Abstract
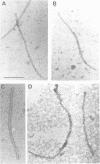
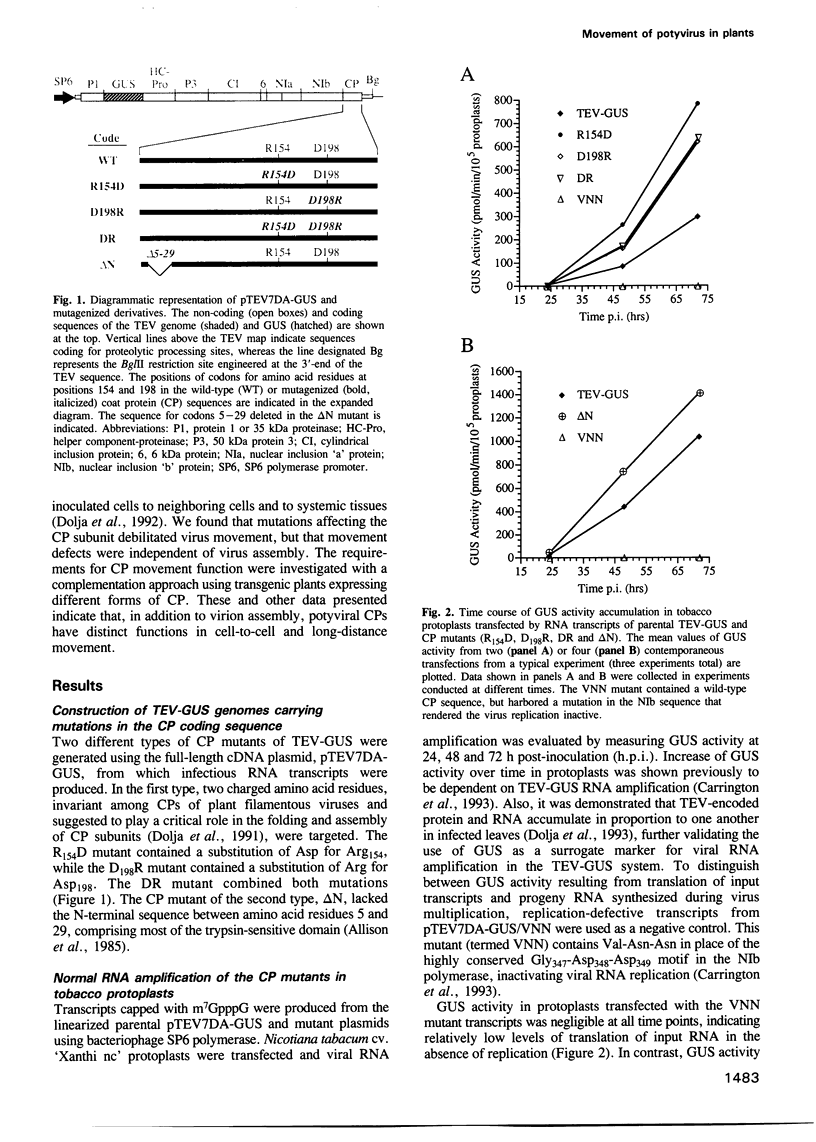
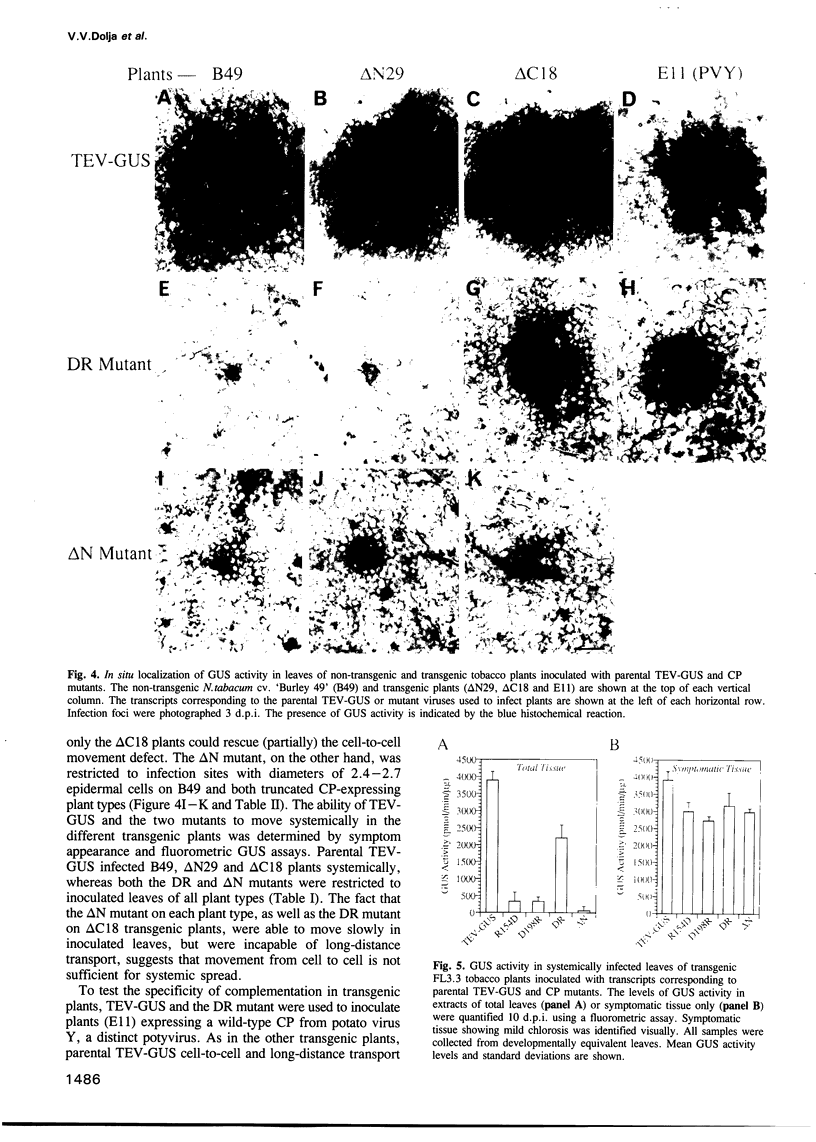
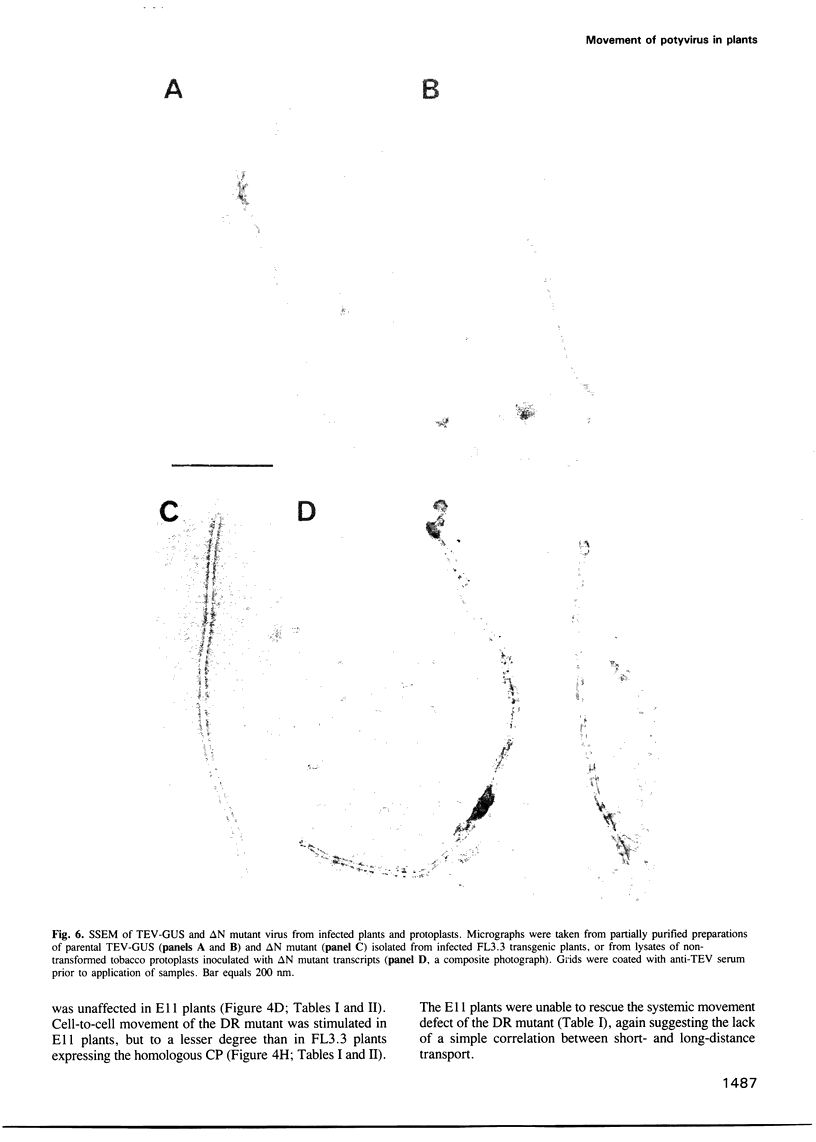
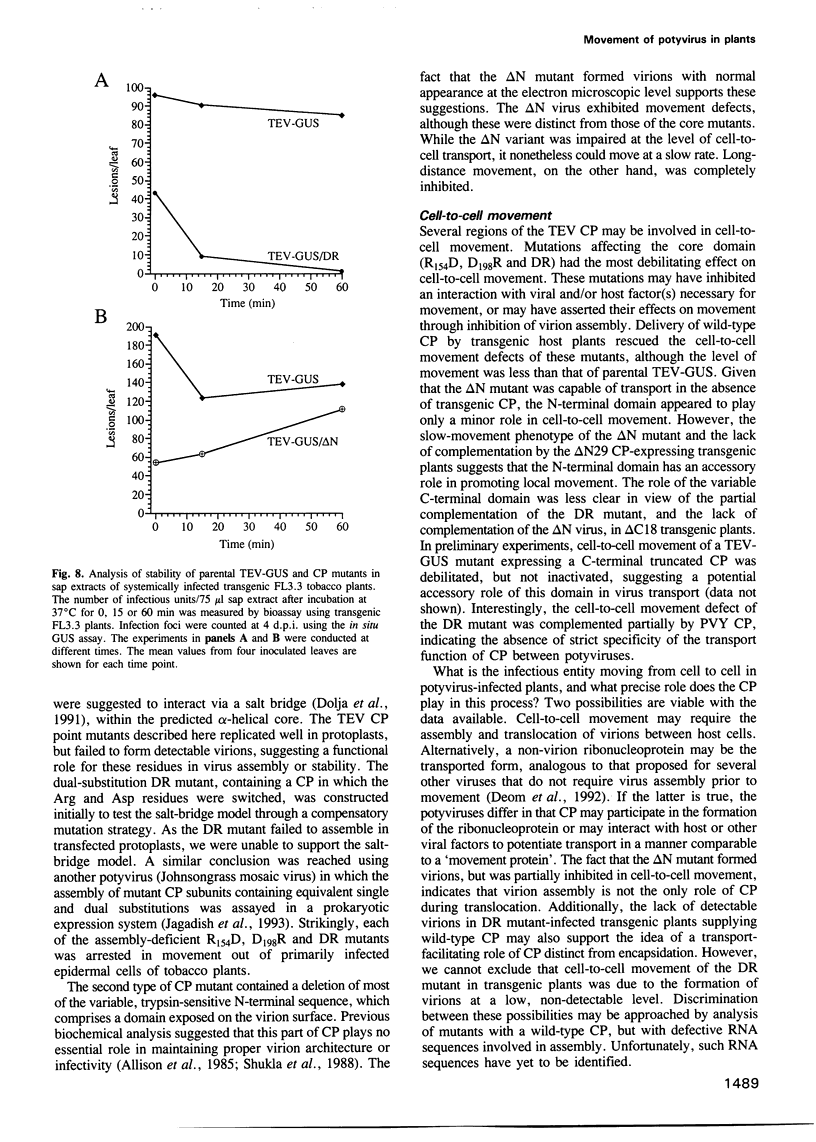
Tobacco etch potyvirus engineered to express the reporter protein beta-glucuronidase (TEV-GUS) was used for direct observation and quantitation of virus translocation in plants. Four TEV-GUS mutants were generated containing capsid proteins (CPs) with single amino acid substitutions (R154D and D198R), a double substitution (DR), or a deletion of part of the N-terminal domain (delta N). Each modified virus replicated as well as the parental virus in protoplasts, but was defective in cell-to-cell movement through inoculated leaves. The R154D, D198R and DR mutants were restricted essentially to single, initially infected cells. The delta N variant exhibited slow cell-to-cell movement in inoculated leaves, but was unable to move systemically due to a lack of entry into or replication in vascular-associated cells. Both cell-to-cell and systemic movement defects of each mutant were rescued in transgenic plants expressing wild-type TEV CP. Cell-to-cell movement, but not systemic movement, of the DR mutant was rescued partially in transgenic plants expressing TEV CP lacking the C-terminal domain, and in plants expressing CP from the heterologous potyvirus, potato virus Y. Despite comparable levels of accumulation of parental virus and each mutant in symptomatic tissue of TEV CP-expressing transgenic plants, virions were detected only in parental virus- and delta N mutant-infected plants, as revealed using three independent assays. These data suggest that the potyvirus CP possesses distinct, separable activities required for virion assembly, cell-to-cell movement and long-distance transport.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Atabekov J. G., Taliansky M. E. Expression of a plant virus-coded transport function by different viral genomes. Adv Virus Res. 1990;38:201–248. doi: 10.1016/s0065-3527(08)60863-5. [DOI] [PubMed] [Google Scholar]
- Atreya P. L., Atreya C. D., Pirone T. P. Amino acid substitutions in the coat protein result in loss of insect transmissibility of a plant virus. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7887–7891. doi: 10.1073/pnas.88.17.7887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Freed D. D. Cap-independent enhancement of translation by a plant potyvirus 5' nontranslated region. J Virol. 1990 Apr;64(4):1590–1597. doi: 10.1128/jvi.64.4.1590-1597.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrington J. C., Haldeman R., Dolja V. V., Restrepo-Hartwig M. A. Internal cleavage and trans-proteolytic activities of the VPg-proteinase (NIa) of tobacco etch potyvirus in vivo. J Virol. 1993 Dec;67(12):6995–7000. doi: 10.1128/jvi.67.12.6995-7000.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman S., Hills G., Watts J., Baulcombe D. Mutational analysis of the coat protein gene of potato virus X: effects on virion morphology and viral pathogenicity. Virology. 1992 Nov;191(1):223–230. doi: 10.1016/0042-6822(92)90183-p. [DOI] [PubMed] [Google Scholar]
- De Jong W., Ahlquist P. A hybrid plant RNA virus made by transferring the noncapsid movement protein from a rod-shaped to an icosahedral virus is competent for systemic infection. Proc Natl Acad Sci U S A. 1992 Aug 1;89(15):6808–6812. doi: 10.1073/pnas.89.15.6808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deom C. M., Lapidot M., Beachy R. N. Plant virus movement proteins. Cell. 1992 Apr 17;69(2):221–224. doi: 10.1016/0092-8674(92)90403-y. [DOI] [PubMed] [Google Scholar]
- Deom C. M., Oliver M. J., Beachy R. N. The 30-kilodalton gene product of tobacco mosaic virus potentiates virus movement. Science. 1987 Jul 24;237(4813):389–394. doi: 10.1126/science.237.4813.389. [DOI] [PubMed] [Google Scholar]
- Derrick K. S. Quantitative assay for plant viruses using serologically specific electron microscopy. Virology. 1973 Dec;56(2):652–653. doi: 10.1016/0042-6822(73)90068-8. [DOI] [PubMed] [Google Scholar]
- Dolja V. V., Boyko V. P., Agranovsky A. A., Koonin E. V. Phylogeny of capsid proteins of rod-shaped and filamentous RNA plant viruses: two families with distinct patterns of sequence and probably structure conservation. Virology. 1991 Sep;184(1):79–86. doi: 10.1016/0042-6822(91)90823-t. [DOI] [PubMed] [Google Scholar]
- Dolja V. V., Herndon K. L., Pirone T. P., Carrington J. C. Spontaneous mutagenesis of a plant potyvirus genome after insertion of a foreign gene. J Virol. 1993 Oct;67(10):5968–5975. doi: 10.1128/jvi.67.10.5968-5975.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolja V. V., McBride H. J., Carrington J. C. Tagging of plant potyvirus replication and movement by insertion of beta-glucuronidase into the viral polyprotein. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10208–10212. doi: 10.1073/pnas.89.21.10208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domier L. L., Franklin K. M., Shahabuddin M., Hellmann G. M., Overmeyer J. H., Hiremath S. T., Siaw M. F., Lomonossoff G. P., Shaw J. G., Rhoads R. E. The nucleotide sequence of tobacco vein mottling virus RNA. Nucleic Acids Res. 1986 Jul 11;14(13):5417–5430. doi: 10.1093/nar/14.13.5417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forster R. L., Beck D. L., Guilford P. J., Voot D. M., Van Dolleweerd C. J., Andersen M. T. The coat protein of white clover mosaic potexvirus has a role in facilitating cell-to-cell transport in plants. Virology. 1992 Nov;191(1):480–484. doi: 10.1016/0042-6822(92)90215-b. [DOI] [PubMed] [Google Scholar]
- Jagadish M. N., Huang D., Ward C. W. Site-directed mutagenesis of a potyvirus coat protein and its assembly in Escherichia coli. J Gen Virol. 1993 May;74(Pt 5):893–896. doi: 10.1099/0022-1317-74-5-893. [DOI] [PubMed] [Google Scholar]
- Kasteel D., Wellink J., Verver J., van Lent J., Goldbach R., van Kammen A. The involvement of cowpea mosaic virus M RNA-encoded proteins in tubule formation. J Gen Virol. 1993 Aug;74(Pt 8):1721–1724. doi: 10.1099/0022-1317-74-8-1721. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A., Roberts J. D., Zakour R. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Methods Enzymol. 1987;154:367–382. doi: 10.1016/0076-6879(87)54085-x. [DOI] [PubMed] [Google Scholar]
- Lindbo J. A., Dougherty W. G. Pathogen-derived resistance to a potyvirus: immune and resistant phenotypes in transgenic tobacco expressing altered forms of a potyvirus coat protein nucleotide sequence. Mol Plant Microbe Interact. 1992 Mar-Apr;5(2):144–153. doi: 10.1094/mpmi-5-144. [DOI] [PubMed] [Google Scholar]
- Lindbo J. A., Dougherty W. G. Untranslatable transcripts of the tobacco etch virus coat protein gene sequence can interfere with tobacco etch virus replication in transgenic plants and protoplasts. Virology. 1992 Aug;189(2):725–733. doi: 10.1016/0042-6822(92)90595-g. [DOI] [PubMed] [Google Scholar]
- Oh C. S., Carrington J. C. Identification of essential residues in potyvirus proteinase HC-Pro by site-directed mutagenesis. Virology. 1989 Dec;173(2):692–699. doi: 10.1016/0042-6822(89)90582-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petty I. T., French R., Jones R. W., Jackson A. O. Identification of barley stripe mosaic virus genes involved in viral RNA replication and systemic movement. EMBO J. 1990 Nov;9(11):3453–3457. doi: 10.1002/j.1460-2075.1990.tb07553.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Restrepo M. A., Freed D. D., Carrington J. C. Nuclear transport of plant potyviral proteins. Plant Cell. 1990 Oct;2(10):987–998. doi: 10.1105/tpc.2.10.987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riechmann J. L., Laín S., García J. A. Highlights and prospects of potyvirus molecular biology. J Gen Virol. 1992 Jan;73(Pt 1):1–16. doi: 10.1099/0022-1317-73-1-1. [DOI] [PubMed] [Google Scholar]
- Saito T., Yamanaka K., Okada Y. Long-distance movement and viral assembly of tobacco mosaic virus mutants. Virology. 1990 Jun;176(2):329–336. doi: 10.1016/0042-6822(90)90002-9. [DOI] [PubMed] [Google Scholar]
- Takamatsu N., Ishikawa M., Meshi T., Okada Y. Expression of bacterial chloramphenicol acetyltransferase gene in tobacco plants mediated by TMV-RNA. EMBO J. 1987 Feb;6(2):307–311. doi: 10.1002/j.1460-2075.1987.tb04755.x. [DOI] [PMC free article] [PubMed] [Google Scholar]