Abstract
The intricate and complex interaction between different populations of neurons in the brain has imposed limits on our ability to gain detailed understanding of synaptic transmission and its integration when employing classical electrophysiological approaches. Indeed, electrical field stimulation delivered via traditional microelectrodes does not permit the targeted, precise and selective control of neuronal activity amongst a varied population of neurons and their inputs (eg, cholinergic, dopaminergic or glutamatergic neurons). Recently established optogenetic techniques overcome these limitations allowing precise control of the target neuron populations, which is essential for the elucidation of the neural substrates underlying complex animal behaviors. Indeed, by introducing light-activated channels (ie, microbial opsin genes) into specific neuronal populations, optogenetics enables non-invasive optical control of specific neurons with milliseconds precision. These approaches can readily be applied to freely behaving live animals. Recently there is increased interests in utilizing optogenetics tools to understand synaptic plasticity and learning/memory. Here, we summarize recent progress in applying optogenetics in in the study of synaptic plasticity.
Keywords: synaptic plasticity, optogenetics, opsin, channelrhodopsin, halorhodopsin, LTP, LTD, learning and memory
Introduction
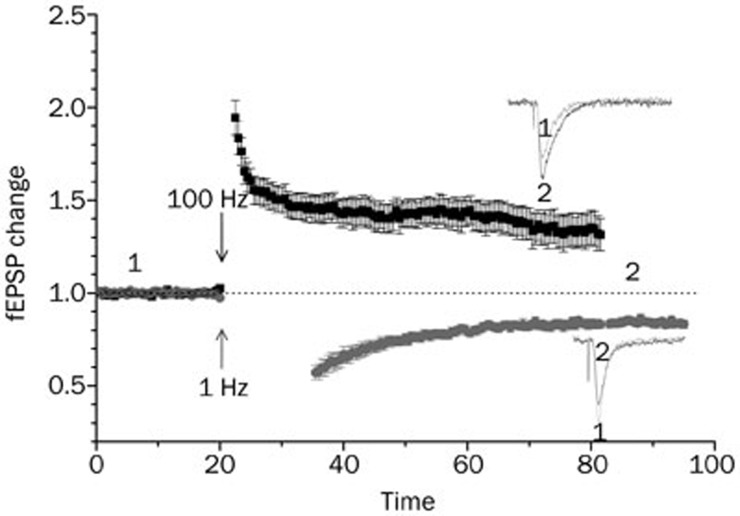
As a fundamental mechanism of learning and memory, synaptic plasticity has been extensively studied using conventional electrophysiological and pharmacological techniques in naïve or transgenic animals, as shown in Figure 11. However, the precise mechanism of synaptic plasticity has not yet been elucidated in detail. Furthermore, findings from studies employing these traditional approaches sometimes remain controversial and require further study2,3,4. The controversial results may at least partially be accounted for by the limitations of classical electrophysiological or pharmacological protocols that induce synaptic plasticity. Such limitations include difficulties in the isolation of individual synaptic responses or different neuronal populations within the electrical field or chemical perfusion area, identification of response location, and loss of an effective time window for LTP or LTD induction.
Figure 1.
The NMDAR-dependent LTP and LTD induced by different electrical stimulation protocols. Inserts represent fEPSP before (1) and after (2) electrical stimulation at different frequencies.
These limitations may be overcome by a recently established technique termed optogenetics. Delivery of genes that encode light-activated channels (opsins) to be expressed within specific neuron populations allows these neurons to be selectively activated or inhibited by different wavelengths of light5. Because of its specificity, precise reversibility, and success in rat models6, this technique will open new avenues for the study of synaptic plasticity and learning/memory-related diseases.
Opsins, optogenetics, and their application in neuroscience
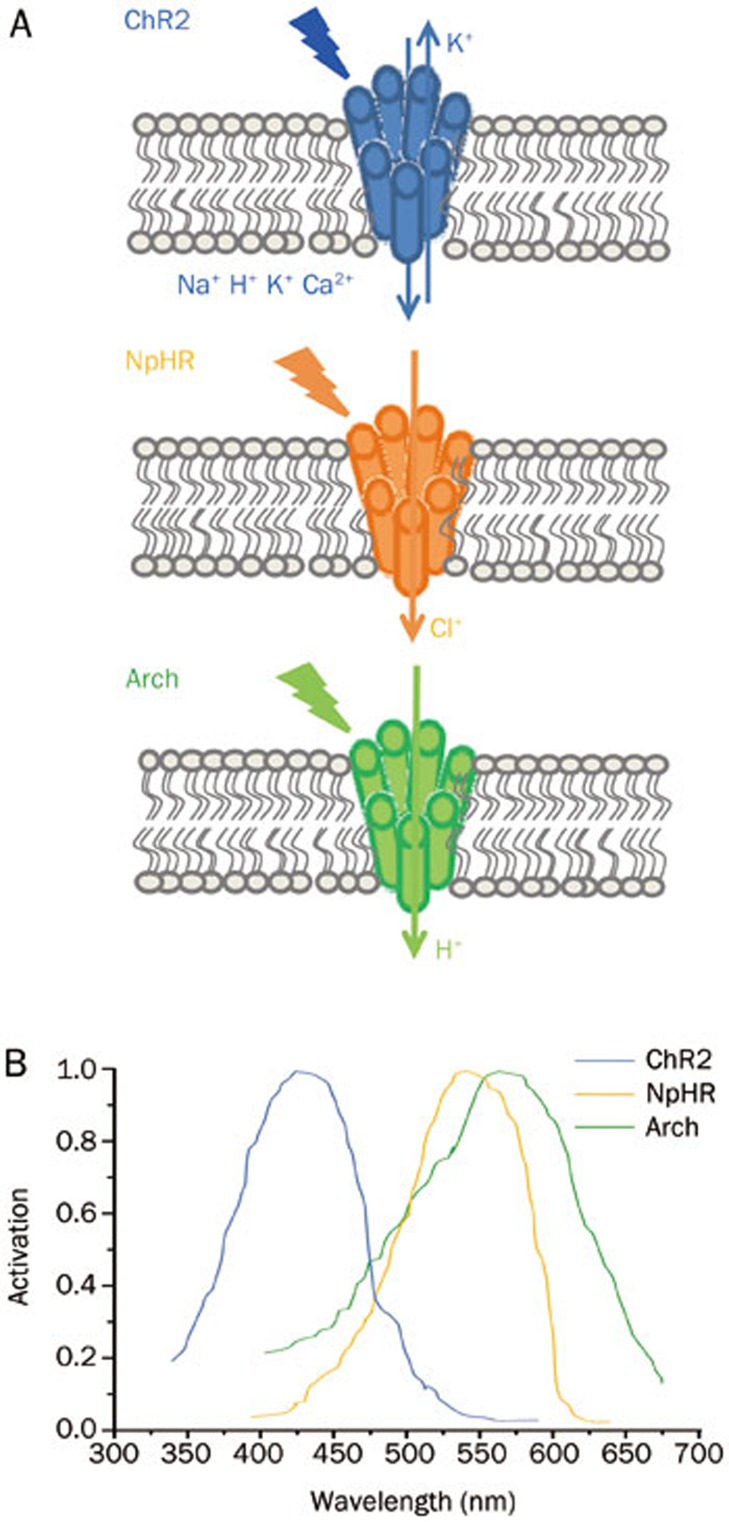
Optogenetics is a technique that takes advantage of opsins, which are found in retinal photoreceptor cells and are activated by light at specific wavelengths. This technology combines genetic targeting of specific neurons with optical control of those neurons to achieve spatiotemporally precise control of specific neurons with millisecond-scale pulses of light. Several families of opsins have been developed through modifications by multiple engineering methods, including channelrhodopsin (ChR), halorhodopsin (HR) and bacteriorhodopsin (BR)7. ChR 2 (ChR2), the first opsin used in neurons to induce depolarization8, is a cation-selective channel for Na+, H+, K+, and Ca2+ following activation by blue light at 475 nm wavelength9 (Figure 2). In addition, different variants of ChR have been engineered to develop an expanded toolkit of channels with distinct electrical characteristics, including CatCh (calcium permeability-enhanced ChR)10, ChIEF11, ChETA12, and VChR113. HR is a light-driven inward chloride pump14 derived from the archaeon Natronomonas pharaonis (Halo/NpHR) and is genetically expressed in neurons to induce hyperpolarization by yellow light at 593 nm15 (Figure 2). BR is microbial single-component proton pumps activated by light7, represented by Arch which is activated by yellow or green light at 575 nm16 (Figure 2). In addition to being light-sensing proteins, opsins are also G-protein coupled receptors (GPCRs) and capable of binding G-proteins intracellularly17. Recently, GPCRs, like alpha-1 and beta-2 adrenergic receptors and 5-HT1a receptor, have been successfully engineered into light-activated regulators named OptoXRs17,18. These examples highlight the diversity and expand the repertoire of genetically encoded proteins allowing electrical activity within targeted neuronal populations to be precisely regulated by light.
Figure 2.
The schematic structures (A) and spectrum activation (B) of ChR2, NpHR, and Arch.
Since its introduction in hippocampal neurons in 2005 by Boyden et al8, optogenetics has been widely used to study GABAergic, cholinergic, serotonergic/glutamatergic and dopaminergic transmitter systems in a variety of CNS regions, including the cerebral cortex, cerebellum, hippocampus, amygdala, striatum and brainstem. This has been undertaken in species ranging from rodents, worms, and non-human primates with multiple expression systems and opsins19,20,21,22,23,24,25,26,27,28,29. The advantage of optogenetics is that it may avoid the local release of neurotransmitters and the damage evoked by electrical stimuli22,23, which introduce high-frequency, sustained stimulation23. This type of neuron population-specific approach will facilitate the precise mapping of neuronal connectivity and will more clearly elucidate the mechanisms of synaptic plasticity and, consequently, learning and memory.
Optogenetics in synaptic plasticity
Optogenetics in neuronal activity
By applying optogenetics, Higley and Sabatini find that light stimulation of motor cortex neurons projecting to the striatal medium spiny neurons (MSNs) reliably evokes EPSPs and EPSCs that are reduced by the D2R agonist quinpirole. In contrast, quinpirole fails to change EPSPs evoked by electrical stimulation22. These results may imply that optogenetic approaches have great potential in the study of the bidirectional synaptic plasticity induced by electrical stimulation of dopamine in striatum30. This conjecture is further supported by the work of Goodl and Nicoll31. AMPA and NMDAR are well known to be involved in synaptic plasticity, and Goodl and Nicoll used optogenetics to detect a compensatory depression of postsynaptic AMPAR and NMDAR following 24 h of optical stimulation of CA1 pyramidal neurons with blue light at 50 ms, 1-3 Hz31. Optogenetics is also used to study presynaptic activity in neurons. Photostimulation of proopiomelanocortin (POMC) neurons expressing ChR2 significantly reduces the frequency of miniature EPSCs (mEPSCs)32, which is also used to study synaptic plasticity33, while POMC neurons are capable of storing a memory of specific hormonal states32. The presynaptic activity induced by photostimulation can be modulated by glial cells. Additionally, activation of ChR2 expressed in astrocytes can trigger enough glutamate release to activate neuronal AMPA receptors34. These results suggest that it is possible to apply optogenetics in synaptic plasticity studies by selectively modulating specific neuronal populations.
Optogenetics and in vitro plasticity
Because of the high selectiveness of specific neuronal populations, optogenetics can facilitate the investigation of synaptic plasticity in different brain areas by selective photostimulation of specific neurons/fibers. The opsin genes can be introduced into specific neuronal populations by in utero electroporation or in vivo injections of viral vectors26. Brain slices containing areas of interest can also be prepared for studies of synaptic plasticity. For example, in striatal D1 receptor dominant MSNs expressing ChR2, 100 photostimulations at 1 Hz could induce NMDAR-dependent LTD35. In cortical slices from transgenic mice expressing ChR2-YFP, 1 Hz blue light stimulation of layer V evokes LTD in layer II/III36. In slices of auditory cortex, optical stimulation of basal forebrain cholinergic input modulates the metabolic glutamate receptor-dependent LTP through activation of the M1 receptor in the thalamocortical projection and presynaptic A1 receptors37. In the anterior cingulate cortex (ACC)-LA pathway, LTP could be induced by stimulation of ChR2 in the presence or absence of GABAAR antagonist38. These results indicate a wide range of applications for optogenetics in synaptic plasticity. Furthermore, when ChR2 is expressed in septal cholinergic neurons, specific photostimulation of septal fibers could induce LTP in CA1 pyramidal neurons that are similar to those induced by electrical stimulation or short-term depression, depending on the optical stimulation timing39. This study highlights the temporal advantage of optogenetics in synaptic plasticity. Optogenetics also shows spatial advantages in the study of synaptic plasticity. The asymmetrical distribution of NR2B subunit-containing NMDARs in CA1 pyramidal neurons40 has been confirmed using optogenetics to contribute to the asymmetry of synaptic plasticity. By combing optogenetics and traditional electrical stimulation, Kohl et al found that left CA3 input contributes more to the expression of LTP in CA1 pyramidal neurons41. These results pave an avenue for optogenetics in investigations of the left-right asymmetric functions of the hippocampus. In addition, photostimulation of astrocytes expressing ChR2 produces LTD in Purkinje cell of cortical slices by activating metabotropic glutamate receptors34, further expanding the range of applications for optogenetics in synaptic plasticity.
Since in vitro study of synaptic plasticity requires slice preparation while slice quality is closely related with the animal age, infection of neurons with virus vector to express opsins will postpone the window of time for experiment. This will increase the difficulty in some studies of synaptic plasticity, particular for LTD which is not easy to be induced in matured animals42,43,44. So, transgenic animals expressing different opsins will represent a better experimental tool for studying synaptic plasticity.
Optogenetics and in vivo plasticity
Cognitive functions require more detailed investigations at the systems level. The advantages of optogenetics in the precise control of specific neuronal populations will help improve the study of the plasticity, learning and memory and the control of the brain in vivo. Liewald et al examined the synaptic function in Caenorhabditis elegans using an optogenetic approach and suggested its use in the study of synaptic plasticity in that system23. Using a slow ChR2 mutant C128X, long-term photostimulation of C elegans command interneurons could induce long-lasting behavioral plasticity in locomotion45. In a rodent study performed in vivo, optogenetics also demonstrated a promising future in plasticity and memory research. Stimulation of dopaminergic neurons in the ventral tegmental area (VTA) that express ChR2 facilitates the development of positive reinforcement during reward-seeking and of behavioral flexibility in freely moving mice46. Photostimulation of striatal D1 receptor-dominant MSNs expressing ChR2 in freely moving mice restores the high-frequency stimulation-induced LTP that is abolished by cocaine treatment35. Optogenetic manipulation of dopamine neurons expressing eNpHR and tyrosine hydroxylase in VTA can modulate depression-like behaviors in mice47. In addition, photostimulation of mouse astrocytes expressing ChR2 can perturb motor behavioral plasticity modulated by the cerebellum34. These studies imply a wide range of applications for optogenetics for the study of plasticity in many brain areas.
Furthermore, optogenetics has also shown exciting results in studies of several memory-related brain areas. It is found that the memory consolidation is impaired in mice upon optical stimulation of hypocretin/orexin (Hcrt) neurons expressing ChR2 at 60-s intervals but not 120-s intervals, mainly through changing the degree of sleep fragmentation48. Consistently, memory retention and working memory formation are blocked by optical stimulation of basolateral amygdala neurons expressing Arch in rats49 and by silencing medial prefrontal cortex neurons expressing Arch in rats50, respectively. However, a study in rats indicates that memory retention is enhanced by optical stimulation of basolateral amygdala neurons expressing ChR249. By expressing eNpHR3.1 in dorsal hippocampus to inhibit the CA1 excitatory neurons, it is found that contextual fear acquisition and retrieval are blocked, the remote fear memory recall is reversibly impaired, and the remote contextual fear recall is blocked only by precise but not prolonged inhibition51. The contextual fear recall may also be modulated by the memory-engram cells in the dentate gyrus. Light stimulation of ChR2 expressed in a small subpopulation of granule cells in the dentate gyrus can produce context-specific memory recall52. However, a false memory can be created by the manipulation of ChR2-expressing neurons in the dentate gyrus but not in the ChR2-expressing neurons of CA153. This finding suggests that memory formation is spatiotemporally precise and that further research is needed. Taken together, these studies suggest that optogenetics has a promising future in studies of plasticity and memory in vivo.
Limitations of optogenetics
One of the limitations of ChR2 optogenetics is that photostimulation will activate voltage-gated calcium channels (VGCCs)54 if the neuron is held at membrane potentials away from −65 mV. The kinetics of ChR2, such as activation and deactivation, can be doubled at 37 °C when compared with 22 °C, and they can be affected by depolarization55. Another possible problem with ChR2 is its low conductance, which will limit its application for direct, efficient and fast stimulation of a single cell, and its alternate function as an outwardly driven H+ pump when expressed in HEK293 cells56. In addition, the transmission and intensity of light is reduced as it passes through multiple levels of tissue, although it can still evoke neuron spiking from up to a 1.4-mm distance57. The behavioral interference from intrinsic phototactic reactions induced by ionic exchanges through opsin channels may also complicate the interpretation of results. Phototoxicity due to the long-term illumination required in some studies may also be a concern. Fortunately, some of these problems can be overcome. For example, temporally focused laser pulses (TEFO) can simultaneously excite large numbers of channels on individual neurons, leading to strong (up to 15 mV), rapid depolarizations (≤1 ms)58. In the case of wild ChR2, the ChR2 mutant C128X can avoid the phototoxicity and intrinsic phototactic reaction induced by long-term photodepolarization45.
Conclusion
The improvement of techniques and the availability of more convenient and applicable opsins will allow multiplexing of light-stimulated channels and thus more precise control of specific neuronal populations. Such experiments will allow clearer elucidation of the mechanisms of neuronal plasticity. These advances will be very helpful in the understanding of brain functions and for the study of learning- and memory-related diseases like Alzheimer's disease.
Acknowledgments
This study was supported by grant (R4321A07) to John F MacDonald by Canadian Institutes of Health Research (CIHR).
References
- Xie YF, Belrose JC, Lei G, Tymianski M, Mori Y, MacDonald JF, et al. Dependence of NMDA/GSK-3beta mediated metaplasticity on TRPM2 channels at hippocampal CA3-CA1 synapses. Mol Brain. 2011;4:44. doi: 10.1186/1756-6606-4-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Wong TP, Pozza MF, Lingenhoehl K, Wang Y, Sheng M, et al. Role of NMDA receptor subtypes in governing the direction of hippocampal synaptic plasticity. Science. 2004;304:1021–4. doi: 10.1126/science.1096615. [DOI] [PubMed] [Google Scholar]
- Morishita W, Lu W, Smith GB, Nicoll RA, Bear MF, Malenka RC. Activation of NR2B-containing NMDA receptors is not required for NMDA receptor-dependent long-term depression. Neuropharmacology. 2007;52:71–6. doi: 10.1016/j.neuropharm.2006.07.005. [DOI] [PubMed] [Google Scholar]
- Yang K, Trepanier C, Sidhu B, Xie YF, Li H, Lei G, et al. Metaplasticity gated through differential regulation of GluN2A versus GluN2B receptors by Src family kinases. EMBO J. 2012;31:805–16. doi: 10.1038/emboj.2011.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson JM, Federici T, Boulis N. Optogenetic neuromodulation. Neurosurgery. 2009;64:796–804. doi: 10.1227/01.NEU.0000339171.87593.6A. [DOI] [PubMed] [Google Scholar]
- Witten IB, Steinberg EE, Lee SY, Davidson TJ, Zalocusky KA, Brodsky M, et al. Recombinase-driver rat lines: tools, techniques, and optogenetic application to dopamine-mediated reinforcement. Neuron. 2011;72:721–33. doi: 10.1016/j.neuron.2011.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yizhar O, Fenno LE, Davidson TJ, Mogri M, Deisseroth K. Optogenetics in neural systems. Neuron. 2011;71:9–34. doi: 10.1016/j.neuron.2011.06.004. [DOI] [PubMed] [Google Scholar]
- Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci. 2005;8:1263–8. doi: 10.1038/nn1525. [DOI] [PubMed] [Google Scholar]
- Nagel G, Szellas T, Huhn W, Kateriya S, Adeishvili N, Berthold P, et al. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci U S A. 2003;100:13940–5. doi: 10.1073/pnas.1936192100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinlogel S, Feldbauer K, Dempski RE, Fotis H, Wood PG, Bamann C, et al. Ultra light-sensitive and fast neuronal activation with the Ca2+-permeable channelrhodopsin CatCh. Nat Neurosci. 2011;14:513–8. doi: 10.1038/nn.2776. [DOI] [PubMed] [Google Scholar]
- Lin JY, Lin MZ, Steinbach P, Tsien RY. Characterization of engineered channelrhodopsin variants with improved properties and kinetics. Biophys J. 2009;96:1803–14. doi: 10.1016/j.bpj.2008.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunaydin LA, Yizhar O, Berndt A, Sohal VS, Deisseroth K, Hegemann P. Ultrafast optogenetic control. Nat Neurosci. 2010;13:387–92. doi: 10.1038/nn.2495. [DOI] [PubMed] [Google Scholar]
- Zhang F, Prigge M, Beyriere F, Tsunoda SP, Mattis J, Yizhar O, et al. Red-shifted optogenetic excitation: a tool for fast neural control derived from Volvox carteri. Nat Neurosci. 2008;11:631–3. doi: 10.1038/nn.2120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schobert B, Lanyi JK. Halorhodopsin is a light-driven chloride pump. J Biol Chem. 1982;257:10306–13. [PubMed] [Google Scholar]
- Zhang F, Wang LP, Brauner M, Liewald JF, Kay K, Watzke N, et al. Multimodal fast optical interrogation of neural circuitry. Nature. 2007;446:633–9. doi: 10.1038/nature05744. [DOI] [PubMed] [Google Scholar]
- Chow BY, Han X, Dobry AS, Qian X, Chuong AS, Li M, et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature. 2010;463:98–102. doi: 10.1038/nature08652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Airan RD, Thompson KR, Fenno LE, Bernstein H, Deisseroth K. Temporally precise in vivo control of intracellular signalling. Nature. 2009;458:1025–9. doi: 10.1038/nature07926. [DOI] [PubMed] [Google Scholar]
- Oh E, Maejima T, Liu C, Deneris E, Herlitze S. Substitution of 5-HT1A receptor signaling by a light-activated G protein-coupled receptor. J Biol Chem. 2010;285:30825–36. doi: 10.1074/jbc.M110.147298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berndt A, Schoenenberger P, Mattis J, Tye KM, Deisseroth K, Hegemann P, et al. High-efficiency channelrhodopsins for fast neuronal stimulation at low light levels. Proc Natl Acad Sci U S A. 2011;108:7595–600. doi: 10.1073/pnas.1017210108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sohal VS, Zhang F, Yizhar O, Deisseroth K. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature. 2009;459:698–702. doi: 10.1038/nature07991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varga V, Losonczy A, Zemelman BV, Borhegyi Z, Nyiri G, Domonkos A, et al. Fast synaptic subcortical control of hippocampal circuits. Science. 2009;326:449–53. doi: 10.1126/science.1178307. [DOI] [PubMed] [Google Scholar]
- Higley MJ, Sabatini BL. Competitive regulation of synaptic Ca2+ influx by D2 dopamine and A2A adenosine receptors. Nat Neurosci. 2010;13:958–66. doi: 10.1038/nn.2592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liewald JF, Brauner M, Stephens GJ, Bouhours M, Schultheis C, Zhen M, et al. Optogenetic analysis of synaptic function. Nat Methods. 2008;5:895–902. doi: 10.1038/nmeth.1252. [DOI] [PubMed] [Google Scholar]
- Johansen JP, Hamanaka H, Monfils MH, Behnia R, Deisseroth K, Blair HT, et al. Optical activation of lateral amygdala pyramidal cells instructs associative fear learning. Proc Natl Acad Sci U S A. 2010;107:12692–7. doi: 10.1073/pnas.1002418107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witten IB, Lin SC, Brodsky M, Prakash R, Diester I, Anikeeva P, et al. Cholinergic interneurons control local circuit activity and cocaine conditioning. Science. 2010;330:1677–81. doi: 10.1126/science.1193771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao S, Ting JT, Atallah HE, Qiu L, Tan J, Gloss B, et al. Cell type-specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function. Nat Methods. 2011;8:745–52. doi: 10.1038/nmeth.1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutierrez DV, Mark MD, Masseck O, Maejima T, Kuckelsberg D, Hyde RA, et al. Optogenetic control of motor coordination by Gi/o protein-coupled vertebrate rhodopsin in cerebellar Purkinje cells. J Biol Chem. 2011;286:25848–58. doi: 10.1074/jbc.M111.253674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutierrez R, Lobo MK, Zhang F, de Lecea L. Neural integration of reward, arousal, and feeding: Recruitment of VTA, lateral hypothalamus, and ventral striatal neurons. IUBMB Life. 2011;63:824–30. doi: 10.1002/iub.539. [DOI] [PubMed] [Google Scholar]
- Diester I, Kaufman MT, Mogri M, Pashaie R, Goo W, Yizhar O, et al. An optogenetic toolbox designed for primates. Nat Neurosci. 2011;14:387–97. doi: 10.1038/nn.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen W, Flajolet M, Greengard P, Surmeier DJ. Dichotomous dopaminergic control of striatal synaptic plasticity. Science. 2008;321:848–51. doi: 10.1126/science.1160575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goold CP, Nicoll RA. Single-cell optogenetic excitation drives homeostatic synaptic depression. Neuron. 2010;68:512–28. doi: 10.1016/j.neuron.2010.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Atasoy D, Su HH, Sternson SM. Hunger states switch a flip-flop memory circuit via a synaptic AMPK-dependent positive feedback loop. Cell. 2011;146:992–1003. doi: 10.1016/j.cell.2011.07.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicoll RA, Malenka RC. Expression mechanisms underlying NMDA receptor-dependent long-term potentiation. Ann N Y Acad Sci. 1999;868:515–25. doi: 10.1111/j.1749-6632.1999.tb11320.x. [DOI] [PubMed] [Google Scholar]
- Sasaki T, Beppu K, Tanaka KF, Fukazawa Y, Shigemoto R, Matsui K. Application of an optogenetic byway for perturbing neuronal activity via glial photostimulation. Proc Natl Acad Sci U S A. 2012;109:20720–5. doi: 10.1073/pnas.1213458109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pascoli V, Turiault M, Luscher C. Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature. 2011;481:71–5. doi: 10.1038/nature10709. [DOI] [PubMed] [Google Scholar]
- Xiong W, Jin X. Optogenetic field potential recording in cortical slices. J Neurosci Methods. 2012;210:119–24. doi: 10.1016/j.jneumeth.2012.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chun S, Bayazitov IT, Blundon JA, Zakharenko SS. Thalamocortical long-term potentiation becomes gated after the early critical period in the auditory cortex. J Neurosci. 2013;33:7345–57. doi: 10.1523/JNEUROSCI.4500-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morozov A, Sukato D, Ito W. Selective suppression of plasticity in amygdala inputs from temporal association cortex by the external capsule. J Neurosci. 2011;31:339–45. doi: 10.1523/JNEUROSCI.5537-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu Z, Yakel JL. Timing-dependent septal cholinergic induction of dynamic hippocampal synaptic plasticity. Neuron. 2011;71:155–65. doi: 10.1016/j.neuron.2011.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinohara Y, Hirase H, Watanabe M, Itakura M, Takahashi M, Shigemoto R. Left-right asymmetry of the hippocampal synapses with differential subunit allocation of glutamate receptors. Proc Natl Acad Sci U S A. 2008;105:19498–503. doi: 10.1073/pnas.0807461105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohl MM, Shipton OA, Deacon RM, Rawlins JN, Deisseroth K, Paulsen O. Hemisphere-specific optogenetic stimulation reveals left-right asymmetry of hippocampal plasticity. Nat Neurosci. 2011;14:1413–5. doi: 10.1038/nn.2915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner AJ, Cummings DM, Spencer JP, Murphy KP. Bi-directional plasticity and age-dependent long-term depression at mouse CA3-CA1 hippocampal synapses. Neurosci Lett. 2004;367:1–5. doi: 10.1016/j.neulet.2004.04.056. [DOI] [PubMed] [Google Scholar]
- Kemp N, McQueen J, Faulkes S, Bashir ZI. Different forms of LTD in the CA1 region of the hippocampus: role of age and stimulus protocol. Eur J Neurosci. 2000;12:360–6. doi: 10.1046/j.1460-9568.2000.00903.x. [DOI] [PubMed] [Google Scholar]
- Dudek SM, Bear MF. Bidirectional long-term modification of synaptic effectiveness in the adult and immature hippocampus. J Neurosci. 1993;13:2910–8. doi: 10.1523/JNEUROSCI.13-07-02910.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultheis C, Liewald JF, Bamberg E, Nagel G, Gottschalk A. Optogenetic long-term manipulation of behavior and animal development. PLoS One 2011. 6:e18766. doi: 10.1371/journal.pone.0018766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adamantidis AR, Tsai HC, Boutrel B, Zhang F, Stuber GD, Budygin EA, et al. Optogenetic interrogation of dopaminergic modulation of the multiple phases of reward-seeking behavior. J Neurosci. 2011;31:10829–35. doi: 10.1523/JNEUROSCI.2246-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tye KM, Mirzabekov JJ, Warden MR, Ferenczi EA, Tsai HC, Finkelstein J, et al. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature. 2013;493:537–41. doi: 10.1038/nature11740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolls A, Colas D, Adamantidis A, Carter M, Lanre-Amos T, Heller HC, et al. Optogenetic disruption of sleep continuity impairs memory consolidation. Proc Natl Acad Sci U S A. 2011;108:13305–10. doi: 10.1073/pnas.1015633108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huff ML, Miller RL, Deisseroth K, Moorman DE, LaLumiere RT. Posttraining optogenetic manipulations of basolateral amygdala activity modulate consolidation of inhibitory avoidance memory in rats. Proc Natl Acad Sci U S A. 2013;110:3597–602. doi: 10.1073/pnas.1219593110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilmartin MR, Miyawaki H, Helmstetter FJ, Diba K. Prefrontal activity links nonoverlapping events in memory. J Neurosci. 2013;33:10910–4. doi: 10.1523/JNEUROSCI.0144-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goshen I, Brodsky M, Prakash R, Wallace J, Gradinaru V, Ramakrishnan C, et al. Dynamics of retrieval strategies for remote memories. Cell. 2011;147:678–89. doi: 10.1016/j.cell.2011.09.033. [DOI] [PubMed] [Google Scholar]
- Liu X, Ramirez S, Pang PT, Puryear CB, Govindarajan A, Deisseroth K, et al. Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature. 2012;484:381–5. doi: 10.1038/nature11028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez S, Liu X, Lin PA, Suh J, Pignatelli M, Redondo RL, et al. Creating a false memory in the hippocampus. Science. 2013;341:387–91. doi: 10.1126/science.1239073. [DOI] [PubMed] [Google Scholar]
- Zhang YP, Oertner TG. Optical induction of synaptic plasticity using a light-sensitive channel. Nat Methods. 2007;4:139–41. doi: 10.1038/nmeth988. [DOI] [PubMed] [Google Scholar]
- Chater TE, Henley JM, Brown JT, Randall AD. Voltage- and temperature-dependent gating of heterologously expressed channelrhodopsin-2. J Neurosci Methods. 2010;193:7–13. doi: 10.1016/j.jneumeth.2010.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldbauer K, Zimmermann D, Pintschovius V, Spitz J, Bamann C, Bamberg E. Channelrhodopsin-2 is a leaky proton pump. Proc Natl Acad Sci U S A. 2009;106:12317–22. doi: 10.1073/pnas.0905852106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aravanis AM, Wang LP, Zhang F, Meltzer LA, Mogri MZ, Schneider MB, et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J Neural Eng. 2007;4:S143–56. doi: 10.1088/1741-2560/4/3/S02. [DOI] [PubMed] [Google Scholar]
- Andrasfalvy BK, Zemelman BV, Tang J, Vaziri A. Two-photon single-cell optogenetic control of neuronal activity by sculpted light. Proc Natl Acad Sci U S A. 2010;107:11981–6. doi: 10.1073/pnas.1006620107. [DOI] [PMC free article] [PubMed] [Google Scholar]