Abstract
A cDNA clone containing sequences complementary to the mRNA coding for anemic rat spleen porphobilinogen deaminase (EC 4.3.1.8) has been isolated. A cDNA library was prepared from partially purified mRNA (1% purity). This library was then screened by colony hybridization, using a cDNA probe derived from porphobilinogen deaminase mRNA further enriched (10-20% purity) by gel electrophoresis in the presence of methylmercury hydroxide. Colonies hybridizing with the probe were analyzed by hybrid-selected translation using anemic rat spleen mRNA. Four recombinant plasmids containing porphobilinogen deaminase cDNA sequences were identified by specific immunoprecipitation of the translational product from hybrid-selected mRNA. Porphobilinogen deaminase mRNA was shown to contain 1800 bases by blot hybridization analysis. The cloned cDNA sequence consists of 1500 bases. Hybridization analysis of poly(A)+ RNA from uninduced and induced mouse erythroleukemic cells indicated that induction to erythroid differentiation by dimethyl sulfoxide results in a 10-fold increase of porphobilinogen deaminase mRNA. The rat cDNA clones hybridize to the corresponding sequences encoding human porphobilinogen deaminase. This property will be useful for isolation of human gene(s) and further characterization of the molecular lesion(s) responsible for acute intermittent porphyria.
Full text
PDF


Images in this article
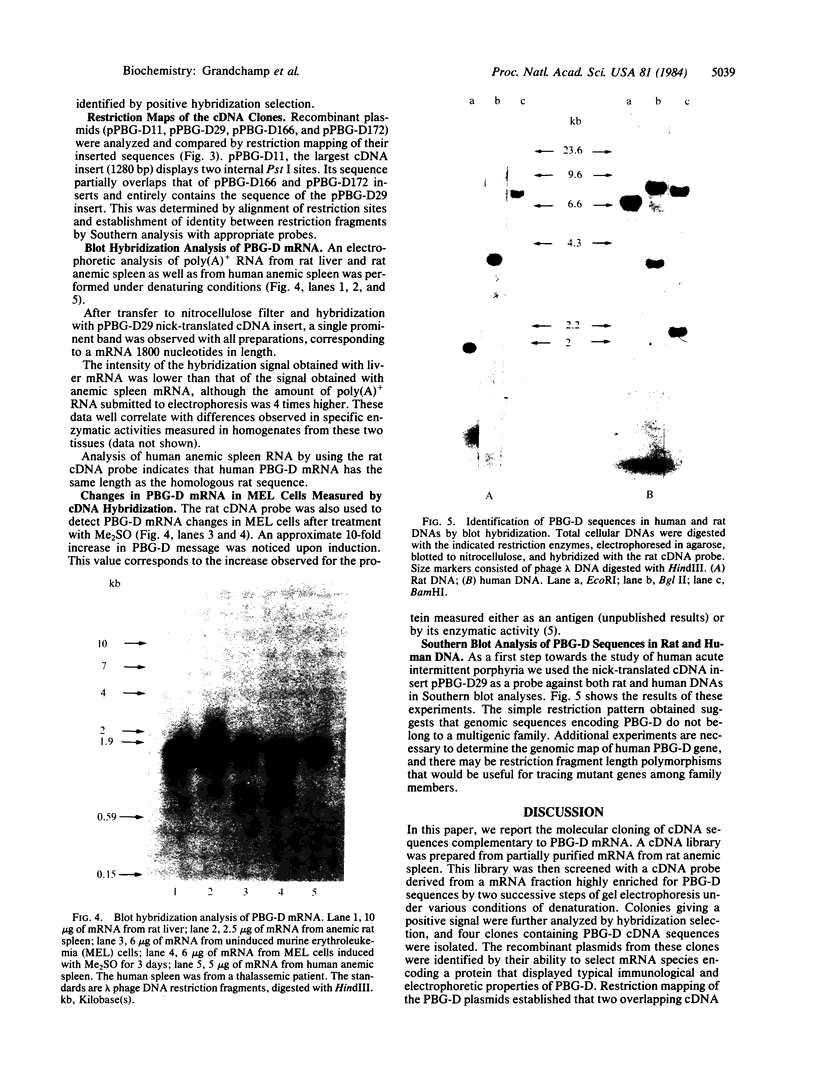
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson P. M., Desnick R. J. Purification and properties of uroporphyrinogen I synthase from human erythrocytes. Identification of stable enzyme-substrate intermediates. J Biol Chem. 1980 Mar 10;255(5):1993–1999. [PubMed] [Google Scholar]
- Anderson P. M., Reddy R. M., Anderson K. E., Desnick R. J. Characterization of the porphobilinogen deaminase deficiency in acute intermittent porphyria. Immunologic evidence for heterogeneity of the genetic defect. J Clin Invest. 1981 Jul;68(1):1–12. doi: 10.1172/JCI110223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bailey J. M., Davidson N. Methylmercury as a reversible denaturing agent for agarose gel electrophoresis. Anal Biochem. 1976 Jan;70(1):75–85. doi: 10.1016/s0003-2697(76)80049-8. [DOI] [PubMed] [Google Scholar]
- Brûlet P., Roskam W. G. Improved method for cloning DNA complementary to minor mRNAs: preparation of a hybridization probe from purified mRNAs encoding intermediate filament proteins. Gene. 1983 Apr;22(1):59–68. doi: 10.1016/0378-1119(83)90064-1. [DOI] [PubMed] [Google Scholar]
- Chandler P. M., Rimkus D., Davidson N. Gel electrophoretic fractionation of RNAs by partial denaturation with methylmercuric hydroxide. Anal Biochem. 1979 Oct 15;99(1):200–206. doi: 10.1016/0003-2697(79)90063-0. [DOI] [PubMed] [Google Scholar]
- Curtis P. J. Cloning of mouse carbonic anhydrase mRNA and its induction in mouse erythroleukemic cells. J Biol Chem. 1983 Apr 10;258(7):4459–4463. [PubMed] [Google Scholar]
- Dottavio-Martin D., Ravel J. M. Radiolabeling of proteins by reductive alkylation with [14C]formaldehyde and sodium cyanoborohydride. Anal Biochem. 1978 Jul 1;87(2):562–565. doi: 10.1016/0003-2697(78)90706-6. [DOI] [PubMed] [Google Scholar]
- Goossens M., Kan Y. Y. DNA analysis in the diagnosis of hemoglobin disorders. Methods Enzymol. 1981;76:805–817. doi: 10.1016/0076-6879(81)76159-7. [DOI] [PubMed] [Google Scholar]
- Hanahan D., Meselson M. Plasmid screening at high colony density. Gene. 1980 Jun;10(1):63–67. doi: 10.1016/0378-1119(80)90144-4. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hofer E., Hofer-Warbinek R., Darnell J. E., Jr Globin RNA transcription: a possible termination site and demonstration of transcriptional control correlated with altered chromatin structure. Cell. 1982 Jul;29(3):887–893. doi: 10.1016/0092-8674(82)90450-0. [DOI] [PubMed] [Google Scholar]
- Holmes D. S., Quigley M. A rapid boiling method for the preparation of bacterial plasmids. Anal Biochem. 1981 Jun;114(1):193–197. doi: 10.1016/0003-2697(81)90473-5. [DOI] [PubMed] [Google Scholar]
- Itoh N., Nose K., Okamoto H. Purification and characterization of proinsulin mRNA from rat B-cell tumor. Eur J Biochem. 1979 Jun;97(1):1–9. doi: 10.1111/j.1432-1033.1979.tb13079.x. [DOI] [PubMed] [Google Scholar]
- Kahn A., Cottreau D., Daegelen D., Dreyfus J. C. Cell-free translation of messenger RNAs from adult and fetal human muscle. Characterization of neosynthesized glycogen phosphorylase, phosphofructokinase and glucose phosphate isomerase. Eur J Biochem. 1981 May;116(1):7–12. doi: 10.1111/j.1432-1033.1981.tb05293.x. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Maniatis T., Jeffrey A., Kleid D. G. Nucleotide sequence of the rightward operator of phage lambda. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1184–1188. doi: 10.1073/pnas.72.3.1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michelson A. M., Orkin S. H. Characterization of the homopolymer tailing reaction catalyzed by terminal deoxynucleotidyl transferase. Implications for the cloning of cDNA. J Biol Chem. 1982 Dec 25;257(24):14773–14782. [PubMed] [Google Scholar]
- Miyagi K., Kaneshima M., Kawakami J., Nakada F., Petryka Z. J., Watson C. J. Uroporphyrinogen I synthase from human erythrocytes: separation, purification, and properties of isoenzymes. Proc Natl Acad Sci U S A. 1979 Dec;76(12):6172–6176. doi: 10.1073/pnas.76.12.6172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oppermann H., Levinson A. D., Varmus H. E. The structure and protein kinase activity of proteins encoded by nonconditional mutants and back mutants in the sec gene of avian sarcoma virus. Virology. 1981 Jan 15;108(1):47–70. doi: 10.1016/0042-6822(81)90526-2. [DOI] [PubMed] [Google Scholar]
- Parnes J. R., Velan B., Felsenfeld A., Ramanathan L., Ferrini U., Appella E., Seidman J. G. Mouse beta 2-microglobulin cDNA clones: a screening procedure for cDNA clones corresponding to rare mRNAs. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2253–2257. doi: 10.1073/pnas.78.4.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham H. R., Jackson R. J. An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem. 1976 Aug 1;67(1):247–256. doi: 10.1111/j.1432-1033.1976.tb10656.x. [DOI] [PubMed] [Google Scholar]
- Rovera G., Bonaiuto J. The phenotypes of variant clones of Friend mouse erythroleukemic cells resistant to dimethyl sulfoxide. Cancer Res. 1976 Nov;36(11 Pt 1):4057–4061. [PubMed] [Google Scholar]
- Rutherford T., Thompson G. G., Moore M. R. Heme biosynthesis in Friend erythroleukemia cells: control by ferrochelatase. Proc Natl Acad Sci U S A. 1979 Feb;76(2):833–836. doi: 10.1073/pnas.76.2.833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sassa S. Sequential induction of heme pathway enzymes during erythroid differentiation of mouse Friend leukemia virus-infected cells. J Exp Med. 1976 Feb 1;143(2):305–315. doi: 10.1084/jem.143.2.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sassa S., Urabe A. Uroporphyrinogen I synthase induction in normal human bone marrow cultures: an early and quantitative response of erythroid differentiation. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5321–5325. doi: 10.1073/pnas.76.10.5321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon M. P., Besmond C., Cottreau D., Weber A., Chaumet-Riffaud P., Dreyfus J. C., Trépat J. S., Marie J., Kahn A. Molecular cloning of cDNA for rat L-type pyruvate kinase and aldolase B. J Biol Chem. 1983 Dec 10;258(23):14576–14584. [PubMed] [Google Scholar]
- Thayer R. E. An improved method for detecting foreign DNA in plasmids of Escherichia coli. Anal Biochem. 1979 Sep 15;98(1):60–63. doi: 10.1016/0003-2697(79)90705-x. [DOI] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickens M. P., Buell G. N., Schimke R. T. Synthesis of double-stranded DNA complementary to lysozyme, ovomucoid, and ovalbumin mRNAs. Optimization for full length second strand synthesis by Escherichia coli DNA polymerase I. J Biol Chem. 1978 Apr 10;253(7):2483–2495. [PubMed] [Google Scholar]
- Zasloff M., Ginder G. D., Felsenfeld G. A new method for the purification and identification of covalently closed circular DNA molcules. Nucleic Acids Res. 1978 Apr;5(4):1139–1152. doi: 10.1093/nar/5.4.1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Verneuil H., Phung N., Nordmann Y., Allard D., Leprince F., Jérome H., Aurias A., Rethoré M. O. Assignment of human uroporphyrinogen I synthase locus to region 11qter by gene dosage effect. Hum Genet. 1982;60(3):212–213. doi: 10.1007/BF00303004. [DOI] [PubMed] [Google Scholar]