Systems analysis of stomatal kinetics identifies the gating of the dominant K+ channels as a promising target for genetic manipulations directed to improving water use efficiency in two plant models.
Abstract
Stomatal transpiration is at the center of a crisis in water availability and crop production that is expected to unfold over the next 20 to 30 years. Global water usage has increased 6-fold in the past 100 years, twice as fast as the human population, and is expected to double again before 2030, driven mainly by irrigation and agriculture. Guard cell membrane transport is integral to controlling stomatal aperture and offers important targets for genetic manipulation to improve crop performance. However, its complexity presents a formidable barrier to exploring such possibilities. With few exceptions, mutations that increase water use efficiency commonly have been found to do so with substantial costs to the rate of carbon assimilation, reflecting the trade-off in CO2 availability with suppressed stomatal transpiration. One approach yet to be explored in detail relies on quantitative systems analysis of the guard cell. Our deep knowledge of transport and homeostasis in these cells gives real substance to the prospect for reverse engineering of stomatal responses, using in silico design in directing genetic manipulation for improved water use and crop yields. Here we address this problem with a focus on stomatal kinetics, taking advantage of the OnGuard software and models of the stomatal guard cell recently developed for exploring stomatal physiology. Our analysis suggests that manipulations of single transporter populations are likely to have unforeseen consequences. Channel gating, especially of the dominant K+ channels, appears the most favorable target for experimental manipulation.
Stomata are pores that provide the major route for gaseous exchange across the impermeable cuticle of leaves and stems (Hetherington and Woodward, 2003). They open and close in response to exogenous and endogenous signals and thereby control the exchange of gases, most importantly water vapor and CO2, between the interior of the leaf and the atmosphere. Stomata exert major controls on the water and carbon cycles of the world (Schimel et al., 2001) and can limit photosynthetic rates by 50% or more when demand exceeds water supply (Ni and Pallardy, 1992). Stomatal transpiration is at the center of a crisis in water availability and crop production that is expected to unfold over the next 20 to 30 years; indeed, global water usage has increased 6-fold in the past 100 years, twice as fast as the human population, and is expected to double again before 2030, driven mainly by irrigation and agriculture (United Nations Educational, Scientific and Cultural Organization, 2009).
Guard cell transport is integral to controlling stomatal aperture. Guard cells surround the stomatal pore and respond in a well-defined manner to an array of extracellular signals, including light, to regulate its aperture. Guard cells coordinate membrane transport within a complex network of intracellular signals (Willmer and Fricker, 1996; Blatt, 2000a, 2000b; Hetherington and Woodward, 2003; Shimazaki et al., 2007) to regulate fluxes, mainly of K+, Cl−, and malate, driving cell turgor and stomatal aperture. Our deep knowledge of these processes has made the guard cell the best known of plant cell models for membrane transport, signaling, and homeostasis (Willmer and Fricker, 1996; Blatt, 2000b; Roelfsema and Hedrich, 2010; Hills et al., 2012). This knowledge gives real substance to the prospect for reverse engineering of stomatal responses, using in silico design in directing genetic manipulation for improved crop yields, especially under water-limited conditions.
Water use efficiency (WUE; defined as the amount of dry matter produced per unit of water transpired) is directly related to stomatal function. Thus, at the practical level, stomata represent an important target for breeders interested in manipulating crop performance. A large body of data relates stomata, transpiration, and carbon assimilation (Willmer and Fricker, 1996; Farquhar et al., 2001; Hetherington and Woodward, 2003; Lawson et al., 2011). Several examples illustrate how manipulating of stomatal characteristics can affect WUE (Fischer et al., 1998; Rebetzke et al., 2002; Masle et al., 2005; Eisenach et al., 2012). With few exceptions, however, mutations that increase WUE commonly do so at the expense of carbon assimilation, reflecting the trade-off in CO2 availability with suppressed stomatal transpiration.
Stomatal movements generally lag behind short-term changes in available light associated with sunflecks and shadeflecks (Pearcy, 1990; Lawson et al., 2012; Lawson and Blatt, 2014). This hysteresis in response, between stomatal aperture and gas exchange on one hand and photosynthetic capacity on the other, can lead alternately to periods of assimilation limited by stomatal conductance, and of high transpiration without corresponding rates of assimilation (Lawson et al., 2011). It has been argued that such hysteresis in stomatal responsiveness with the demand for CO2 erodes assimilation and WUE, with substantial consequences for long-term yield (Vico et al., 2011; Eisenach et al., 2012; Lawson et al., 2012; Lawson and Blatt, 2014). If so, then improving WUE with gains in assimilation should be possible if the speed of stomatal responsiveness can be enhanced. However, the complexity of guard cell transport presents a formidable barrier to exploring such possibilities. Here we address this problem, taking advantage of OnGuard models of the stomatal guard cell. We explore in silico the potential for enhancing stomatal kinetics through single transporter (single gene product) manipulations. Our results identify the gating of the dominant K+ channels as the most promising target for experimental manipulation.
RESULTS AND DISCUSSION
We previously developed the OnGuard software for quantitative dynamic systems modeling of the guard cell (Chen et al., 2012; Hills et al., 2012). OnGuard models incorporate explicitly all of the wealth of molecular, biophysical, and kinetic knowledge for guard cell transport, osmolyte metabolism, and H+ and Ca2+ buffering, and they couple this knowledge to guard cell volume, turgor, and stomatal aperture. OnGuard applications have yielded an abundance of counterintuitive, emergent characteristics in the function of wild-type guard cells of the crop Vicia and the model plant Arabidopsis (Arabidopsis thaliana; Chen et al., 2012; Wang et al., 2012). Applied to the Arabidopsis slac1 anion channel and ost2 ATPase mutants, the latter has also demonstrated new and unexpected connections between membrane transport, metabolism, and stomatal dynamics, uncovering a previously unrecognized homeostatic network that was subsequently verified experimentally (Wang et al., 2012; Blatt et al., 2013). Thus, OnGuard models incorporate the predictive power needed to guide reverse engineering of stomatal function starting with molecular manipulations in silico.
We carried out an analysis of stomatal opening and closing, using OnGuard to predict the consequences of targeted genetic manipulations. At present, OnGuard reflects the characteristics of stomata in isolation (Hills et al., 2012) and does not incorporate feedback from the transpiration stream nor from internal pCO2 in the leaf. As a consequence, kinetic relaxations of OnGuard models are prolonged, especially during opening, but are nonetheless scaled appropriately. The simulations presented here were designed to test the sensitivity to changes over a narrow range of parameters potentially tractable through genetic manipulation for each of the major transporters. We compared these outputs with similar analyses carried out for the wild-type (control) parameter set. For simplicity, each of the transporters was tested with population densities adjusted by factors of 0.5- and 2-fold. We followed a similar approach in varying the macroscopic gating characteristics of several voltage-sensitive ion channels. Outputs were quantified on the basis of the maximum rates of stomatal opening and closing, and on the dynamic range of apertures. Simulations used a standard diurnal cycle with a saw-tooth rise and fall in light over 12 h followed by 12 h of dark; as before, light was used as the driver for Suc and malate synthesis, and for the primary ATPase activities at the two membranes (Chen et al., 2012) employing, as a starting point, the Arabidopsis and Vicia models previously described (Hills et al., 2012; Wang et al., 2012). Analysis of the two models yielded similar results when scaled to the guard cell dimensions. To avoid duplication, only the results for the Arabidopsis model are reported here.
Manipulating Transporter Populations
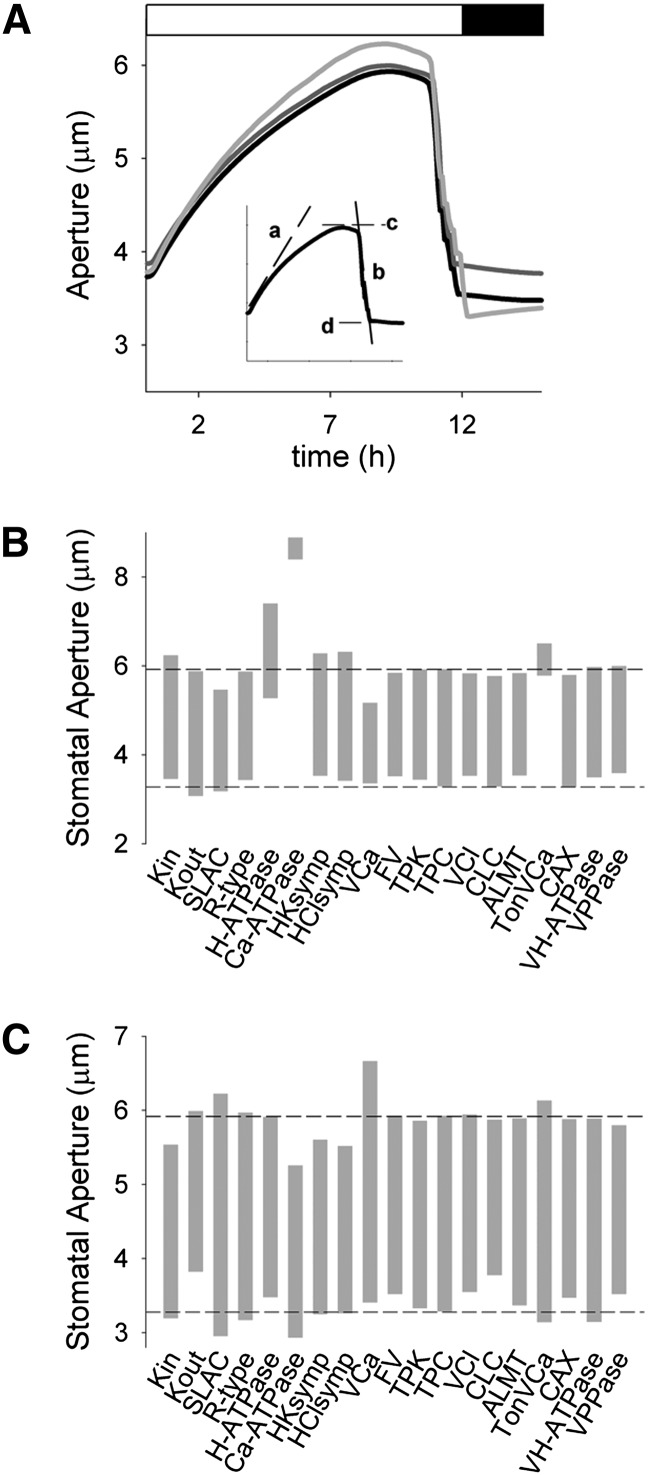
These manipulations are comparable with variations in the steady-state levels of transporter expression such as might be achieved through moderated overexpression and RNA interference-mediated suppression. Figure 1A shows the simulation outputs for the wild type (control) and for 0.5x Kout and SLAC, equivalent to suppressing the population of outward-rectifying K+ channels (Hosy et al., 2003) and SLAC1 anion channels (Negi et al., 2008; Wang et al., 2012) in the guard cell to 50% of the wild type. The daylight period is marked by stomatal opening that progresses over the first 6 to 8 h and, as the light decayed to near its midpoint (K1/2) for activation of malate synthesis and the ATPases at its end, by stomatal closing (Chen et al., 2012; Wang et al., 2012). The Figure 1A inset also provides the outputs used for comparisons. Reducing the K+ and anion channel populations had visible effects on the dynamic range of stomatal movements. It also had substantial, although less obvious, effects on stomatal kinetics. These characteristics are summarized in Figure 1, B and C, and Figures 2 and 3 together with those for each of the other component transporters at the plasma membrane and tonoplast. A full description of each simulation and its outputs is unnecessary, and here we address only the most notable and intriguing of the results.
Figure 1.
Selectively manipulating transporter population identifies a subset of transport activities that strongly influence stomatal dynamics. A, Aperture dynamics, in overlaid sections of 24-h cycles, of stomatal opening and closing simulated under control (black line) conditions and with the population of Kout (dark gray line) and anion channels (SLAC; light gray line) reduced by 50%. Analysis parameters of initial rates of opening (a) and closing (b) and maximum (c) and minimum (d) apertures as indicated. As previously described (Chen et al., 2012; Wang et al., 2012), light was used as a driver for Suc and malic acid synthesis, and for the ion-transporting ATPases at both membranes, and employed a K1/2 for light intensity. The light cycle was driven through a sawtooth rise and fall over 12 h followed by 12 h of dark. Only part of the 12-h-dark period is shown, as indicated above with the white and black bars showing the transition times between light and dark. B and C, Dynamic range of apertures on elevating by 2-fold (B) and reducing to 50% (C) the population of each transporter. Horizontal lines indicate the dynamic range of the control simulation for comparison. Transporters at the plasma membrane are as follows: HClsymp, H+-Cl− symport; HKsymp, H+-K+ symport; R-type, R-type anion channel; SLAC, SLAC1-type anion channel; VCa, voltage-gated (inward-rectifying) Ca2+ channel. Transporters at the tonoplast are as follows: ALMT, voltage-gated ALMT-type (malate-permeable) anion channel; CAX, Ca2+-H+ antiporter; CLC, CLC-type H+-Cl− antiporter; FV, FV-type K+ channel; TonVCa, voltage- and Ca2+-gated Ca2+ channel; TPC, TPC1-type cation channel; TPK, TPK1-type K+ channel; VCl, voltage-gated (inward-rectifying) Cl− channel; VH-ATPase, V-type H+-ATPase; VPPase, H+-PPase. The properties and functions of each of these transporters are summarized in Hills et al. (2012).
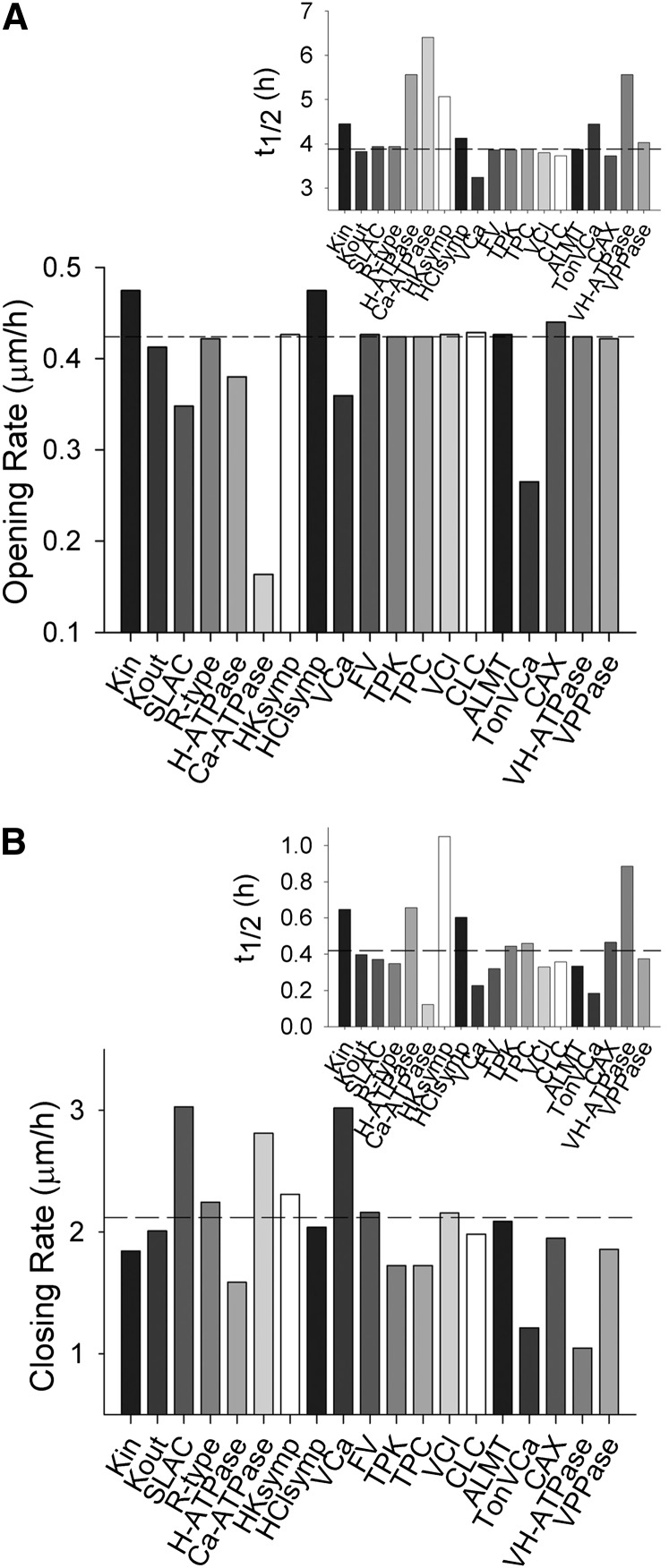
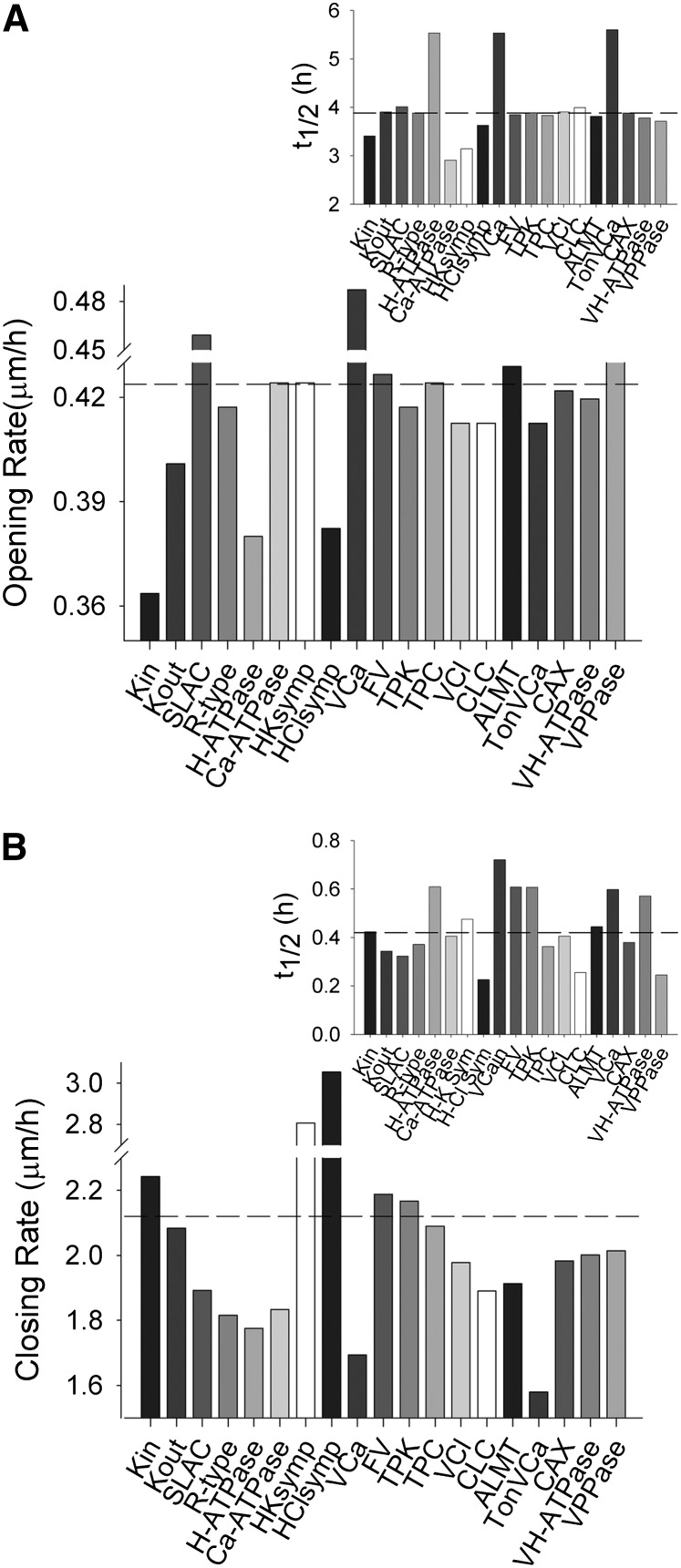
Figure 2.
Selectively elevating transporter populations by 2-fold in simulations yields a subset of transport activities that affect the initial kinetics and the halftimes for response, but invariably affect both stomatal opening and closing. Transporter types are described in the legend to Figure 1. A, Initial rates of stomatal opening for elevation of each transporter population. The inset shows halftimes for opening. Horizontal lines in each case indicate the opening rate and halftime for the control simulation. B, Initial rates of stomatal closing for elevation of each transporter population. The inset shows halftimes for closing. Horizontal lines in each case indicate the closing rate and halftime for the control simulation.
Figure 3.
Selectively reducing transporter populations by 50% in simulations yields a subset of transport activities that affect the initial kinetics and the halftimes for response, but invariably affect both stomatal opening and closing. Transporter types are described in the legend to Figure 1. A, Initial rates of stomatal opening for elevation of each transporter population. The inset shows halftimes for opening. Horizontal lines in each case indicate the opening rate and halftime for the control simulation. B, Initial rates of stomatal closing for elevation of each transporter population. The inset shows halftimes for closing. Horizontal lines in each case indicate the closing rate and halftime for the control simulation.
Figure 1, B and C, highlight both the sensitivity of simulations to a subset of transporters, primarily at the plasma membrane, and an asymmetry in efficacy between 0.5- and 2-fold changes in transporter populations. It also underscores the counterintuitive effects of manipulating many of these transporters. For example, the inward-rectifying K+ channel (Kin), characterized by KAT1 in Arabidopsis (Nakamura et al., 1995), and the H+-K+ symporter (Rodriguez-Navarro et al., 1986; Blatt and Slayman, 1987; Maathuis and Sanders, 1994) serve as the two pathways for K+ uptake. Increasing the densities of either of these transporters elevated the aperture maximum without effect on the minimum (Fig. 1B); however, reducing their densities suppressed both the maximum and minimum aperture values (Fig. 1C). An equally unexpected but inverted pattern was evident for Kout. By contrast, elevating the SLAC density reduced the aperture minimum and maximum, but reducing SLAC density led to an expanded dynamic range with a higher aperture maximum and, counterintuitively, a lower minimum. This asymmetry could be traced to the predicted dynamics in cytosolic-free [Ca2+] ([Ca2+]i), which is subject to membrane voltage (Grabov and Blatt, 1998; Sokolovski and Blatt, 2007) and feeds back on plasma membrane anion channel activities (Chen et al., 2012; Wang et al., 2012). In support of this conclusion, we note the parallel in asymmetry of manipulating transport affecting [Ca2+]i. For example, increasing the density of the Ca2+-ATPase strongly elevated stomatal apertures, both the minimum and maximum; reducing the pump density reduced both the minimum and maximum, albeit with little effect on the dynamic range. Conversely, increasing the density of the hyperpolarization-activated Ca2+ channels (VCa) at the plasma membrane (Hamilton et al., 2000) suppressed the aperture maximum and dynamic range, whereas reducing Ca2+ channel density had the opposite effect. These characteristics are wholly consistent with the roles of these transporters in facilitating [Ca2+]i elevation (low Ca2+-ATPase, high Ca2+ channel densities) or its suppression (high Ca2+-ATPase, low Ca2+ channel densities; McAinsh and Pittman, 2009; Pittman, 2011).
One fundamental prediction of OnGuard models, still to be demonstrated experimentally, is the existence of the endomembrane (vacuolar) Ca2+ channel, TonVCa (Chen et al., 2012; Hills et al., 2012). Essential features of this channel are its activation by [Ca2+]i at submicromolar free concentrations and its inactivation at micromolar [Ca2+]i, analogous to the inositol-trisphosphate receptor channels in animals (Bezprozvanny et al., 1991; Hills et al., 2012). These characteristics give rise to prolonged oscillations in [Ca2+]i that are thought to be essential in driving K+ and Cl− efflux for rapid stomatal closure (Grabov and Blatt, 1998; Blatt, 2000b; McAinsh and Pittman, 2009; Chen et al., 2012; Blatt et al., 2013). The counterintuitive effect of increasing TonVCa on aperture can be ascribed directly to the loss in these oscillations, arising from a reduction in the time-averaged total [Ca2+] from roughly 24 mm to 13 mm and a corresponding reduction in Ca2+ release. By contrast, decreasing the TonVCa population increased Ca2+ accumulation in the vacuole and the dynamic range of apertures. In short, manipulations affecting [Ca2+]i are predicted to have the most profound effects on aperture, again in a seemingly counterintuitive manner. We note that Conn et al. (2011) reported an opposing effect associated with the Arabidopsis cax1/cax3 double mutant lacking two of the dominant Ca2+/H+ exchangers that normally serve to sequester Ca2+ in endomembrane compartments. This mutant reduced Ca2+ accumulation in the leaf epidermis, the bulk of this in epidermal cells, and impaired stomatal dynamics and gas exchange. Their interpretation centered on elevated apoplastic [Ca2+], which is known to reduce stomatal apertures (de Silva et al., 1996; Webb et al., 2001). A direct comparison is complicated by a lack of data on the Ca2+ contents of the guard cells, and by the fixed apoplastic [Ca2+] in our simulations. Nonetheless, the much reduced dynamic range in simulated apertures is consistent with the experimental findings.
In simulations with the H+-ATPase, a 2-fold increase in population at the plasma membrane displaced the dynamic range of apertures to higher values, whereas decreasing the number of pumps to 50% of the control was marginal in effect. Here the asymmetry is the direct consequence of the nonlinearity in pump capacity (Blatt, 1987, 1988) and its parallel to that anticipated for charge balance through H+-coupled transport (Sanders et al., 1985; Blatt and Slayman, 1987; Blatt and Clint, 1989; Maathuis and Sanders, 1994; Hills et al., 2012) and the inward-rectifying K+ channels. Among others, the rise in aperture with H+-ATPase activity is consistent with the known effects of locking the H+-ATPase in an active state, either with the fungal toxin fusicoccin or in the ost2 mutant (MacRobbie, 1988; Fuglsang et al., 1999; Merlot et al., 2007).
A complementary pattern was evident in the predicted rates for stomatal opening and closing. Figures 2 and 3 show the predicted rates for 2- and 0.5-fold changes in the numbers of each transporter, and insets within each figure summarize the halftimes for opening and closing. Again, the consequences of any one manipulation are generally evident in both opening and closing rates, often in counterintuitive ways that connect through changes in cytosolic pH and [Ca2+]i. For example, elevating the H+-ATPase population led to decreases in both initial opening and closing rates and substantial increases in the halftimes for both processes (Fig. 2). These effects arose, in part, from a small elevation in cytosolic pH and the enhanced H+ flux through coupled transport and malate metabolism, from plasma membrane hyperpolarization facilitating Ca2+ entry and [Ca2+]i elevation, and from the reduced dynamic range of apertures. Even more surprising, modest reductions in opening and closing rates were also observed when the H+-ATPase density was reduced (Fig. 3). In both cases, the effects can be ascribed to 0.1- to 0.2-pH unit shifts in cytosolic pH and, most important, in changes to [Ca2+]i arising from the effects on the driving force for Ca2+ entry across the plasma membrane; in turn, these effects on cytosolic pH and [Ca2+]i alternately affected K+ flux through Kin and Kout and the balancing activities of the SLAC and R-type anion currents, much as documented by Wang et al. (2012).
It is noteworthy that several of the comparative predictions in Figure 2 are now confirmed. Coincident with the review of this article, independent experimental work was published by Wang et al. (2014), who demonstrated that a 3-fold overexpression of the AHA2 H+-ATPase driven by a guard cell-specific promoter significantly enhanced stomatal conductance and opening in the light, and led to a small increase in carbon assimilation. Similar overexpression of two inward-rectifying K+ channels, KAT1, which is normally expressed in guard cells, and AKT1, which is primarily found in roots, were without substantial effect on stomatal opening. They also found that overexpression of the H+-ATPase reduced WUE, suggesting that this enhanced activity had other consequences for stomatal behavior, possibly in altering stomatal closure (Lawson and Blatt, 2014).
Manipulating Channel Gating
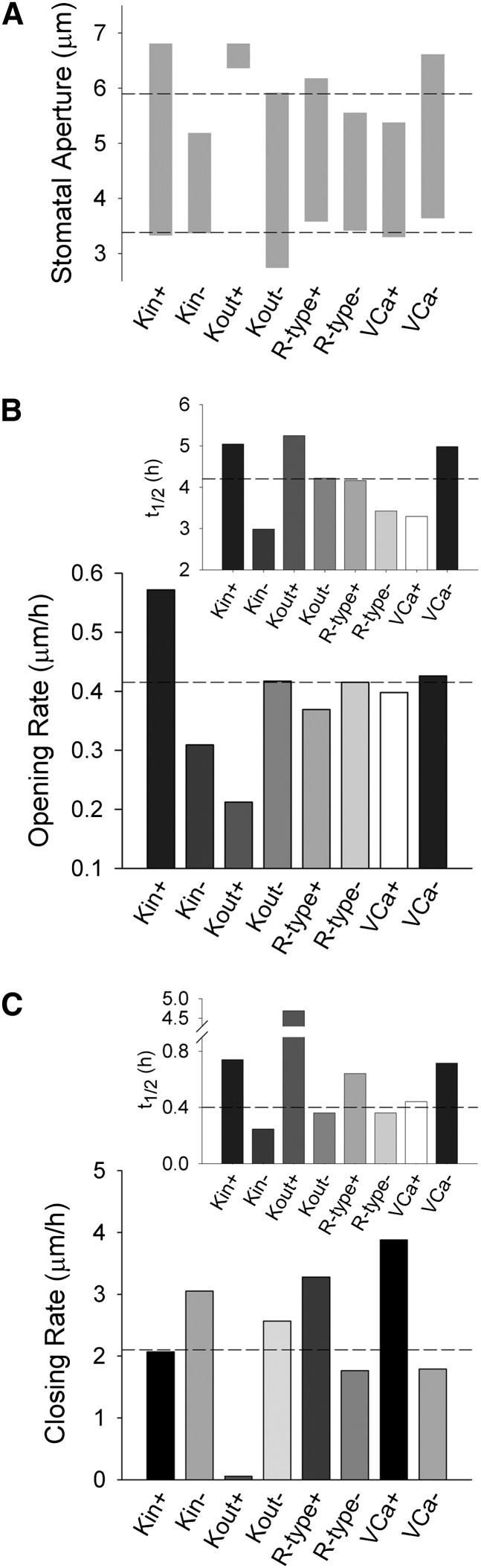
The midpoint voltage (V1/2) for the gating of voltage-sensitive channels is subject to experimental modification (Tao et al., 2010; Pless et al., 2011), and mutations affecting V1/2 are known for KAT1 (C. Lefoulon, C. Grefen, and M.R. Blatt, unpublished data). We found that stomatal aperture was equally sensitive in simulation to altering this parameter in the dominant ion channels at the plasma membrane. Uniquely for the K+ channels, the effects were restricted primarily either to the opening or to the closing process. Most notable, manipulations of Kout to give an −18 mV shift in V1/2 reduced the aperture minimum without substantial change to its maximum and accelerated closing (Fig. 4A). A corresponding +18 mV shift in V1/2 for Kin affected primarily the aperture maximum, again without a proportional change in the minimum; similarly, it accelerated the initial opening rate by roughly 35% compared with the control without affecting closing. For manipulations of both Kin and Kout, the increases in halftimes related directly to the corresponding expansion in the dynamic ranges of apertures. These findings contrast with altering the gating characteristics of the SLAC current, which had compound effects on both opening and closing kinetics when assessed against changes in the dynamic ranges of apertures. Much the same conclusion was drawn from analysis of Ca2+ channel gating.
Figure 4.
Altering the voltage ranges activating the major ion channels at the plasma membrane in simulations identifies the K+ channels as targets for selectively enhancing stomatal opening and closing. Label signs (+ and −) refer to the midpoint voltage for gating (V1/2) displacement from the control, either +18 mV or −18 mV. A, Dynamic range of apertures. Horizontal lines indicate the dynamic range of the control simulation for comparison. B, Initial rates of stomatal opening. The inset shows halftimes for opening. Horizontal lines in each case indicate the opening rate and halftime for the control simulation. C, Initial rates of stomatal closing. The inset shows halftimes for closing. Horizontal lines in each case indicate the closing rate and halftime for the control simulation.
What defines the singular nature of Kin, and especially of Kout, to delimit the effects of manipulating their gating? Analysis of the underlying flux homeostasis for Kout, illustrated in Supplemental Figures S1 to S6, shows principally an absence of effect on either cytosolic pH or [Ca2+]i. Details are provided in the legends to Supplemental Figures S1 to S6, but can be summarized here. Kout normally activates together with anion currents by depolarization of the plasma membrane. Within the range of membrane voltages that activate Kout, changes to its activity have little impact on H+ or Ca2+ flux across the plasma membrane, either through primary or secondary transport. Consequently, any changes in K+ current are balanced primarily by alterations in anion efflux. So virtually all of the anticipated benefit of increasing the Kout population is offset by a change in free-running voltage as K+ efflux adjusts with that of the anions. The voltage dependence of Kout and the anion channels are complementary, but for Kout this dependence is offset to voltages substantially positive of the anion channels, especially at millimolar K+ concentrations and above. In these circumstances, displacing V1/2 for Kout by −18 mV allows for K+ and anion fluxes to balance at marginally more negative voltages, thereby yielding an enhanced capacity for the combined ion efflux. Otherwise, changes to Kout gating had no substantial consequences for the Ca2+ and voltage oscillations that are thought to drive solute efflux and stomatal closure.
In conclusion, we suggest that the gating characteristics of the dominant K+ channels offer potential targets for genetic manipulation as a proof-of-principle in improving the WUE of plants. For reasons outlined above, it has been hypothesized that temporal mismatches between stomatal aperture and available light for photosynthesis significantly erode assimilation and WUE in fluctuating environments (Pearcy, 1990; Lawson et al., 2012; Lawson and Blatt, 2014). Thus, the challenge reduces to that of identifying, as candidates for manipulation, transporters that accelerate opening and closing, ideally both, without substantial constriction in the dynamic range of apertures. Achieving this goal has proven difficult, at least in simulation, because changes in transporter populations that were most effective in accelerating stomatal movements also led to counterintuitive and opposing effects on the complementary process, whether closing or opening. These findings lead us to discount simpler approaches that rely on altered expression of the wild-type transporters. It is important to recognize that these simulations are dissociated from feedback mechanisms affecting stomatal response through relative humidity, water supply, and pCO2 within the leaf. In addition, the simulations do not account for the integrated sink strength of a crop (Baldocchi and Bowling, 2003), which may amplify the hysteresis in stomatal kinetics at leaf level by interactions between the multiple layers of a crop canopy. The consequences of manipulating channel gating as described here may well underestimate the impact on stomatal movements.
MATERIALS AND METHODS
OnGuard Modeling
The OnGuard software and model was driven through a diurnal 12-h-light/12-h-dark cycle as previously described (Chen et al., 2012) and all model outputs were derived from this cycle. Model parameters for Vicia (Hills et al., 2012) and Arabidopsis (Arabidopsis thaliana; Wang et al., 2012) were as described, the latter adjusted to reflect the physical dimensions of the Arabidopsis stomatal complex and transporter numbers scaled accordingly. Light sensitivity was assigned with a K1/2 of 50 μmol m−2 s−1 solely to the plasma membrane H+-ATPase and Ca2+-ATPase, the vacuolar V-type H+-ATPase, H+-PPase and Ca2+-ATPase, and to Suc (and, hence, malic acid) synthesis in accordance with experimental observation (Chen et al., 2012). All other model parameters were fixed, the properties of the individual transporters and buffering reactions thus responding only to changes in model variables arising from the kinetic features encoded in the model. The OnGuard software and the Arabidopsis model used in these simulations are available at http://www.psrg.org.uk.
Supplemental Data
The following materials are available in the online version of this article.
Supplemental Figure S1. Macroscopic outputs from the OnGuard model.
Supplemental Figure S2. K+ contents and analysis of K+ fluxes at the plasma membrane and tonoplast.
Supplemental Figure S3. Cytosolic and vacuolar pH, and H+ fluxes across the plasma membrane and tonoplast.
Supplemental Figure S4. Chloride contents and analysis of Cl− fluxes at the plasma membrane and tonoplast.
Supplemental Figure S5. Malic acid synthesis, total malate (Mal) contents, and analysis of Mal2− fluxes at the plasma membrane and tonoplast.
Supplemental Figure S6. Cytosolic and vacuolar [Ca2+], cytosolic-free [Ca2+], and analysis of Ca2+ fluxes across the plasma membrane and tonoplast.
Supplementary Material
Glossary
- WUE
water use efficiency
- Kout
outward-rectifying K+ channel
- K1/2
midpoint K
- Kin
inward-rectifying K+ channel
- [Ca2+]i
cytosolic-free [Ca2+]
- H+-ATPase
proton-translocating adenosine triphosphatase
- V1/2
midpoint voltage
Footnotes
This work was supported by the Biotechnology and Biological Sciences Research Council (grant nos. BB/H024867/1, BB/F001630/1, BB/L001276/1, and BB/H009817/1 to M.R.B.).
The online version of this article contains Web-only data.
Articles can be viewed online without a subscription.
References
- Baldocchi DD, Bowling DR. (2003) Modelling the discrimination of 13CO2 above and within a temperate broad-leaved forest canopy on hourly to seasonal time scales. Plant Cell Environ 26: 231–244 [Google Scholar]
- Bezprozvanny I, Watras J, Ehrlich BE. (1991) Bell-shaped calcium-response curves of Ins(1,4,5)P3- and calcium-gated channels from endoplasmic reticulum of cerebellum. Nature 351: 751–754 [DOI] [PubMed] [Google Scholar]
- Blatt MR. (1987) Electrical characteristics of stomatal guard cells: the contribution of ATP-dependent, “electrogenic” transport revealed by current-voltage and difference-current-voltage analysis. J Membr Biol 98: 257–274 [Google Scholar]
- Blatt MR. (1988) Mechanisms of fusicoccin action: a dominant role for secondary transport in a higher-plant cell. Planta 174: 187–200 [DOI] [PubMed] [Google Scholar]
- Blatt MR. (2000a) Ca(2+) signalling and control of guard-cell volume in stomatal movements. Curr Opin Plant Biol 3: 196–204 [PubMed] [Google Scholar]
- Blatt MR. (2000b) Cellular signaling and volume control in stomatal movements in plants. Annu Rev Cell Dev Biol 16: 221–241 [DOI] [PubMed] [Google Scholar]
- Blatt MR, Clint GM. (1989) Mechanisms of fusicoccin action: kinetic modification and inactivation of K(+) channels in guard cells. Planta 178: 509–523 [DOI] [PubMed] [Google Scholar]
- Blatt MR, Slayman CL. (1987) Role of “active” potassium transport in the regulation of cytoplasmic pH by nonanimal cells. Proc Natl Acad Sci USA 84: 2737–2741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blatt MR, Wang Y, Leonhardt N, Hills A. (2013) Exploring emergent properties in cellular homeostasis using OnGuard to model K(+) and other ion transport in guard cells. J Plant Physiol (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen ZH, Hills A, Bätz U, Amtmann A, Lew VL, Blatt MR. (2012) Systems dynamic modeling of the stomatal guard cell predicts emergent behaviors in transport, signaling, and volume control. Plant Physiol 159: 1235–1251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conn SJ, Gilliham M, Athman A, Schreiber AW, Baumann U, Moller I, Cheng NH, Stancombe MA, Hirschi KD, Webb AAR, et al. (2011) Cell-specific vacuolar calcium storage mediated by CAX1 regulates apoplastic calcium concentration, gas exchange, and plant productivity in Arabidopsis. Plant Cell 23: 240–257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Silva DLR, Honour SJ, Mansfield TA. (1996) Estimations of apoplastic concentrations of K+ and Ca2+ in the vicinity of stomatal guard cells. New Phytol 134: 463–469 [Google Scholar]
- Eisenach C, Chen ZH, Grefen C, Blatt MR. (2012) The trafficking protein SYP121 of Arabidopsis connects programmed stomatal closure and K+ channel activity with vegetative growth. Plant J 69: 241–251 [DOI] [PubMed] [Google Scholar]
- Farquhar GD, von Caemmerer S, Berry JA, (2001) Models of photosynthesis. Plant Physiol 125: 42–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer MJE, Paulussen JJC, Tollenaere JP, De Mol NJ, Janssen LHM. (1998) Structure-activity relationships of astemizole derivatives for inhibition of store operated Ca2+ channels and exocytosis. Eur J Pharmacol 350: 353–361 [DOI] [PubMed] [Google Scholar]
- Fuglsang AT, Visconti S, Drumm K, Jahn T, Stensballe A, Mattei B, Jensen ON, Aducci P, Palmgren MG. (1999) Binding of 14-3-3 protein to the plasma membrane H(+)-ATPase AHA2 involves the three C-terminal residues Tyr(946)-Thr-Val and requires phosphorylation of Thr(947). J Biol Chem 274: 36774–36780 [DOI] [PubMed] [Google Scholar]
- Grabov A, Blatt MR. (1998) Membrane voltage initiates Ca2+ waves and potentiates Ca2+ increases with abscisic acid in stomatal guard cells. Proc Natl Acad Sci USA 95: 4778–4783 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton DWA, Hills A, Kohler B, Blatt MR. (2000) Ca2+ channels at the plasma membrane of stomatal guard cells are activated by hyperpolarization and abscisic acid. Proc Natl Acad Sci USA 97: 4967–4972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hetherington AM, Woodward FI. (2003) The role of stomata in sensing and driving environmental change. Nature 424: 901–908 [DOI] [PubMed] [Google Scholar]
- Hills A, Chen ZH, Amtmann A, Blatt MR, Lew VL. (2012) OnGuard, a computational platform for quantitative kinetic modeling of guard cell physiology. Plant Physiol 159: 1026–1042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosy E, Vavasseur A, Mouline K, Dreyer I, Gaymard F, Porée F, Boucherez J, Lebaudy A, Bouchez D, Very AA, et al. (2003) The Arabidopsis outward K+ channel GORK is involved in regulation of stomatal movements and plant transpiration. Proc Natl Acad Sci USA 100: 5549–5554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson T, Blatt MR. (February 27, 2014) Size, speed and responsiveness of stomata impact on photosynthesis and water use efficiency. Plant Physiol [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson T, Kramer DM, Raines CA. (2012) Improving yield by exploiting mechanisms underlying natural variation of photosynthesis. Curr Opin Biotechnol 23: 215–220 [DOI] [PubMed] [Google Scholar]
- Lawson T, von Caemmerer S, Baroli I. (2011) Photosynthesis and stomatal behaviour. Prog Bot 72: 265–304 [Google Scholar]
- Maathuis FJ, Sanders D. (1994) Mechanism of high-affinity potassium uptake in roots of Arabidopsis thaliana. Proc Natl Acad Sci USA 91: 9272–9276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacRobbie EAC. (1988) Stomatal guard cells. In Baker DA, Hall JL, eds, Solute Transport in Plant Cells and Tissues, Ed 1 Longman Press, Harlow, UK, pp 453–497 [Google Scholar]
- Masle J, Gilmore SR, Farquhar GD. (2005) The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature 436: 866–870 [DOI] [PubMed] [Google Scholar]
- McAinsh MR, Pittman JK. (2009) Shaping the calcium signature. New Phytol 181: 275–294 [DOI] [PubMed] [Google Scholar]
- Merlot S, Leonhardt N, Fenzi F, Valon C, Costa M, Piette L, Vavasseur A, Genty B, Boivin K, Müller A, et al. (2007) Constitutive activation of a plasma membrane H(+)-ATPase prevents abscisic acid-mediated stomatal closure. EMBO J 26: 3216–3226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura RL, McKendree WL, Jr, Hirsch RE, Sedbrook JC, Gaber RF, Sussman MR. (1995) Expression of an Arabidopsis potassium channel gene in guard cells. Plant Physiol 109: 371–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negi J, Matsuda O, Nagasawa T, Oba Y, Takahashi H, Kawai-Yamada M, Uchimiya H, Hashimoto M, Iba K. (2008) CO2 regulator SLAC1 and its homologues are essential for anion homeostasis in plant cells. Nature 452: 483–486 [DOI] [PubMed] [Google Scholar]
- Ni BR, Pallardy SG. (1992) Stomatal and nonstomatal limitations to net photosynthesis in seedlings of woody angiosperms. Plant Physiol 99: 1502–1508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearcy RW. (1990) Sunflecks and photosynthesis in plant canopies. Ann Rev Plant Physiol Plant Mol Biol 41: 421–453 [Google Scholar]
- Pittman JK. (2011) Vacuolar Ca(2+) uptake. Cell Calcium 50: 139–146 [DOI] [PubMed] [Google Scholar]
- Pless SA, Galpin JD, Niciforovic AP, Ahern CA. (2011) Contributions of counter-charge in a potassium channel voltage-sensor domain. Nat Chem Biol 7: 617–623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rebetzke GJ, Condon AG, Richards RA, Farquhar GD. (2002) Selection for reduced carbon isotope discrimination increases aerial biomass and grain yield of rainfed bread wheat. Crop Sci 42: 739–745 [Google Scholar]
- Rodriguez-Navarro A, Blatt MR, Slayman CL. (1986) A potassium-proton symport in Neurospora crassa. J Gen Physiol 87: 649–674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roelfsema MR, Hedrich R. (2010) Making sense out of Ca(2+) signals: their role in regulating stomatal movements. Plant Cell Environ 33: 305–321 [DOI] [PubMed] [Google Scholar]
- Sanders D, Smith FA, Walker NA. (1985) Proton/chloride cotransport in Chara: mechanism of enhanced influx after rapid external acidification. Planta 163: 411–418 [DOI] [PubMed] [Google Scholar]
- Schimel DS, House JI, Hibbard KA, Bousquet P, Ciais P, Peylin P, Braswell BH, Apps MJ, Baker D, Bondeau A, et al. (2001) Recent patterns and mechanisms of carbon exchange by terrestrial ecosystems. Nature 414: 169–172 [DOI] [PubMed] [Google Scholar]
- Shimazaki KI, Doi M, Assmann SM, Kinoshita T. (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58: 219–247 [DOI] [PubMed] [Google Scholar]
- Sokolovski S, Blatt MR. (2007) Nitric oxide and plant ion channel control. In Lamattina L, Polacco JC, eds, Nitric Oxide in Plant Growth, Development and Stress Physiology, Ed 1 Springer, Berlin, pp 153–172 [Google Scholar]
- Tao X, Lee A, Limapichat W, Dougherty DA, MacKinnon R. (2010) A gating charge transfer center in voltage sensors. Science 328: 67–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- United Nations Educational, Scientific and Cultural Organization (2009) Water in a Changing World–UN World Water Development Report 3. United Nations Educational, Scientific and Cultural Organization, New York [Google Scholar]
- Vico G, Manzoni S, Palmroth S, Katul G. (2011) Effects of stomatal delays on the economics of leaf gas exchange under intermittent light regimes. New Phytol 192: 640–652 [DOI] [PubMed] [Google Scholar]
- Wang Y, Noguchi K, Ono N, Inoue S, Terashima I, Kinoshita T. (2014) Overexpression of plasma membrane H+-ATPase in guard cells promotes light-induced stomatal opening and enhances plant growth. Proc Natl Acad Sci USA 111: 533–538 (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Papanatsiou M, Eisenach C, Karnik R, Williams M, Hills A, Lew VL, Blatt MR. (2012) Systems dynamic modeling of a guard cell Cl- channel mutant uncovers an emergent homeostatic network regulating stomatal transpiration. Plant Physiol 160: 1956–1967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webb AAR, Larman MG, Montgomery LT, Taylor JE, Hetherington AM. (2001) The role of calcium in ABA-induced gene expression and stomatal movements. Plant J 26: 351–362 [DOI] [PubMed] [Google Scholar]
- Willmer C, Fricker MD. (1996) Stomata, Vol 2 Chapman and Hall, London [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.