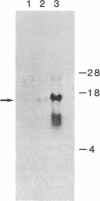
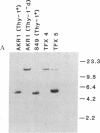
Abstract
We isolated a gene encoding the Thy-1.2 glycoprotein from a recombinant library constructed from BALB/c mouse DNA. To evaluate the expression of this cloned gene in different genomic environments, we introduced it into cell lines derived from fibroblast, lymphoid, and neuronal tissues by DNA-mediated gene transfer. When integrated into the genome of mouse L cells, cell-surface Thy-1 can be detected with anti-Thy-1 monoclonal antibodies. These L-cell lines contain between two and four copies of the cloned Thy-1 gene stably integrated in the host genome. After subcloning into a plasmid vector containing the bacterial Eco-gpt gene as a selectable marker, the Thy-1 gene was introduced into the Thy-1-deficient mouse lymphoma AKR1 (Thy-1-d), and the rat neuronal cell line, B50. The resulting transformants also contain two to four copies of the cloned Thy-1 gene but express up to 50-fold more cell-surface Thy-1.2 than the L cell transformants. The expression of vastly differing amounts of cell-surface Thy-1 from similar numbers of genes suggests that the gene encoding this differentiation antigen is under tissue-specific regulation.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acton R. T., Morris R. J., Williams A. F. Estimation of the amount and tissue distribution of rat Thy-1.1 antigen. Eur J Immunol. 1974 Sep;4(9):598–602. doi: 10.1002/eji.1830040904. [DOI] [PubMed] [Google Scholar]
- Basch R. S., Berman J. W. Thy-1 determinants are present on many murine hematopoietic cells other than T cells. Eur J Immunol. 1982 May;12(5):359–364. doi: 10.1002/eji.1830120502. [DOI] [PubMed] [Google Scholar]
- Blankenhorn E. P., Douglas T. C. Location of the gene for Theta antigen in the mouse. J Hered. 1972 Sep-Oct;63(5):259–263. doi: 10.1093/oxfordjournals.jhered.a108290. [DOI] [PubMed] [Google Scholar]
- Cohen F. E., Novotný J., Sternberg M. J., Campbell D. G., Williams A. F. Analysis of structural similarities between brain Thy-1 antigen and immunoglobulin domains. Evidence for an evolutionary relationship and a hypothesis for its functional significance. Biochem J. 1981 Apr 1;195(1):31–40. doi: 10.1042/bj1950031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dennert G., Hyman R., Lesley J., Trowbridge I. S. Effects of cytotoxic monoclonal antibody specific for T200 glycoprotein on functional lymphoid cell populations. Cell Immunol. 1980 Aug 1;53(2):350–364. doi: 10.1016/0008-8749(80)90335-4. [DOI] [PubMed] [Google Scholar]
- Derman E., Krauter K., Walling L., Weinberger C., Ray M., Darnell J. E., Jr Transcriptional control in the production of liver-specific mRNAs. Cell. 1981 Mar;23(3):731–739. doi: 10.1016/0092-8674(81)90436-0. [DOI] [PubMed] [Google Scholar]
- Enquist L. W., Vande Woude G. F., Wagner M., Smiley J. R., Summers W. C. Construction and characterization of a recombinant plasmid encoding the gene for the thymidine kinase of Herpes simplex type 1 virus. Gene. 1979 Nov;7(3-4):335–342. doi: 10.1016/0378-1119(79)90052-0. [DOI] [PubMed] [Google Scholar]
- Fields K. L., Brockes J. P., Mirsky R., Wendon L. M. Cell surface markers for distinguishing different types of rat dorsal root ganglion cells in culture. Cell. 1978 May;14(1):43–51. doi: 10.1016/0092-8674(78)90299-4. [DOI] [PubMed] [Google Scholar]
- Folsom V., Gold D. P., White J., Marrack P., Kappler J., Tonegawa S. Functional and inducible expression of a transfected murine class II major histocompatibility complex gene. Proc Natl Acad Sci U S A. 1984 Apr;81(7):2045–2049. doi: 10.1073/pnas.81.7.2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillies S. D., Morrison S. L., Oi V. T., Tonegawa S. A tissue-specific transcription enhancer element is located in the major intron of a rearranged immunoglobulin heavy chain gene. Cell. 1983 Jul;33(3):717–728. doi: 10.1016/0092-8674(83)90014-4. [DOI] [PubMed] [Google Scholar]
- Goodenow R. S., McMillan M., Nicolson M., Sher B. T., Eakle K., Davidson N., Hood L. Identification of the class I genes of the mouse major histocompatibility complex by DNA-mediated gene transfer. Nature. 1982 Nov 18;300(5889):231–237. doi: 10.1038/300231a0. [DOI] [PubMed] [Google Scholar]
- Hansen J. N. Use of solubilizable acrylamide disulfide gels for isolation of DNA fragments suitable for sequence analysis. Anal Biochem. 1981 Sep 1;116(1):146–151. doi: 10.1016/0003-2697(81)90337-7. [DOI] [PubMed] [Google Scholar]
- Hyman R., Cunningham K., Stallings V. Effect of gene dosage on cell-surface expression of Thy-1 antigen in somatic cell hybrids between Thy-1- Abelson-leukemia-virus induced lymphomas and Thy-1+ mouse lymphomas. Immunogenetics. 1981;12(3-4):381–395. doi: 10.1007/BF01561678. [DOI] [PubMed] [Google Scholar]
- Itakura K., Hutton J. J., Boyse E. A., Old L. J. Genetic linkage relationships of loci specifying differentiation alloantigens in the mouse. Transplantation. 1972 Mar;13(3):239–243. doi: 10.1097/00007890-197203000-00007. [DOI] [PubMed] [Google Scholar]
- Lake P., Clark E. A., Khorshidi M., Sunshine G. H. Production and characterization of cytotoxic Thy-1 antibody-secreting hybrid cell lines. Detection of T cell subsets. Eur J Immunol. 1979 Nov;9(11):875–886. doi: 10.1002/eji.1830091109. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Herzenberg L. A. Xenogeneic monoclonal antibodies to mouse lymphoid differentiation antigens. Immunol Rev. 1979;47:63–90. doi: 10.1111/j.1600-065x.1979.tb00289.x. [DOI] [PubMed] [Google Scholar]
- Lesley J. F., Lennon V. A. Thy-L antigen expression on rat brain cell lines. Brain Res. 1978 Sep 15;153(1):109–120. doi: 10.1016/0006-8993(78)91132-0. [DOI] [PubMed] [Google Scholar]
- Lesley J., Hyman R., Schulte R., Trotter J. Expression of transferrin receptor on murine hematopoietic progenitors. Cell Immunol. 1984 Jan;83(1):14–25. doi: 10.1016/0008-8749(84)90220-x. [DOI] [PubMed] [Google Scholar]
- Margulies D. H., Evans G. A., Ozato K., Camerini-Otero R. D., Tanaka K., Appella E., Seidman J. G. Expression of H-2Dd and H-2Ld mouse major histocompatibility antigen genes in L cells after DNA-mediated gene transfer. J Immunol. 1983 Jan;130(1):463–470. [PubMed] [Google Scholar]
- Margulies D. H., Parnes J. R., Johnson N. A., Seidman J. G. Linkage of beta 2-microglobulin and ly-m11 by molecular cloning and DNA-mediated gene transfer. Proc Natl Acad Sci U S A. 1983 Apr;80(8):2328–2331. doi: 10.1073/pnas.80.8.2328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marshak-Rothstein A., Fink P., Gridley T., Raulet D. H., Bevan M. J., Gefter M. L. Properties and applications of monoclonal antibodies directed against determinants of the Thy-1 locus. J Immunol. 1979 Jun;122(6):2491–2497. [PubMed] [Google Scholar]
- Mercola M., Wang X. F., Olsen J., Calame K. Transcriptional enhancer elements in the mouse immunoglobulin heavy chain locus. Science. 1983 Aug 12;221(4611):663–665. doi: 10.1126/science.6306772. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Moriuchi T., Chang H. C., Denome R., Silver J. Thy-1 cDNA sequence suggests a novel regulatory mechanism. Nature. 1983 Jan 6;301(5895):80–82. doi: 10.1038/301080a0. [DOI] [PubMed] [Google Scholar]
- Mulligan R. C., Berg P. Selection for animal cells that express the Escherichia coli gene coding for xanthine-guanine phosphoribosyltransferase. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2072–2076. doi: 10.1073/pnas.78.4.2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuberger M. S. Expression and regulation of immunoglobulin heavy chain gene transfected into lymphoid cells. EMBO J. 1983;2(8):1373–1378. doi: 10.1002/j.1460-2075.1983.tb01594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumann E., Schaefer-Ridder M., Wang Y., Hofschneider P. H. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1982;1(7):841–845. doi: 10.1002/j.1460-2075.1982.tb01257.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owen J. J., Raff M. C. Studies on the differentiation of thymus-derived lymphocytes. J Exp Med. 1970 Dec 1;132(6):1216–1232. doi: 10.1084/jem.132.6.1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruss R. M. Thy-1 antigen on astrocytes in long-term cultures of rat central nervous system. Nature. 1979 Aug 23;280(5724):688–690. doi: 10.1038/280688a0. [DOI] [PubMed] [Google Scholar]
- REIF A. E., ALLEN J. M. THE AKR THYMIC ANTIGEN AND ITS DISTRIBUTION IN LEUKEMIAS AND NERVOUS TISSUES. J Exp Med. 1964 Sep 1;120:413–433. doi: 10.1084/jem.120.3.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruddle F. H. A new era in mammalian gene mapping: somatic cell genetics and recombinant DNA methodologies. Nature. 1981 Nov 12;294(5837):115–120. doi: 10.1038/294115a0. [DOI] [PubMed] [Google Scholar]
- Schubert D., Heinemann S., Carlisle W., Tarikas H., Kimes B., Patrick J., Steinbach J. H., Culp W., Brandt B. L. Clonal cell lines from the rat central nervous system. Nature. 1974 May 17;249(454):224–227. doi: 10.1038/249224a0. [DOI] [PubMed] [Google Scholar]
- Wahl G. M., Stern M., Stark G. R. Efficient transfer of large DNA fragments from agarose gels to diazobenzyloxymethyl-paper and rapid hybridization by using dextran sulfate. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3683–3687. doi: 10.1073/pnas.76.8.3683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. F., Gagnon J. Neuronal cell Thy-1 glycoprotein: homology with immunoglobulin. Science. 1982 May 14;216(4547):696–703. doi: 10.1126/science.6177036. [DOI] [PubMed] [Google Scholar]