Abstract
Streptococcus suis (S. suis) is a family of pathogenic gram-positive bacterial strains that represents a primary health problem in the swine industry worldwide. S. suis is also an emerging zoonotic pathogen that causes severe human infections clinically featuring with varied diseases/syndromes (such as meningitis, septicemia, and arthritis). Over the past few decades, continued efforts have made significant progress toward better understanding this zoonotic infectious entity, contributing in part to the elucidation of the molecular mechanism underlying its high pathogenicity. This review is aimed at presenting an updated overview of this pathogen from the perspective of molecular epidemiology, clinical diagnosis and typing, virulence mechanism, and protective antigens contributing to its zoonosis.
Keywords: Streptococcus suis, pathogenesis, zoonosis
Introduction
Streptococcosis is regarded as a leading infectious disease in the swine industry, that clinically features with meningitis, septicemia, or arthritis and annually results in significant economic loss worldwide.1 Streptococcus suis (S. suis) that was initially reported in 19542 has been demonstrated as an etiological agent for this kind of frequently-occurring bacterial infection.1,3 Indeed, S. suis, a complex population consisting of heterogeneous strains,4 can be classified into 35 serotypes (1–34, 1/2) based on the differentiation of capsule antigens.1,3 Based on the varied virulence of these bacteria, they may be categorized into highly-pathogenic, weakly-pathogenic (hypo-virulent), and nonpathogenic (avirulent) strains.1 Generally, serotype 2 of S. suis (SS2) is considered to be the most virulent, and is frequently isolated from clinically-diseased piglets.1 In fact, serotype 9 of S. suis is also one of the most important serotypes in several countries. Of particular note, SS2 seems to be a previously neglected but recently emerging human pathogen,5 whose infection has become increasingly potent, especially in the southeast Asian countries like Thailand,6 Vietnam,7 and China.8,9
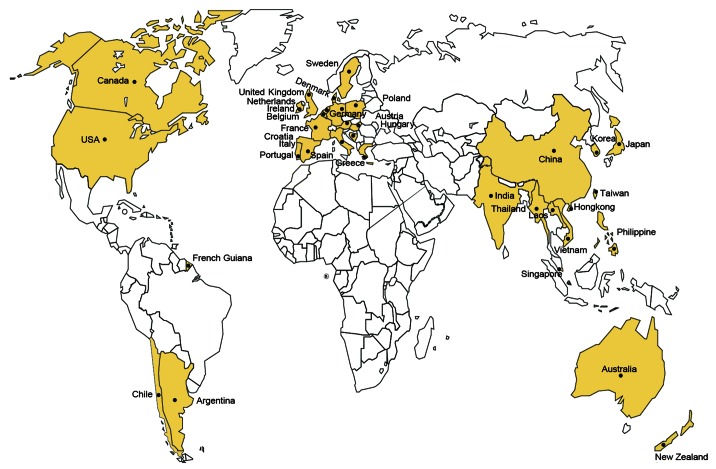
As the primary agent of meningitis, septicemia, arthritis and as an opportunistic pathogen in the case of pneumonia,1,5 S. suis have been reported to have spread over 30 countries and/or regions (Fig. 1) and has claimed no less than 1600 human cases, some of which were fatal.2 Also, similar clinical symptoms including bacterial meningitis, septicemia, and arthritis are frequently observed in human SS2 infections.2,3 Occasionally, serotypes other than SS2, including SS1,10 SS4,10 SS5,11,12 SS14,13,14 SS16,15,16 and SS2411 can also be found to function as the causative agents responsible for sporadic cases of human S. suis infection.3 Of note, two big outbreaks of human SS2 endemics which occurred in China, in 1998 and 2005, respectively,9,17,18 have raised serious concerns in public health and have challenged the conventional opinion that human SS2 infections are only present in sporadic cases.2,8,19 Unfortunately, no specific/effective human therapeutics or vaccine against SS2 infections is available thus far. Considering the severity (high mortality and modality) of SS2 infection in humans,5,8 it is important to develop a method for convenient and quick diagnosis, which can be applied toward local SS2 detection.4,18
Figure 1. Global epidemiology of human SS2 infections. Countries/regions with human cases of SS2 infections were labeled and highlighted in yellow. Adapted from references 2 and 48 with permission.
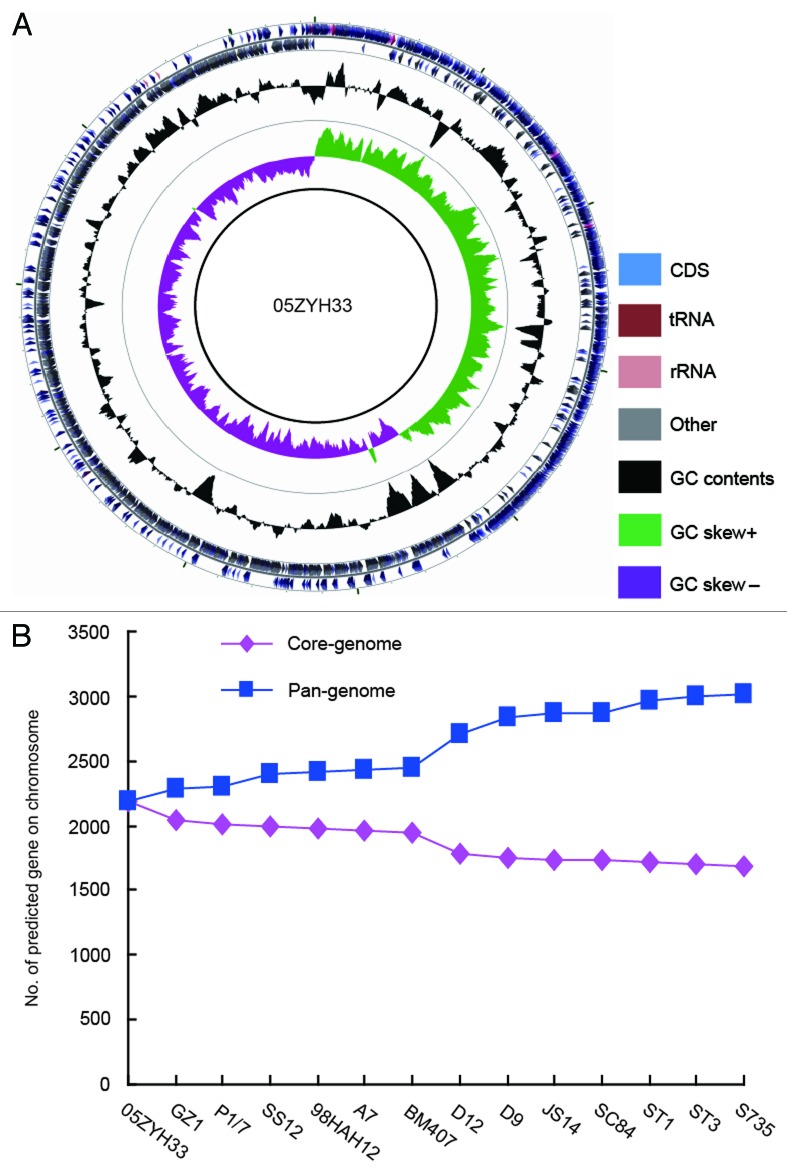
Over the past four decades, significant progress has been made toward better understanding the highly infectious clones of S. suis. At the time of formulating this review, 1104 articles were available in PubMed regarding S. suis (http://www.ncbi.nlm.nih.gov/pubmed/?term=Streptococcus+suis).Totally, over 20 bacterial virulence-associated factors have been identified that include capsular polysaccharides (CPS),20 Muramidase-released protein (MRP),21 and Suilysin (SLY).22 To date, genomic sequences of a collection of S. suis strains are available (Fig. 2), the majority of which are derived from SS2 species,23,24 except two newly-released genomes which correspond to SS325 and SS14,26 respectively. Genomic mining combined with bacterial genetics have elucidated that Chinese epidemic strains of highly pathogenic S. suis 2 carry a specific 89K PAI (pathogenicity island).23,27 Further studies suggested that 89K PAI with a transposon-like essence can undergo GI-type T4SS-mediated horizontal transfer in epidemic SS2 species.28 The systematic elucidation of the of S. suis pathogenesis in the Omics Era was illustrated by functional definition of a collection of other new genes or putative orthologs (such as Zur, a zinc uptake regulator,29 CovR, an orphan response regulator,30 and Rgg-like transcription factor31) following the release of the genome sequence of SS2 (e.g., 05ZYH33).23 Although we have gained a partial glimpse of the molecular mechanism underlying the high pathogenicity of SS2 itself, we are still lacking further insights into the interface between the SS2 pathogen and the host it infects.3,8
Figure 2. Circular diagram and pan-genome analyses of SS2 genome. (A) Circular diagram of representative SS2 genome. (B) Pan-genome analyses of S. suis 2 species.
In this review, we aim to describe an updated but partial picture of SS2 as an emerging infectious agent, which centers on five aspects: global epidemiology/distribution, clinical diagnostics/typing, pathogenesis, protective antigen/candidate vaccine, and zoonotic potential.
An Overview of S. suis
General microbiology of S. suis
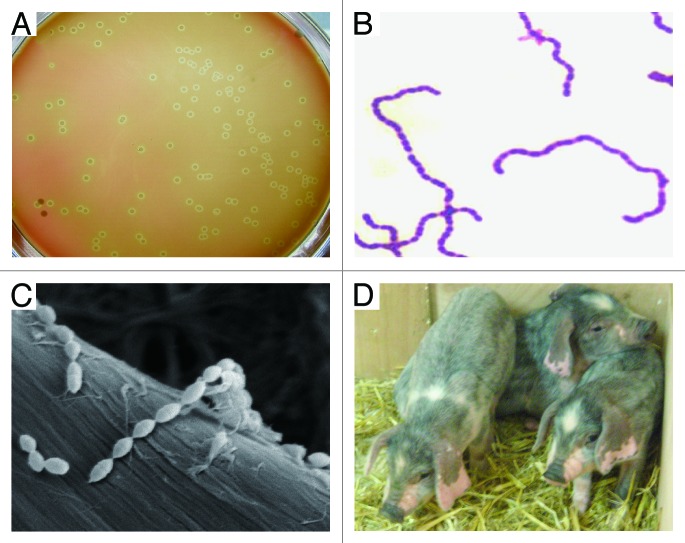
S. suis is a group of heterogeneous gram-positive bacteria that were earlier classified into Lancefield groups R, S, and T (Fig. 3).1 These bacteria are facultative anaerobes with a spherical/ovoid shape which exist in pairs and/or short chains (Fig. 3B and C). Generally, these microorganisms show either α-hemolysis when growing on selective plates of horse blood agar1 (Fig. 3A). Given the variation in their CPS antigens, 35 serotypes have been proposed for S. suis population.1 Very few studies of pathogenicity have been done for serotypes other than serotypes 2, 1, and 7.1 Among them, SS2 is recognized as the most virulent species that is frequently associated with diseased pigs and often causes an opportunistic infection of adults having occupational contact with pig carcass or pork-related products.2,5
Figure 3. Characterization of S. suis and its natural host piglets. (A) Colony phenotype of S. suis serotype 2 grown on THB plate with 5% sheep blood. (B) Gram staining analyses of S. suis serotype 2 grown in liquid THB media. It was adapted from reference 18 with permission. (C) Scanning electronic microscopic analyses of S. suis serotype 2 collected from overnight culture in THB media. (D) Phenotypic characterization of the reservoir of S. suis serotype 2, piglets maintained in backyard.
S. suis, an important animal pathogen, naturally inhabits in the upper respiratory (particularly the tonsils and nasal cavities), genital, and alimentary tracts of piglets.1,32 In addition to the natural host swine (Fig. 3D), this pathogen has been suggested to be isolated from a wide range of other animals, such as horses, dogs, and cats.1,33 Of note, some variants of S. suis probably have evolved into highly infectious zoonotic agents that can cause meningitis, septicemia, arthritis, and even streptococcal toxic shock-like syndrome (which can cause rapid death) in humans.9,17,18,34 Soon after the big outbreak of human SS2 infections in China, in 2005, serious concerns from both the public health and scientific community have been raised.18 Toward better understanding and prevention/control of SS2 infections, multiple lines of new bacterial virulence determinants (such as salK–salR two component system,27 FeoB transporter,35 and Rgg regulator31) have been identified, and fast assays using PCR-based molecular detection18 as well as ELISA (enzyme-linked immunosorbent assay)-guided diagnostics were established36 (Table 1). Most recently, epidemiological investigations conducted in Vietnam proposed that (1) pig population in slaughterhouses is a major reservoir of SS2 with a capacity to cause human infections37 and (2) the most important risk factors of human S. suis infections are consecutively eating “high risk” pork-derived dishes, occupational exposure to pigs and pork-related products, and preparation of pork in the presence of skin lesions.38 The availability of the epidemiological knowledge on human SS2 infections is critical to improve the current situation of public awareness of SS2 infections and to effectively prevent the potential occupational infections caused by SS2.
Table 1. Approaches for detection, identification and typing of Streptococcus suis.
| Different approaches | Description | Year | References |
|---|---|---|---|
| Microbiological methods | |||
| Selective medium-based cultivation | A modified Todd-Hewitt Broth agar containing 5% defibrinated sheep blood and crystal violet | 1991 | 193 |
| Molecular tests | |||
| Conventional PCR | Only gdh as target gene | 2003 | 194 |
| Swabs PCR | Only epf as target gene | 2005 | 195 |
| Nested PCR | To differentiate S. suis from other bacteria like Hemophilus parasuis | 2012 | 196 |
| Single-tube LAMP assay | Loop-mediated isothermal amplification (LAMP) with 100–1000 times higher sensitivity than the conventional PCR assay | 2012/2013 | 197 and 198 |
| Real-time PCR | cps2J, glutamate dehydrogenase (gdh) | 2010/2011 | 199 and 200 |
| Multiplex-PCR | mrp, epf | 1998/ | 201 and 202 |
| cps, epf | 2002 | 203 | |
| mrp, epf, sly | 2000/2003 | 204 and 205 | |
| cps, epf, mrp, sly, arcA, gdh | 2006/2013 | 206 and 207 | |
| 16s, cps2J | 2004 | 208 | |
| RFLP | Used for ribotyping | 1995 | 209 |
| PFGE | Pulsed-field gel electrophoresis | 2002–2011 | 37 and 210–217 |
| ISR-RFLP | PCP amplification of 16S-23S rDNA intergenic spacer region (ISR) that was followed by restriction fragment length polymorphism (RFLP) analysis (ISR-RFLP) | 2006/2007 | 210 and 218 |
| MLST | Multi-Locus Sequence Typing | 2002/2007–2012 | 37, 67, 211, 213, and 219–221 |
| MLVA | multiple-locus variable tandem repeat number analysis | 2010 | 222 |
| RAPD | randomly amplified polymorphic DNA (RAPD) | 1999 | 223 |
| Immunological assays | |||
| An enzyme-based in situ hybridization method | 16S rRNA as target gene | 2000/2001 | 224 and 225 |
| MRP/EF-based ELISA | MRP/EF protein as capture antigen | 1993 | 226 |
| CPS-based indirect ELISA | CPS as capture antigen | 1996 | 227 and 228 |
| Sao-based ELISA | SAO protein as capture antigen | 2007 | 36 |
| SERS | surface enhanced Raman scattering with MRP protein as capture antigen | 2012 | 229 |
| Indirect immunofluorescence assay | / | 2000 | 225 |
| Peroxidase-antiperoxidase method | / | 2000 | 225 |
| ICS (Immunochromatographic strip) | To detect anti-CPS antibody | 2007 | 98 |
| Colloidal gold immunochromatographic strips | To direct detection of the S. suis serotype 2 antigen | 2010 | 97 |
| Electrochemiluminescence (ECL) immunosensor | It is based on l-cysteine combined with mimicking bi-enzyme synergetic catalysis | 2012 | 99 |
/, not listed
Genomics/proteomics-based glimpse of S. suis
Genomics/proteomics approaches to probe S. suis have yielded unprecedented comprehensive information/knowledge about this pathogen.8,39 At the time of writing this review, no less than 14 genomes of S. suis strains have been available in PubMed (http://www.ncbi.nlm.nih.gov/sites/entrez?db=genome&cmd=DetailsSearch&term=streptococcus+suis&save_search=false), most of which were completed by the research groups in China (Fig. 2). Genomic sequence analyses of S. suis showed that (1) all the sequenced genomes feature with the nearly same average GC content, ~41%, indicating slight evolutionary conservation; (2) genome size varies markedly from 1 640 446 nt (05HAS68, NZ_AARD00000000) to 2 146 229 nt (BM407, NC_012926), reflecting the genomic flexibility present in these species (Fig. 2B); (3) the number of putative protein-encoding genes is dramatically different (1559 for 05HAS68, and 1932 for BM407), suggesting that nearly 1/5–1/6 of total genes are not essential (redundant) for bacterial viability, and might confer some new/unknown functions to adapt to varied environmental niches. Functional/comparative genomics of S. suis has defined a series of new infection/virulence-related determinants such as ArgR regulator,40 SspA, a subtilisin-like protease,41 CiaRH two-component regulatory system,42 and so on.
On the other hand, proteomics analyses of Chinese epidemic SS2 strain 05ZYH33 identified 373 proteins in total from 834 processed spots.43 Using an immune-proteomic approach, Lu’s group revealed 11 membrane-related proteins from Chinese vaccine strain SS2-HA980144 and 9 extracellular antigenic proteins from the virulent Chinese SS2 strain ZY05719,45 respectively. Using a similar strategy, Wu and coauthors from the same group addressed two strains of SS9, another prevalent serotype of S. suis (GZ0565 and SH040197), and observed 13 candidate proteins, five of which are virulence-associated factors.46 Additionally, eight immunogenic proteins localized on bacterial surface were determined in strain GZ0565, including extracellular solute-binding protein.47 These findings might provide a solid/reasonable basis for development of subunit vaccine candidates.39
Epidemiology of S. suis Infection
Geographic distribution of human SS2 infections
As a swine pathogen, S. suis was first reported by a vegetarian in 1954,2 while its zoonotic role could be traced to a human SS2 meningitis case in Denmark, in 1968.1 In light of the available literature with human S. suis infection recorded thus far, we expect that S. suis infections have been involved in no less than 30 countries and/or regions (Fig. 1), and resulted in around 1600 cases of severe human infections.2,8,48 In North America (United States49,50 and Canada51,52) and the South American countries (Argentina,53,54 Chile,55 and French Guiana56) only a very few cases of human SS2 infections were reported. Human SS2 infections are featuring with sporadic cases in Europe (Ireland,3 the United Kingdom [UK],3 France3,, Spain,57 Netherlands,58 Belgium,3 Poland,61 Sweden,3 Denmark,1 Germany,62 Hungary,3 Austria,63 Croatia,64 Italy65-67, Greece,68 and Portugal1), some Asian countries (Laos,1 Singapore,33,69 India,2,3,70 Korea,71-73 Japan,74 Hong Kong,75-80 Taiwan,33,81,82 and Philippine2,3), Australia,83,84 and New Zealand.85,86 So far, endemics of human SS2 infections was only observed in two Asian counties Vietnam2,7,16,37,38 and Thailand.6,11,14,87 Of particular note, coexistence of sporadic cases and epidemics of human SS2 infections were present in China.4,8,9,17-19,33,48 It seemed true that the majority of human SS2 infection cases occurred in southeast Asia (especially Vietnam, Thailand, and China), indicating an obvious geographic tropism (Fig. 1). Although we are not quite sure what mechanism can explain such kind of tropism, we anticipate that the following factors are probably correlated with frequent occurrence of human SS2 infections in above countries, which include (1) similar local climates and/or environments; (2) backyard cultivation of pigs; and (3) popular consumption of raw pork sold in the wet market.
Current situation of human S. suis infections in Europe and North America
During the past 40 years, around 100 cases of human S. suis infections were estimated in European countries. Among them, the top three countries in the history of human SS2 infections recorded are Netherlands (41 cases), United Kingdom (15 cases), and Denmark (12 cases).2 The rest of the European countries (such as France and Germany) had less than ten sporadic cases of human infection.2 Moreover, the prevalent type of clinical disease caused by S. suis infections in these countries is bacterial meningitis.
Although human S. suis infections are considered sporadic cases in most countries,2,5,8 it is unbelievable that its zoonotic infectious events are rarely reported in North America (Canada and USA), two huge countries with numerous, large, and frequent swine operations.3,5 To the best of our knowledge, only three human SS2 meningitis cases were confirmed in USA,49,50,89 and three cases were diagnosed in Canada (one case was due to SS14, and the other 2 cases were caused by SS251,52). Gottschalk and coworkers3 believe that this small number of reported cases from these two huge countries with a big industry for pig cultivation might be attributed to the following two major reasons: (1) Clinical under-diagnosis and/or misdiagnosis of S. suis infection, rather than true absence of this infectious disease, (2) S. suis isolates in North America are less virulent, relative to those from Europe and Asia. A pilot study conducted by Smith et al.90 recently indicated that human infection with S. suis is more common in the United States than what it is generally thought. The reason might lie in underdiagnosis or misdiagnosis, rather than a real lacking of disease.3,91,92 On this issue, we doubt the current situation of S. suis infections as well, though we agree that good hygiene conditions and prevention strategies during the whole process of pork operation (e.g., wearing gloves) might secure the major route of bacterial entry into blood by small cuts in the skins, and greatly decrease the incidents of S. suis infections in North America. Therefore, we believe that it is necessary to employ combined specific approaches (like multiplex PCR plus ELISA) for the re-evaluation of the epidemiological aspects of human S. suis infections in North America. Interestingly, Schmid et al.93 recently demonstrated that American SS2 isolates do not carry 89K PAI, a DNA fragment present in Chinese epidemic strain, which might be helpful for development of an American SS2 strain-specific PCR detection assay.
Situation of human S. suis infections in southeast Asia
Accumulated epidemiological data suggests that over 85% of total cases have occurred in Asian countries.2 The cases of human SS2 infections in the mainland of China and Vietnam are comparable.2,8,16 Relative to the above 2 countries, the human cases clinically infected by S. suis are second.6,14,87 Of being noteworthy, SS2 has been recognized as a pathogen with the mostly-relevance to human bacterial meningitis in southern Vietnam.16 In Hongkong, the specialized administrative region of China, the first human case of S. suis infection was recorded in 1983,78,80 and the accumulated number of S. suis infections were estimated to be about 60 cases.75,78 In particular, two big outbreaks of human SS2 infections in China (1998 and 2005) seriously challenged public health.9,18 In the 1998 epidemic, 14 out of 25 SS2-infected persons died along with an estimated 80 000 pigs.18 In the 2005 epidemic, totally 215 patients had SS2 infections, 38 of which are dead. A similar scenario was also observed when more than 600 pigs were demonstrated to be infected by SS2.9,18 The causative agents of the both epidemics were subsequently determined to be highly invasive clones of strong virulent SS2 strain that seemed to have acquired a new pathogenicity island 89K.8,23,94 Additionally, sporadic human meningitis cases caused by SS2 infections were observed in three other cities of China (Shenzhen City, Chongqing City, and Nanjing City) in 2007,4,95 implying that the situation of SS2 infection in China is complicated. This could be due to variants of S. suis 2 identified in subsequent investigations. It might be of much interest to unveil the possible evolutionary relationship of the Chinese epidemic strain with those of the neighboring country like Vietnam through comparative genomics.
Detection and Typing of S. suis
Fast/effective detection and analyses of S. suis is critical for the prevention and/or diagnosis of endemic S. suis 2 infection in the swine industry as well as for S. suis infected patients. Three types of experimental approaches are available thus far that consist of (1) selective media-based microbiological cultivation, (2) molecular tests, and (3) immunological assays (Table 1). Given its advantage in sensitivity and fastness, the second method has been experimentally developed into two subgroups (PCR-based detection plus typing-oriented analyses like pulsed-field gel electrophoresis [PFGE] and restriction fragment length polymorphism [RFLP]) and might be potential in clinical re-confirmation and/or re-valiadtion. Generally, PCR assays can be classified into six kinds among which multiplex-PCR (using sets of specific primers including epf, mrp, and gdh) is appreciably-valid approach to assay SS2 and applied in some countries (Table 1). The major four kinds of typing methods include PFGE, RFLP, multi-locus sequence typing (MLST) and random amplified polymorphic DNA (RAPD), some of which have derivatives such as ISR-RFLP (Table 1). In general, the three experimental methods (PFGE, RFLP, and RAPD) can return clues about genomic differences between different strains/serotypes. Given direct sequencing of multiple loci (cpn60, dpr, recA, aroA, thrA, gki, and mutS), MLST can directly capture the nucleotide sequence deviation used for typing purpose. Among the immunological approaches, ELISA could be the most popular way to address S. suis infection in some experimental tests (Table 1). In fact, different versions of ELISA have been developed that are based on various capture antigens identified to be specifically against S. suis 2. We have also established two kinds ELISA assays (one is based on SAO protein,4,36 the other on Enolase surface antigen96), both of which work well in our trials during field screening and clinical detections. Recently, two more new derivative methods were reported, which are immunochromatographic strip97,98 and electrochemiluminescence immunosensor,99 respectively (Table 1).
Molecular Mechanism for Streptococcus suis Infection
Bacterial virulence determinants
The clinical consequence of S. suis infection is determined by the complicated interplay between this zoonotic pathogen and its host. A series of bacterial components as well as a collection of host cell factors contribute to this pathogenesis-related process. So far, almost 60 bacterial components have been identified to be involved in the infection and/or pathogenicity of S. suis (Table 2). Of particular note, Wilson and coworkers100 developed a powerful signature-tagged mutagenesis (STM) system for S. suis and identified nearly 20 potential virulence associate elements through screening the library consisting of approximately 2600 mutants (Table 2). However, exact roles of these genes needed further verification. According to their general roles in the context of bacterial life cycles, these bacterial virulence-associated factors were temporarily classified into the following three sub-groups (of note some genes probably can be attributed to two different classifications due to their dual characteristics): (1) surface/secreted elements; (2) enzymes/proteases; (3) transcription factors/regulatory systems; and (4) others (transporters/secretion systems) (Table 2).
Table 2. Bacterial and host components associated with Streptococcus suis infectivity.
| Gene | Functional annotation | Structural information | SS2/host | References |
|---|---|---|---|---|
| Bacterial virulence-associated determinants | ||||
| Surface/secreted components (17) | ||||
| cps | Capsular polysaccharide (CPS) | Known | Strain S735 (Netherlands) | 20 and 101 |
| epf | Extra-cellular protein factor (EF) | Unknown | Pig isolate (Netherlands) | 102 |
| fbp | Fibronectin binding protein (FBP) | Unknown | Pig isolate (Netherlands) | 103 |
| mrp | Muramidase-released protein (MRP) | Unknown | Pig isolate (Netherlands) | 102 |
| 38 kDa | A protein of 38 kDa localized on bacterial surface and/or cell wall | Unknown | An avirulent Strain 1933 (Kansas, USA) | 104 |
| sly | Suilysin, thio-activated hemolysin | X-ray crystal structure at 2.85 A | Strain P1/7 (Netherlands) | 105–107, and 230 |
| SspA | A surface-associated subtilisin-like serine protease (SspA), SSU0757 | Unknown | SC-19 (China) and P1/7 (Canada) | 41 and 114 |
| 103 | Zinc-binding lipoprotein 103 | Unknown | P1/7 (Canada) | 126 |
| SSU05_1311 | A surface anchored fibronectin-binding protein | Unknown | SC-19 (China) | 113 |
| HP0197 | A surface protective antigen | Crystal structure | ZYS (China) | 115–117 |
| htpS | A histine triad surface protein | Unknown | 05ZYH33 (China) | 110 |
| hp272 or sat | HP272 or Sat surface protein | Unknown | Strain P1/7 (Netherlands); Strain 05ZYH33 (China) | |
| trag | Trag antigen | Unknown | Strain HA9801 (China) | 124 |
| ofs | OFS, a novel serum opacity factor of S. suis | Unknown | Strain 10 (Netherlands) | 120 and 231 |
| sao | Surface antigen protein (SAO), a minor virulence factor | Unknown | Strains 89/1591 (Canada) and 05ZYH33 (China) | 36, 121, and 123 |
| ssu05_0473 | PAPI-2b, a surface protein as an ancillary pilus subunit | Unknown | Strain 235/02 (pig isolate in Spain) | 125 |
| / | HP0245 | Unknown | SC-19 (China) | 232 |
| Enzymes/protease (22) | ||||
| Ssads | Adenosine synthase | Unknown | 05ZYH33 (China) | 147 |
| SsnA | DNase | Unknown | Strain 10 (Netherlands) | 233 |
| endo D | Endo-β-N-acetylglucosaminidase | Unknown | Strain S735 (Netherlands) | 100 |
| gtfA | Sucrose phosphorylase | Unknown | Strain S735 (Netherlands) | 100 |
| purA | Adenylosuccinate synthetase | Unknown | Strain S735 (Netherlands) | 100 |
| purD | Phosphoribosylamine-glycine ligase | Unknown | Strain S735 (Netherlands) | 100 |
| scrB | Sucrose-6phosphate hydrolase | Unknown | Strain S735 (Netherlands) | 100 |
| cdd | Cytidine deaminase | Unknown | Strain S735 (Netherlands) | 100 |
| neuB | Sialic acid synthase | Modeled structure | O5ZYH33 (China) | 100 and 141 |
| neuC | UDP N-Acetylglucosamine 2-Epimerase | Unknown | P1/7 (Canada) | 142 |
| luxS | S-ribosylhomocysteinase | Modeled structure | HA9801, 05ZYH33 (China) | 139 and 140 |
| igA1 | IgA1 protease | Unknown | 05ZYS (China) | 138 |
| glnA | Glutamine synthetase | Unknown | SC19a (China) | 127 |
| apuA | A multifunctional α-glucan-degrading enzyme | Unknown | Strain 10 (Netherlands) | 134 |
| gdh | Glutamate dehydrogenase (GDH) | Unknown | Strain 1933 (USA) | 128 |
| srtA | Transpeptidase mediating covalent linkage of surface proteins to peptidoglycan | Unknown | Strains NCTC10234 (Canada) and 05ZYH33 (China) | 136 and 137 |
| dltA | Enzyme catalyzing lipoteichoic acid D-alanylation | Unknown | Strain 31533 (France) | 132 |
| pgdA | Peptidoglycan N-acetylglucosamine deacetylase | Unknown | Strain 31533 (France) | 133 |
| dppIV | Di-peptidyl peptidase IV | Unknown | Strain 05ZYH33 (China) | 135 |
| eno | Enolase for dehydration of 2-phosphoglycerate to phosphoenolpyruvate (Eno) | Crystal structure (2.4A) | Strain 166 (France) and 05ZYH33 (China) | 96, 129, 143, and 144 |
| Impdh | Inosine 5-monophosphate dehydrogenase | Unknown | Strain SS2-Ha (China) | 131 |
| arcABC | An operon encoding arginine deiminase system (ADS) | Unknown | 146 | |
| Transcriptional factors/two component signal transduction systems (17) | ||||
| ccpA | Catabolite control protein A | Unknown | Strain 10 (Netherlands) and ZJJX081101 (China) | 118 and 119 |
| adcR | AdcR, a pleiotropic regulator | Unknown | Strain P1/7 (Netherlands) | 149 |
| 05SSU0053 | A predicted transcription factor that is similar to S. mutans SMU_61 | Unknown | Strain S735 (Netherlands) | 100 |
| perR | PerR, a Fur-like regulator | Unknown | Strain SC-19 (China) | 150 |
| argR | An ADS-associated repressor of the ArgR/AhrC arginine family | Unknown | Strain 10 (Netherlands) | 40 |
| rgg | Rgg transcription factor | Unknown | 05ZYH33 (China) | 31 |
| treR | Transcriptional factor | Unknown | Strain S735 (Netherlands) | 100 |
| nadR | Transcriptional factor | Unknown | Strain S735 (Netherlands) | 100 |
| scrR | A repressor for sucrose operon | Unknown | Strain S735 (Netherlands) | 100 |
| nisK-nisR | A two-component system | Unknown | 05ZYH33 (China) | 154 |
| salK-salR (suiK-suiR) | A two-component system of the 89K PAI regulating the bioactive lantibiotic suicin production | Unknown | 05ZYH33 (China) | 27, 151, and 158 |
| ciaR-ciaH | A two-component system | Unknown | SC19 (China) | 42 |
| ihk-ihr | A two-component system | Unknown | 05ZYH33 (China) | 152 |
| virR/virS | A two-component system | Unknown | 05ZYH33 (China) | 153 |
| covR | Orphan response regulator (CovR) | Unknown | Strain 05ZYH33 (China) | 30 |
| revSC21 | Orphan response regulator RevSC21 | Unknown | Strain SC21 (China) | 155 |
| revS | Orphan response regulator | Unknown | Strain 10 (Netherlands) | 156 |
| Others (5) | ||||
| 05SSU0660 | An uncharacterized protein homologous to S. pneumonia spr1018 | Unknown | Strain S735 (Netherlands) | 100 |
| feoB | FeoB transporter | Unknown | Strain P1/7 (Netherlands) | 35 |
| virD4-virB4 | Two elements of the T4SS-like system(VirD4–89K/VirB4–89K) | Unknown | 05ZYH33 (China) | 160 |
| virA | VirA, virulence factor | Unknown | Strain ZY458 (China) | 122 |
| Tig | Trigger factor | Unknown | SC21 (China) | 159 |
| Host immunological/inflammatory factors | ||||
| alI | Novel murine ribonuclease, angiogenin inhibitor 1 (AI1), interacting with hyaluronidase (Hyl) of Streptococcus suis serotype 2 | / | Murine brain | 167 |
| Pro-inflammatory cytokines tumor necrosis factor α (TNF-α), interleukin-1 (IL-1); IL-6, the chemokines IL-8 and monocyte chemotactic protein-1 (MCP-1) | / | Human brain microvascular endothelial cells | 161 | |
| tlr2 | Toll-like receptor (TLR)2 | / | Murine infection model | 165 |
| / | NFκB and MAP-kinases | / | 3D4 porcine alveolar macrophages cell line | 166 |
| cd14 | CD14 | / | 234 | |
| il-8 | Interleukin-8 (IL-8) | / | Porcine brain microvascular endothelial cell | 234 and 235 |
| / | Balance between the increased arachidonic acid, a proinflammatory ω-6 polyunsaturated fatty acid (PUFA) and decreased docosahexaenoic acid, an anti-inflammatory ω-3 PUFA | / | Macrophage | 236 |
/, not listed
Surface/secreted components
For Subgroup 1, a total of 17 genes/gene clusters have been determined thus far to contribute to bacterial pathogenicity (Table 2). In addition to six previously identified elements (capsular polysaccharides [CPS],20,101 extracellular protein factor [EF],102 fibronectin binding factor [FBP],103 muramidase-released protein [MRP],102 a protein of 38 kDa localized on bacterial surface [abbreviated 38 kDa],104 and thio-activated hemolysin with known crystal structure [Suilysin, SLY])105,106 plus implication into bacterial meningitis,107 11 more members have been supplemented into this group that include SspA, the surface-associated subtilisin-like serine protease,41,108,109 HtpS, a novel immunogenic cell surface-exposed protein,110 and Sat surface protein111,112(Table 2). Like FBP,103 the gene SSU05_1311 with unknown function was determined to be one more surface anchored fibronectin-binding protein. More importantly, this surface protein functions in vivo in crossing the mucosal epithelia to disseminate, suggesting it is a novel virulence factor.113
Two independent research groups (one from Canada41 and another from China114) identified the subtilisin-like protease (SspA)-encoding gene (sspA, SSU0757) from the S. suis organism by screening the mutant library and genomic expression library, respectively. SspA proteinase possesses a typical cell wall anchoring signal, LPXTG, at its C-terminus, implying it is a cell surface-displayed protein.108 Infection assays have demonstrated that SspA protein plays critical roles in SS2 pathogenicity.108,109,114 A subsequent study further revealed that S. suis SspA protease might modulate cytokine secretion by macrophages, and thus trigger central nervous system inflammation associated with bacterial meningitis, frequently observed in SS2-infected patients as well as piglets.41
HP0197 is a new a surface protective antigen that was originally identified by Jin’s group in 2009. This immunogenic antigen can elicit obvious humoral antibody response and confer efficient protection against SS2 challenge in the infection models of both mice and pigs.115 Subsequently, HP0197 protein was found to interact with host cell surface glycosaminoglycans (GAGs), and the binding sites were further proved by solving the X-ray structure of N-terminal GAG-binding domain combined with site-directed mutagenesis plus indirect immunofluorescence assay.116 Very recently, the same research group reported that HP0197 is involved in bacterial virulence by evaluating the performance of the isogenic mutant Δhp0197 in both mice and pigs.117 A reasonable interpretation would be that virulence attenuation is due to easier clearance of the Δhp0197 mutant by host immunological system during the stage of infection, and the fast clearance is attributed to the reduced CPS thickness and decreased resistance to phagocytosis.117 Further comparative transcriptomics-based analyses elucidated that expression of CPS synthetic operon is downregulated in the Δhp0197 mutant relative to the wild-type strain.117 Also the introduction of plasmid-borne hp0197 gene into the Δhp0197 mutant can restore the decreased expression level of cps operon to those seen with the wild type strain.117 This observation is consistent with the fact that CcpA, a global carbon catabolite regulator, contributes to bacterial infectivity/pathogenicity.118,119 Preliminary evidence pointed out that HP0197 might determine the Ser-46 phosphorylation level of phospho carrier protein (HPr-46), a partner protein of CcpA in binding the catabolite-responsive elements (cre) of the target operons. Together, it suggested that integration of the posttranslational modification of HPr-46 and CcpA-mediated transcriptional regulation is linked to regulatory network of bacterial virulence.
The serum opacity factor of S. suis (OFS) is a putative member belonging to the family of MSCRAMM (Microbial Surface Components Recognizing Adhesive Matrix Molecules).120 N-terminal region of OFS exhibits similarity to the serum opacity factor of Streptococcus pyogenes and fibronectin-binding protein A (FnBA) of Streptococcus dysgalactiae, and its C-terminus harbors repetitive sequence elements. Crude extract of S. suis and the recombinant OFS protein both possessed serum opacification activity. Experimental infections demonstrated that the deletion of the ofs gene severely impairs S. suis virulence.120
Sao is a surface antigen first identified by Li et al.,121 which can react with convalescent-phase sera from pigs clinically infected by S. suis type 2. Subsequently recombinant Sao formulated with Quil A was found to induce potent opsonizing antibody responses, and confer cross-protection of mice against challenges of virulent heterogeneous S. suis.122 We also discovered that three allelic variants of the sao gene (namely sao-S, sao-M, and sao-L) are present in S. suis population, and we have developed an efficient ELISA method in which Sao-M protein serves as the capture antigen.36 Recent further genetic study suggested that SAO protein is only a minor virulence factor123
Sat (HP272) is another newly-identified surface protein from the S. suis serotype 2,111,112 and two different research groups have shown evidence that recombinant Sat protein can confer effective immuno-protection of mice against SS2 infections, implying that it is a vaccine molecule candidate.111,112 In addition, HtpS, a putative member of histidine triad protein family, was determined to be cell surface-associated protein that was expressed during the infection of Chinese SS2 strain 05ZYH33.110 Moreover, recombinant HtpS protein was demonstrated to function as a protective antigen against SS2 infections.110 Similarly, Zhang et al.124 recently defined another new infection-associated antigen protein, Trag, using in vivo-induced antigen technology (IVIAT). An inactivation of the trag gene was observed to attenuate full virulence of Chinese SS2 strain in the experimental model of Zebrafish.124 As a surface protein, PAPI-2 was found to constitute an ancillary pilus subunit and exhibit ability to confer protection of mice against serious challenge of S. suis.125
Although we have never reported evidence that Zur, a zinc uptake regulator is essential for S. suis virulence,29 Aranda et al. very recently reported that the zinc-binding lipoprotein 103, the structural component of zinc uptake system is associated with the infectivity of Streptococcus suis, implying that zinc uptake system might be involved into bacterial pathogenesis.126 This discrepancy may be attributed to the fact that the deletion of zur only partially affect expression of zinc uptake system genes, whereas inactivation of “103” completely impairs zinc uptake system. That is why the latter could more seriously disrupt the normal zinc metabolism, and in turn lead to virulence attenuation in this sick bacterium.
Enzymes and proteinases
Accumulated data has suggested that no less than 20 bacterial enzymes might be implicated in the manifestation of S. suis virulence (Table 2). Among them, eight enzymes are proposed to be virulence factors by Wilson et al.100 using the system of signature-tagged mutagenesis (Table 2). Four of them are generally regarded as enzymes of central metabolism, which separately correspond to (1) GlnA, glutamine synthetase,127 (2) Gdh, glutamate dehydrogenase,128 (3) enolase catalyzing dehydration of 2-phosphoglycerate to phosphor-enolpyruvate,96,129,130 and (4) Impdh, inosine 5-monophosphate dehydrogenase.131 Five of these enzymes are directly or indirectly related to synthesis and/or modification of bacterial surface structure, including DltA, an enzyme catalyzing lipoteichoic d-alanylation,132 PgdA, peptidoglycan N-acetylglucosamine deacetylase,133 ApuA, a bifunctional amylopullulanase,134 DPP IV, di-peptidyl peptidase IV,135 and sortase A, a transpeptidase.136,137 One of the remaining two enzymes is IgA1 protease, which has highly immune-reactive activity to convalescent sera,138 and the other is S-ribosylhomocycteinase (LuxS) catalyzing synthesis of auto-inducer 2 (AI-2) utilized in interspecies quorum sensing.139,140 As follows, we will discuss the roles of these enzymes in S. suis pathogenesis.
With the exceptions of neuB and neuC, the remaining six among the eight enzymes proposed by Wilson (including endo-β-N-acetylglucosaminidase [endo D], sucrose phosphorylase [gtfA], adenylosuccinate synthetase [purA], phosphoribosylamine-glycine ligase [purD], sucrose-6phosphate hydrolase [scrB], cytidine deaminase [cdd]) are poorly addressed and require further experimental validation.100 NeuB is a sialic acid synthase that catalyzes the last committed step of the de novo biosynthetic pathway of sialic acid, a major element of bacterial surface structure. Recently, we systematically addressed its molecular and immunological role in bacterial virulence and claimed that an altered architecture of S. suis surface attenuates its virulence.100,141 Similarly, neuC that encodes UDP N-Acetylglucosamine 2-Epimerase with an involvement in sialic acid biosynthesis is also found to be essential for capsule production and required for virulence in a mouse infection model.142
Gdh, the glutamine dehydrogenase-encoding gene, was originally known as a virulence factor. It has been widely applied to develop effective methods for differentiate and detect virulent Streptococcus suis species.128 Si et al.127 demonstrated that glutamine synthetase, the glnA-encoding product, is associated with S. suis virulence using a mouse model. We and two other research groups96,129,143 have reported that S. suis enolase acts as an octamer,144 and can exported to the bacterial surface with capability of binding to host fibronectin, indicating its possible role in crosstalk between pathogen and host. However, the protective efficiency of recombinant enolase with different bacterial origins does not seem consistent.96,129,130 Similarly, the gene encoding Inosine 5-monophosphate dehydrogenase, a nucleotide metabolism-related enzyme, was initially cloned by Lu’s research group, and was subsequently suggested to be involved in full virulence of SS2-H, a Chinese strain in the infection model of piglets.131
Modification of the bacterial surface is critical for successful invasion and entry of pathogens into host cells. Gottschalk’s group systemically evaluated contributions of two kinds of modification systems to S. suis pathogenicity: lipoteichoic acid (LTA)-d-alanylation, and peptidoglycan (PG) N-deacetylation, respectively.132,133 The ΔdltA mutant with defection in LTA d-alanylation was found to be attenuated in its virulence, which can be correlated with its diminished adherence/invasion of porcine brain microvascular endothelial cells, and decreased capacity to escape immune clearance or killing by porcine neutrophils.132 Fittipaldi et al.133 observed that the expression level of the pgdA gene can be induced upon interaction of SS2 with neutrophils in vitro and infected mice in vivo, implying that PG N-deacetylation is tightly involved in SS2 infections. This hypothesis was further validated by virulence attenuation of the ΔpgdA mutant of SS2. Not only does ApuA behave like a bacterial surface protein with a LPKTGE cell-wall-anchoring motif at C-terminus, but it also functions as a bi-functional amylopullulanase.134 Further genetic study showed that the multifunctional α-glucan-degrading enzyme ApuA can promote adhesion to porcine epithelium and mucus, which might link bacterial carbohydrate utilization to its capability of colonization and invasiveness into hosts. We and the other research group demonstrated that Sortase A (SrtA, originally referred to as a transpeptidase in Staphylococcus aureus) is essential for full virulence of SS2.136,137 However, other sortase paralogs SrtBCD are not associated with bacterial virulence.145 In addition, we reported the functional definition of di-peptidyl peptidase IV (DPP IV) in S. suis 2, and also confirmed that it does contribute greatly to bacterial virulence.135
Quorum sensing is a method of bacterial cell density-dependent communication, using secreted chemical molecules (like the auto-inducer) as a form of “language”. The luxS gene product is the synthetase of autoinducer 2 (AI-2) that is required for interspecies communication. Recently, we and Lu’s group both defined a functional LuxS member present in Chinese isolates of S. suis 2, and demonstrated its relevance to bacterial virulence.139,140 Zhang et al. identified that IgA1 protease, an immune-dominant antigen, is necessary for full virulence of S. suis 2.138\ Additionally, Gruening et al.146 proved that the arginine deiminase system (ADS), encoded by arcABC operon, is necessary for S. suis survival in acidic stress, indicating its possibility of correlating with bacterial successful infection.
Very recently, it was discovered that the adenosine synthase functions as an effector in evasion of PMNs-mediated innate immunity147. It might suggest possibility that cyclic AMP (cAMP)-dependent signaling is linked to streptococcal infectivity. Given the fact that CadD enzyme, cAMP deaminase from Leptospira,148 can quench cAMP-dependent signaling, it would be of interest to test the hypothesis that in vivo expression of CadD protein in S. suis modulate bacterial pathogenesis or not?
Transcriptional factors/regulators
No less than 16 pleiotropic regulators have been suggested to be involved in modulation of S. suis virulence, which consist of nine transcription factors (five well-studied ones [adcR,149 ccpA,118,119 argR,40 rgg,31 a Fur-like repressor PerR150] plus four poorly-known transcription factors such as 00SSU0053, treR, nadR, and scrR100), five TCS systems (salK-salR [renamed as suiK-suiR],27,151 ciaR-ciaH,42 ihk-ihr,152 virR/virS,153 and nisK-nisR154), and three orphan regulators (CovR,30 RevSC21,155 and RevS156,157).
AdcR is a regulator controlling zinc transport in S. suis. Aranda and coauthors observed that disruption of this transcription factor can attenuate bacterial virulence in mouse model.149 In contrast, Zur, the other zinc uptake regulator from 05ZYH33 strain of S. suis 2 is not essential for strong pathogenicity in porcine models.29 Driven by the idea that host environment is critical for expression of bacterial virulence factors during the process of infection, Willenborg et al.118 evaluated the effect of the sugar metabolism regulator catabolite control protein A (CcpA) on S. suis pathogenesis. As anticipated, expression levels of several virulence factors (such as ArcB, Sao, and Enolase) were altered in the ΔccpA mutant. Of particular note, a recent study implied that Sao protective antigen plays a limited role in bacterial virulence.123 Moreover, the deletion of ccpA led to significant reduction of both capsule thickness and resistance to killing by porcine neutrophils118 and impaired bacterial virulence.119 ArgR, a member of ArgR/AhrC arginine repressor family, was recently proved to regulate expression of arcABC operon encoding an arginine deiminase system that is recognized as a putative virulence factor.40,146 Therefore, it is of interest to test the role of argR in S. suis virulence. Similar to the scenario observed with Rgg regulators present in other gram-positive pathogen, Zheng et al. defined an rgg-like ortholog in S. suis 05ZYH33, and observed its multiple roles in bacterial metabolism. More importantly, it was verified to be a virulence determinant of S. suis 2 in experimental models of piglets.31 Interestingly, an H2O2-responsive Fur-like regulator, PerR was confirmed to determine bacterial virulence through regulating expression of both dpr, a Dps-like peroxide resistance protein-encoding gene, and metQIN encoding a methionine transporter.150
Among the 15 putative two-component signal transduction systems in Chinese virulent strain of S. suis 2,23 five have been proposed to be correlated with manifestation of strong virulence.27,42,152,153 In 2008, we reported the salK-salR system present in the 89K pathogenicity island. The deletion of this TCS system resulted in significant downregulation of 26 genes’ expression level, and increased its susceptibility to polymorphonuclear leukocyte (PMN)-mediated killing. Consequently, the virulence of the ΔsalK-R mutant was seriously attenuated.27 Shen et al.158 conducted a follow-up study to address the regulatory network of SalK-SalR TCS system. As expected, proteomics-based investigation revealed 14 downregulated proteins and 1 upregulated protein, which partially agreed with our former microarray analysis.158 To our much surprise, Zhong’s group very recently elucidated the physiological role of this SalK-SalR TCS in regulating a bioactive lantibiotic suicin production, thereby replaced it with SuiK-SuiR.151
Li and coworkers42 reported the second TCS system, ciaK-ciaR, which is required for pathogenicity of SS2 in the infection models of both CD1 mice and piglets. Han et al. showed that Ihk/Irr TCS contributes to full virulence of Streptococcus suis serotype 2 strain 05ZYH33 via regulating bacterial cell metabolism.152 Very recently, two more TCS (VirR/VirS153 and NisK/R154) was demonstrated to be essential for SS2 pathogenicity.
Also, an orphan response regulator revSC21 was determined by the same research group.155 This regulator revSC21 positively modulates expression levels of virulence factors (e.g., mrp, sly, and cps) and is required for bacterial pathogenesis.155 In contrast, Pan et al. reported another orphan response regulator CovR with an opposite effect on S. suis pathogenicity.30 The covR-defective (ΔcovR) mutant displayed thicker capsules and increased hemolytic activity. Furthermore, adherence of this mutant to epithelial cells was greatly increased, as well as its resistance to phagocytosis and killing by neutrophils and monocytes. Eventually, the removal of covR gene was found to be correlated with increased lethality of piglets, relative to those inoculated with its parent virulent strain 05ZYH33.30
Others
VirA is a newly-determined virulence factor that is exclusively present in virulent SS2 strain.122 Trigger factor is also a virulence determinant which acts by controlling expression of a collection of known virulence factors, such as cps, mrp, and sly.159 FeoBA encoding an iron transporter system might represent the first example of the fact that the machinery of a transporter can be involved in S. suis pathogenicity.35 After Li et al.28 defined transposal characteristics of 89K PAI in epidemic strain of Chinese virulent SS2, Hu’s group recently obtained further insights into 89K PAI. In addition to virulence-determining element, salK-salR TCS system, 89K PAI also carried a second virulence factor, virD4-virB4, a type IV-like secretion system. Unexpectedly, this T4SS-like system is responsible for stimulating host immune reaction observed in mice infected with Chinese streptococcal toxic shock-like syndrome (STSLS)-causing strain of S. suis 2 160, raising a possible relationship of 89K to molecular/immunological machinery by which this non-group A streptococcus (GAS), SS2 to cause STSLS in infected patients (Fig. 4). Finally, Wilson et al. also suggested that 05SSU0660, an uncharacterized protein homologous to S. pneumonia spr1018 might be a virulence factor.100
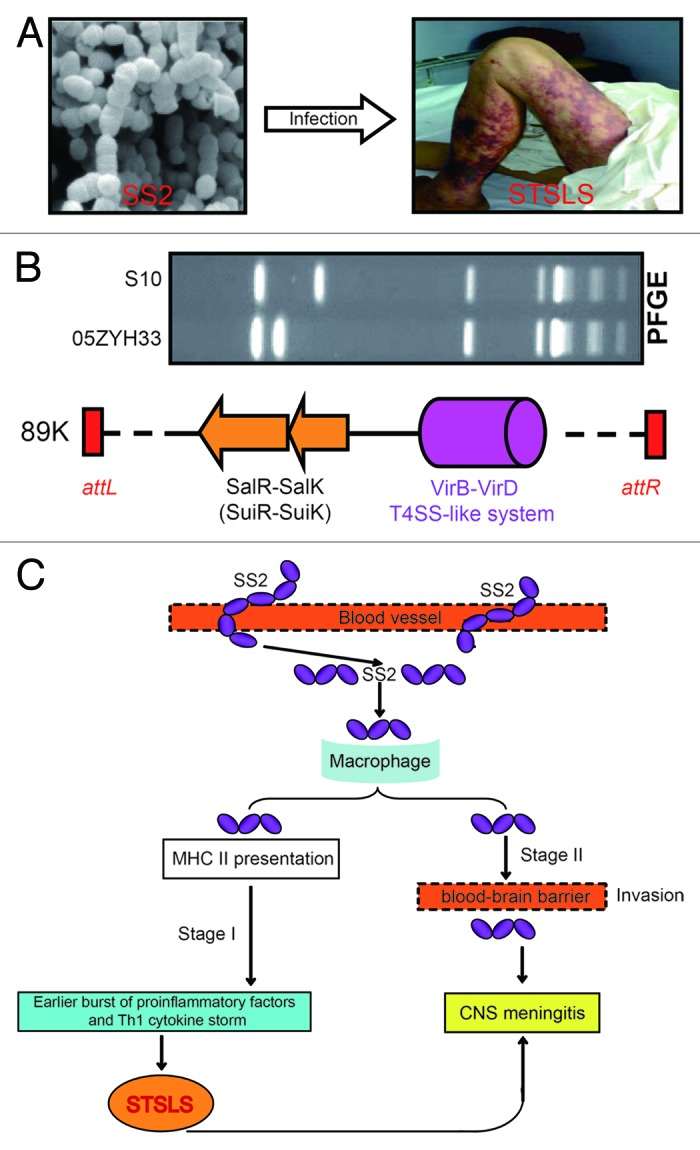
Figure 4. Clinical syndromes, genetic basis and working model for streptococcal toxic shock-like syndrome (STSLS) caused by S. suis 2. (A) Clinical visualization of a representative SS2-infected patient with streptococcal toxic shock-like syndrome. It clearly shows purpura and gangrenous changes on this patient’s legs. Partially adapted from reference 9 with permission. (B) Genetic evidence that 89K pathogenicity island carries elements associated with bacterial virulence and the clinical consequence of streptococcal toxic shock-like syndrome. PFGE assay reveals Chinese epidemic strain 05ZYH33 is distinct from the international strain S10.4 The two genetic elements of the transposable 89K pathogenicity island23,28 have been functionally defined: one is SalK-SalR (renamed as SuiK-SuiR151) TCS with requirement for full virulence,27 and other is VirD4-VirB4 T4SS-like system associated with clinical manifestation of STSLS in mouse model.160 (C) The proposed model of the two-stage hypothesis for STSLS. SS2 gets into blood vessels in stage I and results in an early burst of proinflammatory factors and Th1 cytokine storms. Such kind of inflammatory super-responses lead to STSLS with death as early as 13 h after SS2 infection. In stage II (i.e., several days post-infection), SS2 might use virulence factors like suilysin to cause disease, particularly meningitis.94 CNS, central nervous system. Integrated and modified from references 4, 23, 27, 28, 94, and 160 with permission.
Host Immunological/Inflammatory Factors
Pathogens have evolved capacity to recruit/hijack host cell factors for their successful infections/invasions. So far, the majority of host cell factors identified to be involved in S. suis pathogenesis are immunological/apoptotic/inflammatory factors.
Nearly ten years ago, Gottschalk et al.161 had observed that cell wall components of S. suis can induce releases of interleukin-1 (IL-1), IL-8, monocyte chemotactic protein-1 (MCP-1) in human brain microvascular endothelial cells (BMEC), which might increase the permeability of the blood-brain barrier. Using the experimental model of CD1 mice, the same group observed that (1) ~20% animals with sudden death exhibit high levels of systemic TNF-α, IL-6, IL-12, IFN-γ, CCL2, CXCL1, and CCL5 24 h after infection; (2) infected mice that survived the early sepsis later developed clinical signs of meningitis present in the transcriptional activation of TLR2, TLR3, CD14, NFκB, IL-1β, CCL2, and TNF-α, mainly in myeloid cells located in affected cerebral structures.162 Apparently, the inflammatory response plays important roles in S. suis infection of CD1 mice. Similar observation was also reported by Scherk et al.163 that S. suis infections are correlated with the release of pro-inflammatory cytokines and chemokines (e.g., IL6 and IL8). Gottschalk et al. found that the toll-like receptor 2 (TLR-2)-deficient exhibit significantly reduced production of astrocytes,164 and proposed that bacterial polysaccharides probably modulates TLR2-dependent recognition of S. suis entry/invasiveness into CD1 mice.165
Using a transcriptomic approach, de Greeff et al.166 identified macrophage-specific genes (IL-1-β, MIP-2-α, and TNF-α) with significantly different expression upon S. suis infection, suggesting that MAP-kinase signaling pathway and NFκB signaling are implicated into response of porcine alveolar macrophages to S. suis infections. Additionally, Wu et al.167 verified that a novel murine ribonuclease, angiogenin inhibitor 1 (AI1) can bind to S. suis hyaluronidase (Hyl), and hypothesized that this interaction between host AI1 partner and bacterial Hyl protein might contribute to S. suis meningitis.
Protective Antigens
Development of a safe and efficient vaccine is a useful strategy to combat against S. suis infection. In contrast to conventional killed/live whole-bacteria vaccines,168-170 engineering of subunit vaccine exhibits significant advantage in its safety and its large-scale producibility.171 Identification of protective antigens is a prerequisite for identifying candidate vaccine molecules. Totally, there are no less than 15 protective antigens identified thus far (Table 3). Among them, four molecules of protective antigen are well-known virulence associated factors and are MRP, EF, 38 kDa, and SLY (Table 3). Of note, most of the remaining 10 protein antigens were elucidated by research groups in China. In 2009, in addition to identification of the known protective antigen SLY, Liu’s report verified three more new protective antigens that are RTX family exoprotein A (RfeA), epidermal surface antigen (ESA), and immunoglobulin G (IgG)-binding protein (IBP).172 Two different research groups from China confirmed that enolase, an enzyme of central metabolism, acts as a protective antigen displayed on bacterial surface.96,129 However, Esgleas and coworker130 reported an opposite result regarding the protective efficiency of enolase in mice. This discrepancy could be due to different versions of recombinant protein, different strains plus deviations in animal vaccination protocols. In particular note, all the three newly-identified immunogenic antigens (6PGD, HP0197, and HP245) are elucidated by the same Chen’s research group in China (Table 3). Interestingly, proteomics also facilitated to discover three new immunogenic proteins SsPepO,173 Sat (HP0272),112,174 and the pilus subunit PAPI-2b125 (Table 3). In disagreement with their former observation,121 Li and coworkers122 demonstrated that SAO surface antigen can confer efficient protection in both mice and piglets against virulent SS2 infection (Table 3).
Table 3. List of protective antigens and/or candidate vaccine molecules from Streptococcus suis.
| Candidate vaccine molecules | Properties | Animal model | Year | References |
|---|---|---|---|---|
| EF | Extra-cellular factor | Pigs | 2001 | 171 |
| 38 kDa | A protein of 38 kDa localized on bacterial surface and/or cell wall | Pigs | 2005 | 104 |
| SLY | Suilysin | Mice | 2009/2013 | 96, 172, and 230 |
| MRP | Muramidase-released protein | Pigs, CD1 mice | 2001, 2012 | 171 and 237 |
| SAO | Surface antigen protein | Mice and piglets | 2007 | 121 and 122 |
| 6PGD | 6-Phosphogluconate dehydrogenase (6PGD) localized on cell surface | Piglets | 2009 | 238 |
| Enolase | A surface-localized enzyme of central metabolism, catalyzing the dehydration of 2-phospho-D-glycerate to phosphoenolpyruvate | Mice | 2009 | 96, 129, 130, and 143 |
| RfeA | RTX family exoprotein A | Mice | 2009 | 172 |
| ESA | Epidermal surface antigen | Mice | 2009 | 172 |
| IBP | Immunoglobulin G (IgG)-binding protein | Mice | 2009 | 172 |
| PAPI-2b | Pilus subunit | Mice | 2010 | 125 |
| SsPepO | A secretary immunogenic protein discovered by immuno-proteomic techniques | Mice/pigs | 2011 | 173 |
| HP0197 | An immunogenic protein | Mice/piglets | 2009 | 115 |
| HP0245 | in vivo-induced protein located on the cell surface | Mice | 2011 | 239 |
| Lmb | A surface protein | Mice | 2013 | 240 |
| Sat (HP0272) | Immunogenic surface protein Sat | Mice | 2010 | 112 and 174 |
Zoonotic Potential of S. suis
Clinical consequence of human SS2 infections
It is accepted that S. suis has developed into a significant human pathogen, especially in southeast Asia, posing a great challenge to public health.2 Clinically, a collection of disease types can be observed in those patients with S. suis infections, including meningitis, septicemia, and pneumonia. In terms of epidemiological/clinical statistics, bacterial meningitis is the most prevalent symptom caused by S. suis infection.
Meningitis
Meningitis is medically defined as an inflammation of the lining that covers the brain and spinal cord (the meninges).10,16,60 This kind of inflammation usually initiates with a brief influenza-like prodroma and results in hearing impairment or loss.77,175 Generally, meningitis can be grouped into two types, bacterial and non-bacterial (e.g., viral or fungal meningitis).77 For the former, no less than 10 kinds of bacterial pathogens (such as Mycobacteria tuberculosis, Streptococcus pneumonia, and Staphylococcus aureus) have been determined as the causatives of development of human meningitis.33,77
Although SS2 is a swine pathogen, we have come to know that SS2 can also be a severe agent for human bacterial meningitis.60,88 The first case of human SS2 infection worldwide was recorded in Denmark in 1968.1 Hui et al. declared that SS2 would be another leading cause of the so-called community-acquired meningitis, which is only inferior to M. tuberculosis and S. pneumonia.77 A systematic survey of bacterial meningitis in Vietnamese adults recently suggested that (1) SS2 with multiple virulence factors (e.g., EPF, SLY, MRP) is the most common pathogen and (2) its mortality is relatively low (2.6%), but hearing loss occurs at high percentage (66.4%).16 Moreover, Hoa et al.176 addressed the antimicrobial susceptibility profile of S. suis strains from meningitis patients, an important question, using comprehensive approaches to analyze bacterial isolates in Vietnam from 1997 to 2008. As result, they found that (1) multidrug resistance in S. suis 2 causing meningitis in southern Vietnam has increased over the 11-y period studied; (2) the tet(L) carried in these bacteria is functionally expressed, and multiple other genes are probably co-expressed.176 These findings alerted us again that it is important to minimize abuse of antibiotics in treatment of bacterial infectious diseases, and that the development of new therapeutics against SS2 infections are in great demand. Fortunately, Ho et al.177 reported the largest prospective epidemiological survey of human SS2 meningitis in Vietnam, and pointed out that three important risk factors associated with human SS2 meningitis included (1) eating “high risk” dishes popular in southeast Asia, (2) occupational exposure to pigs and/or pork-related products, and (3) preparation of pork in the presence of skin lesions. Further investigations from the same research group demonstrated that slaughterhouse pigs are a major reservoir of SS2 that led to human infections of SS2 meningitis in southern Vietnam.178 It highlighted (1) the importance of an improved hygiene at pork processing facilities, and (2) the necessity for education programs concerning food safety and proper handling of pork.178
Similar findings were also noted in the Netherlands59 and New Zealand.86,179 In the Netherlands, researchers have clearly indicated that the risk of developing SS2 meningitis among abattoir workers, butchers, and pig breeders are above 1000 times higher than that among persons without close contact with pigs or their unprocessed pork products.59 To our surprise, a study performed in New Zealand revealed that a high ratio of farmers and meat inspectors in markets were sero-positive to SS2, suggesting the presence of human sub-clinical SS2 infections to a small extent.86,179
Septicemia
Septicemia is a serious and life-threatening infection, in which a large amount of bacteria are present in the blood. It is commonly referred to as “blood poisoning” or “bacteremia with sepsis”.1 This disease often causes multi-organ failure by reducing the amount of blood reaching vital organs such as the liver and kidneys.1,48,75 In addition to the known bacterial pathogens (such as Staphylococcus) with capability of leading to septicemia with about 25% lethal rate,1,180 SS2 is also a causative agent of this disease.1 In general, SS2-caused septicemia arises as a result of the localized infection in the body, especially cut skin.1 In patients with septicemia, some of the following symptoms can be observed: fever and chills, rising heart/respiratory rate, cold and clammy feeling, fallen blood pressure, paled ,and petechial skin, and ultimately even unconsciousness. SS2 most likely releases some toxins into the blood that break down the walls of blood vessels which allows blood to leak out under the skin (it is this leaking that causes the rash or petechiae).33 In some cases, SS2 infects the bloodstream and the meninges at the same time, causing both septicemia and meningitis.33,181 Due to the rapidly progressive condition of septicemia, it can evolve into an irreversible toxic shock and even acute death if sufferers do not receive urgent treatment.33,181,182 Certainly, an antibiotic remedy could be altered quickly to treat the target bacteria agents, when medical tests have identified which bacteria cause the septicemia and which antibiotics are best effective.33,182 Of note, some medical treatments themselves are the inducers of septicemia, e.g., dental treatment, long-term use of intravenous needles, a colostomy, and so on.33,175 Collectively, septicemia is so serious and complex that it deserves a lot of attention worldwide.
Others
In addition to the above two major forms of SS2-caused diseases, there are some other clinical types that occur less frequently in the cases of human SS2 infections, including arthritis, endocarditis, streptococcal toxic shock-like syndrome (STSLS), etc. Since STSLS is a newly-described disease form of SS2 infections (Fig. 4), here we have discussed it in detail. Toxic-shock syndrome refers to a highly invasive infection of deep tissues, and can be correlated with production of bacterial super-antigens (e.g., staphylococcal and streptococcal exotoxins).183,184 Before Tang et al.18 formally proposed that SS2 is another bacterial non-GAS agent responsible for STSLS, we had been aware that Staphylococcus185,186 and Streptococcus pyogenes, a group A streptococcus (GAS)183,184 are both leading pathogens with a capability of causing STSLS. The clinical criteria for diagnosis of STSLS disease can be described as follows: (1) clear erythematous blanching rash (Fig. 4), (2) sudden onset of high fever, (3) hypotension diarrhea, (4) blood spots and petechiae, and (5) dysfunction of multiple organs (e.g., disseminated intravascular coagulation and acute renal failure).8,9,18 According to above clinical criteria, two independent research groups in China reported two big outbreaks of human SS2-caused STSLS, which occurred in Jiangsu Province, 1998, and Sichuan Province, 2005, respectively.9,17-19,34 Two models have been proposed to explain the molecular mechanism by which SS2 triggers STSLS in its infected patients, one of which is a two-stage hypothesis,94 the other is a specific 89K PAI.23,160 Although we came to know that similar findings were also recorded in Thailand,187 France,188 and Australia,84 we did not gain any information on difference of these SS2 strains abroad in comparison with Chinese STSLS-causing isolates. Given that the above two models are based on evidence from studying the Chinese SS2 strains, we can’t conclude presently that they represent common or strain-specific mechanisms.
Two kinds of non-SS2 serotypes have been determined in sporadic cases of human S. suis infections: One of these is SS14,13 and the other is SS16.16 Poggenborg and coworker189 reported a pig-farm worker with SS14 meningitis and septicaemia complicated with thoracal and lumbar spine spondylodiscitis. Similarly, Ahmed et al.13 also documented a meningitis case caused by SS14 infections, in a female patient with occupational exposure to piglets each day. Of particular note, totally 12 cases of human SS14 infections were identified in Thailand during 2006–2008, and their clinical presentations included meningitis, septic arthritis and sepsis.14 Further analyses demonstrated that 11 of the 12 SS14 isolates from these patients belonged to the multilocus sequence types (ST) 105, suggesting clonal dissemination of ST105 strains in Thailand. In another neighboring Asian country, Vietnam, a fatal case of human SS16 infection was revealed by Schultsz’s research group,16 indicating multiple serotypes of S. suis are developing into human pathogens circulating in this country. Together, non-SS2 serotypes of S. suis have also exhibited its zoonotic potential, highlighting a great demand for monitoring their epidemiology and development of relevant approaches applied toward prevention and therapeutics.
Concluding Remarks and Perspectives
As a zoonotic agent, S. suis is gathering increasing accumulated attention from public health officials as well as the relevant academic community.2,3,8 With respect to S. suis epidemiology, we believe that current situation of human SS2 infections should be re-evaluated using comprehensive approaches. To minimize the occupational infections by S. suis, it is suggested that advances be made to improve public awareness of S. suis infection by science education and popularization. Although progress has been obtained toward understanding S. suis pathogenicity (especially identifying a group of bacterial virulence factors), it still sounds fragmentary, and lacks an insightful dissection of integrated regulatory networks of bacterial virulence. In the future, an important direction would be to link posttranscriptional regulation (e.g., ncRNA and riboswitch) and modification (such as acetylation and de-acetylation) to the bacterial virulence of Streptococcus suis.190 Moreover, structural information on these virulence factors is very limited (Table 2). We therefore believe it is necessary to strengthen the study of pathogenesis-related structural biology, which could establish a solid basis for design of small molecule drugs targeting bacterial virulence factors.
It is of interest to search for other virulence-related or immunological regulatory elements, different from 89K PAI, because of recent exciting findings on this issue.160 Safe and effective vaccines that can be used for patients is still not available, which is due to lack of comprehensive knowledge of S. suis infections. Description of a full picture of S. suis surface antigen proteins might be helpful to screen and/or design vaccine molecules candidate. The fact that non-SS2 serotypes of S. suis can affect humans and even lead to fatal infections, has put the situation of S. suis infection in a much more complicated and serious status in public health. This might be an alternate way to decode the genome sequences of all 35 kinds of different serotypes, providing new insights into evolution and diversity of heterogeneous species, as well as distinct clues for preventing and controlling severe infections by these pathogens.191,192 Systematic proteomic approaches are also encouraged to further address this question, which can complement genomics-based explorations.138
In summary, our understanding and response to the situation of S. suis infections occurring especially in southeast Asia is not satisfactory. It would be helpful to integrate representative virulent/avirulent strains from different countries/regions for collaborative investigations. Therefore, there is a long way to go toward the complete conquest of S. suis, an emerging human pathogen.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Acknowledgments
We apologize for not citing all relevant important references due to the space limit. We would like to thank Mr Derek Caetano-Anolles and Dr Swaminath Srinivas for the critical reading of this manuscript. It was partially supported by the startup package of Zhejiang University, China, the National Natural Science Foundation of China (31170124, 81371768, 81172794 and 31300119), National S&T Project for Infectious Diseases Control (2013ZX10004-103, 2013ZX10004-203, and 2013ZX10004-218). Y.F. is a recipient of the “Young 1000 Talents” Award, and has also been awarded as a visiting scholar in Department of Epidemiology, Research Institute for Medicine of Nanjing Command.
References
- 1.Staats JJ, Feder I, Okwumabua O, Chengappa MM. Streptococcus suis: past and present. Vet Res Commun. 1997;21:381–407. doi: 10.1023/A:1005870317757. [DOI] [PubMed] [Google Scholar]
- 2.Wertheim HF, Nghia HD, Taylor W, Schultsz C. Streptococcus suis: an emerging human pathogen. Clin Infect Dis. 2009;48:617–25. doi: 10.1086/596763. [DOI] [PubMed] [Google Scholar]
- 3.Gottschalk M, Segura M, Xu J. Streptococcus suis infections in humans: the Chinese experience and the situation in North America. Anim Health Res Rev. 2007;8:29–45. doi: 10.1017/S1466252307001247. [DOI] [PubMed] [Google Scholar]
- 4.Feng Y, Shi X, Zhang H, Zhang S, Ma Y, Zheng B, Han H, Lan Q, Tang J, Cheng J, et al. Recurrence of human Streptococcus suis infections in 2007: three cases of meningitis and implications that heterogeneous S. suis 2 circulates in China. Zoonoses Public Health. 2009;56:506–14. doi: 10.1111/j.1863-2378.2008.01225.x. [DOI] [PubMed] [Google Scholar]
- 5.Gottschalk M, Xu J, Calzas C, Segura M. Streptococcus suis: a new emerging or an old neglected zoonotic pathogen? Future Microbiol. 2010;5:371–91. doi: 10.2217/fmb.10.2. [DOI] [PubMed] [Google Scholar]
- 6.Suankratay C, Intalapaporn P, Nunthapisud P, Arunyingmongkol K, Wilde H. Streptococcus suis meningitis in Thailand. Southeast Asian J Trop Med Public Health. 2004;35:868–76. [PubMed] [Google Scholar]
- 7.Wertheim HF, Nguyen HN, Taylor W, Lien TT, Ngo HT, Nguyen TQ, Nguyen BN, Nguyen HH, Nguyen HM, Nguyen CT, et al. Streptococcus suis, an important cause of adult bacterial meningitis in northern Vietnam. PLoS One. 2009;4:e5973. doi: 10.1371/journal.pone.0005973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Feng Y, Zhang H, Ma Y, Gao GF. Uncovering newly emerging variants of Streptococcus suis, an important zoonotic agent. Trends Microbiol. 2010;18:124–31. doi: 10.1016/j.tim.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 9.Yu H, Jing H, Chen Z, Zheng H, Zhu X, Wang H, Wang S, Liu L, Zu R, Luo L, et al. Streptococcus suis study groups Human Streptococcus suis outbreak, Sichuan, China. Emerg Infect Dis. 2006;12:914–20. doi: 10.3201/eid1206.051194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arends JP, Zanen HC. Meningitis caused by Streptococcus suis in humans. Rev Infect Dis. 1988;10:131–7. doi: 10.1093/clinids/10.1.131. [DOI] [PubMed] [Google Scholar]
- 11.Kerdsin A, Dejsirilert S, Sawanpanyalert P, Boonnark A, Noithachang W, Sriyakum D, Simkum S, Chokngam S, Gottschalk M, Akeda Y, et al. Sepsis and spontaneous bacterial peritonitis in Thailand. Lancet. 2011;378:960. doi: 10.1016/S0140-6736(11)60923-9. [DOI] [PubMed] [Google Scholar]
- 12.Gustavsson C, Ramussen M. Septic arthritis caused by Streptococcus suis serotype 5 in pig farmer. Emerg Infect Dis. 2014;20:489–91. doi: 10.3201/eid2003.130535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Haleis A, Alfa M, Gottschalk M, Bernard K, Ronald A, Manickam K. Meningitis caused by Streptococcus suis serotype 14, North America. Emerg Infect Dis. 2009;15:350–2. doi: 10.3201/eid1502.080842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kerdsin A, Oishi K, Sripakdee S, Boonkerd N, Polwichai P, Nakamura S, Uchida R, Sawanpanyalert P, Dejsirilert S. Clonal dissemination of human isolates of Streptococcus suis serotype 14 in Thailand. J Med Microbiol. 2009;58:1508–13. doi: 10.1099/jmm.0.013656-0. [DOI] [PubMed] [Google Scholar]
- 15.Nghia HD, Hoa NT, Linh D, Campbell J, Diep TS, Chau NV, Mai NT, Hien TT, Spratt B, Farrar J, et al. Human case of Streptococcus suis serotype 16 infection. Emerg Infect Dis. 2008;14:155–7. doi: 10.3201/eid1401.070534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mai NT, Hoa NT, Nga TV, Linh D, Chau TT, Sinh DX, Phu NH, Chuong LV, Diep TS, Campbell J, et al. Streptococcus suis meningitis in adults in Vietnam. Clin Infect Dis. 2008;46:659–67. doi: 10.1086/527385. [DOI] [PubMed] [Google Scholar]
- 17.Ye C, Zhu X, Jing H, Du H, Segura M, Zheng H, Kan B, Wang L, Bai X, Zhou Y, et al. Streptococcus suis sequence type 7 outbreak, Sichuan, China. Emerg Infect Dis. 2006;12:1203–8. doi: 10.3201/eid1708.060232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tang J, Wang C, Feng Y, Yang W, Song H, Chen Z, Yu H, Pan X, Zhou X, Wang H, et al. Streptococcal toxic shock syndrome caused by Streptococcus suis serotype 2. PLoS Med. 2006;3:e151. doi: 10.1371/journal.pmed.0030151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ma Y, Feng Y, Liu D, Gao GF. Avian influenza virus, Streptococcus suis serotype 2, severe acute respiratory syndrome-coronavirus and beyond: molecular epidemiology, ecology and the situation in China. Philos Trans R Soc Lond B Biol Sci. 2009;364:2725–37. doi: 10.1098/rstb.2009.0093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smith HE, Damman M, van der Velde J, Wagenaar F, Wisselink HJ, Stockhofe-Zurwieden N, Smits MA. Identification and characterization of the cps locus of Streptococcus suis serotype 2: the capsule protects against phagocytosis and is an important virulence factor. Infect Immun. 1999;67:1750–6. doi: 10.1128/iai.67.4.1750-1756.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith HE, Vecht U, Wisselink HJ, Stockhofe-Zurwieden N, Biermann Y, Smits MA. Mutants of Streptococcus suis types 1 and 2 impaired in expression of muramidase-released protein and extracellular protein induce disease in newborn germfree pigs. Infect Immun. 1996;64:4409–12. doi: 10.1128/iai.64.10.4409-4412.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jacobs AA, van den Berg AJ, Loeffen PL. Protection of experimentally infected pigs by suilysin, the thiol-activated haemolysin of Streptococcus suis. Vet Rec. 1996;139:225–8. doi: 10.1136/vr.139.10.225. [DOI] [PubMed] [Google Scholar]
- 23.Chen C, Tang J, Dong W, Wang C, Feng Y, Wang J, Zheng F, Pan X, Liu D, Li M, et al. A glimpse of streptococcal toxic shock syndrome from comparative genomics of S. suis 2 Chinese isolates. PLoS One. 2007;2:e315. doi: 10.1371/journal.pone.0000315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Holden MT, Hauser H, Sanders M, Ngo TH, Cherevach I, Cronin A, Goodhead I, Mungall K, Quail MA, Price C, et al. Rapid evolution of virulence and drug resistance in the emerging zoonotic pathogen Streptococcus suis. PLoS One. 2009;4:e6072. doi: 10.1371/journal.pone.0006072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hu P, Yang M, Zhang A, Wu J, Chen B, Hua Y, Yu J, Chen H, Xiao J, Jin M. Complete genome sequence of Streptococcus suis serotype 3 strain ST3. J Bacteriol. 2011;193:3428–9. doi: 10.1128/JB.05018-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu P, Yang M, Zhang A, Wu J, Chen B, Hua Y, Yu J, Xiao J, Jin M. Complete genome sequence of Streptococcus suis serotype 14 strain JS14. J Bacteriol. 2011;193:2375–6. doi: 10.1128/JB.00083-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li M, Wang C, Feng Y, Pan X, Cheng G, Wang J, Ge J, Zheng F, Cao M, Dong Y, et al. SalK/SalR, a two-component signal transduction system, is essential for full virulence of highly invasive Streptococcus suis serotype 2. PLoS One. 2008;3:e2080. doi: 10.1371/journal.pone.0002080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li M, Shen X, Yan J, Han H, Zheng B, Liu D, Cheng H, Zhao Y, Rao X, Wang C, et al. GI-type T4SS-mediated horizontal transfer of the 89K pathogenicity island in epidemic Streptococcus suis serotype 2. Mol Microbiol. 2011;79:1670–83. doi: 10.1111/j.1365-2958.2011.07553.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Feng Y, Li M, Zhang H, Zheng B, Han H, Wang C, Yan J, Tang J, Gao GF. Functional definition and global regulation of Zur, a zinc uptake regulator in a Streptococcus suis serotype 2 strain causing streptococcal toxic shock syndrome. J Bacteriol. 2008;190:7567–78. doi: 10.1128/JB.01532-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pan X, Ge J, Li M, Wu B, Wang C, Wang J, Feng Y, Yin Z, Zheng F, Cheng G, et al. The orphan response regulator CovR: a globally negative modulator of virulence in Streptococcus suis serotype 2. J Bacteriol. 2009;191:2601–12. doi: 10.1128/JB.01309-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zheng F, Ji H, Cao M, Wang C, Feng Y, Li M, Pan X, Wang J, Qin Y, Hu F, et al. Contribution of the Rgg transcription regulator to metabolism and virulence of Streptococcus suis serotype 2. Infect Immun. 2011;79:1319–28. doi: 10.1128/IAI.00193-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gottschalk M, Segura M. The pathogenesis of the meningitis caused by Streptococcus suis: the unresolved questions. Vet Microbiol. 2000;76:259–72. doi: 10.1016/S0378-1135(00)00250-9. [DOI] [PubMed] [Google Scholar]
- 33.Huang YT, Teng LJ, Ho SW, Hsueh PR. Streptococcus suis infection. J Microbiol Immunol Infect. 2005;38:306–13. [PubMed] [Google Scholar]
- 34.Ye C, Bai X, Zhang J, Jing H, Zheng H, Du H, Cui Z, Zhang S, Jin D, Xu Y, et al. Spread of Streptococcus suis sequence type 7, China. Emerg Infect Dis. 2008;14:787–91. doi: 10.3201/eid1405.070437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Aranda J, Cortés P, Garrido ME, Fittipaldi N, Llagostera M, Gottschalk M, Barbé J. Contribution of the FeoB transporter to Streptococcus suis virulence. Int Microbiol. 2009;12:137–43. [PubMed] [Google Scholar]
- 36.Feng Y, Zheng F, Pan X, Sun W, Wang C, Dong Y, Ju AP, Ge J, Liu D, Liu C, et al. Existence and characterization of allelic variants of Sao, a newly identified surface protein from Streptococcus suis. FEMS Microbiol Lett. 2007;275:80–8. doi: 10.1111/j.1574-6968.2007.00859.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ngo TH, Tran TB, Tran TT, Nguyen VD, Campbell J, Pham HA, Huynh HT, Nguyen VV, Bryant JE, Tran TH, et al. Slaughterhouse pigs are a major reservoir of Streptococcus suis serotype 2 capable of causing human infection in southern Vietnam. PLoS One. 2011;6:e17943. doi: 10.1371/journal.pone.0017943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nghia HD, Tu TP, Wolbers M, Thai CQ, Hoang NV, Nga TV, Thao TP, Phu NH, Chau TT, Sinh DX, et al. Risk factors of Streptococcus suis infection in Vietnam. A case-control study. PLoS One. 2011;6:e17604. doi: 10.1371/journal.pone.0017604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Feng Y, Cao M, Wu Z, Chu F, Ma Y, Wang C, Zhang H, Pan X, Mao X, Zou Q. Streptococcus suis in Omics-Era: Where do we stand? J Bacteriol Parasitol. 2011;S2:001. [Google Scholar]
- 40.Fulde M, Willenborg J, de Greeff A, Benga L, Smith HE, Valentin-Weigand P, Goethe R. ArgR is an essential local transcriptional regulator of the arcABC operon in Streptococcus suis and is crucial for biological fitness in an acidic environment. Microbiology. 2011;157:572–82. doi: 10.1099/mic.0.043067-0. [DOI] [PubMed] [Google Scholar]
- 41.Bonifait L, Grenier D. The SspA subtilisin-like protease of Streptococcus suis triggers a pro-inflammatory response in macrophages through a non-proteolytic mechanism. BMC Microbiol. 2011;11:47. doi: 10.1186/1471-2180-11-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li J, Tan C, Zhou Y, Fu S, Hu L, Hu J, Chen H, Bei W. The two-component regulatory system CiaRH contributes to the virulence of Streptococcus suis 2. Vet Microbiol. 2011;148:99–104. doi: 10.1016/j.vetmic.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 43.Jing HB, Yuan J, Wang J, Yuan Y, Zhu L, Liu XK, Zheng YL, Wei KH, Zhang XM, Geng HR, et al. Proteome analysis of Streptococcus suis serotype 2. Proteomics. 2008;8:333–49. doi: 10.1002/pmic.200600930. [DOI] [PubMed] [Google Scholar]
- 44.Zhang W, Lu CP. Immunoproteomic assay of membrane-associated proteins of Streptococcus suis type 2 China vaccine strain HA9801. Zoonoses Public Health. 2007;54:253–9. doi: 10.1111/j.1863-2378.2007.01056.x. [DOI] [PubMed] [Google Scholar]
- 45.Zhang W, Lu CP. Immunoproteomics of extracellular proteins of Chinese virulent strains of Streptococcus suis type 2. Proteomics. 2007;7:4468–76. doi: 10.1002/pmic.200700294. [DOI] [PubMed] [Google Scholar]
- 46.Wu Z, Zhang W, Lu C. Comparative proteome analysis of secreted proteins of Streptococcus suis serotype 9 isolates from diseased and healthy pigs. Microb Pathog. 2008;45:159–66. doi: 10.1016/j.micpath.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 47.Wu Z, Zhang W, Lu C. Immunoproteomic assay of surface proteins of Streptococcus suis serotype 9. FEMS Immunol Med Microbiol. 2008;53:52–9. doi: 10.1111/j.1574-695X.2008.00401.x. [DOI] [PubMed] [Google Scholar]
- 48.Lun ZR, Wang QP, Chen XG, Li AX, Zhu XQ. Streptococcus suis: an emerging zoonotic pathogen. Lancet Infect Dis. 2007;7:201–9. doi: 10.1016/S1473-3099(07)70001-4. [DOI] [PubMed] [Google Scholar]
- 49.Willenburg KS, Sentochnik DE, Zadoks RN. Human Streptococcus suis meningitis in the United States. N Engl J Med. 2006;354:1325. doi: 10.1056/NEJMc053089. [DOI] [PubMed] [Google Scholar]
- 50.Lee GT, Chiu CY, Haller BL, Denn PM, Hall CS, Gerberding JL. Streptococcus suis meningitis, United States. Emerg Infect Dis. 2008;14:183–5. doi: 10.3201/eid1401.070930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Trottier S, Higgins R, Brochu G, Gottschalk M. A case of human endocarditis due to Streptococcus suis in North America. Rev Infect Dis. 1991;13:1251–2. doi: 10.1093/clinids/13.6.1251. [DOI] [PubMed] [Google Scholar]
- 52.Michaud S, Duperval R, Higgins R. Streptococcus suis meningitis: First case reported in Quebec. Can J Infect Dis. 1996;7:329–31. doi: 10.1155/1996/354693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lopreto C, Lopardo HA, Bardi MC, Gottschalk M. [Primary Streptococcus suis meningitis: first case in humans described in Latin America.] Enferm Infecc Microbiol Clin. 2005;23:110. doi: 10.1157/13071618. [DOI] [PubMed] [Google Scholar]
- 54.Callejo R, Prieto M, Salamone F, Auger JP, Goyette-Desjardins G, Gottschalk M. Atypical Streptococcus suis in man, Argentina, 2013. Emerg Infect Dis. 2014;20:500–2. doi: 10.3201/eid2003.131148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Koch E, Fuentes G, Carvajal R, Palma R, Aguirre V, Cruz C, Henríquez R, Calvo M. [Streptococcus suis meningitis in pig farmers: report of first two cases in Chile] Rev Chilena Infectol. 2013;30:557–61. doi: 10.4067/S0716-10182013000500015. [DOI] [PubMed] [Google Scholar]
- 56.Demar M, Belzunce C, Simonnet C, Renaux A, Abboud P, Okandze A, Marois-Créhan C, Djossou F. Streptococcus suis meningitis and bacteremia in man, French Guiana. Emerg Infect Dis. 2013;19:1545–6. doi: 10.3201/eid1909.121872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Geffner Sclarsky DE, Moreno Muñoz R, Campillo Alpera MS, Pardo Serrano FJ, Gómez Gómez A, Martínez-Lozano MD. [Streptococcus suis meningitis] An Med Interna. 2001;18:317–8. doi: 10.4321/S0212-71992001000600007. [DOI] [PubMed] [Google Scholar]
- 58.van de Beek D, Spanjaard L, de Gans J. Streptococcus suis meningitis in the Netherlands. J Infect. 2008;57:158–61. doi: 10.1016/j.jinf.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 59.Halaby T, Hoitsma E, Hupperts R, Spanjaard L, Luirink M, Jacobs J. Streptococcus suis meningitis, a poacher’s risk. Eur J Clin Microbiol Infect Dis. 2000;19:943–5. doi: 10.1007/PL00011230. [DOI] [PubMed] [Google Scholar]
- 60.de Ceuster LM, van Dillen JJ, Wever PC, Rozemeijer W, Louwerse ES. [Streptococcus suis meningitis in a meat factory employee] Ned Tijdschr Geneeskd. 2012;156:A5080. [PubMed] [Google Scholar]
- 61.Zalas-Wiecek P, Michalska A, Grabczewska E, Olczak A, Pawlowska M, Gospodarek E. Human meningitis casued by Streptococcus suis - case report from Poland. J Med Microbiol. 2013;62:483, 5. doi: 10.1099/jmm.0.046599-0. [DOI] [PubMed] [Google Scholar]
- 62.Rosenkranz M, Elsner HA, Stürenburg HJ, Weiller C, Röther J, Sobottka I. Streptococcus suis meningitis and septicemia contracted from a wild boar in Germany. J Neurol. 2003;250:869–70. doi: 10.1007/s00415-003-1103-3. [DOI] [PubMed] [Google Scholar]
- 63.Spiss HK, Kofler M, Hausdorfer H, Pfausler B, Schmutzhard E. [Streptococcus suis meningitis and neurophysiology of the acoustic system. First case report from Austria] Nervenarzt. 1999;70:738–41. doi: 10.1007/s001150050503. [DOI] [PubMed] [Google Scholar]
- 64.Kopić J, Paradzik MT, Pandak N. Streptococcus suis infection as a cause of severe illness: 2 cases from Croatia. Scand J Infect Dis. 2002;34:683–4. doi: 10.1080/00365540210147769. [DOI] [PubMed] [Google Scholar]
- 65.Camporese A, Tizianel G, Bruschetta G, Cruciatti B, Pomes A. Human meningitis caused by Streptococcus suis: the first case report from north-eastern Italy. Infez Med. 2007;15:111–4. [PubMed] [Google Scholar]
- 66.Manzin A, Palmieri C, Serra C, Saddi B, Princivalli MS, Loi G, Angioni G, Tiddia F, Varaldo PE, Facinelli B. Streptococcus suis meningitis without history of animal contact, Italy. Emerg Infect Dis. 2008;14:1946–8. doi: 10.3201/eid1412.080679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Princivalli MS, Palmieri C, Magi G, Vignaroli C, Manzin A, Camporese A, Barocci S, Magistrali C, Facinelli B. Genetic diversity of Streptococcus suis clinical isolates from pigs and humans in Italy (2003-2007) Euro Surveill. 2009;14:14. doi: 10.2807/ese.14.33.19310-en. [DOI] [PubMed] [Google Scholar]
- 68.Mazokopakis EE, Kofteridis DP, Papadakis JA, Gikas AH, Samonis GJ. First case report of Streptococcus suis septicaemia and meningitis from Greece. Eur J Neurol. 2005;12:487–9. doi: 10.1111/j.1468-1331.2005.00998.x. [DOI] [PubMed] [Google Scholar]
- 69.Tambyah PA, Kumarasinghe G, Chan HL, Lee KO. Streptococcus suis infection complicated by purpura fulminans and rhabdomyolysis: case report and review. Clin Infect Dis. 1997;24:710–2. doi: 10.1093/clind/24.4.710. [DOI] [PubMed] [Google Scholar]
- 70.Shneerson JM, Chattopadhyay B, Murphy MF, Fawcett IW. Permanent perceptive deafness due to Streptococcus suis type II infection. J Laryngol Otol. 1980;94:425–7. doi: 10.1017/S0022215100089040. [DOI] [PubMed] [Google Scholar]
- 71.Huh HJ, Park KJ, Jang JH, Lee M, Lee JH, Ahn YH, Kang CI, Ki CS, Lee NY. Streptococcus suis meningitis with bilateral sensorineural hearing loss. Korean J Lab Med. 2011;31:205–11. doi: 10.3343/kjlm.2011.31.3.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kim H, Lee SH, Moon HW, Kim JY, Lee SH, Hur M, Yun YM. Streptococcus suis causes septic arthritis and bacteremia: phenotypic characterization and molecular confirmation. Korean J Lab Med. 2011;31:115–7. doi: 10.3343/kjlm.2011.31.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Oh YJ, Song SH. A case of Streptococcus suis infection causing pneumonia with empyema in Korea. Tuberc Respir Dis (Seoul) 2012;73:178–81. doi: 10.4046/trd.2012.73.3.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ibaraki M, Fujita N, Tada M, Ohtaki O, Nagai H. [A Japanese case of Streptococcus suis meningitis associated with lumbar epidural abscess] Rinsho Shinkeigaku. 2003;43:176–9. [PubMed] [Google Scholar]
- 75.Ma E, Chung PH, So T, Wong L, Choi KM, Cheung DT, Kam KM, Chuang SK, Tsang T, Collaborative Study Group on Streptococcus suis infection in Hong Kong Streptococcus suis infection in Hong Kong: an emerging infectious disease? Epidemiol Infect. 2008;136:1691–7. doi: 10.1017/S0950268808000332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ip M, Fung KS, Chi F, Cheuk ES, Chau SS, Wong BW, Lui S, Hui M, Lai RW, Chan PK. Streptococcus suis in Hong Kong. Diagn Microbiol Infect Dis. 2007;57:15–20. doi: 10.1016/j.diagmicrobio.2006.05.011. [DOI] [PubMed] [Google Scholar]
- 77.Hui AC, Ng KC, Tong PY, Mok V, Chow KM, Wu A, Wong LK. Bacterial meningitis in Hong Kong: 10-years’ experience. Clin Neurol Neurosurg. 2005;107:366–70. doi: 10.1016/j.clineuro.2004.10.006. [DOI] [PubMed] [Google Scholar]
- 78.Kay R, Cheng AF, Tse CY. Streptococcus suis infection in Hong Kong. QJM. 1995;88:39–47. [PubMed] [Google Scholar]
- 79.Woo J. Streptococcus suis meningitis in man in Hong Kong. Trans R Soc Trop Med Hyg. 1986;80:848–9. doi: 10.1016/0035-9203(86)90403-7. [DOI] [PubMed] [Google Scholar]
- 80.Chau PY, Huang CY, Kay R. Streptococcus suis meningitis. An important underdiagnosed disease in Hong Kong. Med J Aust. 1983;1:414–6, 417. [PubMed] [Google Scholar]
- 81.Tsai HY, Liao CH, Liu CY, Huang YT, Teng LJ, Hsueh PR. Streptococcus suis infection in Taiwan, 2000-2011. Diagn Microbiol Infect Dis. 2012;74:75–7. doi: 10.1016/j.diagmicrobio.2012.05.013. [DOI] [PubMed] [Google Scholar]
- 82.Yen MY, Liu YC, Wang JH, Chen YS, Wang YH, Cheng DL. Streptococcus suis meningitis complicated with permanent perceptive deafness: report of a case. J Formos Med Assoc. 1994;93:349–51. [PubMed] [Google Scholar]
- 83.Kennedy KJ, Jadeer AA, Ong CW, Senanayake SN, Collignon PJ. Two cases of Streptococcus suis endocarditis in Australian piggery workers. Med J Aust. 2008;189:413. doi: 10.5694/j.1326-5377.2008.tb02100.x. [DOI] [PubMed] [Google Scholar]
- 84.Tramontana AR, Graham M, Sinickas V, Bak N. An Australian case of Streptococcus suis toxic shock syndrome associated with occupational exposure to animal carcasses. Med J Aust. 2008;188:538–9. doi: 10.5694/j.1326-5377.2008.tb01771.x. [DOI] [PubMed] [Google Scholar]
- 85.Robertson ID. Streptococcus suis type 2--a zoonotic agent in New Zealand. N Z Med J. 1986;99:167–8. [PubMed] [Google Scholar]
- 86.Dickie AS, Bremner DA, Wong PY, North JD, Robertson ID. Streptococcus suis bacteraemia. N Z Med J. 1987;100:677–8. [PubMed] [Google Scholar]
- 87.Teekakirikul P, Wiwanitkit V. Streptococcus suis infection: overview of case reports in Thailand. Southeast Asian J Trop Med Public Health. 2003;34(Suppl 2):178–83. [PubMed] [Google Scholar]
- 88.Ibaraki M, Fujita N, Tada M, Ohtaki O, Nagai H. [A Japanese case of Streptococcus suis meningitis associated with lumbar epidural abscess] Rinsho Shinkeigaku. 2003;43:176–9. [PubMed] [Google Scholar]
- 89.Fittipaldi N, Collis T, Prothero B, Gottschalk M. Streptococcus suis meningitis, Hawaii. Emerg Infect Dis. 2009;15:2067–9. doi: 10.3201/eid1512.090825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Smith TC, Capuano AW, Boese B, Myers KP, Gray GC. Exposure to Streptococcus suis among US swine workers. Emerg Infect Dis. 2008;14:1925–7. doi: 10.3201/eid1412.080162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Donsakul K, Dejthevaporn C, Witoonpanich R. Streptococcus suis infection: clinical features and diagnostic pitfalls. Southeast Asian J Trop Med Public Health. 2003;34:154–8. [PubMed] [Google Scholar]
- 92.Heidt MC, Mohamed W, Hain T, Vogt PR, Chakraborty T, Domann E. Human infective endocarditis caused by Streptococcus suis serotype 2. J Clin Microbiol. 2005;43:4898–901. doi: 10.1128/JCM.43.9.4898-4901.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Schmid S, O’Connor M, Okwumabua O. The pathogenicity island-like DNA segment associated with Chinese outbreak strain of Streptococcus suis serotype 2 is absent in the United States isolates. Int J Mol Epidemiol Genet. 2011;2:56–60. [PMC free article] [PubMed] [Google Scholar]
- 94.Ye C, Zheng H, Zhang J, Jing H, Wang L, Xiong Y, Wang W, Zhou Z, Sun Q, Luo X, et al. Clinical, experimental, and genomic differences between intermediately pathogenic, highly pathogenic, and epidemic Streptococcus suis. J Infect Dis. 2009;199:97–107. doi: 10.1086/594370. [DOI] [PubMed] [Google Scholar]
- 95.Wang C, Zheng F, Pan X, Dong R, Hu D, Qin Y, Tang J. Identification of a virulent Streptococcus suis serotype 2 strain 07NJH06 isolated from patient with streptococcal encephalomeningitis syndrome. Journal of pathogen biology. 2009;4:161, 5. [Google Scholar]
- 96.Feng Y, Pan X, Sun W, Wang C, Zhang H, Li X, Ma Y, Shao Z, Ge J, Zheng F, et al. Streptococcus suis enolase functions as a protective antigen displayed on the bacterial cell surface. J Infect Dis. 2009;200:1583–92. doi: 10.1086/644602. [DOI] [PubMed] [Google Scholar]
- 97.Ju Y, Hao HJ, Xiong GH, Geng HR, Zheng YL, Wang J, Cao Y, Yang YH, Cai XH, Jiang YQ. Development of colloidal gold-based immunochromatographic assay for rapid detection of Streptococcus suis serotype 2. Vet Immunol Immunopathol. 2010;133:207–11. doi: 10.1016/j.vetimm.2009.08.010. [DOI] [PubMed] [Google Scholar]
- 98.Yang J, Jin M, Chen J, Yang Y, Zheng P, Zhang A, Song Y, Zhou H, Chen H. Development and evaluation of an immunochromatographic strip for detection of Streptococcus suis type 2 antibody. J Vet Diagn Invest. 2007;19:355–61. doi: 10.1177/104063870701900403. [DOI] [PubMed] [Google Scholar]
- 99.Wang H, Yuan R, Chai Y, Cao Y, Gan X, Chen Y, Wang Y. An ultrasensitive peroxydisulfate electrochemiluminescence immunosensor for Streptococcus suis serotype 2 based on L-cysteine combined with mimicking bi-enzyme synergetic catalysis to in situ generate coreactant. Biosens Bioelectron. 2013;43:63–8. doi: 10.1016/j.bios.2012.11.038. [DOI] [PubMed] [Google Scholar]
- 100.Wilson TL, Jeffers J, Rapp-Gabrielson VJ, Martin S, Klein LK, Lowery DE, Fuller TE. A novel signature-tagged mutagenesis system for Streptococcus suis serotype 2. Vet Microbiol. 2007;122:135–45. doi: 10.1016/j.vetmic.2006.12.025. [DOI] [PubMed] [Google Scholar]
- 101.Segura M, Gottschalk M, Olivier M. Encapsulated Streptococcus suis inhibits activation of signaling pathways involved in phagocytosis. Infect Immun. 2004;72:5322–30. doi: 10.1128/IAI.72.9.5322-5330.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Vecht U, Wisselink HJ, Jellema ML, Smith HE. Identification of two proteins associated with virulence of Streptococcus suis type 2. Infect Immun. 1991;59:3156–62. doi: 10.1128/iai.59.9.3156-3162.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.de Greeff A, Buys H, Verhaar R, Dijkstra J, van Alphen L, Smith HE. Contribution of fibronectin-binding protein to pathogenesis of Streptococcus suis serotype 2. Infect Immun. 2002;70:1319–25. doi: 10.1128/IAI.70.3.1319-1325.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Okwumabua O, Chinnapapakkagari S. Identification of the gene encoding a 38-kilodalton immunogenic and protective antigen of Streptococcus suis. Clin Diagn Lab Immunol. 2005;12:484–90. doi: 10.1128/CDLI.12.4.484-490.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lun S, Perez-Casal J, Connor W, Willson PJ. Role of suilysin in pathogenesis of Streptococcus suis capsular serotype 2. Microb Pathog. 2003;34:27–37. doi: 10.1016/S0882-4010(02)00192-4. [DOI] [PubMed] [Google Scholar]
- 106.Xu L, Huang B, Du H, Zhang XC, Xu J, Li X, Rao Z. Crystal structure of cytotoxin protein suilysin from Streptococcus suis. Protein Cell. 2010;1:96–105. doi: 10.1007/s13238-010-0012-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Takeuchi D, Akeda Y, Nakayama T, Kerdsin A, Sano Y, Kanda T, Hamada S, Dejsirilert S, Oishi K. The contribution of suilysin to the pathogenesis of Streptococcus suis meningitis. J Infect Dis. 2014 doi: 10.1093/infdis/jit661. forthcoming. [DOI] [PubMed] [Google Scholar]
- 108.Bonifait L, de la Cruz Dominguez-Punaro M, Vaillancourt K, Bart C, Slater J, Frenette M, Gottschalk M, Grenier D. The cell envelope subtilisin-like proteinase is a virulence determinant for Streptococcus suis. BMC Microbiol. 2010;10:42. doi: 10.1186/1471-2180-10-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bonifait L, Vaillancourt K, Gottschalk M, Frenette M, Grenier D. Purification and characterization of the subtilisin-like protease of Streptococcus suis that contributes to its virulence. Vet Microbiol. 2011;148:333–40. doi: 10.1016/j.vetmic.2010.09.024. [DOI] [PubMed] [Google Scholar]
- 110.Shao Z, Pan X, Li X, Liu W, Han M, Wang C, Wang J, Zheng F, Cao M, Tang J. HtpS, a novel immunogenic cell surface-exposed protein of Streptococcus suis, confers protection in mice. FEMS Microbiol Lett. 2011;314:174–82. doi: 10.1111/j.1574-6968.2010.02162.x. [DOI] [PubMed] [Google Scholar]
- 111.Chen B, Zhang A, Li R, Mu X, He H, Chen H, Jin M. Evaluation of the protective efficacy of a newly identified immunogenic protein, HP0272, of Streptococcus suis. FEMS Microbiol Lett. 2010;307:12–8. doi: 10.1111/j.1574-6968.2010.01944.x. [DOI] [PubMed] [Google Scholar]
- 112.Mandanici F, Gómez-Gascón L, Garibaldi M, Olaya-Abril A, Luque I, Tarradas C, Mancuso G, Papasergi S, Bárcena JA, Teti G, et al. A surface protein of Streptococcus suis serotype 2 identified by proteomics protects mice against infection. J Proteomics. 2010;73:2365–9. doi: 10.1016/j.jprot.2010.07.009. [DOI] [PubMed] [Google Scholar]
- 113.Li W, Wan Y, Tao Z, Chen H, Zhou R. A novel fibronectin-binding protein of Streptococcus suis serotype 2 contributes to epithelial cell invasion and in vivo dissemination. Vet Microbiol. 2013;162:186, 94. doi: 10.1016/j.vetmic.2012.09.004. [DOI] [PubMed] [Google Scholar]
- 114.Hu P, Yang M, Zhang A, Wu J, Chen B, Hua Y, Yu J, Xiao J, Jin M. Complete genome sequence of Streptococcus suis serotype 14 strain JS14. J Bacteriol. 2011;193:2375–6. doi: 10.1128/JB.00083-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhang A, Chen B, Li R, Mu X, Han L, Zhou H, Chen H, Meilin J. Identification of a surface protective antigen, HP0197 of Streptococcus suis serotype 2. Vaccine. 2009;27:5209–13. doi: 10.1016/j.vaccine.2009.06.074. [DOI] [PubMed] [Google Scholar]
- 116.Yuan ZZ, Yan XJ, Zhang AD, Chen B, Shen YQ, Jin ML. The molecular mechanism by which surface antigen HP0197 mediates host cell attachment in the pathogenic bacteria Streptococcus suis. J Biol Chem. 2013;288:956, 63. doi: 10.1074/jbc.M112.388686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang A, Chen B, Yuan Z, Li R, Liu C, Zhou H, Chen H, Jin M. HP0197 contributes to CPS synthesis and the virulence of Streptococcus suis via CcpA. PLoS One. 2012;7:e50987. doi: 10.1371/journal.pone.0050987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Willenborg J, Fulde M, de Greeff A, Rohde M, Smith HE, Valentin-Weigand P, Goethe R. Role of glucose and CcpA in capsule expression and virulence of Streptococcus suis. Microbiology. 2011;157:1823–33. doi: 10.1099/mic.0.046417-0. [DOI] [PubMed] [Google Scholar]
- 119.Tang Y, Wu W, Zhang X, Lu Z, Chen J, Fang W. Catabolite control protein A of Streptococcus suis type 2 contributes to sugar metabolism and virulence. J Microbiol. 2012;50:994–1002. doi: 10.1007/s12275-012-2035-3. [DOI] [PubMed] [Google Scholar]
- 120.Baums CG, Kaim U, Fulde M, Ramachandran G, Goethe R, Valentin-Weigand P. Identification of a novel virulence determinant with serum opacification activity in Streptococcus suis. Infect Immun. 2006;74:6154–62. doi: 10.1128/IAI.00359-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Li Y, Martinez G, Gottschalk M, Lacouture S, Willson P, Dubreuil JD, Jacques M, Harel J. Identification of a surface protein of Streptococcus suis and evaluation of its immunogenic and protective capacity in pigs. Infect Immun. 2006;74:305–12. doi: 10.1128/IAI.74.1.305-312.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Li Y, Gottschalk M, Esgleas M, Lacouture S, Dubreuil JD, Willson P, Harel J. Immunization with recombinant Sao protein confers protection against Streptococcus suis infection. Clin Vaccine Immunol. 2007;14:937–43. doi: 10.1128/CVI.00046-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Roy D, Fittipaldi N, Dumesnil A, Lacouture S, Gottschalk M. The protective protein Sao (surface antigen one) is not a critical virulence factor for Streptococcus suis serotype 2. Microb Pathog. 2014;67-68C:31, 35. doi: 10.1016/j.micpath.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 124.Zhang H, Fan H, Lu C. Identification of a novel virulence-related gene in Streptococcus suis type 2 strains. Curr Microbiol. 2010;61:494–9. doi: 10.1007/s00284-010-9643-0. [DOI] [PubMed] [Google Scholar]
- 125.Garibaldi M, Rodríguez-Ortega MJ, Mandanici F, Cardaci A, Midiri A, Papasergi S, Gambadoro O, Cavallari V, Teti G, Beninati C. Immunoprotective activities of a Streptococcus suis pilus subunit in murine models of infection. Vaccine. 2010;28:3609–16. doi: 10.1016/j.vaccine.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 126.Aranda J, Teixidó L, Fittipaldi N, Cortés P, Llagostera M, Gottschalk M, Barbé J. Inactivation of the gene encoding zinc-binding lipoprotein 103 impairs the infectivity of Streptococcus suis. Can J Vet Res. 2012;76:72–6. [PMC free article] [PubMed] [Google Scholar]
- 127.Si Y, Yuan F, Chang H, Liu X, Li H, Cai K, Xu Z, Huang Q, Bei W, Chen H. Contribution of glutamine synthetase to the virulence of Streptococcus suis serotype 2. Vet Microbiol. 2009;139:80–8. doi: 10.1016/j.vetmic.2009.04.024. [DOI] [PubMed] [Google Scholar]
- 128.Okwumabua O, Persaud JS, Reddy PG. Cloning and characterization of the gene encoding the glutamate dehydrogenase of Streptococcus suis serotype 2. Clin Diagn Lab Immunol. 2001;8:251–7. doi: 10.1128/CDLI.8.2.251-257.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhang A, Chen B, Mu X, Li R, Zheng P, Zhao Y, Chen H, Jin M. Identification and characterization of a novel protective antigen, Enolase of Streptococcus suis serotype 2. Vaccine. 2009;27:1348–53. doi: 10.1016/j.vaccine.2008.12.047. [DOI] [PubMed] [Google Scholar]
- 130.Esgleas M, Dominguez-Punaro MdeL, Li Y, Harel J, Dubreuil JD, Gottschalk M. Immunization with SsEno fails to protect mice against challenge with Streptococcus suis serotype 2. FEMS Microbiol Lett. 2009;294:82–8. doi: 10.1111/j.1574-6968.2009.01551.x. [DOI] [PubMed] [Google Scholar]
- 131.Zhang XH, He KW, Duan ZT, Zhou JM, Yu ZY, Ni YX, Lu CP. Identification and characterization of inosine 5-monophosphate dehydrogenase in Streptococcus suis type 2. Microb Pathog. 2009;47:267–73. doi: 10.1016/j.micpath.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 132.Fittipaldi N, Sekizaki T, Takamatsu D, Harel J, Domínguez-Punaro MdeL, Von Aulock S, Draing C, Marois C, Kobisch M, Gottschalk M. D-alanylation of lipoteichoic acid contributes to the virulence of Streptococcus suis. Infect Immun. 2008;76:3587–94. doi: 10.1128/IAI.01568-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Fittipaldi N, Sekizaki T, Takamatsu D, de la Cruz Domínguez-Punaro M, Harel J, Bui NK, Vollmer W, Gottschalk M. Significant contribution of the pgdA gene to the virulence of Streptococcus suis. Mol Microbiol. 2008;70:1120–35. doi: 10.1111/j.1365-2958.2008.06463.x. [DOI] [PubMed] [Google Scholar]
- 134.Ferrando ML, Fuentes S, de Greeff A, Smith H, Wells JM. ApuA, a multifunctional alpha-glucan-degrading enzyme of Streptococcus suis, mediates adhesion to porcine epithelium and mucus. Microbiology. 2010;156:2818–28. doi: 10.1099/mic.0.037960-0. [DOI] [PubMed] [Google Scholar]
- 135.Ge J, Feng Y, Ji H, Zhang H, Zheng F, Wang C, Yin Z, Pan X, Tang J. Inactivation of dipeptidyl peptidase IV attenuates the virulence of Streptococcus suis serotype 2 that causes streptococcal toxic shock syndrome. Curr Microbiol. 2009;59:248–55. doi: 10.1007/s00284-009-9425-8. [DOI] [PubMed] [Google Scholar]
- 136.Wang C, Li M, Feng Y, Zheng F, Dong Y, Pan X, Cheng G, Dong R, Hu D, Feng X, et al. The involvement of sortase A in high virulence of STSS-causing Streptococcus suis serotype 2. Arch Microbiol. 2009;191:23–33. doi: 10.1007/s00203-008-0425-z. [DOI] [PubMed] [Google Scholar]
- 137.Vanier G, Sekizaki T, Domínguez-Punaro MC, Esgleas M, Osaki M, Takamatsu D, Segura M, Gottschalk M. Disruption of srtA gene in Streptococcus suis results in decreased interactions with endothelial cells and extracellular matrix proteins. Vet Microbiol. 2008;127:417–24. doi: 10.1016/j.vetmic.2007.08.032. [DOI] [PubMed] [Google Scholar]
- 138.Zhang A, Mu X, Chen B, Han L, Chen H, Jin M. IgA1 protease contributes to the virulence of Streptococcus suis. Vet Microbiol. 2011;148:436–9. doi: 10.1016/j.vetmic.2010.09.027. [DOI] [PubMed] [Google Scholar]
- 139.Wang Y, Zhang W, Wu Z, Zhu X, Lu C. Functional analysis of luxS in Streptococcus suis reveals a key role in biofilm formation and virulence. Vet Microbiol. 2011;152:151–60. doi: 10.1016/j.vetmic.2011.04.029. [DOI] [PubMed] [Google Scholar]
- 140.Cao M, Feng Y, Wang C, Zheng F, Li M, Liao H, Mao Y, Pan X, Wang J, Hu D, et al. Functional definition of LuxS, an autoinducer-2 (AI-2) synthase and its role in full virulence of Streptococcus suis serotype 2. J Microbiol. 2011;49:1000–11. doi: 10.1007/s12275-011-1523-1. [DOI] [PubMed] [Google Scholar]
- 141.Feng Y, Cao M, Shi J, Zhang H, Hu D, Zhu J, Zhang X, Geng M, Zheng F, Pan X, et al. Attenuation of Streptococcus suis virulence by the alteration of bacterial surface architecture. Sci Rep. 2012;2:710. doi: 10.1038/srep00710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Lecours MP, Fittipaldi N, Takamatsu D, Okura M, Segura M, Goyette-Desjardins G, Van Calsteren MR, Gottschalk M. Sialylation of Streptococcus suis serotype 2 is essential for capsule expression but is not responsible for the main capsular epitope. Microbes Infect. 2012;14:941–50. doi: 10.1016/j.micinf.2012.03.008. [DOI] [PubMed] [Google Scholar]
- 143.Esgleas M, Li Y, Hancock MA, Harel J, Dubreuil JD, Gottschalk M. Isolation and characterization of alpha-enolase, a novel fibronectin-binding protein from Streptococcus suis. Microbiology. 2008;154:2668–79. doi: 10.1099/mic.0.2008/017145-0. [DOI] [PubMed] [Google Scholar]
- 144.Lu Q, Lu H, Qi J, Lu G, Gao GF. An octamer of enolase from Streptococcus suis. Protein Cell. 2012;3:769–80. doi: 10.1007/s13238-012-2040-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Chen H, Liao H, Wang C, Pan X, Tang J. [Construction and in vitro assay of the sortase BCD geneknock-out mutant of Streptococcus suis 2] Wei Sheng Wu Xue Bao. 2011;51:386–92. [PubMed] [Google Scholar]
- 146.Gruening P, Fulde M, Valentin-Weigand P, Goethe R. Structure, regulation, and putative function of the arginine deiminase system of Streptococcus suis. J Bacteriol. 2006;188:361–9. doi: 10.1128/JB.188.2.361-369.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Liu P, Pian Y, Li X, Liu R, Xie W, Zhang C, Zheng Y, Jiang Y, Yuan Y. Streptococcus suis adenosine synthase functions as an effector in evasion of PMNs-mediated innate immunity. J Infect Dis. 2014 doi: 10.1093/infdis/jiu050. forthcoming. [DOI] [PubMed] [Google Scholar]
- 148.Goble AM, Feng Y, Raushel FM, Cronan JE. Discovery of a cAMP deaminase that quenches cyclic AMP-dependent regulation. ACS Chem Biol. 2013;8:2622–9. doi: 10.1021/cb4004628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Aranda J, Garrido ME, Fittipaldi N, Cortés P, Llagostera M, Gottschalk M, Barbé J. The cation-uptake regulators AdcR and Fur are necessary for full virulence of Streptococcus suis. Vet Microbiol. 2010;144:246–9. doi: 10.1016/j.vetmic.2009.12.037. [DOI] [PubMed] [Google Scholar]
- 150.Zhang T, Ding Y, Li T, Wan Y, Li W, Chen H, Zhou R. A Fur-like protein PerR regulates two oxidative stress response related operons dpr and metQIN in Streptococcus suis. BMC Microbiol. 2012;12:85. doi: 10.1186/1471-2180-12-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Wang J, Gao Y, Teng K, Zhang J, Sun S, Zhong J. Restoration of bioactive lantibiotic suicin from a remnant lan locus of pathogenic Streptococcus suis serotype 2. Appl Environ Microbiol. 2014;80:1062–71. doi: 10.1128/AEM.03213-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Han H, Liu C, Wang Q, Xuan C, Zheng B, Tang J, Yan J, Zhang J, Li M, Cheng H, et al. The two-component system Ihk/Irr contributes to the virulence of Streptococcus suis serotype 2 strain 05ZYH33 through alteration of the bacterial cell metabolism. Microbiology. 2012;158:1852–66. doi: 10.1099/mic.0.057448-0. [DOI] [PubMed] [Google Scholar]
- 153.Wang H, Shen X, Zhao Y, Wang M, Zhong Q, Chen T, Hu F, Li M. Identification and proteome analysis of the two-component VirR/VirS system in epidemic Streptococcus suis serotype 2. FEMS Microbiol Lett. 2012;333:160–8. doi: 10.1111/j.1574-6968.2012.02611.x. [DOI] [PubMed] [Google Scholar]
- 154.Xu J, Fu S, Liu M, Xu Q, Bei W, Chen H, Tan C. The two-component system NisK/NisR contributes to the virulence of Streptococcus suis serotype 2. Microbiol Res. 2013 doi: 10.1016/j.micres.2013.11.002. forthcoming. [DOI] [PubMed] [Google Scholar]
- 155.Wu T, Chang H, Tan C, Bei W, Chen H. The orphan response regulator RevSC21 controls the attachment of Streptococcus suis serotype-2 to human laryngeal epithelial cells and the expression of virulence genes. FEMS Microbiol Lett. 2009;292:170–81. doi: 10.1111/j.1574-6968.2008.01486.x. [DOI] [PubMed] [Google Scholar]
- 156.de Greeff A, Buys H, van Alphen L, Smith HE. Response regulator important in pathogenesis of Streptococcus suis serotype 2. Microb Pathog. 2002;33:185–92. doi: 10.1016/s0882-4010(02)90526-7. [DOI] [PubMed] [Google Scholar]
- 157.Ju AP, Wang CJ, Li M, Cheng G, Zheng F, Pan XZ, Lu CP, Tang JQ. [Construction of RevS gene knock-out mutant of Streptococcus suis serotype 2] Zhonghua Liu Xing Bing Xue Za Zhi. 2008;29:59–64. [PubMed] [Google Scholar]
- 158.Shen X, Zhong Q, Zhao Y, Yin S, Chen T, Hu F, Li M. Proteome analysis of the two-component SalK/SalR system in Epidemic Streptococcus suis serotype 2. Curr Microbiol. 2013;67:118–22. doi: 10.1007/s00284-013-0343-4. [DOI] [PubMed] [Google Scholar]
- 159.Wu T, Zhao Z, Zhang L, Ma H, Lu K, Ren W, Liu Z, Chang H, Bei W, Qiu Y, et al. Trigger factor of Streptococcus suis is involved in stress tolerance and virulence. Microb Pathog. 2011;51:69–76. doi: 10.1016/j.micpath.2010.10.001. [DOI] [PubMed] [Google Scholar]
- 160.Zhao Y, Liu G, Li S, Wang M, Song J, Wang J, Tang J, Li M, Hu F. Role of a type IV-like secretion system of Streptococcus suis 2 in the development of streptococcal toxic shock syndrome. J Infect Dis. 2011;204:274–81. doi: 10.1093/infdis/jir261. [DOI] [PubMed] [Google Scholar]
- 161.Vadeboncoeur N, Segura M, Al-Numani D, Vanier G, Gottschalk M. Pro-inflammatory cytokine and chemokine release by human brain microvascular endothelial cells stimulated by Streptococcus suis serotype 2. FEMS Immunol Med Microbiol. 2003;35:49–58. doi: 10.1111/j.1574-695X.2003.tb00648.x. [DOI] [PubMed] [Google Scholar]
- 162.Domínguez-Punaro MC, Segura M, Plante MM, Lacouture S, Rivest S, Gottschalk M. Streptococcus suis serotype 2, an important swine and human pathogen, induces strong systemic and cerebral inflammatory responses in a mouse model of infection. J Immunol. 2007;179:1842–54. doi: 10.4049/jimmunol.179.3.1842. [DOI] [PubMed] [Google Scholar]
- 163.Schwerk C, Adam R, Borkowski J, Schneider H, Klenk M, Zink S, Quednau N, Schmidt N, Stump C, Sagar A, et al. In vitro transcriptome analysis of porcine choroid plexus epithelial cells in response to Streptococcus suis: release of pro-inflammatory cytokines and chemokines. Microbes Infect. 2011;13:953–62. doi: 10.1016/j.micinf.2011.05.012. [DOI] [PubMed] [Google Scholar]
- 164.Zheng H, Punaro MC, Segura M, Lachance C, Rivest S, Xu J, Houde M, Gottschalk M. Toll-like receptor 2 is partially involved in the activation of murine astrocytes by Streptococcus suis, an important zoonotic agent of meningitis. J Neuroimmunol. 2011;234:71–83. doi: 10.1016/j.jneuroim.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 165.Graveline R, Segura M, Radzioch D, Gottschalk M. TLR2-dependent recognition of Streptococcus suis is modulated by the presence of capsular polysaccharide which modifies macrophage responsiveness. Int Immunol. 2007;19:375–89. doi: 10.1093/intimm/dxm003. [DOI] [PubMed] [Google Scholar]
- 166.de Greeff A, Benga L, Wichgers Schreur PJ, Valentin-Weigand P, Rebel JM, Smith HE. Involvement of NF-kappaB and MAP-kinases in the transcriptional response of alveolar macrophages to Streptococcus suis. Vet Microbiol. 2010;141:59–67. doi: 10.1016/j.vetmic.2009.07.031. [DOI] [PubMed] [Google Scholar]
- 167.Wu T, Yuan F, Chang H, Zhang L, Chen G, Tan C, Chen H, Bei W. Identification of a novel angiogenin inhibitor 1 and its association with hyaluronidase of Streptococcus suis serotype 2. Microb Pathog. 2010;49:32–7. doi: 10.1016/j.micpath.2010.03.002. [DOI] [PubMed] [Google Scholar]
- 168.Wisselink HJ, Stockhofe-Zurwieden N, Hilgers LA, Smith HE. Assessment of protective efficacy of live and killed vaccines based on a non-encapsulated mutant of Streptococcus suis serotype 2. Vet Microbiol. 2002;84:155–68. doi: 10.1016/S0378-1135(01)00452-7. [DOI] [PubMed] [Google Scholar]
- 169.Busque P, Higgins R, Caya F, Quessy S. Immunization of pigs against Streptococcus suis serotype 2 infection using a live avirulent strain. Can J Vet Res. 1997;61:275–9. [PMC free article] [PubMed] [Google Scholar]
- 170.Andresen LO, Tegtmeier C. Passive immunization of pigs against experimental infection with Streptococcus suis serotype 2. Vet Microbiol. 2001;81:331–44. doi: 10.1016/S0378-1135(01)00359-5. [DOI] [PubMed] [Google Scholar]
- 171.Wisselink HJ, Vecht U, Stockhofe-Zurwieden N, Smith HE. Protection of pigs against challenge with virulent Streptococcus suis serotype 2 strains by a muramidase-released protein and extracellular factor vaccine. Vet Rec. 2001;148:473–7. doi: 10.1136/vr.148.15.473. [DOI] [PubMed] [Google Scholar]
- 172.Liu L, Cheng G, Wang C, Pan X, Cong Y, Pan Q, Wang J, Zheng F, Hu F, Tang J. Identification and experimental verification of protective antigens against Streptococcus suis serotype 2 based on genome sequence analysis. Curr Microbiol. 2009;58:11–7. doi: 10.1007/s00284-008-9258-x. [DOI] [PubMed] [Google Scholar]
- 173.Li J, Xia J, Tan C, Zhou Y, Wang Y, Zheng C, Chen H, Bei W. Evaluation of the immunogenicity and the protective efficacy of a novel identified immunogenic protein, SsPepO, of Streptococcus suis serotype 2. Vaccine. 2011;29:6514–9. doi: 10.1016/j.vaccine.2011.07.010. [DOI] [PubMed] [Google Scholar]
- 174.Chen B, Zhang A, Li R, Mu X, He H, Chen H, Jin M. Evaluation of the protective efficacy of a newly identified immunogenic protein, HP0272, of Streptococcus suis. FEMS Microbiol Lett. 2010;307:12–8. doi: 10.1111/j.1574-6968.2010.01944.x. [DOI] [PubMed] [Google Scholar]
- 175.Dragojlović J, Milosević B, Sasić N, Pelemis M, Sasić M. [Streptococcus suis infection--clinical manifestations] Med Pregl. 2005;58:236–9. doi: 10.2298/MPNS0506236D. [DOI] [PubMed] [Google Scholar]
- 176.Hoa NT, Chieu TT, Nghia HD, Mai NT, Anh PH, Wolbers M, Baker S, Campbell JI, Chau NV, Hien TT, et al. The antimicrobial resistance patterns and associated determinants in Streptococcus suis isolated from humans in southern Vietnam, 1997-2008. BMC Infect Dis. 2011;11:6. doi: 10.1186/1471-2334-11-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Nghia HD, Tu TP, Wolbers M, Thai CQ, Hoang NV, Nga TV, Thao TP, Phu NH, Chau TT, Sinh DX, et al. Risk factors of Streptococcus suis infection in Vietnam. A case-control study. PLoS One. 2011;6:e17604. doi: 10.1371/journal.pone.0017604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Ngo TH, Tran TB, Tran TT, Nguyen VD, Campbell J, Pham HA, Huynh HT, Nguyen VV, Bryant JE, Tran TH, et al. Slaughterhouse pigs are a major reservoir of Streptococcus suis serotype 2 capable of causing human infection in southern Vietnam. PLoS One. 2011;6:e17943. doi: 10.1371/journal.pone.0017943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Coolen L, Dens J, Baeck E, Claes C, Lins RL, Verbraeken H, Daelemans R. Streptococcus suis meningitis, permanent perceptive deafness and endophthalmitis. Intensive Care Med. 1989;15:545. [PubMed] [Google Scholar]
- 180.Watkins EJ, Brooksby P, Schweiger MS, Enright SM. Septicaemia in a pig-farm worker. Lancet. 2001;357:38. doi: 10.1016/S0140-6736(00)03570-4. [DOI] [PubMed] [Google Scholar]
- 181.Arend SM, van Buchem MA, van Ogtrop ML, Thompson J. Septicaemia, meningitis and spondylodiscitis caused by Streptococcus suis type 2. Infection. 1995;23:128. doi: 10.1007/BF01833885. [DOI] [PubMed] [Google Scholar]
- 182.Maher D. Streptococcus suis septicaemia presenting as severe acute gastro-enteritis. J Infect. 1991;22:303–4. doi: 10.1016/S0163-4453(05)80022-2. [DOI] [PubMed] [Google Scholar]
- 183.Bergdoll MS, Crass BA, Reiser RF, Robbins RN, Davis JP. A new staphylococcal enterotoxin, enterotoxin F, associated with toxic-shock-syndrome Staphylococcus aureus isolates. Lancet. 1981;1:1017–21. doi: 10.1016/S0140-6736(81)92186-3. [DOI] [PubMed] [Google Scholar]
- 184.Cohen ML, Falkow S. Protein antigens from Staphylococcus aureus strains associated with toxic-shock syndrome. Science. 1981;211:842–4. doi: 10.1126/science.7466361. [DOI] [PubMed] [Google Scholar]
- 185.Bassaris HP, Venezio FR, Morlock BA, Phair JP. Staphylococcus aureus in toxic-shock syndrome. J Infect Dis. 1981;144:386–7. doi: 10.1093/infdis/144.4.386. [DOI] [PubMed] [Google Scholar]
- 186.Schutzer SE, Fischetti VA, Zabriskie JB. Toxic shock syndrome and lysogeny in Staphylococcus aureus. Science. 1983;220:316–8. doi: 10.1126/science.6220467. [DOI] [PubMed] [Google Scholar]
- 187.Leelarasamee A, Nilakul C, Tien-Grim S, Srifuengfung S, Susaengrat W. Streptococcus suis toxic-shock syndrome and meningitis. J Med Assoc Thai. 1997;80:63–8. [PubMed] [Google Scholar]
- 188.François B, Gissot V, Ploy MC, Vignon P. Recurrent septic shock due to Streptococcus suis. J Clin Microbiol. 1998;36:2395. doi: 10.1128/jcm.36.8.2395-2395.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Poggenborg R, Gaïni S, Kjaeldgaard P, Christensen JJ. Streptococcus suis: meningitis, spondylodiscitis and bacteraemia with a serotype 14 strain. Scand J Infect Dis. 2008;40:346–9. doi: 10.1080/00365540701716825. [DOI] [PubMed] [Google Scholar]
- 190.Feng Y, Zhang H. M. C, Wang C. Regulation of Virulence in Streptococcus suis. J Bacteriol Parasitol. 2012;3:e108. [Google Scholar]
- 191.Zheng X, Zheng H, Lan R, Ye C, Wang Y, Zhang J, Jing H, Chen C, Segura M, Gottschalk M, et al. Identification of genes and genomic islands correlated with high pathogenicity in Streptococcus suis using whole genome tiling microarrays. PLoS One. 2011;6:e17987. doi: 10.1371/journal.pone.0017987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wu Z, Li M, Wang C, Li J, Lu N, Zhang R, Jiang Y, Yang R, Liu C, Liao H, et al. Probing genomic diversity and evolution of Streptococcus suis serotype 2 by NimbleGen tiling arrays. BMC Genomics. 2011;12:219. doi: 10.1186/1471-2164-12-219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Kataoka Y, Sugimoto C, Nakazawa M, Kashiwazaki M. Detection of Streptococcus suis type 2 in tonsils of slaughtered pigs using improved selective and differential media. Vet Microbiol. 1991;28:335–42. doi: 10.1016/0378-1135(91)90068-Q. [DOI] [PubMed] [Google Scholar]
- 194.Okwumabua O, O’Connor M, Shull E. A polymerase chain reaction (PCR) assay specific for Streptococcus suis based on the gene encoding the glutamate dehydrogenase. FEMS Microbiol Lett. 2003;218:79–84. doi: 10.1111/j.1574-6968.2003.tb11501.x. [DOI] [PubMed] [Google Scholar]
- 195.Swildens B, Wisselink HJ, Engel B, Smith HE, Nielen M, Verheijden JH, Stegeman JA. Detection of extracellular factor-positive Streptococcus suis serotype 2 strains in tonsillar swabs of live sows by PCR. Vet Microbiol. 2005;109:223–8. doi: 10.1016/j.vetmic.2005.04.024. [DOI] [PubMed] [Google Scholar]
- 196.Kang I, Kim D, Han K, Seo HW, Oh Y, Park C, Lee J, Gottschalk M, Chae C. Optimized protocol for multiplex nested polymerase chain reaction to detect and differentiate Haemophilus parasuis, Streptococcus suis, and Mycoplasma hyorhinis in formalin-fixed, paraffin-embedded tissues from pigs with polyserositis. Can J Vet Res. 2012;76:195–200. [PMC free article] [PubMed] [Google Scholar]
- 197.Huy NT, Hang TT, Boamah D, Lan NT, Van Thanh P, Watanabe K, Huong VT, Kikuchi M, Ariyoshi K, Morita K, et al. Development of a single-tube loop-mediated isothermal amplification assay for detection of four pathogens of bacterial meningitis. FEMS Microbiol Lett. 2012;337:25–30. doi: 10.1111/1574-6968.12002. [DOI] [PubMed] [Google Scholar]
- 198.Zhang J, Zhu J, Ren H, Zhu S, Zhao P, Zhang F, Lv H, Hu D, Hao L, Geng M, et al. Rapid visual detection of highly pathogenic Streptococcus suis serotype 2 isolates by use of loop-mediated isothermal amplification. J Clin Microbiol. 2013;51:3250–6. doi: 10.1128/JCM.01183-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Nga TV, Nghia HD, Tu TP, Diep TS, Mai NT, Chau TT, Sinh DX, Phu NH, Nga TT, Chau NV, et al. Real-time PCR for detection of Streptococcus suis serotype 2 in cerebrospinal fluid of human patients with meningitis. Diagn Microbiol Infect Dis. 2011;70:461–7. doi: 10.1016/j.diagmicrobio.2010.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Yang W, Cai X, Hao Y, Liu Y, Wang S, Xing R, Gu J, Li C, Yue X, Yuan C, et al. Characterization of Streptococcus suis serotype 2 blood infections using RT-qPCR to quantify glutamate dehydrogenase copy numbers. J Microbiol Methods. 2010;83:326–9. doi: 10.1016/j.mimet.2010.09.013. [DOI] [PubMed] [Google Scholar]
- 201.Wisselink HJ, Reek FH, Vecht U, Stockhofe-Zurwieden N, Smits MA, Smith HE. Detection of virulent strains of Streptococcus suis type 2 and highly virulent strains of Streptococcus suis type 1 in tonsillar specimens of pigs by PCR. Vet Microbiol. 1999;67:143–57. doi: 10.1016/S0378-1135(99)00036-X. [DOI] [PubMed] [Google Scholar]
- 202.Gottschalk M, Lebrun A, Wisselink H, Dubreuil JD, Smith H, Vecht U. Production of virulence-related proteins by Canadian strains of Streptococcus suis capsular type 2. Can J Vet Res. 1998;62:75–9. [PMC free article] [PubMed] [Google Scholar]
- 203.Wisselink HJ, Joosten JJ, Smith HE. Multiplex PCR assays for simultaneous detection of six major serotypes and two virulence-associated phenotypes of Streptococcus suis in tonsillar specimens from pigs. J Clin Microbiol. 2002;40:2922–9. doi: 10.1128/JCM.40.8.2922-2929.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Berthelot-Hérault F, Morvan H, Kéribin AM, Gottschalk M, Kobisch M. Production of muraminidase-released protein (MRP), extracellular factor (EF) and suilysin by field isolates of Streptococcus suis capsular types 2, 1/2, 9, 7 and 3 isolated from swine in France. Vet Res. 2000;31:473–9. doi: 10.1051/vetres:2000133. [DOI] [PubMed] [Google Scholar]
- 205.Martinez G, Pestana de Castro AF, Ribeiro Pagnani KJ, Nakazato G, Dias da Silveira W, Gottschalk M. Clonal distribution of an atypical MRP+, EF*, and suilysin+ phenotype of virulent Streptococcus suis serotype 2 strains in Brazil. Can J Vet Res. 2003;67:52–5. [PMC free article] [PubMed] [Google Scholar]
- 206.Silva LM, Baums CG, Rehm T, Wisselink HJ, Goethe R, Valentin-Weigand P. Virulence-associated gene profiling of Streptococcus suis isolates by PCR. Vet Microbiol. 2006;115:117–27. doi: 10.1016/j.vetmic.2005.12.013. [DOI] [PubMed] [Google Scholar]
- 207.Liu Z, Zheng H, Gottschalk M, Bai X, Lan R, Ji S, Liu H, Xu J. Development of multiplex PCR assays for the identification of the 33 serotypes of Streptococcus suis. PLoS One. 2013;8:e72070. doi: 10.1371/journal.pone.0072070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Marois C, Bougeard S, Gottschalk M, Kobisch M. Multiplex PCR assay for detection of Streptococcus suis species and serotypes 2 and 1/2 in tonsils of live and dead pigs. J Clin Microbiol. 2004;42:3169–75. doi: 10.1128/JCM.42.7.3169-3175.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Okwumabua O, Staats J, Chengappa MM. Detection of genomic heterogeneity in Streptococcus suis isolates by DNA restriction fragment length polymorphisms of rRNA genes (ribotyping) J Clin Microbiol. 1995;33:968–72. doi: 10.1128/jcm.33.4.968-972.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Marois C, Le Devendec L, Gottschalk M, Kobisch M. Detection and molecular typing of Streptococcus suis in tonsils from live pigs in France. Can J Vet Res. 2007;71:14–22. [PMC free article] [PubMed] [Google Scholar]
- 211.Luey CK, Chu YW, Cheung TK, Law CC, Chu MY, Cheung DT, Kam KM. Rapid pulsed-field gel electrophoresis protocol for subtyping of Streptococcus suis serotype 2. J Microbiol Methods. 2007;68:648–50. doi: 10.1016/j.mimet.2006.10.010. [DOI] [PubMed] [Google Scholar]
- 212.Wang LL, Ye CY, Xu YM, Cui ZG, Jing HQ, Jin D, Du HM, Zhang SY, Bai XM, Zhao AL, et al. [Development of a protocol on pulsed field gel electrophoresis analysis for Streptococcus suis.] Zhonghua Liu Xing Bing Xue Za Zhi. 2008;29:473–7. [PubMed] [Google Scholar]
- 213.Blume V, Luque I, Vela AI, Borge C, Maldonado A, Domínguez L, Tarradas C, Fernández-Garayzábal JF. Genetic and virulence-phenotype characterization of serotypes 2 and 9 of Streptococcus suis swine isolates. Int Microbiol. 2009;12:161–6. [PubMed] [Google Scholar]
- 214.Luque I, Blume V, Borge C, Vela AI, Perea JA, Márquez JM, Fernández-Garayzábal JF, Tarradas C. Genetic analysis of Streptococcus suis isolates recovered from diseased and healthy carrier pigs at different stages of production on a pig farm. Vet J. 2010;186:396–8. doi: 10.1016/j.tvjl.2009.09.005. [DOI] [PubMed] [Google Scholar]
- 215.Chang B, Wada A, Ikebe T, Ohnishi M, Mita K, Endo M, Matsuo H, Asatuma Y, Kuramoto S, Sekiguchi H, et al. Characteristics of Streptococcus suis isolated from patients in Japan. Jpn J Infect Dis. 2006;59:397–9. [PubMed] [Google Scholar]
- 216.Vela AI, Goyache J, Tarradas C, Luque I, Mateos A, Moreno MA, Borge C, Perea JA, Domínguez L, Fernández-Garayzábal JF. Analysis of genetic diversity of Streptococcus suis clinical isolates from pigs in Spain by pulsed-field gel electrophoresis. J Clin Microbiol. 2003;41:2498–502. doi: 10.1128/JCM.41.6.2498-2502.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Berthelot-Hérault F, Marois C, Gottschalk M, Kobisch M. Genetic diversity of Streptococcus suis strains isolated from pigs and humans as revealed by pulsed-field gel electrophoresis. J Clin Microbiol. 2002;40:615–9. doi: 10.1128/JCM.40.2.615-6192002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Marois C, Le Devendec L, Gottschalk M, Kobisch M. Molecular characterization of Streptococcus suis strains by 16S-23S intergenic spacer polymerase chain reaction and restriction fragment length polymorphism analysis. Can J Vet Res. 2006;70:94–104. [PMC free article] [PubMed] [Google Scholar]
- 219.Wang HM, Ke CW, Pan WB, Ke BX, Chen JD, Deng XL, Liu MZ, Chen GR, Yang XF, Zhu ZY. [MLST typing of Streptococcus suis isolated from clinical patients in Guangdong Province in 2005] Nan Fang Yi Ke Da Xue Xue Bao. 2008;28:1438–41. [PubMed] [Google Scholar]
- 220.Chen L, Song Y, Wei Z, He H, Zhang A, Jin M. Antimicrobial susceptibility, tetracycline and erythromycin resistance genes, and multilocus sequence typing of Streptococcus suis isolates from diseased pigs in China. J Vet Med Sci. 2013;75:583, 7. doi: 10.1292/jvms.12-0279. [DOI] [PubMed] [Google Scholar]
- 221.King SJ, Leigh JA, Heath PJ, Luque I, Tarradas C, Dowson CG, Whatmore AM. Development of a multilocus sequence typing scheme for the pig pathogen Streptococcus suis: identification of virulent clones and potential capsular serotype exchange. J Clin Microbiol. 2002;40:3671–80. doi: 10.1128/JCM.40.10.3671-3680.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Li W, Ye C, Jing H, Cui Z, Bai X, Jin D, Zheng H, Zhao A, Xu Y, Gottschalk M, et al. Streptococcus suis outbreak investigation using multiple-locus variable tandem repeat number analysis. Microbiol Immunol. 2010;54:380–8. doi: 10.1111/j.1348-0421.2010.00228.x. [DOI] [PubMed] [Google Scholar]
- 223.Chatellier S, Gottschalk M, Higgins R, Brousseau R, Harel J. Relatedness of Streptococcus suis serotype 2 isolates from different geographic origins as evaluated by molecular fingerprinting and phenotyping. J Clin Microbiol. 1999;37:362–6. doi: 10.1128/jcm.37.2.362-366.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Madsen LW, Boye M, Jensen HE. An enzyme-based in situ hybridisation method for the identification of Streptococcus suis. APMIS. 2001;109:665–9. doi: 10.1034/j.1600-0463.2001.d01-130.x. [DOI] [PubMed] [Google Scholar]
- 225.Boye M, Feenstra AA, Tegtmeier C, Andresen LO, Rasmussen SR, Bille-Hansen V. Detection of Streptococcus suis by in situ hybridization, indirect immunofluorescence, and peroxidase-antiperoxidase assays in formalin-fixed, paraffin-embedded tissue sections from pigs. J Vet Diagn Invest. 2000;12:224–32. doi: 10.1177/104063870001200305. [DOI] [PubMed] [Google Scholar]
- 226.Vecht U, Wisselink HJ, Anakotta J, Smith HE. Discrimination between virulent and nonvirulent Streptococcus suis type 2 strains by enzyme-linked immunosorbent assay. Vet Microbiol. 1993;34:71–82. doi: 10.1016/0378-1135(93)90008-U. [DOI] [PubMed] [Google Scholar]
- 227.del Campo Sepúlveda EM, Altman E, Kobisch M, D’Allaire S, Gottschalk M. Detection of antibodies against Streptococcus suis capsular type 2 using a purified capsular polysaccharide antigen-based indirect ELISA. Vet Microbiol. 1996;52:113–25. doi: 10.1016/0378-1135(96)00056-9. [DOI] [PubMed] [Google Scholar]
- 228.Kataoka Y, Yamashita T, Sunaga S, Imada Y, Ishikawa H, Kishima M, Nakazawa M. An enzyme-linked immunosorbent assay (ELISA) for the detection of antibody against Streptococcus suis type 2 in infected pigs. J Vet Med Sci. 1996;58:369–72. doi: 10.1292/jvms.58.369. [DOI] [PubMed] [Google Scholar]
- 229.Chen K, Han H, Luo Z. Streptococcus suis II immunoassay based on thorny gold nanoparticles and surface enhanced Raman scattering. Analyst. 2012;137:1259–64. doi: 10.1039/c2an15997j. [DOI] [PubMed] [Google Scholar]
- 230.Du H, Huang W, Xie H, Ye C, Jing H, Ren Z, Xu J. The genetically modified suilysin, rSLY(P353L), provides a candidate vaccine that suppresses proinflammatory response and reduces fatality following infection with Streptococcus suis. Vaccine. 2013;31:4209–15. doi: 10.1016/j.vaccine.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 231.Takamatsu D, Osaki M, Tharavichitkul P, Takai S, Sekizaki T. Allelic variation and prevalence of serum opacity factor among the Streptococcus suis population. J Med Microbiol. 2008;57:488–94. doi: 10.1099/jmm.0.47755-0. [DOI] [PubMed] [Google Scholar]
- 232.Li W, Hu X, Liu L, Chen H, Zhou R. Induction of protective immune response against Streptococcus suis serotype 2 infection by the surface antigen HP0245. FEMS Microbiol Lett. 2011;316:115–22. doi: 10.1111/j.1574-6968.2010.02200.x. [DOI] [PubMed] [Google Scholar]
- 233.de Buhr N, Neumann A, Jerjomiceva N, von Köckritz-Blickwede M, Baums CG. Streptococcus suis DNase SsnA contributes to degradation of neutrophil extracellular traps (NETs) and evasion of NET-mediated antimicrobial activity. Microbiology. 2014;160:385–95. doi: 10.1099/mic.0.072199-0. [DOI] [PubMed] [Google Scholar]
- 234.Segura M, Vadeboncoeur N, Gottschalk M. CD14-dependent and -independent cytokine and chemokine production by human THP-1 monocytes stimulated by Streptococcus suis capsular type 2. Clin Exp Immunol. 2002;127:243–54. doi: 10.1046/j.1365-2249.2002.01768.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Vanier G, Segura M, Lecours MP, Grenier D, Gottschalk M. Porcine brain microvascular endothelial cell-derived interleukin-8 is first induced and then degraded by Streptococcus suis. Microb Pathog. 2009;46:135–43. doi: 10.1016/j.micpath.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 236.Lachance C, Segura M, Dominguez-Punaro MC, Wojewodka G, De Sanctis JB, Radzioch D, Gottschalk M. Deregulated balance of omega-6 and omega-3 following infection by the zoonotic pathogen Streptococcus suis. Infect Immun. 2014(forthcoming doi: 10.1128/IAI.01524-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Huang D, Zhu H, Lin H, Xu J, Lu C. First insights into the protective effects of a recombinant swinepox virus expressing truncated MRP of Streptococcus suis type 2 in mice. Berl Munch Tierarztl Wochenschr. 2012;125:144–52. [PubMed] [Google Scholar]
- 238.Tan C, Liu M, Liu J, Yuan F, Fu S, Liu Y, Jin M, Bei W, Chen H. Vaccination with Streptococcus suis serotype 2 recombinant 6PGD protein provides protection against S. suis infection in swine. FEMS Microbiol Lett. 2009;296:78–83. doi: 10.1111/j.1574-6968.2009.01617.x. [DOI] [PubMed] [Google Scholar]
- 239.Li W, Hu X, Liu L, Chen H, Zhou R. Induction of protective immune response against Streptococcus suis serotype 2 infection by the surface antigen HP0245. FEMS Microbiol Lett. 2011;316:115–22. doi: 10.1111/j.1574-6968.2010.02200.x. [DOI] [PubMed] [Google Scholar]
- 240.Zhang YM, Shao ZQ, Wang J, Wang L, Li X, Wang C, Tang J, Pan X. Prevalent distribution and conservation of Streptococcus suis Lmb protein and its protective capacity against the Chinese highly virulent strain infection. Microbiol Res. 2013 doi: 10.1016/j.micres.2013.09.007. forthcoming. [DOI] [PubMed] [Google Scholar]