Abstract
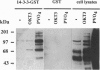
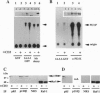
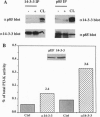
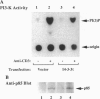
Proteins of the 14-3-3 family can associate with, and/or modulate the activity of, several protooncogene and oncogene products and, thus, are implicated in regulation of signaling pathways. We report that 14-3-3 is associated with another important transducing enzyme, phosphatidylinositol 3-kinase (PI3-K). A recombinant 14-3-3 fusion protein bound several tyrosine-phosphorylated proteins from antigen receptor-stimulated T lymphocytes. PI3-K was identified by immunoblotting and enzymatic assays as one of the 14-3-3-binding proteins in resting or activated cells. Moreover, endogenous 14-3-3 and PI3-K were coimmunoprecipitated from intact T cells. Far-Western blots of gel-purified, immunoprecipitated PI3-K with a recombinant 14-3-3 fusion protein revealed direct binding of 14-3-3 to the catalytic subunit (p110) of PI3-K. Finally, anti-phosphotyrosine immunoprecipitates from activated, 14-3-3-overexpressing cells contained lower PI3-K enzymatic activity than similar immunoprecipitates from control cells. These findings suggest that association of 14-3-3 with PI3-K in hematopoietic (and possibly other) cells regulates the enzymatic activity of PI3-K during receptor-initiated signal transduction.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aitken A. 14-3-3 proteins on the MAP. Trends Biochem Sci. 1995 Mar;20(3):95–97. doi: 10.1016/s0968-0004(00)88971-9. [DOI] [PubMed] [Google Scholar]
- Auger K. R., Cantley L. C. Novel polyphosphoinositides in cell growth and activation. Cancer Cells. 1991 Jul;3(7):263–270. [PubMed] [Google Scholar]
- Bohen S. P., Kralli A., Yamamoto K. R. Hold 'em and fold 'em: chaperones and signal transduction. Science. 1995 Jun 2;268(5215):1303–1304. doi: 10.1126/science.7761850. [DOI] [PubMed] [Google Scholar]
- Burbelo P. D., Hall A. 14-3-3 proteins. Hot numbers in signal transduction. Curr Biol. 1995 Feb 1;5(2):95–96. doi: 10.1016/s0960-9822(95)00022-4. [DOI] [PubMed] [Google Scholar]
- Davis R. J. The mitogen-activated protein kinase signal transduction pathway. J Biol Chem. 1993 Jul 15;268(20):14553–14556. [PubMed] [Google Scholar]
- Fantl W. J., Muslin A. J., Kikuchi A., Martin J. A., MacNicol A. M., Gross R. W., Williams L. T. Activation of Raf-1 by 14-3-3 proteins. Nature. 1994 Oct 13;371(6498):612–614. doi: 10.1038/371612a0. [DOI] [PubMed] [Google Scholar]
- Finney R. E., Robbins S. M., Bishop J. M. Association of pRas and pRaf-1 in a complex correlates with activation of a signal transduction pathway. Curr Biol. 1993 Dec 1;3(12):805–812. doi: 10.1016/0960-9822(93)90214-9. [DOI] [PubMed] [Google Scholar]
- Freed E., Symons M., Macdonald S. G., McCormick F., Ruggieri R. Binding of 14-3-3 proteins to the protein kinase Raf and effects on its activation. Science. 1994 Sep 16;265(5179):1713–1716. doi: 10.1126/science.8085158. [DOI] [PubMed] [Google Scholar]
- Fu H., Coburn J., Collier R. J. The eukaryotic host factor that activates exoenzyme S of Pseudomonas aeruginosa is a member of the 14-3-3 protein family. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2320–2324. doi: 10.1073/pnas.90.6.2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu H., Xia K., Pallas D. C., Cui C., Conroy K., Narsimhan R. P., Mamon H., Collier R. J., Roberts T. M. Interaction of the protein kinase Raf-1 with 14-3-3 proteins. Science. 1994 Oct 7;266(5182):126–129. doi: 10.1126/science.7939632. [DOI] [PubMed] [Google Scholar]
- Fukui Y., Hanafusa H. Phosphatidylinositol kinase activity associates with viral p60src protein. Mol Cell Biol. 1989 Apr;9(4):1651–1658. doi: 10.1128/mcb.9.4.1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukui Y., Hanafusa H. Requirement of phosphatidylinositol-3 kinase modification for its association with p60src. Mol Cell Biol. 1991 Apr;11(4):1972–1979. doi: 10.1128/mcb.11.4.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furukawa Y., Ikuta N., Omata S., Yamauchi T., Isobe T., Ichimura T. Demonstration of the phosphorylation-dependent interaction of tryptophan hydroxylase with the 14-3-3 protein. Biochem Biophys Res Commun. 1993 Jul 15;194(1):144–149. doi: 10.1006/bbrc.1993.1796. [DOI] [PubMed] [Google Scholar]
- Hayashi H., Nishioka Y., Kamohara S., Kanai F., Ishii K., Fukui Y., Shibasaki F., Takenawa T., Kido H., Katsunuma N. The alpha-type 85-kDa subunit of phosphatidylinositol 3-kinase is phosphorylated at tyrosines 368, 580, and 607 by the insulin receptor. J Biol Chem. 1993 Apr 5;268(10):7107–7117. [PubMed] [Google Scholar]
- Irie K., Gotoh Y., Yashar B. M., Errede B., Nishida E., Matsumoto K. Stimulatory effects of yeast and mammalian 14-3-3 proteins on the Raf protein kinase. Science. 1994 Sep 16;265(5179):1716–1719. doi: 10.1126/science.8085159. [DOI] [PubMed] [Google Scholar]
- Isobe T., Hiyane Y., Ichimura T., Okuyama T., Takahashi N., Nakajo S., Nakaya K. Activation of protein kinase C by the 14-3-3 proteins homologous with Exo1 protein that stimulates calcium-dependent exocytosis. FEBS Lett. 1992 Aug 17;308(2):121–124. doi: 10.1016/0014-5793(92)81257-m. [DOI] [PubMed] [Google Scholar]
- Johnson G. L., Vaillancourt R. R. Sequential protein kinase reactions controlling cell growth and differentiation. Curr Opin Cell Biol. 1994 Apr;6(2):230–238. doi: 10.1016/0955-0674(94)90141-4. [DOI] [PubMed] [Google Scholar]
- Kavanaugh W. M., Klippel A., Escobedo J. A., Williams L. T. Modification of the 85-kilodalton subunit of phosphatidylinositol-3 kinase in platelet-derived growth factor-stimulated cells. Mol Cell Biol. 1992 Aug;12(8):3415–3424. doi: 10.1128/mcb.12.8.3415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura Y., Yahara I., Lindquist S. Role of the protein chaperone YDJ1 in establishing Hsp90-mediated signal transduction pathways. Science. 1995 Jun 2;268(5215):1362–1365. doi: 10.1126/science.7761857. [DOI] [PubMed] [Google Scholar]
- Kodaki T., Woscholski R., Hallberg B., Rodriguez-Viciana P., Downward J., Parker P. J. The activation of phosphatidylinositol 3-kinase by Ras. Curr Biol. 1994 Sep 1;4(9):798–806. doi: 10.1016/s0960-9822(00)00177-9. [DOI] [PubMed] [Google Scholar]
- Li S., Janosch P., Tanji M., Rosenfeld G. C., Waymire J. C., Mischak H., Kolch W., Sedivy J. M. Regulation of Raf-1 kinase activity by the 14-3-3 family of proteins. EMBO J. 1995 Feb 15;14(4):685–696. doi: 10.1002/j.1460-2075.1995.tb07047.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Marengere L. E., Koch C. A., Pawson T. The v-Src SH3 domain binds phosphatidylinositol 3'-kinase. Mol Cell Biol. 1993 Sep;13(9):5225–5232. doi: 10.1128/mcb.13.9.5225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. C., Kawagishi M., Mikayama T., Inagaki Y., Takeuchi T., Ohashi H. Processing of a fusion protein by endoprotease in COS-1 cells for secretion of mature peptide by using a chimeric expression vector. Proc Natl Acad Sci U S A. 1993 Oct 1;90(19):8957–8961. doi: 10.1073/pnas.90.19.8957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Y., Phillips C. A., Trevillyan J. M. Phosphatidylinositol 3-kinase activity is not essential for CD28 costimulatory activity in Jurkat T cells: studies with a selective inhibitor, wortmannin. Eur J Immunol. 1995 Feb;25(2):533–537. doi: 10.1002/eji.1830250234. [DOI] [PubMed] [Google Scholar]
- Michaud N. R., Fabian J. R., Mathes K. D., Morrison D. K. 14-3-3 is not essential for Raf-1 function: identification of Raf-1 proteins that are biologically activated in a 14-3-3- and Ras-independent manner. Mol Cell Biol. 1995 Jun;15(6):3390–3397. doi: 10.1128/mcb.15.6.3390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moodie S. A., Willumsen B. M., Weber M. J., Wolfman A. Complexes of Ras.GTP with Raf-1 and mitogen-activated protein kinase kinase. Science. 1993 Jun 11;260(5114):1658–1661. doi: 10.1126/science.8503013. [DOI] [PubMed] [Google Scholar]
- Morrison D. 14-3-3: modulators of signaling proteins? Science. 1994 Oct 7;266(5182):56–57. doi: 10.1126/science.7939645. [DOI] [PubMed] [Google Scholar]
- Nielsen P. J. Primary structure of a human protein kinase regulator protein. Biochim Biophys Acta. 1991 Mar 26;1088(3):425–428. doi: 10.1016/0167-4781(91)90136-a. [DOI] [PubMed] [Google Scholar]
- O'Shea J. J., McVicar D. W., Bailey T. L., Burns C., Smyth M. J. Activation of human peripheral blood T lymphocytes by pharmacological induction of protein-tyrosine phosphorylation. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10306–10310. doi: 10.1073/pnas.89.21.10306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pallas D. C., Fu H., Haehnel L. C., Weller W., Collier R. J., Roberts T. M. Association of polyomavirus middle tumor antigen with 14-3-3 proteins. Science. 1994 Jul 22;265(5171):535–537. doi: 10.1126/science.8036498. [DOI] [PubMed] [Google Scholar]
- Panayotou G., Waterfield M. D. Phosphatidyl-inositol 3-kinase: a key enzyme in diverse signalling processes. Trends Cell Biol. 1992 Dec;2(12):358–360. doi: 10.1016/0962-8924(92)90042-l. [DOI] [PubMed] [Google Scholar]
- Pleiman C. M., Hertz W. M., Cambier J. C. Activation of phosphatidylinositol-3' kinase by Src-family kinase SH3 binding to the p85 subunit. Science. 1994 Mar 18;263(5153):1609–1612. doi: 10.1126/science.8128248. [DOI] [PubMed] [Google Scholar]
- Prasad K. V., Kapeller R., Janssen O., Repke H., Duke-Cohan J. S., Cantley L. C., Rudd C. E. Phosphatidylinositol (PI) 3-kinase and PI 4-kinase binding to the CD4-p56lck complex: the p56lck SH3 domain binds to PI 3-kinase but not PI 4-kinase. Mol Cell Biol. 1993 Dec;13(12):7708–7717. doi: 10.1128/mcb.13.12.7708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reuther G. W., Fu H., Cripe L. D., Collier R. J., Pendergast A. M. Association of the protein kinases c-Bcr and Bcr-Abl with proteins of the 14-3-3 family. Science. 1994 Oct 7;266(5182):129–133. doi: 10.1126/science.7939633. [DOI] [PubMed] [Google Scholar]
- Robinson K., Jones D., Patel Y., Martin H., Madrazo J., Martin S., Howell S., Elmore M., Finnen M. J., Aitken A. Mechanism of inhibition of protein kinase C by 14-3-3 isoforms. 14-3-3 isoforms do not have phospholipase A2 activity. Biochem J. 1994 May 1;299(Pt 3):853–861. doi: 10.1042/bj2990853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Viciana P., Warne P. H., Dhand R., Vanhaesebroeck B., Gout I., Fry M. J., Waterfield M. D., Downward J. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature. 1994 Aug 18;370(6490):527–532. doi: 10.1038/370527a0. [DOI] [PubMed] [Google Scholar]
- Rutherford S. L., Zuker C. S. Protein folding and the regulation of signaling pathways. Cell. 1994 Dec 30;79(7):1129–1132. doi: 10.1016/0092-8674(94)90003-5. [DOI] [PubMed] [Google Scholar]
- Secrist J. P., Burns L. A., Karnitz L., Koretzky G. A., Abraham R. T. Stimulatory effects of the protein tyrosine phosphatase inhibitor, pervanadate, on T-cell activation events. J Biol Chem. 1993 Mar 15;268(8):5886–5893. [PubMed] [Google Scholar]
- Takebe Y., Seiki M., Fujisawa J., Hoy P., Yokota K., Arai K., Yoshida M., Arai N. SR alpha promoter: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and the R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol Cell Biol. 1988 Jan;8(1):466–472. doi: 10.1128/mcb.8.1.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Aelst L., Barr M., Marcus S., Polverino A., Wigler M. Complex formation between RAS and RAF and other protein kinases. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):6213–6217. doi: 10.1073/pnas.90.13.6213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogel L. B., Fujita D. J. The SH3 domain of p56lck is involved in binding to phosphatidylinositol 3'-kinase from T lymphocytes. Mol Cell Biol. 1993 Dec;13(12):7408–7417. doi: 10.1128/mcb.13.12.7408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vojtek A. B., Hollenberg S. M., Cooper J. A. Mammalian Ras interacts directly with the serine/threonine kinase Raf. Cell. 1993 Jul 16;74(1):205–214. doi: 10.1016/0092-8674(93)90307-c. [DOI] [PubMed] [Google Scholar]
- Warne P. H., Viciana P. R., Downward J. Direct interaction of Ras and the amino-terminal region of Raf-1 in vitro. Nature. 1993 Jul 22;364(6435):352–355. doi: 10.1038/364352a0. [DOI] [PubMed] [Google Scholar]
- Whitman M., Kaplan D. R., Schaffhausen B., Cantley L., Roberts T. M. Association of phosphatidylinositol kinase activity with polyoma middle-T competent for transformation. Nature. 1985 May 16;315(6016):239–242. doi: 10.1038/315239a0. [DOI] [PubMed] [Google Scholar]
- Yamamori B., Kuroda S., Shimizu K., Fukui K., Ohtsuka T., Takai Y. Purification of a Ras-dependent mitogen-activated protein kinase kinase kinase from bovine brain cytosol and its identification as a complex of B-Raf and 14-3-3 proteins. J Biol Chem. 1995 May 19;270(20):11723–11726. doi: 10.1074/jbc.270.20.11723. [DOI] [PubMed] [Google Scholar]
- Yano H., Nakanishi S., Kimura K., Hanai N., Saitoh Y., Fukui Y., Nonomura Y., Matsuda Y. Inhibition of histamine secretion by wortmannin through the blockade of phosphatidylinositol 3-kinase in RBL-2H3 cells. J Biol Chem. 1993 Dec 5;268(34):25846–25856. [PubMed] [Google Scholar]
- Zhang X. F., Settleman J., Kyriakis J. M., Takeuchi-Suzuki E., Elledge S. J., Marshall M. S., Bruder J. T., Rapp U. R., Avruch J. Normal and oncogenic p21ras proteins bind to the amino-terminal regulatory domain of c-Raf-1. Nature. 1993 Jul 22;364(6435):308–313. doi: 10.1038/364308a0. [DOI] [PubMed] [Google Scholar]
- von Willebrand M., Baier G., Couture C., Burn P., Mustelin T. Activation of phosphatidylinositol-3-kinase in Jurkat T cells depends on the presence of the p56lck tyrosine kinase. Eur J Immunol. 1994 Jan;24(1):234–238. doi: 10.1002/eji.1830240137. [DOI] [PubMed] [Google Scholar]