Abstract
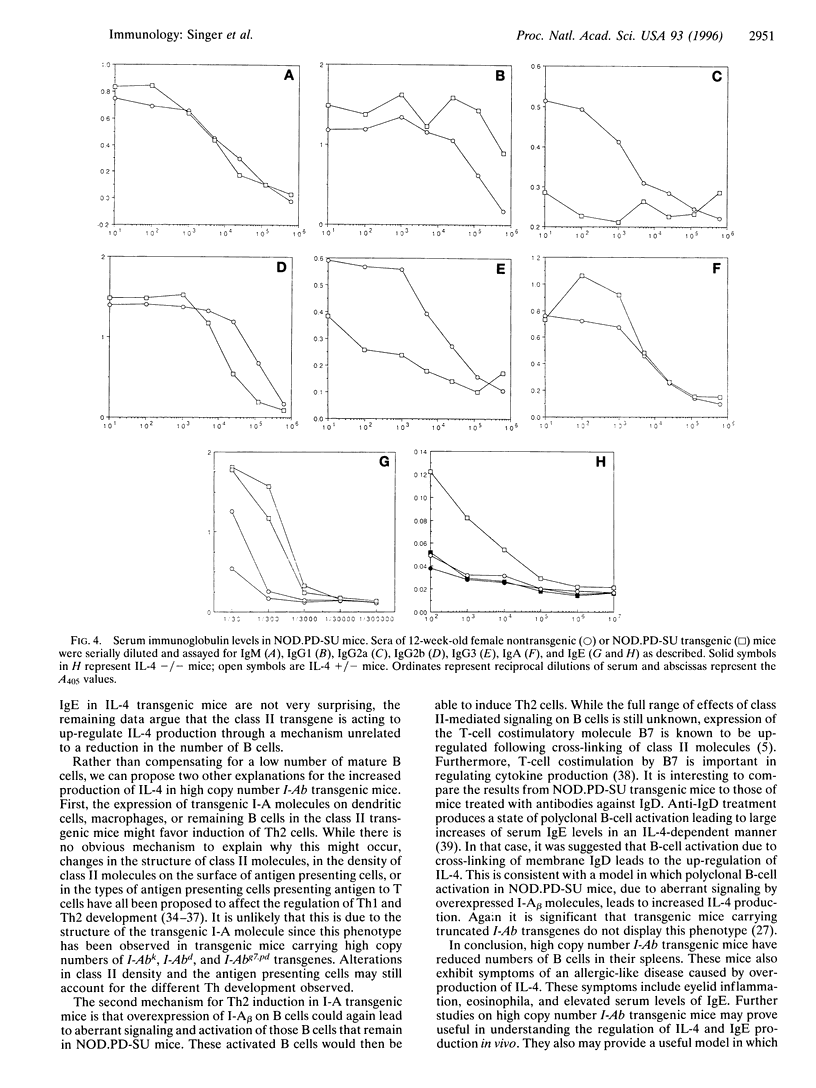
To better understand the role of class II major histocompatibility complex molecules in both normal and autoimmune responses, we have produced a series of I-Ab transgenic mice. One of these transgenic constructs, designated NOD.PD, has the sequence of the NOD beta chain (Abeta(g7)) except at positions 56 and 57, where Pro-Asp replaces His-Ser. Several NOD.PD transgenic lines have been produced. One line of these mice carried a very high number of copies (>50) of the NOD.PD transgene. As has been described in other mice carrying high copy numbers of I-Ab transgenes, B-cell development was abnormal. The steady state numbers of mature B cells (IgM+/IgD(hi)) in the periphery were greatly reduced in transgenic mice compared to nontransgenic littermates. Surprisingly, rather than being accompanied by a generalized hypogammaglobulinemia, this B-cell deficiency was accompanied by elevated concentrations of IgG1 and IgE in the serum. Conversely, the levels of IgG2a were reduced in transgenic mice compared to nontransgenic littermates. Because this isotype pattern was characteristic of interleukin (IL)-4-induced class-switching, we then investigated the role of IL-4 in causing the observed phenotype. We crossed the high copy number transgenic mice with an IL-4-deficient strain of mice. As expected, the elevated levels of IgE in high copy number transgenic mice were eliminated when the IL-4 gene was inactivated. However, the reduction in the number of B cells was not ameliorated. These data indicate that the primary defect caused by the transgene was to reduce the number of B cells in these mice. This reduction was accompanied by a secondary increase in IL-4 production, which drove the remaining B cells toward the production of IgGl and IgE.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baniyash M., Eshhar Z. Inhibition of IgE binding to mast cells and basophils by monoclonal antibodies to murine IgE. Eur J Immunol. 1984 Sep;14(9):799–807. doi: 10.1002/eji.1830140907. [DOI] [PubMed] [Google Scholar]
- Benoist C., Mathis D. Regulation of major histocompatibility complex class-II genes: X, Y and other letters of the alphabet. Annu Rev Immunol. 1990;8:681–715. doi: 10.1146/annurev.iy.08.040190.003341. [DOI] [PubMed] [Google Scholar]
- Bishop G. A., Haughton G. Induced differentiation of a transformed clone of Ly-1+ B cells by clonal T cells and antigen. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7410–7414. doi: 10.1073/pnas.83.19.7410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosma G. C., Custer R. P., Bosma M. J. A severe combined immunodeficiency mutation in the mouse. Nature. 1983 Feb 10;301(5900):527–530. doi: 10.1038/301527a0. [DOI] [PubMed] [Google Scholar]
- Bottomly K. A functional dichotomy in CD4+ T lymphocytes. Immunol Today. 1988 Sep;9(9):268–274. doi: 10.1016/0167-5699(88)91308-4. [DOI] [PubMed] [Google Scholar]
- Buus S., Sette A., Colon S. M., Miles C., Grey H. M. The relation between major histocompatibility complex (MHC) restriction and the capacity of Ia to bind immunogenic peptides. Science. 1987 Mar 13;235(4794):1353–1358. doi: 10.1126/science.2435001. [DOI] [PubMed] [Google Scholar]
- Cambier J. C., Lehmann K. R. Ia-mediated signal transduction leads to proliferation of primed B lymphocytes. J Exp Med. 1989 Sep 1;170(3):877–886. doi: 10.1084/jem.170.3.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cambier J. C., Newell M. K., Justement L. B., McGuire J. C., Leach K. L., Chen Z. Z. Ia binding ligands and cAMP stimulate nuclear translocation of PKC in B lymphocytes. Nature. 1987 Jun 18;327(6123):629–632. doi: 10.1038/327629a0. [DOI] [PubMed] [Google Scholar]
- Cerny A., Heusser C., Sutter S., Huegin A. W., Bazin H., Hengartner H., Zinkernagel R. M. Generation of agammaglobulinaemic mice by prenatal and postnatal exposure to polyclonal or monoclonal anti-IgM antibodies. Scand J Immunol. 1986 Oct;24(4):437–445. doi: 10.1111/j.1365-3083.1986.tb02132.x. [DOI] [PubMed] [Google Scholar]
- Chen J., Trounstine M., Alt F. W., Young F., Kurahara C., Loring J. F., Huszar D. Immunoglobulin gene rearrangement in B cell deficient mice generated by targeted deletion of the JH locus. Int Immunol. 1993 Jun;5(6):647–656. doi: 10.1093/intimm/5.6.647. [DOI] [PubMed] [Google Scholar]
- Clare-Salzler M. J., Brooks J., Chai A., Van Herle K., Anderson C. Prevention of diabetes in nonobese diabetic mice by dendritic cell transfer. J Clin Invest. 1992 Sep;90(3):741–748. doi: 10.1172/JCI115946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coffman R. L., Seymour B. W., Lebman D. A., Hiraki D. D., Christiansen J. A., Shrader B., Cherwinski H. M., Savelkoul H. F., Finkelman F. D., Bond M. W. The role of helper T cell products in mouse B cell differentiation and isotype regulation. Immunol Rev. 1988 Feb;102:5–28. doi: 10.1111/j.1600-065x.1988.tb00739.x. [DOI] [PubMed] [Google Scholar]
- DeKruyff R. H., Fang Y., Umetsu D. T. IL-4 synthesis by in vivo primed keyhole limpet hemocyanin-specific CD4+ T cells. I. Influence of antigen concentration and antigen-presenting cell type. J Immunol. 1992 Dec 1;149(11):3468–3476. [PubMed] [Google Scholar]
- Finkelman F. D., Snapper C. M., Mountz J. D., Katona I. M. Polyclonal activation of the murine immune system by a goat antibody to mouse IgD. IX. Induction of a polyclonal IgE response. J Immunol. 1987 May 1;138(9):2826–2830. [PubMed] [Google Scholar]
- Forsgren S., Pobor G., Coutinho A., Pierres M. The role of I-A/E molecules in B lymphocyte activation. I. Inhibition of lipopolysaccharide-induced responses by monoclonal antibodies. J Immunol. 1984 Oct;133(4):2104–2110. [PubMed] [Google Scholar]
- Gajewski T. F., Pinnas M., Wong T., Fitch F. W. Murine Th1 and Th2 clones proliferate optimally in response to distinct antigen-presenting cell populations. J Immunol. 1991 Mar 15;146(6):1750–1758. [PubMed] [Google Scholar]
- Gilfillan S., Aiso S., Michie S. A., McDevitt H. O. Immune deficiency due to high copy numbers of an Ak beta transgene. Proc Natl Acad Sci U S A. 1990 Sep;87(18):7319–7323. doi: 10.1073/pnas.87.18.7319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilfillan S., Aiso S., Michie S. A., McDevitt H. O. The effect of excess beta-chain synthesis on cell-surface expression of allele-mismatched class II heterodimers in vivo. Proc Natl Acad Sci U S A. 1990 Sep;87(18):7314–7318. doi: 10.1073/pnas.87.18.7314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakobovits A., Vergara G. J., Kennedy J. L., Hales J. F., McGuinness R. P., Casentini-Borocz D. E., Brenner D. G., Otten G. R. Analysis of homozygous mutant chimeric mice: deletion of the immunoglobulin heavy-chain joining region blocks B-cell development and antibody production. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2551–2555. doi: 10.1073/pnas.90.6.2551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- June C. H., Ledbetter J. A., Linsley P. S., Thompson C. B. Role of the CD28 receptor in T-cell activation. Immunol Today. 1990 Jun;11(6):211–216. doi: 10.1016/0167-5699(90)90085-n. [DOI] [PubMed] [Google Scholar]
- Kitamura D., Kudo A., Schaal S., Müller W., Melchers F., Rajewsky K. A critical role of lambda 5 protein in B cell development. Cell. 1992 May 29;69(5):823–831. doi: 10.1016/0092-8674(92)90293-l. [DOI] [PubMed] [Google Scholar]
- Kitamura D., Roes J., Kühn R., Rajewsky K. A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin mu chain gene. Nature. 1991 Apr 4;350(6317):423–426. doi: 10.1038/350423a0. [DOI] [PubMed] [Google Scholar]
- Kühn R., Rajewsky K., Müller W. Generation and analysis of interleukin-4 deficient mice. Science. 1991 Nov 1;254(5032):707–710. doi: 10.1126/science.1948049. [DOI] [PubMed] [Google Scholar]
- Mombaerts P., Iacomini J., Johnson R. S., Herrup K., Tonegawa S., Papaioannou V. E. RAG-1-deficient mice have no mature B and T lymphocytes. Cell. 1992 Mar 6;68(5):869–877. doi: 10.1016/0092-8674(92)90030-g. [DOI] [PubMed] [Google Scholar]
- Murray J. S., Madri J., Tite J., Carding S. R., Bottomly K. MHC control of CD4+ T cell subset activation. J Exp Med. 1989 Dec 1;170(6):2135–2140. doi: 10.1084/jem.170.6.2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nabavi N., Freeman G. J., Gault A., Godfrey D., Nadler L. M., Glimcher L. H. Signalling through the MHC class II cytoplasmic domain is required for antigen presentation and induces B7 expression. Nature. 1992 Nov 19;360(6401):266–268. doi: 10.1038/360266a0. [DOI] [PubMed] [Google Scholar]
- Nitschke L., Kosco M. H., Köhler G., Lamers M. C. Immunoglobulin D-deficient mice can mount normal immune responses to thymus-independent and -dependent antigens. Proc Natl Acad Sci U S A. 1993 Mar 1;90(5):1887–1891. doi: 10.1073/pnas.90.5.1887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oi V. T., Jones P. P., Goding J. W., Herzenberg L. A., Herzenberg L. A. Properties of monoclonal antibodies to mouse Ig allotypes, H-2, and Ia antigens. Curr Top Microbiol Immunol. 1978;81:115–120. doi: 10.1007/978-3-642-67448-8_18. [DOI] [PubMed] [Google Scholar]
- Osmond D. G. The turnover of B-cell populations. Immunol Today. 1993 Jan;14(1):34–37. doi: 10.1016/0167-5699(93)90322-C. [DOI] [PubMed] [Google Scholar]
- Paul W. E. Pleiotropy and redundancy: T cell-derived lymphokines in the immune response. Cell. 1989 May 19;57(4):521–524. doi: 10.1016/0092-8674(89)90121-9. [DOI] [PubMed] [Google Scholar]
- Scher I. CBA/N immune defective mice; evidence for the failure of a B cell subpopulation to be expressed. Immunol Rev. 1982;64:117–136. doi: 10.1111/j.1600-065x.1982.tb00421.x. [DOI] [PubMed] [Google Scholar]
- Shinkai Y., Rathbun G., Lam K. P., Oltz E. M., Stewart V., Mendelsohn M., Charron J., Datta M., Young F., Stall A. M. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell. 1992 Mar 6;68(5):855–867. doi: 10.1016/0092-8674(92)90029-c. [DOI] [PubMed] [Google Scholar]
- Sinha A. A., Lopez M. T., McDevitt H. O. Autoimmune diseases: the failure of self tolerance. Science. 1990 Jun 15;248(4961):1380–1388. doi: 10.1126/science.1972595. [DOI] [PubMed] [Google Scholar]
- Smiley S. T., Laufer T. M., Lo D., Glimcher L. H., Grusby M. J. Transgenic mice expressing MHC class II molecules with truncated A beta cytoplasmic domains reveal signaling-independent defects in antigen presentation. Int Immunol. 1995 Apr;7(4):665–677. doi: 10.1093/intimm/7.4.665. [DOI] [PubMed] [Google Scholar]
- St-Pierre Y., Nabavi N., Ghogawala Z., Glimcher L. H., Watts T. H. A functional role for signal transduction via the cytoplasmic domains of MHC class II proteins. J Immunol. 1989 Aug 1;143(3):808–812. [PubMed] [Google Scholar]
- Tepper R. I., Levinson D. A., Stanger B. Z., Campos-Torres J., Abbas A. K., Leder P. IL-4 induces allergic-like inflammatory disease and alters T cell development in transgenic mice. Cell. 1990 Aug 10;62(3):457–467. doi: 10.1016/0092-8674(90)90011-3. [DOI] [PubMed] [Google Scholar]
- Vasicek T. J., Levinson D. A., Schmidt E. V., Campos-Torres J., Leder P. B-less: a strain of profoundly B cell-deficient mice expressing a human lambda transgene. J Exp Med. 1992 May 1;175(5):1169–1180. doi: 10.1084/jem.175.5.1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Boehmer H., Kisielow P. Self-nonself discrimination by T cells. Science. 1990 Jun 15;248(4961):1369–1373. doi: 10.1126/science.1972594. [DOI] [PubMed] [Google Scholar]