Abstract
Excess body weight due to obesity has traditionally been considered to have a positive effect on bone; however, more recent findings suggest that bone quality is compromised. Both obesity and caloric restriction increase fracture risk and are regulated by endocrine factors and cytokines that have direct and indirect effects on bone and calcium absorption. Weight reduction will decrease bone mass and mineral density, but this varies by the individual’s age, gender, and adiposity. Dietary modifications, exercise, and medications have been shown to attenuate the bone loss associated with weight reduction. Future obesity and weight loss trials would benefit from assessment of key hormones, adipokine and gut peptides that regulate calcium absorption, and bone mineral density and quality by using sensitive techniques in high-risk populations.
Keywords: body weight, bone, caloric restriction, hormones, calcium, nutrients
INTRODUCTION
Obesity and osteoporosis are growing concerns worldwide, and both are attributed to a poor diet, excess caloric intake, and/or lower physical activity. An estimated 1.5 billion people were obese in the year 2008 worldwide (191), and in the United States, 34% of the population is obese, and 68% is either overweight or obese. This has led to an increased risk for several comorbidities, including cardiovascular disease (CVD), type 2 diabetes, and certain cancers (48). Osteoporosis is often referred to as a silent disease and therefore contrasts the high visibility of obesity. In the United States, osteoporosis affects 55% of the population over 50 years of age (116). One out of three women will suffer a fracture compared to one out of five men, but mortality afterward is greater in men. Neither obesity nor osteoporosis is considered to be a part of normal aging, and both conditions have common hormonal alterations and are associated with increased proinflammatory cytokines and oxidative stress. These factors contribute to increased fat acquisition and loss of bone mass.
Body weight is directly associated with bone mineral density (BMD). A low body mass index (BMI) has been identified as an important risk factor for lower BMD and predicts greater bone loss in older age (118, 138) and in younger persons in the absence of menses and/or an eating disorder (113). On the other hand, a high body weight can be due to increased physical activity or obesity, and both will increase BMD, but there is increasing evidence that excess weight due to adiposity is detrimental to bone and fracture risk. In addition, although weight reduction is recommended to reduce comorbidities related to obesity, it also induces bone loss and increases risk of fracture in older individuals. Dietary factors, exercise, and some medications will attenuate bone loss due to weight reduction. This review addresses how obesity due to greater adiposity and weight reduction separately and together influence bone quality, bone turnover, and bone loss and examines how interventions influence bone metabolism during weight reduction.
OBESITY AND BONE
Cellular Relationship of Adiposity and Bone
The pluripotent stromal cell differentiates into mature cell types—adipocytes, osteoblasts, and chondrocytes. Because the stromal cell can differentiate into an osteoblast or adipocyte, this can eventually determine the balance between bone and adipose tissue. This is further evident in multiple clinical conditions that show a relationship between bone marrow fat and BMD. Both osteoporosis and aging-related bone loss are associated with an increase in marrow adipogenesis (108), which may suggest a conversion of stromal cells to adipocytes rather than osteoblasts. It remains unclear whether adipocytes and osteoblasts respond in a unique and possibly opposite manner to the same factor that is regulating differentiation to ultimately affect the bone and fat relationship.
The peroxisome proliferator–activated receptor-δ (PPARδ) plays a central role in initiating adipogenesis and inhibiting osteoblastogenesis (145). Several drugs act as ligands for PPARδ. These include the thiazolidinedione class of antidiabetic drugs (i.e., rosiglitazone) that increase insulin sensitivity (175) and have also been shown to increase adiposity, reduce bone mass, and increase fracture risk (68). Another example of drugs that increase adiposity, especially visceral fat, at the expense of osteoblast differentiation are glucocorticoids. In addition, low-density lipoprotein oxidation products promote osteoporotic bone loss by directing progenitor marrow stromal cells to undergo adipogenic instead of osteogenic differentiation (127). Furthermore, skeletal unloading due to immobilization or inactivity increases adipocyte differentiation and inhibits osteoblast differentiation (1). There is also evidence that the higher adiposity in obesity is associated with a lower rate of bone formation (126). Overall, a further understanding of the cellular connection between adipocytes and osteoblasts could result in drugs to inhibit marrow adipogenesis and attenuate age-related bone loss.
Factors Influencing Bone in Obesity
Hormonal and mechanical loading
The higher BMD and bone mineral content (BMC) in obesity are attributed to multiple factors (153). These include a greater mechanical loading on bone (53, 198) and the altered hormonal milieu and higher serum levels of adipokines associated with obesity (41, 153, 197). Other genetic and environmental factors such as smoking, dietary intake, and lifestyle also play an independent role in influencing bone mass in obesity.
In comparison with leaner individuals, in obese persons higher levels of serum estrogen and parathyroid hormone (PTH), lower 25 hydroxyvitamin D (25OHD) and sex hormone–binding globulin (SHBG), and possibly lower 1,25 dihydroxyvitamin D3 (1,25(OH)2D3) are found (95, 153, 197), and all have specific actions on bone. Higher levels of serum estrogens in obese postmenopausal women and men in comparison with leaner individuals are largely derived from the metabolism of circulating androstenedione by adipose tissue. The lower serum levels of 25OHD in obesity are likely due to storage in adipose tissue (193). In contrast, it is not clear why excess body fat is associated with higher PTH (16, 131), but PTH affects calcium metabolism and proinflammatory cytokines (65, 164), which would have a detrimental effect on bone.
Obesity is also associated with higher levels of pancreatic hormones such as insulin, amylin, and preptin (86) that are anabolic to bone (21, 30, 35). Levels of several adipose-derived peptides and enzymes, such as aromatase, hydroxyl steroid dehydrogenase, leptin, and resistin are higher in obesity and have specific anabolic or catabolic actions on the osteoblast (87, 129, 130). Adiponectin is reduced in obesity (5), and in vitro observations show it increases osteoblastic activity (104), yet clinical studies do not necessarily support a positive effect on bone (6, 7, 85, 100) or fracture risk. Obesity is also associated with higher circulating concentrations of inflammatory cytokines, such as interleukin-6 (IL-6), monocyte chemoattractant protein-1, and C-reactive protein (CRP) (67). In leaner populations, higher inflammatory cytokines have been associated with higher bone turnover (11, 83, 93, 114), and their role in regulating bone in obesity is likely also important (159) but is not clearly understood. It is possible that the low level of chronic inflammation in obesity is counterbalanced by adipose-derived estrogen and/or other factors such as weight bearing activities to prevent bone loss that occurs in leaner populations.
Mechanical loading
The skeletal tissue is highly responsive to its mechanical environment, and strain will maintain mineral homeostasis (52). In addition, mechanical loading also stimulates bone formation by decreasing apoptosis and increasing the proliferation and differentiation of osteoblasts and osteocytes (43). Mechanical loading, in fact, favors osteoblastogenesis at the expense of adipogenesis by downregulating PPARδ in bone marrow stromal cells (37). It is well established that dynamic loads due to muscle contraction are more anabolic to bone than are static loads (177) induced by excess adipose tissue and relates to the differential mechanostatic sensitivity of these tissues (52, 156). Thus, obesity may not confer a mechanical advantage to the bone if not accompanied by greater lean mass and a physically active (nonsedentary) lifestyle.
Body Composition and Bone
Lean and fat masses are both independent determinants of bone mass. Lean mass is a reflection of physical activity, whereas adipose tissue acts as an endocrine organ with an important influence on the bone, as described above (41, 153, 197). In premenopausal women, low muscle mass is associated with low BMD (162), and the positive effect of a higher body weight on bone occurs only when it is primarily composed of lean mass (149). The excess weight in obesity is primarily attributed to excess adipose tissue, yet in general, there is also higher fat-free soft tissue to support the weight. The positive effect of a higher lean mass on BMD may be attributed to lifestyle factors including exercise and diet, estrogen sufficiency, genetic influences, or a combination of these factors (152). Also, greater muscle mass has an independent effect on better balance and therefore reduces frailty and falls associated with osteoporotic fracture. Fat mass shows a direct correlation with bone in some studies of postmenopausal women (26, 42, 102), but not necessarily in children and young adults (63, 80), and may differ by skeletal site and amount of trabecular/cortical bone (89). At least some of these studies have corrected for muscle mass, so the independent effect of fat on bone could be determined, which can explain some of the different conclusions in these studies. In addition, the location of fat accumulation, including visceral adipose tissue (VAT) compared to subcutaneous tissue, may differentially influence bone. For example, studies in postmenopausal women suggest a positive relationship between VAT and bone, but not in children or men, and again findings may depend on the specific bone site that is measured (56, 96, 105, 187). However, many of these studies measure trunk fat using dual-energy x-ray absorptiometry (DXA), which includes both subcutaneous and visceral depots, unlike quantitative computed tomography (QCT), which can distinguish between fat depots. Higher VAT is associated with greater dyslipidemia, insulin resistance, and inflammatory cytokines, which would be expected to have a detrimental effect on bone. Nevertheless, a decrease in VAT due to weight loss has been shown to decrease BMD (18), suggesting that other factors unrelated to VAT are regulating bone during caloric restriction (CR). Besides white adipose tissue, increased bone marrow fat tissue has been shown to be associated with lower BMD (157), whereas brown adipose tissue may be important in maintaining bone (20). Understanding how lean tissue and the type and amount of adipose tissue influence BMD in different populations is important to better estimate fracture risk under conditions where body composition is altered.
Bone Mass, Quality, and Fracture
There is greater calcium retention during growth in obese children (74) and higher bone mass in obese adults (197). Not surprisingly, the higher BMD in obesity is not proportional to the increase in body weight because adiposity is the major component of excess weight gain. Therefore, BMD per unit BMI is lower in obesity (38). Indeed, studies suggest that excess adipose tissue may have a negative impact on bone quality in adults and children (80, 132, 165, 198). Also, studies show lower bone formation relative to resorption markers in obese compared with leaner populations in adults (27, 126) and children (40). Excess adiposity and a high-fat diet may be particularly detrimental to BMD and bone quality during growth, as demonstrated in rodent studies (24, 128, 190).
Bone quality is a composite of BMD (cortical and trabecular), bone geometry, and bone strength and is considered a major determinant of fracture risk. Several techniques are used to measure BMD (19) and other aspects of bone quality. DXA is the gold standard method of measuring areal BMD (aBMD), BMC, and soft tissue composition. Low aBMD predicts fractures, but the sensitivity and specificity of the prediction are low since more than half of all fractures occur in persons without osteoporosis, as defined by aBMD. Newer noninvasive imaging techniques, including volumetric assessments of bone density (vBMD) and geometry, bone compartments (trabecular and cortical), and microarchitecture, and measurements of bone strength such as finite element analysis, provide estimates of bone fragility (19). These in vivo high-resolution bone-imaging techniques, such as QCT and high-resolution magnetic resonance imaging assessed at axial (hip and spine) or peripheral (radius and tibia) sites, combined with DXA can improve estimates of bone strength and fracture risk in the future.
There is evidence that bone quality is altered in obesity. The obese have lower cortical BMD, as measured by peripheral QCT in comparison with age-matched leaner individuals (132, 165, 169, 188). Serum PTH, which is elevated in obesity (16, 165), has known catabolic actions on cortical bone, with less evidence for an effect on trabecular bone (25). Other obesity-induced factors may also affect bone quality and may contribute to the higher-than-expected fracture risk for a given BMD in this population (133). The extent to which an altered bone quality and/or balance contribute to fractures in the obese is not known and should be addressed in prospective studies.
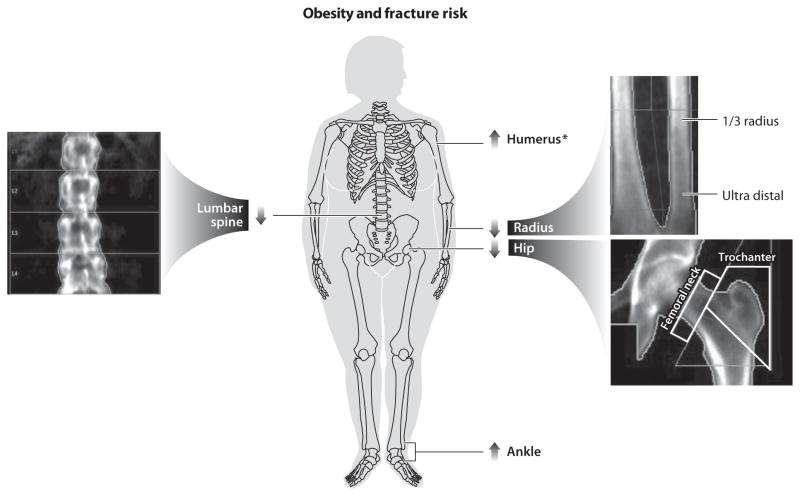
There is consistent evidence for a higher fracture risk in the obese pediatric population that may be explained by an altered bone quality or mineralization (40). It is not clear why obese children have a higher fracture risk, but it could be related to abnormal bone mineralization (39, 40), poor balance (62), or force upon falling creating a greater risk for injury and fracture (62, 137). In older women, there is a higher site-specific fracture risk with higher BMI at the humerus, and in both men and women at the ankle (58, 78, 32, 134), and the higher fracture risk occurs in the presence of normal BMD (119, 133) (Figure 1). In contrast, there is a lower risk of hip, vertebral, and forearm fractures with higher BMI (58, 78). In addition, BMI adjusted for BMD is associated with a higher risk of hip fracture when BMI is greater than 35 kg/m2 (38). The high BMI–fracture link in obesity may be influenced by altered bone quality, different patterns or forces upon falling, or the greater padding by adipose tissue at specific bone sites.
Figure 1.
Arrows indicate direction of fracture risk at bone sites measured by dual-energy x-ray absorptiometry showing axial (lumbar spine and hip) and peripheral sites (humerus, radius, and tibia/ankle) due to increased body mass index (obesity). *Higher humerus fracture due to obesity is found in women but not in men (32, 58, 78, 119, 134).
WEIGHT REDUCTION AND BONE
A weight loss of ~10% will have a positive effect on the comorbidities associated with obesity. However, this also results in a 1% to 2% bone loss at the hip and total body and a 3% to 4% loss at highly trabecular sites such as the trochanter and radius (143, 163, 182). The weight-loss-induced BMD reduction has been found in peri- and postmenopausal women and older men (14, 45, 98, 118, 148, 153, 184) and is more than double the annual rate of bone loss in weight-stable individuals. In addition, it is unclear whether the rate and amount of weight loss influence the loss of bone, since in the Comprehensive Assessment of Long-Term Effects of Reducing Intake of Energy (CALERIE) trial, middle-aged individuals on a very-low-energy diet who rapidly lost 15% of their body weight did not show a greater bone loss than did individuals in a slower, moderate weight loss intervention (139). Furthermore, the long-term effect of weight loss continues, based on one-year follow-up studies (75, 182). Short-term (less than six months) weight loss studies are not reported in this review because bone remodeling requires a minimum of four to six months and may continue for one to two years (72).
Several epidemiological studies have addressed the influence of voluntary or involuntary weight loss on bone over a long period of time. In one study, approximately 1,500 men were followed up for 30 years to assess the influence on bone with a 5% to 10% weight loss or regain (109). The prevalence of osteoporosis in the lowest quartile of baseline BMI was 31% in persons losing weight compared with 4% in those who gained weight (109). In another study, men who lost greater than 5% of body weight had a 1.7% total hip BMD loss/year compared with 0.1% loss in men who were weight stable (45). A recent study in elderly women identified risk factors for hip BMD loss over four years and concluded that women who gain weight show attenuated BMD loss at the trochanter, femoral neck, and total hip (66). Importantly, high rates of BMD loss, weight loss, and weight fluctuation are also predictors of all-cause mortality in elderly men and women (117).
Fracture Risk
Weight loss of only about 5% is associated with bone loss and increased fracture risk in both men and women (36, 97, 98, 110, 111). The risk of fracture also increases in individuals of low body weight and in those who have a history of weight loss (44, 76, 97, 115). In community dwelling older women (98) and men (115), those who lost greater than 10% of body weight had the highest risk of hip fracture. In a study examining ~11,000 men and women, a 10-year reduction in BMI (−2 kg/m2) compared to an increase (1 kg/m2) was associated with an elevated nonvertebral fracture risk of about 1.7 (hazard ratio) in nonsmokers (189). Another longitudinal study with a 17-year follow-up period in over 7,500 postmenopausal women who lost 5% or more of their weight showed a 33% increased risk of distal forearm fractures compared with those who lost less than 5% of their weight (123). Both voluntary and involuntary weight reduction result in loss of BMD and increase fracture risk in older men and women at several bone sites.
Gender, Age, and Initial Body Weight
Bone loss and increased fracture risk due to weight reduction occur in both older women and men, but neither has been demonstrated in younger individuals (139, 144, 156, 179). The absence of bone loss may be due to the well-maintained muscle in men and young women (180). In a controlled six-month weight loss trial, 38 obese (average age 44 years; BMI of 35 kg/m2) premenopausal women lost 8% of their body weight without significant BMD loss (156). Because a lower initial body weight could result in bone loss, another study was conducted in 44 overweight premenopausal women (average age 38 years; BMI of 28 kg/m2) (144). In this study, six months of weight reduction resulted in 7% loss of body weight and no loss of BMD at any site (144). Others also found that bone is preserved in younger men and women with adequate calcium intake during moderate weight loss (139), yet bone is not spared under conditions of extreme weight loss (see Bariatric Surgery and Extreme Weight Loss section below). In rodents, biomechanical properties of bone are reduced more in older compared with younger energy-restricted rats (171). This is consistent with evidence of a higher fracture risk in the older populations that lose weight (36, 97, 98, 110, 111).
Studies show that initial body weight is not only a predictor of bone loss due to aging (76, 118), but that weight reduction also causes greater loss of bone in leaner populations. In a large study of older women and weight loss (≤5%), thinner women (<60 kg) lost more femoral neck BMD (3%/year) compared with heavier women (>70 kg) who lost <1%/year (118). There is also some evidence that initial body weight results in a greater loss of trabecular than cortical bone in rodent studies (71). Multiple mechanisms may influence a differential response of bone to energy restriction in obese compared with leaner individuals, such as a greater decline in serum estrogen and other sex steroids.
Both obesity (132, 165) and weight reduction (69) are associated with reduced cortical bone density, and the combination may pose a unique risk to cortical bone. Randomized clinical trials have assessed how dietary or exercise interventions during CR or surgical interventions influence bone, and findings are discussed below.
Factors Leading to Bone Loss During Weight Loss
Hormones, cytokines, and peptides influencing bone during CR
Several bone-regulating hormones are altered during CR and with weight reduction. A reduction in adiposity decreases circulating estrogen and other sex hormones (121) and increases sex hormone–binding globulin. These changes negatively influence bone osteoblastic (46) and osteoclastic activity directly or indirectly (73) due to increased levels of cytokines (i.e., IL-1, IL-6, and tumor necrosis factor-α). Weight loss studies show that a decline in estradiol correlates with BMD loss (141, 151). Even small decreases in estradiol and other sex steroids lead to a rise in bone resorption and loss in both older women and in men (47, 90), so small changes due to loss of adiposity are important in older individuals.
There is a rise in the Ca-PTH axis during CR in women consuming low Ca, which would increase bone resorption (141, 153). Serum 25OHD rises with weight loss (106, 143) due to its release from adipose tissue (22), but this should not have a detrimental effect on bone. Short-term CR also increases serum levels of cortisol (143, 174), and this may be linked to bone loss (9). Also, during CR there is a decrease in serum insulin-like growth factor-1 (IGF-1) (125). Serum IGF-1 increases osteoblast differentiation and BMD (120), and hence a decrease in IGF-1 associated with CR would negatively influence bone.
The adipocyte-derived hormones may also play a role in bone metabolism during weight reduction. Leptin is an anorexic hormone with both central and peripheral effects to inhibit bone formation, possibly through sympathetic signaling (170), and direct effects on osteoblasts (172) and osteoclasts (77). The role of reduced leptin on bone in clinical weight loss trials is likely complicated due to its varied peripheral and central effects and leptin resistance in the obese population. For example, leptin administration does not prevent a rise in bone resorption and decrease in formation due to weight reduction in obese patients (34), yet it increases BMD in amenorrheic women in energy-deficient states (158). Adiponectin, the anti-inflammatory adipokine, is low in obesity and increases with 10% or more weight loss (92), but its role in the regulation of bone mass is not clear (6). Because adiponectin suppresses osteoclast number and activates osteoblastogenesis (124), it is possible that a rise due to weight reduction would have a beneficial effect on bone mass. Other adipokines such as resistin and visfatin that are altered due to weight reduction do not consistently show an association with BMD (12).
Several gut peptides [i.e., ghrelin, incretins, cholecystokinin (CCK), peptide YY (PYY), and pancreatic polypeptide (PPY)] that regulate satiety are altered during CR and also regulate bone. Ghrelin, an appetite-regulating hormone, increases in response to weight reduction (91, 166) and modulates the proliferation and differentiation of osteoblasts (54). However, a recent systematic review examining 59 studies concluded that there is a lack of convincing data to support an association between ghrelin and BMD or fractures (12). The incretin hormones, gastric inhibitory polypeptide and glucagon-like peptide-1 and -2, which are released upon meal ingestion and decrease with weight loss, appear to have an anabolic effect on bone (195). PYY and PPY are neuropeptides implicated in the regulation of energy balance and bone mass. For example, PYY-deficient mice show a reduction in trabecular bone mass and deficit in bone strength (192), and PYY levels decrease following weight reduction (166). Serum levels of amylin and CCK are both reduced due to weight reduction (166). Amylin is a peptide hormone cosecreted with insulin from pancreatic beta cells and has been shown to stimulate bone formation and decrease bone resorption (21), whereas the role of CCK on bone is not well understood. Currently, there is no clear link between the change in gut peptides during CR and its influence on bone metabolism and loss.
Calcium absorption and other factors influencing bone during CR
Energy restriction also reduces calcium absorption efficiency (28, 29), and it is another mechanism whereby weight reduction could regulate bone loss. Several of the endocrine changes due to CR may also be regulating the decrease in calcium absorption. For example, reduced estrogen levels (29, 71, 122, 141) and a rise in cortisol may have a direct detrimental effect on calcium absorption (28). Consistent with these observations, the high serum PTH that has been shown to occur with weight reduction (141, 153) may be a response to decreased calcium absorption. In addition, the reduced weight bearing and the loss of lean mass due to CR may contribute to bone loss (see Exercise-Induced Weight Loss section). Insufficient micronutrient intake during CR could contribute to bone loss yet cannot explain it entirely because micronutrient replacement in clinical trials (143, 163) and in rodent studies also resulted in bone loss (69, 171). The loss of mass resulting from weight reduction would decrease mechanical loading to bone, especially if the loss consisted of lean soft tissue in addition to adipose tissue.
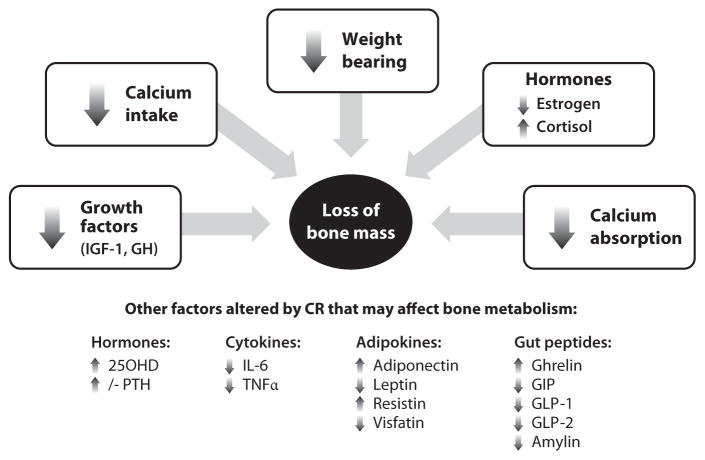
Overall, the interaction between hormones, growth factors, and cytokines and their effects on calcium absorption and bone during caloric restriction are complex (Figure 2), and this is influenced by other factors such as initial body weight/composition, the amount of weight loss, gender, age, and ethnicity. A change in the endocrine profile due to energy restriction can affect bone both directly and indirectly. Targeting the primary regulators and/or organs that respond to energy restriction and influence calcium metabolism are approaches to minimize or prevent bone loss.
Figure 2.
Mechanisms leading to bone loss due to caloric restriction (CR) and weight reduction. 25OHD, 25 hydroxyvitamin D; GIP, gastric inhibitory polypeptide; GLP, glucagon-like peptide; IL-6, interleukin 6; PTH, parathyroid hormone.
Intervention Studies: Strategies to Reduce Bone Loss During Caloric Restriction
Dietary and clinical interventions can help attenuate bone loss with weight reduction, such as increasing a specific micro- or macronutrient and/or physical activity.
Calcium
Randomized controlled trials (RCTs) have shown that calcium supplementation will attenuate the increased activity of the calcium-PTH axis and decrease bone turnover and loss at multiple sites in post-menopausal women who reduce their weight. One study in obese postmenopausal women showed that 1g Ca/day compared with placebo suppressed bone turnover and total body BMD loss (140). In another double-blind RCT study, 66 overweight postmenopausal women (age 61 years) reduced body weight by 9% over six months (143). This resulted in 4% loss of trochanter BMD when consuming 1 g Ca/d but not in women consuming a higher Ca intake of 1.7 g/d (143). Even with supplementation of calcium (1.7 g/d), postmenopausal women with moderate activity level continue to lose trochanter and spine BMD. In another six-month randomized CR trial, 52 women (14 postmenopausal) were assigned to Ca supplement or no treatment (81). A loss of 5.5% of body weight resulted in 2.2% total body BMC loss and 4% loss of hip BMC only in the untreated group of women (81). In contrast, double-blind RCT studies of premenopausal women showed no bone loss over a six-month period irrespective of Ca intake (<0.8 g–1.8 g/d) (144, 156). In one of these weight-reduction studies, 1 g of Ca supplementation tended to increase lumbar spine BMD by 1.7% compared with the placebo. However, this was likely due to low Ca intake before the study and filling of resorptive space (156). In studies of extreme weight loss and bariatric surgery, calcium and vitamin D supplementation is less effective in suppressing bone turnover and loss (see Bariatric Surgery and Extreme Weight Loss section below). The effect of Ca intake in men during CR has not been specifically examined, but it is hypothesized that older men losing weight would also benefit from supplemental Ca intake.
Protein
The positive effect of a higher protein intake on bone has been established in several epidemiological and clinical studies (33). A higher-protein weight loss diet may preserve bone mass during caloric restriction by various mechanisms such as attenuating the decrease in IGF-1 or Ca absorption or by other physiological changes accompanying a higher protein intake. Caloric restriction leads to a decrease in serum IGF-1 and IGF-1/IGF binding protein (BP)-3 ratio, whereas a higher protein intake raises these levels. Additionally, a higher protein intake increases Ca absorption (88), which thus may attenuate the decrease in Ca absorption associated with caloric restriction. There are reports showing that a higher-protein diet will decrease bone turnover and loss during CR (154), but these studies failed to control for Ca or other nutrients (161, 173). In a one-year randomized controlled trial in our laboratory, 47 postmenopausal women either received a higher-protein (24% of total calories) or a normal-protein (18% of total calories) diet with a controlled Ca intake totaling 1.2 g/day in both groups (163). After one year, women lost 7% of body weight. The higher-protein (86 g/d) compared with the normal-protein (60 g/d) diet attenuated (a) the loss of BMD at the ultradistal radius and lumbar spine, (b) total hip and trabecular volumetric BMD, and (c) BMC of the tibia. This was attributed to the higher final values of IGF-1 and IGFBP-3 and lower bone resorption in the higher-protein diet. Thus, a dietary protein intake of up to 24% of total calories, even without higher Ca intake than recommended, will attenuate bone loss due to weight reduction.
Vitamin D
The effect of vitamin D on BMD in combination with Ca is well established (79). To address the 10% reduction in calcium absorption caused by caloric restriction (29), we conducted a double-blind RCT in 82 overweight/obese women who were assigned to 2,500 IU of vitamin D/d (Hi-D) compared to 400 IU/d. We used dual stable isotopes of calcium and measured true fractional calcium absorption (TFCA) at baseline and after six weeks of CR. Our preliminary results showed a greater rise in 25OHD (+8 ng/ml) and no reduction in TFCA in the Hi-D group, whereas the 400 IU/d vitamin D group showed no change in 25OHD (+1 ng/ml) and the expected CR-induced reduction in TFCA (155). The results of this study indicate that CR is not associated with a decline in Ca absorption with higher vitamin D, and the possibility that this will attenuate bone loss due to weight reduction is currently being addressed.
Other nutrients and foods
The role of several other nutrients on bone has been examined in long-term clinical or animal studies. Studies consistently show a positive influence of dairy on BMD (107). Other foods, such as dietary amylase-resistant starch (15), also attenuate bone loss associated with weight reduction (and cycling) in rodents. Phytoestrogens and functional foods such as dried plum, flaxseed, and garlic have shown some positive effects on bone yet have not been studied specifically during CR. In addition, other vitamins and minerals have smaller effects on bone health. Overall, with the exception of vitamin D and calcium, there is little or no evidence for a role of other foods or nutrients in attenuating bone loss during CR.
Exercise-induced weight loss
The mechanical load on the skeleton may contribute to the higher bone mass in obesity and the bone loss due to weight reduction. Exercise training in adults reduces BMD loss in the presence of adequate calcium (135). The types of physical activity most likely to have beneficial effects on reducing fracture risk are those that include impact forces that generate both gravitation and muscle loading (94).
Exercise added to weight loss regimens attenuates bone loss at the hip or femoral neck compared with diet alone (147, 159, 184). Yet as compared with a weight-stable group, weight reduction with exercise in older individuals (148, 151, 167, 185) is associated with bone loss, suggesting that it can be attenuated but not prevented. When CR is combined with exercise, a few factors may attenuate the bone loss, including an increase in IGF-1 and a reduction in inflammation in adults (159) and children (10). In addition, exercise attenuates lumbar spine bone loss with 8% weight reduction in relatively lean (BMI of 25 kg/m2) perimenopausal women (148). It is possible that the improvement in physical function associated with exercise during dieting (150, 183) is just as important as increasing BMD to prevent falls and fractures. In children, weight loss may slow bone growth (146), and although it would be expected that exercise would have a positive effect, this has not been specifically addressed.
The mechanical loading advantage of excess body weight on bone may be reduced with weight reduction. However, weight loss resulting primarily from loss of fat rather than lean tissue (such as with exercise) should maintain this mechanical advantage on bone. Overall, exercise attenuates bone loss during conditions of weight stability and loss in older individuals, but some bone loss will occur nevertheless. Hence, factors in addition to reduced weight bearing are also contributing to the loss of bone associated with weight reduction.
Medication and other factors
Medications to treat osteoporosis, such as estrogen and raloxifene, during weight reduction will prevent bone loss (64), and a study in rodents shows that low-dose PTH can maintain normal bone formation during rapid weight loss (178). Several herbal supplements (194) have also shown a positive influence on BMD; however, use of medications or herbal remedies to prevent bone loss during caloric restriction remains an experimental approach.
Bariatric Surgery and Extreme Weight Loss
An increasing number of individuals with severe obesity are undergoing bariatric surgical procedures that are associated with substantial weight reduction and amelioration of obesity-related comorbidities. Bariatric surgery also increases the risk for development of metabolic bone disease (59, 60). The malabsorptive procedures [Roux-en-Y gastric bypass (GB) and duodenal switch] compared with those that only reduce stomach size (gastric banding) induce greater weight reduction as well as bone loss, persistence of high serum PTH, and a rise in 25OHD (60, 142, 160, 181). One study also found a rise in serum levels of 1,25OHD (160). A 27% to 35% weight loss six to 18 months after GB surgery results in BMD loss of 9% to 10% at the hip and 3% at the lumbar spine, despite adequate calcium intake of 1.2 g/d or more (31, 49, 57, 84, 181). In a longer (three-year) study, GB individuals lost 30 kg body weight, and BMD decreased by 13.6% and 5.5% at the femoral neck and lumbar spine, respectively (181), suggesting that bone loss continues but at a less rapid rate after weight stabilizes. The amount of bone lost from different sites may differ: Four years after GB surgery, women have higher lumbar spine BMD and lower femoral neck BMD in comparison with weight-matched controls who never had surgery (60). The differential bone loss in GB women may be attributed to the high serum PTH (60) or the presence of spinal osteoarthritis that overestimates BMD and is more common in patients with a history of severe obesity (103). The bone loss due to extreme weight reduction is not simply proportional to the loss in body weight. For example, BMD loss at the hip is two to three times greater than expected after GB compared with 1% to 2% BMD loss found with 10% weight loss. This may be partially related to the 33% reduction in Ca absorption due to GB surgery (142). However, because absolute absorption (24%) is not especially low after GB, other factors are also contributing to bone loss, such as the potential incidence of other micronutrient deficiencies (4), hormonal factors, and/or reduced weight bearing.
It does not appear that serum 25OHD influences bone loss, at least during the first year, since 25OHD levels rise after Roux-en-Y GB or gastric banding (49, 84, 160). In addition, supplementing with higher vitamin D does not further raise serum 25OHD (49, 160), although this has not been examined in a controlled trial. Although the American Society for Metabolic and Bariatric Surgery recommends high levels of Ca (1.5–2 g/day) and vitamin D intake (2,000 IU/day) following surgery (2), exact levels associated with beneficial outcomes are not known due to the lack of RCTs.
Weight Cycling
Weight cycling, defined as weight loss followed by regain (one or more times), is a concern because lean body mass is not typically regained as much as fat is with weight regain (8, 99). For example, Fogelholm et al. (51) found that women with a history of weight cycling have lower spine and distal radius BMD in comparison with those who are not weight cyclers. Importantly, Meyer et al. (111) showed that greater weight variability was associated with increased hip fracture incidence in women and men over a 12-year period (ages 50–62 years). Restrained eaters who chronically diet or who weight cycle may be at greater risk of low bone mass or osteoporosis (3, 15, 111). Other researchers have not found an effect of weight cycling on bone, such as a cross-sectional study in younger pre-menopausal women (50, 55). After successful weight reduction in the obese individual, maintaining the reduced body weight over a longer duration may result in further bone loss even after weight stabilizes (182, 186) or when preserving muscle mass with the addition of exercise (75). In addition, individuals with lower compared with higher body weight who weight cycle would be expected to lose more bone (118).
Bone Measurement Concerns: Obesity and Weight Loss
Excess fat tissue surrounding the bone in obesity may result in DXA measurement errors for BMD (13, 17, 70, 176). The lack of homogeneity of the adipose tissue and fat overlying a specific bone site will affect the amount of error. Furthermore, measurement of sites with less surrounding fat thickness (for example, a peripheral site such as forearm or leg rather than axial sites such as spine) may reduce BMD artifacts in the obese (70). In addition, QCT, which measures true volumetric bone density (mg/cm3) and uses the density of fat tissue as zero, can reduce artifacts associated with a two-dimensional measurement of areal bone density (g/cm2) by DXA (196). Reporting BMC (g) is helpful because it is not an areal measurement, as is BMD; BMC (g) is typically reported in children due to its higher reproducibility (61). In general, measurement error in the lumbar spine (196) due to excess abdominal fat in the obese makes this site less desirable than the hip or other sites. In addition, obese individuals are at greater risk of spinal osteoarthritis and osteophytes (103), which contribute to the overestimation of bone mass. In our experience, the prevalence of spinal osteoarthritis in post-menopausal obese women is ~43% (140, 141). The International Society for Clinical Densitometry established criteria for vertebral exclusion to increase precision in patients with spinal deformities (101). An additional concern about BMD measurements in the obese is the difficulty of positioning them within the scan area on the DXA bed (23).
Weight reduction also alters the precision of DXA because of changes in the soft tissue surrounding the bone (136, 168, 176), but it does not cause systematic errors in longitudinal results in DXA studies (13). Experiments that simulate increases in body fat by sequential fat layering on bone show that this overestimates BMD measurements by DXA (82, 112, 196). In general, the accuracy of DXA measurements in extreme obesity (BMI >40 kg/m2) is reduced, and studies examining extreme weight loss (typically about 35 kg) after gastric bypass would be expected to overestimate the decrease in BMD. Importantly, a reduction in BMD as a result of weight loss is not questioned because of the evidence in animal models and the higher risk of fracture, but there remain concerns about precision and the intrinsic limitations of measuring bone due to the varying amounts of excess soft tissue over time. However, this would primarily affect the interpretation of studies examining bone changes after extreme weight loss or studies of very lean individuals with weight gain because the errors are greater in individuals at the extremes of body weight. There is less concern about measurement error in studies with moderate weight loss or in studies with a control group. Instruments such as QCT, peripheral QCT, and magnetic resonance imaging can also reduce measurement error while addressing bone quality and the location of fat and should be considered for future trials.
CONCLUSION
Obesity and weight loss are both associated with alterations in bone metabolism. With a significant percentage of the population being either obese and/or undergoing weight reduction, the influence of these conditions on bone metabolism requires attention. The poor bone quality and higher fracture risk in obese individuals are only beginning to be understood, and well-designed prospective trials are required to address these issues and to explicate the mechanisms that regulate the effect of excess adiposity on bone. The use of additional sensitive imaging techniques to measure bone quality and the interaction of bone with the amount and location of adipose tissue will improve our understanding of fracture risk in different populations. Several clinical trials have investigated the effect of nutrient supplementation on bone during CR, and nutrients such as calcium, protein, and vitamin D alone or in combination have shown positive results. However, bone loss is not completely attenuated with nutrient supplementation during CR. The use of weight reduction and/or lifestyle changes to reduce the comorbidities associated with obesity needs to be encouraged, but it should be balanced with methods to prevent the risk of osteoporosis. Overall, understanding the mechanisms regulating bone in obesity and during CR should help address concerns about declining bone health with aging and in achieving peak bone density and strength in children.
SUMMARY POINTS.
Adipogenesis and osteoblastogenesis share a common cellular relationship.
Endocrine factors and body composition influence bone mineral density in obesity.
Bone quality is compromised in obesity.
Caloric restriction leads to changes in calcium metabolism, resulting in bone loss. Endocrine and other physiological changes during weight reduction regulate bone loss.
Nutrient, exercise, and drug interventions partially attenuate bone loss associated with caloric restriction.
FUTURE ISSUES.
Well-designed prospective trials are required to investigate mechanisms regulating bone in the presence of excess adiposity.
The mechanisms regulating bone during caloric restriction can only partially explain the bone loss during and after weight loss and require further study.
Our understanding of fracture risk in different populations will be improved with the use of additional sensitive techniques to measure bone quality and its interaction with the amount as well as type of fat.
Glossary
- BMD
bone mineral density
- BMC
bone mineral content
- PTH
parathyroid hormone
- 25OHD
25 hydroxyvitamin D
- DXA
dual-energy x-ray absorptiometry
LITERATURE CITED
- 1.Ahdjoudj S, Lasmoles F, Holy X, Zerath E, Marie PJ. Transforming growth factor beta2 inhibits adipocyte differentiation induced by skeletal unloading in rat bone marrow stroma. J Bone Miner Res. 2002;17:668–77. doi: 10.1359/jbmr.2002.17.4.668. [DOI] [PubMed] [Google Scholar]
- 2.Aills L, Blankenship J, Buffington C, Furtado M, Parrott J. ASMBS Allied Health Nutritional Guidelines for the Surgical Weight Loss Patient. Surg Obes Relat Dis. 2008;4:S73–108. doi: 10.1016/j.soard.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 3.Bacon L, Stern JS, Keim NL, Van Loan MD. Low bone mass in premenopausal chronic dieting obese women. Eur J Clin Nutr. 2004;58:966–71. doi: 10.1038/sj.ejcn.1601922. [DOI] [PubMed] [Google Scholar]
- 4.Bal BS, Finelli FC, Koch TR. Origins of and recognition of micronutrient deficiencies after gastric bypass surgery. Curr Diab Rep. 2011;11:136–41. doi: 10.1007/s11892-010-0169-4. [DOI] [PubMed] [Google Scholar]
- 5.Balistreri CR, Caruso C, Candore G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediators Inflamm. 2010;2010:802078. doi: 10.1155/2010/802078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barbour KE, Zmuda JM, Boudreau R, Strotmeyer ES, Horwitz MJ, et al. Adipokines and the risk of fracture in older adults. J Bone Miner Res. 2011;26:1568–76. doi: 10.1002/jbmr.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barbour KE, Zmuda JM, Boudreau R, Strotmeyer ES, Horwitz MJ, et al. The effects of adiponectin and leptin on changes in bone mineral density. Osteoporos Int. 2011 doi: 10.1007/s00198-011-1768-x. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Beavers KM, Lyles MF, Davis CC, Wang X, Beavers DP, Nicklas BJ. Is lost lean mass from intentional weight loss recovered during weight regain in postmenopausal women? Am J Clin Nutr. 2011;94:767–74. doi: 10.3945/ajcn.110.004895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bedford JL, Barr SI. The relationship between 24-h urinary cortisol and bone in healthy young women. Int J Behav Med. 2010;17:207–15. doi: 10.1007/s12529-009-9064-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ben OO, Elloumi M, Zouhal H, Makni E, Denguezli M, et al. Effect of individualized exercise training combined with diet restriction on inflammatory markers and IGF-1/IGFBP-3 in obese children. Ann Nutr Metab. 2010;56:260–66. doi: 10.1159/000275888. [DOI] [PubMed] [Google Scholar]
- 11.Bertolini DR, Nedwin GE, Bringman TS, Smith DD, Mundy GR. Stimulation of bone resorption and inhibition of bone formation in vitro by human tumour necrosis factors. Nature. 1986;319:516–18. doi: 10.1038/319516a0. [DOI] [PubMed] [Google Scholar]
- 12.Biver E, Salliot C, Combescure C, Gossec L, Hardouin P, et al. Influence of adipokines and ghrelin on bone mineral density and fracture risk: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2011;96:2703–13. doi: 10.1210/jc.2011-0047. [DOI] [PubMed] [Google Scholar]
- 13.Blake GM, Fogelman I. How important are BMD accuracy errors for the clinical interpretation of DXA scans? J Bone Miner Res. 2008;23:457–62. doi: 10.1359/jbmr.071119. [DOI] [PubMed] [Google Scholar]
- 14.Bleicher K, Cumming RG, Naganathan V, Travison TG, Sambrook PN, et al. The role of fat and lean mass in bone loss in older men: findings from the CHAMP study. Bone. 2011;49:1299–305. doi: 10.1016/j.bone.2011.08.026. [DOI] [PubMed] [Google Scholar]
- 15.Bogden JD, Kemp FW, Huang AE, Shapses SA, Ambia-Sobhan H, et al. Bone mineral density and content during weight cycling in female rats: effects of dietary amylase-resistant starch. Nutr Metab (Lond) 2008;5:34. doi: 10.1186/1743-7075-5-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bolland MJ, Grey AB, Ames RW, Horne AM, Gamble GD, Reid IR. Fat mass is an important predictor of parathyroid hormone levels in postmenopausal women. Bone. 2006;38:317–21. doi: 10.1016/j.bone.2005.08.018. [DOI] [PubMed] [Google Scholar]
- 17.Bolotin HH. A new perspective on the causal influence of soft tissue composition on DXA-measured in vivo bone mineral density. J Bone Miner Res. 1998;13:1739–46. doi: 10.1359/jbmr.1998.13.11.1739. [DOI] [PubMed] [Google Scholar]
- 18.Bosy-Westphal A, Later W, Schautz B, Lagerpusch M, Goele K, et al. Impact of intra- and extra-osseous soft tissue composition on changes in bone mineral density with weight loss and regain. Obesity (Silver Spring) 2011;19:1503–10. doi: 10.1038/oby.2011.40. [DOI] [PubMed] [Google Scholar]
- 19.Bouxsein ML, Seeman E. Quantifying the material and structural determinants of bone strength. Best Pract Res Clin Rheumatol. 2009;23:741–53. doi: 10.1016/j.berh.2009.09.008. [DOI] [PubMed] [Google Scholar]
- 20.Bredella MA, Fazeli PK, Freedman LM, Calder G, Lee H, et al. Young women with cold-activated brown adipose tissue have higher bone mineral density and lower Pref-1 than women without brown adipose tissue: a study in women with anorexia nervosa, women recovered from anorexia nervosa, and normal-weight women. J Clin Endocrinol Metab. 2012;97:E584–90. doi: 10.1210/jc.2011-2246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bronsky J, Prusa R, Nevoral J. The role of amylin and related peptides in osteoporosis. Clin Chim Acta. 2006;373:9–16. doi: 10.1016/j.cca.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 22.Brouwer DA, van Beek J, Ferwerda H, Brugman AM, van der Klis FR, et al. Rat adipose tissue rapidly accumulates and slowly releases an orally-administered high vitamin D dose. Br J Nutr. 1998;79:527–32. doi: 10.1079/bjn19980091. [DOI] [PubMed] [Google Scholar]
- 23.Brownbill RA, Ilich JZ. Measuring body composition in overweight individuals by dual energy x-ray absorptiometry. BMC Med Imaging. 2005;5:1. doi: 10.1186/1471-2342-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cao JJ, Sun L, Gao H. Diet-induced obesity alters bone remodeling leading to decreased femoral trabecular bone mass in mice. Ann N Y Acad Sci. 2010;1192:292–97. doi: 10.1111/j.1749-6632.2009.05252.x. [DOI] [PubMed] [Google Scholar]
- 25.Charopoulos I, Tournis S, Trovas G, Raptou P, Kaldrymides P, et al. Effect of primary hyper-parathyroidism on volumetric bone mineral density and bone geometry assessed by peripheral quantitative computed tomography in postmenopausal women. J Clin Endocrinol Metab. 2006;91:1748–53. doi: 10.1210/jc.2005-2102. [DOI] [PubMed] [Google Scholar]
- 26.Chen Z, Lohman TG, Stini WA, Ritenbaugh C, Aickin M. Fat or lean tissue mass: Which one is the major determinant of bone mineral mass in healthy postmenopausal women? J Bone Miner Res. 1997;12:144–51. doi: 10.1359/jbmr.1997.12.1.144. [DOI] [PubMed] [Google Scholar]
- 27.Cifuentes M, Johnson MA, Lewis RD, Heymsfield SB, Chowdhury HA, et al. Bone turnover and body weight relationships differ in normal-weight compared with heavier postmenopausal women. Osteoporos Int. 2003;14:116–22. doi: 10.1007/s00198-002-1324-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cifuentes M, Morano AB, Chowdhury HA, Shapses SA. Energy restriction reduces fractional calcium absorption in mature obese and lean rats. J Nutr. 2002;132:2660–66. doi: 10.1093/jn/132.9.2660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cifuentes M, Riedt CS, Brolin RE, Field MP, Sherrell RM, Shapses SA. Weight loss and calcium intake influence calcium absorption in overweight postmenopausal women. Am J Clin Nutr. 2004;80:123–30. doi: 10.1093/ajcn/80.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Clowes JA, Khosla S, Eastell R. Potential role of pancreatic and enteric hormones in regulating bone turnover. J Bone Miner Res. 2005;20:1497–506. doi: 10.1359/JBMR.050524. [DOI] [PubMed] [Google Scholar]
- 31.Coates PS, Fernstrom JD, Fernstrom MH, Schauer PR, Greenspan SL. Gastric bypass surgery for morbid obesity leads to an increase in bone turnover and a decrease in bone mass. J Clin Endocrinol Metab. 2004;89:1061–65. doi: 10.1210/jc.2003-031756. [DOI] [PubMed] [Google Scholar]
- 32.Compston JE, Watts NB, Chapurlat R, Cooper C, Boonen S, et al. Obesity is not protective against fracture in postmenopausal women: GLOW. Am J Med. 2011;124:1043–50. doi: 10.1016/j.amjmed.2011.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Conigrave AD, Brown EM, Rizzoli R. Dietary protein and bone health: roles of amino acid-sensing receptors in the control of calcium metabolism and bone homeostasis. Annu Rev Nutr. 2008;28:131–55. doi: 10.1146/annurev.nutr.28.061807.155328. [DOI] [PubMed] [Google Scholar]
- 34.Conroy R, Girotra M, Shane E, McMahon DJ, Pavlovich KH, et al. Leptin administration does not prevent the bone mineral metabolism changes induced by weight loss. Metabolism. 2011;60:1222–26. doi: 10.1016/j.metabol.2011.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cornish J, Callon KE, Bava U, Watson M, Xu X, et al. Preptin, another peptide product of the pancreatic beta-cell, is osteogenic in vitro and in vivo. Am J Physiol Endocrinol Metab. 2007;292:E117–22. doi: 10.1152/ajpendo.00642.2005. [DOI] [PubMed] [Google Scholar]
- 36.Cummings SR, Nevitt MC. Non-skeletal determinants of fractures: the potential importance of the mechanics of falls. Study of Osteoporotic Fractures Research Group. Osteoporos Int. 1994;4(Suppl 1):67–70. doi: 10.1007/BF01623439. [DOI] [PubMed] [Google Scholar]
- 37.David V, Martin A, Lafage-Proust MH, Malaval L, Peyroche S, et al. Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis. Endocrinology. 2007;148:2553–62. doi: 10.1210/en.2006-1704. [DOI] [PubMed] [Google Scholar]
- 38.De Laet C, Kanis JA, Oden A, Johanson H, Johnell O, et al. Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporos Int. 2005;16:1330–38. doi: 10.1007/s00198-005-1863-y. [DOI] [PubMed] [Google Scholar]
- 39.Dimitri P, Bishop N, Walsh JS, Eastell R. Obesity is a risk factor for fracture in children but is protective against fracture in adults: a paradox. Bone. 2012;50:457–66. doi: 10.1016/j.bone.2011.05.011. [DOI] [PubMed] [Google Scholar]
- 40.Dimitri P, Wales JK, Bishop N. Fat and bone in children: differential effects of obesity on bone size and mass according to fracture history. J Bone Miner Res. 2010;25:527–36. doi: 10.1359/jbmr.090823. [DOI] [PubMed] [Google Scholar]
- 41.Dimitri P, Wales JK, Bishop N. Adipokines, bone-derived factors and bone turnover in obese children; evidence for altered fat-bone signalling resulting in reduced bone mass. Bone. 2011;48:189–96. doi: 10.1016/j.bone.2010.09.034. [DOI] [PubMed] [Google Scholar]
- 42.Douchi T, Yamamoto S, Oki T, Maruta K, Kuwahata R, et al. Difference in the effect of adiposity on bone density between pre- and postmenopausal women. Maturitas. 2000;34:261–66. doi: 10.1016/s0378-5122(99)00114-0. [DOI] [PubMed] [Google Scholar]
- 43.Ehrlich PJ, Noble BS, Jessop HL, Stevens HY, Mosley JR, Lanyon LE. The effect of in vivo mechanical loading on estrogen receptor alpha expression in rat ulnar osteocytes. J Bone Miner Res. 2002;17:1646–55. doi: 10.1359/jbmr.2002.17.9.1646. [DOI] [PubMed] [Google Scholar]
- 44.Ensrud KE, Cauley J, Lipschutz R, Cummings SR. Weight change and fractures in older women. Study of Osteoporotic Fractures Research Group. Arch Intern Med. 1997;157:857–63. [PubMed] [Google Scholar]
- 45.Ensrud KE, Fullman RL, Barrett-Connor E, Cauley JA, Stefanick ML, et al. Voluntary weight reduction in older men increases hip bone loss: the Osteoporotic Fractures in Men study. J Clin Endocrinol Metab. 2005;90:1998–2004. doi: 10.1210/jc.2004-1805. [DOI] [PubMed] [Google Scholar]
- 46.Eriksen EF, Colvard DS, Berg NJ, Graham ML, Mann KG, et al. Evidence of estrogen receptors in normal human osteoblast-like cells. Science. 1988;241:84–86. doi: 10.1126/science.3388021. [DOI] [PubMed] [Google Scholar]
- 47.Ettinger B, Pressman A, Sklarin P, Bauer DC, Cauley JA, Cummings SR. Associations between low levels of serum estradiol, bone density, and fractures among elderly women: the Study of Osteoporotic Fractures. J Clin Endocrinol Metab. 1998;83:2239–43. doi: 10.1210/jcem.83.7.4708. [DOI] [PubMed] [Google Scholar]
- 48.FastStats. Obesity and Overweight. Atlanta, GA: CDC; 2011. http://www.cdc.gov/nchs/fastats/overwt.htm. [Google Scholar]
- 49.Fleischer J, Stein EM, Bessler M, Della BM, Restuccia N, et al. The decline in hip bone density after gastric bypass surgery is associated with extent of weight loss. J Clin Endocrinol Metab. 2008;93:3735–40. doi: 10.1210/jc.2008-0481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fogelholm GM, Sievanen HT, Kukkonen-Harjula TK, Pasanen ME. Bone mineral density during reduction, maintenance and regain of body weight in premenopausal, obese women. Osteoporos Int. 2001;12:199–206. doi: 10.1007/s001980170130. [DOI] [PubMed] [Google Scholar]
- 51.Fogelholm M, Sievanen H, Heinonen A, Virtanen M, Uusi-Rasi K, et al. Association between weight cycling history and bone mineral density in premenopausal women. Osteoporos Int. 1997;7:354–58. doi: 10.1007/BF01623777. [DOI] [PubMed] [Google Scholar]
- 52.Frost HM. Bone “mass” and the “mechanostat”: a proposal. Anat Rec. 1987;219:1–9. doi: 10.1002/ar.1092190104. [DOI] [PubMed] [Google Scholar]
- 53.Frost HM. Obesity, and bone strength and “mass”: a tutorial based on insights from a new paradigm. Bone. 1997;21:211–14. doi: 10.1016/s8756-3282(97)00124-5. [DOI] [PubMed] [Google Scholar]
- 54.Fukushima N, Hanada R, Teranishi H, Fukue Y, Tachibana T, et al. Ghrelin directly regulates bone formation. J Bone Miner Res. 2005;20:790–98. doi: 10.1359/JBMR.041237. [DOI] [PubMed] [Google Scholar]
- 55.Gallagher KI, Jakicic JM, Kiel DP, Page ML, Ferguson ES, Marcus BH. Impact of weight-cycling history on bone density in obese women. Obes Res. 2002;10:896–902. doi: 10.1038/oby.2002.123. [DOI] [PubMed] [Google Scholar]
- 56.Gilsanz V, Chalfant J, Mo AO, Lee DC, Dorey FJ, Mittelman SD. Reciprocal relations of subcutaneous and visceral fat to bone structure and strength. J Clin Endocrinol Metab. 2009;94:3387–93. doi: 10.1210/jc.2008-2422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Giusti V, Gasteyger C, Suter M, Heraief E, Gaillard RC, Burckhardt P. Gastric banding induces negative bone remodelling in the absence of secondary hyperparathyroidism: potential role of serum C telopeptides for follow-up. Int J Obes (Lond) 2005;29:1429–35. doi: 10.1038/sj.ijo.0803040. [DOI] [PubMed] [Google Scholar]
- 58.Gnudi S, Sitta E, Lisi L. Relationship of body mass index with main limb fragility fractures in postmenopausal women. J Bone Miner Metab. 2009;27:479–84. doi: 10.1007/s00774-009-0056-8. [DOI] [PubMed] [Google Scholar]
- 59.Goldner WS, O’Dorisio TM, Dillon JS, Mason EE. Severe metabolic bone disease as a long-term complication of obesity surgery. Obes Surg. 2002;12:685–92. doi: 10.1381/096089202321019693. [DOI] [PubMed] [Google Scholar]
- 60.Goode LR, Brolin RE, Chowdhury HA, Shapses SA. Bone and gastric bypass surgery: effects of dietary calcium and vitamin D. Obes Res. 2004;12:40–47. doi: 10.1038/oby.2004.7. [DOI] [PubMed] [Google Scholar]
- 61.Gordon CM, Baim S, Bianchi ML, Bishop NJ, Hans DB, et al. Special report on the 2007 Pediatric Position Development Conference of the International Society for Clinical Densitometry. South Med J. 2008;101:740–43. doi: 10.1097/SMJ.0b013e31817a8b55. [DOI] [PubMed] [Google Scholar]
- 62.Goulding A, Jones IE, Taylor RW, Piggot JM, Taylor D. Dynamic and static tests of balance and postural sway in boys: effects of previous wrist bone fractures and high adiposity. Gait Posture. 2003;17:136–41. doi: 10.1016/s0966-6362(02)00161-3. [DOI] [PubMed] [Google Scholar]
- 63.Goulding A, Jones IE, Taylor RW, Williams SM, Manning PJ. Bone mineral density and body composition in boys with distal forearm fractures: a dual-energy x-ray absorptiometry study. J Pediatr. 2001;139:509–15. doi: 10.1067/mpd.2001.116297. [DOI] [PubMed] [Google Scholar]
- 64.Gozansky WS, Van Pelt RE, Jankowski CM, Schwartz RS, Kohrt WM. Protection of bone mass by estrogens and raloxifene during exercise-induced weight loss. J Clin Endocrinol Metab. 2005;90:52–59. doi: 10.1210/jc.2004-0275. [DOI] [PubMed] [Google Scholar]
- 65.Grey A, Mitnick MA, Shapses S, Ellison A, Gundberg C, Insogna K. Circulating levels of interleukin-6 and tumor necrosis factor-alpha are elevated in primary hyperparathyroidism and correlate with markers of bone resorption—a clinical research center study. J Clin Endocrinol Metab. 1996;81:3450–54. doi: 10.1210/jcem.81.10.8855783. [DOI] [PubMed] [Google Scholar]
- 66.Gudmundsdottir SL, Oskarsdottir D, Indridason OS, Franzson L, Sigurdsson G. Risk factors for bone loss in the hip of 75-year-old women: a 4-year follow-up study. Maturitas. 2010;67:256–61. doi: 10.1016/j.maturitas.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 67.Guri AJ, Bassaganya-Riera J. Systemic effects of white adipose tissue dysregulation and obesity-related inflammation. Obesity (Silver Spring) 2011;19:689–700. doi: 10.1038/oby.2010.230. [DOI] [PubMed] [Google Scholar]
- 68.Habib ZA, Havstad SL, Wells K, Divine G, Pladevall M, Williams LK. Thiazolidinedione use and the longitudinal risk of fractures in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab. 2010;95:592–600. doi: 10.1210/jc.2009-1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hamrick MW, Ding KH, Ponnala S, Ferrari SL, Isales CM. Caloric restriction decreases cortical bone mass but spares trabecular bone in the mouse skeleton: implications for the regulation of bone mass by body weight. J Bone Miner Res. 2008;23:870–78. doi: 10.1359/jbmr.080213. [DOI] [PubMed] [Google Scholar]
- 70.Hangartner TN, Johnston CC. Influence of fat on bone measurements with dual-energy absorptiometry. Bone Miner. 1990;9:71–81. doi: 10.1016/0169-6009(90)90101-k. [DOI] [PubMed] [Google Scholar]
- 71.Hawkins J, Cifuentes M, Pleshko NL, Ambia-Sobhan H, Shapses SA. Energy restriction is associated with lower bone mineral density of the tibia and femur in lean but not obese female rats. J Nutr. 2010;140:31–37. doi: 10.3945/jn.109.111450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Heaney RP. The bone-remodeling transient: implications for the interpretation of clinical studies of bone mass change. J Bone Miner Res. 1994;9:1515–23. doi: 10.1002/jbmr.5650091003. [DOI] [PubMed] [Google Scholar]
- 73.Heino TJ, Hentunen TA, Vaananen HK. Osteocytes inhibit osteoclastic bone resorption through transforming growth factor-beta: enhancement by estrogen. J Cell Biochem. 2002;85:185–97. doi: 10.1002/jcb.10109. [DOI] [PubMed] [Google Scholar]
- 74.Hill KM, Braun MM, Egan KA, Martin BR, McCabe LD, et al. Obesity augments calcium-induced increases in skeletal calcium retention in adolescents. J Clin Endocrinol Metab. 2011;96:2171–77. doi: 10.1210/jc.2010-2709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hinton PS, Rector RS, Linden MA, Warner SO, Dellsperger KC, et al. Weight-loss-associated changes in bone mineral density and bone turnover after partial weight regain with or without aerobic exercise in obese women. Eur J Clin Nutr. 2012;66:606–12. doi: 10.1038/ejcn.2011.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Holbrook TL, Barrett-Connor E. The association of lifetime weight and weight control patterns with bone mineral density in an adult community. Bone Miner. 1993;20:141–49. doi: 10.1016/s0169-6009(08)80023-2. [DOI] [PubMed] [Google Scholar]
- 77.Holloway WR, Collier FM, Aitken CJ, Myers DE, Hodge JM, et al. Leptin inhibits osteoclast generation. J Bone Miner Res. 2002;17:200–9. doi: 10.1359/jbmr.2002.17.2.200. [DOI] [PubMed] [Google Scholar]
- 78.Holmberg AH, Johnell O, Nilsson PM, Nilsson J, Berglund G, Akesson K. Risk factors for fragility fracture in middle age. A prospective population-based study of 33,000 men and women. Osteoporos Int. 2006;17:1065–77. doi: 10.1007/s00198-006-0137-7. [DOI] [PubMed] [Google Scholar]
- 79.Inst. Med. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: Natl. Acad. Press; 2011. [Google Scholar]
- 80.Janicka A, Wren TA, Sanchez MM, Dorey F, Kim PS, et al. Fat mass is not beneficial to bone in adolescents and young adults. J Clin Endocrinol Metab. 2007;92:143–47. doi: 10.1210/jc.2006-0794. [DOI] [PubMed] [Google Scholar]
- 81.Jensen LB, Kollerup G, Quaade F, Sorensen OH. Bone minerals changes in obese women during a moderate weight loss with and without calcium supplementation. J Bone Miner Res. 2001;16:141–47. doi: 10.1359/jbmr.2001.16.1.141. [DOI] [PubMed] [Google Scholar]
- 82.Jensen LB, Quaade F, Sorensen OH. Bone loss accompanying voluntary weight loss in obese humans. J Bone Miner Res. 1994;9:459–63. doi: 10.1002/jbmr.5650090404. [DOI] [PubMed] [Google Scholar]
- 83.Jilka RL, Hangoc G, Girasole G, Passeri G, Williams DC, et al. Increased osteoclast development after estrogen loss: mediation by interleukin-6. Science. 1992;257:88–91. doi: 10.1126/science.1621100. [DOI] [PubMed] [Google Scholar]
- 84.Johnson JM, Maher JW, Samuel I, Heitshusen D, Doherty C, Downs RW. Effects of gastric bypass procedures on bone mineral density, calcium, parathyroid hormone, and vitamin D. J Gastrointest Surg. 2005;9:1106–10. doi: 10.1016/j.gassur.2005.07.012. [DOI] [PubMed] [Google Scholar]
- 85.Jurimae J, Rembel K, Jurimae T, Rehand M. Adiponectin is associated with bone mineral density in perimenopausal women. Horm Metab Res. 2005;37:297–302. doi: 10.1055/s-2005-861483. [DOI] [PubMed] [Google Scholar]
- 86.Karra E, Batterham RL. The role of gut hormones in the regulation of body weight and energy homeostasis. Mol Cell Endocrinol. 2010;316:120–28. doi: 10.1016/j.mce.2009.06.010. [DOI] [PubMed] [Google Scholar]
- 87.Karsenty G, Elefteriou F. Neuronal regulation of bone remodeling. In: Rosen CJ, Compston JE, Lian JB, editors. Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism. 7 Vol. 1. Hoboken, NJ: Wiley; 2008. pp. 56–60. [Google Scholar]
- 88.Kerstetter JE, O’Brien KO, Caseria DM, Wall DE, Insogna KL. The impact of dietary protein on calcium absorption and kinetic measures of bone turnover in women. J Clin Endocrinol Metab. 2005;90:26–31. doi: 10.1210/jc.2004-0179. [DOI] [PubMed] [Google Scholar]
- 89.Khosla S, Atkinson EJ, Riggs BL, Melton LJ., III Relationship between body composition and bone mass in women. J Bone Miner Res. 1996;11:857–63. doi: 10.1002/jbmr.5650110618. [DOI] [PubMed] [Google Scholar]
- 90.Khosla S, Melton LJ, III, Atkinson EJ, O’Fallon WM, Klee GG, Riggs BL. Relationship of serum sex steroid levels and bone turnover markers with bone mineral density in men and women: a key role for bioavailable estrogen. J Clin Endocrinol Metab. 1998;83:2266–74. doi: 10.1210/jcem.83.7.4924. [DOI] [PubMed] [Google Scholar]
- 91.Kim HJ, Lee S, Kim TW, Kim HH, Jeon TY, et al. Effects of exercise-induced weight loss on acylated and unacylated ghrelin in overweight children. Clin Endocrinol (Oxf) 2008;68:416–22. doi: 10.1111/j.1365-2265.2007.03058.x. [DOI] [PubMed] [Google Scholar]
- 92.Klempel MC, Varady KA. Reliability of leptin, but not adiponectin, as a biomarker for diet-induced weight loss in humans. Nutr Rev. 2011;69:145–54. doi: 10.1111/j.1753-4887.2011.00373.x. [DOI] [PubMed] [Google Scholar]
- 93.Koh JM, Khang YH, Jung CH, Bae S, Kim DJ, et al. Higher circulating hsCRP levels are associated with lower bone mineral density in healthy pre- and postmenopausal women: evidence for a link between systemic inflammation and osteoporosis. Osteoporos Int. 2005;16:1263–71. doi: 10.1007/s00198-005-1840-5. [DOI] [PubMed] [Google Scholar]
- 94.Kohrt WM, Barry DW, Schwartz RS. Muscle forces or gravity: What predominates mechanical loading on bone? Med Sci Sports Exerc. 2009;41:2050–55. doi: 10.1249/MSS.0b013e3181a8c717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Konradsen S, Ag H, Lindberg F, Hexeberg S, Jorde R. Serum 1,25-dihydroxyvitamin D is inversely associated with body mass index. Eur J Nutr. 2008;47:87–91. doi: 10.1007/s00394-008-0700-4. [DOI] [PubMed] [Google Scholar]
- 96.Kuwahata A, Kawamura Y, Yonehara Y, Matsuo T, Iwamoto I, Douchi T. Non-weight-bearing effect of trunk and peripheral fat mass on bone mineral density in pre- and post-menopausal women. Maturitas. 2008;60:244–47. doi: 10.1016/j.maturitas.2008.07.005. [DOI] [PubMed] [Google Scholar]
- 97.Langlois JA, Harris T, Looker AC, Madans J. Weight change between age 50 years and old age is associated with risk of hip fracture in white women aged 67 years and older. Arch Intern Med. 1996;156:989–94. [PubMed] [Google Scholar]
- 98.Langlois JA, Mussolino ME, Visser M, Looker AC, Harris T, Madans J. Weight loss from maximum body weight among middle-aged and older white women and the risk of hip fracture: the NHANES I Epidemiologic Follow-Up Study. Osteoporos Int. 2001;12:763–68. doi: 10.1007/s001980170053. [DOI] [PubMed] [Google Scholar]
- 99.Lee JS, Visser M, Tylavsky FA, Kritchevsky SB, Schwartz AV, et al. Weight loss and regain and effects on body composition: the Health, Aging, and Body Composition Study. J Gerontol A Biol Sci Med Sci. 2010;65:78–83. doi: 10.1093/gerona/glp042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Lenchik L, Register TC, Hsu FC, Lohman K, Nicklas BJ, et al. Adiponectin as a novel determinant of bone mineral density and visceral fat. Bone. 2003;33:646–51. doi: 10.1016/s8756-3282(03)00237-0. [DOI] [PubMed] [Google Scholar]
- 101.Lewiecki EM, Gordon CM, Baim S, Leonard MB, Bishop NJ, et al. International Society for Clinical Densitometry 2007 Adult and Pediatric Official Positions. Bone. 2008;43:1115–21. doi: 10.1016/j.bone.2008.08.106. [DOI] [PubMed] [Google Scholar]
- 102.Lindsay R, Cosman F, Herrington BS, Himmelstein S. Bone mass and body composition in normal women. J Bone Miner Res. 1992;7:55–63. doi: 10.1002/jbmr.5650070109. [DOI] [PubMed] [Google Scholar]
- 103.Liu G, Peacock M, Eilam O, Dorulla G, Braunstein E, Johnston CC. Effect of osteoarthritis in the lumbar spine and hip on bone mineral density and diagnosis of osteoporosis in elderly men and women. Osteoporos Int. 1997;7:564–69. doi: 10.1007/BF02652563. [DOI] [PubMed] [Google Scholar]
- 104.Luo XH, Guo LJ, Yuan LQ, Xie H, Zhou HD, et al. Adiponectin stimulates human osteoblasts proliferation and differentiation via the MAPK signaling pathway. Exp Cell Res. 2005;309:99–109. doi: 10.1016/j.yexcr.2005.05.021. [DOI] [PubMed] [Google Scholar]
- 105.Makovey J, Naganathan V, Sambrook P. Gender differences in relationships between body composition components, their distribution and bone mineral density: a cross-sectional opposite sex twin study. Osteoporos Int. 2005;16:1495–505. doi: 10.1007/s00198-005-1841-4. [DOI] [PubMed] [Google Scholar]
- 106.Mason C, Xiao L, Imayama I, Duggan CR, Bain C, et al. Effects of weight loss on serum vitamin D in postmenopausal women. Am J Clin Nutr. 2011;94:95–103. doi: 10.3945/ajcn.111.015552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.McCabe LD, Martin BR, McCabe GP, Johnston CC, Weaver CM, Peacock M. Dairy intakes affect bone density in the elderly. Am J Clin Nutr. 2004;80:1066–74. doi: 10.1093/ajcn/80.4.1066. [DOI] [PubMed] [Google Scholar]
- 108.Meunier P, Aaron J, Edouard C, Vignon G. Osteoporosis and the replacement of cell populations of the marrow by adipose tissue. A quantitative study of 84 iliac bone biopsies. Clin Orthop Relat Res. 1971;80:147–54. doi: 10.1097/00003086-197110000-00021. [DOI] [PubMed] [Google Scholar]
- 109.Meyer HE, Sogaard AJ, Falch JA, Jorgensen L, Emaus N. Weight change over three decades and the risk of osteoporosis in men: the Norwegian Epidemiological Osteoporosis Studies (NOREPOS) Am J Epidemiol. 2008;168:454–60. doi: 10.1093/aje/kwn151. [DOI] [PubMed] [Google Scholar]
- 110.Meyer HE, Tverdal A, Falch JA. Changes in body weight and incidence of hip fracture among middle-aged Norwegians. BMJ. 1995;311:91–92. doi: 10.1136/bmj.311.6997.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Meyer HE, Tverdal A, Selmer R. Weight variability, weight change and the incidence of hip fracture: a prospective study of 39,000 middle-aged Norwegians. Osteoporos Int. 1998;8:373–78. doi: 10.1007/s001980050077. [DOI] [PubMed] [Google Scholar]
- 112.Milliken LA, Going SB, Lohman TG. Effects of variations in regional composition on soft tissue measurements by dual-energy X-ray absorptiometry. Int J Obes Relat Metab Disord. 1996;20:677–82. [PubMed] [Google Scholar]
- 113.Misra M, Klibanski A. Bone metabolism in adolescents with anorexia nervosa. J Endocrinol Invest. 2011;34:324–32. doi: 10.3275/7505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Mundy GR. Osteoporosis and inflammation. Nutr Rev. 2007;65:S147–51. doi: 10.1111/j.1753-4887.2007.tb00353.x. [DOI] [PubMed] [Google Scholar]
- 115.Mussolino ME, Looker AC, Madans JH, Langlois JA, Orwoll ES. Risk factors for hip fracture in white men: the NHANES I Epidemiologic Follow-Up Study. J Bone Miner Res. 1998;13:918–24. doi: 10.1359/jbmr.1998.13.6.918. [DOI] [PubMed] [Google Scholar]
- 116.Natl. Osteoporos. Found. Prevalence Report. Washington, DC: Natl. Osteoporos. Found; 2011. http://www.nof.org/advocacy/resources/prevalencereport. [Google Scholar]
- 117.Nguyen ND, Center JR, Eisman JA, Nguyen TV. Bone loss, weight loss, and weight fluctuation predict mortality risk in elderly men and women. J Bone Miner Res. 2007;22:1147–54. doi: 10.1359/jbmr.070412. [DOI] [PubMed] [Google Scholar]
- 118.Nguyen TV, Sambrook PN, Eisman JA. Bone loss, physical activity, and weight change in elderly women: the Dubbo Osteoporosis Epidemiology Study. J Bone Miner Res. 1998;13:1458–67. doi: 10.1359/jbmr.1998.13.9.1458. [DOI] [PubMed] [Google Scholar]
- 119.Nielson CM, Marshall LM, Adams AL, Leblanc ES, Cawthon PM, et al. BMI and fracture risk in older men: the Osteoporotic Fractures in Men (MrOS) study. J Bone Miner Res. 2011;26(3):496–502. doi: 10.1002/jbmr.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Niu T, Rosen CJ. The insulin-like growth factor-I gene and osteoporosis: a critical appraisal. Gene. 2005;361:38–56. doi: 10.1016/j.gene.2005.07.016. [DOI] [PubMed] [Google Scholar]
- 121.O’Dea JP, Wieland RG, Hallberg MC, Llerena LA, Zorn EM, Genuth SM. Effect of dietery weight loss on sex steroid binding sex steroids, and gonadotropins in obese postmenopausal women. J Lab Clin Med. 1979;93:1004–8. [PubMed] [Google Scholar]
- 122.O’Loughlin PD, Morris HA. Oestrogen deficiency impairs intestinal calcium absorption in the rat. J Physiol. 1998;511(Pt. 1):313–22. doi: 10.1111/j.1469-7793.1998.313bi.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Omsland TK, Schei B, Gronskag AB, Langhammer A, Forsen L, et al. Weight loss and distal forearm fractures in postmenopausal women: the Nord-Trondelag health study, Norway. Osteoporos Int. 2009;20:2009–16. doi: 10.1007/s00198-009-0894-1. [DOI] [PubMed] [Google Scholar]
- 124.Oshima K, Nampei A, Matsuda M, Iwaki M, Fukuhara A, et al. Adiponectin increases bone mass by suppressing osteoclast and activating osteoblast. Biochem Biophys Res Commun. 2005;331:520–26. doi: 10.1016/j.bbrc.2005.03.210. [DOI] [PubMed] [Google Scholar]
- 125.Oster MH, Fielder PJ, Levin N, Cronin MJ. Adaptation of the growth hormone and insulin-like growth factor-I axis to chronic and severe calorie or protein malnutrition. J Clin Invest. 1995;95:2258–65. doi: 10.1172/JCI117916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Papakitsou EF, Margioris AN, Dretakis KE, Trovas G, Zoras U, et al. Body mass index (BMI) and parameters of bone formation and resorption in postmenopausal women. Maturitas. 2004;47:185–93. doi: 10.1016/S0378-5122(03)00282-2. [DOI] [PubMed] [Google Scholar]
- 127.Parhami F, Jackson SM, Tintut Y, Le V, Balucan JP, et al. Atherogenic diet and minimally oxidized low density lipoprotein inhibit osteogenic and promote adipogenic differentiation of marrow stromal cells. J Bone Miner Res. 1999;14:2067–78. doi: 10.1359/jbmr.1999.14.12.2067. [DOI] [PubMed] [Google Scholar]
- 128.Patsch JM, Kiefer FW, Varga P, Pail P, Rauner M, et al. Increased bone resorption and impaired bone microarchitecture in short-term and extended high-fat diet-induced obesity. Metabolism. 2011;60:243–49. doi: 10.1016/j.metabol.2009.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Peng XD, Xie H, Zhao Q, Wu XP, Sun ZQ, Liao EY. Relationships between serum adiponectin, leptin, resistin, visfatin levels and bone mineral density, and bone biochemical markers in Chinese men. Clin Chim Acta. 2008;387:31–35. doi: 10.1016/j.cca.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 130.Pistilli EE, Gordish-Dressman H, Seip RL, Devaney JM, Thompson PD, et al. Resistin polymorphisms are associated with muscle, bone, and fat phenotypes in white men and women. Obesity (Silver Spring) 2007;15:392–402. doi: 10.1038/oby.2007.543. [DOI] [PubMed] [Google Scholar]
- 131.Pitroda AP, Harris SS, Dawson-Hughes B. The association of adiposity with parathyroid hormone in healthy older adults. Endocrine. 2009;36:218–23. doi: 10.1007/s12020-009-9231-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Pollock NK, Laing EM, Baile CA, Hamrick MW, Hall DB, Lewis RD. Is adiposity advantageous for bone strength? A peripheral quantitative computed tomography study in late adolescent females. Am J Clin Nutr. 2007;86:1530–38. doi: 10.1093/ajcn/86.5.1530. [DOI] [PubMed] [Google Scholar]
- 133.Premaor MO, Pilbrow L, Tonkin C, Parker RA, Compston J. Obesity and fractures in post-menopausal women. J Bone Miner Res. 2010;25:292–97. doi: 10.1359/jbmr.091004. [DOI] [PubMed] [Google Scholar]
- 134.Prieto-Alhambra D, Premaor MO, Fina Avilés F, Hermosilla E, Martinez-Laguna D, et al. The association between fracture and obesity is site-dependent: a population-based study in postmenopausal women. J Bone Miner Res. 2012;27:294–300. doi: 10.1002/jbmr.1466. [DOI] [PubMed] [Google Scholar]
- 135.Prince RL, Smith M, Dick IM, Price RI, Webb PG, et al. Prevention of postmenopausal osteoporosis. A comparative study of exercise, calcium supplementation, and hormone-replacement therapy. N Engl J Med. 1991;325:1189–95. doi: 10.1056/NEJM199110243251701. [DOI] [PubMed] [Google Scholar]
- 136.Rajamanohara R, Robinson J, Rymer J, Patel R, Fogelman I, Blake GM. The effect of weight and weight change on the long-term precision of spine and hip DXA measurements. Osteoporos Int. 2011;22:1503–12. doi: 10.1007/s00198-010-1339-6. [DOI] [PubMed] [Google Scholar]
- 137.Rana AR, Michalsky MP, Teich S, Groner JI, Caniano DA, Schuster DP. Childhood obesity: a risk factor for injuries observed at a level-1 trauma center. J Pediatr Surg. 2009;44:1601–5. doi: 10.1016/j.jpedsurg.2008.11.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Ravn P, Cizza G, Bjarnason NH, Thompson D, Daley M, et al. Low body mass index is an important risk factor for low bone mass and increased bone loss in early postmenopausal women. Early Postmenopausal Intervention Cohort (EPIC) study group. J Bone Miner Res. 1999;14:1622–27. doi: 10.1359/jbmr.1999.14.9.1622. [DOI] [PubMed] [Google Scholar]
- 139.Redman LM, Rood J, Anton SD, Champagne C, Smith SR, Ravussin E. Calorie restriction and bone health in young, overweight individuals. Arch Intern Med. 2008;168:1859–66. doi: 10.1001/archinte.168.17.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ricci TA, Chowdhury HA, Heymsfield SB, Stahl T, Pierson RN, Jr, Shapses SA. Calcium supplementation suppresses bone turnover during weight reduction in postmenopausal women. J Bone Miner Res. 1998;13:1045–50. doi: 10.1359/jbmr.1998.13.6.1045. [DOI] [PubMed] [Google Scholar]
- 141.Ricci TA, Heymsfield SB, Pierson RN, Jr, Stahl T, Chowdhury HA, Shapses SA. Moderate energy restriction increases bone resorption in obese postmenopausal women. Am J Clin Nutr. 2001;73:347–52. doi: 10.1093/ajcn/73.2.347. [DOI] [PubMed] [Google Scholar]
- 142.Riedt CS, Brolin RE, Sherrell RM, Field MP, Shapses SA. True fractional calcium absorption is decreased after Roux-en-Y gastric bypass surgery. Obesity (Silver Spring) 2006;14:1940–48. doi: 10.1038/oby.2006.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Riedt CS, Cifuentes M, Stahl T, Chowdhury HA, Schlussel Y, Shapses SA. Overweight post-menopausal women lose bone with moderate weight reduction and 1 g/day calcium intake. J Bone Miner Res. 2005;20:455–63. doi: 10.1359/JBMR.041132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Riedt CS, Schlussel Y, von Thun N, Ambia-Sobhan H, Stahl T, et al. Premenopausal overweight women do not lose bone during moderate weight loss with adequate or higher calcium intake. Am J Clin Nutr. 2007;85:972–80. doi: 10.1093/ajcn/85.4.972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Rosen CJ, Bouxsein ML. Mechanisms of disease: Is osteoporosis the obesity of bone? Nat Clin Pract Rheumatol. 2006;2:35–43. doi: 10.1038/ncprheum0070. [DOI] [PubMed] [Google Scholar]
- 146.Rourke KM, Brehm BJ, Cassell C, Sethuraman G. Effect of weight change on bone mass in female adolescents. J Am Diet Assoc. 2003;103:369–72. doi: 10.1053/jada.2003.50051. [DOI] [PubMed] [Google Scholar]
- 147.Ryan AS, Nicklas BJ, Dennis KE. Aerobic exercise maintains regional bone mineral density during weight loss in postmenopausal women. J Appl Physiol. 1998;84:1305–10. doi: 10.1152/jappl.1998.84.4.1305. [DOI] [PubMed] [Google Scholar]
- 148.Salamone LM, Cauley JA, Black DM, Simkin-Silverman L, Lang W, et al. Effect of a lifestyle intervention on bone mineral density in premenopausal women: a randomized trial. Am J Clin Nutr. 1999;70:97–103. doi: 10.1093/ajcn/70.1.97. [DOI] [PubMed] [Google Scholar]
- 149.Salamone LM, Glynn N, Black D, Epstein RS, Palermo L, et al. Body composition and bone mineral density in premenopausal and early perimenopausal women. J Bone Miner Res. 1995;10:1762–68. doi: 10.1002/jbmr.5650101120. [DOI] [PubMed] [Google Scholar]
- 150.Santanasto AJ, Glynn NW, Newman MA, Taylor CA, Brooks MM, et al. Impact of weight loss on physical function with changes in strength, muscle mass, and muscle fat infiltration in overweight to moderately obese older adults: a randomized clinical trial. J Obes. 2011;2011:516576. doi: 10.1155/2011/516576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Shah K, Armamento-Villareal R, Parimi N, Chode S, Sinacore DR, et al. Exercise training in obese older adults prevents increase in bone turnover and attenuates decrease in hip bone mineral density induced by weight loss despite decline in bone-active hormones. J Bone Miner Res. 2011;26:2851–59. doi: 10.1002/jbmr.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Shapses SA, Lewis RD, Pollack N. Relationships between fat and bone in children and adults. In: Anderson JB, Garner SC, Klemmer PJ, editors. Calcium and Phosphorus in Health and Disease. Boca Raton, FL: CRC Press; 2011. pp. 325–68. [Google Scholar]
- 153.Shapses SA, Riedt CS. Bone, body weight, and weight reduction: What are the concerns? J Nutr. 2006;136:1453–56. doi: 10.1093/jn/136.6.1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Shapses SA, Sukumar D. Protein intake during weight loss: effects on bone. In: Burckhardt P, Dawson-Hughes B, Weaver C, editors. Nutritional Influences on Bone Health. London: Springer-Verlag; 2010. pp. 27–33. [Google Scholar]
- 155.Shapses SA, Sukumar D, Schlussel Y, Schneider SH, Ambia-Sobhan H. Vitamin D supplementation increases true fractional calcium absorption in the absence of caloric restriction. FASEB J. 2012;26:386.6. [Google Scholar]
- 156.Shapses SA, Von Thun NL, Heymsfield SB, Ricci TA, Ospina M, et al. Bone turnover and density in obese premenopausal women during moderate weight loss and calcium supplementation. J Bone Miner Res. 2001;16:1329–36. doi: 10.1359/jbmr.2001.16.7.1329. [DOI] [PubMed] [Google Scholar]
- 157.Shen W, Chen J, Punyanitya M, Shapses S, Heshka S, Heymsfield SB. MRI-measured bone marrow adipose tissue is inversely related to DXA-measured bone mineral in Caucasian women. Osteoporos Int. 2007;18:641–47. doi: 10.1007/s00198-006-0285-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Sienkiewicz E, Magkos F, Aronis KN, Brinkoetter M, Chamberland JP, et al. Long-term metreleptin treatment increases bone mineral density and content at the lumbar spine of lean hypoleptinemic women. Metabolism. 2011;60:1211–21. doi: 10.1016/j.metabol.2011.05.016. [DOI] [PubMed] [Google Scholar]
- 159.Silverman NE, Nicklas BJ, Ryan AS. Addition of aerobic exercise to a weight loss program increases BMD, with an associated reduction in inflammation in overweight postmenopausal women. Calcif Tissue Int. 2009;84:257–65. doi: 10.1007/s00223-009-9232-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Sinha N, Shieh A, Stein EM, Strain G, Schulman A, et al. Increased PTH and 1.25(OH)(2)D levels associated with increased markers of bone turnover following bariatric surgery. Obesity (Silver Spring) 2011;19:2388–93. doi: 10.1038/oby.2011.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Skov AR, Haulrik N, Toubro S, Molgaard C, Astrup A. Effect of protein intake on bone mineralization during weight loss: a 6-month trial. Obes Res. 2002;10:432–38. doi: 10.1038/oby.2002.60. [DOI] [PubMed] [Google Scholar]
- 162.Sowers MF, Kshirsagar A, Crutchfield MM, Updike S. Joint influence of fat and lean body composition compartments on femoral bone mineral density in premenopausal women. Am J Epidemiol. 1992;136:257–65. doi: 10.1093/oxfordjournals.aje.a116491. [DOI] [PubMed] [Google Scholar]
- 163.Sukumar D, Ambia-Sobhan H, Zurfluh R, Schlussel Y, Stahl TJ, et al. Areal and volumetric bone mineral density and geometry at two levels of protein intake during caloric restriction: a randomized, controlled trial. J Bone Miner Res. 2011;26:1339–48. doi: 10.1002/jbmr.318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Sukumar D, Partridge NC, Wang X, Shapses SA. The high serum monocyte chemoattractant protein-1 in obesity is influenced by high parathyroid hormone and not adiposity. J Clin Endocrinol Metab. 2011;96:1852–58. doi: 10.1210/jc.2010-2781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Sukumar D, Schlussel Y, Riedt CS, Gordon C, Stahl T, Shapses SA. Obesity alters cortical and trabecular bone density and geometry in women. Osteoporos Int. 2011;22:635–45. doi: 10.1007/s00198-010-1305-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Sumithran P, Prendergast LA, Delbridge E, Purcell K, Shulkes A, et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med. 2011;365:1597–604. doi: 10.1056/NEJMoa1105816. [DOI] [PubMed] [Google Scholar]
- 167.Svendsen OL, Hassager C, Christiansen C. Effect of an energy-restrictive diet, with or without exercise, on lean tissue mass, resting metabolic rate, cardiovascular risk factors, and bone in overweight postmenopausal women. Am J Med. 1993;95:131–40. doi: 10.1016/0002-9343(93)90253-l. [DOI] [PubMed] [Google Scholar]
- 168.Svendsen OL, Hendel HW, Gotfredsen A, Pedersen BH, Andersen T. Are soft tissue composition of bone and non-bone pixels in spinal bone mineral measurements by DXA similar? Impact of weight loss. Clin Physiol Funct Imaging. 2002;22:72–77. doi: 10.1046/j.1475-097x.2002.00398.x. [DOI] [PubMed] [Google Scholar]
- 169.Taes YE, Lapauw B, Vanbillemont G, Bogaert V, De Bacquer D, et al. Fat mass is negatively associated with cortical bone size in young healthy male siblings. J Clin Endocrinol Metab. 2009;94:2325–31. doi: 10.1210/jc.2008-2501. [DOI] [PubMed] [Google Scholar]
- 170.Takeda S, Elefteriou F, Levasseur R, Liu X, Zhao L, et al. Leptin regulates bone formation via the sympathetic nervous system. Cell. 2002;111:305–17. doi: 10.1016/s0092-8674(02)01049-8. [DOI] [PubMed] [Google Scholar]
- 171.Talbott SM, Cifuentes M, Dunn MG, Shapses SA. Energy restriction reduces bone density and biomechanical properties in aged female rats. J Nutr. 2001;131:2382–87. doi: 10.1093/jn/131.9.2382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Thomas T, Gori F, Khosla S, Jensen MD, Burguera B, Riggs BL. Leptin acts on human marrow stromal cells to enhance differentiation to osteoblasts and to inhibit differentiation to adipocytes. Endocrinology. 1999;140:1630–38. doi: 10.1210/endo.140.4.6637. [DOI] [PubMed] [Google Scholar]
- 173.Thorpe MP, Jacobson EH, Layman DK, He X, Kris-Etherton PM, Evans EM. A diet high in protein, dairy, and calcium attenuates bone loss over twelve months of weight loss and maintenance relative to a conventional high-carbohydrate diet in adults. J Nutr. 2008;138:1096–100. doi: 10.1093/jn/138.6.1096. [DOI] [PubMed] [Google Scholar]
- 174.Tomiyama AJ, Mann T, Vinas D, Hunger JM, Dejager J, Taylor SE. Low-calorie dieting increases cortisol. Psychosom Med. 2010;72:357–64. doi: 10.1097/PSY.0b013e3181d9523c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Tontonoz P, Spiegelman BM. Fat and beyond: the diverse biology of PPARgamma. Annu Rev Biochem. 2008;77:289–312. doi: 10.1146/annurev.biochem.77.061307.091829. [DOI] [PubMed] [Google Scholar]
- 176.Tothill P. Dual-energy x-ray absorptiometry measurements of total-body bone mineral during weight change. J Clin Densitom. 2005;8:31–38. doi: 10.1385/jcd:8:1:031. [DOI] [PubMed] [Google Scholar]
- 177.Turner CH, Pavalko FM. Mechanotransduction and functional response of the skeleton to physical stress: the mechanisms and mechanics of bone adaptation. J Orthop Sci. 1998;3:346–55. doi: 10.1007/s007760050064. [DOI] [PubMed] [Google Scholar]
- 178.Turner RT, Iwaniec UT. Low dose parathyroid hormone maintains normal bone formation in adult male rats during rapid weight loss. Bone. 2011;48:726–32. doi: 10.1016/j.bone.2010.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Uusi-Rasi K, Rauhio A, Kannus P, Pasanen M, Kukkonen-Harjula K, et al. Three-month weight reduction does not compromise bone strength in obese premenopausal women. Bone. 2010;46:1286–93. doi: 10.1016/j.bone.2009.10.013. [DOI] [PubMed] [Google Scholar]
- 180.Uusi-Rasi K, Sievanen H, Kannus P, Pasanen M, Kukkonen-Harjula K, Fogelholm M. Influence of weight reduction on muscle performance and bone mass, structure and metabolism in obese premenopausal women. J Musculoskelet Neuronal Interact. 2009;9:72–80. [PubMed] [Google Scholar]
- 181.Vilarrasa N, San Jose P, Garcia I, Gomez-Vaquero C, Miras PM, et al. Evaluation of bone mineral density loss in morbidly obese women after gastric bypass: 3-year follow-up. Obes Surg. 2011;21:465–72. doi: 10.1007/s11695-010-0338-1. [DOI] [PubMed] [Google Scholar]
- 182.Villalon KL, Gozansky WS, Van Pelt RE, Wolfe P, Jankowski CM, et al. A losing battle: weight regain does not restore weight loss-induced bone loss in postmenopausal women. Obesity (Silver Spring) 2011;19:2345–50. doi: 10.1038/oby.2011.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Villareal DT, Chode S, Parimi N, Sinacore DR, Hilton T, et al. Weight loss, exercise, or both and physical function in obese older adults. N Engl J Med. 2011;364:1218–29. doi: 10.1056/NEJMoa1008234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Villareal DT, Fontana L, Weiss EP, Racette SB, Steger-May K, et al. Bone mineral density response to caloric restriction-induced weight loss or exercise-induced weight loss: a randomized controlled trial. Arch Intern Med. 2006;166:2502–10. doi: 10.1001/archinte.166.22.2502. [DOI] [PubMed] [Google Scholar]
- 185.Villareal DT, Shah K, Banks MR, Sinacore DR, Klein S. Effect of weight loss and exercise therapy on bone metabolism and mass in obese older adults: a one-year randomized controlled trial. J Clin Endocrinol Metab. 2008;93:2181–87. doi: 10.1210/jc.2007-1473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.VonThun N, Sukumar D, Heymsfield S, Shapses S. Weight reduction followed by regain in postmenopausal women only partially attenuates bone loss: a two year case control trial. Obesity (Silver Spring) 2012 In press. [Google Scholar]
- 187.Warming L, Ravn P, Christiansen C. Visceral fat is more important than peripheral fat for endometrial thickness and bone mass in healthy postmenopausal women. Am J Obstet Gynecol. 2003;188:349–53. doi: 10.1067/mob.2003.93. [DOI] [PubMed] [Google Scholar]
- 188.Wetzsteon RJ, Petit MA, Macdonald HM, Hughes JM, Beck TJ, McKay HA. Bone structure and volumetric BMD in overweight children: a longitudinal study. J Bone Miner Res. 2008;23:1946–53. doi: 10.1359/jbmr.080810. [DOI] [PubMed] [Google Scholar]
- 189.Wilsgaard T, Jacobsen BK, Ahmed LA, Joakimsen RM, Stormer J, Jorgensen L. BMI change is associated with fracture incidence, but only in non-smokers. The Tromso Study Osteoporos Int. 2011;22:1237–45. doi: 10.1007/s00198-010-1318-y. [DOI] [PubMed] [Google Scholar]
- 190.Woo DG, Lee BY, Lim D, Kim HS. Relationship between nutrition factors and osteopenia: effects of experimental diets on immature bone quality. J Biomech. 2009;42:1102–7. doi: 10.1016/j.jbiomech.2009.02.020. [DOI] [PubMed] [Google Scholar]
- 191.World Health Org. Obesity and Overweight. Geneva: World Health Org; 2011. http://www.who.int/mediacentre/factsheets/fs311/en. [Google Scholar]
- 192.Wortley KE, Garcia K, Okamoto H, Thabet K, Anderson KD, et al. Peptide YY regulates bone turnover in rodents. Gastroenterology. 2007;133:1534–43. doi: 10.1053/j.gastro.2007.08.024. [DOI] [PubMed] [Google Scholar]
- 193.Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr. 2000;72:690–93. doi: 10.1093/ajcn/72.3.690. [DOI] [PubMed] [Google Scholar]
- 194.Xu M, Qi C, Deng B, Deng PX, Mo CW. Phytotherapy versus hormonal therapy for post-menopausal bone loss: a meta-analysis. Osteoporos Int. 2009;20:519–26. doi: 10.1007/s00198-008-0724-x. [DOI] [PubMed] [Google Scholar]
- 195.Yamada C. Role of incretins in the regulation of bone metabolism (Transl from Japanese.) Nihon Rinsho. 2011;69:842–47. [PubMed] [Google Scholar]
- 196.Yu EW, Thomas BJ, Brown JK, Finkelstein JS. Simulated increases in body fat and errors in bone mineral density measurements by DXA and QCT. J Bone Miner Res. 2012;27:119–24. doi: 10.1002/jbmr.506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Zhao LJ, Jiang H, Papasian CJ, Maulik D, Drees B, et al. Correlation of obesity and osteoporosis: effect of fat mass on the determination of osteoporosis. J Bone Miner Res. 2008;23:17–29. doi: 10.1359/JBMR.070813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Zhao LJ, Liu YJ, Liu PY, Hamilton J, Recker RR, Deng HW. Relationship of obesity with osteoporosis. J Clin Endocrinol Metab. 2007;92:1640–46. doi: 10.1210/jc.2006-0572. [DOI] [PMC free article] [PubMed] [Google Scholar]