Abstract
Cytoplasmic dynein is a multisubunit, microtubule-associated, mechanochemical enzyme that has been identified as a retrograde transporter of various membranous organelles. Dynactin, an additional multisubunit complex, is required for efficient dynein-mediated transport of vesicles in vitro. Recently, we showed that three genes defined by a group of phenotypically identical mutants of the filamentous fungus Neurospora crassa encode proteins that are apparent subunits of either cytoplasmic dynein or dynactin. These mutants, designated ropy (ro), display abnormal hyphal growth and are defective in nuclear distribution. We propose that mutations in other genes encoding dynein/dynactin subunits are likely to result in a ropy phenotype and have devised a genetic screen for the isolation of additional ro mutants. Cytoplasmic dynein/dynactin is the largest and most complex of the cytoplasmic motor proteins, and the genetic system described here is unique in its potentiality for identifying mutations in undefined genes encoding dynein/dynactin subunits or regulators. We used this screen to isolate > 1000 ro mutants, which were found to define 23 complementation groups. Unexpectedly, interallelic complementation was observed with some allele pairs of ro-1 and ro-3, which are predicted to encode the largest subunits of cytoplasmic dynein and dynactin, respectively. The results suggest that the Ro1 and Ro3 polypeptides may consist of multiple, functionally independent domains. In addition, approximately 10% of all newly isolated ro mutantsdisplay unlinked noncomplementation with two or more of the mutants that define the 23 complementation groups. The frequent appearance of ro mutants showing noncomplementation with multiple ro mutants having unlinked mutations suggests that nuclear distribution in filamentous fungi is a process that is easily disrupted by affecting either dosage or activity of cytoplasmic dynein, dynactin, and perhaps other cytoskeletal proteins or regulators.
Full text
PDF

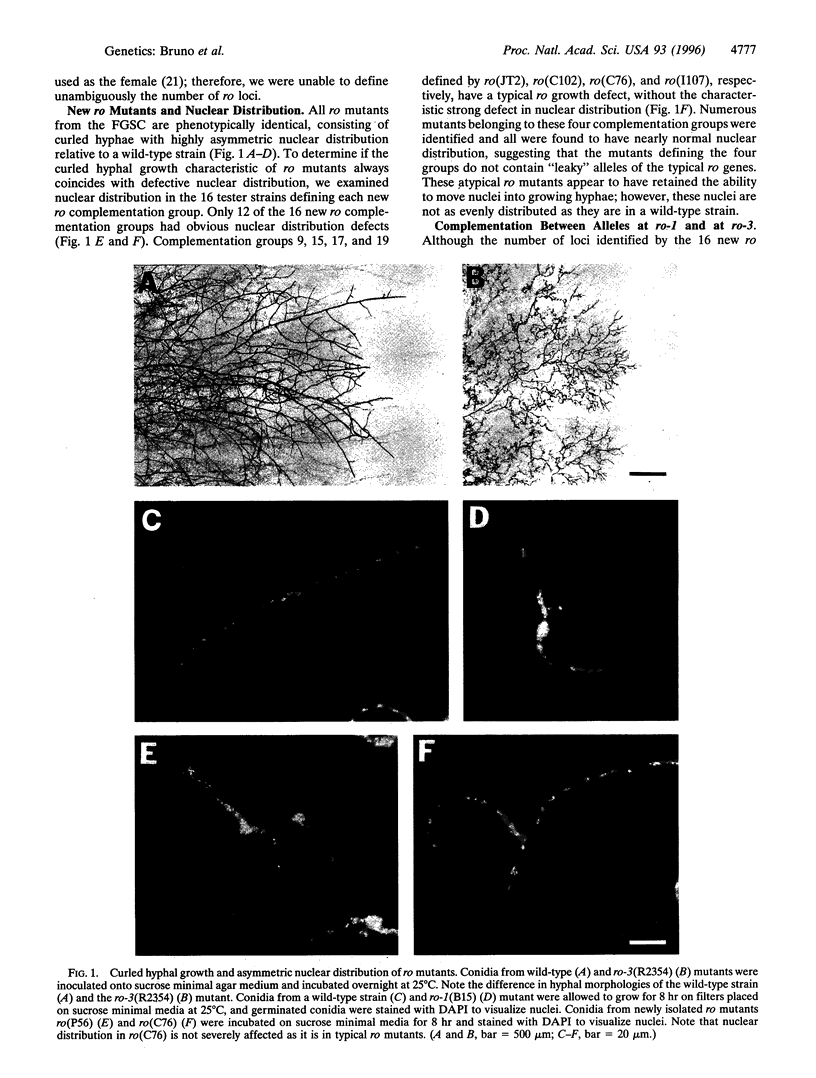

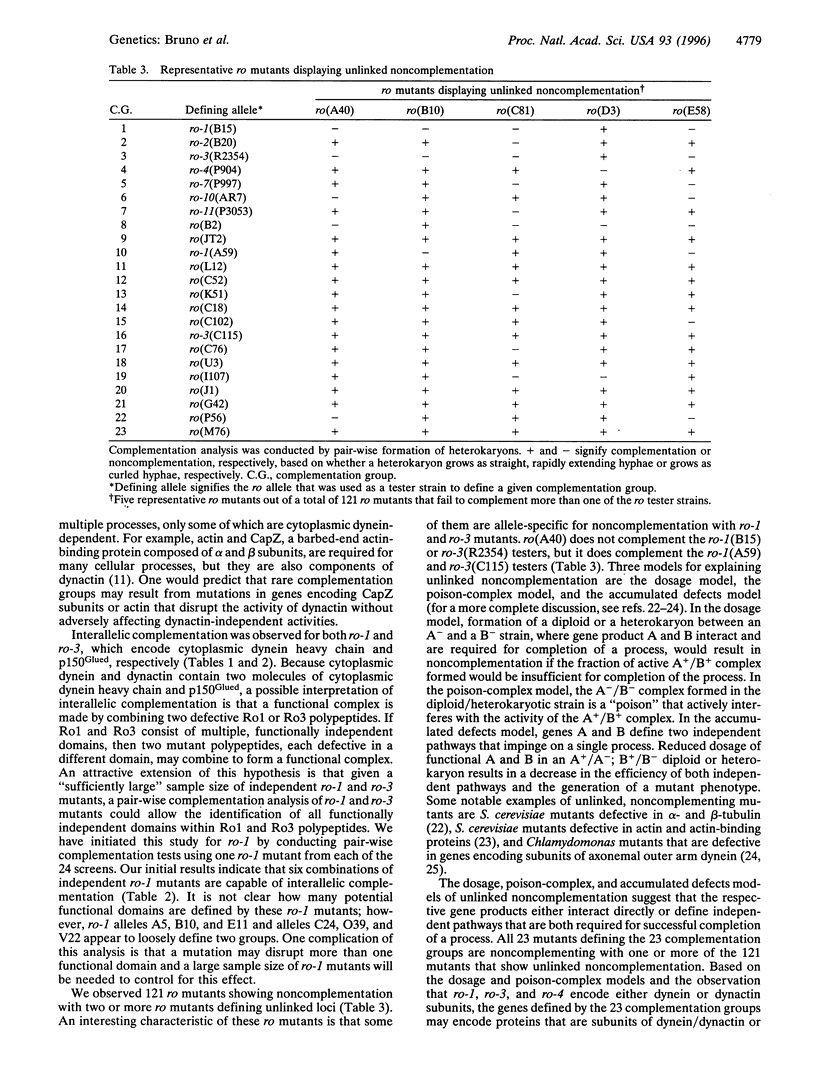

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Clark S. W., Meyer D. I. ACT3: a putative centractin homologue in S. cerevisiae is required for proper orientation of the mitotic spindle. J Cell Biol. 1994 Oct;127(1):129–138. doi: 10.1083/jcb.127.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dutcher S. K., Lux F. G., 3rd Genetic interactions of mutations affecting flagella and basal bodies in Chlamydomonas. Cell Motil Cytoskeleton. 1989;14(1):104–117. doi: 10.1002/cm.970140120. [DOI] [PubMed] [Google Scholar]
- Eshel D., Urrestarazu L. A., Vissers S., Jauniaux J. C., van Vliet-Reedijk J. C., Planta R. J., Gibbons I. R. Cytoplasmic dynein is required for normal nuclear segregation in yeast. Proc Natl Acad Sci U S A. 1993 Dec 1;90(23):11172–11176. doi: 10.1073/pnas.90.23.11172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garen A., Miller B. R., Paco-Larson M. L. Mutations affecting functions of the Drosophila gene glued. Genetics. 1984 Aug;107(4):645–655. doi: 10.1093/genetics/107.4.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garnjobst L., Tatum E. L. A survey of new morphological mutants in Neurospora crassa. Genetics. 1967 Nov;57(3):579–604. doi: 10.1093/genetics/57.3.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill S. R., Schroer T. A., Szilak I., Steuer E. R., Sheetz M. P., Cleveland D. W. Dynactin, a conserved, ubiquitously expressed component of an activator of vesicle motility mediated by cytoplasmic dynein. J Cell Biol. 1991 Dec;115(6):1639–1650. doi: 10.1083/jcb.115.6.1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harte P. J., Kankel D. R. Genetic analysis of mutations at the Glued locus and interacting loci in Drosophila melanogaster. Genetics. 1982 Jul-Aug;101(3-4):477–501. doi: 10.1093/genetics/101.3-4.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holzbaur E. L., Hammarback J. A., Paschal B. M., Kravit N. G., Pfister K. K., Vallee R. B. Homology of a 150K cytoplasmic dynein-associated polypeptide with the Drosophila gene Glued. Nature. 1991 Jun 13;351(6327):579–583. doi: 10.1038/351579a0. [DOI] [PubMed] [Google Scholar]
- Holzbaur E. L., Vallee R. B. DYNEINS: molecular structure and cellular function. Annu Rev Cell Biol. 1994;10:339–372. doi: 10.1146/annurev.cb.10.110194.002011. [DOI] [PubMed] [Google Scholar]
- Kamiya R. Mutations at twelve independent loci result in absence of outer dynein arms in Chylamydomonas reinhardtii. J Cell Biol. 1988 Dec;107(6 Pt 1):2253–2258. doi: 10.1083/jcb.107.6.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y. Y., Yeh E., Hays T., Bloom K. Disruption of mitotic spindle orientation in a yeast dynein mutant. Proc Natl Acad Sci U S A. 1993 Nov 1;90(21):10096–10100. doi: 10.1073/pnas.90.21.10096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muhua L., Karpova T. S., Cooper J. A. A yeast actin-related protein homologous to that in vertebrate dynactin complex is important for spindle orientation and nuclear migration. Cell. 1994 Aug 26;78(4):669–679. doi: 10.1016/0092-8674(94)90531-2. [DOI] [PubMed] [Google Scholar]
- Paschal B. M., Holzbaur E. L., Pfister K. K., Clark S., Meyer D. I., Vallee R. B. Characterization of a 50-kDa polypeptide in cytoplasmic dynein preparations reveals a complex with p150GLUED and a novel actin. J Biol Chem. 1993 Jul 15;268(20):15318–15323. [PubMed] [Google Scholar]
- Perkins D. D., Radford A., Newmeyer D., Björkman M. Chromosomal loci of Neurospora crassa. Microbiol Rev. 1982 Dec;46(4):426–570. doi: 10.1128/mr.46.4.426-570.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plamann M., Minke P. F., Tinsley J. H., Bruno K. S. Cytoplasmic dynein and actin-related protein Arp1 are required for normal nuclear distribution in filamentous fungi. J Cell Biol. 1994 Oct;127(1):139–149. doi: 10.1083/jcb.127.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders W. S., Koshland D., Eshel D., Gibbons I. R., Hoyt M. A. Saccharomyces cerevisiae kinesin- and dynein-related proteins required for anaphase chromosome segregation. J Cell Biol. 1995 Feb;128(4):617–624. doi: 10.1083/jcb.128.4.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer D. A., Gill S. R., Cooper J. A., Heuser J. E., Schroer T. A. Ultrastructural analysis of the dynactin complex: an actin-related protein is a component of a filament that resembles F-actin. J Cell Biol. 1994 Jul;126(2):403–412. doi: 10.1083/jcb.126.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroer T. A., Sheetz M. P. Two activators of microtubule-based vesicle transport. J Cell Biol. 1991 Dec;115(5):1309–1318. doi: 10.1083/jcb.115.5.1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stearns T., Botstein D. Unlinked noncomplementation: isolation of new conditional-lethal mutations in each of the tubulin genes of Saccharomyces cerevisiae. Genetics. 1988 Jun;119(2):249–260. doi: 10.1093/genetics/119.2.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaisberg E. A., Koonce M. P., McIntosh J. R. Cytoplasmic dynein plays a role in mammalian mitotic spindle formation. J Cell Biol. 1993 Nov;123(4):849–858. doi: 10.1083/jcb.123.4.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vinh D. B., Welch M. D., Corsi A. K., Wertman K. F., Drubin D. G. Genetic evidence for functional interactions between actin noncomplementing (Anc) gene products and actin cytoskeletal proteins in Saccharomyces cerevisiae. Genetics. 1993 Oct;135(2):275–286. doi: 10.1093/genetics/135.2.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker R. A., Sheetz M. P. Cytoplasmic microtubule-associated motors. Annu Rev Biochem. 1993;62:429–451. doi: 10.1146/annurev.bi.62.070193.002241. [DOI] [PubMed] [Google Scholar]
- Xiang X., Beckwith S. M., Morris N. R. Cytoplasmic dynein is involved in nuclear migration in Aspergillus nidulans. Proc Natl Acad Sci U S A. 1994 Mar 15;91(6):2100–2104. doi: 10.1073/pnas.91.6.2100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yarden O., Plamann M., Ebbole D. J., Yanofsky C. cot-1, a gene required for hyphal elongation in Neurospora crassa, encodes a protein kinase. EMBO J. 1992 Jun;11(6):2159–2166. doi: 10.1002/j.1460-2075.1992.tb05275.x. [DOI] [PMC free article] [PubMed] [Google Scholar]



