Abstract
Understanding the intrinsic and extrinsic signals that regulate the molecular basis of the pluripotent state may improve our understanding of mammalian embryogenesis, different states of pluripotency, and our ability to tailor lineage differentiation. Although the role of the PI3K/Akt pathway in the self-renewal and maintenance of mESCs is well-established, the specific contribution of the pathway or of its negative regulator, PTEN, in the maintenance of the human pluripotent state is less understood. To explore the PI3K/AKT pathway in human embryonic stem cell (hESC) pluripotency and differentiation, we generated stable PTEN knockdown (KD) hESCs using short hairpin RNA. Similar to mESCs, we found that PTEN KD hESCs have increased self-renewal, cell survival, and proliferation over multiple passages compared to control cells. However, in contrast to mESCs, in vitro, PTEN KD hESCs differentiated inefficiently in directed differentiation assays, in part due to the continued maintenance of OCT4 and NANOG expression. In teratoma assays, PTEN KD hESCs generated tissues from the three germ layers, although with a bias toward neuroectoderm differentiation. These results demonstrate that PTEN is a key regulator of hESC growth and differentiation, and manipulation of this pathway may improve our ability to regulate and understand the pluripotent state.
Keywords: Pluripotent stem cells, Embryonic stem cells, Self-renewal, Differentiation, Proliferation, Cell signaling
Introduction
Human embryonic stem cells (hESCs) and their derivatives are able to differentiate into the three embryonic germ layers, making them a potential source of cells for regenerative medicine. The pluripotent state is balanced by the coordinated actions of extrinsic signaling pathways such as fibroblast growth factor (FGF), transforming growth factor β, and IGF, as well as intrinsic transcriptional networks such as OCT4, NANOG, and SOX2. Extensive efforts are being put forth to determine how to maintain pluripotency in the absence of exogenous self-renewing factors. An understanding of how these signaling pathways regulate ESC fate may provide inroads to the universal maintenance of the pluripotent state [1, 2], help minimize differences in mouse ESCs (mESCs) and human ESC culture systems, and help standardize approaches used in reprogramming.
One extrinsic signaling pathway that is a common mediator of pluripotency in human and mESCs is the phosphoinositide 3-kinase (PI3K)/Akt pathway [3–6]. The PI3K/Akt pathway regulates a range of physiological processes including proliferation, development, growth, and migration [7]. Akt activity results in the phosphorylation of a myriad of downstream effectors, including mammalian target of rapamycin (mTOR) and glycogen synthase kinase 3 beta (GSK3β) [8]. A major negative regulator of PI3K/Akt signaling is phosphatase and tensin homolog (Pten), a ubiquitously expressed protein and lipid phosphatase with essential roles during embryonic development as well as in adult tissues [9]. The importance of Pten in tissue homeostasis is highlighted by its function as a tumor suppressor, with its deletion or mutation found in a wide range of tumors [10].
Numerous studies have established a role for PI3K/Akt/Pten in stem cells. In mESCs and adult stem cells, the loss of Pten is correlated with the activation of Akt, increased self-renewal, and proliferation [11–14]. It is well documented in mESCs that PI3K-dependent signaling is required for maintenance of the mESC pluripotent state [15]. For example, PI3K/Akt signaling regulates Nanog and Nanog target genes in mESCs and embryonal carcinoma (EC) cells [16]. Moreover, Akt interacts with and phosphorylates Sox2, providing a direct mechanism by which PI3K/Akt regulates the pluripotency network [17].
We have previously shown that inhibition of PI3K signaling using the PI3K-specific inhibitor LY294002 leads to loss of pluripotent stem cell (PSC) colonies in low density dissociation assays, suggesting a critical role in survival or self-renewal in hESCs [5, 18]. Others have shown that AKT activation, downstream of IGF and FGF signaling, promotes hESC survival and pluripotency [19, 20]. Moreover, the prevention of anoikis, detachment-mediated cell death, in stem cells is mediated through mitogen-activated protein kinase–extracellular signal-regulated kinase or AKT signaling [21–23]. The importance of the PI3K/AKT pathway to the hESC pluripotent state is further supported by the demonstration that its downregulation is required prior to initiation of differentiation toward endoderm [24]. Therefore, a role for PI3K/Akt activity has been established in the regulation of self-renewal and pluripotency in mESCs and hESCs. However, the connection between PI3K and the pluripotency network in human PSCs (hPSCs) remains unclear.
In this work, we define how PTEN activity regulates transcription factor networks to control pluripotency in hESCs and further, how PTEN levels regulate lineage fate choices during hESC differentiation. Here we report that knockdown of PTEN in hESCs leads to activation of subsets of the pluripotency network genes, more homogeneous pluripotent stem cell growth, and aberrant differentiation. Understanding PTEN control of pluripotency networks will provide novel targets for improving our understanding of lineage differentiation, reprogramming, and establishment of the pluripotent state.
Materials and Methods
Additional methods are listed in Supporting Information materials—see Supporting Information Methods Doc.
Cell Culture and Generation of Genetically Modified hESCs
HESCs used in this study were from lines H9 (Wicell, Madison, WI, www.wicell.org) between passages 37 and 50 and HSF1 (University of California San Francisco) between passages 65 and 75. When hESCs reached beyond these passages, new hESCs were thawed from the original stocks to ensure that similar passage numbers were used in each experiment. HESCs were cultured on gelatin-coated plates with mitomycin C-treated mouse embryonic fibroblasts (MEFs) in hESC medium consisting of Dulbecco’s Modified Eagle Medium (DMEM)-F12 (Invitrogen, Carlsbad, CA, www.invitrogen.com) supplemented with 20% knockout serum replacement (Invitrogen), 1 mM l-glutamine, 2 mM 2-mercaptoethanol, 1 mM nonessential amino acids, penicillin/ streptomycin (Hyclone, Rockford, IL, www.thermoscientific.com), and 4 ng/ml basic FGF (bFGF, NCI Biological Resources Branch, Pre-Clinical Repository). Karyotype analysis was performed by Cell Line Genetics (Madison, WI, www.clgenetics. com). Cells were prepared and sent according to their protocol. Nontarget short hairpin RNA (shRNA; control) and shPTEN knockdown hESCs (PTEN KD hESCs) were generated by lentiviral-mediated transduction of short hairpin pLKO.1 vectors (Sigma, St. Louis, MO, www.sigmaaldrich.com) and subsequent puromycin selection for 4 weeks.
Lentivirus Production
Virus was produced using the calcium phosphate transfection Kit (Clontech, Mountain View, CA, www.clontech.com). Virus was concentrated by centrifugation at 17,000 rpm for 90 minutes at 4°C in a Beckman ultracentrifuge with a SW28 rotor.
Self-Renewal and Survival Assays
HESCs were dissociated into single cells using 0.05% trypsin-EDTA, rinsed with phosphate-buffered saline solution (PBS), and strained through a 40 µm mesh prior to centrifugation. For counting, cells were resuspended in 0.4% trypan blue stain (Invitrogen), and live cells were counted using a hemocytometer. A total of 1 × 103 or 15 × 103 cells were cultured under self-renewing conditions (in 12-well plates) for 5–7 days. For multiple passage self-renewal assays, 100,000 single hESCs were plated in 12-well plates and cultured under self-renewing conditions for 1 week. These experiments were repeated for five passages. Prior to staining, colonies were fixed using 4% paraformaldehyde (PFA) for 30 minutes at room temperature. Colonies were stained with alkaline phosphatase (AP) buffer (0.01% naphthol AS-MX phosphate alkaline solution/1 mg/ml nuclear fast red) for 30 minutes to 1 hour at room temperature. AP+ colonies were visually counted under a microscope. To evaluate cell death after multiple passages, 1 × 105 cells (in six-well plates) were cultured in self-renewing conditions for 5–7 days and stained for TRA-1-81 to identify stem cells. Cell death was measured using the fluorescein isothiocyanate (FITC) Annexin V apoptosis detection kit (BD Pharmingen, San Diego, CA, www.bdbiosciences.com/pharmingen) and analyzed using flow cytometry.
Cell Proliferation and Cell Cycle Experiments
For cell proliferation analysis, the bromodeoxyuridine (BrdU) kit (BD Biosciences, San Diego, CA, www.bdbiosciences.com) was used according to manufacturer’s instructions. Briefly, hESCs were incubated with 10 µm BrdU for 3 hours at 37°C, rinsed in PBS, and then stained for TRA-1-81. Fluorescence-activated cell sorting (FACS) analysis was used to determine the percentage of TRA-1-81+ cells that had incorporated BrdU. For cell cycle analysis, cells were stained for TRA-1-81 to identify stem cells. Stained cells were resuspended in 0.875 ml of cold PBS, mixed, and then fixed by adding 0.125 ml of cold 2% PFA for 30 minutes at 4°C. After washing with buffer (1% fetal bovine serum [FBS] in PBS), cells were permeabilized using 0.2% Tween-20 in PBS for 15 minutes at 37°C. After buffer wash, cells were resuspended in 10 µg/ml of propidium iodide (PI) and 11.25 Kunitz units of RNase (Sigma), and incubated for 30 minutes at 4°C prior to FACS analysis. Modfit LT software was used to quantify the percentage of cells in each stage of the cell cycle.
Embryoid Body Formation and Directed Differentiation
Confluent hESC colonies were detached by incubation with 1 mg/ml collagenase (Invitrogen) for 30–60 minutes and replated onto low-attachment six-well plates (Fisher, Chino, CA, www.fishersci.com) in embryoid body (EB) medium consisting of DMEM-F12 (Invitrogen) supplemented with 15% defined fetal bovine serum (Hyclone), 5% knockout serum replacement (Invitrogen), 1 mM l-glutamine (Invitrogen), 2 mM 2-mercaptoethanol, 0.1 mM nonessential amino acids (Invitrogen), and 1 mM penicillin/streptomycin (Hyclone). For neural-directed differentiation, day 5 EBs were plated on fibronectin (20 µg/ml)-coated dishes and cultured in DMEM/F12 supplemented with N2 and B27 (Invitrogen), 10 ng/ml bFGF, 1 ng/ml IGF, 1 ng/ml platelet-derived growth factor α polypeptide, and 10 ng/ml epidermal growth factor (all from Peprotech, Rocky Hill, NJ, www.peprotech.com) for an additional 5–7 days [25]. For mesoderm differentiation [26], EBs were cultured for 8 days in Stem Line II medium (Sigma) that was supplemented with 1× CD lipid concentrates, 2 mM GlutaMAX, 1× insulin transferrin selenium, penicillin/streptomycin (100 units/100 mg/ml) all from Invitrogen, 400 µM monothioglycerol, and 50 mg/ml ascorbic acid (Sigma). The following growth factors were added: 10 ng/ml bone morphogenetic protein (BMP-4) (R&D Systems, Minneapolis, MN, www.rndsystems.com), 5 ng/ml bFGF (Invitrogen), and 20 ng/ml vascular endothelial growth factor (R&D Systems). For endoderm differentiation, hESCs were treated with 100 ng/ml activin (Peprotech) for 1 day and with 1% FBS and 100 ng/ml of activin for the next 2 days in DMEM/F12.
Teratomas
To prepare hESCs for injection, two confluent wells were collected using 1 mg/ml of collagenase type IV (Invitrogen) for 5 minutes at 37°C. Colonies were dissociated using a 5 ml pipette and strained using a 100 µm nylon strainer before centrifugation at 1,000 rpm. Cell pellets were resuspended in 50 µl of PBS and injected into the testes of 4–8-week-old SCID BEIGE mice (Charles River, Wilmington, MA, www.criver.com) as described previously [27]. After 8 weeks, teratomas were isolated and fixed in 4% PFA for 24 hours. Fixed teratomas were embedded and processed by the Translational Pathology Core Laboratory at the David Geffen School of Medicine (TPCL). If teratomas were large, four to six quadrants were isolated and fixed in PFA as described above.
For endoderm and mesoderm lineage quantification, five random areas from four different H&E-stained sections per teratoma (n = 6) were visually analyzed for gut formation (endoderm) or bone and muscle (mesoderm). For quantification of ectoderm in teratoma sections, control and PTEN KD sections (n = 3) were immunostained for NESTIN and microtubule-associated protein 2 (MAP2). Images of these teratoma sections were acquired using the Ariol SL-50 scanner (Applied Imaging Corp., Grand Rapids, MI, www.applied imaging.com). After optimal exposure times were determined, slides were scanned at 20× magnification for 4′,6-diamidino-2-phenylindole (DAPI), FITC (NESTIN), and tetramethyl rhodamine isothiocyanate (TRITC) (MAP2) signals. Threshold levels of individual signals were optimized prior to final analysis and quantification. The Ariol software was then used to quantify the levels of FITC and TRITC signals relative to DAPI signals. Scanning and analysis were performed through the TPCL. For all teratoma quantification assays, statistical analysis was performed using Student’s t test.
Results
PTEN KD hESCs Have Increased Self-Renewal, Survival, and Proliferation
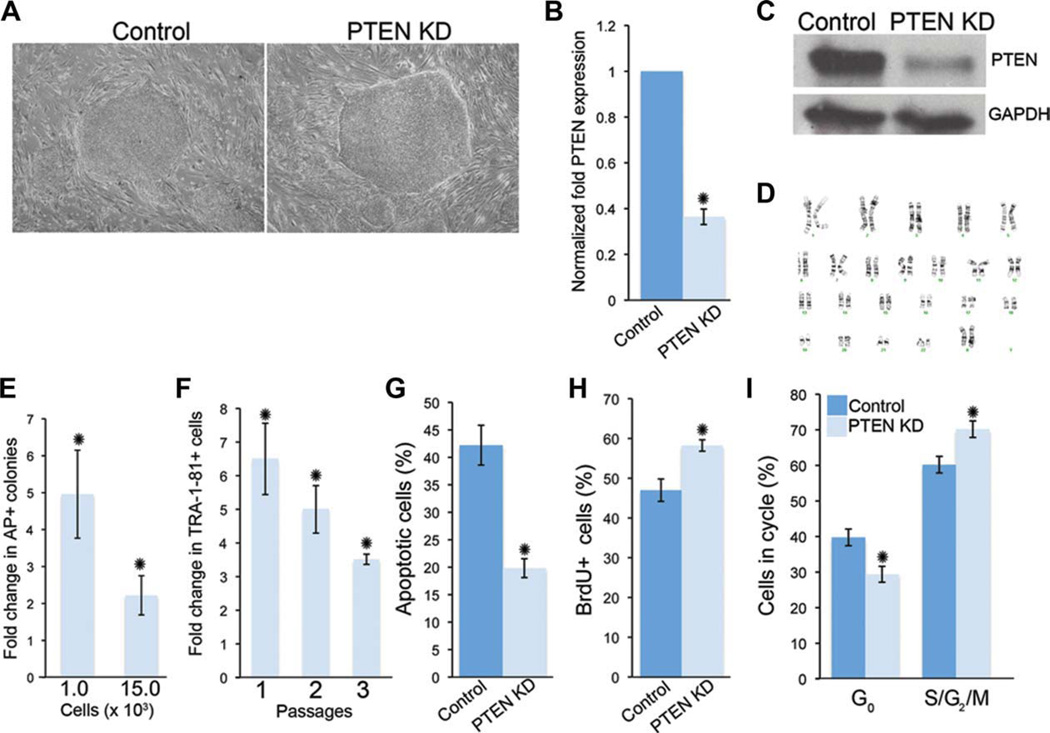
Pten has a well-known role in the negative regulation of self-renewal in mouse embryonic, neural, and mammary stem cells [9, 13, 14]. Furthermore, studies have shown that PI3K/Akt signaling is an important regulator of cell fate decisions in mESCs and hESCs [19, 28]. Therefore, to evaluate the role of PTEN in hESC self-renewal and survival, we generated H9 (data shown in Figs. 1–7, Supporting Information Figs. 3, 5, and 6) or HSF1 (Supporting Information Figs. 1, 2, 4 and, 7) hESCs with a knockdown of PTEN (PTEN KD) using lentiviral mediated shRNA (pLKO.1-shRNA PTEN) (Fig. 1A and Supporting Information Fig. 1a). This system resulted in a 60–80% reduction of PTEN expression compared to nontarget controls (control) as shown by quantitative real time polymerase chain reaction (qPCR) and Western blot analyses (Fig. 1B, 1C). Similar results were seen with HSF1 PTEN KD hESCs (Supporting Information Fig. 1). PTEN KD hESCs are karyotypically stable after multiple passages (Fig. 1D). Interestingly, control but not PTEN KD hESCs were more prone to spontaneously differentiate (preferentially toward a fibroblast-like morphology) when grown in defined conditions (Supporting Information Fig. 2). To examine the effects on cell survival or self-renewal on PTEN knockdown, we used a low-density assay in which 1,000 or 15,000 control and PTEN KD single cells were plated on mouse embryonic fibroblasts (MEFs). After 5 days under self-renewing conditions, colonies were identified with AP staining. There was a fivefold and twofold increase, respectively, in the number of PTEN KD colonies compared to control hESCs (Fig. 1E) in these assays. In a similar experiment, we plated 105 single hESCs on MEFs, and after 5 days of culture under self-renewal conditions, the percentage of stem cells was evaluated using the pluripotency marker, TRA-1-81. As shown in Figure 1F, PTEN KD hESCs have a sixfold increase in TRA-1-81+ cells compared to control hESCs. A significant increase in TRA-1-81+ cells was sustained over three passages (Fig. 1F). To confirm that PTEN KD hESCs have increased self-renewal compared to control cells, we repeated similar experiments for five passages and found that at every passage, PTEN KD hESCs have between a threefold and fourfold increase in the number of colonies after each passage (Supporting Information Fig. 3a, 3b). In addition, by passage 5, control hESCs began to differentiate into fibroblasts-like cells, generating smaller colonies compared to PTEN KD hESCs (Supporting Information Fig. 3a). Because PTEN regulates cell survival in a number of stem cell populations, we evaluated changes in cell death in these assays. Under self-renewing conditions, TRA-1-81+ PTEN KD hESCs had a twofold decrease in the number of cells undergoing apoptosis as measured by PI and Annexin V staining (Fig. 1G). Therefore, PTEN KD hESCs have increased survival, which may enable their increased proliferation or self-renewal. PTEN modulates cell proliferation and entry into the cell cycle in multiple cell types in vitro and in vivo [29]. We therefore evaluated cell proliferation using BrdU pulsing in PTEN KD hESCs. When cultured in bulk, TRA-1-81+ PTEN KD hESCs have a significantly higher percentage of cells incorporating BrdU (Fig. 1H). Further, PTEN KD TRA-1-81+ hESCs have a decreased percentage of cells in G0/G1 and increased S/G2/M populations (Fig. 1I). Similar alterations in cell cycle profiles have been observed in PTEN-deficient mESCs [9]. Taken together, these results demonstrate that PTEN KD hESCs have increased self-renewal, survival, and proliferation.
Figure 1.
PTEN KD human embryonic stem cells (hESCs) have reduced apoptotic cells and increased AP-positive cells over continuous passage in culture. (A): Bright-field images of non-target control (control) and PTEN KD hESCs, passaged on a mouse feeder layer under self-renewing conditions. (B): Quantitative real time PCR (qPCR) was used to evaluate PTEN levels in PTEN KD hESCs relative to control hESCs. (C): Western blot analysis of PTEN protein levels in control and PTEN KD hESCs. (D): Metaphase spread of PTEN KD hESCs at passage 40 indicates normal karyotype. (E): To evaluate self-renewal of PTEN KD hESCs, 1.0 × 103 or 15.0 × 103 single cells were cultured under self-renewing conditions for 5–7 days. The numbers of AP-positive colonies were counted and the fold increase in PTEN KD colonies relative to control is shown. (F): To assess changes in the number of stem cells over multiple passages, 1 × 105 single cells were cultured under self-renewing conditions for three passages [1–3] and FACS analysis for TRA-1-81 was performed to determine the percentage of hESCs after 5–7 days. Graph depicts the fold increase in TRA-1-81-positive hESCs relative to control hESCs. (G): To evaluate changes in cell survival, TRA-1-81-positive hESCs in (F) were stained with Annexin V and PI and percentages were determined using FACS analysis. (H): To evaluate cell proliferation, incorporated BrdU in TRA-1-81-positive hESCs was evaluated using FACS analysis. (I): To determine the percentage of hESCs in each stage of the cell cycle, TRA-1-81-positive control and PTEN KD hESCs were stained for PI and analyzed via FACS. Experiments were performed using control and PTEN KD passage 40–45 hESCs. Error bars represent standard error of three independent experiments and asterisks denote a p value <.05. Abbreviations: AK, alkaline phosphatase; BrdU, bromodeoxyuridine; FACS, fluorescence-activated cell sorting; GADPH, glyceraldehyde-3-phosphate dehydrogenase; PI, propidium iodide; PTEN KD, PTEN knockdown.
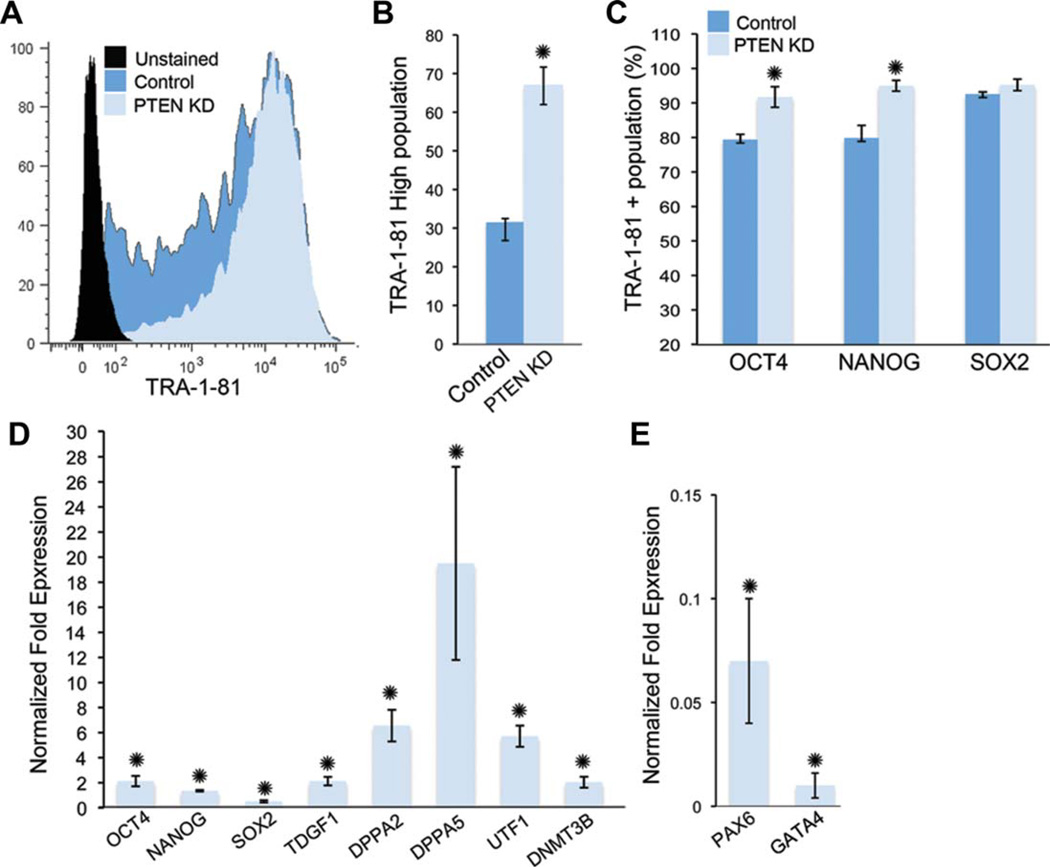
Figure 7.
PTEN KD human embryonic stem cells (hESCs) are more homogeneous and have increased pluripotency gene expression under self-renewing conditions. (A): FACS histogram of unstained control and PTEN KD hESCs cultured under self-renewing conditions showing total TRA-1-81-positive events. (B): The percentage of high TRA1-81-positive hESCs in control and PTEN KD hESCs. (C): FACS analysis of nuclear protein expression of OCT4, NANOG, and SOX2 in control and PTEN KD hESCs. Graph shows the percentage of TRA-1-81-positive hESCs that were positive for OCT4, NANOG, or SOX2. (D): Expression of pluripotency markers was evaluated using quantitative PCR (qPCR). (E): Expression of the early progenitor markers, PAX6 and GATA4, in control and PTEN KD hESCs. Graphs show PTEN KD fold over control. All experiments were performed using control and PTEN KD passage 37–45 hESCs. Error bars represent standard errors of three independent experiments and asterisks denote p value <.05. Abbreviations: FACS, fluorescence-activated cell sorting; PCR, polymerase chain reaction; PTEN KD, PTEN knockdown; UTF, undifferentiated embryonic cell transcription factor.
PTEN KD hESCs Have Increased Activity of AKT and S6 But Not Active β-Catenin
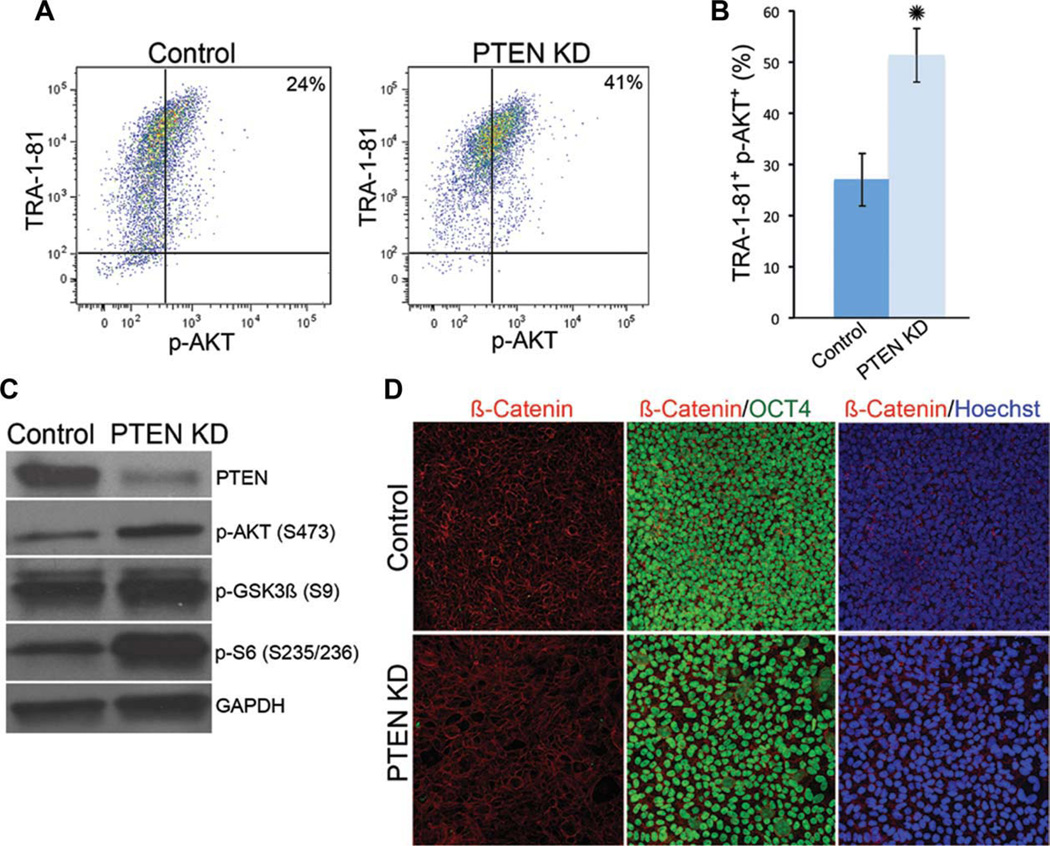
To determine the signaling mechanisms by which PTEN regulates hESC self-renewal and survival, we performed Western blot analysis to assess the phosphorylation status of components that are downstream of PTEN, such as AKT, S6, and GSK3β. As expected, PTEN KD hESCs have increased levels of p-AKT (Fig. 2C). We used FACS analysis to quantify the percentage of PTEN KD hESCs that have p-AKT (Fig. 2A, 2B). Levels of p-S6, the ribosomal substrate of S6 kinase, were higher in PTEN KD hESCs than in control hESCs (Fig. 2C). These studies suggest that the AKT/mTOR pathway is activated in the absence of PTEN in hESCs. Pten activity modulates Wnt signaling via indirect effects of Akt activity in mouse stem cells [12, 13]. To determine if PTEN KD hESCs have changes in components of the WNT pathway, we evaluated the levels of phosphorylated GSK3β and localization of β-catenin in the PTEN KD hESCs. We did not observe a significant difference in the levels of p-GSK3β in the PTEN KD hESCs (Fig. 2C). Furthermore, β-catenin localized to the plasma membrane of OCT4-positive hESCs in both H9 control and PTEN KD hESCs (Fig. 2D), suggesting that β-catenin is not activated on PTEN knockdown as has been demonstrated in other stem cell systems [12, 13]. It was previously reported that β-catenin was localized to both the plasma membrane and nucleus of hESCs grown in conditioned media. However, recent studies have performed a more detailed analysis of β-catenin localization, demonstrating that OCT4-positive hESCs do not express nuclear β-catenin and that the stabilization of β-catenin in hESCs leads to disruption of self-renewal [30, 31]. To confirm this, we examined the localization of β-catenin in HSF1 control and PTEN KD hESCs grown on MEFs and found that β-catenin was localized to the plasma membrane (Supporting Information Fig. 1E). Interestingly, differentiated cells within hESC colonies (OCT4-negative) had accumulated β-catenin in the nucleus (data not shown). Further continuous passage of PTEN KD hESCs does not result in loss of membrane localization of β-catenin (Supporting Information Fig. 3c). These results demonstrate that the level of PTEN knockdown in our system results in the phosphorylation of AKT and S6 but not activation of WNT signaling.
Figure 2.
Signaling downstream of PTEN KD in human embryonic stem cells (hESCs) includes activation of AKT and S6, but not β-catenin. (A): FACS plots showing the percentage of TRA-1-81-positive hESCs that are positive for p-AKT S473. (B): Quantification of FACS analysis in (A). (C): Western blot analysis of PTEN, p-AKT, phospho-GSK3β (p-GSK3β S9), and phospho-S6 (p-S6S235/236) protein levels in control and PTEN KD hESCs. (D): Immunofluorescent staining of β-catenin (red), OCT4 (green), and Hoechst (blue) shows similar membrane localization of β-catenin in control and PTEN KD hESCs. Experiments were performed using control and PTEN KD passage 40–45 hESCs. FACS plot, immunofluorescent staining, and Western blots are representative of three independent experiments. Error bars represent standard error of three independent experiments and asterisks denote a p value <.05. Abbreviations: FACS, fluorescence-activated cell sorting; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GSK, glycogen synthase kinase; PCR, polymerase chain reaction; p-AKT, phospho-AKT; PTEN KD, PTEN knockdown.
PTEN KD hESCs Have Altered Differentiation Potential
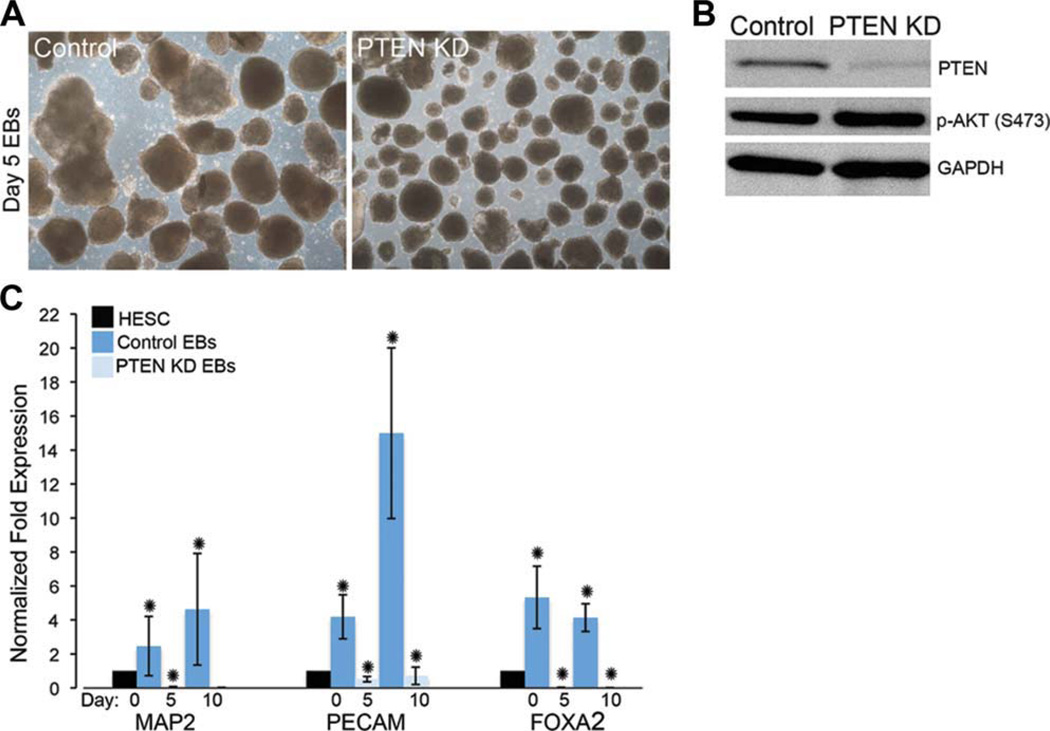
Gene ablation studies in mice defined a role for Pten in regulating embryonic differentiation [32]. To determine whether PTEN KD hESCs have aberrant differentiation, we performed bulk differentiation using EB formation (Fig. 3A). To assess differentiation of PTEN KD hESCs, we differentiated hESCs into EBs for 5 and 10 days. PTEN KD hESCs form uniform EBs, however, decreased differentiation of all three germ lineages was observed by qPCR of selected lineage markers (Fig. 3C). In the evaluation of HSF1 control and PTEN KD day 5 EBs, we similarly observed decreased expression of endoderm and mesoderm makers, forkhead box A2 (FOXA2) and platelet/endothelial cell adhesion molecule (PECAM) (Supporting Information Fig. 4a). However, we observed an increase in the expression of the mature neural marker, neurogenic differentiation 1 (NEUROD1), in HSF1-day 5 EBs compared to control cells, suggesting there may be a bias toward ectoderm differentiation in some lines on PTEN KD (Supporting Information Fig. 4b). We further verified that PTEN expression remained decreased after EB differentiation using Western blot analysis. Importantly, in H9 PTEN KD EBs, we found increased p-AKT expression levels (Fig. 3B), demonstrating that PTEN knockdown and downstream signaling activity is still maintained in the EB system. Interestingly, in the HSF1 PTEN KD EBs, while PTEN protein expression was reduced compared to controls, we found minimal increase in p-AKT or OCT4 levels but an increase in differentiation, suggesting that levels of p-AKT are critical in regulating the differentiated state (Supporting Information Fig. 4c). We did however find significant expression of p-AKT, OCT4, and NANOG maintained in the H9 PTEN KD differentiated cells using Western blot assays (Supporting Information Fig. 5 and data not shown). H9 PTEN KD EBs maintained OCT4 expression over the course of the directed differentiation assays, suggesting that PTEN KD hESCs maintain expression of pluripotency network genes and have reduced ability to differentiate compared to control hESCs when high levels of p-AKT are maintained.
Figure 3.
PTEN KD hESCs differentiate less efficiently as EBs compared to control hESCs. (A): Bright-field images of control and PTEN KD day 5 EBs generated from passage 45 hESCs. (B): Western blot analysis of PTEN and p-AKT in control and PTEN KD hESCs. (C): To evaluate the efficiency of EB differentiation, quantitative PCR (qPCR) was used to evaluate gene expression of the lineage-specific makers, MAP2 (ectoderm), PECAM (mesoderm), and FOXA2 (endoderm). Expression levels of control and PTEN KD Day 5 and Day 10 EBs are shown relative to control hESCs. EBs and Western blots are representative of three independent experiments. Error bars represent standard error of three independent experiments and asterisks denote p values <.05. Abbreviations: EBs, embryoid bodies; GADPH, glyceraldehyde-3-phosphate dehydrogenase; FOXA2, forkhead box A2; hESCs, human embryonic stem cells; MAP2, microtubule-associated protein 2; p-AKT, phospho-AKT; PCR, polymerase chain reaction; PECAM, platelet/endothelial cell adhesion molecule.
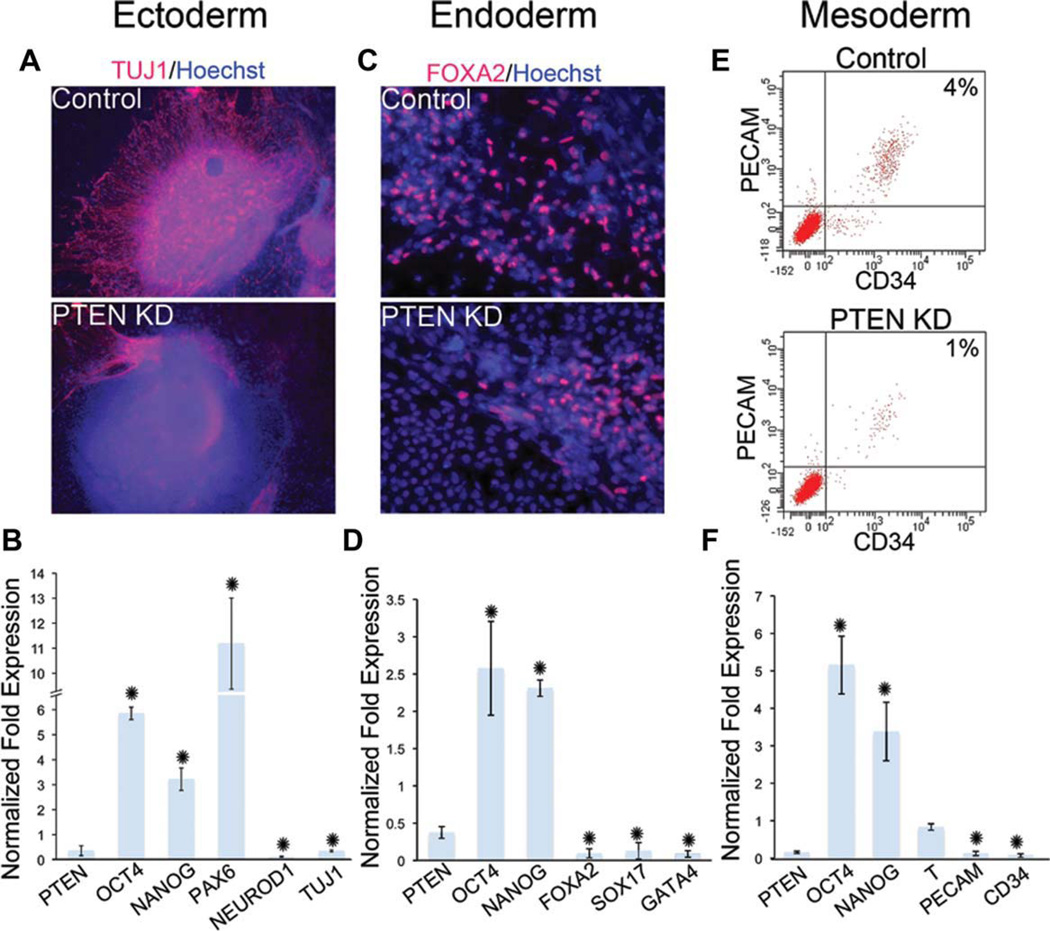
To perform a more comprehensive analysis of differentiation in PTEN KD hESCs with high levels of p-AKT, we performed directed differentiation toward ectoderm, endoderm, and mesoderm by growth factor supplementation and growth on defined matrices using established protocols [25, 26]. In all cases, PTEN KD hESCs differentiated less efficiently than control hESCs. To visualize the expression of neural markers after neural differentiation, we performed immunocytochemisty using TUJ1, a mature neural marker and PAX6, a neural progenitor marker. Compared to control, PTEN KD hESCs differentiated into PAX6-positive cells but had reduced TUJ1-positive staining (Fig. 4A and Supporting Information Fig. 6). To examine the expression of additional markers after neural differentiation, we performed qPCR for stem cell, progenitor, and mature lineage markers. As shown in Figure 4B, while the expression of the mature neural markers NEUROD1 and TUJ1 were far below control levels, we observed an increase in OCT4, NANOG, and PAX6, suggesting an increase in the presence of neural progenitors and maintenance of pluripotency gene expression on PTEN knockdown using directed differentiation. To examine endoderm potential, we used immunofluorescence analysis to determine the presence of the endoderm marker FOXA2. PTEN KD hESCs have reduced ability to differentiate into FOXA2-positive cells (Fig. 4C), and the reduction in endoderm potential was confirmed using qPCR for additional markers (Fig. 4D). Interestingly, increased expression of the pluripotency markers, OCT4 and NANOG, was also observed (Fig. 4D). Under conditions that promoted mesoderm differentiation, PTEN KD hESCs were less efficient than control cells in differentiating into CD34-positive and PECAM-positive cells, as demonstrated using FACS analysis (Fig. 4E). This aberrant differentiation was confirmed by the examination of additional mesoderm and pluripotency markers using qPCR (Fig. 4F). Similar to the results found after neural and endodermal lineage differentiation, OCT4 and NANOG gene expression was increased in the PTEN KD cells under mesoderm promoting conditions compared to control differentiated cells.
Figure 4.
PTEN KD human embryonic stem cells (hESCs) have a decreased potential to differentiate into late-stage neuroectoderm, and early and late-stage endoderm and mesoderm. Control and PTEN KD hESCs were differentiated into neural (A, B), endoderm (C, D), and mesoderm (E, F) lineages. (A): Immunofluorescent staining of TUJ1 (red) and Hoechst (blue) after neural directed differentiation of control and PTEN KD differentiated cells. (B): Quantification of the experiment in (A) using qPCR of neuroectoderm markers PAX6, NEUROD1, and TUJ1. (C): Immunofluorescent staining of FOXA2 (red) and Hoechst (blue) after endoderm differentiation of control and PTEN KD differentiated cells. (D): Quantification of the experiment in (C) using quantitative PCR (qPCR) of endoderm makers FOXA2, SOX17, and GATA4. (E): FACS analysis of PECAM and CD34 expression in control and PTEN KD cells after mesoderm differentiation. (F): Quantification of the experiment in (E) using qPCR of mesoderm markers, BRACHYURY (T), PECAM, and CD34 in PTEN KD cells relative to control. qPCR graphs show expression of PTEN KD cells relative to control cells. In all differentiation experiments, the pluripotency genes, OCT4 and NANOG, remain highly expressed in PTEN KD cells. All experiments were performed using control and PTEN KD passage 37–45 hESCs. Immunofluorescent staining and FACS analysis are representative of three independent experiments yielding similar results. qPCR graphs represent data from three independent experiments. Error bars represent standard error and asterisks denote p values <.05. Abbreviations: FACS, fluorescence-activated cell sorting; FOXA2, forkhead box A2; NEUROD1, neurogenic differentiation 1; PCR, polymerase chain reaction; PECAM, platelet/endothelial cell adhesion molecule; PTEN KD, PTEN knockdown.
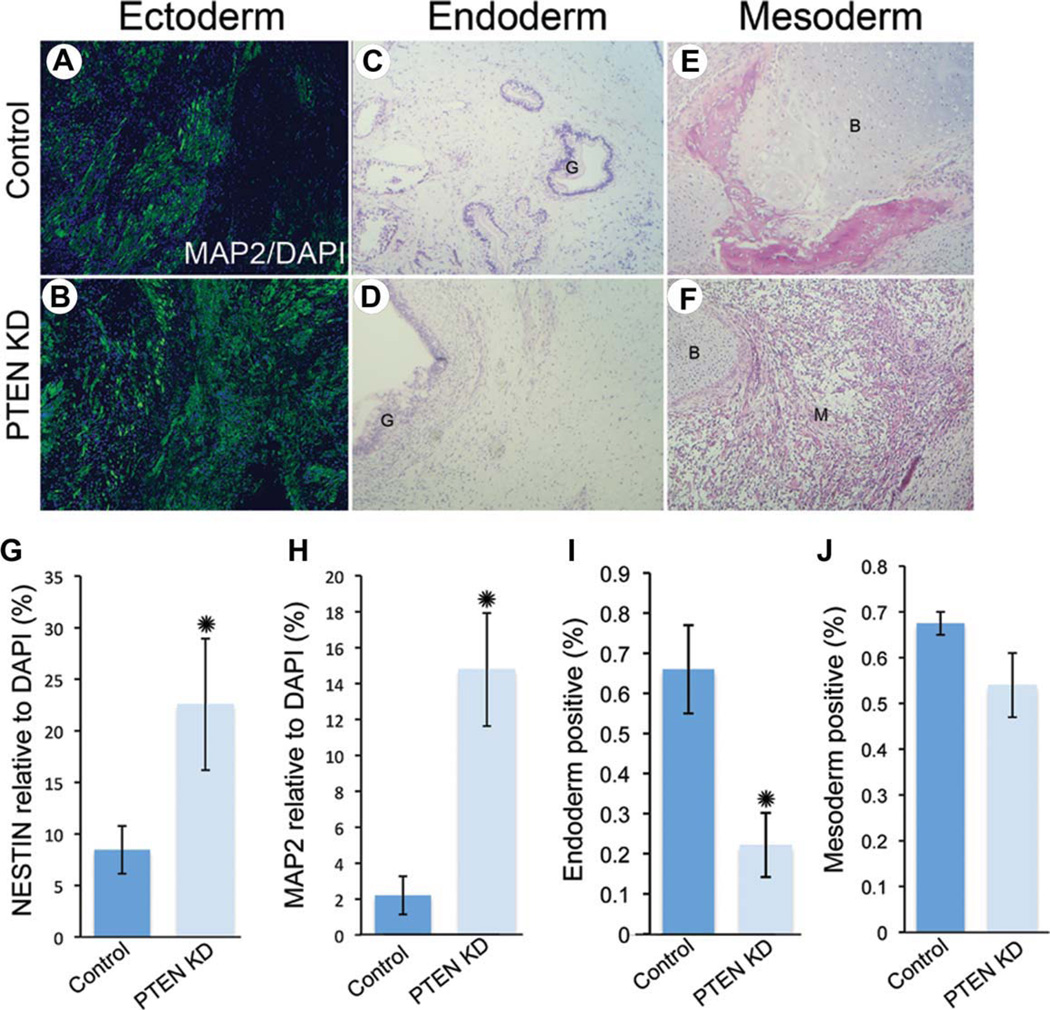
Figure 6.
PTEN KD human embryonic stem cells (hESCs) generate teratomas containing all three germ layers with increased ectoderm and reduced endoderm potential. Control (A) and PTEN KD (B) teratoma sections were stained for MAP2 (green) and DAPI (blue) to show neuroectoderm regions. H & E stained control (C, E) and PTEN KD (D, F) teratoma sections showing gut (G), bone (B), and muscle (M) formation. Teratoma sections were immunostained for NESTIN (G) and MAP2 (H) and quantified using Ariol software (n = 4). Quantification of endoderm (I) and mesoderm (J) using H & E staining (for gut or bone and muscle) in control and PTEN KD teratoma sections (n = 6). Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; MAP2, microtubule-associated protein 2; PTEN KD, PTEN knockdown.
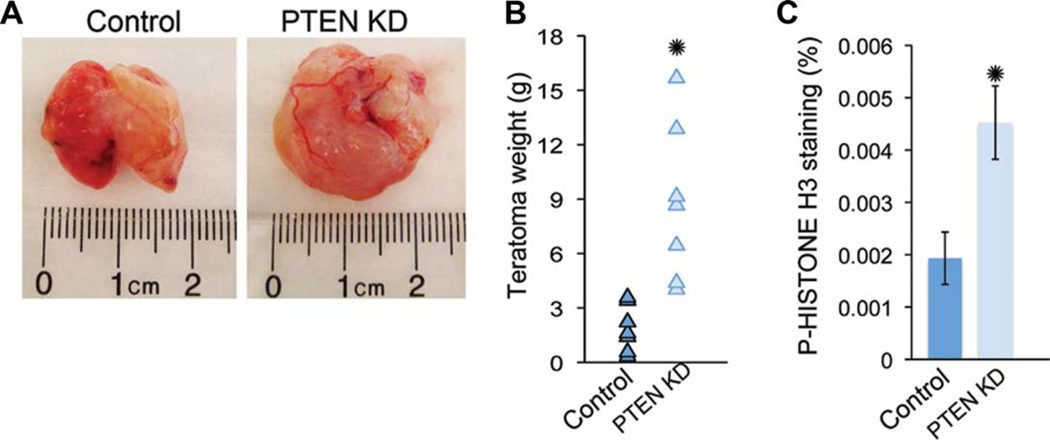
As in vitro assays may not recapitulate differentiation capabilities in vivo, we performed teratoma analysis of H9 PTEN KD and control hESCs. To examine in vivo differentiation capability on PTEN knockdown, we injected approximately 1 × 106 hESCs per testis in SCID BEIGE 4–6-week-old mice. Two months after the survival surgery, we isolated, sectioned, and stained teratomas using H&E or immunofluorescence. Interestingly, teratoma size was increased in PTEN KD teratomas in seven of seven experiments compared to controls (Fig. 5A, 5B). A corresponding increase in p-histone H3 was seen (Fig. 5C), demonstrating that PTEN KD hESCs generate larger and more proliferative teratomas compared to control hESCs. To quantify differentiation in vivo, we counted H&E-stained structures resembling gut or bone/cartilage for endoderm and mesoderm, respectively. For ectoderm quantification, we stained teratoma sections with two markers for early and late differentiation using immunofluorescence and analyzed using Ariol software. PTEN KD teratomas had increased expression of both NESTIN and MAP2 (Fig. 6A, 6B, 6G, 6H) and reduced formation of gut-like structures (Fig. 6C, 6D, 6I). No change was seen in mesoderm potential as measured by counting H&E-stained bone/cartilage-like structures (Fig. 6E, 6F, 6J). In summary, PTEN KD hESCs generate more proliferative teratomas that are much larger in size compared to controls. PTEN KD hESCs can differentiate into all three germ layers but have increased potential for ectoderm and reduced potential for endoderm fate in vivo.
Figure 5.
PTEN KD human embryonic stem cells (hESCs) generate large teratomas when injected into immunocompromised mice. (A): Bright-field images of control and PTEN KD teratomas. (B): Weight in grams (g) of control and PTEN KD teratomas (n = 7). (C): To evaluate cell proliferation in teratomas, paraffin-embedded sections from control and PTEN KD teratomas (n = 3) were stained for p-HISTONE H3 and DAPI and quantified using Ariol software. Teratomas were generated using passage 40–45 hESCs. Error bars represent standard errors and asterisks denote p value <0.05. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; PTEN KD, PTEN knockdown.
PTEN Regulates Levels of Pluripotency Network Genes in hESCs
To improve our understanding of the mechanism by which PTEN regulates pluripotency and differentiation, we extended our analysis to evaluate pluripotency regulators in PTEN KD hESCs cultured in vitro under self-renewing conditions. Recent studies have shown that hPSCs are heterogeneous with regard to expression of pluripotency and lineage commitment markers. Furthermore, regulation of extrinsic signals may modify the heterogeneous hPSC state [33, 34]. To determine if PTEN KD hESCs have reduced heterogeneity of marker expression, we examined a panel of pluripotency markers. Remarkably, PTEN KD hESCs have a more uniform expression (RNA, protein, or both) of a subset of pluripotency markers compared to control hESCs (Fig. 7). TRA-1-81 expression is more uniform (Fig. 7A) with an increase in the percentage of PTEN KD hESCs expressing higher levels of TRA-1-81 (Fig. 7B). Furthermore, performing intracellular FACS analysis of the pluripotency markers demonstrated a significant increase in the percentage of cells positive for OCT4 and NANOG but not SOX2 (Fig. 7C). In addition, qPCR validated that these and additional markers are expressed at higher levels, with the greatest increase seen in a subsets of pluripotency markers including teratocarcinomaderived growth factor (TDGF), undifferentiated embryonic cell transcription factor (UTF), DPPA2, DPPA5, and DNMT3B (Fig. 7D). Finally, we examined the expression of early progenitor markers in the PTEN KD hESCs. The expression of early progenitor markers, such as PAX6 and GATA4, is minimal in hESCs that express the highest levels of pluripotency genes [35]. Likewise, hESCs that are less pluripotent express higher levels of these early progenitor markers. Supporting the notion that PTEN KD hESCs are more homogeneous with respect to pluripotency gene expression, we found minimal PAX6 and GATA4 gene expression in the PTEN KD hESCs cultured under self-renewing conditions relative to control cells (Fig. 7E). Similar results were found in HSF1 PTEN KD hESCs (Supporting Information Fig. 7). Taken together, this data suggests that regulation through PI3K/AKT signaling can modify the expression of pluripotency markers and the pluripotent state.
Discussion
Previous studies have established that the PI3K/AKT pathway is important for survival and self-renewal in hESCs; however, how this regulation is achieved and its relationship to the pluripotency network is not fully understood. This work demonstrates for the first time a clear role for PTEN, a negative regulator of PI3K/AKT signaling, in modulating the hESC pluripotent state and lineage specification. Using hESCs with a knockdown of PTEN, and consequently activation of AKT, we found that PTEN KD hESCs have increased self-renewal, survival, and proliferation over multiple passages, as well as reduced spontaneous differentiation. These hESCs have drastically increased p-S6 levels, indicative of activation of the mTOR/S6 pathway. When assayed for differentiation in vitro, PTEN KD hESCs maintain high expression levels of OCT4 and NANOG with lower expression of lineage-specific markers. Moreover, PTEN KD hESCs display inefficient terminal differentiation, as shown in the directed differentiation assays. Interestingly, PTEN KD hESCs have increased neural progenitor differentiation in vitro, demonstrated by the increased levels of PAX6 taken together with lower levels of mature neural markers. In vivo, PTEN KD hESCs differentiate into teratomas with greater cell proliferation, and consequently, larger mass compared to controls. In addition, PTEN KD hESCs display a bias toward neuroectoderm in vivo, with greater neural tissue and less endodermal tissue in the teratomas. Furthermore, PTEN KD hESCs consistently show significant increases in expression of pluripotency network genes, accompanied by a decrease in early progenitor gene expression. Taken together, these results suggest that PTEN is a key regulator of hESC pluripotent state.
Downstream of PI3K, Akt activity regulates multiple proteins such as GSK3β, mTOR, and S6K. In mESCs, it has been shown that PI3K/Akt signaling activates Wnt/GSK3β/β-catenin signaling to regulate self-renewal [36, 37]. In this study, using shRNA-mediated knockdown of PTEN in hESCs, we found no evidence of WNT signaling in the presence of p-AKT, as β-catenin localization was observed at cell membranes. PTEN KD hESCs, however, have activation of the mTOR/S6 pathway, which has a direct role in the regulation of protein synthesis in hESCs [38]. It is becoming increasingly clear that regulation of ESC fate is not only due to mRNA expression levels but also the post-translational mechanisms regulate both self-renewal and differentiation [39]. Therefore, one key regulator of the pluripotent state in PTEN KD hESCs may be through the S6 pathway. Indeed, mTOR has previously been shown to be important for hESC self-renewal and repression of mesoderm and endoderm differentiation [40].
The PTEN phosphatase is also well known for its contribution to tumorigenesis, as its deregulation causes cellular transformation in a variety of cell types, including testicular germ cells [41]. Although PTEN KD hESCs exhibit properties that may provide a growth advantage such as increased survival and proliferation, no aneuploidy was observed. In addition, although able to differentiate, PTEN KD hESCs exhibit a bias toward neuroectoderm in the teratoma assays. PTEN KD hESCs have increased expression of NANOG, and overexpression of NANOG in hESCs has been shown to enable feeder-free growth, as well as differentiation toward primitive ectoderm [42]. Alternatively, enhanced PI3K and NANOG activity during teratoma formation could lead to transformation. Previous work has shown that transformed mESCs can result in formation of failed-to-differentiate stem cells similar to generation of EC cells during teratocarcinoma formation [27]. Indeed, recent work has shown that loss of Pten in mESCs causes tumor initiation due to increased Nanog expression, even when cells are induced to differentiate with retinoic acid [43]. Transformation assays in vivo remain to be explored in PTEN KD hESCs.
Knockdown of Pten in the mouse brain has been shown to increase the proliferation of progenitor cells but not result in a bias of terminal differentiation [14, 29]. In directed differentiation assays, the greater expression of PAX6 found in PTEN KD hESCs is indicative of an increase in neural progenitor differentiation. As PAX6 was recently identified as the essential transcription factor required for the transition of pluripotent cells to the neuroectoderm fate [44], the regulation of PTEN activity may be crucial in controlling neuroectoderm fate specification in vitro and in vivo. These studies suggest that PTEN could prove to be an important regulator of direct reprogramming strategies, where it may be required to generate human neural progenitor cells rather than terminally differentiated neurons from fibroblasts as has previously been shown [45].
Remarkably, hESCs with a knockdown of PTEN showed a higher percentage of cells expressing PSC markers including TRA-1-81 and showed consistent and statistically significant increases in expression of pluripotency network genes, including OCT4, NANOG, TDGF, DPPA5, DPPA2, UTF, and DNMT3B. This increase in expression of pluripotency network genes was accompanied by a decrease in early progenitor gene expression. Recent studies have shown that hESCs are heterogeneous with regard to expression of pluripotency and lineage commitment markers and that this can be modified by regulation of extrinsic signals [33, 46, 47]. This data suggests that PTEN is a key regulator of and can alone modify the hESC pluripotent state. Interestingly, a number of recent studies have provided insight into the different states of mPSCs and hPSCs [2, 47]. Small molecules targeting several different pathways were required in transition of hESCs to a more mESC-like or naïve state; however, the role of the PI3K/AKT pathway in this transition was not examined. In addition, maintenance of the naïve state is difficult to achieve without the use of continuous forced overexpression of pluripotency transcription factors. This work suggests that by modifying the PTEN pathway, we may be one step closer in directing the naïve state with less need to perform multiple pathway modifications, as PTEN KD hESCs already have increased expression of subsets of shared pluripotency network genes, including OCT4 and NANOG and intrinsic signaling molecules such as AKT.
Although we have shown a direct correlation with enhanced PI3K/AKT signaling and expression of core pluripotency network genes, the mechanism by which this pathway signals to the core regulatory circuitry in hESCs is not known. In mESCs, it has been shown that the transcription factors associated with the Lif, Wnt, and Bmp4 signaling pathways (Stat3, Tcf3, and Smad1) tend to co-occupy enhancers bound by Oct4, Sox2, and Nanog, thereby allowing direct control of genes within the core circuitry by these signaling pathways [48–53]. It is assumed that signals mediated by these pathways are delivered directly to the enhancers of genes within the core regulatory circuitry, which then regulates the pluripotent state. Recent work further suggests that post-translational modifications provide another level of regulation of mESCs, as it has recently been shown that Akt directly interacts with Sox2 and promotes its stabilization through phosphorylation at Thr118. This results in enhanced protein abundance and transcriptional activity of Sox2 in mESCs [17]. In this work, we did not see an increase in SOX2 as measured by protein and RNA expression levels; however, it will be important to determine if post-translational modifications of the core pluripotency transcription factors are seen in future studies.
Conclusions
In conclusion, we have shown that PTEN is a critical regulator of hESC fate, as PTEN KD hESCs have pronounced changes in differentiation potential and maintenance of the pluripotency program. Regulation of PTEN activity and associated downstream signals will be an important parameter to assess in routine pluripotent stem cell cultures, and understanding this regulation could reduce heterogeneity often seen across multiple hPSC lines. This work suggests that PTEN/PI3K/AKT activity is likely to be an important mediator of the ground state of pluripotency as well as play a key role in direct reprogramming strategies.
Supplementary Material
Acknowledgments
We would like to thank Dr. Amander Clark for providing feedback throughout this study; Dr. Anne Lindgren in Dr. Clark’s laboratory for providing technical assistance with the survival surgeries for teratoma formation; Dr. Gautam Dravid in Dr. Crook’s laboratory for assistance with the mesoderm directed differentiation assays and David Chan in Dr. Lowry’s laboratory for helpful discussions regarding the endoderm directed differentiation assays; Clara Magyar from the TPCL for providing assistance with the analysis of immunofluorescent teratoma sections; and the members of the Pyle laboratory for providing constructive criticism and technical support for these studies. This work was supported by funds from the UCLA Jonsson Comprehensive Cancer Center (JCCC), The Eli & Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA and the University of California Cancer Research Coordinating Committee (CRCC) to ADP. J.A. was funded by the UCLA Tumor Immunology Training Grant Fellowship (T32CA009120-33) and the University of California Regents President’s Postdoctoral Fellowship for these studies.
Footnotes
Author contributions: J.A.A. and G.E.L.: conception and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing, and final approval of manuscript; E.E.E.: collection and/or assembly of data; A.D.P.: conception and design, financial support, data analysis and interpretation, manuscript writing, and final approval of manuscript.
Disclosure of Potential Conflicts of Interest
The authors indicate no potential conflicts of interest.
References
- 1.Ying QL, Wray J, Nichols J, et al. The ground state of embryonic stem cell self-renewal. Nature. 2008;453:519–523. doi: 10.1038/nature06968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nichols J, Smith A. Naive and primed pluripotent states. Cell Stem Cell. 2009;4:487–492. doi: 10.1016/j.stem.2009.05.015. [DOI] [PubMed] [Google Scholar]
- 3.Armstrong L, Hughes O, Yung S, et al. The role of PI3K/AKT, MAPK/ERK And Nfkappabeta Signalling in the maintenance of human embryonic stem cell pluripotency and viability highlighted by transcriptional profiling and functional analysis. Hum Mol Genet. 2006;15:1894–1913. doi: 10.1093/hmg/ddl112. [DOI] [PubMed] [Google Scholar]
- 4.Paling NR, Wheadon H, Bone HK, et al. Regulation of embryonic stem cell self-renewal by phosphoinositide 3-kinase-dependent signaling. J Biol Chem. 2004;279:48063–48070. doi: 10.1074/jbc.M406467200. [DOI] [PubMed] [Google Scholar]
- 5.Pyle AD, Lock LF, Donovan PJ. Neurotrophins mediate human embryonic stem cell survival. Nat Biotechnol. 2006;24:344–350. doi: 10.1038/nbt1189. [DOI] [PubMed] [Google Scholar]
- 6.Watanabe S, Umehara H, Murayama K, et al. Activation of Akt signaling is sufficient to maintain pluripotency in mouse and primate embryonic stem cells. Oncogene. 2006;25:2697–2707. doi: 10.1038/sj.onc.1209307. [DOI] [PubMed] [Google Scholar]
- 7.Engelman JA, Luo J, Cantley LC. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet. 2006;7:606–619. doi: 10.1038/nrg1879. [DOI] [PubMed] [Google Scholar]
- 8.Manning BD, Cantley LC. AKT/PKB signaling: Navigating downstream. Cell. 2007;129:1261–1274. doi: 10.1016/j.cell.2007.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stiles B, Groszer M, Wang S, et al. PTENless means more. Dev Biol. 2004;273:175–184. doi: 10.1016/j.ydbio.2004.06.008. [DOI] [PubMed] [Google Scholar]
- 10.Salmena L, Carracedo A, Pandolfi PP. Tenets of PTEN tumor suppression. Cell. 2008;133:403–414. doi: 10.1016/j.cell.2008.04.013. [DOI] [PubMed] [Google Scholar]
- 11.Sun H, Lesche R, Li DM, et al. PTEN modulates cell cycle progression and cell survival by regulating phosphatidylinositol 3,4,5,-trisphosphate and Akt/protein kinase B signaling pathway. Proc Natl Acad Sci USA. 1999;96:6199–6204. doi: 10.1073/pnas.96.11.6199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.He XC, Yin T, Grindley JC, et al. PTEN-deficient intestinal stem cells initiate intestinal polyposis. Nat Genet. 2007;39:189–198. doi: 10.1038/ng1928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Korkaya H, Paulson A, Charafe-Jauffret E, et al. Regulation of mammary stem/progenitor cells by PTEN/Akt/beta-catenin signaling. PLoS Biol. 2009;7:1–14. doi: 10.1371/journal.pbio.1000121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Groszer M, Erickson R, Scripture-Adams DD, et al. PTEN negatively regulates neural stem cell self-renewal by modulating G0–G1 cell cycle entry. Proc Natl Acad Sci USA. 2006;103:111–116. doi: 10.1073/pnas.0509939103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Welham MJ, Kingham E, Sanchez-Ripoll Y, et al. Controlling embryonic stem cell proliferation and pluripotency: The role of PI3K- and GSK-3-dependent signalling. Biochem Soc Trans. 2011;39:674–678. doi: 10.1042/BST0390674. [DOI] [PubMed] [Google Scholar]
- 16.Kim JS, Kim BS, Kim J, et al. The phosphoinositide-3-kinase/Akt pathway mediates the transient increase in Nanog expression during differentiation of F9 cells. Arch Pharm Res. 2010;33:1117–1125. doi: 10.1007/s12272-010-0719-y. [DOI] [PubMed] [Google Scholar]
- 17.Jeong CH, Cho YY, Kim MO, et al. Phosphorylation of Sox2 cooperates in reprogramming to pluripotent stem cells. Stem Cells. 2010;28:2141–2150. doi: 10.1002/stem.540. [DOI] [PubMed] [Google Scholar]
- 18.Damoiseaux R, Sherman SP, Alva JA, et al. Integrated chemical genomics reveals modifiers of survival in human embryonic stem cells. Stem Cells. 2009;27:533–542. doi: 10.1634/stemcells.2008-0596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ding VM, Ling L, Natarajan S, et al. FGF-2 modulates Wnt signaling in undifferentiated hESC and iPS cells through activated PI3-K/GSK3beta signaling. J Cell Physiol. 2010;225:417–428. doi: 10.1002/jcp.22214. [DOI] [PubMed] [Google Scholar]
- 20.Bendall SC, Stewart MH, Menendez P, et al. IGF and FGF cooperatively establish the regulatory stem cell niche of pluripotent human cells in vitro. Nature. 2007;448:1015–1021. doi: 10.1038/nature06027. [DOI] [PubMed] [Google Scholar]
- 21.Wang X, Lin G, Martins-Taylor K, et al. Inhibition of caspase-mediated anoikis is critical for basic fibroblast growth factor-sustained culture of human pluripotent stem cells. J Biol Chem. 2009;284:34054–34064. doi: 10.1074/jbc.M109.052290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wong RC, Tellis I, Jamshidi P, et al. Anti-apoptotic effect of sphingosine-1-phosphate and platelet-derived growth factor in human embryonic stem cells. Stem Cells Dev. 2007;16:989–1001. doi: 10.1089/scd.2007.0057. [DOI] [PubMed] [Google Scholar]
- 23.Soh BS, Song CM, Vallier L, et al. Pleiotrophin enhances clonal growth and long-term expansion of human embryonic stem cells. Stem Cells. 2007;25:3029–3037. doi: 10.1634/stemcells.2007-0372. [DOI] [PubMed] [Google Scholar]
- 24.McLean AB, D’Amour KA, Jones KL, et al. Activin a efficiently specifies definitive endoderm from human embryonic stem cells only when phosphatidylinositol 3-kinase signaling is suppressed. Stem Cells. 2007;25:29–38. doi: 10.1634/stemcells.2006-0219. [DOI] [PubMed] [Google Scholar]
- 25.Carpenter MK, Inokuma MS, Denham J, et al. Enrichment of neurons and neural precursors from human embryonic stem cells. Exp Neurol. 2001;172:383–397. doi: 10.1006/exnr.2001.7832. [DOI] [PubMed] [Google Scholar]
- 26.Dravid G, Zhu Y, Scholes J, et al. Dysregulated gene expression during hematopoietic differentiation from human embryonic stem cells. Mol Ther. 2011;19:768–781. doi: 10.1038/mt.2010.281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Conway AE, Lindgren A, Galic Z, et al. A self-renewal program controls the expansion of genetically unstable cancer stem cells in pluripotent stem cell-derived tumors. Stem Cells. 2009;27:18–28. doi: 10.1634/stemcells.2008-0529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Storm MP, Bone HK, Beck CG, et al. Regulation of Nanog expression by phosphoinositide 3-kinase-dependent signaling in murine embryonic stem cells. J Biol Chem. 2007;282:6265–6273. doi: 10.1074/jbc.M610906200. [DOI] [PubMed] [Google Scholar]
- 29.Groszer M, Erickson R, Scripture-Adams DD, et al. Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science. 2001;294:2186–2189. doi: 10.1126/science.1065518. [DOI] [PubMed] [Google Scholar]
- 30.Lam H, Patel S, Wong J, et al. Localized decrease of beta-catenin contributes to the differentiation of human embryonic stem cells. Biochem Biophys Res Commun. 2008;372:601–606. doi: 10.1016/j.bbrc.2008.05.116. [DOI] [PubMed] [Google Scholar]
- 31.Sumi T, Tsuneyoshi N, Nakatsuji N, et al. Defining early lineage specification of human embryonic stem cells by the orchestrated balance of canonical Wnt/beta-catenin. Activin/Nodal and Bmp Signaling Development. 2008;135:2969–2979. doi: 10.1242/dev.021121. [DOI] [PubMed] [Google Scholar]
- 32.Di Cristofano A, Pesce B, Cordon-Cardo C, et al. Pten is essential for embryonic development and tumour suppression. Nat Genet. 1998;19:348–355. doi: 10.1038/1235. [DOI] [PubMed] [Google Scholar]
- 33.Hough SR, Laslett AL, Grimmond SB, et al. A continuum of cell states spans pluripotency and lineage commitment in human embryonic stem cells. PLoS ONE. 2009;4:1–10. doi: 10.1371/journal.pone.0007708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pera MF, Tam PP. Extrinsic regulation of pluripotent stem cells. Nature. 2010;465:713–720. doi: 10.1038/nature09228. [DOI] [PubMed] [Google Scholar]
- 35.Laslett AL, Grimmond S, Gardiner B, et al. Transcriptional analysis of early lineage commitment in human embryonic stem cells. BMC Dev Biol. 2007;7:12. doi: 10.1186/1471-213X-7-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sato N, Meijer L, Skaltsounis L, et al. Maintenance of pluripotency in human and mouse embryonic stem cells through activation of Wnt signaling by a pharmacological GSK-3-specific inhibitor. Nat Med. 2004;10:55–63. doi: 10.1038/nm979. [DOI] [PubMed] [Google Scholar]
- 37.Hao J, Li TG, Qi X, et al. WNT/beta-catenin pathway up-regulates Stat3 and converges on LIF to prevent differentiation of mouse embryonic stem cells. Dev Biol. 2006;290:81–91. doi: 10.1016/j.ydbio.2005.11.011. [DOI] [PubMed] [Google Scholar]
- 38.Easley CAt, Ben-Yehudah A, Redinger CJ, et al. mTOR-mediated activation of p70 S6K induces differentiation of pluripotent human embryonic stem cells. Cell Reprogram. 2010;12:263–273. doi: 10.1089/cell.2010.0011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sampath P, Pritchard DK, Pabon L, et al. A hierarchical network controls protein translation during murine embryonic stem cell self-renewal and differentiation. Cell Stem Cell. 2008;2:448–460. doi: 10.1016/j.stem.2008.03.013. [DOI] [PubMed] [Google Scholar]
- 40.Zhou J, Su P, Wang L, et al. mTOR supports long-term self-renewal and suppresses mesoderm and endoderm activities of human embryonic stem cells. Proc Natl Acad Sci USA. 2009;106:7840–7845. doi: 10.1073/pnas.0901854106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Di Vizio D, Cito L, Boccia A, et al. Loss of the tumor suppressor gene PTEN marks the transition from intratubular germ cell neoplasias (ITGCN) to invasive germ cell tumors. Oncogene. 2005;24:1882–1894. doi: 10.1038/sj.onc.1208368. [DOI] [PubMed] [Google Scholar]
- 42.Darr H, Mayshar Y, Benvenisty N. Overexpression of NANOG in human ES cells enables feeder-free growth while inducing primitive ectoderm features. Development. 2006;133:1193–1201. doi: 10.1242/dev.02286. [DOI] [PubMed] [Google Scholar]
- 43.Lindgren AG, Natsuhara K, Tian E, et al. Loss of Pten causes tumor initiation following differentiation of murine pluripotent stem cells due to failed repression of Nanog. PLoS ONE. 2011;6:1–10. doi: 10.1371/journal.pone.0016478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang X, Huang CT, Chen J, et al. Pax6 is a human neuroectoderm cell fate determinant. Cell Stem Cell. 2010;7:90–100. doi: 10.1016/j.stem.2010.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vierbuchen T, Ostermeier A, Pang ZP, et al. Direct conversion of fibroblasts to functional neurons by defined factors. Nature. 2010;463:1035–1041. doi: 10.1038/nature08797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Buecker C, Geijsen N. Different flavors of pluripotency, molecular mechanisms, and practical implications. Cell Stem Cell. 2010;7:559–564. doi: 10.1016/j.stem.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 47.Hanna J, Cheng AW, Saha K, et al. Human embryonic stem cells with biological and epigenetic characteristics similar to those of mouse ESCs. Proc Natl Acad Sci USA. 2010;107:9222–9227. doi: 10.1073/pnas.1004584107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chen X, Vega VB, Ng HH. Transcriptional regulatory networks in embryonic stem cells. Cold Spring Harb Symp Quant Biol. 2008;73:203–209. doi: 10.1101/sqb.2008.73.026. [DOI] [PubMed] [Google Scholar]
- 49.Chen X, Xu H, Yuan P, et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell. 2008;133:1106–1117. doi: 10.1016/j.cell.2008.04.043. [DOI] [PubMed] [Google Scholar]
- 50.Tam WL, Lim CY, Han J, et al. T-cell factor 3 regulates embryonic stem cell pluripotency and self-renewal by the transcriptional control of multiple lineage pathways. Stem Cells. 2008;26:2019–2031. doi: 10.1634/stemcells.2007-1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu Q, Chen X, Zhang J, et al. Sall4 interacts with Nanog and cooccupies Nanog genomic sites in embryonic stem cells. J Biol Chem. 2006;281:24090–24094. doi: 10.1074/jbc.C600122200. [DOI] [PubMed] [Google Scholar]
- 52.Zhang J, Tam WL, Tong GQ, et al. Sall4 modulates embryonic stem cell pluripotency and early embryonic development by the transcriptional regulation of Pou5f1. Nat Cell Biol. 2006;8:1114–1123. doi: 10.1038/ncb1481. [DOI] [PubMed] [Google Scholar]
- 53.Young RA. Control of the embryonic stem cell state. Cell. 2011;144:940–954. doi: 10.1016/j.cell.2011.01.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.