnotes
In memory of Peter F. Baker, David E. Goldman, Alan L. Hodgkin, Howard A. Schneiderman, Daniel C. Tosteson, and Mani Matter.
References
- Altamirano J, Li Y, DeSantiago J, Piacentino V, Houser SR, Bers DM. The inotropic effect of cardioactive glycosides in ventricular myocytes requires Na+-Ca2+ exchanger function. J. Physiol. 2006;575:845–854. doi: 10.1113/jphysiol.2006.111252. 3rd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnon A, Hamlyn JM, Blaustein MP. Na+ entry via store-operated channels modulates Ca(2+) signaling in arterial myocytes. Am. J. Physiol. Cell Physiol. 2000a;278:C163–173. doi: 10.1152/ajpcell.2000.278.1.C163. [DOI] [PubMed] [Google Scholar]
- Arnon A, Hamlyn JM, Blaustein MP. Ouabain augments Ca2+ transients in arterial smooth muscle without raising cytosolic Na+ Am. J. Physiol. Heart Circ. Physiol. 2000b;279:H679–691. doi: 10.1152/ajpheart.2000.279.2.H679. [DOI] [PubMed] [Google Scholar]
- Bae YM, Kim A, Lee YJ, Lim W, Noh YH, Kim EJ, Kim J, Kim TK, Park SW, Kim B, Cho SI, Kim DK, Ho WK. Enhancement of receptor-operated cation current and TRPC6 expression in arterial smooth muscle cells of deoxycorticosterone acetate-salt hypertensive rats. J Hypertens. 2007;25:809–817. doi: 10.1097/HJH.0b013e3280148312. [DOI] [PubMed] [Google Scholar]
- Baker PF, Blaustein MP, Hodgkin AL, Steinhardt RA. The influence of calcium on sodium efflux in squid axons. J. Physiol. 1969;200:431–458. doi: 10.1113/jphysiol.1969.sp008702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaustein MP. Barbiturates block sodium and potassium conductance increases in voltage-clamped lobster axons. J. Gen. Physiol. 1968;51:293–307. doi: 10.1085/jgp.51.3.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaustein MP. The interrelationship between sodium and calcium fluxes across cell membranes. Rev. Physiol. Biochem. Pharmacol. 1974;70:33–82. doi: 10.1007/BFb0034293. [DOI] [PubMed] [Google Scholar]
- Blaustein MP. Sodium ions, calcium ions, blood pressure regulation, and hypertension: a reassessment and a hypothesis. Am. J. Physiol. 1977;232:C165–173. doi: 10.1152/ajpcell.1977.232.5.C165. [DOI] [PubMed] [Google Scholar]
- Blaustein MP, Ambesi A, Bloch RJ, Goldman WF, Juhaszova M, Lindenmayer GE, Weiss DN. Regulation of vascular smooth muscle contractility: roles of the sarcoplasmic reticulum (SR) and the sodium/calcium exchanger. Jpn. J. Pharmacol. 1992;58(Suppl 2):107P–114P. [PubMed] [Google Scholar]
- Blaustein MP, Goldman DE. Action of anionic and cationic nerve-blocking agents: experiment and interpretation. Science. 1966;153:429–432. doi: 10.1126/science.153.3734.429. [DOI] [PubMed] [Google Scholar]
- Blaustein MP, Goldring JM. Membrane potentials in pinched-off presynaptic nerve ternimals monitored with a fluorescent probe: evidence that synaptosomes have potassium diffusion potentials. J. Physiol. 1975;247:589–615. doi: 10.1113/jphysiol.1975.sp010949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaustein MP, Hamlyn JM. Signaling mechanisms that link salt retention to hypertension: endogenous ouabain, the Na+ pump, the Na+/Ca2+ exchanger and TRPC proteins. Biochim. Biophys. Acta. 2010;1802:1219–1229. doi: 10.1016/j.bbadis.2010.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaustein MP, Juhaszova M, Golovina VA. The cellular mechanism of action of cardiotonic steroids: a new hypothesis. Clin. Exp. Hypertens. 1998;20:691–703. doi: 10.3109/10641969809053247. [DOI] [PubMed] [Google Scholar]
- Blaustein MP, Lederer WJ. Sodium/calcium exchange: its physiological implications. Physiol. Rev. 1999;79:763–854. doi: 10.1152/physrev.1999.79.3.763. [DOI] [PubMed] [Google Scholar]
- Blaustein MP, Leenen FH, Chen L, Golovina VA, Hamlyn JM, Pallone TL, Van Huysse JW, Zhang J, Wier WG. How NaCl raises blood pressure: A new paradigm for the pathogenesis of salt-dependent hypertension. Am. J. Physiol. Heart Circ. Physiol. 2011 doi: 10.1152/ajpheart.00899.2011. doi:10.1152, in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaustein MP, Rogowski RS, Schneider MJ, Krueger BK. Polypeptide toxins from the venoms of Old World and New World scorpions preferentially block different potassium channels. Mol. Pharmacol. 1991;40:932–942. [PubMed] [Google Scholar]
- Blaustein MP, Wiesmann WP. Effect of sodium ions on calcium movements in isolated synaptic terminals. Proc. Natl. Acad. Sci. U.S.A. 1970;66:664–671. doi: 10.1073/pnas.66.3.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao YQ, Tsien RW. Different relationship of N- and P/Q-type Ca2+ channels to channel-interacting slots in controlling neurotransmission at cultured hippocampal synapses. J. Neurosci. 2010;30:4536–4546. doi: 10.1523/JNEUROSCI.5161-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catterall WA. Structure and function of neuronal Ca2+ channels and their role in neurotransmitter release. Cell Calcium. 1998;24:307–323. doi: 10.1016/s0143-4160(98)90055-0. [DOI] [PubMed] [Google Scholar]
- Drapeau P, Blaustein MP. Initial release of [3H]dopamine from rat striatal synaptosomes: correlation with calcium entry. J. Neurosci. 1983;3:703–713. doi: 10.1523/JNEUROSCI.03-04-00703.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eder P, Poteser M, Romanin C, Groschner K. Na+ entry and modulation of Na+/Ca2+ exchange as a key mechanism of TRPC signaling. Pflugers Arch. 2005;451:99–104. doi: 10.1007/s00424-005-1434-2. [DOI] [PubMed] [Google Scholar]
- Fontana G, Rogowski RS, Blaustein MP. Kinetic properties of the sodium-calcium exchanger in rat brain synaptosomes. J. Physiol. 1995;485:349–364. doi: 10.1113/jphysiol.1995.sp020734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fried RC, Blaustein MP. Synaptic vesicle recycling in synaptosomes in vitro. Nature. 1976;261:255–256. doi: 10.1038/261255a0. [DOI] [PubMed] [Google Scholar]
- Giachini FR, Tostes RC. Does Na+ really play a role in Ca2+ homeostasis in hypertension? Am. J. Physiol. Heart Circ. Physiol. 2010;299:H602–604. doi: 10.1152/ajpheart.00542.2010. [DOI] [PubMed] [Google Scholar]
- Golovina VA. Visualization of localized store-operated calcium entry in mouse astrocytes. Close proximity to the endoplasmic reticulum. J. Physiol. 2005;564:737–749. doi: 10.1113/jphysiol.2005.085035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guyton AC. Dominant role of the kidneys and accessory role of whole-body autoregulation in the pathogenesis of hypertension. Am. J. Hypertens. 1989;2:575–585. doi: 10.1093/ajh/2.7.575. [DOI] [PubMed] [Google Scholar]
- Hamlyn JM, Blaustein MP, Bova S, DuCharme DW, Harris DW, Mandel F, Mathews WR, Ludens JH. Identification and characterization of a ouabain-like compound from human plasma. Proc. Natl. Acad. Sci. U.S.A. 1991;88:6259–6263. doi: 10.1073/pnas.88.14.6259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgkin AL, Keynes RD. Experiments on the injection of substances into squid giant axons by means of a microsyringe. J. Physiol. 1956;131:592–616. doi: 10.1113/jphysiol.1956.sp005485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwamoto T, Kita S, Zhang J, Blaustein MP, Arai Y, Yoshida S, Wakimoto K, Komuro I, Katsuragi T. Salt-sensitive hypertension is triggered by Ca2+ entry via Na+/Ca2+ exchanger type-1 in vascular smooth muscle Nature Med. 2004;10:1193–1199. doi: 10.1038/nm1118. [DOI] [PubMed] [Google Scholar]
- Juhaszova M, Ambesi A, Lindenmayer GE, Bloch RJ, Blaustein MP. Na+-Ca2+ exchanger in arteries: identification by immunoblotting and immunofluorescence microscopy. Am. J. Physiol. 1994;266:C234–242. doi: 10.1152/ajpcell.1994.266.1.C234. [DOI] [PubMed] [Google Scholar]
- Juhaszova M, Blaustein MP. Distinct distribution of different Na+ pump alpha subunit isoforms in plasmalemma. Physiological implications. Ann. N.Y. Acad. Sci. 1997a;834:524–536. doi: 10.1111/j.1749-6632.1997.tb52310.x. [DOI] [PubMed] [Google Scholar]
- Juhaszova M, Blaustein MP. Na+ pump low and high ouabain affinity alpha subunit isoforms are differently distributed in cells. Proc. Natl. Acad. Sci. U.S.A. 1997b;94:1800–1805. doi: 10.1073/pnas.94.5.1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawamura A, Guo J, Itagaki Y, Bell C, Wang Y, Haupert GT, Jr., Magil S, Gallagher RT, Berova N, Nakanishi K. On the structure of endogenous ouabain. Proc. Natl. Acad. Sci. U.S.A. 1999;96:6654–6659. doi: 10.1073/pnas.96.12.6654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kendrick NC, Blaustein MP, Fried RC, Ratzlaff RW. ATP-dependent calcium storage in presynaptic nerve terminals. Nature. 1977;265:246–248. doi: 10.1038/265246a0. [DOI] [PubMed] [Google Scholar]
- Krueger BK, Blaustein MP, Ratzlaff RW. Sodium channels in presynaptic nerve terminals. Regulation by neurotoxins. J. Gen. Physiol. 1980;76:287–313. doi: 10.1085/jgp.76.3.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laredo J, Hamilton BP, Hamlyn JM. Ouabain is secreted by bovine adrenocortical cells. Endocrinology. 1994;135:794–797. doi: 10.1210/endo.135.2.8033829. [DOI] [PubMed] [Google Scholar]
- Lee MY, Song H, Nakai J, Ohkura M, Kotlikoff MI, Kinsey SP, Golovina VA, Blaustein MP. Local subplasma membrane Ca2+ signals detected by a tethered Ca2+ sensor. Proc. Natl. Acad. Sci. U.S.A. 2006;103:13232–13237. doi: 10.1073/pnas.0605757103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lencesova L, O’Neill A, Resneck WG, Bloch RJ, Blaustein MP. Plasma membrane-cytoskeleton-endoplasmic reticulum complexes in neurons and astrocytes. J. Biol. Chem. 2004;279:2885–2893. doi: 10.1074/jbc.M310365200. [DOI] [PubMed] [Google Scholar]
- Liu D, Yang D, He H, Chen X, Cao T, Feng X, Ma L, Luo Z, Wang L, Yan Z, Zhu Z, Tepel M. Increased transient receptor potential canonical type 3 channels in vasculature from hypertensive rats. Hypertension. 2009;53:70–76. doi: 10.1161/HYPERTENSIONAHA.108.116947. [DOI] [PubMed] [Google Scholar]
- Liu J, Xie ZJ. The sodium pump and cardiotonic steroids-induced signal transduction protein kinases and calcium-signaling microdomain in regulation of transporter trafficking. Biochim. Biophys. Acta. 2010;1802:1237–1245. doi: 10.1016/j.bbadis.2010.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Ivanov AV, Gable ME, Jolivel F, Morrill GA, Askari A. Comparative properties of caveolar and noncaveolar preparations of kidney Na+/K+-ATPase. Biochemistry. 2011;50:8664–8673. doi: 10.1021/bi2009008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Zhao X, Pierre SV, Askari A. Association of PI3K-Akt signaling pathway with digitalis-induced hypertrophy of cardiac myocytes. Am. J. Physiol. Cell Physiol. 2007;293:C1489–1497. doi: 10.1152/ajpcell.00158.2007. [DOI] [PubMed] [Google Scholar]
- Luttgau HC, Niedergerke R. The antagonism between Ca and Na ions on the frog’s heart. J. Physiol. 1958;143:486–505. doi: 10.1113/jphysiol.1958.sp006073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manunta P, Hamilton J, Rogowski AC, Hamilton BP, Hamlyn JM. Chronic hypertension induced by ouabain but not digoxin in the rat: antihypertensive effect of digoxin and digitoxin. Hypertens. Res. 2000;23(Suppl):S77–85. doi: 10.1291/hypres.23.supplement_s77. [DOI] [PubMed] [Google Scholar]
- Moore JW, Blaustein MP, Anderson NC, Narahashi T. Basis of tetrodotoxin’s selectivity in blockage of squid axons. J. Gen. Physiol. 1967;50:1401–1411. doi: 10.1085/jgp.50.5.1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy RA. Contraction of muscle cells. In: Berne RM, Levy MN, editors. Physiology. 2nd Mosby; Washington, D.C.: 1988. pp. 315–342. [Google Scholar]
- Murphy RA. Smooth Muscle. In: Berne RM, Levy MN, editors. Physiology. 3rd Mosby Year Book; St. Louis: 1993. pp. 309–324. [Google Scholar]
- Nachshen DA, Blaustein MP. The effects of some organic “calcium antagonists” on calcium influx in presynaptic nerve terminals. Mol. Pharmacol. 1979;16:576–586. [PubMed] [Google Scholar]
- Nicoll DA, Longoni S, Philipson KD. Molecular cloning and functional expression of the cardiac sarcolemmal Na+-Ca2+ exchanger. Science. 1990;250:562–565. doi: 10.1126/science.1700476. [DOI] [PubMed] [Google Scholar]
- Owsianik G, Talavera K, Voets T, Nilius B. Permeation and selectivity of TRP channels. Annu. Rev. Physiol. 2006;68:685–717. doi: 10.1146/annurev.physiol.68.040204.101406. [DOI] [PubMed] [Google Scholar]
- Poburko D, Liao CH, Lemos VS, Lin E, Maruyama Y, Cole WC, van Breemen C. Transient receptor potential channel 6-mediated, localized cytosolic [Na+] transients drive Na+/Ca2+ exchanger-mediated Ca2+ entry in purinergically stimulated aorta smooth muscle cells. Circ. Res. 2007;101:1030–1038. doi: 10.1161/CIRCRESAHA.107.155531. [DOI] [PubMed] [Google Scholar]
- Pulina MV, Zulian A, Berra-Romani R, Beskina O, Mazzocco-Spezzia A, Baryshnikov SG, Papparella I, Hamlyn JM, Blaustein MP, Golovina VA. Upregulation of Na+ and Ca2+ transporters in arterial smooth muscle from ouabain-induced hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2010a;298:H263–274. doi: 10.1152/ajpheart.00784.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulina MV, Zulian A, Berra-Romani R, Beskina O, Mazzocco-Spezzia A, Baryshnikov SG, Papparella I, Hamlyn JM, Blaustein MP, Golovina VA. Upregulation of Na+ and Ca2+ transporters in arterial smooth muscle from ouabain-induced hypertensive rats. Am J Physiol Heart Circ Physiol. 2010b;298:H263–274. doi: 10.1152/ajpheart.00784.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reuter H, Blaustein MP, Haeusler G. Na-Ca exchange and tension development in arterial smooth muscle. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 1973;265:87–94. doi: 10.1098/rstb.1973.0011. [DOI] [PubMed] [Google Scholar]
- Reuter H, Henderson SA, Han T, Ross RS, Goldhaber JI, Philipson KD. The Na+-Ca2+ exchanger is essential for the action of cardiac glycosides. Circ. Res. 2002;90:305–308. doi: 10.1161/hh0302.104562. [DOI] [PubMed] [Google Scholar]
- Reuter H, Seitz N. The dependence of calcium efflux from cardiac muscle on temperature and external ion composition. J. Physiol. 1968;195:451–470. doi: 10.1113/jphysiol.1968.sp008467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi G, Manunta P, Hamlyn JM, Pavan E, De Toni R, Semplicini A, Pessina AC. Immunoreactive endogenous ouabain in primary aldosteronism and essential hypertension: relationship with plasma renin, aldosterone and blood pressure levels. J. Hypertens. 1995;13:1181–1191. doi: 10.1097/00004872-199510000-00013. [DOI] [PubMed] [Google Scholar]
- Schneider R, Wray V, Nimtz M, Lehmann WD, Kirch U, Antolovic R, Schoner W. Bovine adrenals contain, in addition to ouabain, a second inhibitor of the sodium pump. J. Biol. Chem. 1998;273:784–792. doi: 10.1074/jbc.273.2.784. [DOI] [PubMed] [Google Scholar]
- Skou JC. The influence of some cations on an adenosine triphosphatase from peripheral nerves. Biochim. Biophys. Acta. 1957;23:394–401. doi: 10.1016/0006-3002(57)90343-8. [DOI] [PubMed] [Google Scholar]
- Somlyo AP, Broderick R, Somlyo AV. Calcium and sodium in vascular smooth muscle. Ann. N.Y. Acad. Sci. 1986;488:228–239. doi: 10.1111/j.1749-6632.1986.tb46561.x. [DOI] [PubMed] [Google Scholar]
- Somlyo AP, Somlyo AV. Signal transduction and regulation in smooth muscle. Nature. 1994;372:231–236. doi: 10.1038/372231a0. [DOI] [PubMed] [Google Scholar]
- Song H, Lee MY, Kinsey SP, Weber DJ, Blaustein MP. An N-terminal sequence targets and tethers Na+ pump α2 subunits to specialized plasma membrane microdomains. J. Biol. Chem. 2006;281:12929–12940. doi: 10.1074/jbc.M507450200. [DOI] [PubMed] [Google Scholar]
- Sophocleous A, Elmatzoglou I, Souvatzoglou A. Circulating endogenous digitalis-like factor(s) (EDLF) in man is derived from the adrenals and its secretion is ACTH-dependent. J. Endocrinol. Invest. 2003;26:668–674. doi: 10.1007/BF03347027. [DOI] [PubMed] [Google Scholar]
- Tamura M, Konishi F, Sakakibara M, Inagami T. Bamberg E, Schoner W, editors. Large scale purification of an endogenous Na+/K+-pump inhibitor from bovine adrenal glands. The Sodium Pump: Structure, Mechanism, Hormonal Control and its Role in Disease, Steinkopff, Darmstadt. 1994:763–766. in. [Google Scholar]
- Tanaka H, Shimada H, Namekata I, Kawanishi T, Iida-Tanaka N, Shigenobu K. Involvement of the Na+/Ca2+ exchanger in ouabain-induced inotropy and arrhythmogenesis in guinea-pig myocardium as revealed by SEA0400. J. Pharmacol. Sci. 2007;103:241–246. doi: 10.1254/jphs.fp0060911. [DOI] [PubMed] [Google Scholar]
- Taniguchi S, Furukawa K, Sasamura S, Ohizumi Y, Seya K, Motomura S. Gene expression and functional activity of sodium/calcium exchanger enhanced in vascular smooth muscle cells of spontaneously hypertensive rats. J Cardiovasc Pharmacol. 2004;43:629–637. doi: 10.1097/00005344-200405000-00004. [DOI] [PubMed] [Google Scholar]
- van Breemen C, Chen Q, Laher I. Superficial buffer barrier function of smooth muscle sarcoplasmic reticulum. Trends Pharmacol. Sci. 1995;16:98–105. doi: 10.1016/s0165-6147(00)88990-7. [DOI] [PubMed] [Google Scholar]
- van Breemen C, Lukeman S, Leijten P, Yamamoto H, Loutzenhiser R. The role of superficial SR in modulating force development induced by Ca entry into arterial smooth muscle. J. Cardiovasc. Pharmacol. 1986;8(Suppl 8):S111–116. doi: 10.1097/00005344-198600088-00023. [DOI] [PubMed] [Google Scholar]
- Vemuri R, Haberland ME, Fong D, Philipson KD. Identification of the cardiac sarcolemmal Na+-Ca2+ exchanger using monoclonal antibodies. J. Membr. Biol. 1990;118:279–283. doi: 10.1007/BF01868612. [DOI] [PubMed] [Google Scholar]
- Wier WG, Hess P. Excitation-contraction coupling in cardiac Purkinje fibers. Effects of cardiotonic steroids on the intracellular [Ca2+] transient, membrane potential, and contraction. J. Gen. Physiol. 1984;83:395–415. doi: 10.1085/jgp.83.3.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie Z, Askari A. Na+/K+-ATPase as a signal transducer. Eur. J. Biochem. 2002;269:2434–2439. doi: 10.1046/j.1432-1033.2002.02910.x. [DOI] [PubMed] [Google Scholar]
- Yu Y, Fantozzi I, Remillard CV, Landsberg JW, Kunichika N, Platoshyn O, Tigno DD, Thistlethwaite PA, Rubin LJ, Yuan JX. Enhanced expression of transient receptor potential channels in idiopathic pulmonary arterial hypertension. Proc Natl Acad Sci U S A. 2004;101:13861–13866. doi: 10.1073/pnas.0405908101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Ren C, Chen L, Navedo MF, Antos LK, Kinsey SP, Iwamoto T, Philipson KD, Kotlikoff MI, Santana LF, W.G. W, Matteson DR, Blaustein MP. Knockout of Na+/Ca2+ exchanger in smooth muscle attenuates vasoconstriction and L-type Ca2+ channel current, and lowers blood pressure. Am J Physiol Heart Circ Physiol. 2010;298:H1472–H1483. doi: 10.1152/ajpheart.00964.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S, Dong H, Rubin LJ, Yuan JX. Upregulation of Na+/Ca2+ exchanger contributes to the enhanced Ca2+ entry in pulmonary artery smooth muscle cells from patients with idiopathic pulmonary arterial hypertension. Am J Physiol Cell Physiol. 2007a;292:C2297–2305. doi: 10.1152/ajpcell.00383.2006. [DOI] [PubMed] [Google Scholar]
- Zhang S, Patel HH, Murray F, Remillard CV, Schach C, Thistlethwaite PA, Insel PA, Yuan JX. Pulmonary artery smooth muscle cells from normal subjects and IPAH patients show divergent cAMP-mediated effects on TRPC expression and capacitative Ca2+ entry. Am J Physiol Lung Cell Mol Physiol. 2007b;292:L1202–1210. doi: 10.1152/ajplung.00214.2006. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Cao C, Lee-Kwon W, Pallone TL. Descending vasa recta pericytes express voltage operated Na+ conductance in the rat. J. Physiol. 2005;567:445–457. doi: 10.1113/jphysiol.2005.091538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zulian A, Baryshnikov SG, Linde CI, Hamlyn JM, Ferrari P, Golovina VA. Upregulation of Na+/Ca2+ exchanger and TRPC6 contributes to abnormal Ca2+ homeostasis in arterial smooth muscle cells from Milan hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2010a;299:H624–633. doi: 10.1152/ajpheart.00356.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zulian A, Pulina MV, Linde CI, Baryshnikov S, Hamlyn JM, Blaustein MP, Golovina VA. Digoxin Blocks Ouabain-induced Hypertension by Preventing Up-regulation of the Na/Ca Exchanger-1/TRPC6 Ca2+ Signaling Pathway. Hypertension. 2010b;56:e57. [Google Scholar]