Abstract
Obesity has increased dramatically over the past decades, reaching epidemic proportions. The reasons are likely multifactorial. One of the suggested causes is the accelerated exposure to obesity-inducing chemicals (obesogens). However, out of the tens of thousands of industrial chemicals humans are exposed to, very few have been tested for their obesogenic potential, mostly due to the limited availability of appropriate in vivo screening models. In this study, we investigated whether two commonly used flame retardants, the halogenated bisphenol-A (BPA) analogs tetrabromobisphenol-A (TBBPA) and tetrachlorobisphenol-A (TCBPA), could act as obesogens using zebrafish larvae as an in vivo animal model. The effect of embryonic exposure to these chemicals on lipid accumulation was analyzed by Oil Red-O staining, and correlated to their capacity to activate human and zebrafish peroxisome proliferator-activated receptor gamma (PPARγ) in zebrafish and in reporter cell lines. Then, the metabolic fate of TBBPA and TCBPA in zebrafish larvae was analyzed by high-performance liquid chromatography (HPLC) . TBBPA and TCBPA were readily taken up by the fish embryo and both compounds were biotransformed to sulfate-conjugated metabolites. Both halogenated-BPAs, as well as TBBPA-sulfate induced lipid accumulation in zebrafish larvae. TBBPA and TCBPA also induced late-onset weight gain in juvenile zebrafish. These effects correlated to their capacity to act as zebrafish PPARγ agonists. Screening of chemicals for inherent obesogenic capacities through the zebrafish lipid accumulation model could facilitate prioritizing chemicals for further investigations in rodents, and ultimately, help protect humans from exposure to environmental obesogens.
Keywords: zebrafish, lipid, obesity, PPARγ, TBBPA, TCBPA
Over the past decades, obesity has been on a rising epidemic worldwide, with more than 10% of the world's adult population being obese in 2008 (WHO, 2013). In the United States, more than one third of the adult population and almost 17% of children are obese (Ogden et al., 2012). Obesity is associated with an increasing risk for adverse medical outcomes such as type 2 diabetes, cardiovascular diseases, and certain cancers (Aballay et al., 2013; Flegal et al., 2007). In the United States, the medical expenditures for treating obesity-related diseases were $147 billion in 2008 accounting for 9% of medical costs (Finkelstein et al., 2012).
Although the onset of obesity is mainly attributed to high-calorie intake, lack of exercise, and sedentary lifestyle, exposure to obesogenic chemicals present in human environment could potentially be an additional cause (Baillie-Hamilton, 2002). Obesogens act by modulating lipid homeostasis, adipocyte numbers, lipid storage, or regulation of appetite and satiety (Grun and Blumberg, 2007; Janesick and Blumberg, 2011a). The environment is increasingly filled with chemicals, produced for a variety of industrial applications, that are deemed to be hazardous to human health, but our knowledge regarding obesogens is still limited. So far, obesogenic chemicals tend to fall under the group of endocrine disruptors either as environmental estrogens [bisphenol-A (BPA), genistein, diethylstilbestrol (DES)] (Newbold et al., 2009), or activators of the peroxisome proliferator-activated receptor gamma (PPARγ) signaling pathway (Janesick and Blumberg, 2011b). PPARγ, a nuclear receptor family member, heterodimerizing with the retinoid X receptor (RXR), has been widely recognized as a master regulator of adipogenesis. One of the most studied obesogens is tributyltin (TBT), an organotin used as a fungicide or as an antifouling agent on boat hulls. Mechanistically, TBT is a ligand for PPARγ and RXR, reflecting that these receptors play important roles in the regulation of adipogenesis and lipid homeostasis (Janesick and Blumberg, 2011b; le Maire et al., 2009). Through this mechanism, demonstrated in in vitro assays, TBT promotes the differentiation of preadipocytes (Li et al., 2011). Other environmental contaminants have also been proposed as obesogen candidates. We previously reported that two halogenated bisphenol-A analogs (halogenated-BPAs), namely tetrabromobisphenol-A (TBBPA) and tetrachlorobisphenol-A (TCBPA), commonly used as flame retardants, induce adipogenesis in 3T3-L1 cells through the activation of PPARγ (Riu et al., 2011a). These chemicals are found in the environment (Fan et al., 2013) and the presence of TBBPA has been detected in human samples (Cariou et al., 2008). In vitro and in vivo, TBBPA undergoes phase II metabolism to form glucuronic acid and sulfate conjugates (Fini et al., 2012; Riu et al., 2011b; Schauer et al., 2006). For this reason, it has been suggested that rapid metabolism by conjugation of the absorbed TBBPA results in a low systemic bioavailability of TBBPA. However, the monosulfated conjugates of TBBPA and TCBPA also act as human PPARγ agonists, suggesting that they are still biologically active (Riu et al., 2011b).
Some studies have reported obesogenic effects of exposure to xenobiotics in vivo. In particular, prenatal or perinatal exposure to obesogenic chemicals may predispose the individual to become obese later in life (Janesick and Blumberg, 2012). In rodents, perinatal exposure to BPA results in a higher body fat percentage by increasing the size of adipocytes at adult age (Wei et al., 2011). Similarly, exposure to TBT in utero causes predisposition to obesity in mice (Grun et al., 2006; Kirchner et al., 2010). More recently, other animal models have been used as whole organisms to screen for chemicals inducing fat accumulation, such as the nonvertebrate nematode (Caenorhabditis elegans) or the vertebrate zebrafish (Lemieux et al., 2011; Tingaud-Sequeira et al., 2011).
Similar to mammals, despite the lack of brown adipose tissue in poikilotherms, zebrafish store neutral lipid triglycerides in the visceral, intramuscular, and subcutaneous adipocyte depots. Studies of the zebrafish embryo, which is optically transparent facilitating the detection of lipid depots using lipid stains (Minchin and Rawls, 2011), have shown that white adipose tissue appearance correlates with size rather than age of the fish. The first adipocytes, which can be observed from 8 to 12 days post fertilization (dpf), or at a minimal size of about 5 mm (Imrie and Sadler, 2010), appear in the pancreatic region, then in the viscera, and later on, in the subcutaneous and cranial regions (Flynn et al., 2009; Imrie and Sadler, 2010).
In this report, we describe that zebrafish is a suitable animal model for metabolic studies, sharing similar capabilities as mammals for the biotransformation of halogenated bisphenols. We first examined the metabolic fate of TBPPA and TCBPA in order to validate the use of zebrafish as a vertebrate model for mammalian endocrine disruption. Then, we investigated whether pharmaceutical PPARγ ligands as well as the environmental pollutants TBBPA, TCBPA, and TBT or their metabolites were able to activate human and zebrafish PPARγ in vitro and in vivo. We used a screening method for lipid accumulation in zebrafish in order to assess the obesogenic properties induced by the exposure to halogenated-BPAs and TBT at an early developmental stage. Finally, we assessed whether an early exposure to the halogenated-BPAs and TBT induced late onset weight gain in juvenile zebrafish.
MATERIALS AND METHODS
Fish Maintenance
Wild type, AB/Tuebingen (TAB) and transgenic (Tg) zebrafish (zf), were housed at 28.5°C in a Tecniplast system (Tecniplast USA Inc., West Chester, PA) on a 14 h/10 h light/dark cycle. They were used according to the maintenance and experimental protocols approved by the Institutional Animal Care and Use Committee at the University of Houston (protocol numbers 12-042 and 10-040). We used the LT-hPPARγ transgenic zebrafish (Tg(hPPARγ-eGFP)) previously described in Tiefenbach et al. (2010). In this fish, the expression of GAL4 DNA binding domain (DBD) fused in-frame with the human PPARγ ligand binding domain (hPPARγ-LBD) is driven by the zebrafish hsp70 promoter. Upon heat pulse, GAL4-hPPARγ-LBD is ubiquitously expressed and GAL4 binds the upstream activating sequence (UAS) response element, leading to the expression of the reporter gene nuclear enhanced Green Fluorescent Protein (eGFP), in the presence of active hPPARγ ligands. On the contrary, RXR ligands are not active on this system (data not shown).
Chemicals
Rosiglitazone (RGZ) 5-[[4-[2-(methyl-2-pyridinylamino)ethoxy]phenyl]methyl]-2,4-thiazolidinedione, TBBPA [2,2-bis(3,5-dibromo-4-hydroxyphenyl)propane] and tributyltin chloride and all solvents (analytical grade) were purchased from Sigma-Aldrich (St. Louis, MO). TCBPA [2,2-bis(3,5-dichloro-4-hydroxyphenyl)propane] was purchased from TCI America (Portland, OR) and 15-deoxy-D12,14-prostaglandin J2 (15d-PGJ2) was purchased from Cayman chemical (Ann Arbor, MI). The other thiazolidinediones (pioglitazone, troziglitazone, and ciglitazone) were obtained from Tocris Bioscience (Lille, France).
PPARγ Activation In Vitro and In Vivo
Reporter cell lines and stable gene expression assays
We used reporter cell lines stably transfected with the human (Seimandi et al., 2005) or the zebrafish PPARγ-LBD. The stably transfected reporter cell line HG5LN-GAL4-zfPPARγ was generated using the same procedure as described in Seimandi et al. (2005) for the HG5LN-GAL4-hPPARγ. These reporter cell lines, contrary to the reporter cell line HGELN-GAL4-hPPARγ described in le Maire et al. (2009), are not responsive to RXR ligands (data not shown). The HG5LN cells were originally derived from human cervical cancer HeLa cells.
Reporter cells were seeded in 96-well white opaque tissue culture plates (Greiner Bio-one, Monroe, NC) at a density of 40,000 cells per well and maintained in Dulbecco's Modified Eagle Medium (DMEM) without phenol red, supplemented with 5% dextran-coated, charcoal-treated FCS. After 24 h, culture medium was replaced by DMEM containing test chemicals. Assays were performed in absence of serum in order to avoid ligand capture by serum proteins. Cells were exposed to chemicals during 16 h, and then the medium was replaced by medium containing 0.3mM luciferin. Luminescence was measured in intact living cells for 2 s in a plate reader (2030 multilabel reader, Perkin-Elmer, Waltham, MA)
In vivo assay using Tg(hPPARγ-eGFP) zebrafish
Embryos were collected after spawning. At 3 h post fertilization (hpf), embryos were transferred to 12-well plates (10 embryos/well) in 1 ml of embryo medium (E3: 5mM NaCl, 0.17mM KCl, 0.33mM CaCl2, 0.3mM MgSO4). At 4 hpf, embryos were heat shocked at 37°C for 45 min. After heat pulse, embryos were treated with vehicle only (0.1% Dimethylsulfoxide; DMSO) or chemicals solubilized in DMSO (0.1% DMSO final concentration). After 24 h of treatment, solution was removed and replaced with fresh E3. For GFP quantification, embryos were transferred to 96-well black plates, 1 embryo/well in 100 μl E3 (8 embryos/treatment) and GFP expression was quantified using a plate reader. Embryos were manually removed from the chorion and mounted in 3% 3-methyl cellulose for 3D microscopy live imaging using a Nikon AZ100M microscope set up with a fluorescence camera (CoolSNAP HQ2, Photometrics) and the NIS-Elements AR analyzing program (Nikon, New York, NY). Images for all samples were captured using identical exposure and camera settings.
Zebrafish Treatment For Oil Red O (ORO) Staining
Embryos were collected after spawning. At 24 hpf, 200μM phenylthiourea (PTU) was added to E3 in order to inhibit embryo melanization. At 3 dpf, larvae were transferred into 6-well plates (20 larvae/well) in 3 ml of E3 with PTU, and treated with vehicle only (0.1% DMSO) or with chemicals solubilized in DMSO and left in the same medium until 6 dpf. By then, larvae were daily fed an egg yolk diet (EYD) every morning, followed by E3 renewal and chemical treatment, 8 h after feeding, until 11 dpf (n = 6). At 11 dpf, larvae were euthanized with tricaine methylsulfonate (MS-222, Sigma-Aldrich) and fixed in 4% paraformaldehyde overnight at 4°C for further staining or transferred to the fish housing system and raised until 30 dpf for weight and length scoring. Fixed larvae were then washed in PBS with 0.1% Tween-20 (PBT) and stained with a filtered 0.3% ORO solution for 3 h at room temperature (RT). After staining, larvae were washed three times in PBT and stored in 90% glycerol before imaging. Larvae were scored for fat accumulation and imaged using a Nikon AZ100M microscope set up with a color camera DS Fi1 (Nikon) and the NIS-Elements AR analyzing program. The nonspecific staining in the zebrafish swimbladder by ORO was not classified as lipid staining in our scoring.
BMI Analysis of 1 Month Old Fish
Fish embryos were exposed as described above for the ORO staining. At 11 dpf, fish were transferred and raised on the fish housing system and fed a regular diet. At 1 month of age, the length of the fish was measured with a ruler, and the wet weight was recorded using a precision balance (Mettler Toledo, Columbus, OH) after all the water had been removed from the fish. The BMI was calculated as weight/(length)2.
Metabolic Fate of TBBPA and TCBPA in Zebrafish
Zebrafish treatment
First, we assessed the uptake kinetic of TBBPA and TCBPA in zebrafish larvae exposed at 3 dpf. Larvae were placed in 6-well plates (20 embryos/well) in 3 ml of E3 and exposed to a single dose of 1μM TBBPA or TCBPA for 48 h. At different time points after exposure, E3 was collected in glass vials and stored at 4°C for high-performance liquid chromatography (HPLC) analysis.
In a second experiment, the same exposure protocol as used for ORO staining was applied, in which embryos were exposed at 3 dpf and then daily from 6 dpf to TBBPA or TBBPA. E3 was collected in glass vials every 24 h and stored at 4°C for HPLC analysis. At 10 dpf, larvae were euthanized. The experiments were replicated in three independent experiments. As a control, we analyzed E3 that was incubated with compounds without the addition of fish.
Analytical procedure
HPLC analyzes were performed on a Binary HPLC pump 1525 (Waters, Milford, MA) equipped with an autosampler 2707 injector and a photodiode array detector (PDA) 2998 set at 290 nm (Waters). HPLC procedures were adapted from Riu et al. (2011b) and carried out with an HPLC system based on a Nucleodur C8 column (250 × 4 mm, 5 μm, Macherey Nagel, Bethlehem, PA) using the following conditions: mobile phases: A: 20mM ammonium acetate pH 3.5/acetonitrile (95/5, vol/vol), B: 100% acetonitrile; gradient: 0–4 min, A: 100% isocratic; 4–6 min, linear gradient from A: 100% to A:B 70:30; 6–21 min, A:B 70:30 isocratic; 21–23 min, linear gradient from A:B 70:30 to A:B: 50:50; 23–33 min, A:B 50:50 isocratic; 33–34 min, linear gradient from A:B 50:50 to B 100%; 34–43 min, B: 100% isocratic. TBBPA, TCBPA, and their metabolites were quantified by measuring the area under the peak, based on their respective standard curves as previously established.
TBBPA-S synthesis and purification
We synthesized TBBPA-S from the biotransformation of TBBPA by the zebrafish, as follows: 60 zebrafish larvae (3 dpf) were placed in a Petri dish (60 mm) with 9 ml of E3, and treated with 9 μl of 1mM TBBPA dissolved in DMSO, or DMSO only (control). After 72 h treatment, E3 was collected and analyzed using HPLC with Ultra Violet (UV) detector (UV-HPLC). Only TBBPA-S was detectable on the UV-chromatogram. Then E3 containing synthesized TBBPA-S and control were stored at −20°C until further utilization.
Purification of TBBPA-S was carried out by solid-phase extraction (SPE). SPE: chromabond 1 g C8 glass cartridges (Macherey Nagel, Hoerdt, France) were washed with 3 ml methanol and equilibrated with 3 ml of fresh E3. Then 8 ml of E3 containing synthesized TBBPA-S were loaded on cartridge. Elution was performed successively with 3 ml ultrapure water, 3 ml ultrapure water:acetonitrile (90:10, vol/vol), 900 μl 100% acetonitrile and 2 ml 100% acetonitrile. TBBPA-S was eluted in the last fraction. Acetonitrile was evaporated using a vacuum concentrator Speedvac (ThermoScientific, Waltham, MA). TBBPA-S was then dissolved in DMSO and its purity was analyzed by HPLC-UV.
RESULTS
Halogenated-BPAs are hPPARγ Ligands In Vivo
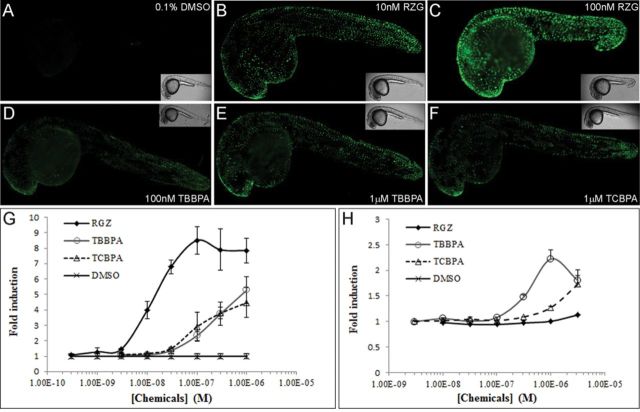
TBBPA and TBBPA have been described as hPPARγ ligands in vitro (100-fold less potent than Rosiglitazone, RGZ) using the stably transfected HGELN-hPPARγ cell lines (Riu et al., 2011a). We used the Tg(hPPARγ-eGFP) zebrafish generated by Tiefenbach et al. (2010) to investigate whether these compounds were absorbed by zebrafish embryos and were able to activate hPPARγ-LBD in a whole organism. Following a 24-h exposure to RGZ, we observed a strong GFP expression in the epidermis of the whole embryo (Figs. 1B and C), whereas no GFP expression was detected in the untreated group (Fig. 1A). GFP expression was also detected in embryos treated with 100nM and 1μM TBBPA or 1μM TCBPA, primarily in the head and tail regions (Figs. 1D–F). Quantification of GFP expression using a fluorescence reader showed that RGZ exposure induced GFP expression in a dose-dependent manner with an EC50 of 12nM (Fig. 1G), similarly to what was shown using hPPARγ reporter cell lines (Supplementary fig. 1A). Both TBBPA and TCBPA exposure induced GFP expression in a dose-dependent manner, but to a lesser extent than RGZ, reaching approximately 65 and 50% of RGZ activation at 1μM, respectively (Fig. 1G), whereas TBT exposure did not. Concentrations above 1μM for TBBPA and TCBPA were not used because of their lethal effects in zebrafish embryos. These results showed that TBBPA and TCBPA acted as hPPARγ agonists in transgenic zebrafish embryos.
FIG. 1.
Human and zebrafish PPARγ activation by halogenated-BPAs in vivo and in vitro. (A–F) GFP fluorescence and bright field (insert) images (lateral view) of 28 hpf Tg(hPPARγ-eGFP) zebrafish embryos treated for 24 h with DMSO (A), RGZ (B, C), TBBPA (D, E) and TCBPA (F). (G) Dose-response curves of quantified GFP expression in 28 hpf Tg(hPPARγ-eGFP) zebrafish embryos exposed to 0.1% DMSO, RGZ, TBBPA, and TCBPA. (H) Results of luciferase assays showing dose-response curves for 0.1% DMSO, RGZ, TBBPA, and TCBPA in HG5LN-GAL4-zfPPARγ reporter cell line. Results are expressed as fold induction above control (mean ± SD from three independent experiments).
Halogenated-BPAs are Zebrafish PPARγ Ligands In Vitro
To investigate whether the chemicals found to be hPPARγ ligands also activated zfPPARγ, we generated a reporter cell line using HeLa cells stably transfected with the zfPPARγ-LBD. We then characterized the activity of several thiazolidinediones (TZDs: RGZ, pioglitazone, ciglitazone, and troziglitazone), 15-deoxy-Δ12,14-prostaglandin-J2 (15d-PGJ2), TBT, TBBPA, and TCBPA on the HG5LN-zfPPARγ cell line. Neither of the well-known high affinity hPPARγ ligands nor TBT were able to activate the zebrafish counterpart (Supplementary Fig. 1). Interestingly, TBBPA and TCBPA displayed a weak and specific agonistic activity towards zfPPARγ (Supplementary Fig. 2). TBBPA was a more potent zfPPARγ agonist than TCBPA with EC50 of 0.297 and 23.2μM, respectively. We observed cytotoxic effects after treatment with concentrations of 10μM and higher for both halogenated-BPAs. The results showed that the halogenated-BPAs are zfPPARγ agonists in vitro, in accordance to previous results obtained using other cell-based assays (Riu et al., 2011a). The known hPPARγ high affinity ligands did not activate zfPPARγ. Consequently, the halogenated-BPAs we tested are the most potent zfPPARγ ligands reported till date.
Uptake and Metabolism of Halogenated-BPAs by Zebrafish Embryos
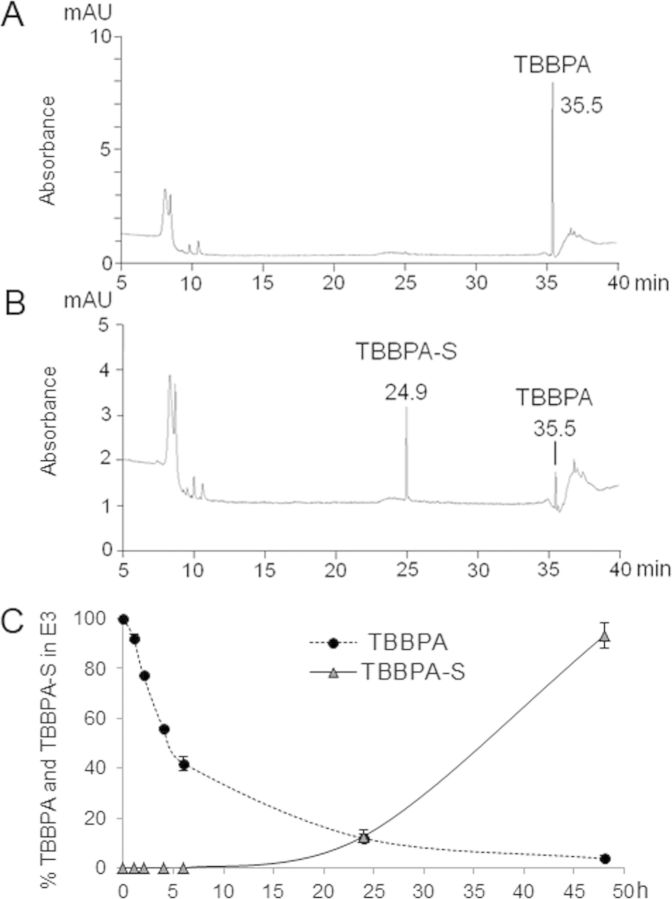
Taking into account the rapid uptake and biotransformation of TBBPA by different animal models and humans (Fini et al., 2012; Hakk et al., 2000; Schauer et al., 2006), we assessed the uptake and fate of TBBPA and TCBPA in zebrafish using an HPLC system coupled to a UV detector set at 290 nm (maximal absorption wavelength for TBBPA and TCBPA). Under our analytical conditions, TBBPA was eluted at a retention time (Rt) of 35.5 min (Fig. 2A) and TCBPA at a Rt of 33.8 min (Supplementary fig. 3A). This system was used to investigate the metabolic fate of TBBPA and TCBPA in zebrafish by testing embryo media (E3) that was collected 24 h after exposure. One major metabolite was observed on the UV-chromatogram together with unchanged TBBPA (Fig. 2B). The metabolite recovered in water was identified as TBBPA-monosulfate (TBBPA-S), based on its coelution with the authentic standard (synthesized and characterized as described in Riu et al. (2011b)) at Rt = 24.9 min. Similarly, only one major metabolite was recovered in the water of TCBPA-treated larvae, monosulfated TCBPA (TCBPA-S), which eluted at Rt = 24.3 min (Supplementary Fig. 3B). As controls, the compounds were incubated with E3 without the addition of zebrafish. No metabolites were formed in the controls.
FIG. 2.
Uptake and metabolism of TBBPA in early zebrafish larvae. (A) A typical HPLC-UV chromatogram of unchanged TBBPA in embryo media (E3) and (B) a typical HPLC-UV chromatogram from an E3 sample from 8 dpf wild-type zebrafish larvae exposed to 1μM TBBPA for 24 h. (C) TBBPA uptake and TBBPA-S formation kinetics recovered in E3 from zebrafish exposed to TBBPA from 3 dpf for 48 h. Results are expressed as mean ± SD from three independent experiments.
In order to establish TBBPA and TCBPA uptake kinetics, zebrafish larvae at 3 dpf were incubated in E3 supplemented with 1μM TBBPA or TCBPA, then E3 aliquots were taken at different time points and analyzed using HPLC-UV. The relative proportion of unchanged parent compounds and metabolites in E3 were quantified by integrating the areas under the peaks. TBBPA and TCBPA were rapidly absorbed by zebrafish at 3 dpf (Fig. 2C and Supplementary Fig. 3C). After 6 h of exposure, more than 50% of TBBPA was recovered in E3. After 24 h of exposure, only 11.9 ± 1.8% of TBBPA was found in E3, and by 48 h, most of the compound was absorbed by the larvae with only 3.8 ± 1.1% TBBPA remaining in E3. Following this rapid absorption, TBBPA was metabolized by zebrafish into TBBPA-S which was gradually released in E3, such that 12.7 ± 2.7 and 93.2 ± 5.0% of TBBPA-S was released in E3 after 24 and 48 h of exposure, respectively (Fig. 2C). Similar results were observed for TCBPA, which was taken up more quickly than its brominated analog, with only 13.9 ± 3.5 and 2.3 ± 1.0% of TCBPA recovered in E3 after 8 and 24 h of exposure, respectively. However, unlike TBBPA, TCBPA was not rapidly metabolized, with only 13.4 ± 3.1% of TCBPA-S released in E3 after 48 h of exposure (Supplementary Fig. 3C).
We also investigated the uptake and metabolism of TBBPA and TCBPA using repeated chemical exposures from 3 dpf. Both compounds were efficiently taken up and metabolites recovered, as shown in Supplementary Figure 4. At 6 dpf after 72 h of exposure, almost all TBBPA was absorbed and metabolized (97.7 ± 0.5%), leading to the formation and release of TBBPA-S with more than 50% recovered in E3 (Supplementary Fig. 4A). After a daily E3 renewal and repeated exposure starting at 6 dpf, more than 90% of TBBPA was taken up every day. A slight decrease of TBBPA-S elimination from the fish into E3 was observed at 7 and 8 dpf with 43.7 ± 2.4 and 29.0 ± 2.0% of TBBPA-S recovered in E3, respectively, to reach a plateau at 9 dpf. For larvae treated at 3 dpf for 72 h with 1μM TCBPA, 5% of the parental TCBPA remained in E3 at 6 dpf, whereas 47.1 ± 10.2% of its metabolite, TCBPA-S, was recovered (Supplementary Fig. 4B). Like TBBPA, at 7, 8, and 9 dpf, almost 90% of TCBPA was taken up after 24 h treatment, and more than 40% of the TCBPA absorbed was found in E3 in its TCBPA-S form.
Lipid Accumulation Induced by Chemical Exposure in Zebrafish
After demonstrating that halogenated-BPAs were extensively absorbed by zebrafish at early stages of development, and activated zfPPARγ, we investigated the effects of repeated exposure to these compounds on lipid accumulation. Larvae were exposed daily to compounds from 3 dpf and simultaneously fed a EYD from 6 to 10 dpf. At 11 dpf, lipid accumulation was assessed by ORO staining. Chemical-treated and control larvae were first scored for positive ORO staining (Fig. 3). Larvae exposed daily to TBBPA, TCBPA, or TBT had markedly increased levels of lipid accumulation at 11 dpf, compared with vehicle-treated (0.1% DMSO) or RGZ-treated larvae (Fig. 3A). Following EYD feeding, less than 14% of the vehicle-treated or RGZ-treated larvae displayed ORO staining. A statistically significant increase in the percentage of ORO-stained larvae was observed after exposure to 0.1 and 1nM TBT, and 10 and 100nM TBBPA (29 and 38%, and 34 and 30% of stained larvae, respectively). Only 25, 22, and 9% of larvae exhibited lipid accumulation following 1μM TBBPA, 100nM and 1μM TCBPA exposure (Fig. 3A); this decrease in the percentage of stained larvae compared with those treated at lower concentrations was likely caused by the toxicity of TBBPA and TCBPA at these concentrations, possibly leading to decreased feeding behavior and hence less lipid accumulation at high concentrations. TCBPA was more toxic in zebrafish than TBBPA, and induced larvae lethality to a high extent by the end of the treatment (about 50% lethality at 1μM), which likely explains the decrease in lipid accumulation in surviving zebrafish (Supplementary Table 1). When fed with a regular diet (Hatchfry encapsulon, grade 0, Argent Chemical Laboratories, Redmond, WA), none of the chemically exposed or control larvae were stained (data not shown).
FIG. 3.
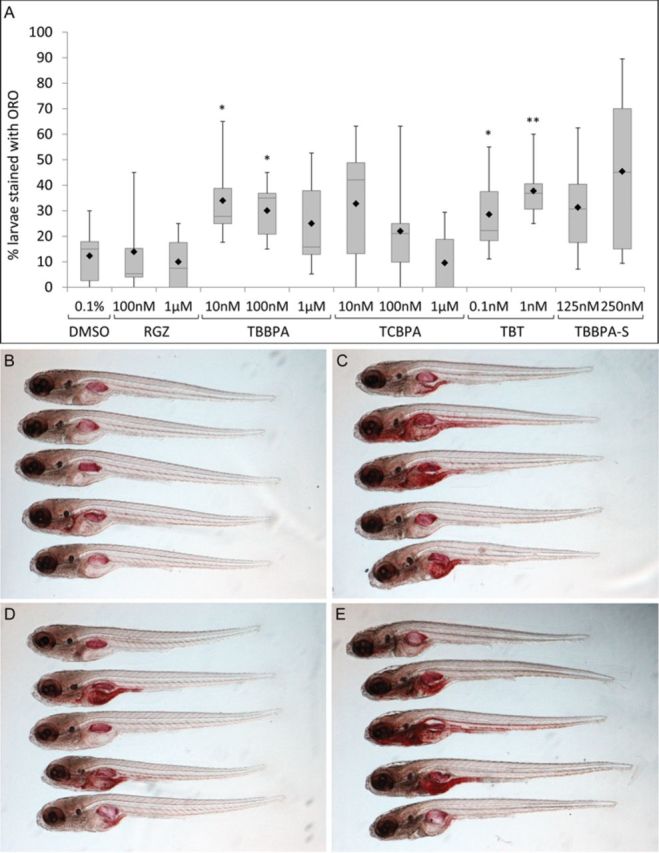
Effects of exposure to TBBPA, TCBPA, TBT, and the TBBPA metabolite, TBBPA-S, on lipid accumulation in 11 dpf zebrafish larvae. (A) Box-plot diagram showing the quantification of the percentage of zebrafish larvae (11 dpf) stained with ORO after 8 days of exposure to RGZ, TBBPA, TCBPA, TBT, and TBBPA-S. The box-plot depicts six values, namely the minimum and maximum values, the upper (Q3) and lower (Q1) quartiles, the median and the mean. The length of the box represents the interquartile range. The median is identified by a line inside the box and the mean is represented by a black diamond. (B–E) Lateral view of a sample of fixed and ORO-stained 11 dpf zebrafish larvae after treatment with 0.1% DMSO (B), 100nM TBBPA (C), 100nM TCBPA (D) and 1nM TBT (E). Statistics were done using a Student's t-test, *p < 0.05, **p < 0.01.
Next, each larva was scored for intensity in ORO staining ranging from no staining (−) to strong staining (+++) (Figs. 4A–D). In the mildly stained larvae, ORO staining appeared in the liver, anterior intestine, gut region and jaw (Fig. 4B). Moderately stained larvae included the same stained regions as seen in the mild category with additional staining in the heart and major blood vessels (dorsal aorta and posterior cardinal vein) (Fig. 4C), whereas strongly stained larvae displayed staining that extended to the whole vasculature, including the intersegmental vessels, and the brain (Fig. 4D). ORO also stained the swimbladder nonspecifically; however, this staining was not included in the positive lipid staining score. The distribution of ORO staining in larvae at 11 dpf was dependent on the compound and its concentration (Fig. 4E). We observed a higher proportion of larvae displaying a mild, moderate or strong staining after chemical exposure compared with controls, except for 1μM TBBPA or TCBPA due to their toxicity at high concentration. Taken together, these results showed that repeated exposures to TBBPA, TCBPA, or TBT induced lipid accumulation in 11 dpf zebrafish.
FIG. 4.
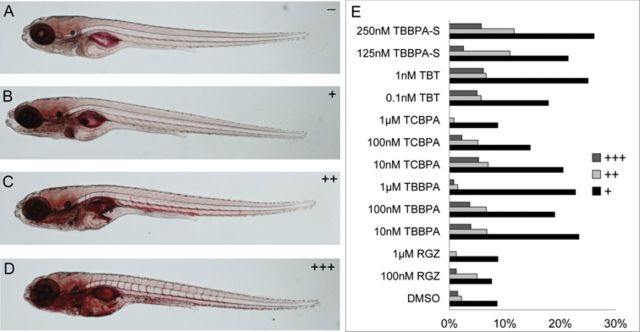
Distribution of ORO staining in 11 dpf treated zebrafish. ORO staining was scored from no staining (A), mild (B), moderate (C), and strong staining (D). Distribution of ORO staining after exposure of zebrafish from 3 dpf to 11 dpf to vehicle (0.1% DMSO), and to different concentrations of RGZ, TBBPA, TCBPA, TBT, and TBBPA-S (E).
TBBPA-Monosulfate Induces Lipid Accumulation in Zebrafish
Because TBBPA-S is the major metabolite formed in zebrafish embryos/larvae, and it has been previously described as a hPPARγ agonist in vitro (Riu et al., 2011b), we assessed the ability of TBBPA-S to bind to and activate this nuclear receptor in vivo. Tg(hPPARγ-eGFP) zebrafish were exposed to TBBPA-S at 5 hpf and GFP expression was quantified after 24 h of treatment. TBBPA-S was able to activate hPPARγ in vivo in a dose-dependent manner (Fig. 5A). Also, using zfPPARγ reporter cell lines, we observed that TBBPA-S activated zfPPARγ in vitro at the highest concentration 1μM (Fig. 5B). These results demonstrated that TBBPA-S can act as zfPPARγ and hPPARγ ligand, but displaying a lower potency than the parent compound, TBBPA (Fig. 1H).
FIG. 5.
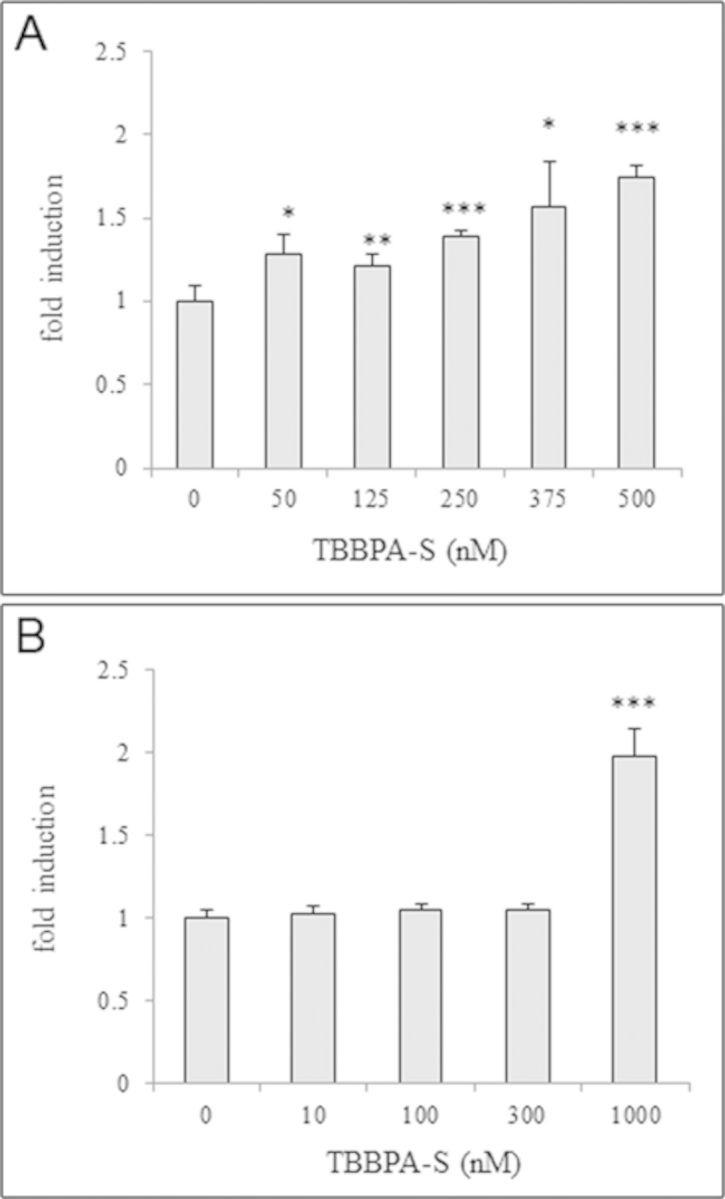
Activation of hPPARγ by TBBPA-S in vitro and in vivo. (A) Dose-response curve of quantified GFP expression of 28 hpf Tg(hPPARγ-eGFP) zebrafish embryos exposed to TBBPA-S (mean ± SD from three independent experiments). (B) Results of luciferase assays showing dose-response curves for TBBPA-S in HG5LN-GAL4-zfPPARγ reporter cell line. Results are expressed as fold induction above control (mean ± SD from three independent experiments). Statistics were done using a Student's t-test, *p < 0.05, **p < 0.01, and ***p < 0.001.
We then assessed lipid accumulation using the methods described above in zebrafish treated with TBBPA-S. We first generated TBBPA-S by treating zebrafish larvae with 1μM TBPPA at 3 dpf for 72 h, taking advantage of the complete biotransformation of TBBPA into TBBPA-S at that time point (Fig. 2C). More than 31 and 45% of 11 dpf larvae displayed lipid accumulation visualized by ORO staining after 8 days of exposure to 125 and 250nM TBBPA-S, respectively, whereas only 14% of ORO-stained larvae were observed in the control group (Fig. 3A). Moreover, the distribution pattern (mild to strong staining) was similar for TBBPA-S and for parent TBBPA, with a dose-dependent increase in larvae displaying mild, moderate, and strong ORO staining (Fig. 4E). We conclude that TBBPA-S had the capacity to increase lipid accumulation in vivo in zebrafish, like TBBPA itself, which correlated to their affinities as ligands to zfPPARγ observed in vitro in our transactivation assay.
Early Exposure to Halogenated-BPAs Induces Late-Onset Weight Gain
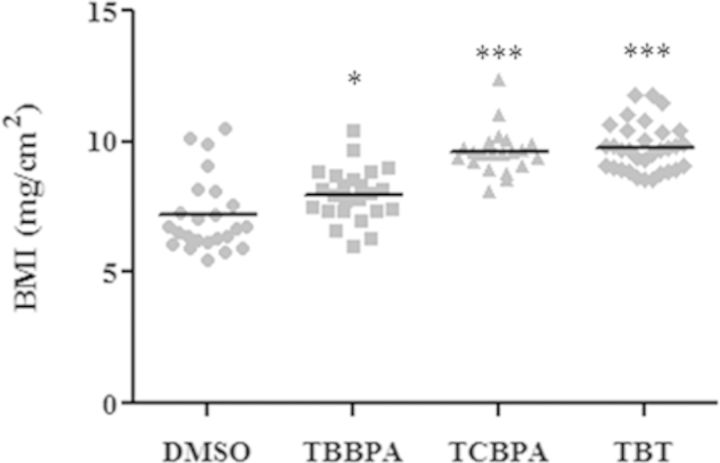
It has been reported that exposure to endocrine disrupting chemicals early in life can lead to obesity later in life in rodents (Janesick and Blumberg, 2012). To investigate whether zebrafish could be used as a late onset obesity model, we performed grow-out studies of exposed fish. Larvae were exposed to 100nM TBBPA, 100nM TCBPA, or 1nM TBT from 3 dpf to 11 dpf and fed EYD. Then they were raised in a chemical-free fish housing system and fed regular diet until 30 dpf. At this age, the length and weight of the fish were recorded and the body mass index [BMI = weight/(length)2] calculated. The BMI of fish exposed to TBBPA, TCBPA, or TBT was significantly higher than that of vehicle-treated fish (Fig. 6). Thus, we conclude that exposure to halogenated-BPAs during early development can induce weight gain in juvenile zebrafish at 1 month of age.
FIG. 6.
Effect of early chemical exposure on BMI of 30 dpf zebrafish. Changes in BMI (mg/cm2) in 30 dpf juvenile zebrafish exposed to vehicle (0.1% DMSO), 100nM TBBPA, 100nM TCBPA, and 1nM TBT from 3 to 11 dpf and fed an egg yolk diet from 6 to 11 dpf, followed by transfer to the regular housing system and regular feeding regiment until 30 dpf. Control group: n = 24, TBBPA group: n = 24, TCBPA group: n = 21, TBT group: n = 30. Statistics were done using a Student's t-test, *p < 0.05, ***p < 0.001.
DISCUSSION
Because of the worldwide obesity epidemic, and the recognition of obesogens as a potential cause of it, it is important to develop assays based on in vivo models in order to screen for obesogens. In this study, we confirm that zebrafish is a suitable animal model for in vivo lipid accumulation screening, and show that halogenated-BPAs are new obesogen candidates to which early exposure can lead to weight gain later in life.
Metabolic Fate of Halogenated-BPAs in Zebrafish
We describe the metabolic fate of TBBPA and TCBPA in zebrafish after hatching stage (3 dpf). Similarly to what has been recently shown in amphibians (Fini et al., 2012), TBBPA is extensively and quickly absorbed by zebrafish larvae. The route of TBBPA absorption in zebrafish is not known. Several absorption routes are possible, such as an active oral absorption via ingestion, a passive absorption via the gills (which is often a major route of absorption in fish), or through skin diffusion. The distribution of TBBPA in tissues was recently reported in different wild fish species such as Cyprinus carpio, Culter alburnus, Carssius auratus, and Silurus asotus exposed to TBBPA from the environment (Yang et al., 2012). In those species, TBBPA was recovered in the kidneys, liver, muscle, adipose tissue, and gills, suggesting that the gills might be one of the major routes of absorption in fish for TBBPA. A similar distribution of TBBPA in tissues could be expected in zebrafish, although this has not been studied. TBBPA rapidly undergoes phase II enzyme biotransformation (sulfation), leading to the formation of a single metabolite, the corresponding sulfated conjugate (TBBPA-S). In humans, TBBPA is also rapidly absorbed and is undetectable in the plasma of volunteers exposed orally to 100 mg/kg TBBPA after 8 h. The major detected metabolite is TBBPA-S (Schauer et al., 2006). In bile duct-cannulated rats treated with radiolabeled 14C-TBBPA, TBBPA-S was identified as the major metabolite together with glucurono-conjugated metabolites of TBBPA, although these metabolites are minor ones (Hakk et al., 2000). Thus, studies in fish, amphibians, rodents, and human demonstrate that the predominant biotransformation pathway of TBBPA is sulfation, and suggest that this pathway is conserved between species. The same phase II metabolic pathway is also predominant for TCBPA. Here, we show that the metabolic fate of TCBPA is very similar to that of TBBPA, with an extensive uptake of TCBPA in the first hours of exposure, leading to the formation of one major metabolite, the TCBPA-S. These results suggest parallel metabolic pathways for TBBPA and TCBPA in vivo, and strong similarities between the metabolism of halogenated-BPAs in mammals and zebrafish.
Halogenated-BPAs are Zebrafish PPARγ Agonists
Using stably transfected cell lines with zebrafish PPARγ, we have found that neither synthetic TZDs, nor the endogenous 15-deoxy-Δ12,14-prostaglandin-J2, which are known to be high affinity ligands to hPPARγ (Forman et al., 1995), are able to activate the zebrafish homologue, probably due to the high divergence of PPARγ-LBDs that are known to be fast evolving in vertebrates (Bertrand et al., 2007). We demonstrate that these well-known hPPARγ ligands cannot be used as positive controls to study the effects driven by PPARγ in zebrafish. On the other hand, we show that TBBPA and TCBPA activate both human and zebrafish PPARγ in vitro. Despite the weak agonistic effect of these halogenated-BPAs on zfPPARγ, they are the most potent zfPPARγ agonists identified till date. The halogenated-BPAs, just like RGZ, were able to bind to and activate hPPARγ in 1 dpf transgenic reporter zebrafish, demonstrating that they were readily absorbed by the zebrafish embryos at a very early stage of development. The Tg(hPPARγ-eGFP) zebrafish appears to be a suitable humanized in vivo screening model for endocrine disruptor chemicals, integrating the effects of both parental chemicals and their biotransformation products. Indeed, TBBPA activated hPPARγ in vivo in a dose-dependent manner, as did its sulfate-conjugated major metabolite (Figs. 1G and 5A). Sulfation is an important step in the detoxification process of xenobiotics in vivo. The role of TBBPA as an endocrine disruptor of hPPARγ in vivo is further emphasized by the generation of its sulfate-conjugated metabolite, which is itself biologically active either directly through the activation of PPARγ, or indirectly following the hydrolysis of TBBPA-S, converting it back into its parental form in vivo. When TBBPA-S was administered to the larvae, no TBBPA was detected in E3 after 24 h treatment (data not shown); we suggest that a direct PPARγ activity of TBBPA-S is involved in the observed effects, because TBBPA-S activated zfPPARγ in vitro (Fig. 5B).
Halogenated-BPAs are New Obesogens
TBBPA and TCBPA are known to induce lipid accumulation in murine 3T3-L1 cells through mouse PPARγ (Riu et al., 2011a). Here, we report that these chemicals could act as obesogens in vivo. The zebrafish lipid accumulation assay that we have developed in this study allowed us to demonstrate that relatively low concentrations (10–100nM) of TCBPA, TBBPA, and TBBPA-sulfate were able to induce lipid accumulation in zebrafish treated during early development. Likewise, TBT induced lipid accumulation in zebrafish at very low concentrations (0.1 and 1nM) in accordance with a previous study (Tingaud-Sequeira et al., 2011). Similarly to humans or mice, this induction of lipid accumulation observed in zebrafish could be due to direct RXRs activation. We have shown that TBT activates human RXR-PPARγ heterodimers, primarily through its interaction with RXR (le Maire et al., 2009). If this mechanism of action is conserved in fish, TBT would bind to RXR and indirectly activate PPARγ signaling and lipid accumulation.
Two previous studies using zebrafish as a model organism have reported an lipid accumulation following exposure to TZDs (1μM RGZ or 10nM Troziglitazone) or 15d-PGJ2 (Jones et al., 2008; Tingaud-Sequeira et al., 2011). However, we demonstrate in our study that these hPPARγ activators are either not zfPPARγ agonists or they induce a weak zfPPARγ activation only at high concentrations (troziglitazone at 3μM), which might be due to the replacement of Gly284 and Cys285 in hPPARγ-LBD by serine and tyrosine residues in zfPPARγ-LBD (Riu et al., 2011a). An in vitro assay using the full-length zfPPARγ would be needed to confirm the differences in affinities for TZDs, 15d-PGJ2 and other environmental compounds such as TBBPA and TCBPA on hPPARγ and zfPPARγ. RGZ did not induce lipid accumulation in our zebrafish assay. In the light of our results, the previously described effects of TZDs on lipid accumulation might be caused by indirect zfPPARγ activation, or by the regulation of other signaling pathways. The differences between our investigation and the two previous studies could depend on differences in exposure and scoring time points. As the mechanisms leading to obesity are complex, it would be valuable to investigate obesogenic pathways and identify obesogen targeted genes in zebrafish.
We further showed that the zebrafish can be used as a model for late onset weight gain after embryonic exposure. TBBPA, TCBPA, and TBT exposure during the embryonic period (3–11 dpf) resulted in an increased BMI at 1 month of age, similar to what has been shown for compounds such as BPA and DES in rodents (Newbold et al., 2009). Possibly, the increased BMI could be the result of a “head-start” in growth caused by early-life lipid accumulation, which may be induced by a change in feeding behavior or an effect on systemic lipid metabolism. There is growing evidence suggesting that an early (perinatal) imprinting by xeno-estrogens such as BPA and DES could contribute to the etiology of obesity (Newbold et al., 2005; Wei et al., 2011). Less is known about PPARγ agonists, although both types of endocrine disruption likely involve a disruption of energy metabolism. In our experiment, zebrafish were exposed daily to 100nM halogenated-BPAs, which corresponds to 8.1 and 5.4 ng/larva/day for TBBPA and TCBPA, respectively. TBBPA is a high production chemical, produced at around 150,000 tons/year (EFSA, 2011), whereas TCBPA production is much lower at around 10,000 tons/year. Significant levels of TBBPA have been found in human cord blood (Cariou et al., 2008) and recently, based on levels of TBBPA in breast milk, it has been estimated that breast-fed 3-month-old infants have daily exposures of 0.28–257 ng/kg body weight (EFSA, 2011), demonstrating both prenatal and postnatal exposure. Exposure to halogenated-BPAs during critical windows of development could disrupt the mechanisms for controlling energy balance and facilitate weight gain later in life. From our studies, we conclude that halogenated flame retardants act as obesogens in zebrafish. It will be important to investigate which organs are the targets of obesogens in fish and whether obesogens induce lipid accumulation in both adipose tissues and nonadipose tissues, such as intestine and liver. Whether halogenated flame retardants would have similar obesogenic effects in rodents or humans remains to be determined.
By applying zebrafish as a screening model, we show for the first time that halogenated-BPA analogs are inducers of lipid accumulation in vivo in zebrafish larvae. Both TBBPA and TCBPA act as zfPPARγ ligands in vitro in reporter cell lines. We also show that the sulfated metabolite of TBBPA, which is readily produced in zebrafish as in mammals, also causes lipid accumulation in zebrafish larvae. Activation of zfPPARγ by the halogenated-BPAs correlates to increased lipid accumulation at the larval stage as well as weight gain at 1 month of age. Thus, we conclude that halogenated flame retardants have the potential to act as obesogens in zebrafish.
SUPPLEMENTARY DATA
Supplementary data are available online at http://toxsci.oxfordjournals.org/.
FUNDING
National Institutes of Health/National Institute of Environmental Health Sciences (1R21ES020036); Environmental Protection Agency (R834289); Programme National de Recherche sur les Perturbateurs Endocriniens and Agence Nationale de Recherches Contaminants, Ecosystèmes, Sante Project “BISCOT” 2010 Contaminants et Environnements: Métrologie, Santé, Adaptabilité, Comportements et Usages 004 02 (France).
Supplementary Material
Acknowledgments
We thank Dr Jens Tiefenbach and Dr Henry Krause for the gift of the pLT PPARγ transgenic zebrafish line.
Footnotes
1These authors contributed equally to this study.
Disclaimer: The views expressed in the article do not necessarily reflect the views of the funders.
REFERENCES
- Aballay L. R., Eynard A. R., Diaz Mdel P., Navarro A., Munoz S. E. Overweight and obesity: A review of their relationship to metabolic syndrome, cardiovascular disease, and cancer in South America. Nutr. Rev. 2013;71:168–179. doi: 10.1111/j.1753-4887.2012.00533.x. [DOI] [PubMed] [Google Scholar]
- Baillie-Hamilton P. F. Chemical toxins: A hypothesis to explain the global obesity epidemic. J. Altern. Complement. Med. 2002;8:185–192. doi: 10.1089/107555302317371479. [DOI] [PubMed] [Google Scholar]
- Bertrand S., Thisse B., Tavares R., Sachs L., Chaumot A., Bardet P. L., Escriva H., Duffraisse M., Marchand O., Safi R., et al. Unexpected novel relational links uncovered by extensive developmental profiling of nuclear receptor expression. PLoS Genet. 2007;3:e188. doi: 10.1371/journal.pgen.0030188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cariou R., Antignac J. P., Zalko D., Berrebi A., Cravedi J. P., Maume D., Marchand P., Monteau F., Riu A., Andre F., et al. Exposure assessment of French women and their newborns to tetrabromobisphenol-A: Occurrence measurements in maternal adipose tissue, serum, breast milk and cord serum. Chemosphere. 2008;73:1036–1041. doi: 10.1016/j.chemosphere.2008.07.084. [DOI] [PubMed] [Google Scholar]
- EFSA (European Food Safety Authority) Scientific opinion on tetrabromobisphenol A (TBBPA) and its derivatives in food. EFSA J. 2011;9:2477. [Google Scholar]
- Fan Z., Hu J., An W., Yang M. Detection and occurrence of chlorinated by-products of bisphenol A, nonylphenol and estrogens in drinking water of China: Comparison to the parent compounds. Environ. Sci. Technol. 2013;47:10841–10850. doi: 10.1021/es401504a. [DOI] [PubMed] [Google Scholar]
- Fini J. B., Riu A., Debrauwer L., Hillenweck A., Le Mevel S., Chevolleau S., Boulahtouf A., Palmier K., Balaguer P., Cravedi J. P., et al. Parallel biotransformation of tetrabromobisphenol A in Xenopus laevis and mammals: Xenopus as a model for endocrine perturbation studies. Toxicol. Sci. 2012;125:359–367. doi: 10.1093/toxsci/kfr312. [DOI] [PubMed] [Google Scholar]
- Finkelstein E. A., Khavjou O. A., Thompson H., Trogdon J. G., Pan L., Sherry B., Dietz W. Obesity and severe obesity forecasts through 2030. Am. J. Prev. Med. 2012;42:563–570. doi: 10.1016/j.amepre.2011.10.026. [DOI] [PubMed] [Google Scholar]
- Flegal K. M., Graubard B. I., Williamson D. F., Gail M. H. Cause-specific excess deaths associated with underweight, overweight, and obesity. JAMA. 2007;298:2028–2037. doi: 10.1001/jama.298.17.2028. [DOI] [PubMed] [Google Scholar]
- Flynn E. J., III, Trent C. M., Rawls J. F. Ontogeny and nutritional control of adipogenesis in zebrafish (Danio rerio) J. Lipid Res. 2009;50:1641–1652. doi: 10.1194/jlr.M800590-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forman B. M., Tontonoz P., Chen J., Brun R. P., Spiegelman B. M., Evans R. M. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR gamma. Cell. 1995;83:803–812. doi: 10.1016/0092-8674(95)90193-0. [DOI] [PubMed] [Google Scholar]
- Grun F., Blumberg B. Perturbed nuclear receptor signaling by environmental obesogens as emerging factors in the obesity crisis. Rev. Endocr. Metab. Disord. 2007;8:161–171. doi: 10.1007/s11154-007-9049-x. [DOI] [PubMed] [Google Scholar]
- Grun F., Watanabe H., Zamanian Z., Maeda L., Arima K., Cubacha R., Gardiner D. M., Kanno J., Iguchi T., Blumberg B. Endocrine-disrupting organotin compounds are potent inducers of adipogenesis in vertebrates. Mol. Endocrinol. 2006;20:2141–2155. doi: 10.1210/me.2005-0367. [DOI] [PubMed] [Google Scholar]
- Hakk H., Larsen G., Bergman A., Orn U. Metabolism, excretion and distribution of the flame retardant tetrabromobisphenol-A in conventional and bile-duct cannulated rats. Xenobiotica. 2000;30:881–890. doi: 10.1080/004982500433309. [DOI] [PubMed] [Google Scholar]
- Imrie D., Sadler K. C. White adipose tissue development in zebrafish is regulated by both developmental time and fish size. Dev. Dyn. 2010;239:3013–3023. doi: 10.1002/dvdy.22443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janesick A., Blumberg B. Endocrine disrupting chemicals and the developmental programming of adipogenesis and obesity. Birth Defects Res. C Embryo Today. 2011a;93:34–50. doi: 10.1002/bdrc.20197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janesick A., Blumberg B. Minireview: PPARgamma as the target of obesogens. J. Steroid Biochem. Mol. Biol. 2011b;127:4–8. doi: 10.1016/j.jsbmb.2011.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janesick A., Blumberg B. Obesogens, stem cells and the developmental programming of obesity. Int. J. Androl. 2012;35:437–448. doi: 10.1111/j.1365-2605.2012.01247.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones K. S., Alimov A. P., Rilo H. L., Jandacek R. J., Woollett L. A., Penberthy W. T. A high throughput live transparent animal bioassay to identify non-toxic small molecules or genes that regulate vertebrate fat metabolism for obesity drug development. Nutr. Metab. (Lond.) 2008;5:23. doi: 10.1186/1743-7075-5-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirchner S., Kieu T., Chow C., Casey S., Blumberg B. Prenatal exposure to the environmental obesogen tributyltin predisposes multipotent stem cells to become adipocytes. Mol. Endocrinol. 2010;24:526–539. doi: 10.1210/me.2009-0261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- le Maire A., Grimaldi M., Roecklin D., Dagnino S., Vivat-Hannah V., Balaguer P., Bourguet W. Activation of RXR-PPAR heterodimers by organotin environmental endocrine disruptors. EMBO Rep. 2009;10:367–373. doi: 10.1038/embor.2009.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemieux G. A., Liu J., Mayer N., Bainton R. J., Ashrafi K., Werb Z. A whole-organism screen identifies new regulators of fat storage. Nat. Chem. Biol. 2011;7:206–213. doi: 10.1038/nchembio.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Ycaza J., Blumberg B. The environmental obesogen tributyltin chloride acts via peroxisome proliferator activated receptor gamma to induce adipogenesis in murine 3T3-L1 preadipocytes. J. Steroid Biochem. Mol. Biol. 2011;127:9–15. doi: 10.1016/j.jsbmb.2011.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minchin J. E., Rawls J. F. In vivo analysis of white adipose tissue in zebrafish. Methods Cell Biol. 2011;105:63–86. doi: 10.1016/B978-0-12-381320-6.00003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newbold R. R., Padilla-Banks E., Jefferson W. N. Environmental estrogens and obesity. Mol. Cell. Endocrinol. 2009;304:84–89. doi: 10.1016/j.mce.2009.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newbold R. R., Padilla-Banks E., Snyder R. J., Jefferson W. N. Developmental exposure to estrogenic compounds and obesity. Birth Defects Res. A Clin. Mol. Teratol. 2005;73:478–480. doi: 10.1002/bdra.20147. [DOI] [PubMed] [Google Scholar]
- Ogden C. L., Carroll M. D., Kit B. K., Flegal K. M. Prevalence of obesity in the United States, 2009–2010. NCHS Data Brief. 2012;82:1–8. [PubMed] [Google Scholar]
- Riu A., Grimaldi M., le Maire A., Bey G., Phillips K., Boulahtouf A., Perdu E., Zalko D., Bourguet W., Balaguer P. Peroxisome proliferator-activated receptor gamma is a target for halogenated analogs of bisphenol A. Environ. Health Perspect. 2011a;119:1227–1232. doi: 10.1289/ehp.1003328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riu A., le Maire A., Grimaldi M., Audebert M., Hillenweck A., Bourguet W., Balaguer P., Zalko D. Characterization of novel ligands of ERalpha, Erbeta, and PPARgamma: The case of halogenated bisphenol A and their conjugated metabolites. Toxicol. Sci. 2011b;122:372–382. doi: 10.1093/toxsci/kfr132. [DOI] [PubMed] [Google Scholar]
- Schauer U. M., Volkel W., Dekant W. Toxicokinetics of tetrabromobisphenol a in humans and rats after oral administration. Toxicol. Sci. 2006;91:49–58. doi: 10.1093/toxsci/kfj132. [DOI] [PubMed] [Google Scholar]
- Seimandi M., Lemaire G., Pillon A., Perrin A., Carlavan I., Voegel J. J., Vignon F., Nicolas J. C., Balaguer P. Differential responses of PPARalpha, PPARdelta, and PPARgamma reporter cell lines to selective PPAR synthetic ligands. Anal. Biochem. 2005;344:8–15. doi: 10.1016/j.ab.2005.06.010. [DOI] [PubMed] [Google Scholar]
- Tiefenbach J., Moll P. R., Nelson M. R., Hu C., Baev L., Kislinger T., Krause H. M. A live zebrafish-based screening system for human nuclear receptor ligand and cofactor discovery. PLoS One. 2010;5:e9797. doi: 10.1371/journal.pone.0009797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tingaud-Sequeira A., Ouadah N., Babin P. J. Zebrafish obesogenic test: A tool for screening molecules that target adiposity. J. Lipid Res. 2011;52:1765–1772. doi: 10.1194/jlr.D017012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei J., Lin Y., Li Y., Ying C., Chen J., Song L., Zhou Z., Lv Z., Xia W., Chen X., et al. Perinatal exposure to bisphenol A at reference dose predisposes offspring to metabolic syndrome in adult rats on a high-fat diet. Endocrinology. 2011;152:3049–3061. doi: 10.1210/en.2011-0045. [DOI] [PubMed] [Google Scholar]
- WHO. Obesity and overweight. 2013 Fact sheet no. 311. [Google Scholar]
- Yang S., Wang S., Liu H., Yan Z. Tetrabromobisphenol A: Tissue distribution in fish, and seasonal variation in water and sediment of Lake Chaohu, China. Environ. Sci. Pollut. Res. Int. 2012;19:4090–4096. doi: 10.1007/s11356-012-1023-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.