Abstract
The conventional approach to cytotoxic T-lymphocyte (CTL) induction uses maximal antigen concentration with the intent of eliciting more CTL. However, the efficacy of this approach has not been systematically explored with regard to the quality of the CTLs elicited or their in vivo functionality. Here, we show that a diametrically opposite approach elicits CTLs that are much more effective at clearing virus. CTLs specific for a defined peptide epitope were selectively expanded with various concentrations of peptide antigen. CTLs generated with exceedingly low-dose peptide lysed targets sensitized with > 100-fold less peptide than CTLs generated with high-dose peptide. Differences in expression of T-cell antigen receptors or a number of other accessory molecules did not account for the functional differences. Further, high-avidity CTLs adoptively transferred into severe combined immunodeficient mice were 100- to 1000-fold more effective at viral clearance than the low-avidity CTLs, despite the fact that all CTL lines lysed virus-infected targets in vitro. Thus, the quality of CTLs is as important as the quantity of CTLs for adoptive immunotherapy, and the ability to kill virally infected targets in vitro is not predictive of in vivo efficacy, whereas the determinant density requirement described here is predictive. Application of these principles may be critical in developing effective adoptive cellular immunotherapy for viral infections and cancer.
Full text
PDF
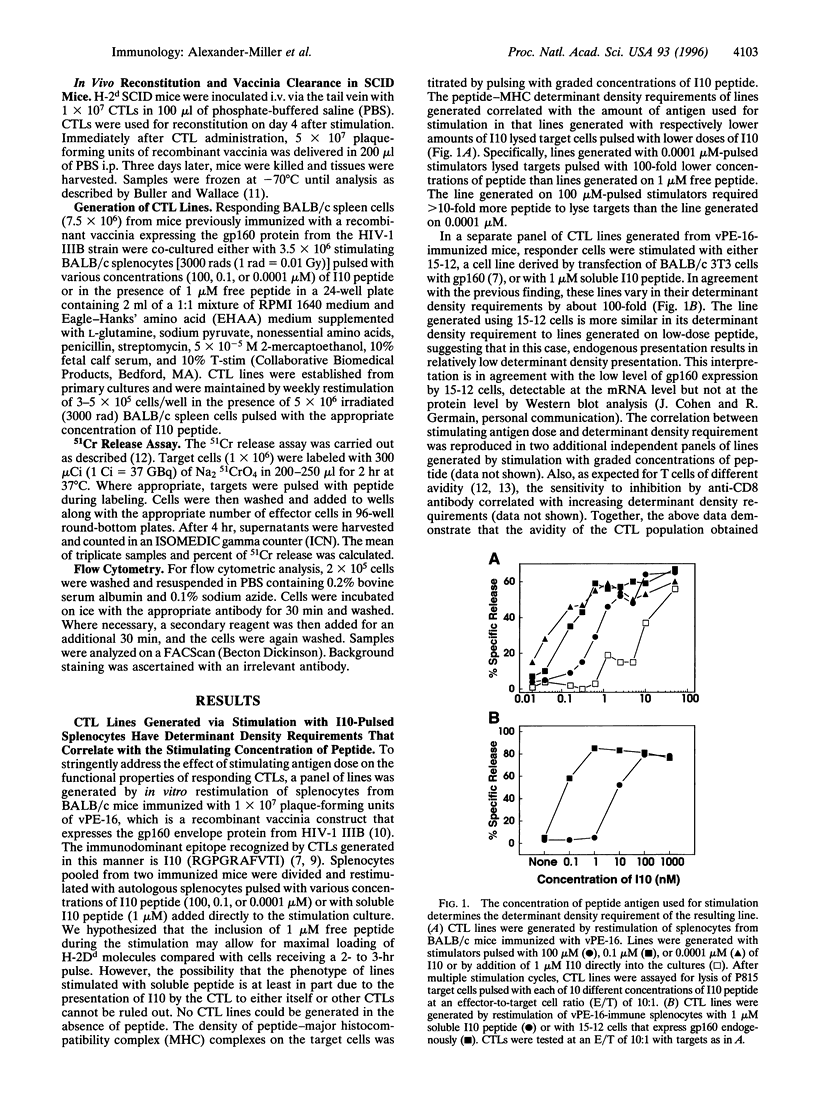
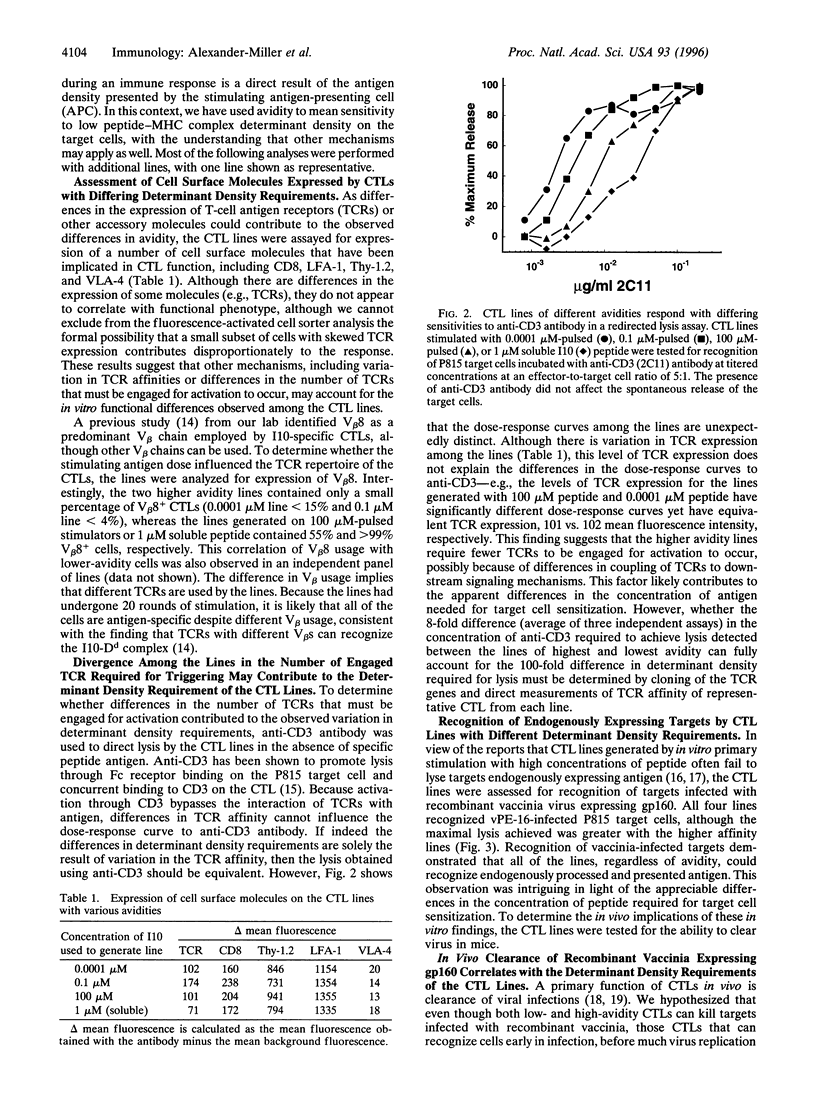
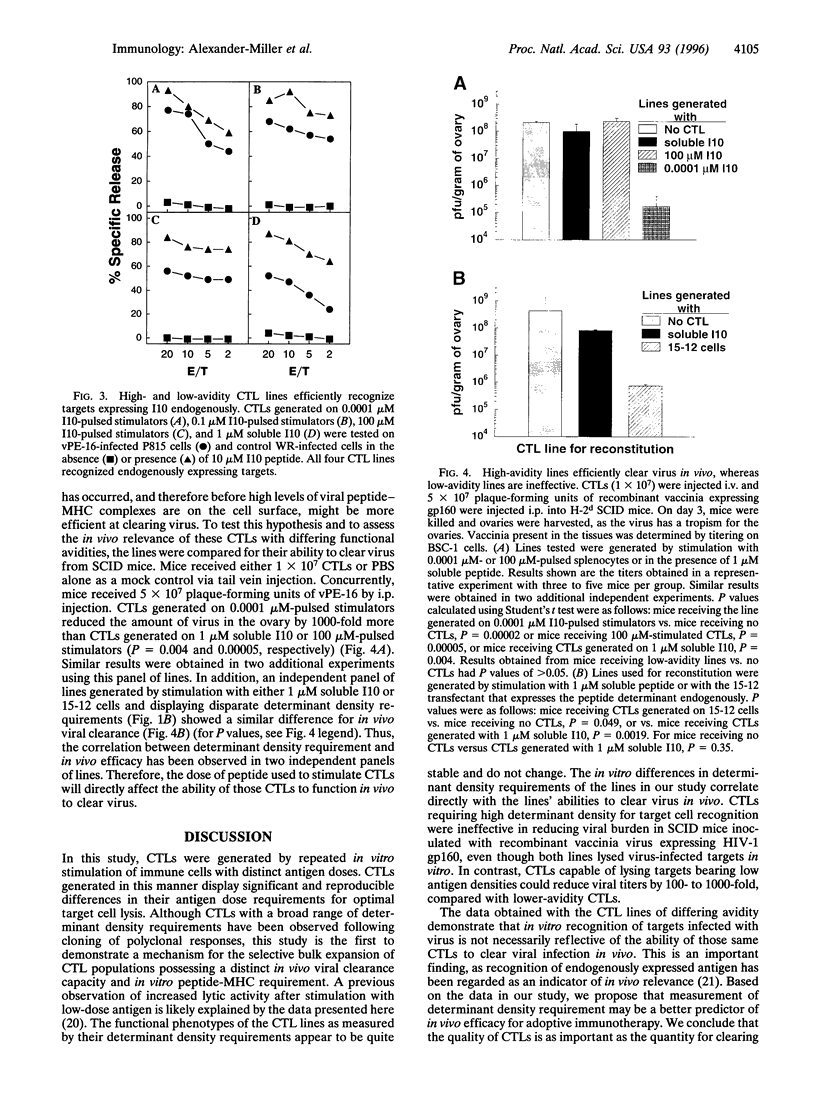


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander M. A., Damico C. A., Wieties K. M., Hansen T. H., Connolly J. M. Correlation between CD8 dependency and determinant density using peptide-induced, Ld-restricted cytotoxic T lymphocytes. J Exp Med. 1991 Apr 1;173(4):849–858. doi: 10.1084/jem.173.4.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buller R. M., Wallace G. D. Reexamination of the efficacy of vaccination against mousepox. Lab Anim Sci. 1985 Oct;35(5):473–476. [PubMed] [Google Scholar]
- Byrne J. A., Oldstone M. B. Biology of cloned cytotoxic T lymphocytes specific for lymphocytic choriomeningitis virus: clearance of virus in vivo. J Virol. 1984 Sep;51(3):682–686. doi: 10.1128/jvi.51.3.682-686.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbone F. R., Moore M. W., Sheil J. M., Bevan M. J. Induction of cytotoxic T lymphocytes by primary in vitro stimulation with peptides. J Exp Med. 1988 Jun 1;167(6):1767–1779. doi: 10.1084/jem.167.6.1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Earl P. L., Koenig S., Moss B. Biological and immunological properties of human immunodeficiency virus type 1 envelope glycoprotein: analysis of proteins with truncations and deletions expressed by recombinant vaccinia viruses. J Virol. 1991 Jan;65(1):31–41. doi: 10.1128/jvi.65.1.31-41.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houbiers J. G., Nijman H. W., van der Burg S. H., Drijfhout J. W., Kenemans P., van de Velde C. J., Brand A., Momburg F., Kast W. M., Melief C. J. In vitro induction of human cytotoxic T lymphocyte responses against peptides of mutant and wild-type p53. Eur J Immunol. 1993 Sep;23(9):2072–2077. doi: 10.1002/eji.1830230905. [DOI] [PubMed] [Google Scholar]
- Jackson D. C., Ada G. L., Tha Hla R. Cytotoxic T cells recognize very early, minor changes in ectromelia virus-infected target cells. Aust J Exp Biol Med Sci. 1976 Aug;54(4):349–363. doi: 10.1038/icb.1976.35. [DOI] [PubMed] [Google Scholar]
- Kawakami Y., Eliyahu S., Delgado C. H., Robbins P. F., Rivoltini L., Topalian S. L., Miki T., Rosenberg S. A. Cloning of the gene coding for a shared human melanoma antigen recognized by autologous T cells infiltrating into tumor. Proc Natl Acad Sci U S A. 1994 Apr 26;91(9):3515–3519. doi: 10.1073/pnas.91.9.3515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenig S., Conley A. J., Brewah Y. A., Jones G. M., Leath S., Boots L. J., Davey V., Pantaleo G., Demarest J. F., Carter C. Transfer of HIV-1-specific cytotoxic T lymphocytes to an AIDS patient leads to selection for mutant HIV variants and subsequent disease progression. Nat Med. 1995 Apr;1(4):330–336. doi: 10.1038/nm0495-330. [DOI] [PubMed] [Google Scholar]
- Kwan-Lim G. E., Ong T., Aosai F., Stauss H., Zamoyska R. Is CD8 dependence a true reflection of TCR affinity for antigen? Int Immunol. 1993 Oct;5(10):1219–1228. doi: 10.1093/intimm/5.10.1219. [DOI] [PubMed] [Google Scholar]
- Leo O., Foo M., Sachs D. H., Samelson L. E., Bluestone J. A. Identification of a monoclonal antibody specific for a murine T3 polypeptide. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1374–1378. doi: 10.1073/pnas.84.5.1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maryanski J. L., Pala P., Cerottini J. C., MacDonald H. R. Antigen recognition by H-2-restricted cytolytic T lymphocytes: inhibition of cytolysis by anti-CD8 monoclonal antibodies depends upon both concentration and primary sequence of peptide antigen. Eur J Immunol. 1988 Nov;18(11):1863–1866. doi: 10.1002/eji.1830181135. [DOI] [PubMed] [Google Scholar]
- Riddell S. R., Greenberg P. D., Overell R. W., Loughran T. P., Gilbert M. J., Lupton S. D., Agosti J., Scheeler S., Coombs R. W., Corey L. Phase I study of cellular adoptive immunotherapy using genetically modified CD8+ HIV-specific T cells for HIV seropositive patients undergoing allogeneic bone marrow transplant. The Fred Hutchinson Cancer Research Center and the University of Washington School of Medicine, Department of Medicine, Division of Oncology. Hum Gene Ther. 1992 Jun;3(3):319–338. doi: 10.1089/hum.1992.3.3-319. [DOI] [PubMed] [Google Scholar]
- Riddell S. R., Watanabe K. S., Goodrich J. M., Li C. R., Agha M. E., Greenberg P. D. Restoration of viral immunity in immunodeficient humans by the adoptive transfer of T cell clones. Science. 1992 Jul 10;257(5067):238–241. doi: 10.1126/science.1352912. [DOI] [PubMed] [Google Scholar]
- Rodrigues M., Nussenzweig R. S., Romero P., Zavala F. The in vivo cytotoxic activity of CD8+ T cell clones correlates with their levels of expression of adhesion molecules. J Exp Med. 1992 Apr 1;175(4):895–905. doi: 10.1084/jem.175.4.895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg S. A., Packard B. S., Aebersold P. M., Solomon D., Topalian S. L., Toy S. T., Simon P., Lotze M. T., Yang J. C., Seipp C. A. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N Engl J Med. 1988 Dec 22;319(25):1676–1680. doi: 10.1056/NEJM198812223192527. [DOI] [PubMed] [Google Scholar]
- Shirai M., Vacchio M. S., Hodes R. J., Berzofsky J. A. Preferential V beta usage by cytotoxic T cells cross-reactive between two epitopes of HIV-1 gp160 and degenerate in class I MHC restriction. J Immunol. 1993 Aug 15;151(4):2283–2295. [PubMed] [Google Scholar]
- Speiser D. E., Kyburz D., Stübi U., Hengartner H., Zinkernagel R. M. Discrepancy between in vitro measurable and in vivo virus neutralizing cytotoxic T cell reactivities. Low T cell receptor specificity and avidity sufficient for in vitro proliferation or cytotoxicity to peptide-coated target cells but not for in vivo protection. J Immunol. 1992 Aug 1;149(3):972–980. [PubMed] [Google Scholar]
- Staerz U. D., Zepp F., Schmid R., Hill M., Rothbard J. Recruitment of alloreactive cytotoxic T lymphocytes by an antigenic peptide. Eur J Immunol. 1989 Dec;19(12):2191–2196. doi: 10.1002/eji.1830191203. [DOI] [PubMed] [Google Scholar]
- Takahashi H., Cohen J., Hosmalin A., Cease K. B., Houghten R., Cornette J. L., DeLisi C., Moss B., Germain R. N., Berzofsky J. A. An immunodominant epitope of the human immunodeficiency virus envelope glycoprotein gp160 recognized by class I major histocompatibility complex molecule-restricted murine cytotoxic T lymphocytes. Proc Natl Acad Sci U S A. 1988 May;85(9):3105–3109. doi: 10.1073/pnas.85.9.3105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeshita T., Takahashi H., Kozlowski S., Ahlers J. D., Pendleton C. D., Moore R. L., Nakagawa Y., Yokomuro K., Fox B. S., Margulies D. H. Molecular analysis of the same HIV peptide functionally binding to both a class I and a class II MHC molecule. J Immunol. 1995 Feb 15;154(4):1973–1986. [PubMed] [Google Scholar]
- Taylor P. M., Askonas B. A. Influenza nucleoprotein-specific cytotoxic T-cell clones are protective in vivo. Immunology. 1986 Jul;58(3):417–420. [PMC free article] [PubMed] [Google Scholar]
- Topalian S. L., Muul L. M., Solomon D., Rosenberg S. A. Expansion of human tumor infiltrating lymphocytes for use in immunotherapy trials. J Immunol Methods. 1987 Aug 24;102(1):127–141. doi: 10.1016/s0022-1759(87)80018-2. [DOI] [PubMed] [Google Scholar]
- Yannelli J. R. The preparation of effector cells for use in the adoptive cellular immunotherapy of human cancer. J Immunol Methods. 1991 May 17;139(1):1–16. doi: 10.1016/0022-1759(91)90345-g. [DOI] [PubMed] [Google Scholar]
- Zhou X., Berg L., Motal U. M., Jondal M. In vivo primary induction of virus-specific CTL by immunization with 9-mer synthetic peptides. J Immunol Methods. 1992 Aug 30;153(1-2):193–200. doi: 10.1016/0022-1759(92)90322-k. [DOI] [PubMed] [Google Scholar]
- Zinkernagel R. M., Althage A. Antiviral protection by virus-immune cytotoxic T cells: infected target cells are lysed before infectious virus progeny is assembled. J Exp Med. 1977 Mar 1;145(3):644–651. doi: 10.1084/jem.145.3.644. [DOI] [PMC free article] [PubMed] [Google Scholar]