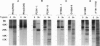Abstract
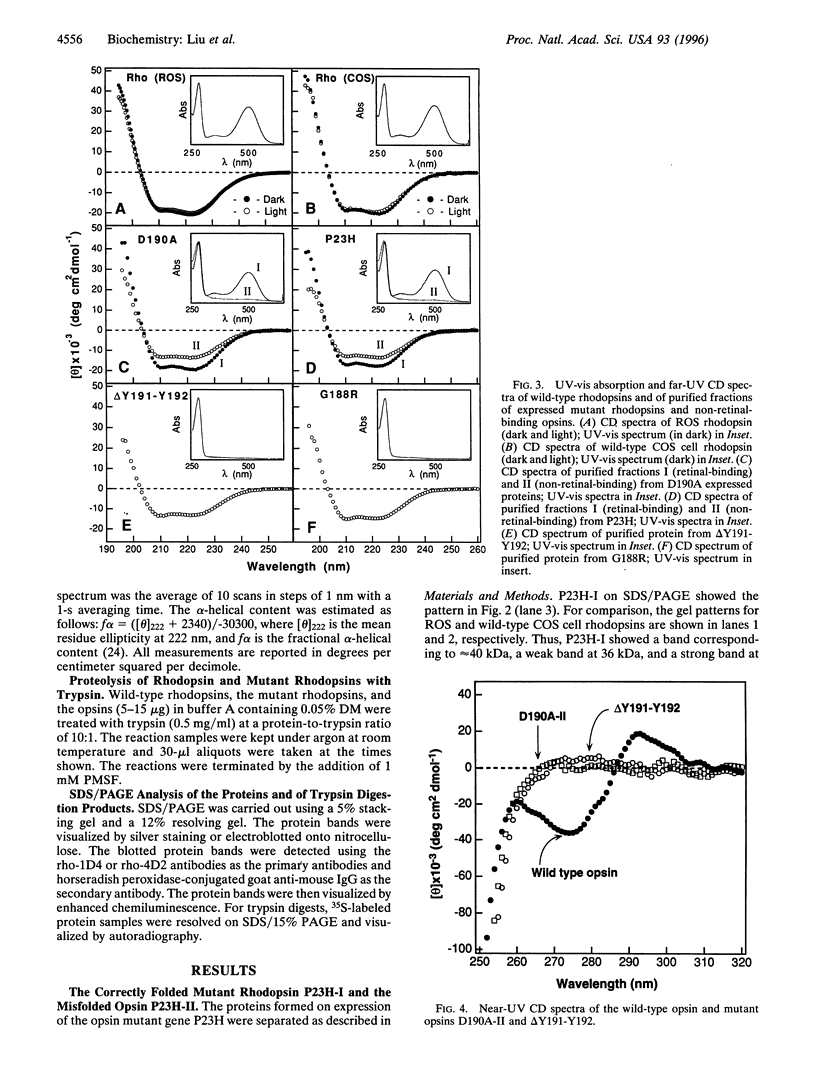
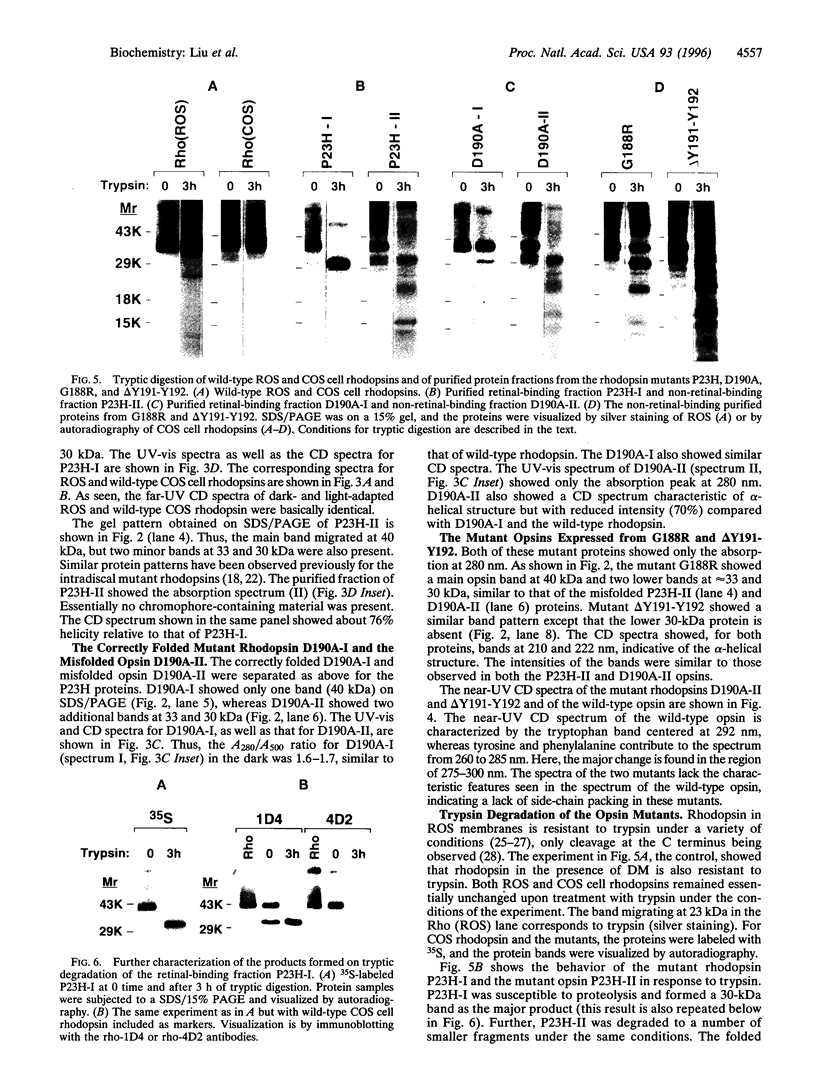
The rhodopsin mutants P23H and G188R, identified in autosomal dominant retinitis pigmentosa (ADRP), and the site-specific mutants D190A and DeltaY191-Y192 were expressed in COS cells from synthetic mutant opsin genes containing these mutations. The proteins expressed from P23H and D190A partially regenerated the rhodopsin chromophore with 11-cis-retinal and were mixtures of the correctly folded (retinal-binding) and misfolded (non-retinal-binding) opsins. The mixtures were separated into pure, correctly folded mutant rhodopsins and misfolded opsins. The proteins expressed from the ADRP mutant G188R and the mutant DeltaY191-Y192 were composed of totally misfolded non-retinal-binding opsins. Far-UV CD spectra showed that the correctly folded mutant rhodopsins had helical content similar to that of the wild-type rhodopsin, whereas the misfolded opsins had helical content 50-70% of the wild type. The near-UV CD spectra of the misfolded mutant proteins lack the characteristic band pattern seen in the wild-type opsin, indicative of a different tertiary structure. Further, whereas the folded mutant rhodopsins were essentially resistant to trypsin digestion, the misfolded opsins were degraded to small fragments under the same conditions. Therefore, the misfolded opsins appear to be less compact in their structures than the correctly folded forms. We suggest that most, if not all, of the point mutations in the intradiscal domain identified in ADRP cause partial or complete misfolding of rhodopsin.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anukanth A., Khorana H. G. Structure and function in rhodopsin. Requirements of a specific structure for the intradiscal domain. J Biol Chem. 1994 Aug 5;269(31):19738–19744. [PubMed] [Google Scholar]
- Berson E. L. Retinitis pigmentosa. The Friedenwald Lecture. Invest Ophthalmol Vis Sci. 1993 Apr;34(5):1659–1676. [PubMed] [Google Scholar]
- Chen Y. H., Yang J. T., Martinez H. M. Determination of the secondary structures of proteins by circular dichroism and optical rotatory dispersion. Biochemistry. 1972 Oct 24;11(22):4120–4131. doi: 10.1021/bi00772a015. [DOI] [PubMed] [Google Scholar]
- Doi T., Molday R. S., Khorana H. G. Role of the intradiscal domain in rhodopsin assembly and function. Proc Natl Acad Sci U S A. 1990 Jul;87(13):4991–4995. doi: 10.1073/pnas.87.13.4991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dryja T. P., Berson E. L. Retinitis pigmentosa and allied diseases. Implications of genetic heterogeneity. Invest Ophthalmol Vis Sci. 1995 Jun;36(7):1197–1200. [PubMed] [Google Scholar]
- Dryja T. P., Hahn L. B., Cowley G. S., McGee T. L., Berson E. L. Mutation spectrum of the rhodopsin gene among patients with autosomal dominant retinitis pigmentosa. Proc Natl Acad Sci U S A. 1991 Oct 15;88(20):9370–9374. doi: 10.1073/pnas.88.20.9370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dryja T. P., McGee T. L., Reichel E., Hahn L. B., Cowley G. S., Yandell D. W., Sandberg M. A., Berson E. L. A point mutation of the rhodopsin gene in one form of retinitis pigmentosa. Nature. 1990 Jan 25;343(6256):364–366. doi: 10.1038/343364a0. [DOI] [PubMed] [Google Scholar]
- Farahbakhsh Z. T., Ridge K. D., Khorana H. G., Hubbell W. L. Mapping light-dependent structural changes in the cytoplasmic loop connecting helices C and D in rhodopsin: a site-directed spin labeling study. Biochemistry. 1995 Jul 11;34(27):8812–8819. doi: 10.1021/bi00027a033. [DOI] [PubMed] [Google Scholar]
- Fuchs S., Kranich H., Denton M. J., Zrenner E., Bhattacharya S. S., Humphries P., Gal A. Three novel rhodopsin mutations (C110F, L131P, A164V) in patients with autosomal dominant retinitis pigmentosa. Hum Mol Genet. 1994 Jul;3(7):1203–1203. doi: 10.1093/hmg/3.7.1203. [DOI] [PubMed] [Google Scholar]
- Hicks D., Molday R. S. Differential immunogold-dextran labeling of bovine and frog rod and cone cells using monoclonal antibodies against bovine rhodopsin. Exp Eye Res. 1986 Jan;42(1):55–71. doi: 10.1016/0014-4835(86)90017-5. [DOI] [PubMed] [Google Scholar]
- Karnik S. S., Sakmar T. P., Chen H. B., Khorana H. G. Cysteine residues 110 and 187 are essential for the formation of correct structure in bovine rhodopsin. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8459–8463. doi: 10.1073/pnas.85.22.8459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaushal S., Khorana H. G. Structure and function in rhodopsin. 7. Point mutations associated with autosomal dominant retinitis pigmentosa. Biochemistry. 1994 May 24;33(20):6121–6128. doi: 10.1021/bi00186a011. [DOI] [PubMed] [Google Scholar]
- Kim R. Y., al-Maghtheh M., Fitzke F. W., Arden G. B., Jay M., Bhattacharya S. S., Bird A. C. Dominant retinitis pigmentosa associated with two rhodopsin gene mutations. Leu-40-Arg and an insertion disrupting the 5'-splice junction of exon 5. Arch Ophthalmol. 1993 Nov;111(11):1518–1524. doi: 10.1001/archopht.1993.01090110084030. [DOI] [PubMed] [Google Scholar]
- Li T., Franson W. K., Gordon J. W., Berson E. L., Dryja T. P. Constitutive activation of phototransduction by K296E opsin is not a cause of photoreceptor degeneration. Proc Natl Acad Sci U S A. 1995 Apr 11;92(8):3551–3555. doi: 10.1073/pnas.92.8.3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macke J. P., Davenport C. M., Jacobson S. G., Hennessey J. C., Gonzalez-Fernandez F., Conway B. P., Heckenlively J., Palmer R., Maumenee I. H., Sieving P. Identification of novel rhodopsin mutations responsible for retinitis pigmentosa: implications for the structure and function of rhodopsin. Am J Hum Genet. 1993 Jul;53(1):80–89. [PMC free article] [PubMed] [Google Scholar]
- Macke J. P., Hennessey J. C., Nathans J. Rhodopsin mutation proline347-to-alanine in a family with autosomal dominant retinitis pigmentosa indicates an important role for proline at position 347. Hum Mol Genet. 1995 Apr;4(4):775–776. doi: 10.1093/hmg/4.4.775. [DOI] [PubMed] [Google Scholar]
- Millán J. M., Fuchs S., Paricio N., Wedemann H., Gal A., Nájera C., Prieto F. Gly114Asp mutation of rhodopsin in autosomal dominant retinitis pigmentosa. Mol Cell Probes. 1995 Feb;9(1):67–69. doi: 10.1016/s0890-8508(95)91052-2. [DOI] [PubMed] [Google Scholar]
- Min K. C., Zvyaga T. A., Cypess A. M., Sakmar T. P. Characterization of mutant rhodopsins responsible for autosomal dominant retinitis pigmentosa. Mutations on the cytoplasmic surface affect transducin activation. J Biol Chem. 1993 May 5;268(13):9400–9404. [PubMed] [Google Scholar]
- Molday R. S., MacKenzie D. Monoclonal antibodies to rhodopsin: characterization, cross-reactivity, and application as structural probes. Biochemistry. 1983 Feb 1;22(3):653–660. doi: 10.1021/bi00272a020. [DOI] [PubMed] [Google Scholar]
- Oprian D. D., Molday R. S., Kaufman R. J., Khorana H. G. Expression of a synthetic bovine rhodopsin gene in monkey kidney cells. Proc Natl Acad Sci U S A. 1987 Dec;84(24):8874–8878. doi: 10.1073/pnas.84.24.8874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridge K. D., Lu Z., Liu X., Khorana H. G. Structure and function in rhodopsin. Separation and characterization of the correctly folded and misfolded opsins produced on expression of an opsin mutant gene containing only the native intradiscal cysteine codons. Biochemistry. 1995 Mar 14;34(10):3261–3267. doi: 10.1021/bi00010a016. [DOI] [PubMed] [Google Scholar]
- Robinson P. R., Cohen G. B., Zhukovsky E. A., Oprian D. D. Constitutively active mutants of rhodopsin. Neuron. 1992 Oct;9(4):719–725. doi: 10.1016/0896-6273(92)90034-b. [DOI] [PubMed] [Google Scholar]
- Saari J. C. The accessibility of bovine rhodopsin in photoreceptor membranes. J Cell Biol. 1974 Nov;63(2 Pt 1):480–491. doi: 10.1083/jcb.63.2.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sung C. H., Davenport C. M., Hennessey J. C., Maumenee I. H., Jacobson S. G., Heckenlively J. R., Nowakowski R., Fishman G., Gouras P., Nathans J. Rhodopsin mutations in autosomal dominant retinitis pigmentosa. Proc Natl Acad Sci U S A. 1991 Aug 1;88(15):6481–6485. doi: 10.1073/pnas.88.15.6481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trayhurn P., Habgood J. O., Virmaux N. The effect of trypsin on retinal rod outer segments: trypsin digestion as a means of isolating viable discs. Exp Eye Res. 1975 May;20(5):479–487. doi: 10.1016/0014-4835(75)90088-3. [DOI] [PubMed] [Google Scholar]
- Vaithinathan R., Berson E. L., Dryja T. P. Further screening of the rhodopsin gene in patients with autosomal dominant retinitis pigmentosa. Genomics. 1994 May 15;21(2):461–463. doi: 10.1006/geno.1994.1301. [DOI] [PubMed] [Google Scholar]
- WALD G., BROWN P. K. The molar extinction of rhodopsin. J Gen Physiol. 1953 Nov 20;37(2):189–200. doi: 10.1085/jgp.37.2.189. [DOI] [PMC free article] [PubMed] [Google Scholar]