Abstract
In plants, the cambium and procambium are meristems from which vascular tissue is derived. In contrast to most plant cells, stem cells within these tissues are thin and extremely long. They are particularly unusual as they divide down their long axis in a highly ordered manner, parallel to the tangential axis of the stem. CLAVATA3-LIKE/ESR-RELATED 41 (CLE41) and PHLOEM INTERCALATED WITH XYLEM (PXY) are a multifunctional ligand-receptor pair that regulate vascular cell division, vascular organisation and xylem differentiation in vascular tissue. A transcription factor gene, WUSCHEL HOMEOBOX RELATED 4 (WOX4) has been shown to act downstream of PXY. Here we show that WOX4 acts redundantly with WOX14 in the regulation of vascular cell division, but that these genes have no function in regulating vascular organisation. Furthermore, we identify an interaction between PXY and the receptor kinase ERECTA (ER) that affects the organisation of the vascular tissue but not the rate of cell division, suggesting that cell division and vascular organisation are genetically separable. Our observations also support a model whereby tissue organisation and cell division are integrated via PXY and ER signalling, which together coordinate development of different cell types that are essential for normal stem formation.
Keywords: Arabidopsis, Cell division, Phloem, Procambium, Signalling, Vascular
INTRODUCTION
Differential growth is the basis of all plant morphology and is determined by cell proliferation, cell expansion and the orientation of cell division. In the cambium and procambium, which are meristems from which vascular tissue is derived, cells divide down their long axis in a highly oriented manner, perpendicular to the radial axis of the stem. These divisions result in highly distinctive cell files that are characteristic of the xylem in most dicots. The cell files that are generated are also essential for maintaining the spatial separation of xylem and phloem (Caño-Delgado et al., 2010). These properties make vascular tissue an ideal system for examining factors that regulate cell division and its effect on tissue organisation during differential growth.
CLE41 (CLAVATA3/ESR LIKE 41) and PXY (PHLOEM INTERCALATED WITH XYLEM) are a ligand-receptor pair, constituting a multifunctional pathway that regulates vascular cell division, vascular bundle organisation and xylem differentiation (Etchells and Turner, 2010; Fisher and Turner, 2007; Hirakawa et al., 2008). Vascular bundle organisation defects in pxy mutants are characterised by intercalation of xylem and phloem, which is in contrast to the wild type where they are spatially separated. Defects in organisation are apparent even in the early stages of pxy vascular tissue differentiation (Fisher and Turner, 2007).
PXY ligand, CLE41, is expressed in the phloem (Etchells and Turner, 2010; Hirakawa et al., 2008) and therefore ideally placed to confer positional information to PXY that is expressed in the adjacent vascular procambium (Fisher and Turner, 2007). Ectopic overexpression of CLE41 using constitutive or xylem-specific promoters results in both increased vascular cell division, and organisational defects in the vascular tissue. This is consistent with a role for localised CLE41 expression providing a positional signal to PXY that is required for correct orientation of the division plane in vascular stem cells (supplementary material Fig. S1A,B) (Etchells and Turner, 2010).
Although these experiments are all consistent with a role for PXY and CLE41 in regulating the orientation of vascular cell division, an alternative model for the intercalation of xylem and phloem in pxy mutants (Hirakawa et al., 2010) followed the identification of a downstream target of CLE41/PXY signalling as WOX4. WOX4 had previously been shown to promote vascular cell division (Ji et al., 2010), and its expression was found to be rapidly increased by exogenous application of TDIF (Hirakawa et al., 2010), the active peptide derived from CLE41, CLE42 and CLE44 (Ito et al., 2006). wox4 pxy mutants were reported with a phenotype stronger than that of pxy single mutants and consequently, an ‘enhanced cavity’ model was put forward whereby adjacent xylem vessels and phloem were proposed to be the result of differentiation of cells in the vascular meristem, resulting in loss of spatial separation of xylem and phloem (Hirakawa et al., 2010) (supplementary material Fig. S1C). Because TDIF has been shown to repress xylem differentiation in cell culture, and plants overexpressing both CLE41 and PXY result in more undifferentiated vascular cells (Etchells and Turner, 2010; Hirakawa et al., 2008; Ito et al., 2006), it was suggested that the ‘cavity’ is the result of the meristem differentiating into xylem and that vascular cell proliferation subsequently reinitiates via a compensatory mechanism between the phloem and cortex (Hirakawa et al., 2010). A compensatory mechanism has recently been described that involves ethylene maintaining vascular cell numbers by promoting vascular cell division in the absence of functional pxy or wox4 (Etchells et al., 2012).
Although WOX4 is reported to be downstream of pxy, no reduction in WOX4 expression has been reported in pxy mutant seedling hypocotyls, demonstrating that the pxy phenotype is not a result of changes in WOX4 expression (Hirakawa et al., 2010). Alternative transcriptional targets of PXY/CLE41 signalling must therefore exist.
ERECTA (ER) encodes a RLK involved in mediating cell-cell communication in several plant developmental contexts (van Zanten et al., 2009), including repression of xylem expansion in hypocotyls (Ragni et al., 2011). The importance of the role of ER in cell-cell communication is perhaps most clearly evident in stomatal spacing, which ER controls together with the related RLK proteins ERECTA-LIKE1 (ERL1) and ERL2 (Shpak et al., 2005), collectively referred to as the ER family (ERf), and with members of the EPIDERMAL PATTERNING FACTOR LIKE (EPFL) family that encode ligands for ERf receptors (Hara et al., 2007; Hara et al., 2009; Hunt and Gray, 2009; Lee et al., 2012). In the cortex, ERf receptors also have a role in promoting cell division (Shpak et al., 2004), controlled by CHALLAH (CHAL), CHALLAH-LIKE1 (CLL1) and CLL2 ligands (EPFL6, EPFL5 and EPFL4, respectively). CHAL and CLL2 are expressed in the endodermis and signal to ER, which is present in the phloem, promoting growth of the inflorescence stem (Abrash et al., 2011; Uchida et al., 2012).
Here, we demonstrate that WOX14 acts redundantly with WOX4 in promoting vascular cell division downstream of PXY signalling. wox4 wox14 double mutants demonstrate similar reductions in vascular cell division to those observed in pxy wox4 lines, but do not have vascular organisation defects. By contrast, er dramatically enhances the effects of pxy on organisation of inflorescence stem vascular bundles, resulting in intercalation of xylem and phloem, but er has no effect on vascular cell number. Our results suggest that vascular organisation can be disrupted in the absence of reductions in vascular cell division, and disrupted rates of cell division do not lead to disrupted vascular organisation.
MATERIALS AND METHODS
Generation of plant stocks
wox14-1 and er-124 T-DNA insertion mutants were identified using the TAIR database (Swarbreck et al., 2008) and confirmed by PCR with primers described in supplementary material Table S1. pxy-1, pxy-3, pxy-5, pxy-3 pxl1-2 pxl2-1, wox4-1, er-105 erl1-2 erl2-1, IRX3::CLE41 and 35S::CLE41 lines have been described previously (Etchells and Turner, 2010; Fisher and Turner, 2007; Shpak et al., 2004). IRX3::CLE41 wox4-1, pxy-5 er-124, pxy-3 wox4-1, wox14-1 wox4-1, pxy-3 wox14-1 and pxy-3 wox4-1 wox14-1 were identified using standard PCR-based screening of segregating F2 populations using primers described in supplementary material Table S1. For WOX14::GUS lines, 2.5 kb of the WOX14 upstream region was PCR amplified using primers described in supplementary material Table S1, and cloned into pKGWFS7 via pENTR-D-TOPO. Transgenic plants were generated using Agrobacterium infiltration (Clough and Bent, 1998). Images shown are representative of eight independent transgenic lines.
TDIF/P9A synthetic peptide treatments
Seeds were plated on 0.5×MS, 1% Agar and stratified for 4 days at 4°C prior to transfer to a Sanyo MLR-351H plant growth chamber set at 23°C with constant light. After 5 days, seedlings were transferred to 0.5×MS liquid media supplemented with either 5 mM TDIF (His-Glu-Val-Hyp-Ser-Gly-Hyp-Asn-Pro-Ile-Ser-Asn) or negative control P9A (His-Glu-Val-Hyp-Ser-Gly-Hyp-Asn-Ala-Ile-Ser-Asn; Bachem, Switzerland) and maintained on a rocking platform for either 1 or 16 hours. Following treatment, samples were snap frozen and RNA was extracted using Trizol (Life Technologies, USA). RNA was DNase treated prior to cDNA synthesis using BioScript reverse transcriptase (Bioline, UK).
Gene expression analysis
qRT-PCR primary inflorescence stem expression analysis, was performed on stems from 5-week old plants (∼10 cm), stripped of side branches and cauline leaves and divided into three equal lengths. In all qRT-PCR analysis, cDNA was prepared as described above. qRT-PCR was carried out using SensiMix SYBR Hi-ROX reaction mix (Bioline), primers described in supplementary material Table S1, and an ABI Prism 7000 machine (Life Technologies) with the standard SYBR green detection programme. Melting curves were analysed to ensure single products were formed. Relative expression was determined using a version of the comparative threshold cycle (Ct) method using average amplification efficiency for each primer pair, determined using LinReg (Hårdstedt et al., 2005). Samples were normalised to 18S rRNA (not shown) and ACT2 (shown). Results were similar independent of the control used. Samples were measured in technical triplicates on biological triplicates.
For determination of spatial gene expression patterns in WOX14::GUS lines, stain was prepared as described previously (Young et al., 2010). Plant tissue was stained overnight and destained in 70% ethanol. Methods for in situ hybridization using digoxigenin-labelled probes were carried out as described by Etchells et al. (Etchells et al., 2010). WOX4 antisense probe was generated from a pT48 template (Suer et al., 2011). WOX14 sense and antisense probes were made by cloning a WOX14 PCR fragment, amplified using primers shown in supplementary material Table S1 into pGEM-T easy (Promega, USA), which was subsequently used in PCR with M13 primers to generate a probe template.
Histology
For thin sections, hypocotyls or tissue 1 cm from the base of inflorescence stems was fixed in FAA and dehydrated through an ethanol series prior to infiltration and subsequent embedding with JB4 embedding media. Sections (4 μm) were stained with 0.05% aqueous Toluidine Blue. Callose sieve plates marking the phloem were observed by staining hand sections with 500 mM Aniline Blue in 100 mM phosphate buffer (pH 7.2) and visualising tissue on a Leica DMR microscope using the UV lamp. Vascular tissue was considered to be ordered in vascular bundles when xylem and phloem could be separated by a simple arced line and in hypocotyls if xylem could be incorporated in an elliptical shape that excluded the phloem.
RESULTS
Evidence for factors downstream of PXY signalling in addition to WOX4
WOX4, a downstream target of PXY signalling, is upregulated by application of CLE ligand in seedling hypocotyls; however, no reduction in WOX4 expression was observed in pxy mutants (Hirakawa et al., 2010). Consequently, pxy phenotypes cannot solely be attributable to changes in WOX4 expression and we examined this possibility by crossing IRX3::CLE41 and wox4, and comparing IRX3::CLE41 wox4 with parental lines and wild-type counterparts. We reasoned that if other factors do act redundantly with WOX4 then wox4 mutants should only partially suppress a CLE41 overexpression phenotype.
wox4 mutants have been described with gross morphology that is similar to wild type but have been reported to contain fewer cells in vasculature undergoing secondary growth, either directly above the rosette leaves in inflorescence stems (Suer et al., 2011) or in seedling hypocotyls (Hirakawa et al., 2010). We measured vascular cell division in areas of primary growth in 6-week-old plants 1 cm above the rosette, avoiding the secondary growth region directly above rosette leaves. At this developmental stage, wox4 mutants demonstrated vascular cell numbers that were not significantly different from wild-type counterparts (Fig. 1A). IRX3::CLE41 plants are characterised with large vascular bundles containing many undifferentiated cells and also have phloem intercalated with xylem (Fig. 1A; supplementary material Fig. S2A,B) (Etchells and Turner, 2010). Analysis of vascular tissue in the stem revealed a significant modification of the IRX3::CLE41 phenotype by wox4, as wox4 IRX3::CLE41 vascular tissue demonstrated a 28.6% reduction in cell number when compared with IRX3::CLE41. However, wox4 did not completely abolish the increase in cell division in IRX3::CLE41 vascular tissue. wox4 IRX3::CLE41 lines still had a further 28.6% more cells than wild type and 53.3% more than wox4 single mutants (Fig. 1A), clearly demonstrating that factors exist that act redundantly with WOX4, downstream of PXY signalling.
Fig. 1.
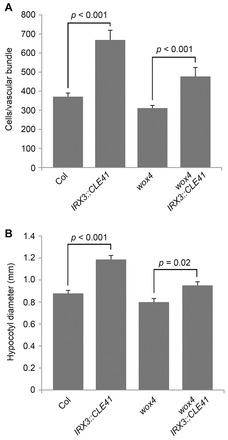
wox4 only partially suppresses the CLE41 overexpression phenotypes. (A) Mean number of cells per vascular bundle 1 cm from the base of inflorescence stems of 6-week-old wild type (Col). (B) Hypocotyl diameter of 6-week-old wild type, IRX3::CLE41, wox4 and wox4 IRX3::CLE41 plants. P values were determined using ANOVA with an LSD post-hoc test. Error bars indicate s.e.m.
We tested whether evidence for WOX4-independent PXY targets was also present in hypocotyls, where vascular tissue undergoes secondary growth. wox4 IRX3::CLE41 hypocotyls were 19.8% smaller than those of IRX3::CLE41 lines, but as was the case in vascular bundles, IRX3::CLE41 wox4 were larger than those observed in wox4 single mutants (Fig. 1B; Fig. 2), demonstrating that WOX4 acts with additional factors. Our previous analysis suggested that increases in vascular cell division associated with CLE41 overexpression is entirely dependent on the presence of a PXY receptor as pxy 35S::CLE41 lines are identical to pxy single mutants in Arabidopsis hypocotyls (Etchells and Turner, 2010). As wox4 acts as only a partial suppressor of IRX3::CLE41, we hypothesised that WOX4 may act with other factors in mediating the promotion of cell division in the procambium in response to and downstream of PXY signalling.
Fig. 2.
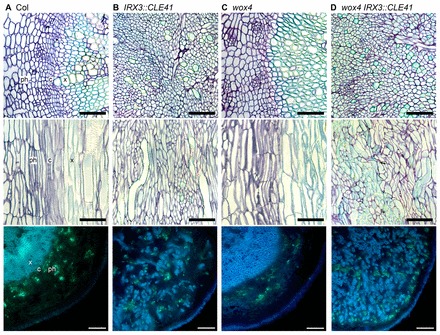
Hypocotyl phenotype of wox4 IRX3::CLE41 lines. (A-D) Vascular tissue from 6-week-old hypocotyls. Upper and middle panels show Toluidine Blue-stained thin sections. Upper section is transverse, middle section is longitudinal. Lower panels are Analine Blue-stained hand sections viewed under a UV light. (A) Col wild-type vascular tissue with phloem (ph), cambium (c) and xylem (x) marked. (B) IRX3::CLE41 plants with intercalated xylem and phloem. (C) wox4 plants are organised similarly to wild type. (D) wox4 IRX3::CLE41 lines, organised similarly to IRX3::CLE41 but with smaller spaces between differentiated xylem and phloem. Scale bars: 50 μm (top and middle); 100 μm (bottom).
Both WOX4 and WOX14 act downstream of PXY signalling
Our genetic analysis (Figs 1, 2; supplementary material Fig. S2) suggested that WOX4 is only partially responsible for promoting cell division downstream of the PXY signalling so must act redundantly with other factor(s). The CLE-RLK-WOX interaction in regulation of stem cell niches has been widely reported (Brand et al., 2000; Hirakawa et al., 2010; Sarkar et al., 2007; Schoof et al., 2000) so we analysed microarray data comparing expression in pxy-3 mutants and wild type (experiment E-MEXP-2639, http://www.ebi.ac.uk/arrayexpress) (Etchells et al., 2012), for changes in WOX expression. WOX14 demonstrated a 1.7-fold reduction in expression in pxy suggesting that it may be regulated by PXY signalling. WOX14 expression was compared with that of WOX4, a previously identified target of PXY signalling (Hirakawa et al., 2010) in 5-day-old wild-type seedlings subjected to a 1- or 16-hour exposure to exogenous application of TDIF, the peptide derived from CLE41 or negative control peptide P9A (Ito et al., 2006). Consistent with the observations of Hirakawa et al. (Hirakawa et al., 2010), WOX4 expression was increased following either a 1- or 16-hour treatment (Fig. 3). Although WOX14 expression appeared increased at 1 hour, differences in expression proved not significant (Fig. 3A); however, at 16 hours a small (1.6-fold) but significant increase in expression was observed (Fig. 3B) that occurred in a PXY-dependent manner, demonstrating that WOX14 acts downstream of PXY signalling. Increases in WOX14 expression were not dependent on functional WOX4, as a clear WOX14 response to TDIF was observed in wox4 mutant lines (Fig. 3), demonstrating that WOX14 is not a downstream target of WOX4.
Fig. 3.
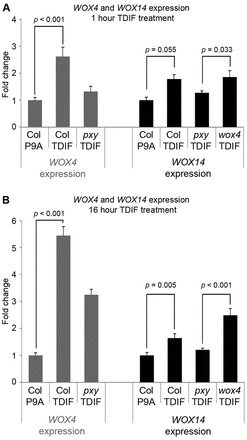
Comparison of WOX4 and WOX14 expression in response to a TDIF stimulus. (A) qRT-PCR showing WOX4 (grey) and WOX14 (black) expression changes in 5-day-old seedlings exposed to a 5 μM TDIF or P9A mock treatment for 1 hour, normalised to ACT2. (B) A similar experiment to A, except that plants were treated for 16 hours with TDIF or P9A. Samples were measured in technical triplicates on biological triplicates. P values were determined using ANOVA with an LSD post-hoc test. Error bars indicate s.e.m.
Inflorescence stems represent a developmental series with most recently derived vascular tissue present in apical regions of the stem and the oldest tissue in basal regions. We compared WOX4 and WOX14 expression in apical, central and basal regions of inflorescence stems from 5-week-old plants using qRT-PCR and found that the dynamics of WOX14 expression differed from that of WOX4. WOX4 was expressed at a relatively consistent level along the length of the inflorescence stem (Fig. 4A); however, WOX14 expression was low at the top but expression increased along the apical-basal axis such that expression was 20-fold higher at the base than at the top (Fig. 4B).
Fig. 4.
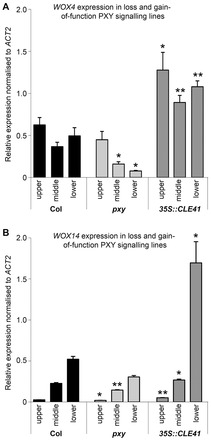
WOX4 and WOX14 expression in inflorescence stems. (A) qRT-PCR showing WOX4 inflorescence expression in 5-week-old Col, pxy or 35S::CLE41 plants normalised to ACT2. (B) WOX14 inflorescence expression in 5-week-old plants normalised to ACT2. P values were determined using ANOVA with an LSD post-hoc test; *P<0.05; **P≤0.01. Error bars indicate s.e.m.
WOX4 and WOX14 expression levels were also compared in pxy and 35S::CLE41 lines. WOX4 expression was maintained at the level of wild-type expression in apical regions of pxy inflorescence stems; however, in central and basal regions WOX4 expression was significantly reduced (Fig. 4A). Consequently, we have demonstrated that PXY signalling is required for normal levels of WOX4 expression. WOX14 expression was also dependent upon PXY as mRNA levels were similarly reduced in pxy mutant stems (Fig. 4B). In the apical third of the stem, WOX14 expression was reduced in pxy by a small (1.3-fold) but significant amount, and this reduction was more apparent in the middle of the stem (Fig. 4B). The trend of reduced WOX14 expression appeared similar in the stem base of pxy mutants, but these differences proved not to be statistically significant.
In 35S::CLE41 lines, WOX4 expression was increased approximately twofold at all stages of stem development relative to wild type (Fig. 4A). WOX14 expression levels were also elevated in these lines, but in this case the largest increase in expression was observed at the base of the stem (Fig. 4B). Wild-type expression levels of WOX4 and WOX14 are therefore dependent on the presence of a PXY receptor kinase and promoted by PXY ligand, demonstrating that both of these WOX genes act downstream of PXY signalling.
WOX14 is expressed predominantly in vascular tissue
We determined the spatial expression pattern of WOX14 by generating eight independent WOX14::GUS lines. A 2.5 kb region of WOX14 upstream regulatory sequence was cloned behind a GUS-GFP cassette in a binary vector and subsequently transformed into Arabidopsis. When subjected to staining, in all cases 14-day-old seedlings demonstrated no expression in leaves, cotyledons or in the shoot apex. Staining was observed where the leaves met the stem (supplementary material Fig. S3A, arrows, upper panel). Strong staining was observed in the vascular cylinder of the hypocotyl and root, and was also apparent in the root elongation zone but not the root tip (supplementary material Fig. S3A). In plants 5 weeks post-germination (inflorescence stem at 10 cm), GUS staining did not occur at the very top of the stem but was present 2 cm below the apex (supplementary material Fig. S3B) where staining was predominantly in vascular strands but also present, albeit at a much lower level, between the vascular strands (arrowhead). At the base of inflorescence stems, 1 cm above the rosette, GUS staining was more intense in vascular strands but, unlike in apical parts of the inflorescence stem, was absent between vascular strands (supplementary material Fig. S3B). In rosette leaves of 5-week-old plants, GUS stain was not observed in expanding leaves (not shown) but was present in midveins (supplementary material Fig. S3B, arrowheads) but no other leaf veins of fully expanded leaves (supplementary material Fig. S3B; arrow in lower panel). These results suggest that WOX14 upstream regulatory sequences do not restrict expression in initiating vascular tissue but do promote WOX14 expression in established vasculature.
To better understand cell-type specific WOX14 expression in stained GUS lines, transverse inflorescence stem sections were taken to determine spatial expression within the vascular tissue. At 2 cm below the shoot apex, GUS staining was expressed broadly in vascular tissue, present in xylem, phloem and procambium; however, in vascular tissue at the base of stems 1 cm above the rosette, staining was restricted to the procambium and adjacent areas of xylem and phloem (supplementary material Fig. S3C). To confirm that the GUS pattern represented a true reflection of WOX14 expression, RNA in situ hybridization was performed on tissue from the same stage in development. In these experiments, WOX14 antisense probes broadly marked the vascular bundles in apical sections, but demonstrated predominantly procambium expression in basal sections (Fig. 5A), in common with the GUS lines (supplementary material Fig. S3C). By contrast, signal was not observed above background levels using a WOX14 sense control (Fig. 5C). To compare WOX4 and WOX14 expression, we also carried out in situ hybridization using an antisense probe previously described for WOX4 (Suer et al., 2011). WOX4 expression was confined to the procambium in both apical and basal sections (Fig. 5B). Consequently, expression of both of these genes overlaps in the procambium.
Fig. 5.
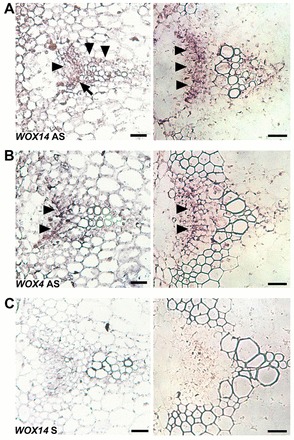
WOX14 expression in inflorescence stem vascular bundles. (A-C) Expression in inflorescence stem vascular bundles from 5-week-old plants (∼10 cm inflorescence); panels on the left show sections 2 cm below the shoot apex; panels on the right show sections 1 cm above the rosette. (A,B) In situ hybridization using antisense WOX14 (A) and WOX4 (C) probes. In apical sections, WOX14 expression was present throughout the vascular bundle (A, LHS; arrowheads) but prominent in the procambium (arrow). WOX4 expression was restricted to the procambium (B, LHS; arrowheads). In basal sections, WOX14 expression was particularly prominent in the procambium (A, RHS; arrowheads). WOX4 expression was procambium specific (B, RHS; arrowheads). (C) WOX14 sense controls did not label above background levels. Scale bars: 25 μm.
WOX14 acts redundantly with WOX4 in regulation of vascular cell division
Expression analysis suggested that WOX14 acts downstream of the PXY receptor (Figs 3, 4, 5). Consequently, we identified a wox14 T-DNA insertion mutant (GabiKat_469C11), renamed it wox14-1 and used it to test for a role for WOX14 in Arabidopsis vascular tissue. These plants exhibited no obvious alteration in plant growth or morphology (not shown), and no alterations in vascular organisation or cell number were observed (Figs 6, 7). We reasoned that WOX4 and WOX14 may act redundantly, so generated wox4 wox14 double mutants. wox4 wox14 gross morphology was also indistinguishable from that of wild-type plants; however, when cell number was analysed in vascular bundles, fewer cells were present in wox4 wox14 than wild type or single mutants (Figs 6, 7), demonstrating that WOX4 and WOX14 act redundantly in promoting vascular cell division.
Fig. 6.
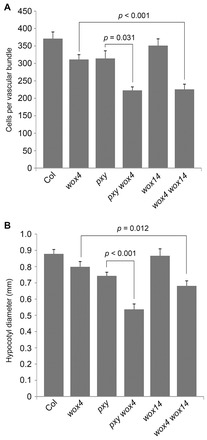
The wox4 phenotype is enhanced by wox14. (A) Mean number of cells per vascular bundle 1 cm from the base of inflorescence stems of 8-week-old wild-type (Col), wox4, pxy, wox4 pxy, wox14 and wox4 wox14 plants. (B) Hypocotyl diameter of 8-week-old plants. P values were determined using ANOVA with an LSD post-hoc test. Error bars are s.e.m.
Fig. 7.
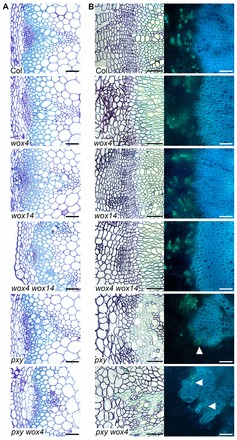
wox4 wox14 double mutants have no vascular organisation defects. (A) Transverse Toluidine Blue-stained sections of vascular tissue from 8-week-old inflorescence stems. (B) Transverse hypocotyl sections. LHS are Toluidine Blue stained; RHS are Analine Blue stained. Arrowheads indicate intercalated xylem and phloem in pxy and pxy wox4. Scales bars: 50 μm.
Strikingly, wox4 wox14 double mutant vascular bundles were smaller than those observed in pxy single mutants, and were a similar size to those observed in pxy wox4 lines (Fig. 6A). In contrast to pxy and pxy wox4 lines, which exhibit characteristic defects in vascular organisation, organisation of wox4 wox14 vascular bundles was similar to the wild type (Fig. 7). A similar situation was observed in hypocotyls, where cambium activity was assessed by measuring hypocotyl diameter. wox4 wox14 mutants demonstrated a similar diameter to that observed in pxy (Fig. 6B), but in contrast to pxy (Fig. 7B; arrowheads) no defects in vascular organisation were observed.
We further characterised the relationship between pxy and wox14 by analysing pxy wox14 and pxy wox4 wox14 mutant combinations. pxy wox14 lines were identical to pxy single mutants for all parameters tested, i.e. for vascular proliferation measures (cell number in inflorescence stem vascular bundles and hypocotyl diameter; supplementary material Figs S4, S5) and for vascular organisation phenotypes (Analine Blue stained sections; supplementary material Fig. S5). This result is in contrast to that observed between pxy and wox4 as pxy wox4 lines demonstrate an enhancement (Hirakawa et al., 2010). Similarly, pxy wox4 wox14 triple mutants were identical to pxy wox4 lines (supplementary material Figs S4, S5). These observations are consistent with WOX14 acting in an epistatic manner to PXY, and suggest that the strong phenotype observed in pxy wox4 may in part be due to the loss of control of WOX14 in pxy mutants.
pxy interacts with er during vascular development
Our results with wox4 and wox14 suggested that the vascular organisation defect in pxy mutants may be genetically separable from proliferation defects. In our original description of pxy, the pxy-1 allele in Ler background demonstrated a stronger organisation defect than mutants in the Col background (Fisher and Turner, 2007) (supplementary material Fig. S6). pxy-1 has a premature stop codon in the kinase domain that is presumably essential for transmitting the CLE41/42 signal through PXY, but a variety of Col pxy alleles, including those that entirely lack the kinase domain do not exhibit similarly strong phenotypes (Fisher and Turner, 2007).
We explored the reasons behind the difference in the strength of Ler and Col pxy alleles to investigate further whether vascular organisation and proliferation are genetically separable. One prominent difference between Ler and Col ecotypes is the presence of a mutant er gene in Ler. ER acts redundantly with closely related RLK proteins ERL1 and ERL2 in promotion of cell division in Arabidopsis (Shpak et al., 2004), including in the vascular tissue (Fig. 8), but is also involved in intertissue communication between endodermis and the phloem, where both ER (Uchida et al., 2012) and CLE41 (Etchells and Turner, 2010) are expressed. We hypothesised that the severity of the pxy-1 Ler allele could be due to a genetic interaction between pxy and er. To test this, we isolated a novel er allele in the Col background which harboured a T-DNA insertion in the LLR region of the gene (GabiKat_364C05) (Rosso et al., 2003), renamed er-124 following the allelic sequence described (Lease et al., 2001). er-124 was indistinguishable from other er alleles - siliques were short, blunt and wider than those of wild type, and inflorescence stems were shorter (supplementary material Figs S7, S8). er single mutants demonstrated no inflorescence stem vascular cell division defects (Fig. 8A) or secondary growth defects, as er hypocotyls were the same size as wild type (Fig. 8B). We nevertheless examined whether ER and PXY interacted by generating pxy er double mutants. These plants accumulated anthocyanin in cotyledons (supplementary material Fig. S7D) in a manner reminiscent of the pxy-1 Ler allele (Fisher and Turner, 2007). pxy er mutants demonstrated a height defect but this was an additive phenotype as average plant height was a combination of the reductions observed in the single mutants (supplementary material Figs S7, S8). Similarly, no interaction was found between pxy and er in silique length (pxy mutants have the same pod length as wild type, er mutants are shorter and er pxy double mutant pods are the same as those of er single mutants; supplementary material Fig. S8). Consequently, pxy is not a general enhancer of er.
Fig. 8.
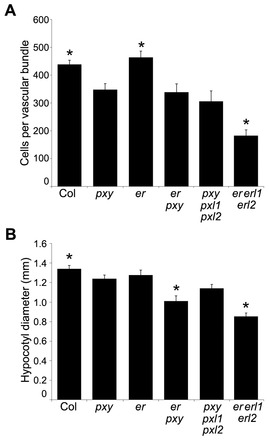
pxy er mutants demonstrate that vascular proliferation and organisation are genetically separable. (A) Mean number of cells per vascular bundle 1 cm from the base of inflorescence stems of 8-week-old wild-type, pxy, er, er pxy, pxy pxl1 pxl2 and er erl1 erl2 plants. (B) Hypocotyl diameter of 8-week-old wild-type, pxy, er, er pxy, pxy pxl1 pxl2 and er erl1 erl2 plants. P values were determined using ANOVA with an LSD post-hoc test. *P<0.05, significantly different from pxy. Error bars indicate s.e.m.
We assayed the average number of cells in inflorescence stem vascular bundles and found that pxy er double mutants had similar numbers of vascular cells to pxy single mutants (Fig. 8) and, therefore, er does not enhance the pxy vascular cell number defect in inflorescence stems. However, vascular organisation has been quantitatively characterised by determining the width:length (tangential:radial) ratio of vascular bundles as a measure of changes in organisation in vascular bundles (Fisher and Turner, 2007). We applied this measure to examine pxy wox4 mutants that have been reported to have more severe vascular bundle disruption than pxy single mutants. Although pxy wox4 did appear to show a small enhancement of the pxy phenotype (Fig. 9A), much more striking changes in the shape of vascular bundles were observed in pxy er double mutants, and this was much greater than that observed for either of the corresponding single mutants (Fig. 9B; Fig. 10A). The large increase in the tangential:radial ratio in pxy er (Fig. 9B) was similar to that of the original pxy-1 allele (Fisher and Turner, 2007). Consequently, pxy er double mutants have the same number of cells as pxy single mutants, but the shape of the vascular bundle is altered. We further investigated vascular organisation in pxy er lines by observing Analine Blue-stained hand sections and found that xylem and phloem were clearly intercalated in pxy er to a greater extent than observed in pxy single mutants (Fig. 10A-D).
Fig. 9.
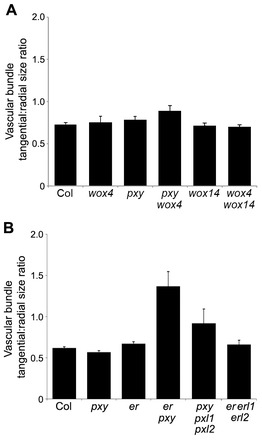
The tangential:radial ratio of inflorescence stem vascular bundles. (A) Wild-type (Col), wox4, pxy, wox4 pxy, wox14 and wox4 wox14 plants. (B) Wild-type, pxy, er, er pxy, pxy pxl1 pxl2 and er erl1 erl2 plants. Error bars indicate s.e.m.
Fig. 10.
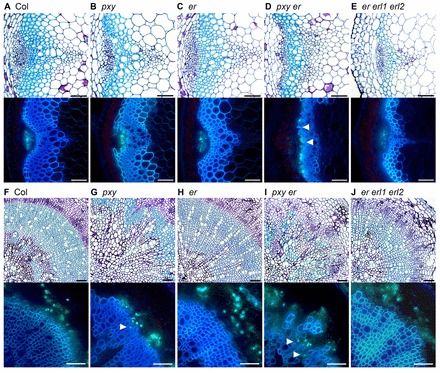
Xylem and phloem are intercalated in pxy er mutants. (A-E) Toluidine Blue-stained thin sections (upper panels); Analine Blue-stained hand sections (lower panels) of Col (A), pxy (B), er (C), pxy er (D) and er erl1 erl2 (E) 8-week-old inflorescence stem vascular tissue. Vascular organisation is perturbed in pxy and severely perturbed in pxy er lines (arrowheads show adjacent xylem and phloem, which are spatially separated in wild type). (F-J) Toluidine Blue (upper) and Analine Blue (lower) stained sections of Col (F), pxy (G), er (H), pxy er (I) and er erl1 erl2 (J) hypocotyl vascular tissue. Arrowheads indicate intercalated xylem and phloem. Scale bars: 50 μm.
Finally, we analysed the pxy er interaction during secondary growth in hypocotyls. In contrast to our observations in the inflorescence stem, pxy er lines has fewer vascular cell divisions than those of pxy and er single mutants, as determined by hypocotyl diameter measurements (Fig. 8B), suggesting that ER and PXY may interact to promote cell division during secondary growth; however, interpretation of this phenotype is not straightforward as ER is known to repress xylem expansion in this context (Ragni et al., 2011).
ER expression overlaps with that of CLE41 (Etchells and Turner, 2010; Uchida et al., 2012) so we tested whether er mutants displayed differences in CLE41, CLE42 or CLE44 expression; however, none was observed (supplementary material Fig. S9). Thin sections were analysed for morphological differences between er and wild type in the phloem, but no clear differences were apparent (supplementary material Fig. S10). Similarly, vasculature of er erl1 erl2 triple mutants was analysed and these were found to retain wild-type organisation (Figs 9, 10), despite large reductions in vascular cell number (Fig. 8).
DISCUSSION
WOX4 acts as a transcriptional target for PXY signalling controlling the rate of vascular cell division; however, vascular proliferation caused by overexpression of the PXY ligand in IRX3::CLE41 lines was only partially abolished in wox4 IRX3::CLE41 (Figs 1, 2; supplementary material Fig. S2), suggesting that other factors must also act downstream of PXY to promote vascular cell divisions. PXY could act redundantly with other RLK proteins such that WOX4 levels are partially maintained in pxy owing to CLE41 acting through redundant receptors and this model would be consistent with transmission of the CLV3 signal in the shoot apical meristem, which acts through the CLV1 receptor (Clark et al., 1997; Fletcher et al., 1999; Ogawa et al., 2008), RECEPTOR-LIKE PROTEIN KINASE2 (Kinoshita et al., 2010) and CLV2-CORYNE receptor heterodimers (Jeong et al., 1999; Müller et al., 2008; Zhu et al., 2010). There is some support for this hypothesis, as levels of WOX4 expression were partially increased upon a 16-hour TDIF induction in pxy mutants (Fig. 3). However, although there is evidence of enhancement of the organisational defect in pxy by mutating related RLK genes PXL1 and PXL2 (Fisher and Turner, 2007), there is no evidence of enhancement of the pxy cell division phenotype (Fisher and Turner, 2007) (Fig. 9), suggesting that if further receptors do confer the TDIF signal promoting cell division they are not closely related to PXY. On the other hand, in our previous analysis, we observed that hypocotyl diameter increases in 35S::CLE41 lines were suppressed in 35S::CLE41 pxy apparently to the level of pxy mutants (Etchells and Turner, 2010), so although this analysis does not completely rule out receptor redundancy in promoting cell division, it suggests that any redundancy is likely to be limited. As WOX4 levels are unchanged in pxy mutant seedlings, despite a clear phenotype (Hirakawa et al., 2010), we hypothesised that WOX4-redundant transcriptional targets of PXY signalling may be present. WOX14 is related to WOX4 (Haecker et al., 2004), downregulated in pxy mutants and upregulated in lines overexpressing CLE41 (Fig. 4). Strikingly, when expression of WOX4 and WOX14 was compared in pxy mutants, WOX4 was significantly reduced at the base of stems, in contrast to WOX14, the expression of which was reduced at the top of stems. These results suggest that PXY-mediated control of WOX expression is dependent on developmental stage (Fig. 4).
wox14 strongly enhanced wox4, such that wox4 wox14 mutants had comparable cell division defects to those observed in pxy wox4 double mutants (Figs 6, 7). However, pxy wox14 plants were indistinguishable from pxy single mutants (supplementary material Figs S4, S5). pxy wox4 wox14 lines appeared identical to pxy wox4 and had vascular cell numbers similar to those of wox4 wox14 (supplementary material Figs S4, S5). These observations are consistent with WOX14 acting downstream of PXY and redundantly with WOX4 (Fig. 11). Strikingly, wox4 wox14 double mutants exhibited no defect in the organisation of vascular tissue in contrast to that observed in pxy, pxy wox14 and pxy wox4 lines (Figs 6, 7; supplementary material Figs S4, S5).
Fig. 11.
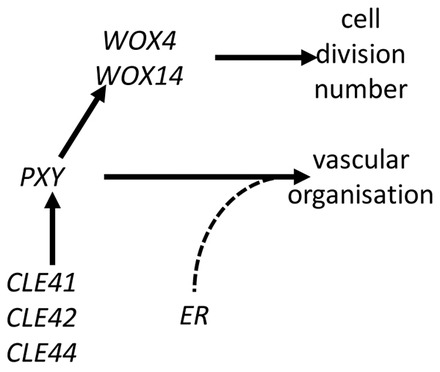
The interaction between PXY, WOX4, WOX14 and ER. PXY and ER signalling modules interacting in the Arabidopsis stem. Procambium-expressed PXY controls cell division number via WOX4 and WOX14 as a separate outcome from vascular organisation. ER, which in stems is expressed in the epidermis, phloem and xylem, is required non-cell autonomously with PXY to control vascular organisation.
Partly based upon the phenotype of pxy wox4 mutants, it has been proposed that the organisation defect in pxy is due to consumption of the vascular meristem in some cell files followed by re-initiation of a vascular developmental programme (cavity model; supplementary material Fig. S1C) (Hirakawa et al., 2010). At the shoot apex, shoot meristemless (Barton and Poethig, 1993), wuschel (Laux et al., 1996) and pinhead (McConnell and Barton, 1995) mutants all have defective apical meristems but in all instances, some level of meristem re-initiation occurs in adventitious positions on the flank of the original meristem. We have previously demonstrated that a mechanism exists whereby procambial cell divisions are promoted by ethylene in the absence of PXY signalling (Etchells et al., 2012). wox4 wox14 double mutants have a more severe proliferation defect to that observed in pxy mutants but vascular organisation is maintained; consequently, our results suggest that if the cavity model can explain the loss of vascular organisation in pxy it must involve factors other than WOX4 and WOX14.
For the ‘localised ligand’ model to be correct, the rate of cell division and vascular organisation must be genetically separable, independent outputs of pxy signalling. Evidence that this is the case comes from analysis of pxy er double mutants. Vascular cell number in pxy er lines was unchanged compared with that observed in pxy single mutants; however, defects in the arrangement of vascular tissue along the tangential and radial axes were observed (Fig. 8B) in addition to more prominent intercalation of xylem and phloem in pxy er compared with pxy. Our model, in which procambium-expressed PXY receives positional information from the phloem is analogous to a WNT signalling-controlled oriented cell division in C. elegans EMS cells (Goldstein, 1995; Schlesinger et al., 1999). ER protein interacts directly with the TOO MANY MOUTHS (TMM) receptor controlling asymmetric cell divisions during stomata development (Abrash et al., 2011; Lee et al., 2012); however, a distinct mechanism must work in the context of vascular tissue. ER is able to affect vascular organisation in the procambium but in stems it is expressed in the xylem, phloem and epidermis (Uchida et al., 2012). As such, it does not overlap with procambium-expressed PXY (Etchells and Turner, 2010), making a physical interaction between PXY and ER proteins unlikely. Although the PXY ligands CLE41/42/44 are also phloem expressed, their mRNA levels are unaffected in er mutants (supplementary material Fig. S9). Within the stem CHAL, CLL1 and CLL2 have been identified as the ligands for ER, yet they are expressed specifically in the endodermis (Abrash et al., 2011; Uchida et al., 2012). Neither chal/cll (Uchida et al., 2012) nor ERf mutants (Figs 9, 10) exhibit defects in the orientation of vascular cell division. Although it may be the case that the change in vascular organisation in pxy er is due to an unknown effect caused by the pleiotropic nature of er loss-of-function phenotypes, we speculate that the interaction between PXY and ER may represent a novel signalling pathway that coordinates differential growth over several cell layers in Arabidopsis via non-cell autonomous factors acting downstream of ER (Fig. 11).
In plants, there are a growing number of cell-surface receptors described as conferring positional information in proliferating cell lineages, possibly as a consequence of interpreting positional information derived from excreted peptide ligands. In the epidermis, stomatal spacing is enforced through EPF ligands signalling to TMM/ERf receptors (Abrash et al., 2011; Hara et al., 2007; Lee et al., 2012) and similarly in anthers, the ligand TAPETUM DETERMINANT 1 (Yang et al., 2003) signals to the EXCESS MICROSPOROCYTES 1 receptor (Yang et al., 2005), resulting in oriented divisions that form the tapetum in a contiguous layer (Canales et al., 2002; Feng and Dickinson, 2010; Zhao et al., 2002). We suggest that this may be a general mechanism for maintaining tissue integrity during cell division and differentiation during differential growth.
Acknowledgments
The authors are grateful to Keiko Torii for sharing er and erl mutants, to the Nottingham Arabidopsis Stock Centre for providing other seed lines, and to Thomas Greb for providing WOX4 in situ probe template. We thank Patrick Gallois and Minsung Kim for critical reading of the manuscript.
Footnotes
Funding
This work was funded by the Biotechnology and Biological Sciences Research Council [BB/H019928]. Deposited in PMC for immediate release.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.091314/-/DC1
References
- Abrash E. B., Davies K. A., Bergmann D. C. (2011). Generation of signaling specificity in Arabidopsis by spatially restricted buffering of ligand-receptor interactions. Plant Cell 23, 2864–2879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barton M. K., Poethig R. S. (1993). Formation of the shoot apical meristem in Arabidopsis thaliana: an analysis of development in the wild type and in the shoot meristemless mutant. Development 119, 823–831 [Google Scholar]
- Brand U., Fletcher J. C., Hobe M., Meyerowitz E. M., Simon R. (2000). Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289, 617–619 [DOI] [PubMed] [Google Scholar]
- Canales C., Bhatt A. M., Scott R., Dickinson H. (2002). EXS, a putative LRR receptor kinase, regulates male germline cell number and tapetal identity and promotes seed development in Arabidopsis. Curr. Biol. 12, 1718–1727 [DOI] [PubMed] [Google Scholar]
- Caño-Delgado A., Lee J.-Y., Demura T. (2010). Regulatory mechanisms for specification and patterning of plant vascular tissues. Annu. Rev. Cell Dev. Biol. 26, 605–637 [DOI] [PubMed] [Google Scholar]
- Clark S. E., Williams R. W., Meyerowitz E. M. (1997). The CLAVATA1 gene encodes a putative receptor kinase that controls shoot and floral meristem size in Arabidopsis. Cell 89, 575–585 [DOI] [PubMed] [Google Scholar]
- Clough S. J., Bent A. F. (1998). Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 [DOI] [PubMed] [Google Scholar]
- Etchells J. P., Turner S. R. (2010). The PXY-CLE41 receptor ligand pair defines a multifunctional pathway that controls the rate and orientation of vascular cell division. Development 137, 767–774 [DOI] [PubMed] [Google Scholar]
- Etchells J. P., Provost C. M., Turner S. R. (2012). Plant vascular cell division is maintained by an interaction between PXY and ethylene signalling. PLoS Genet. 8, e1002997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X., Dickinson H. G. (2010). Tapetal cell fate, lineage and proliferation in the Arabidopsis anther. Development 137, 2409–2416 [DOI] [PubMed] [Google Scholar]
- Fisher K., Turner S. (2007). PXY, a receptor-like kinase essential for maintaining polarity during plant vascular-tissue development. Curr. Biol. 17, 1061–1066 [DOI] [PubMed] [Google Scholar]
- Fletcher J. C., Brand U., Running M. P., Simon R., Meyerowitz E. M. (1999). Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 283, 1911–1914 [DOI] [PubMed] [Google Scholar]
- Goldstein B. (1995). Cell contacts orient some cell division axes in the Caenorhabditis elegans embryo. J. Cell Biol. 129, 1071–1080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haecker A., Gross-Hardt R., Geiges B., Sarkar A., Breuninger H., Herrmann M., Laux T. (2004). Expression dynamics of WOX genes mark cell fate decisions during early embryonic patterning in Arabidopsis thaliana. Development 131, 657–668 [DOI] [PubMed] [Google Scholar]
- Hara K., Kajita R., Torii K. U., Bergmann D. C., Kakimoto T. (2007). The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes Dev. 21, 1720–1725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara K., Yokoo T., Kajita R., Onishi T., Yahata S., Peterson K. M., Torii K. U., Kakimoto T. (2009). Epidermal cell density is autoregulated via a secretory peptide, EPIDERMAL PATTERNING FACTOR 2 in Arabidopsis leaves. Plant Cell Physiol. 50, 1019–1031 [DOI] [PubMed] [Google Scholar]
- Hårdstedt M., Finnegan C. P., Kirchhof N., Hyland K. A., Wijkstrom M., Murtaugh M. P., Hering B. J. (2005). Post-transplant upregulation of chemokine messenger RNA in non-human primate recipients of intraportal pig islet xenografts. Xenotransplantation 12, 293–302 [DOI] [PubMed] [Google Scholar]
- Hirakawa Y., Shinohara H., Kondo Y., Inoue A., Nakanomyo I., Ogawa M., Sawa S., Ohashi-Ito K., Matsubayashi Y., Fukuda H. (2008). Non-cell-autonomous control of vascular stem cell fate by a CLE peptide/receptor system. Proc. Natl. Acad. Sci. USA 105, 15208–15213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirakawa Y., Kondo Y., Fukuda H. (2010). TDIF peptide signaling regulates vascular stem cell proliferation via the WOX4 homeobox gene in Arabidopsis. Plant Cell 22, 2618–2629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L., Gray J. E. (2009). The signaling peptide EPF2 controls asymmetric cell divisions during stomatal development. Curr. Biol. 19, 864–869 [DOI] [PubMed] [Google Scholar]
- Ito Y., Nakanomyo I., Motose H., Iwamoto K., Sawa S., Dohmae N., Fukuda H. (2006). Dodeca-CLE peptides as suppressors of plant stem cell differentiation. Science 313, 842–845 [DOI] [PubMed] [Google Scholar]
- Jeong S., Trotochaud A. E., Clark S. E. (1999). The Arabidopsis CLAVATA2 gene encodes a receptor-like protein required for the stability of the CLAVATA1 receptor-like kinase. Plant Cell 11, 1925–1934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji J., Strable J., Shimizu R., Koenig D., Sinha N., Scanlon M. J. (2010). WOX4 promotes procambial development. Plant Physiol. 152, 1346–1356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinoshita A., Betsuyaku S., Osakabe Y., Mizuno S., Nagawa S., Stahl Y., Simon R., Yamaguchi-Shinozaki K., Fukuda H., Sawa S. (2010). RPK2 is an essential receptor-like kinase that transmits the CLV3 signal in Arabidopsis. Development 137, 3911–3920 [DOI] [PubMed] [Google Scholar]
- Laux T., Mayer K. F., Berger J., Jürgens G. (1996). The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122, 87–96 [DOI] [PubMed] [Google Scholar]
- Lease K. A., Lau N. Y., Schuster R. A., Torii K. U., Walker J. C. (2001). Receptor serine/threonine protein kinases in signalling: analysis of the erecta receptor-like kinase of Arabidopsis thaliana. New Phytol. 151, 133–143 [DOI] [PubMed] [Google Scholar]
- Lee J. S., Kuroha T., Hnilova M., Khatayevich D., Kanaoka M. M., McAbee J. M., Sarikaya M., Tamerler C., Torii K. U. (2012). Direct interaction of ligand-receptor pairs specifying stomatal patterning. Genes Dev. 26, 126–136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McConnell J. R., Barton M. K. (1995). Effect of mutations in the PINHEAD gene of Arabidopsis on the formation of shoot apical meristems. Dev. Genet. 16, 358–366 [Google Scholar]
- Müller R., Bleckmann A., Simon R. (2008). The receptor kinase CORYNE of Arabidopsis transmits the stem cell-limiting signal CLAVATA3 independently of CLAVATA1. Plant Cell 20, 934–946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa M., Shinohara H., Sakagami Y., Matsubayashi Y. (2008). Arabidopsis CLV3 peptide directly binds CLV1 ectodomain. Science 319, 294–294 [DOI] [PubMed] [Google Scholar]
- Ragni L., Nieminen K., Pacheco-Villalobos D., Sibout R., Schwechheimer C., Hardtke C. S. (2011). Mobile gibberellin directly stimulates Arabidopsis hypocotyl xylem expansion. Plant Cell 23, 1322–1336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosso M. G., Li Y., Strizhov N., Reiss B., Dekker K., Weisshaar B. (2003). An Arabidopsis thaliana T-DNA mutagenized population (GABI-Kat) for flanking sequence tag-based reverse genetics. Plant Mol. Biol. 53, 247–259 [DOI] [PubMed] [Google Scholar]
- Sarkar A. K., Luijten M., Miyashima S., Lenhard M., Hashimoto T., Nakajima K., Scheres B., Heidstra R., Laux T. (2007). Conserved factors regulate signalling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 446, 811–814 [DOI] [PubMed] [Google Scholar]
- Schlesinger A., Shelton C. A., Maloof J. N., Meneghini M., Bowerman B. (1999). Wnt pathway components orient a mitotic spindle in the early Caenorhabditis elegans embryo without requiring gene transcription in the responding cell. Genes Dev. 13, 2028–2038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoof H., Lenhard M., Haecker A., Mayer K. F. X., Jürgens G., Laux T. (2000). The stem cell population of Arabidopsis shoot meristems in maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100, 635–644 [DOI] [PubMed] [Google Scholar]
- Shpak E. D., Berthiaume C. T., Hill E. J., Torii K. U. (2004). Synergistic interaction of three ERECTA-family receptor-like kinases controls Arabidopsis organ growth and flower development by promoting cell proliferation. Development 131, 1491–1501 [DOI] [PubMed] [Google Scholar]
- Shpak E. D., McAbee J. M., Pillitteri L. J., Torii K. U. (2005). Stomatal patterning and differentiation by synergistic interactions of receptor kinases. Science 309, 290–293 [DOI] [PubMed] [Google Scholar]
- Suer S., Agusti J., Sanchez P., Schwarz M., Greb T. (2011). WOX4 imparts auxin responsiveness to cambium cells in Arabidopsis. Plant Cell 23, 3247–3259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swarbreck D., Wilks C., Lamesch P., Berardini T. Z., Garcia-Hernandez M., Foerster H., Li D., Meyer T., Muller R., Ploetz L., et al. (2008). The Arabidopsis Information Resource (TAIR): gene structure and function annotation. Nucleic Acids Res. 36, D1009–D1014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uchida N., Lee J. S., Horst R. J., Lai H.-H., Kajita R., Kakimoto T., Tasaka M., Torii K. U. (2012). Regulation of inflorescence architecture by intertissue layer ligand-receptor communication between endodermis and phloem. Proc. Natl. Acad. Sci. USA 109, 6337–6342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Zanten M., Snoek L. B., Proveniers M. C. G., Peeters A. J. M. (2009). The many functions of ERECTA. Trends Plant Sci. 14, 214–218 [DOI] [PubMed] [Google Scholar]
- Yang S.-L., Xie L.-F., Mao H.-Z., Puah C. S., Yang W.-C., Jiang L., Sundaresan V., Ye D. (2003). Tapetum determinant1 is required for cell specialization in the Arabidopsis anther. Plant Cell 15, 2792–2804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S.-L., Jiang L., Puah C. S., Xie L.-F., Zhang X.-Q., Chen L.-Q., Yang W.-C., Ye D. (2005). Overexpression of TAPETUM DETERMINANT1 alters the cell fates in the Arabidopsis carpel and tapetum via genetic interaction with excess microsporocytes1/extra sporogenous cells. Plant Physiol. 139, 186–191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young B., Wightman R., Blanvillain R., Purcel S. B., Gallois P. (2010). pH-sensitivity of YFP provides an intracellular indicator of programmed cell death. Plant Methods 6, 27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao D.-Z., Wang G.-F., Speal B., Ma H. (2002). The excess microsporocytes1 gene encodes a putative leucine-rich repeat receptor protein kinase that controls somatic and reproductive cell fates in the Arabidopsis anther. Genes Dev. 16, 2021–2031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y., Wang Y., Li R., Song X., Wang Q., Huang S., Jin J. B., Liu C.-M., Lin J. (2010). Analysis of interactions among the CLAVATA3 receptors reveals a direct interaction between CLAVATA2 and CORYNE in Arabidopsis. Plant J. 61, 223–233 [DOI] [PubMed] [Google Scholar]


