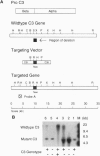
Abstract
Group B streptococci (GBS) cause sepsis and meningitis in neonates and serious infections in adults with underlying chronic illnesses. Specific antibodies have been shown to be an important factor in protective immunity for neonates, but the role of serum complement is less well defined. To elucidate the function of the complement system in immunity to this pathogen, we have used the approach of gene targeting in embryonic stem cells to generate mice totally deficient in complement component C3. Comparison of C3-deficient mice with mice deficient in complement component C4 demonstrated that the 50% lethal dose for GBS infection was reduced by approximately 50-fold and 25-fold, respectively, compared to control mice. GBS were effectively killed in vitro by human blood leukocytes in the presence of specific antibody and C4-deficient serum but not C3-deficient serum. The defective opsonization by C3-deficient serum in vitro was corroborated by in vivo studies in which passive immunization of pregnant dams with specific antibodies conferred protection from GBS challenge to normal and C4-deficient pups but not C3-deficient pups. These results indicate that the alternative pathway is sufficient to mediate effective opsonophagocytosis and protective immunity to GBS in the presence of specific antibody. In contrast, the increased susceptibility to infection of non-immune mice deficient in either C3 or C4 implies that the classical pathway plays an essential role in host defense against GBS infection in the absence of specific immunity.
Full text
PDF

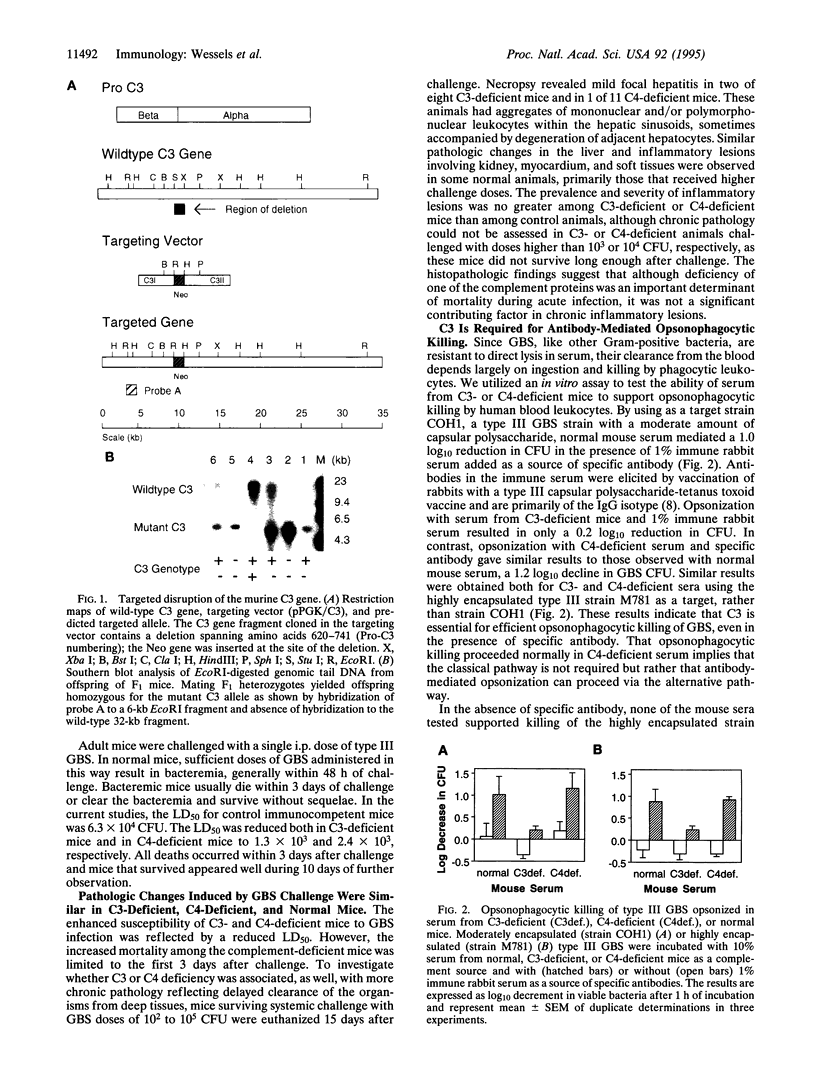
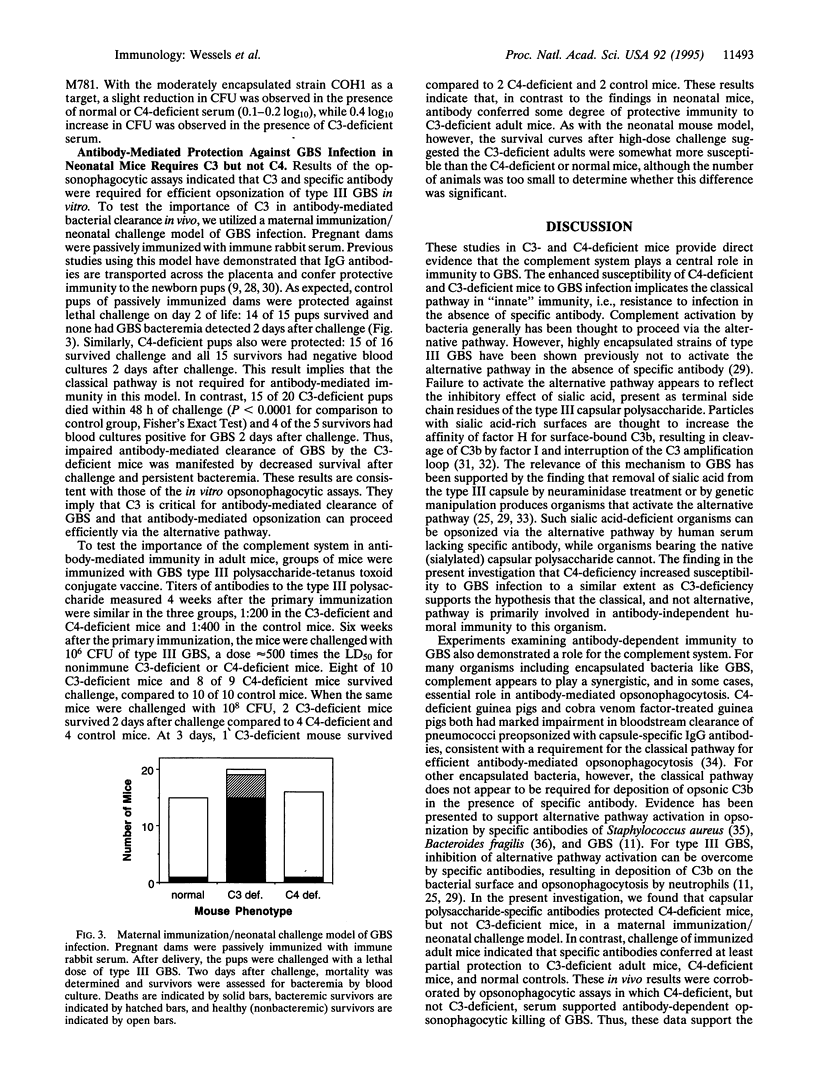

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson D. C., Freeman K. L., Heerdt B., Hughes B. J., Jack R. M., Smith C. W. Abnormal stimulated adherence of neonatal granulocytes: impaired induction of surface Mac-1 by chemotactic factors or secretagogues. Blood. 1987 Sep;70(3):740–750. [PubMed] [Google Scholar]
- Baker C. J., Edwards M. S., Kasper D. L. Role of antibody to native type III polysaccharide of group B Streptococcus in infant infection. Pediatrics. 1981 Oct;68(4):544–549. [PubMed] [Google Scholar]
- Baker C. J., Edwards M. S., Webb B. J., Kasper D. L. Antibody-independent classical pathway-mediated opsonophagocytosis of type Ia, group B streptococcus. J Clin Invest. 1982 Feb;69(2):394–404. doi: 10.1172/JCI110463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker C. J., Kasper D. L. Correlation of maternal antibody deficiency with susceptibility to neonatal group B streptococcal infection. N Engl J Med. 1976 Apr 1;294(14):753–756. doi: 10.1056/NEJM197604012941404. [DOI] [PubMed] [Google Scholar]
- Baltimore R. S., Baker C. J., Kasper D. L. Antibody to group B Streptococcus type III in human sera measured by a mouse protection test. Infect Immun. 1981 Apr;32(1):56–61. doi: 10.1128/iai.32.1.56-61.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baltimore R. S., Kasper D. L., Baker C. J., Goroff D. K. Antigenic specificity of opsonophagocytic antibodies in rabbit anti-sera to group B streptococci. J Immunol. 1977 Feb;118(2):673–678. [PubMed] [Google Scholar]
- Bjornson A. B., Bjornson H. S. Participation of immunoglobulin and the alternative complement pathway in opsonization of Bacteroides fragilis and Bacteroides thetaiotaomicron. J Infect Dis. 1978 Sep;138(3):351–358. doi: 10.1093/infdis/138.3.351. [DOI] [PubMed] [Google Scholar]
- Brown E. J., Hosea S. W., Hammer C. H., Burch C. G., Frank M. M. A quantitative analysis of the interactions of antipneumococcal antibody and complement in experimental pneumococcal bacteremia. J Clin Invest. 1982 Jan;69(1):85–98. doi: 10.1172/JCI110444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruce M. C., Baley J. E., Medvik K. A., Berger M. Impaired surface membrane expression of C3bi but not C3b receptors on neonatal neutrophils. Pediatr Res. 1987 Mar;21(3):306–311. doi: 10.1203/00006450-198703000-00022. [DOI] [PubMed] [Google Scholar]
- Eads M. E., Levy N. J., Kasper D. L., Baker C. J., Nicholson-Weller A. Antibody-independent activation of C1 by type Ia group B streptococci. J Infect Dis. 1982 Nov;146(5):665–672. doi: 10.1093/infdis/146.5.665. [DOI] [PubMed] [Google Scholar]
- Edwards M. S., Kasper D. L., Jennings H. J., Baker C. J., Nicholson-Weller A. Capsular sialic acid prevents activation of the alternative complement pathway by type III, group B streptococci. J Immunol. 1982 Mar;128(3):1278–1283. [PubMed] [Google Scholar]
- Edwards M. S., Nicholson-Weller A., Baker C. J., Kasper D. L. The role of specific antibody in alternative complement pathway-mediated opsonophagocytosis of type III, group B Streptococcus. J Exp Med. 1980 May 1;151(5):1275–1287. doi: 10.1084/jem.151.5.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farley M. M., Harvey R. C., Stull T., Smith J. D., Schuchat A., Wenger J. D., Stephens D. S. A population-based assessment of invasive disease due to group B Streptococcus in nonpregnant adults. N Engl J Med. 1993 Jun 24;328(25):1807–1811. doi: 10.1056/NEJM199306243282503. [DOI] [PubMed] [Google Scholar]
- Fearon D. T. Regulation by membrane sialic acid of beta1H-dependent decay-dissociation of amplification C3 convertase of the alternative complement pathway. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1971–1975. doi: 10.1073/pnas.75.4.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotoff S. P., Odell C., Papierniak C. K., Klegerman M. E., Boyer K. M. Human IgG antibody to group b Streptococcus type III: comparison of protective levels in a murine model with levels in infected human neonates. J Infect Dis. 1986 Mar;153(3):511–519. doi: 10.1093/infdis/153.3.511. [DOI] [PubMed] [Google Scholar]
- Gray B. M., Pritchard D. G., Dillon H. C., Jr Seroepidemiology of group B streptococcus type III colonization at delivery. J Infect Dis. 1989 Jun;159(6):1139–1142. doi: 10.1093/infdis/159.6.1139. [DOI] [PubMed] [Google Scholar]
- Johnston R. B., Jr, Klemperer M. R., Alper C. A., Rosen F. S. The enhancement of bacterial phagocytosis by serum. The role of complement components and two cofactors. J Exp Med. 1969 Jun 1;129(6):1275–1290. doi: 10.1084/jem.129.6.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasper D. L., Baker C. J., Baltimore R. S., Crabb J. H., Schiffman G., Jennings H. J. Immunodeterminant specificity of human immunity to type III group B streptococcus. J Exp Med. 1979 Feb 1;149(2):327–339. doi: 10.1084/jem.149.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K. F., Li E., Huber L. J., Landis S. C., Sharpe A. H., Chao M. V., Jaenisch R. Targeted mutation of the gene encoding the low affinity NGF receptor p75 leads to deficits in the peripheral sensory nervous system. Cell. 1992 May 29;69(5):737–749. doi: 10.1016/0092-8674(92)90286-l. [DOI] [PubMed] [Google Scholar]
- Lundwall A., Wetsel R. A., Domdey H., Tack B. F., Fey G. H. Structure of murine complement component C3. I. Nucleotide sequence of cloned complementary and genomic DNA coding for the beta chain. J Biol Chem. 1984 Nov 25;259(22):13851–13856. [PubMed] [Google Scholar]
- Marques M. B., Kasper D. L., Pangburn M. K., Wessels M. R. Prevention of C3 deposition by capsular polysaccharide is a virulence mechanism of type III group B streptococci. Infect Immun. 1992 Oct;60(10):3986–3993. doi: 10.1128/iai.60.10.3986-3993.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pangburn M. K., Müller-Eberhard H. J. Complement C3 convertase: cell surface restriction of beta1H control and generation of restriction on neuraminidase-treated cells. Proc Natl Acad Sci U S A. 1978 May;75(5):2416–2420. doi: 10.1073/pnas.75.5.2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paoletti L. C., Wessels M. R., Rodewald A. K., Shroff A. A., Jennings H. J., Kasper D. L. Neonatal mouse protection against infection with multiple group B streptococcal (GBS) serotypes by maternal immunization with a tetravalent GBS polysaccharide-tetanus toxoid conjugate vaccine. Infect Immun. 1994 Aug;62(8):3236–3243. doi: 10.1128/iai.62.8.3236-3243.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodewald A. K., Onderdonk A. B., Warren H. B., Kasper D. L. Neonatal mouse model of group B streptococcal infection. J Infect Dis. 1992 Sep;166(3):635–639. doi: 10.1093/infdis/166.3.635. [DOI] [PubMed] [Google Scholar]
- Schuchat A., Oxtoby M., Cochi S., Sikes R. K., Hightower A., Plikaytis B., Broome C. V. Population-based risk factors for neonatal group B streptococcal disease: results of a cohort study in metropolitan Atlanta. J Infect Dis. 1990 Sep;162(3):672–677. doi: 10.1093/infdis/162.3.672. [DOI] [PubMed] [Google Scholar]
- Schwartz B., Schuchat A., Oxtoby M. J., Cochi S. L., Hightower A., Broome C. V. Invasive group B streptococcal disease in adults. A population-based study in metropolitan Atlanta. JAMA. 1991 Aug 28;266(8):1112–1114. [PubMed] [Google Scholar]
- Shigeoka A. O., Jensen C. L., Pincus S. H., Hill H. R. Absolute requirement for complement in monoclonal IgM antibody-mediated protection against experimental infection with type III group B streptococci. J Infect Dis. 1984 Jul;150(1):63–70. doi: 10.1093/infdis/150.1.63. [DOI] [PubMed] [Google Scholar]
- Smith J. B., Campbell D. E., Ludomirsky A., Polin R. A., Douglas S. D., Garty B. Z., Harris M. C. Expression of the complement receptors CR1 and CR3 and the type III Fc gamma receptor on neutrophils from newborn infants and from fetuses with Rh disease. Pediatr Res. 1990 Aug;28(2):120–126. doi: 10.1203/00006450-199008000-00009. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Verhoef J., Peterson P., Kim Y., Sabath L. D., Quie P. G. Opsonic requirements for staphylococcal phagocytosis. Heterogeneity among strains. Immunology. 1977 Aug;33(2):191–197. [PMC free article] [PubMed] [Google Scholar]
- Wessels M. R., Haft R. F., Heggen L. M., Rubens C. E. Identification of a genetic locus essential for capsule sialylation in type III group B streptococci. Infect Immun. 1992 Feb;60(2):392–400. doi: 10.1128/iai.60.2.392-400.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessels M. R., Paoletti L. C., Kasper D. L., DiFabio J. L., Michon F., Holme K., Jennings H. J. Immunogenicity in animals of a polysaccharide-protein conjugate vaccine against type III group B Streptococcus. J Clin Invest. 1990 Nov;86(5):1428–1433. doi: 10.1172/JCI114858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessels M. R., Paoletti L. C., Rodewald A. K., Michon F., DiFabio J., Jennings H. J., Kasper D. L. Stimulation of protective antibodies against type Ia and Ib group B streptococci by a type Ia polysaccharide-tetanus toxoid conjugate vaccine. Infect Immun. 1993 Nov;61(11):4760–4766. doi: 10.1128/iai.61.11.4760-4766.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zangwill K. M., Schuchat A., Wenger J. D. Group B streptococcal disease in the United States, 1990: report from a multistate active surveillance system. MMWR CDC Surveill Summ. 1992 Nov 20;41(6):25–32. [PubMed] [Google Scholar]