Abstract
Mutation of Tar DNA-binding protein 43 (TDP-43) is linked to amyotrophic lateral sclerosis. Although astrocytes have important roles in neuron function and survival, their potential contribution to TDP-43 pathogenesis is unclear. Here, we created novel lines of transgenic rats that express a mutant form of human TDP-43 (M337V substitution) restricted to astrocytes. Selective expression of mutant TDP-43 in astrocytes caused a progressive loss of motor neurons and the denervation atrophy of skeletal muscles, resulting in progressive paralysis. The spinal cord of transgenic rats also exhibited a progressive depletion of the astroglial glutamate transporters GLT-1 and GLAST. Astrocytic expression of mutant TDP-43 led to activation of astrocytes and microglia, with an induction of the neurotoxic factor Lcn2 in reactive astrocytes that was independent of TDP-43 expression. These results indicate that mutant TDP-43 in astrocytes is sufficient to cause non-cell-autonomous death of motor neurons. This motor neuron death likely involves deficiency in neuroprotective genes and induction of neurotoxic genes in astrocytes.
Keywords: amyotrophic lateral sclerosis, astrocytes, Lcn2, rats, TAR DNA-binding protein 43
Introduction
Amyotrophic lateral sclerosis (ALS) results from progressive loss of motor neurons and denervation atrophy of skeletal muscles (Lim et al, 2012). ALS often develops around middle age and inevitably progresses to paralysis during a disease course of 5 years on average. At present, no treatment substantially prolongs patient’s lifespan or significantly improves the quality of life. Although most ALS cases are sporadic, about 10% are associated with pathogenic mutations of individual genes such as superoxide dismutase 1 (SOD1), Tar DNA-binding protein 43 (TDP-43), and C9ORF72 (Deng et al, 1993, 2011; Rosen et al, 1993; Gurney et al, 1994; Kabashi et al, 2008a, 2008b; Rutherford et al, 2008; Sreedharan et al, 2008; Van Deerlin et al, 2008; Kwiatkowski et al, 2009; Vance et al, 2009; DeJesus-Hernandez et al, 2011; Renton et al, 2011; Wu et al, 2012a). The pathogenesis of TDP-43 mutation in ALS has been validated in a variety of animal and cell models. Overexpression of mutant TDP-43 causes neuronal death in Caenorhabditis elegans (Ash et al, 2010; Vaccaro et al, 2012), drosophila (Li et al, 2010; Voigt et al, 2010; Wang et al, 2011), zebrafish (Kabashi et al, 2010), mice (Wegorzewska et al, 2009; Shan et al, 2010; Wils et al, 2010; Xu et al, 2010; Igaz et al, 2011; Swarup et al, 2011), rats (Zhou et al, 2010; Huang et al, 2012b), monkeys (Uchida et al, 2012), and cultured human motor neurons differentiated from reprogrammed stem cells (Bilican et al, 2012). Intriguingly, an increase in the expression of normal TDP-43 is sufficient to kill neurons, although the wild-type form of TDP-43 is less neurotoxic than its mutant forms (Sephton et al, 2010; Tsai et al, 2010; Wu et al, 2012b). Indeed, TDP-43 proteinopathy is observed in patients with ALS or frontotemporal lobar degeneration who have no pathogenic mutation of TDP-43 (Neumann et al, 2006). TDP-43 is a ribonucleoprotein capable of binding to both DNA and RNA targets and is reported to regulate miRNA biogenesis and an enormous number of protein-encoding genes (Polymenidou et al, 2011; Sephton et al, 2011; Tollervey et al, 2011; Kawahara and Mieda-Sato, 2012). In particular, TDP-43 binds to the 3′ untranslational region of its own mRNA and regulates mRNA stability (Ayala et al, 2011; Avendano-Vazquez et al, 2012), thereby finely tuning its own expression. Disturbances to TDP-43 expression, therefore, may tilt the functional balance of the complex pathways that are tightly regulated by TDP-43, resulting in severe outcomes, including neuronal death.
The death of motor neurons in ALS may not require simultaneous TDP-43 alteration in all types of relevant cells including neurons, glia, and muscle cells. Rather, unhealthy cells may interact aberrantly with neighbouring cells to initiate or propagate neuron death signalling. We previously reported that mutant TDP-43 in motor neurons alone is sufficient to initiate and propagate cell-autonomous neurodegeneration in transgenic rats (Huang et al, 2012b); however, less expression of mutant TDP-43 can result in comparable phenotypes when the transgene is expressed simultaneously in multiple types of neurons (Huang et al, 2012b), suggesting that cells other than motor neurons also contribute to TDP-43 pathogenesis. Astrocytes, which are critical for neuron function and survival, may play an important role in TDP-43 pathogenesis. In response to TDP-43 mutation, neurotoxic effects may result from changes in the expression of astrocytic receptors and transporters, the coupling of gap junctions, the release and metabolism of astrocytic transmitters, or the synthesis and release of chemokines, cytokines, and free radicals (Lee et al, 2007, 2009; Escartin and Bonvento, 2008; Landreth et al, 2008; Bi et al, 2013). In rodent models of ALS, the overexpression of mutant SOD1 in astrocytes does not cause motor neuron death in transgenic mice (Gong et al, 2000), but transplanted astrocytes expressing mutant SOD1 induce the death of motor neurons in acceptor rats (Papadeas et al, 2011). It is not clear why different outcomes are obtained with varying approaches. Furthermore, it is not clear how astrocytes contribute to the development and progression of motor neuron degeneration in TDP-43 pathogenesis.
To examine the effects of astrocytic mutant TDP-43 expression on motor neurons, we created novel lines of transgenic rats that express human TDP-43 with a pathogenic mutation (M337V substitution) selectively in astrocytes. This restricted overexpression of mutant TDP-43 in astrocytes caused a progressive loss of spinal motor neurons and denervation atrophy of skeletal muscles, leading to progressive paralysis. Therefore, we found that mutant TDP-43 in astrocytes is sufficient to initiate and propagate non-cell-autonomous motor neuron degeneration.
Results
Selective GFAP promoter-driven transgene expression in spinal cord astrocytes
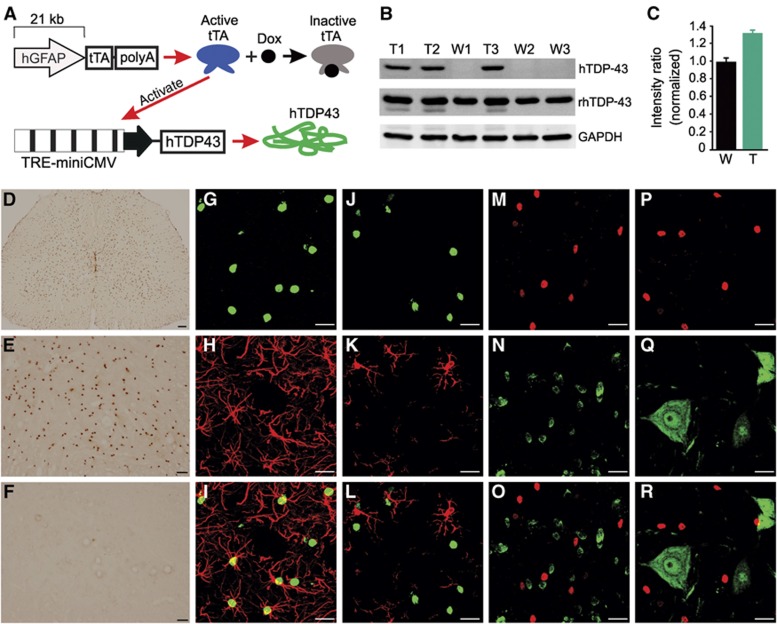
To examine the role of astrocytes in TDP-43 pathogenesis in motor neurons, we selectively expressed mutant TDP-43 (M337V substitution) in astrocytes in the rat spinal cord. Tetracycline (Tet)-inducible transgenic rats carrying a mutant human TDP-43 (TDP-43M337V) transgene under the control of a Tet-responsive element (TRE) were previously created and validated (Zhou et al, 2010; Huang et al, 2012b); therefore, we continued to use the Tet-off system for inducible expression of mutant human TDP-43 in astrocytes (Figure 1A). We also created new lines of transgenic rats that carry a Tet-responsive transactivator (tTA) under the control of the human gfap promoter (Figure 1A). A small fragment of the human GFAP promoter (2.3 kb) was previously reported to drive transgene expression selectively in astrocytes in mice (Gong et al, 2000). To increase the chance of cell-specific transgene expression, we isolated a bulk fragment of the human GFAP promoter (21 kb) from a BAC clone (CHORI: RP11-419E16) and used this promoter to drive tTA transgene expression (Figure 1A). Three transmittable GFAP–tTA lines were created (GFAP–tTA#1, 2, and 8). After crossing GFAP–tTA lines with TRE–TDP-43M337V line-16 (Zhou et al, 2010), we assessed transgene expression in GFAP–tTA/TRE–TDP-43M337V double transgenic rats after doxycycline (Dox) was withdrawn (Supplementary Figure S1). Two of the three transmittable GFAP–tTA lines (GFAP–tTA#1 and 2) expressed human TDP-43 (Supplementary Figure S1).
Figure 1.
Restricted expression of mutant human TDP-43 in astrocytes in transgenic rats. (A) Schematic diagram showing a Tet-off system for inducible gene expression. The transgene tTA is driven by the human GFAP promoter. Expressed tTA binds to the TRE promoter and activates the transcription of the mutant human TDP-43 transgene (hTDP43). Dox binds to tTA and inactivates its transcriptional activity. (B, C) Immunoblotting showed expression levels for hTDP43, total TDP-43 (rat and human TDP-43: rhTDP43), and GAPDH in the spinal cord of transgenic rats (T1, T2, and T3) and nontransgenic littermates (W1, W2, and W3). The intensity of individual rhTDP43 blots was first normalized to that of individual GAPDH and calculated as a ratio of transgenic to nontransgenic rats (n=3). (D) Immunohistochemistry revealed hTDP43 expression in GFAP–tTA/TRE–hTDP-43M337V double transgenic rats (D and E) but not GFAP–tTA#2 single transgenic rats (F). (G–R) Double-labelling fluorescence staining revealed that hTDP43 (G, J, M, and P) was colocalized with the astrocyte marker GFAP (H and I) and that hTDP43 did not colocalize with the microglia marker Iba1 (K and L), oligodendrocyte marker APC (N and O), or neuron marker NeuN (Q and R). Scale bars: 100 μm (D), 50 μm (E, F), 20 μm (G–R).
As the GFAP–tTA#2 line of rats showed substantial expression of the transgene in the spinal cord (Supplementary Figure S1), our study was largely focused on characterizing this desirable transgenic line. In GFAP–tTA#2/TRE–TDP-43M337V double transgenic rats, human TDP-43 was clearly expressed in the spinal cord and brain but barely detectable in the skeletal muscles and sciatic nerves (Supplementary Figure S2). Human TDP-43 was also detected in some non-nervous tissues, including kidney, lung, spleen, stomach, and heart (Supplementary Figure S2), which is consistent with previous reports of GFAP promoter activity in peripheral organs, specifically in hepatic stellate cells and stellate-shaped cells of non-nervous tissues (Gard et al, 1985; Buniatian et al, 2002; Zhao and Burt, 2007; Puche et al, 2013). Quantitative analysis of human TDP-43 expression in transgenic rats (Figure 1B) showed that total TDP-43 protein was increased by ∼30% in the spinal cord (Figure 1C). Immunohistochemistry also confirmed that human TDP-43 was substantially expressed in the spinal cord of double transgenic rats (Figure 1D–F). Double-labelling fluorescence staining revealed that human TDP-43 was expressed selectively in astrocytes (Figure 1G–I) but not microglia (Figure 1J–L), oligodendrocytes (Figure 1M–O), or neurons (Figure 1P–R) in the spinal cord. The predominate expression of GFAP in astrocytic processes (Supplementary Figure S3) rendered the strict colocalization of GFAP and human TDP-43 difficult. Therefore, we used confocal microscopy to assess the relative localization of GFAP with human TDP-43 by continuous scanning (Supplementary Figure S3). Z-stack images show that GFAP fluorescence wrapped around human TDP-43 fluorescence (Supplementary Figure S3), suggesting that GFAP and human TDP-43 were expressed in the same astrocytes. NG2-positive glia, which are a major cell population in the central nervous system (CNS), were increased in the spinal cord of double transgenic rats, but NG2 staining did not colocalize with human TDP-43 staining (Supplementary Figure S4). Taken together, these findings demonstrate that mutant human TDP-43 was expressed selectively in astrocytes in the spinal cord of double transgenic rats.
Restricted expression of mutant TDP-43 in astrocytes causes progressive paralysis
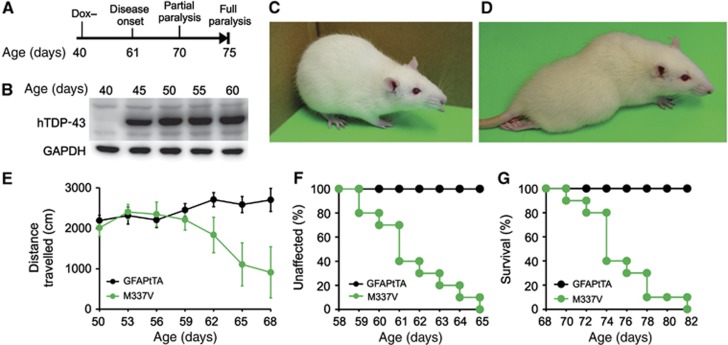
To induce a disease phenotype in adult rats, we administered Dox in rats’ drinking water until 40 days of age (Figure 2A). This Dox treatment effectively suppressed transgene expression, and transgene expression was rapidly restored in the absence of Dox (Figure 2B). When crossed onto a TRE–TDP-43M337V transgenic background, the two GFAP–tTA-expressing lines (Supplementary Figure S1: line #1 and 2) were found to develop paralysis by 80 days of age (Figure 2 and Supplementary Figure S5). The rats with higher transgene expression (line #2) gradually developed paralysis in distinct stages (Figure 2 and Supplementary Video S1). They initially displayed weakness in one leg by 60 days of age as detected in the open field test (Figure 2E and F: disease onset), after which they progressively lost mobility and became fully paralyzed in two or more legs by 80 days of age (Figure 2D and G: disease end stages). Whereas GFAP–tTA#2 rats displayed a phenotype predominantly characterized by paralysis (Figure 2), GFAP–tTA#1 rats developed a more complex phenotype, showing leg weakness by 70 days of age and quickly developing tonic seizures by 80 days of age (Supplementary Figure S5). Therefore, both lines of transgenic rats showed a progressive impairment in movement reminiscent of ALS when they expressed mutant human TDP-43 selectively in astrocytes.
Figure 2.
Restricted expression of mutant TDP-43 in astrocytes causes progressive paralysis in transgenic rats. (A) Diagram showing strategy for inducing human TDP-43 (hTDP43) transgene expression and paralysis in transgenic rats. GFAP–tTA#2/TRE–TDP-43M337V double transgenic (M337V) rats were deprived of Dox (Dox−) at 40 days (d) of age. (B) Immunoblotting showed a rapid induction of the hTDP43 transgene in the spinal cords after Dox withdrawal. Equal loading was probed with an antibody to GAPDH. (C, D) Representative images of a paralyzed M337V rat (D) and an age-matched GFAP–tTA#2 single transgenic rat (C). (E) The open field test measured mobility during 10-min trials. GFAP–tTA denotes GFAP–tTA#2 transgenic rats. Data are shown as mean±s.d. (n=12, equal sex composition). (F, G) Probability of weakness onset (F) and rat mortality (G). Mortality was counted as full paralysis.
Astrocytic TDP-43M337V overexpression causes progressive motor neuron death and denervation atrophy of muscles
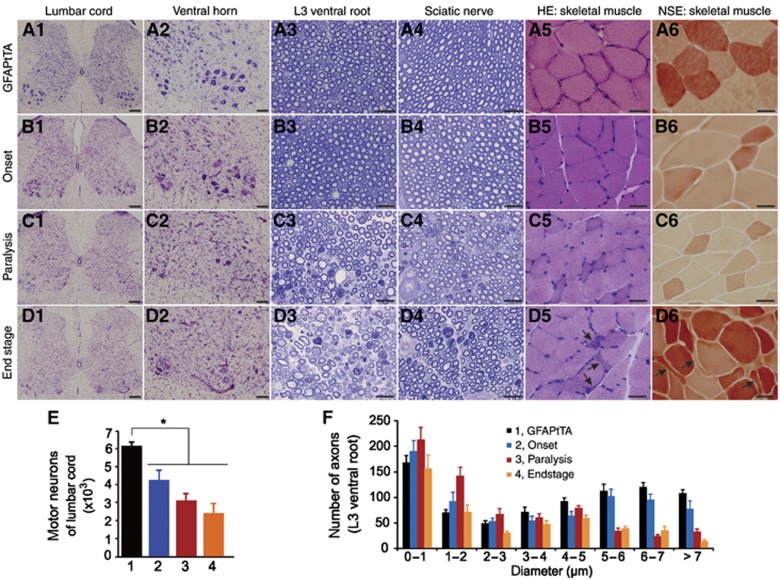
Paralysis in GFAP–tTA/TRE–TDP-43M337V transgenic rats likely resulted from damage to motor units. As the GFAP–tTA#2 rat line developed ALS-like paralysis when carrying the TRE–TDP-43M337V transgene (Figure 2), we continued to characterize this desirable line in greater detail (Figure 3). Cresyl violet staining showed that spinal motor neurons were lost throughout the progression of paralysis in transgenic rats (Figure 3A1–D1 and A2–D2). Also, toluidine blue staining revealed a progressive degeneration of motor axons in the ventral roots (Figure 3A3–D3) and sciatic nerves (Figure 3A4–D4). As a result of motor neuron degeneration, groups of skeletal muscle fibres were atrophied as revealed by H&E staining (Figure 3A5–D5) and nonspecific esterase histochemistry (Figure 3A6–D6). Unbiased stereological cell counting confirmed that motor neurons in the spinal cords were gradually lost in the transgenic rats (Figure 3E). ImageJ analysis of motor axons verified that motor axons were progressively damaged as paralysis progressed (Figure 3F). In contrast to motor neuron axons in the ventral roots, sensory neuron axons in the dorsal roots were not affected (Supplementary Figure S6). Taken together, these findings indicate that paralysis in GFAP–tTA#2/TRE–TDP-43M337V rats results from the progressive loss of motor neurons and denervation atrophy of skeletal muscles. Although GFAP–tTA#1/TRE–TDP-43M337V rats showed a more complex phenotype, motor neuron loss was also detected at advanced disease stages (Supplementary Figure S5). Therefore, we found that astrocytic overexpression of mutant TDP-43 causes progressive motor neuron death.
Figure 3.
Astrocytic TDP-43M337V expression causes progressive motor neuron degeneration and denervation atrophy in transgenic rats. (A1–D1, A2–D2) Cresyl violet staining revealed progressive motor neuron loss in GFAP–tTA#2/TRE–TDP-43M337V transgenic rats (M337V) from the onset of weakness (B1–2) to paralysis (C1–2) and end stage (D1–2) compared with GFAP–tTA#2 single transgenic rats (A1–2). (A3–D3, A4–D4) Toluidine blue staining revealed structures of ventral roots (A3–D3) and sciatic nerves (A4–D4) in M337V rats at varying disease stages and in GFAP–tTA rats at 70 days of age. (A5–D5, A6–D6) H&E staining (A5–D5) and histochemistry for nonspecific esterase (A6–D6) revealed atrophy of skeletal muscle in M337V rats at the disease end stage. Arrows point to atrophied muscle fibres. (E) Stereological cell counting revealed the number of motor neurons (>25 μm) in the L3–L5 lumbar cords of GFAP–tTA and M337V rats. Groups in panel E are identical to those in panel F. Data are shown as mean±s.d. (n=6). *P<0.05. (F) The distribution of motor axons in cross-sections of L3 ventral roots was determined with ImageJ software. Data are shown as mean±s.d. (n=4 rats). Scale bars: 200 μm (A1–D1), 100 μm (A2–D2), 30 μm (A3–D3 to A6–D6).
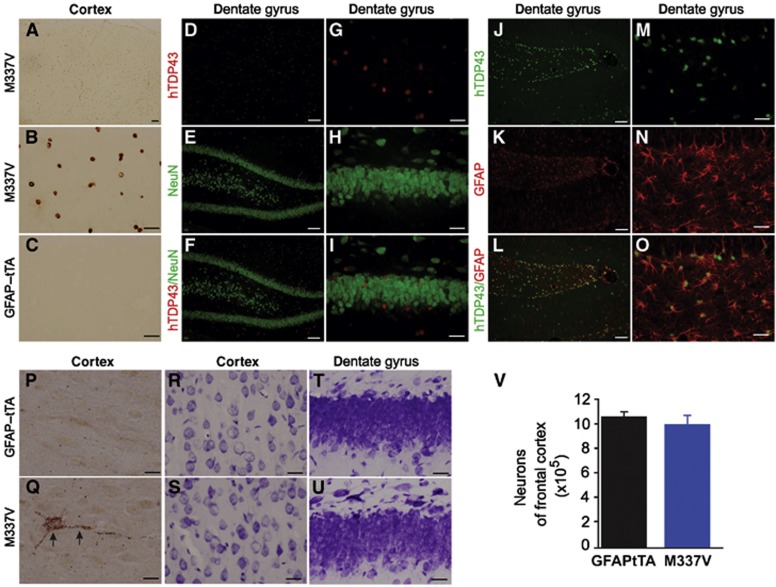
In GFAP–tTA#2/TRE–TDP-43M337V transgenic rats, human TDP-43 was substantially expressed in the brain, but stereological cell counting revealed no significant loss of cortical neurons (Figure 4). However, scattered degenerating neurons were detected by silver staining, which is a classic method for detecting degenerating neurons in the CNS (Figure 4Q). Compared with spinal motor neurons, therefore, neurons in the brain exhibited milder pathologies. This variation in phenotype expression between different populations of neurons may arise from the different patterns of transgene expression in the brain and spinal cord (Figure 5S: A).
Figure 4.
No significant loss of neurons in the brains of GFAP–tTA/TRE–TDP-43M337V transgenic rats. (A–C) Immunohistochemistry revealed expression of mutant human TDP-43 (hTDP43) in the cortex of GFAP–tTA/TRE–TDP-43M337V double transgenic rats (M337V: A and B) but not that of GFAP–tTA single transgenic rats (C). (D–O) Double-labelling fluorescence staining revealed colocalization of hTDP43 with the astrocyte marker GFAP (J–O) but not the neuronal marker NeuN (D–I). Rats were deprived of Dox at 40 days of age, and transgene expression was examined at 50 days of age. (P, Q) FD Silver staining detected scattered degenerating neurons in M337V rats at 70 days of age (Q) but not in age-matched GFAP–tTA rats (P). (R–U) Cresyl violet staining showed cortical (R, S) and hippocampal (T, U) neurons in M337V rats with paralysis. Scale bars: 100 μm (A, D–F, and J–L), 30 μm (B, C, G–I, and M–O), 20 μm (P, Q). (V) Stereological cell counting revealed no significant loss of cortical neurons in M337V rats during paralysis stages (n=4).
Figure 5.
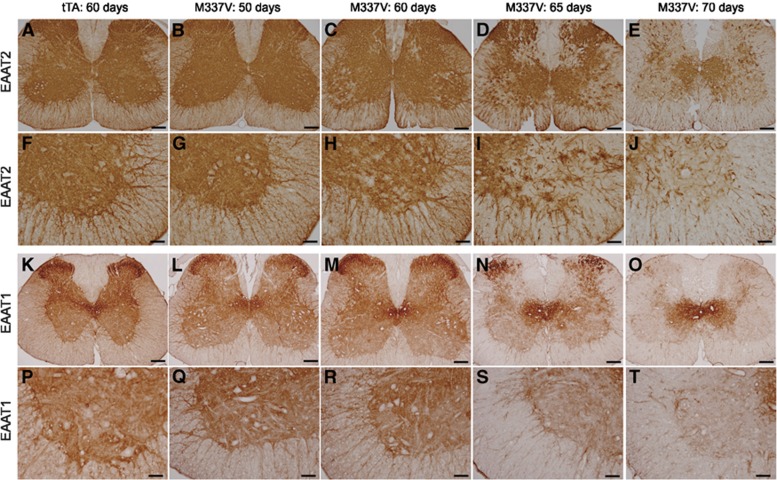
Astrocytic TDP-43M337V expression results in progressive depletion of EAAT1 and EAAT2 in transgenic rats. (A–T) Immunostaining revealed that the glutamate transporters EAAT1 (K–T) and EAAT2 (A–J) were globally and progressively depleted in the spinal cord of GFAP–tTA#2/TRE–TDP-43M337V double transgenic rats (M337V) as compared with GFAP–tTA#2 single transgenic rats (tTA). Representative images show EAAT1 and EAAT2 expression in cross-sections of the whole spinal cords (A–E) as well as the ventral (F–J and P–T) and dorsal (K–O) horns of lumbar cords. Scale bars: 200 μm (A–E), 100 μm (F–T).
Astrocytic TDP-43M337V expression leads to progressive depletion of glutamate transporters EAAT1 and EAAT2
Upper motor neurons signal lower motor neurons by releasing the neurotransmitter glutamate, and excess glutamate in the synaptic cleft is taken up by astrocytes via the glutamate transporters EAAT1 and EAAT2 (Rothstein et al, 1996). Depletion of glutamate transporters causes excitatory neurotoxicity and paralysis in rodents (Rothstein et al, 1996). Overexpression of mutant SOD1 causes a focal depletion of EAAT2 (or GLT-1) but not EAAT1 in the ventral horns of transgenic rats during paralysis stages (Howland et al, 2002; Lepore et al, 2008). We examined the effects of astrocytic TDP-43M337V expression on EAAT1 and EAAT2 in transgenic rats. After the TDP-43M337V transgene was expressed, both EAAT1 and EAAT2 proteins underwent progressive depletion in the spinal cord (Figure 5), indicating that astrocytic TDP-43M337V overexpression causes a progressive loss of astroglial glutamate transporters.
Astrocytic TDP-43M337V expression leads to induction of the neurotoxic gene lcn2
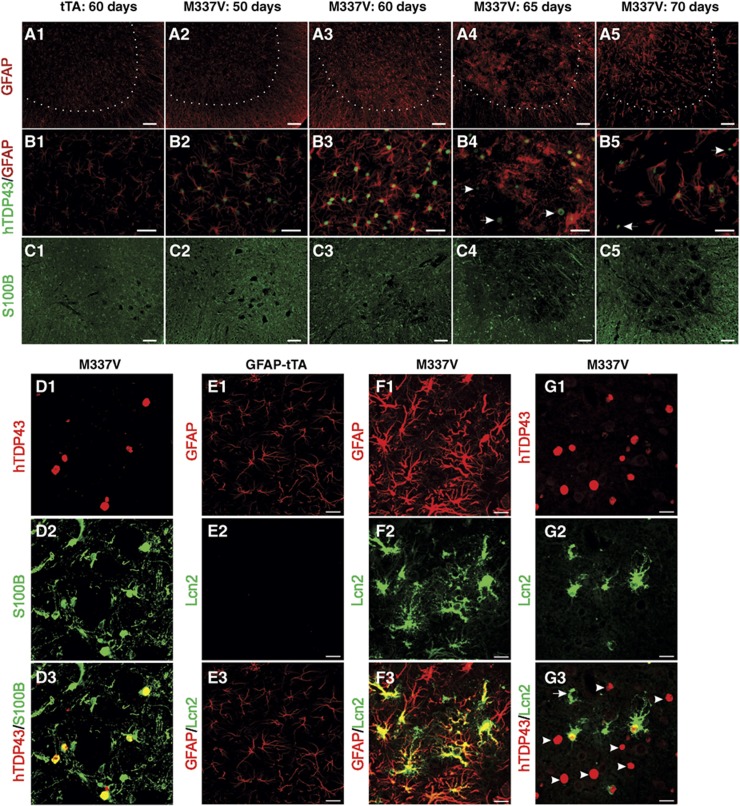
The expression of mutant TDP-43 in astrocytes led to the upregulation of GFAP expression in transgenic rats (Figure 6A1), suggesting that astrocytes became reactive. Intriguingly, some TDP-43M337V-positive cells were partially depleted of GFAP during paralysis stages (Figure 6B4 and B5), suggesting a loss of unhealthy astrocytes consistent with that observed in an in vitro study of mutant TDP-43-expressing astrocytes (Serio et al, 2013). The expression of S100B, which has been used as a marker of astrocyte transformation (Diaz-Amarilla et al, 2011), was altered in TDP-43M337V transgenic rats (Figure 6C1). Furthermore, recent studies show that reactive astrocytes secrete Lcn2 to promote neuronal death (Bi et al, 2013). We found that Lcn2 expression was indeed induced in astrocytes in transgenic rats. Moreover, this induction of Lcn2 was independent of mutant TDP-43 expression (Figure 6E–G), suggesting that it was a response to the reactive status of astrocytes rather than the expression of mutant TDP-43.
Figure 6.
Astrocytic TDP-43M337V overexpression alters astrocyte characteristics in transgenic rats. Gene expression was examined in cross-sections of lumbar cords. (A1–5, B1–5) Immunofluorescence staining revealed that GFAP expression was initially enhanced and later depleted in some astrocytes expressing mutant human TDP-43 (indicated by arrows). (C1–5) Representative images show that S100B expression was also altered in mutant TDP-43 transgenic rats. (D1–3) Immunofluorescence staining revealed the colocalization of S100B with human TDP-43 (hTDP43) in transgenic rats. (E1–3, F1–3, G1–3) Confocal microscopy revealed that Lcn2 was induced in GFAP–tTA#2/TRE–TDP-43M337V double transgenic rats (M337V: F1–3 and G1–3) but not in GFAP–tTA#2 single transgenic rats (tTA: E1–3). Although Lcn2 colocalized well with GFAP (F1–3), it did not fully colocalize with hTDP43 (G1–3). Arrowheads point to cells that express hTDP43 but not Lcn2, and arrows point to cells that express Lcn2 but not hTDP43 (G3). Scale bars: 100 μm (A1–5 and C1–5), 40 μm (B1–5), and 25 μm (D–G).
We also observed the formation of ubiquitin inclusions in astrocytes, but not in neurons (Supplementary Figure S7), again supporting a glial pathology of ALS and confirming that ubiquitin inclusion is not required for neurodegeneration in this disease (Huang et al, 2012b). The activation of microglia, which is a common response to neurodegeneration, was observed in the ventral horn as paralysis progressed (Supplementary Figure S8).
Discussion
How neurodegeneration signalling is initiated and propagated among complex neuron–glia networks in the CNS is not well understood. Astrocytes are critical to neuron function and survival, with a single astrocyte contacting up to thousands of synapses in rodents (Matyash and Kettenmann, 2010); therefore, disturbance to TDP-43 expression in astrocytes may be sufficient to induce neurodegeneration. To test this possibility, we created novel lines of transgenic rats that selectively express mutant human TDP-43 in astrocytes. In one desirable GFAP–tTA line (GFAP–tTA#2), expression of the transgene was restricted to astrocytes in the spinal cord, with no detection of the transgene in other types of cells such as neurons, oligodendrocytes, or microglia. Further, human TDP-43 was barely detectable in sciatic nerves and skeletal muscles along the motor units. Selective expression of mutant TDP-43 in spinal cord astrocytes led to the progressive loss of spinal motor neurons and motor axons and the denervation atrophy of skeletal muscles, resulting in paralysis. Astrocytic TDP-43M337V expression, therefore, is sufficient to kill spinal motor neurons in a non-cell-autonomous manner. Two of the newly created GFAP–tTA lines developed progressive paralysis reminiscent of ALS. Thus, our findings suggest that mutant TDP-43 in astrocytes is sufficient to initiate and propagate neurodegeneration signalling, at least in rats.
Expression of mutant TDP-43 in astrocytes may change their molecular characteristics. EAAT1 (also known as GLAST) and EAAT2 (also known as GLT-1) are globally and progressively depleted in the spinal cord of GFAP–tTA/TRE–TDP-43M337V transgenic rats. GLT-1 and GLAST are two astroglial glutamate transporters that are responsible for synaptic clearance of the neurotransmitter glutamate (Matyash and Kettenmann, 2010). Deficiencies in GLT-1 or GLAST produce chronic excitatory neurotoxicity in animal models (Rothstein et al, 1996; Tanaka et al, 1997). GLT-1 is depleted in the spinal cord of SOD1G93A transgenic rats, and stimulating GLT-1 expression by beta-Lactam antibiotics has therapeutic effects on SOD1G93A transgenic rats (Howland et al, 2002; Rothstein et al, 2005). In addition, GLAST is depleted in transgenic mice expressing a mutant form of ataxin-7 linked to spinocerebellar ataxia (Custer et al, 2006). Although GLT-1 is focally depleted in the ventral horn of rats that ubiquitously express mutant SOD1G93A in the spinal cord (Howland et al, 2002), we found that GLT-1 was globally depleted in the spinal cord of TDP-43M337V rats that selectively express mutant TDP-43 in astrocytes. Distinct patterns of GLT-1 depletion might be attributed (at least partially) to differences in the cell types of transgene expression. Nonetheless, astrocytic expression of mutant TDP-43 causes a deficiency in neuroprotective genes such as GLT-1 and GLAST.
Serio et al (2013) recently reported that TDP-43M337V-expressing astrocytes do not adversely affect the survival of motor neurons in co-culture. As the intrinsic interaction among neurons and glia is disturbed in culturing conditions, disease mechanisms may not be fully uncovered in in vitro models. In addition, astrocytes differentiated from patients’ iPS cells carry only one copy of the disease gene and express mutant TDP-43 at moderate levels. Therefore, insufficient expression of mutant TDP-43 in cultured astrocytes may not affect motor neuron survival. Nonetheless, it is important to emphasize that examining disease mechanisms in an in vivo animal model is an essential step towards understanding disease pathogenesis.
Astrocytic expression of mutant TDP-43 led to activation of astrocytes and microglia in the spinal cord. In a previous study, reactive astrocytes were found to secrete the neurotoxic factor Lcn2 (Bi et al, 2013). In the present study, Lcn2 was induced selectively in astrocytes in GFAP–tTA/TRE–TDP-43M337V transgenic rats, and its induction was independent of mutant TDP-43 expression. Lcn2 is a small lipocalin that is selectively toxic to neurons rather than glial cells and that sensitizes neurons to beta-amyloid toxicity (Naude et al, 2012; Bi et al, 2013). Reactive astrocytes likely secrete molecules such as Lcn2 to exert toxicity in neurons. Astrocytic TDP-43M337V expression may lead to intrinsic activation of neurotoxic genes that may be involved in TDP-43 pathogenesis.
In summary, restricted expression of mutant TDP-43 in astrocytes is sufficient to kill motor neurons in a non-cell-autonomous manner and thus to cause ALS-like paralysis in transgenic rats. This TDP-43 pathogenesis likely involves deficiency in neuroprotective genes and induction of neurotoxic genes in astrocytes.
Materials and methods
Creation of transgenic rats
Transgenic rats were created and maintained in a Sprague Dawley background as previously described (Zhou et al, 2009). The promoter of the human glial fibrillary acidic protein (GFAP) gene was isolated from a BAC clone (CHORI: RP11-419E16). The GFAP promoter of 21 kb was used to drive the expression of a tTA gene. GFAP–tTA-positive rats were identified by PCR analysis of tail DNA with the following primers: 5′-TGAGTGAGATAATGCCTGGG-3′ (forward) and 5′-ACCCTCTCTCTAGGAAGGTG-3′ (reverse). TRE–TDP-43M337V transgenic rats were crossed with those from GFAP–tTA transgenic lines (Zhou et al, 2010), and transgene expression profiles were determined by immunoblotting and immunohistochemistry. To selectively express the human TDP-43 transgene in adulthood, breeding rats and transgenic offspring were given Dox in their drinking water (50 μg/ml) to suppress transgene expression during embryonic and postnatal development. GFAP–tTA/TRE–TDP-43M337V transgenic rats were deprived of Dox at the 40 days of age to induce TDP-43M337V expression.
Behavioural tests and disease stages
Using the open field test (Med Associates), the total distance a rat travelled within 10 min was used to detect the onset of the ALS phenotypes in transgenic rats (Huang et al, 2012b). Disease onset was defined as an unrecoverable reduction in distance travelled, and paralysis was defined as a dragging of legs or an inability to retract individual legs. Disease end stages were defined as an inability to retract two or more legs, frequent tonic seizures, or an inability to right itself when the rat was placed on its side.
Immunoblotting, histochemistry, and fluorescence staining
Human TDP-43 transgene expression was determined by immunoblotting as previously described (Huang et al, 2012a, 2012b). The expression profiles of GLT-1, GLAST, ubiquitin, and human TDP-43M337V were examined with immunohistochemistry, and the colocalization of human TDP-43 with related proteins was examined with double-labelling immunofluorescence staining. Immunohistochemistry and immunofluorescence staining were performed on coronal forebrain sections and cross-sections of the spinal cord. The following primary antibodies were used for immunostaining: chicken anti-ubiquitin (Sigma), guinea pig anti-GLT-1 (Millipore), mouse monoclonal anti-APC (Calbiochem), mouse monoclonal anti-GAPDH (Abcam), mouse monoclonal anti-human TDP-43 (Abnova, clone 2E2-D3), rabbit anti-GFAP (DAKO, North America), and rabbit anti-Iba-1 (Wako Chemicals, USA). Gene expression in immunostained tissue sections was examined with a Nikon microscope or a confocal microscope (Imaging Facility of Kimmel Cancer Center at Jefferson). Single-layer images were scanned with a Zeiss LSM510 META confocal system, and Z-stacks of confocal images (at 1 μm intervals) were projected to reconstruct astrocyte structure. The structure of gastrocnemius muscle was assessed by H&E staining and histochemistry for nonspecific esterase as previously described (Huang et al, 2012b).
Cresyl violet staining and stereological cell counting
Lower motor neurons in the lumbar spinal cord and cortical neurons in the frontal cortex were examined by Cresyl violet staining and quantified by stereological cell counting as previously described (Huang et al, 2012b). Briefly, the lumbar spinal cord (L3–L5) was cut into cross-sections of 30 μm, and motor neurons were counted in every 10th section on both sides of the spinal cord (i.e., 15–20 sections per rat). Cortical neurons in the frontal cortex were quantitated as previously described (Huang et al, 2011). Briefly, coronal forebrain sections of 30 μm were cut between the apical rostral part of the brain and the first occurrence of hippocampus, and cortical neurons were counted in every 12th section (i.e., 15–18 sections per rat).
Toluidine blue and silver staining
The ultrastructure of axons in the ventral and dorsal roots of the spinal nerve was assessed by toluidine blue staining. Rats were deeply anesthetized and perfused with a mixture of 4% paraformaldehyde and 2% glutaraldehyde. Nerve roots were dissected and post-fixed in the same fixative at 4°C overnight. Fixed tissues were embedded in Epon 812 and cut into semithin (1 μm) sections. Semithin sections were stained with 1% toluidine blue and examined under a light microscope. ImageJ software was used to quantify the distribution of motor axons across entire images of nerve roots. Degenerating neurons were examined using the FD NeuroSilver kit (FD Neurotechnologies, Baltimore, MD) as previously described (Zhou et al, 2010).
Statistical analysis
Numbers of motor neurons were compared between different genotypes of rats using unpaired t-tests. A P-value of less than 0.05 was considered statistically significant.
Study approval
Animal use was in accord with NIH guidelines and the animal use protocol was approved by the Institutional Animal Care and Use Committees at Thomas Jefferson University.
Supplementary Material
Acknowledgments
This work is supported by the National Institutes of Health (NIH)/National Institute of Neurological Disorders and Stroke (NS073829 to H.Z., and NS072113 and NS084089 to X.G.X). The content is the author’s responsibility and does not necessarily represent the official view of the NIH Institutes.
Footnotes
The authors declare that they have no conflict of interest.
References
- Ash PE, Zhang YJ, Roberts CM, Saldi T, Hutter H, Buratti E, Petrucelli L, Link CD (2010) Neurotoxic effects of TDP-43 overexpression in C. elegans. Hum Mol Genet 19: 3206–3218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avendano-Vazquez SE, Dhir A, Bembich S, Buratti E, Proudfoot N, Baralle FE (2012) Autoregulation of TDP-43 mRNA levels involves interplay between transcription, splicing, and alternative polyA site selection. Genes Dev 26: 1679–1684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ayala YM, De Conti L, Avendano-Vazquez SE, Dhir A, Romano M, D'Ambrogio A, Tollervey J, Ule J, Baralle M, Buratti E, Baralle FE (2011) TDP-43 regulates its mRNA levels through a negative feedback loop. EMBO J 30: 277–288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bi F, Huang C, Tong J, Qiu G, Huang B, Wu Q, Li F, Xu Z, Bowser R, Xia XG, Zhou H (2013) Reactive astrocytes secrete lcn2 to promote neuron death. Proc Natl Acad Sci USA 110: 4069–4074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilican B, Serio A, Barmada SJ, Nishimura AL, Sullivan GJ, Carrasco M, Phatnani HP, Puddifoot CA, Story D, Fletcher J, Park IH, Friedman BA, Daley GQ, Wyllie DJ, Hardingham GE, Wilmut I, Finkbeiner S, Maniatis T, Shaw CE, Chandran S (2012) Mutant induced pluripotent stem cell lines recapitulate aspects of TDP-43 proteinopathies and reveal cell-specific vulnerability. Proc Natl Acad Sci USA 109: 5803–5808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buniatian GH, Hartmann HJ, Traub P, Wiesinger H, Albinus M, Nagel W, Shoeman R, Mecke D, Weser U (2002) Glial fibrillary acidic protein-positive cells of the kidney are capable of raising a protective biochemical barrier similar to astrocytes: expression of metallothionein in podocytes. Anat Rec 267: 296–306 [DOI] [PubMed] [Google Scholar]
- Custer SK, Garden GA, Gill N, Rueb U, Libby RT, Schultz C, Guyenet SJ, Deller T, Westrum LE, Sopher BL, La Spada AR (2006) Bergmann glia expression of polyglutamine-expanded ataxin-7 produces neurodegeneration by impairing glutamate transport. Nat Neurosci 9: 1302–1311 [DOI] [PubMed] [Google Scholar]
- DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, Nicholson AM, Finch NA, Flynn H, Adamson J, Kouri N, Wojtas A, Sengdy P, Hsiung GY, Karydas A, Seeley WW, Josephs KA, Coppola G, Geschwind DH, Wszolek ZK et al. (2011) Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72: 245–256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N, Yang Y, Fecto F, Shi Y, Zhai H, Jiang H, Hirano M, Rampersaud E, Jansen GH, Donkervoort S, Bigio EH, Brooks BR, Ajroud K, Sufit RL, Haines JL et al. (2011) Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature 477: 211–215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng HX, Hentati A, Tainer JA, Iqbal Z, Cayabyab A, Hung WY, Getzoff ED, Hu P, Herzfeldt B, Roos RP, Warner C, Deng G, Soriano E, Smyth C, Parge HE, Ahmed A, Rose SA, Hallewell RA, Pericak-Vance MA, Siddique T (1993) Amyotrophic lateral sclerosis and structural defects in Cu,Zn superoxide dismutase. Science 261: 1047–1051 [DOI] [PubMed] [Google Scholar]
- Diaz-Amarilla P, Olivera-Bravo S, Trias E, Cragnolini A, Martinez-Palma L, Cassina P, Beckman J, Barbeito L (2011) Phenotypically aberrant astrocytes that promote motoneuron damage in a model of inherited amyotrophic lateral sclerosis. Proc Natl Acad Sci USA 108: 18126–18131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escartin C, Bonvento G (2008) Targeted activation of astrocytes: a potential neuroprotective strategy. Mol Neurobiol 38: 231–241 [DOI] [PubMed] [Google Scholar]
- Gard AL, White FP, Dutton GR (1985) Extra-neural glial fibrillary acidic protein (GFAP) immunoreactivity in perisinusoidal stellate cells of rat liver. J Neuroimmunol 8: 359–375 [DOI] [PubMed] [Google Scholar]
- Gong YH, Parsadanian AS, Andreeva A, Snider WD, Elliott JL (2000) Restricted expression of G86R Cu/Zn superoxide dismutase in astrocytes results in astrocytosis but does not cause motoneuron degeneration. J Neurosci 20: 660–665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurney ME, Pu H, Chiu AY, Dal Canto MC, Polchow CY, Alexander DD, Caliendo J, Hentati A, Kwon YW, Deng HX, Chen W, Zhai P, Sufit RL, Siddique T (1994) Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation. Science 264: 1772–1775 [DOI] [PubMed] [Google Scholar]
- Howland DS, Liu J, She Y, Goad B, Maragakis NJ, Kim B, Erickson J, Kulik J, DeVito L, Psaltis G, DeGennaro LJ, Cleveland DW, Rothstein JD (2002) Focal loss of the glutamate transporter EAAT2 in a transgenic rat model of SOD1 mutant-mediated amyotrophic lateral sclerosis (ALS). Proc Natl Acad Sci USA 99: 1604–1609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, Tong J, Bi F, Wu Q, Huang B, Zhou H, Xia XG (2012a) Entorhinal cortical neurons are the primary targets of FUS mislocalization and ubiquitin aggregation in FUS transgenic rats. Hum Mol Genet 21: 4602–4614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, Tong J, Bi F, Zhou H, Xia XG (2012b) Mutant TDP-43 in motor neurons promotes the onset and progression of ALS in rats. J Clin Invest 122: 107–118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, Zhou H, Tong J, Chen H, Liu YJ, Wang D, Wei X, Xia XG (2011) FUS Transgenic rats develop the phenotypes of amyotrophic lateral sclerosis and frontotemporal lobar degeneration. PLOS Genetics 7: e1002011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igaz LM, Kwong LK, Lee EB, Chen-Plotkin A, Swanson E, Unger T, Malunda J, Xu Y, Winton MJ, Trojanowski JQ, Lee VM (2011) Dysregulation of the ALS-associated gene TDP-43 leads to neuronal death and degeneration in mice. J Clin Invest 121: 726–738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabashi E, Lin L, Tradewell ML, Dion PA, Bercier V, Bourgouin P, Rochefort D, Bel Hadj S, Durham HD, Vande Velde C, Rouleau GA, Drapeau P (2010) Gain and loss of function of ALS-related mutations of TARDBP (TDP-43) cause motor deficits in vivo. Hum Mol Genet 19: 671–683 [DOI] [PubMed] [Google Scholar]
- Kabashi E, Valdmanis PN, Dion P, Spiegelman D, McConkey BJ, Vande Velde C, Bouchard JP, Lacomblez L, Pochigaeva K, Salachas F, Pradat PF, Camu W, Meininger V, Dupre N, Rouleau GA (2008a) TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis. Nat Genet 40: 572–574 [DOI] [PubMed] [Google Scholar]
- Kabashi E, Valdmanis PN, Dion P, Spiegelman D, McConkey BJ, Velde CV, Bouchard J-P, Lacomblez L, Pochigaeva K, Salachas F, Pradat P-F, Camu W, Meininger V, Dupre N, Rouleau GA (2008b) TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis. Nat Genet 40: 572–574 [DOI] [PubMed] [Google Scholar]
- Kawahara Y, Mieda-Sato A (2012) TDP-43 promotes microRNA biogenesis as a component of the Drosha and Dicer complexes. Proc Natl Acad Sci USA 109: 3347–3352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwiatkowski TJ Jr, Bosco DA, Leclerc AL, Tamrazian E, Vanderburg CR, Russ C, Davis A, Gilchrist J, Kasarskis EJ, Munsat T, Valdmanis P, Rouleau GA, Hosler BA, Cortelli P, de Jong PJ, Yoshinaga Y, Haines JL, Pericak-Vance MA, Yan J, Ticozzi N et al. (2009) Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science 323: 1205–1208 [DOI] [PubMed] [Google Scholar]
- Landreth G, Jiang Q, Mandrekar S, Heneka M (2008) PPARgamma agonists as therapeutics for the treatment of Alzheimer's disease. Neurotherapeutics 5: 481–489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S, Lee J, Kim S, Park JY, Lee WH, Mori K, Kim SH, Kim IK, Suk K (2007) A dual role of lipocalin 2 in the apoptosis and deramification of activated microglia. J Immunol 179: 3231–3241 [DOI] [PubMed] [Google Scholar]
- Lee S, Park JY, Lee WH, Kim H, Park HC, Mori K, Suk K (2009) Lipocalin-2 is an autocrine mediator of reactive astrocytosis. J Neurosci 29: 234–249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepore AC, Rauck B, Dejea C, Pardo AC, Rao MS, Rothstein JD, Maragakis NJ (2008) Focal transplantation–based astrocyte replacement is neuroprotective in a model of motor neuron disease. Nat Neurosci 11: 1294–1301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Ray P, Rao EJ, Shi C, Guo W, Chen X, Woodruff EA, Fushimi K, Wu JY (2010) A Drosophila model for TDP-43 proteinopathy. Proc Natl Acad Sci USA 107: 3169–3174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim MA, Selak MA, Xiang Z, Krainc D, Neve RL, Kraemer BC, Watts JL, Kalb RG (2012) Reduced activity of AMP-activated protein kinase protects against genetic models of motor neuron disease. J Neurosci 32: 1123–1141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matyash V, Kettenmann H (2010) Heterogeneity in astrocyte morphology and physiology. Brain Res Rev 63: 2–10 [DOI] [PubMed] [Google Scholar]
- Naude PJ, Nyakas C, Eiden LE, Ait-Ali D, van der Heide R, Engelborghs S, Luiten PG, De Deyn PP, den Boer JA, Eisel UL (2012) Lipocalin 2: Novel component of proinflammatory signaling in Alzheimer's disease. FASEB J 26: 2811–2823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314: 130–133 [DOI] [PubMed] [Google Scholar]
- Papadeas ST, Kraig SE, O'Banion C, Lepore AC, Maragakis NJ (2011) Astrocytes carrying the superoxide dismutase 1 (SOD1G93A) mutation induce wild-type motor neuron degeneration in vivo. Proc Natl Acad Sci USA 108: 17803–17808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polymenidou M, Lagier-Tourenne C, Hutt KR, Huelga SC, Moran J, Liang TY, Ling SC, Sun E, Wancewicz E, Mazur C, Kordasiewicz H, Sedaghat Y, Donohue JP, Shiue L, Bennett CF, Yeo GW, Cleveland DW (2011) Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat Neurosci 14: 459–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puche JE, Lee YA, Jiao J, Aloman C, Fiel MI, Munoz U, Kraus T, Lee T, Yee HF Jr, Friedman SL (2013) A novel murine model to deplete hepatic stellate cells uncovers their role in amplifying liver damage in mice. Hepatology 57: 339–350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renton AE, Majounie E, Waite A, Simon-Sanchez J, Rollinson S, Gibbs JR, Schymick JC, Laaksovirta H, van Swieten JC, Myllykangas L, Kalimo H, Paetau A, Abramzon Y, Remes AM, Kaganovich A, Scholz SW, Duckworth J, Ding J, Harmer DW, Hernandez DG et al. (2011) A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron 72: 257–268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, Donaldson D, Goto J, O'Regan JP, Deng HX et al. (1993) Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362: 59–62 [DOI] [PubMed] [Google Scholar]
- Rothstein JD, Dykes-Hoberg M, Pardo CA, Bristol LA, Jin L, Kuncl RW, Kanai Y, Hediger MA, Wang Y, Schielke JP, Welty DF (1996) Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 16: 675–686 [DOI] [PubMed] [Google Scholar]
- Rothstein JD, Patel S, Regan MR, Haenggeli C, Huang YH, Bergles DE, Jin L, Dykes Hoberg M, Vidensky S, Chung DS, Toan SV, Bruijn LI, Su ZZ, Gupta P, Fisher PB (2005) Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 433: 73–77 [DOI] [PubMed] [Google Scholar]
- Rutherford NJ, Zhang YJ, Baker M, Gass JM, Finch NA, Xu YF, Stewart H, Kelley BJ, Kuntz K, Crook RJ, Sreedharan J, Vance C, Sorenson E, Lippa C, Bigio EH, Geschwind DH, Knopman DS, Mitsumoto H, Petersen RC, Cashman NR et al. (2008) Novel mutations in TARDBP (TDP-43) in patients with familial amyotrophic lateral sclerosis. PLoS Genet 4: e1000193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sephton CF, Cenik C, Kucukural A, Dammer EB, Cenik B, Han Y, Dewey CM, Roth FP, Herz J, Peng J, Moore MJ, Yu G (2011) Identification of neuronal RNA targets of TDP-43-containing ribonucleoprotein complexes. J Biol Chem 286: 1204–1215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sephton CF, Good SK, Atkin S, Dewey CM, Mayer P 3rd, Herz J, Yu G (2010) TDP-43 is a developmentally regulated protein essential for early embryonic development. J Biol Chem 285: 6826–6834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serio A, Bilican B, Barmada SJ, Ando DM, Zhao C, Siller R, Burr K, Haghi G, Story D, Nishimura AL, Carrasco MA, Phatnani HP, Shum C, Wilmut I, Maniatis T, Shaw CE, Finkbeiner S, Chandran S (2013) Astrocyte pathology and the absence of non-cell autonomy in an induced pluripotent stem cell model of TDP-43 proteinopathy. Proc Natl Acad Sci USA 110: 4697–4702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan X, Chiang PM, Price DL, Wong PC (2010) Altered distributions of Gemini of coiled bodies and mitochondria in motor neurons of TDP-43 transgenic mice. Proc Natl Acad Sci USA 107: 16325–16330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreedharan J, Blair IP, Tripathi VB, Hu X, Vance C, Rogelj B, Ackerley S, Durnall JC, Williams KL, Buratti E, Baralle F, de Belleroche J, Mitchell JD, Leigh PN, Al-Chalabi A, Miller CC, Nicholson G, Shaw CE (2008) TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science 319: 1668–1672 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swarup V, Phaneuf D, Bareil C, Robertson J, Rouleau GA, Kriz J, Julien JP (2011) Pathological hallmarks of amyotrophic lateral sclerosis/frontotemporal lobar degeneration in transgenic mice produced with TDP-43 genomic fragments. Brain 134: 2610–2626 [DOI] [PubMed] [Google Scholar]
- Tanaka K, Watase K, Manabe T, Yamada K, Watanabe M, Takahashi K, Iwama H, Nishikawa T, Ichihara N, Kikuchi T, Okuyama S, Kawashima N, Hori S, Takimoto M, Wada K (1997) Epilepsy and exacerbation of brain injury in mice lacking the glutamate transporter GLT-1. Science 276: 1699–1702 [DOI] [PubMed] [Google Scholar]
- Tollervey JR, Curk T, Rogelj B, Briese M, Cereda M, Kayikci M, Konig J, Hortobagyi T, Nishimura AL, Zupunski V, Patani R, Chandran S, Rot G, Zupan B, Shaw CE, Ule J (2011) Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat Neurosci 14: 452–458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai KJ, Yang CH, Fang YH, Cho KH, Chien WL, Wang WT, Wu TW, Lin CP, Fu WM, Shen CK (2010) Elevated expression of TDP-43 in the forebrain of mice is sufficient to cause neurological and pathological phenotypes mimicking FTLD-U. J Exp Med 207: 1661–1673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uchida A, Sasaguri H, Kimura N, Tajiri M, Ohkubo T, Ono F, Sakaue F, Kanai K, Hirai T, Sano T, Shibuya K, Kobayashi M, Yamamoto M, Yokota S, Kubodera T, Tomori M, Sakaki K, Enomoto M, Hirai Y, Kumagai J et al. (2012) Non-human primate model of amyotrophic lateral sclerosis with cytoplasmic mislocalization of TDP-43. Brain 135: 833–846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaccaro A, Tauffenberger A, Ash PE, Carlomagno Y, Petrucelli L, Parker JA (2012) TDP-1/TDP-43 regulates stress signaling and age-dependent proteotoxicity in Caenorhabditis elegans. PLoS Genet 8: e1002806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Deerlin VM, Leverenz JB, Bekris LM, Bird TD, Yuan W, Elman LB, Clay D, Wood EM, Chen-Plotkin AS, Martinez-Lage M, Steinbart E, McCluskey L, Grossman M, Neumann M, Wu IL, Yang WS, Kalb R, Galasko DR, Montine TJ, Trojanowski JQ et al. (2008) TARDBP mutations in amyotrophic lateral sclerosis with TDP-43 neuropathology: a genetic and histopathological analysis. Lancet Neurol 7: 409–416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vance C, Rogelj B, Hortobagyi T, De Vos KJ, Nishimura AL, Sreedharan J, Hu X, Smith B, Ruddy D, Wright P, Ganesalingam J, Williams KL, Tripathi V, Al-Saraj S, Al-Chalabi A, Leigh PN, Blair IP, Nicholson G, de Belleroche J, Gallo JM et al. (2009) Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science 323: 1208–1211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voigt A, Herholz D, Fiesel FC, Kaur K, Muller D, Karsten P, Weber SS, Kahle PJ, Marquardt T, Schulz JB (2010) TDP-43-mediated neuron loss in vivo requires RNA-binding activity. PLoS ONE 5: e12247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang JW, Brent JR, Tomlinson A, Shneider NA, McCabe BD (2011) The ALS-associated proteins FUS and TDP-43 function together to affect Drosophila locomotion and life span. J Clin Invest 121: 4118–4126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wegorzewska I, Bell S, Cairns NJ, Miller TM, Baloh RH (2009) TDP-43 mutant transgenic mice develop features of ALS and frontotemporal lobar degeneration. Proc Natl Acad Sci USA 106: 18809–18814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wils H, Kleinberger G, Janssens J, Pereson S, Joris G, Cuijt I, Smits V, Ceuterick-de Groote C, Van Broeckhoven C, Kumar-Singh S (2010) TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration. Proc Natl Acad Sci USA 107: 3858–3863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CH, Fallini C, Ticozzi N, Keagle PJ, Sapp PC, Piotrowska K, Lowe P, Koppers M, McKenna-Yasek D, Baron DM, Kost JE, Gonzalez-Perez P, Fox AD, Adams J, Taroni F, Tiloca C, Leclerc AL, Chafe SC, Mangroo D, Moore MJ et al. (2012a) Mutations in the profilin 1 gene cause familial amyotrophic lateral sclerosis. Nature 488: 499–503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu LS, Cheng WC, Shen CK (2012b) Targeted depletion of TDP-43 expression in the spinal cord motor neurons leads to the development of amyotrophic lateral sclerosis-like phenotypes in mice. J Biol Chem 287: 27335–27344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu YF, Gendron TF, Zhang YJ, Lin WL, D'Alton S, Sheng H, Casey MC, Tong J, Knight J, Yu X, Rademakers R, Boylan K, Hutton M, McGowan E, Dickson DW, Lewis J, Petrucelli L (2010) Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice. J Neurosci 30: 10851–10859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao L, Burt AD (2007) The diffuse stellate cell system. J Mol Histol 38: 53–64 [DOI] [PubMed] [Google Scholar]
- Zhou H, Huang C, Chen H, Wang D, Landel CP, Xia PY, Bowser R, Liu YJ, Xia XG (2010) transgenic rat model of neurodegeneration caused by mutation in the TDP gene. PLoS Genetics 6: e1000887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H, Huang C, Yang M, Landel CP, Xia PY, Liu YJ, Xia XG (2009) Developing tTA transgenic rats for inducible and reversible gene expression. Int J Biol Sci 2: 171–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.