Abstract
Background
In order to get global molecular understanding of one of the most important crop diseases worldwide, we investigated compatible and incompatible interactions between Phytophthora infestans and potato (Solanum tuberosum). We used the two most field-resistant potato clones under Swedish growing conditions, which have the greatest known local diversity of P. infestans populations, and a reference compatible cultivar.
Results
Quantitative label-free proteomics of 51 apoplastic secretome samples (PXD000435) in combination with genome-wide transcript analysis by 42 microarrays (E-MTAB-1515) were used to capture changes in protein abundance and gene expression at 6, 24 and 72 hours after inoculation with P. infestans. To aid mass spectrometry analysis we generated cultivar-specific RNA-seq data (E-MTAB-1712), which increased peptide identifications by 17%. Components induced only during incompatible interactions, which are candidates for hypersensitive response initiation, include a Kunitz-like protease inhibitor, transcription factors and an RCR3-like protein. More secreted proteins had lower abundance in the compatible interaction compared to the incompatible interactions. Based on this observation and because the well-characterized effector-target C14 protease follows this pattern, we suggest 40 putative effector targets.
Conclusions
In summary, over 17000 transcripts and 1000 secreted proteins changed in abundance in at least one time point, illustrating the dynamics of plant responses to a hemibiotroph. Half of the differentially abundant proteins showed a corresponding change at the transcript level. Many putative hypersensitive and effector-target proteins were single representatives of large gene families.
Electronic supplementary material
The online version of this article (doi:10.1186/1471-2164-15-497) contains supplementary material, which is available to authorized users.
Keywords: Potato, Desiree, Sarpo Mira, SW93-1015, Secretome, Apoplast, Resistance, Hypersensitive response, Phytophthora infestans
Background
The oomycete Phytophthora infestans, the cause of potato (Solanum tuberosum) late blight disease, is one of the most confounding plant pathogens. Despite over a century of resistance breeding, fungicide use, and other control measures, it is still a major threat to sustainable food production worldwide. P. infestans is responsible for global annual costs of at least €5.6 billion in control measures and crop losses and is especially a threat to farmers’ income and food security in developing countries [1]. In addition, new regulations designed to reduce threats to the environment will limit the availability of fungicides, and conditions favoring overwintering spores due to the effects of global warming, are expected to increase problems with this disease [2].
P. infestans has a hemibiotrophic life cycle [3] and bi-phasic growth, with an initial biotrophic phase and subsequent necrotrophic phase of infection [3, 4]. The successful suppression of plant defense by effector molecules leads to a compatible interaction between plant and pathogen, which is referred to as effector-triggered susceptibility (ETS) [5]. A large number of extracellular and cytoplasmic effectors in the P. infestans genome have been identified [6, 7] and increasing evidence for their role in establishing ETS exists. However, knowledge about their targets in the plant host is scarce, mainly because of the limited availability of hemibiotrophic pathogens with susceptible interactions with the model species Arabidopsis [8]. Some of the P. infestans extracellular effectors are inhibitors that target defense-related proteins such as proteases and glucanases, and processes such as programmed cell death in plants. For example, Kazal-like extracellular serine protease inhibitors EPI1 and EPI10 inhibit the P69B subtilisin-like serine protease in tomato [9, 10], while others such as the cystatin-like protein target cysteine-proteases [11] and EPIC1 and EPIC2B inhibit papain-like cysteine protease RCR3, in the same plant [12]. These latter two inhibitors also target cysteine protease C14 by direct binding in the extracellular compartment [13]. In addition to extracellular inhibition of C14 by EPIC1 and EPIC2, an intracellular RXLR effector, AVRblb2, can prevent C14 secretion to the apoplast [14]. On the other hand, P. infestans has several xyloglucan-specific endoglucanases while the potato genome includes ten clustered genes for xyloglucans-specific endoglucanase inhibitor proteins (XEGIPs) [15].
Apart from PAMP (pathogen associated molecular patterns) recognition, plants have evolved a second mechanism of pathogen recognition through resistance genes, via direct or indirect interaction with effectors. Through this mechanism plants can avoid ETS, leading instead to an incompatible interaction between plant and pathogen, referred to as effector-triggered immunity (ETI). This is often accompanied by a hypersensitive response (HR) [5, 16]. PAMP and effector recognition leads to induction of biotic stress signaling involving MAP kinase cascades and induction of defense-related genes through phosphorylation of WRKY [17] and MYB transcription factors [18]. WRKY8 is involved in plant basal defense in tobacco, and is phosphorylated by MAPKs [19]. In addition, both jasmonic acid and salicylic acid are required for activation of PAMP-induced defense responses in potato [20].
Genome-wide transcript profiling has revealed many similarities between incompatible and compatible interactions, but higher numbers of differentially expressed genes were found during an incompatible Hyaloperonospora–Arabidopsis interaction [21]. However, genes with altered expression exclusively in compatible interactions have also been identified [21–23]. Microarrays based on cDNA clones [24–26], and more recently DeepSAGE transcriptome sequencing analysis, [27] have developed an understanding of compatible and incompatible interactions between P. infestans and potato, but none of these studies were based on the sequenced potato genome.
Here, we present a genome-wide expression profiling of compatible and incompatible potato–P. infestans interactions in combination with quantitative apoplastic secretome analyses. Although apoplastic profiling to find representative protein families has been performed in rice, chickpea, alfalfa and Arabidopsis unaffected by plant pathogens [28, 29], and in response to pathogens to a limited extent [30, 31], quantitative analysis of apoplast proteins during the progress of any compatible and incompatible interactions has not been presented previously. The recent release of the potato genome [32], combined with clone-specific sequence information determined by RNA-seq, increased sensitivity of current mass spectrometers, and new methods for label-free quantitative proteomic analysis enabled our study. These technological advances present new possibilities to help understand mechanisms in plant-pathogen interactions as well as to identify candidates for resistance against pathogens.
The Nordic countries have high P. infestans diversity, with both of the P. infestans mating types required for sexual reproduction and a suitable environment for sexual recombination [33]. Since these conditions provide an excellent environment to evaluate sustainable resistance against P. infestans, we have chosen the two most resistant clones based on several years of field trials in Sweden [34]. In addition, we monitor one compatible interaction, allowing us to find putative effector targets, many of which are single proteins from large gene families.
Results
Overview of differentially expressed genes and changes in protein abundance
In order to study stress responses in plant–oomycete interactions, we performed time series analyses of two incompatible interactions (Sarpo Mira and SW93-1015) and one compatible interaction (Desiree) with P. infestans (SE-03058). This was performed at a global level using genome-wide microarrays to determine gene expression changes and short 1D-gel separation followed by MS/MS analysis on a LC-coupled Orbitrap mass spectrometer to determine quantitative changes in apoplastic protein levels. Sarpo Mira and SW93-1015 have similar HR initiation, but later Sarpo Mira has a more typical R gene-mediated expansion in HR lesions [34]. For the compatible interaction, Desiree was chosen because it is the most used potato cultivar in molecular and physiological studies.
In our genome-wide array, 17451 transcripts were significantly differentially expressed relative to uninoculated control in at least one time point in one or more clones during the course of P. infestans challenge (Table 1). This demonstrates that this biotic stress condition has a profound effect on transcriptional responses in potato.
Table 1.
Number of differentially expressed transcripts in the microarray experiments in potato clones Desiree, Sarpo Mira and SW93-1015 and the number of proteins with differential abundance in the three potato clones at 6, 24 and 72 hpi
| Differentially expressed transcripts | ||||||
|---|---|---|---|---|---|---|
| Potato clone | 6 hpi | 24 hpi | 72 hpi | |||
| Up | Down | Up | Down | Up | Down | |
| Desiree | 1449 | 1602 | 633 | 618 | 6009 | 6109 |
| Sarpo Mira | 1362 | 1440 | 543 | 351 | 331 | 417 |
| SW93-1015 | 1169 | 1131 | 1501 | 1782 | 2554 | 2989 |
| Proteins with differential abundance in the secretome | ||||||
| Potato clone | 6 hpi | 24 hpi | 72 hpi | |||
| Up | Down | Up | Down | Up | Down | |
| Desiree | 218 | 76 | 194 | 82 | 289 | 225 |
| Sarpo Mira | 307 | 74 | 353 | 66 | 375 | 127 |
| SW93-1015 | 273 | 45 | 397 | 41 | 307 | 38 |
In order to improve the peptide identification by mass spectrometry, we generated about 52 million clean pair-end reads from RNA-seq data for each of the three potato clones. Contigs were constructed by de novo transcriptome assembly and subsequently ab initio gene prediction algorithms were used to predict ORFs, which were then combined in a customized database for peptide identification. We found more than 3000 transcripts (ORFs) for each potato clone that lacked BLAST hits in the S. tuberosum predicted proteome (Additional file 1: Table S1) and an overall 17% increase in peptide identification was observed. From the apoplast of the three potato clones a total of 12591 peptides were identified. Peptides that appeared in at least two replicates per time point were kept, resulting in 5055 peptides corresponding to 1639 proteins, which were used for quantitative analysis. Of the 1639 identified proteins, 1075 were found to be differentially abundant in at least one time point during the infection, and of these 785 had Potato Genome Sequencing Consortium (PGSC) annotations. In total, 830 of the identified proteins could be classified into 433 different protein families by InterProScan analysis [34, 35]. Based on TargetP [36] analysis for secretory signals, 517 of these proteins were predicted to have a secretory signal, which is in line with other apoplastic secretome studies [29]. Among the most prominent protein families were subtilase family proteins, peroxidases, protease inhibitors, GDSL-lipases, and pectin esterases (Figure 1a). Several proteins that are classified as pathogenesis-related (PR) proteins were identified both in infected and un-infected samples in all three potato clones, including thaumatin, glucanases, glucosidases, P69 proteins, peroxidases, serine proteases, cysteine proteases, PRp27 protein, subtilases, chitinases, osmotins and lipid transfer proteins. Many PR protein family members are thus present in the apoplast already prior to infection and could be putative effector targets very early in plant–pathogen interactions.
Figure 1.
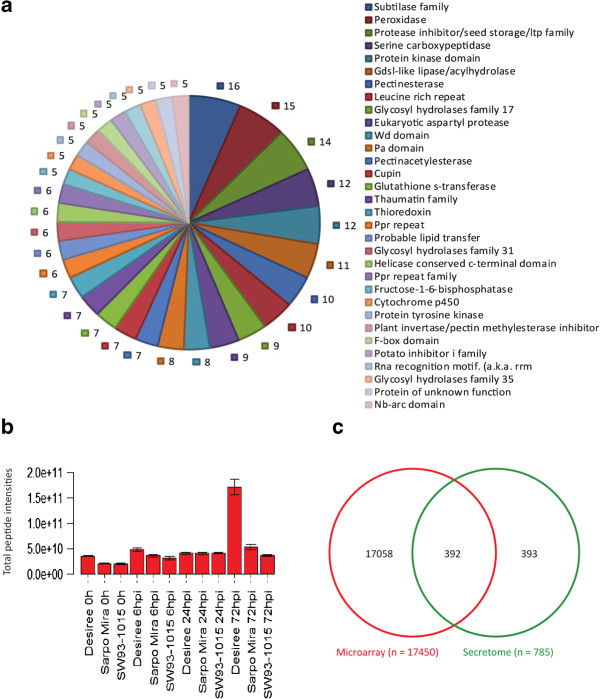
Characterization of secreted proteins. (a) Number of genes found in identified protein families in the apoplastic secretome based on conserved domains and motifs using InterProScan. (b) Mean total peptide intensities for each sample across the four replicates. Error bars indicate standard deviation of the mean. (c) Venn diagrams of differentially expressed transcripts in the microarray and differentially abundant proteins in the apoplast. Only the 785 proteins with Potato Genome Sequencing Consortium (PGSC) annotations from 1075 differentially abundant proteins were used for comparison.
At 6 hours post infection (hpi) and 24 hpi, similar total peptide intensity was observed in both compatible and incompatible interactions, suggesting similar amounts of protein at these time points. However, at 72 hpi we observed significantly higher total peptide intensity in the compatible interaction (Desiree) compared with the two resistant potato clones (Figure 1b) although fewer proteins with higher abundance were observed in Desiree (Table 1). At 72 hpi in Desiree, glucanases and glucosidases comprised a major part of the total apoplast protein intensity, whereas total peptide intensity for all identified proteases and peptidases increased in all three clones (Additional file 2: Figure S1). This indicates a more complex response of the protein composition in the incompatible interactions. For proteins that showed a change in abundance in the apoplast during infection only 392, or 50%, of the corresponding transcripts showed differential expression (Figure 1c).
A large number of transcripts were found to be differentially expressed at 6, 24, and 72 hpi in both compatible and incompatible interactions (Table 1). Different sets of transcripts were differentially expressed between earlier time points (6 and 24 hpi) and later time point (72 hpi) (Figure 2a), indicating a biphasic response to pathogen inoculation in both incompatible and compatible interactions.
Figure 2.
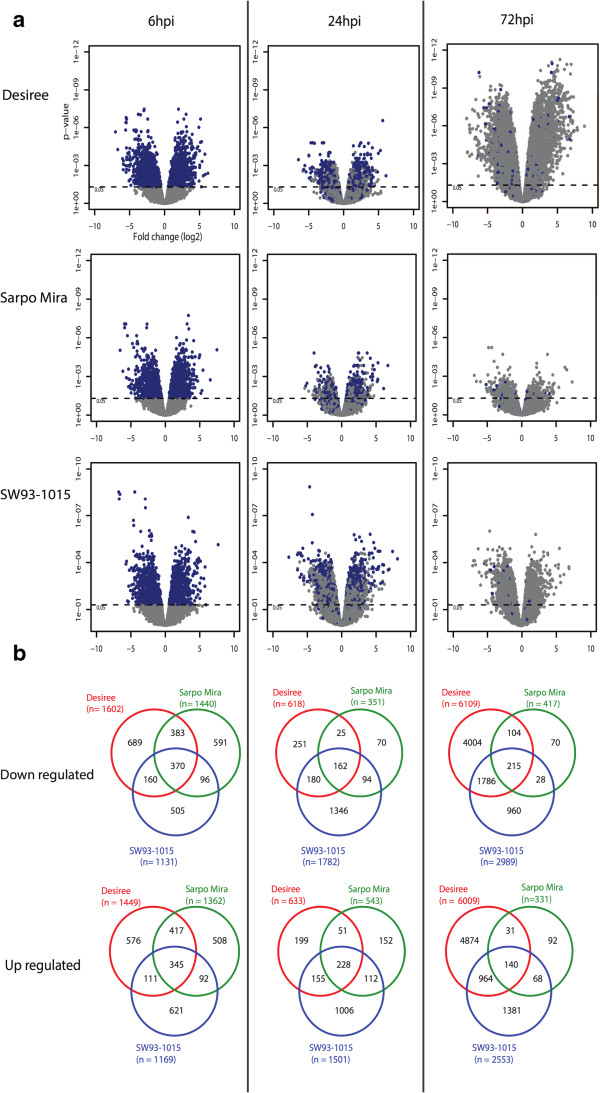
Transcript analysis across the time course in Desiree, Sarpo Mira and SW93-1015. (a) Volcano plots showing differentially expressed transcripts and overlap between the different time points. Blue dots at 24 and 72 hpi indicate transcripts with present at previous time points and grey dots indicate unique transcripts for that time point. (b) Venn diagrams of down-regulated transcripts (upper part) and up-regulated transcripts (lower part) regulated in the microarray experiment in the three potato clones at 6, 24, and 72 hpi from left to right.
Similar numbers of induced transcripts were observed in all three clones at 6 hpi (Figure 2b). However in Sarpo Mira fewer differentially expressed transcripts were observed compared with the other two potato clones at later time-points (Table 1, Figure 2). Uninfected SW93-1015 had around 4000 transcripts that were differentially expressed compared to un-infected Desiree controls. These included SA induced defense marker PR1 gene family members (DMG400005109; DMG400005111), and JA induced defense marker [37] MYC2 (DMG400017535). This supports our earlier conclusion that this clone has a paranoia trait, a weak constitutively active defense [34]. Furthermore, GO terms related to plant cell death and defense such as programmed cell death, methyl salicylate esterase activity, apoptotic process, and innate immune response were enriched in SW93-1015 control samples compared to Desiree control (Additional file 3: Table S2). SW93-1015 generally had more differentially expressed transcripts than Sarpo Mira at 24 and 72 hpi (Table 1), most of which were specific to SW93-1015 (Figure 2b). However, a large number of transcripts with altered expression in SW93-1015 were also differentially expressed in Desiree (Figure 2b), indicating that SW93-1015 might recognize more cues from P. infestans than Sarpo Mira.
Similar to the transcriptome patterns, several proteins that differed in abundance overlapped between 6 and 24 hpi within clones, as seen in the volcano plots (Figure 3a). At 72 hpi, however, several more proteins differentially accumulated compared with earlier time-points (Figure 2a). In addition, a larger number of proteins decreased in abundance in Desiree compared with Sarpo Mira and SW93-1015 (Figure 3a, Table 1). A corresponding difference in down-regulated transcripts was not observed. Instead, a much larger number of up-regulated transcripts was seen in Desiree at 72 hpi compared with the incompatible interactions, as mentioned above. A large number of proteins that increased in abundance were common in all three potato clones (Figure 3b). In addition, a higher percentage of proteins with increased abundance were observed during incompatible interactions in the apoplastic secretome data (Figure 3b) compared with transcript data where larger number of differentially expressed transcripts were observed in the compatible interaction (Table 1).
Figure 3.
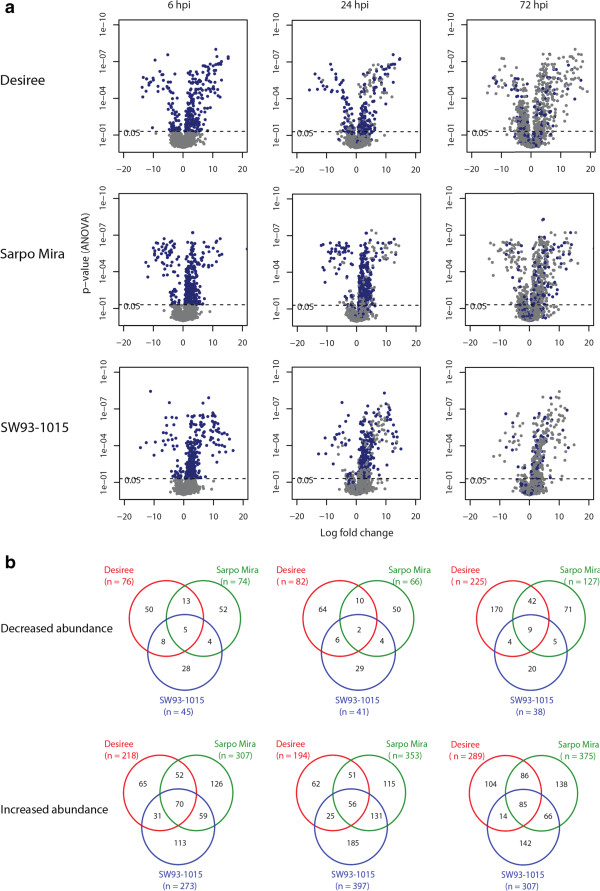
Differentially abundant apoplastic proteins across the time course in Desiree, Sarpo Mira and SW93-1015. (a) Volcano plots showing differentially abundant proteins. Blue dots at 24 and 72 hpi indicate proteins present at previous time points and grey dots indicate unique proteins for that time point. (b) Venn diagrams illustrating numbers of proteins with lower abundance (upper part) and higher abundance (lower part) in the apoplast of Desiree, Sarpo Mira and SW93-1015 from top to bottom at 6, 24, and 72 hpi from left to right.
Responses during incompatible interactions
A hallmark for plant resistance towards pathogens is the initiation of the HR reaction, but in crops relatively little is known about the genes involved. Among the differentially expressed transcripts we found 92, 112, and 68 transcripts at 6, 24, and 72 hpi, respectively, that were up-regulated in both resistant potato clones but not in susceptible Desiree (Figure 2b). Some of these genes such as the MYB transcription factors, glutaredoxins, RING zinc finger proteins and U-box proteins, have members that have previously been associated with resistance (Additional file 4: Table S3). There were 29 different GO terms uniquely enriched for the early (6 hpi) incompatible interaction (Figure 4). Similarly we found 59, 131, and 66 secreted proteins at 6, 24, and 72 hpi, respectively, with increased abundance in the two incompatible interactions but not in the compatible interaction (Figure 3b). These include a subtilisin-like protease, Kunitz-like protease inhibitors, lipid transfer proteins, defensins and strictosidine synthase. These genes and proteins can be regarded as candidate involved in initiation of HR and resistance against P. infestans.
Figure 4.
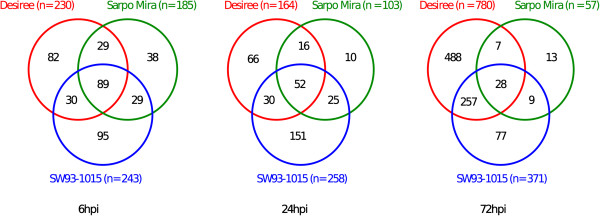
Number of enriched GO terms based on the gene expression analyses of the three potato clones Desiree, Sarpo Mira and SW93-1015 at 6, 24, and 72 hpi.
MapMan pathway analyses reveal that a majority of the represented MYB transcription factors are down-regulated at 6 hpi in Desiree, whereas the opposite is true for Sarpo Mira and SW93-1015 (Additional file 2: Figure S2). In contrast, several WRKY transcription factors are down-regulated in all three clones at 6 hpi.
In order to identify testable HR initiation candidates, we took a conservative approach and identified genes that were 4-fold up-regulated only in the resistant cultivars at 6 or 24 h, at the RNA (Table 2) and protein (Table 3) level. Only four of the 25 transcripts were differentially regulated in the apoplast (highlighted bold in Table 2). The 49 secreted proteins which accumulated early in resistant clones indicate that this compartment is active in HR initiation or other early resistance mechanisms (Table 3). Notable proteins from the apoplast are the RCR3-like Phytophthora-inhibited protease 1 (PIP1), the aspartic protease nepenthesin-1 and a Kunitz-like protease inhibitor. These findings show that the proteolytic machinery of potato plays an important role in defense against pathogens. In line with these findings, tomato plants with mutated Rcr3, which is related to PIP1, have enhanced susceptibility to P. infestans [12]. Several hydrolytic enzymes are also identified and these might exhibit novel catalytic specificities evolved to assist in ETI.
Table 2.
Candidates for hypersensitivity initiation from microarray data
| RNA transcripts | Increased in incompatible clones | ||||
|---|---|---|---|---|---|
| Gene ID | Protein ID | Name | 6 h | 24 h | 72 h |
| PGSC0003DMG401010883 | PGSC0003DMP400019223 | R2r3-myb transcription factor | * | ||
| PGSC0003DMG402004331 | PGSC0003DMP400007701 | Conserved gene of unknown function | * | ||
| PGSC0003DMG400030212 | PGSC0003DMP400052593 | Nitrate reductase | * | ||
| PGSC0003DMG400001855 | PGSC0003DMP400003321 | Beta-amylase PCT-BMYI | * | ||
| PGSC0003DMG400036101 | PGSC0003DMP400058205 | Gene of unknown function | * | ||
| PGSC0003DMG400019518 | PGSC0003DMP400033920 | Pseudo response regulator | * | ||
| PGSC0003DMG400011502 | PGSC0003DMP400020369 | PEP carboxylase kinase | * | ||
| PGSC0003DMG400020209 | PGSC0003DMP400035084 | Nodulin family protein | * | ||
| PGSC0003DMG400001460 | PGSC0003DMP400002648 | Ninja-family protein Os03g0419100 | * | ||
| PGSC0003DMG400025240 | PGSC0003DMP400043812 | Axi 1 protein | * | ||
| PGSC0003DMG400020498 | PGSC0003DMP400035611 | Inositol-1,4,5-triphosphate-5-phosphatase | * | ||
| PGSC0003DMG400023702 | PGSC0003DMP400041014 | Calmodulin binding protein | * | ||
| PGSC0003DMG400023949 | PGSC0003DMP400041401 | Abscisic acid receptor PYL4 | * | ||
| PGSC0003DMG400001771 | PGSC0003DMP400003164 | Dead box ATP-dependent RNA helicase | * | ||
| PGSC0003DMG400001387 | PGSC0003DMP400002507 | Ocs element-binding factor | * | ||
| PGSC0003DMG400010759 | PGSC0003DMP400019038 | Gene of unknown function | * | ||
| PGSC0003DMG400000731 | PGSC0003DMP400001424 | Response to dessication RD2 | * | ||
| PGSC0003DMG400009982 | PGSC0003DMP400017630 | BTB/POZ domain-containing protein | * | ||
| PGSC0003DMG400003993 | PGSC0003DMP400007118 | Citrate binding protein | * | ||
| PGSC0003DMG400003057 | PGSC0003DMP400005490 | Osmotin | * | * | |
| PGSC0003DMG400015267 | PGSC0003DMP400026776 | Kunitz-type protease inhibitor | * | * | |
| PGSC0003DMG400026220 | PGSC0003DMP400045511 | Major pollen allergen Ory s 1 | * | * | |
| PGSC0003DMG400008100 | PGSC0003DMP400014249 | KiTH-2 | * | * | * |
| PGSC0003DMG400008099 | PGSC0003DMP400014248 | KiTH-2 | * | * | * |
| PGSC0003DMG400008098 | PGSC0003DMP400014247 | KiTH-2 | * | * | * |
Genes with increased expression in the two incompatible clones Sarpo Mira and SW93-1015, but not in compatible Desirée. Genes with a minimum 4-fold up-regulation in both Sarpo Mira and SW93-1015 at 6 or 24 hpi and stable or down-regulated in Desirée at 6 and 24 hpi were selected. Up-regulation is indicated with an asterisk. Bolded protein IDs and names indicate proteins for which abundance data was obtained. Symbol (*) indicates the time point in which the change was observed.
Table 3.
Candidates for hypersensitivity initiation from apoplast proteomics data
| Secreted proteins | Increased in incompatible clones | |||
|---|---|---|---|---|
| Protein ID | Name | 6 h | 24 h | 72 h |
| PGSC0003DMP400015021 | Cellulase containing protein | * | ||
| PGSC0003DMP400051976 | Glucan endo-1,3-beta-D-glucosidase | * | ||
| PGSC0003DMP400012140 | Polyamine oxidase | * | ||
| PGSC0003DMP400056168 | Mitogen-activated protein kinase kinase kinase | * | ||
| Q9LTJ3 | Putative uncharacterized protein At5g59350 | * | ||
| Q9SWI1 | Protein PHYTOCHROME KINASE SUBSTRATE 1 | * | ||
| DES_g15011_t1 | Uncharacterized protein | * | ||
| PGSC0003DMP400011041 | Leucine-rich repeat family protein | * | ||
| PGSC0003DMP400015631 | Aldo-keto reductase family 4 member C10 | * | ||
| PGSC0003DMP400027722 | Bacterial-induced peroxidase | * | ||
| PGSC0003DMP400028029 | Alpha-glucosidase | * | ||
| PGSC0003DMP400030201 | Strictosidine synthase | * | ||
| PGSC0003DMP400046981 | Kunitz trypsin inhibitor | * | ||
| PGSC0003DMP400049292 | Pattern formation protein | * | ||
| PGSC0003DMP400051822 | Actin-101 | * | ||
| PGSC0003DMP400001286 † | Conserved gene of unknown function † | * | ||
| PGSC0003DMP400002450 | Conserved gene of unknown function | * | ||
| PGSC0003DMP400003176 | Zinc finger family protein | * | ||
| PGSC0003DMP400008097 | Rho guanine dissociation inhibitor | * | ||
| PGSC0003DMP400009086 † | PR1 protein † | * | ||
| PGSC0003DMP400035459 | Triacylglycerol lipase | * | ||
| PGSC0003DMP400040683 | ATP binding protein | * | ||
| PGSC0003DMP400045856 | Pentatricopeptide repeat-containing protein | * | ||
| PGSC0003DMP400057833 | Polyprotein protein | * | ||
| Q9FJT0 | Putative uncharacterized protein | * | ||
| Q9LDP1 | F28H19.2 protein (F2J6.15 protein) | * | ||
| Q9LVB8 | Probable carboxylesterase 120 (AtCXE20) (EC 3.1.1.1) | * | ||
| Q9SCZ5 | Putative uncharacterized protein F26O13.180 | * | ||
| PGSC0003DMP400005465 | Osmotin | * | * | |
| PGSC0003DMP400018074 | Phytophthora-inhibited protease 1 | * | * | |
| PGSC0003DMP400021164 | Non-specific lipid-transfer protein | * | * | |
| DES_g20722_t1 | Uncharacterized protein | * | * | |
| O22214 | Putative uncharacterized protein At2g41520 | * | * | |
| O64572 | Expressed protein (Uncharacterized protein) | * | * | |
| PGSC0003DMP400006538 | Aspartic proteinase nepenthesin-1 | * | * | |
| PGSC0003DMP400012829 | Leucine-rich repeat receptor kinase | * | * | |
| PGSC0003DMP400026222 | Cytochrome P450 | * | * | |
| PGSC0003DMP400038388 | Rhicadhesin receptor | * | * | |
| PGSC0003DMP400059150 | Gene of unknown function | * | * | |
| PGSC0003DMP400067598 | O-methyltransferase | * | * | |
| PGSC0003DMP400013560 | Gene of unknown function | * | * | |
| PGSC0003DMP400053911 | Beta-galactosidase | * | * | |
| PGSC0003DMP400012597 | Epidermis-specific secreted glycoprotein EP1 | * | * | |
| PGSC0003DMP400016025 | Pentatricopeptide repeat-containing protein | * | * | |
| PGSC0003DMP400006168 | Glycosyl hydrolase family | * | * | * |
| PGSC0003DMP400017287 † | Cytochrome P450 hydroxylase † | * | * | * |
| PGSC0003DMP400035498 † | Peroxidase † | * | * | * |
| PGSC0003DMP400064458 | Gag-pol protein | * | * | * |
| Q9FKV2 | Berberine bridge enzyme (FAD-binding and BBE domain-containing protein) | * | * | * |
Proteins with increased abundance in the secretome for the two incompatible clones Sarpo Mira and SW93-1015, but not for compatible Desirée. Proteins with a minimum 4-fold increase in both Sarpo Mira and SW93-1015 at 6 or 24 hpi and stable or decreased abundance in Desirée at 6 and 24 hpi were selected. Up-regulation is indicated with an asterisk. The symbol † indicates proteins that were not identified using at least one peptide specific for the listed gene. Symbol (*) indicates the time point in which the change was observed.
General defense and the susceptible interaction
There was a clear overlap between differentially expressed transcripts in both the incompatible and compatible interactions (Figure 2b) demonstrating that there are common sets of genes differentially expressed independently of the type of interaction. At 6 hpi there is a large overlap between all three clones in enriched GO terms (Figure 4) with many GO terms linked to primary metabolism, but also general signaling mechanisms such as protein tyrosine and serine/threonine kinase activity. A change in primary metabolism and brassinosteroid-related processes is then seen throughout the time course in all three clones (Additional file 3: Table S2). Between the three clones at 6 hpi, the MapMan pathway analysis shows a striking similarity for C2H2-CO-like and C2H2-Dof zinc finger containing transcription factors indicating that these might be involved in general defense (Additional file 2: Figure S1). There are a large number of GO terms only enriched in the compatible interaction (Figure 4). In hormone-related processes, auxin transport and stimulus GO terms are unique to the compatible interaction, and this is also true for gibberellin biosynthesis and stimulus. At 72 hpi a large number of GO terms are significantly enriched in Desiree. It is the only time point where functional groups associated with jasmonic acid and systemic acquired resistance are present, and is in line with the start of the necrotrophic phase of the pathogen. Also, several histone-related genes can be seen to be up-regulated in Desiree, whereas this is the case for only a few members in SW93-1015 and none in Sarpo Mira.
Screen for putative effector targets in the apoplast
Although the use of effector targets in resistance breeding is desirable [38], very few of such proteins have been identified in oomycete–plant interactions. We detected many secreted proteins that decreased in abundances during the compatible interaction. This is in line with a suppression of some secreted proteins by pathogen effectors or a faster degradation due to protein-complex formation. P. infestans effector can suppress secretion of a defense protein is exemplified by the cysteine protease C14 [14]. We found that the C14 protease decreased sharply at 6 hpi in the Desiree apoplast even though it increased at transcript level at this time point validating Bozkurt et al’s findings in tomato (Figure 5). This inspired two strategies to screen for putative effector targets in our quantitative apoplastic secretome data.
Figure 5.
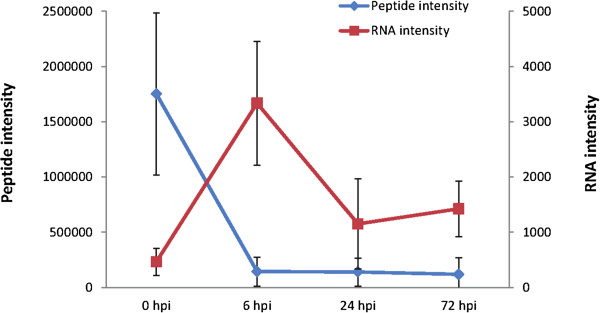
Gene expression and protein abundance in the apoplast of known effector target cysteine protease C14. Transcript and protein levels of the effector target cysteine protease C14 decreased at 6 hpi in the Desiree apoplast and at the same time was transitionally up regulated.
Firstly, we selected proteins with a minimum of 8-fold decreased in abundance in the susceptible clone at 6 and 24 h after infection. This selection produced 40 candidates, many of which are single proteins from large gene families (Table 4). These proteins may also be putative targets in the incompatible interactions because at early time points secreted effectors are expected to be present in all interactions.
Table 4.
Putative effector targets
| Protein ID | Name | Desiree 6 h | Desiree 24 h |
|---|---|---|---|
| PGSC0003DMP400017664 | Transcription factor | * | |
| Q9SZ87 | Reverse transcriptase (RNA-dependent DNA polymerase) | * | |
| Q9SPM5 | GDA1/CD39 (nucleoside phosphatase) family | * | |
| Q8LBK6 | Glutaredoxin | * | |
| Q9SSD1 | Leucine rich repeat | * | |
| PGSC0003DMP400018076 | Cysteine protease | * | |
| PGSC0003DMP400045977 | Cystatin | * | |
| PGSC0003DMP400024264 | Conserved gene of unknown function | * | |
| PGSC0003DMP400020961 | 5′-nucleotidase sure | * | |
| O81459 | ATPase family associated with various cellular activities (AAA) | * | * |
| PGSC0003DMP400049952 | Pectinesterase-2 | * | * |
| F4J1D9 | MutS domain II | * | * |
| PGSC0003DMP400008301 | Pto-interacting protein 1 | * | * |
| Q8VZM7 | Protein of unknown function (DUF1012) | * | * |
| Q9FJV2 | FBD, Leucine Rich Repeat | * | * |
| PGSC0003DMP400006604 | Beta-galactosidase † | * | * |
| PGSC0003DMP400021388 | HSP transcription factor † | * | * |
| PGSC0003DMP400001052 | Peptidyl-prolyl cis-trans isomerase | * | * |
| PGSC0003DMP400054971 | Phospholipase A1 † | * | * |
| PGSC0003DMP400065569 | Gene of unknown function | * | * |
| DES.g31837.t1 | * | * | |
| PGSC0003DMP400056405 | Histidine kinase 3B | * | * |
| PGSC0003DMP400016823 | Kunitz-type proteinase inhibitor | * | * |
| PGSC0003DMP400044843 | Serine-threonine protein kinase, plant-type | * | * |
| PGSC0003DMP400004370 | Glutamate decarboxylase 4a | * | * |
| Q9FGS4 | Fe-S metabolism associated domain, Quinolinate synthetase A protein | * | * |
| A0ME24 | Putative uncharacterized protein | * | * |
| O81861 | Glycosyl hydrolases family 18 | * | * |
| PGSC0003DMP400008705 | Subtilase family protein | * | * |
| PGSC0003DMP400068385 | Conserved gene of unknown function | * | * |
| PGSC0003DMP400004622 | Beta-galactosidase | * | * |
| PGSC0003DMP400055439 | Protein kinase atmrk1 | * | * |
| PGSC0003DMP400017510 | Cysteine protease 14 | * | * |
| PGSC0003DMP400017719 | Multicopper oxidase | * | |
| Q8VZG2 | Domain associated at C-terminal with AAA, ATPase family associated with various cellular activities (AAA) | * | |
| PGSC0003DMP400048051 | Glutathione S-transferase T5 | * | |
| Q4FE22 | Potato inhibitor I family | * | |
| PGSC0003DMP400049086 | Thioredoxin peroxidase | * | |
| Q9FMR7 | Mitochondrial fission ELM1 | * | |
| PGSC0003DMP400025698 | High mobility group protein | * |
Proteins with decreased abundance in the secretome for the compatible clone Desiree, but not for the two incompatible clones Sarpo Mira and SW93-1015. Proteins with a minimum 8-fold down-regulation in Desiree and stable in both Sarpo Mira and SW93-1015 at 6 or 24 hpi were selected. 8-fold down-regulation is indicated with an asterisk. The symbol † indicates proteins that were not identified using at least one peptide specific for the listed gene. Symbol (*) indicates the time point in which the change was observed.
Secondly, we performed clustering by STEM, in order to find protein abundance patterns that occur during compatible and incompatible interactions in the apoplast. Among 13 profiles with significant numbers of associated proteins in Desiree, we found five profiles (number 26, 11, 9, 1, and 12 in order of significance) containing in total 156 proteins with lower abundance in the apoplast. In contrast, only one significant profile in Sarpo Mira (out of 10 profiles) and none in SW93-1015 (out of 11 profiles) represented proteins with lower abundance (Figure 6a). In addition, one of the profiles representing proteins with higher abundance (49), was only significant for SW93-1015 and Sarpo Mira, and represented a large number of proteins for these clones, 203 and 91 proteins respectively (Figure 6a).
Figure 6.
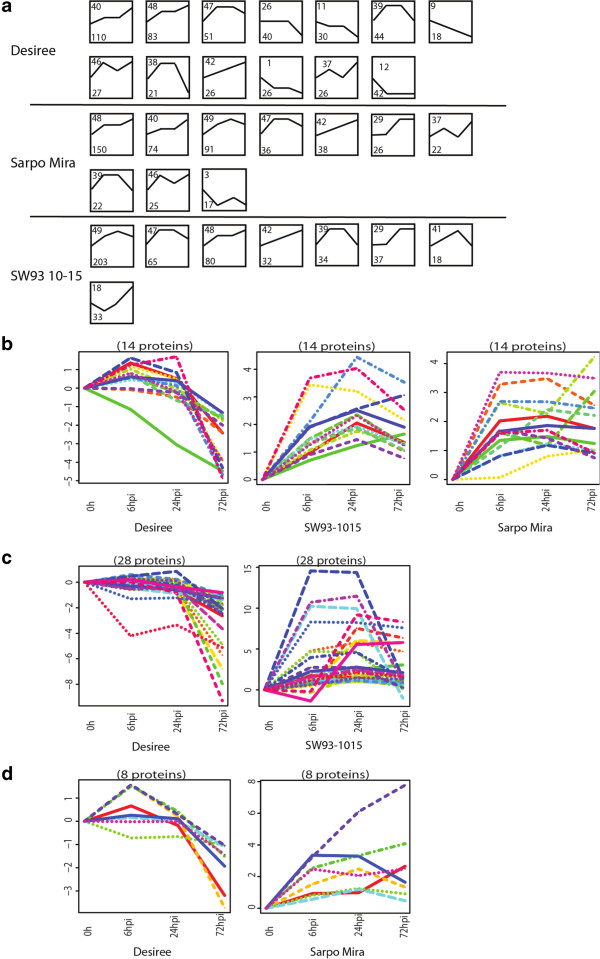
Identification of candidates for effector targets. (a) Significant profiles from STEM clustering of apoplastic proteins at 0, 6, 24, and 72 hpi in the three potato clones. Protein intensities from four replicates for each sample were used for clustering. Only proteins with statistically significant differences for at least at one time point were used. Each box represents a protein abundance pattern during the time course. Top left of each box is the profile number defined in STEM and bottom left of each box indicates the number of proteins that fit the defined profile pattern. (b) Proteins found with decreased abundance profiles in Desiree, but increased abundance profiles in both Sarpo Mira and SW93-1015. (c) Proteins found with decreased abundance profiles in Desiree but with increased abundance profiles in SW93-1015. (d) Proteins found with decreased abundance profiles in Desiree but with increased abundance profiles in Sarpo Mira.
In order to find putative effector-targeted proteins that are differentially regulated in compatible and incompatible interactions, we selected candidates from profiles containing proteins with increased levels in the two incompatible interactions (profile 39, 40, 42, 47, 48, and 49 from Sarpo Mira, and profile 29, 39, 41, 42, 47, 48, and 49 from SW93-1015) and compared them with the proteins associated with profiles representing decreased levels in the compatible interaction (profile 1, 9, 11, 12, 26) (Figure 6a). We found 14 proteins from both incompatible interaction profiles with increased abundance that showed decreased accumulation in the compatible interaction (Figure 6b; Additional file 5: Table S4). These included a lyase, a pectinesterase, a Kunitz trypsin inhibitor and a dehydratase. In addition, we found 28 proteins with decreased abundance profiles in Desiree (Profile 1, 9, 11, 12, 26) but with increased abundance profiles in SW93-1015 (Figure 6c; Additional file 5: Table S4). Eight proteins with decreased abundance profiles in Desiree were found in the increased abundance profiles of Sarpo Mira (profile 39, 40, 42, 47, 48, and 49; Figure 6d; Additional file 5: Table S4).
Discussion
We have carried out time course studies of one compatible and two incompatible interactions between P. infestans and potato by global transcriptomics and apoplastic proteomics using potato clones Desiree, Sarpo Mira, and SW93-1015. In response to P. infestans infection, there was a large overlap of transcripts and proteins with changed abundance irrespective of compatibility, which can be seen as a common signature for challenge with Phytophthora (Figures 2b and 3b). Among the differentially abundant proteins found in the apoplast, 50% were also differentially expressed at the transcript level (Figure 1c). This is in line with previously reported data from yeast that showed that regulation of mRNA explains around 40% of the protein concentration changes [39], and demonstrates the value of measuring both transcript and protein levels.
We identified more than 1500 proteins in the apoplast; almost half of these are associated with 400 conserved functional domains (Figure 1a, Additional file 4: Table S3). The wide range of functional categories among apoplastic secretome suggests a high complexity in this protein fraction. Among the identified proteins 30% had a secretory signal predicted by TargetP, which is consistent with other apoplastic proteomics studies and suggests that a large proportion of the secreted proteins are secreted through unconventional secretory pathways [40].
At 6 hpi, a higher number of apoplast proteins increased in both incompatible interactions than in the compatible interaction, demonstrating a quicker response during the incompatible interaction (Table 1). This difference is even clearer at 24 hpi, when the number of proteins with increased abundance in the incompatible interactions is almost twice that of the compatible interaction. Another notable difference in the apoplast at this stage is that more proteins decreased in abundance in the compatible interaction compared to the incompatible interactions. Interestingly, we did not observe this difference in the transcriptome, indicating a more pronounced suppression by effectors on secreted proteins than on transcription. At 72 hpi, substantially more genes were induced in the compatible interaction than in the incompatible interactions. Most of these genes may have a role in metabolic and structural re-modeling due to infection.
Although the total protein content is greater in the compatible interaction (Figure 1b), fewer proteins show increased abundance. This suggests that the composition of the apoplastic secretome is important in the incompatible interaction and that certain apoplastic proteins are suppressed by the pathogen during the compatible interaction (Figure 1b; Tables 2 and 3). Major proteins that accumulated in the susceptible interaction were glucanases and glucosidases (Additional file 2: Figure S1a) that might be involved in symptom development, as many more cells were visibly affected in the compatible interaction than in the incompatible interactions [34]. Evidence that the apoplastic secretome is altered during pathogen attack has previously been found in cell cultures [41], but such a large-scale perturbation of the host apoplastic secretome in planta is unprecedented.
Little is known about the genes involved in development of HR in potato. There were few genes that were uniquely induced in both resistant potato clones; notably glutaredoxins, MYB transcription factors and a zinc finger protein (Tables 2 and 3; Additional file 4: Table S3). Plants with impaired glutaredoxin activity are resistant to the necrotroph Botrytis [42] and SA-inducible glutaredoxin is involved in suppression of the jasmonic acid-responsive genes in Arabidopsis [43], suggesting a role in hormone crosstalk during the defense response. Plant U-box proteins, also unique to incompatible interactions, is a gene family with diverse functions linked to salicylic acid and PAD4 mediated pathways [44]. WRKY transcription factors did not show this pattern and many were initially down-regulated, although WRKY8 has been implicated in defense-related MAPK signaling and P. infestans resistance in Nicotiana benthamiana [19].
Sarpo Mira, which carries five different resistance genes [45], showed relatively few changes after challenge with P. infestans. This might be due to specific R gene recognition and responses to P. infestans. Although the difference between specific responses in the two resistant genotypes was large, both still display full resistance against the pathogen and initiate similar macroscopic HR in detached leaf assays [34]. Our analysis suggests the possibility that pathogen recognition occurs differently in Sarpo Mira and SW93-1015. Sarpo Mira might mainly depend on effector recognition by resistance proteins, while SW93-1015 may have additional recognition and response mechanisms. At 6 hpi in SW93-1015 two LRR receptor-like kinases are induced, perhaps leading to defense responses unique to SW93-1015.
The fact that certain proteins in the apoplast might be selectively suppressed during the compatible interaction but not the incompatible interactions allows us to screen for putative effector targets. In this unique type of screening described here, we found several candidates that were single proteins from large gene families. In addition to the well characterized cysteine protease 14 (PGSC0003DMP400017510), we found another cysteine protease (PGSC0003DMP400045977) as well as a cystatin homologue (a putative cysteine protease inhibitor PGSC0003DMP400018076). Two other proteins of interest were a Kunitz-type proteinase inhibitor (PGSC0003DMP400016823) and a subtilase family protein (PGSC0003DMP400008705) that also was identified in this screen. With the exact identity of these genes it is now possible to investigate these functionally.
Conclusion
We carried out a time course study of potato transcriptomic and proteomic responses after P. infestans inoculation, both in compatible and incompatible interactions. Studying samples from the same time course for both gene expression and protein abundance lead to identification of potential targets for plant proteins directly manipulated by the pathogen. By using two phenotypically different resistant potato clones we identify several transcripts and proteins that only show increased expression or abundance in both incompatible potato clones that are potentially involved in resistance. In summary, we provide ample number of transcripts and proteins that could be used in targeted studies on one of the most agriculturally important plant-pathogen interactions.
Methods
Plant material and growth conditions
Three potato clones Desiree, Sarpo Mira and SW93-1015 were used. Sarpo Mira and SW93-1015 are two highly resistant potato clones with slightly different resistance reactions [34]. Sarpo Mira is a potato cultivar that recognizes five different effectors from P. infestans [45] and shows a classical HR reaction in response to P. infestans inoculation. SW93-1015 is a breeding potato clone which is consistently resistant to Swedish P. infestans populations with reduced HR expansion and a weak cpr genotype [34]. Plants were grown in a growth chamber with controlled conditions set at 20°C with a 16:8 light:dark cycle and 70% relative humidity. Five-week-old plants were transferred to an infection chamber with 100% humidity and 10:14 light:dark cycle. After 6 hours, plants were sprayed with an encysted zoospore suspension from P. infestans isolate SE-03058 [46] until the leaf surfaces were fully saturated with the zoospore suspension (15000 sporangia/ml). Relative humidity was maintained at 100% for 2 days after inoculation and then adjusted to 90% for the rest of the experiment. Samples were collected at 6, 24, and 72 hpi. For RNA and apoplastic secretome sampling, fully-expanded upper leaves were collected.
RNA isolation and microarray analysis
Samples were collected from three independent biological experiments. Four to six leaves frozen in liquid nitrogen were homogenized to a fine powder using FastPrep-24 (MP-bio, Santa Ana, USA) with 2 mm beads. Leaf tissue (100 mg) was weighed out and put into RNase-free tubes before extraction. RNA extractions were performed using the RNeasy Plant Mini kit (Qiagen GmbH, Hilden, Germany). Samples were DNase treated and cleaned using the Qiagen RNA cleanup kit. RNA concentration and purity (260/280 nm > 1.8) was checked by a ND-1000 NanoDrop (Wilmington, USA) and integrity of the samples were analyzed with an Experion™ Automated Electrophoresis System (Bio-Rad Laboratories, Hercules, USA). For mRNA expression analysis, a custom-made Agilent expression array (JHI Solanum tuberosum 60 k v1; ArrayExpress accession A-MEXP-2272) based on predicted transcripts of the Solanum tuberosum genome (version 3.4) was used according to the supplier’s directions (One-Color Microarray-Based Gene Expression Analysis Low Input Quick Amp Labeling v. 6.5; Agilent). Data were extracted from each scanned array image using Feature Extraction software (v. 10.7.3; Agilent). The array data is deposited in ArrayExpress: E-MTAB-1515.
Apoplastic secretome sample preparation
Three biological replicates of un-infected control samples were collected. For each apoplast sample, four fully expanded middle leaves from two plants were subjected to independent apoplast isolations. In order to collect un-infected control samples, plants were transferred to the humid chamber and kept in the chamber for 6 hours under the same experimental conditions as for infection experiments. Apoplast isolation was performed by using vacuum infiltration as described [34]. Apoplastic fluids collected from two plants for each sample were pooled, dissolved in 6× SDS-PAGE buffer containing DTT, and denatured at 95°C for 3 minutes. Pooled samples (30 μl of each) were loaded on polyacrylamide gels and separated for 2 cm with SDS-PAGE. After staining with Coomassie, the gel lane from each sample was cut into about 1 mm2 pieces. Each lane was kept separate from this point on, generating five subreplicates for each control sample and four subreplicates for each infected sample. Samples were then subjected to in-gel tryptic digestion with incubation (modified sequencing grade; Promega, Madison, WI, USA) overnight at 37°C. Peptides were extracted in 50–80% acetonitrile and excess acetonitrile was vaporized using centrifugation under vacuum. De-salting was performed using UltraMicro spin columns (Nest group). The whole experiment was repeated twice.
Mass spectrometry
MS analysis was performed on a LTQ Orbitrap XL with an Eksigent nano-LC system (Eksigent technologies, Dublin, CA, USA). A 5 μl sample was injected and separated at a flow rate of 300 nl/min with a 90 minute gradient. The four most intense ions were selected in data-dependent mode for fragmentation in the linear ion trap, for details see [47]. Files were converted to mzML [48] and Mascot Generic Format (MGF) using ProteoWizard [49] and uploaded to the Proteios Software Environment [50]. MGF files were used for MS/MS identification, and mzML files for feature detection. Identification searches were performed in Mascot (http://www.matrixscience.com) and X!Tandem (http://www.thegpm.org/tandem) in a database consisting of all Solanum proteins in UniProt (http://www.uniprot.org), all annotated proteins from the potato genome project (http://www.potatogenome.net, [6]) and predicted proteins from de novo assembled transcripts from three potato clones Desiree, Sarpo Mira and SW93-1015, plus an equally sized decoy part consisting of the reversed protein sequences (in total 670584 proteins). The MS mass tolerance was set to 5 ppm and MS/MS fragment tolerance to 0.5 Da, with one missed cleavage allowed. Cysteine carbamidomethylation was set as fixed and methionine oxidation as variable modification. FDR was subsequently calculated for the combined search results at the peptide-spectrum level, and filtered at a FDR of 1% as described previously [50, 51]. To quantify possible peptides, msInspect [52] feature detection was performed on the mzML files from Proteios using default settings. The features were matched to identifications with a retention time tolerance of 0.2 minutes and an m/z tolerance of 0.005 Da as well as a requirement of same charge and LC-MS/MS run. To facilitate the differential expression analysis and to propagate sequences between the runs, alignment was performed using our recently developed algorithm within Proteios [47, 53]. A report of the peptide features corresponding between runs was exported for further analysis. Peptide data used for further analysis is found in Additional file 6: Table S5 and LC-MS runs are listed in supporting information, Additional file 7: Table S6. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the PRIDE partner repository with the dataset identifier PXD000435 and DOI 10.6019/PXD000435.
Bioinformatics and statistical analysis
RNA-sequencing
We obtained approximately 52 million reads for each potato clone from RNA samples collected at 24 hpi using paired-end libraries from Illumina HiSeq 2000 machines. Clean paired-end reads were then de novo assembled using Trinity (version r2011-11-26) to build transcript contigs. Protein coding sequences from these contigs were extracted by gene prediction program Augustus (version 2.5.5) that was used for constructing a proteomics database. BLASTX was used to obtain annotations from the potato genome. The RNA-seq data has been deposited in ArrayExpress: E-MTAB-1712.
Microarray
Probe intensities were background-corrected and normalized using the quantile method in the Limma R package [54]. Genes with p-values below 0.05 after adjustment with the Benjamini-Hochberg method were regarded as significant. The projected GO annotation (Additional file 8: Table S7) [55] for the differentially expressed probes was then analyzed for enrichment of Gene Ontology (GO) terms using GOEast [56] with default settings (adjusted p-value < 0.1).
Apoplastic secretome
Peptides with a FDR of < 0.01 were selected for further analysis. For normalization, we used the Eigen MS method incorporated in DanteR (v0.2) that uses Eigen values to find trends in the data for normalization. Only peptides that were identified in at least two of the subreplicates of any sample were selected and normalized. In DanteR, data was filtered, missing values imputed and protein level intensities for the leading proteins calculated using Q-Rollup function with 2 to 3 peptides per protein based on median peptide intensities [57–59]. Proteins with single peptide hits were included for further analysis. Differentially expressed proteins were calculated using one-way ANOVA for all time points from each potato clone with their relative controls. Fold change estimates were performed based on linear model comparison of each time point with the relative control for each potato clone. After Benjamini-Hochberg (BH) p-value adjustment, differentially expressed proteins with adjusted p-value < 0.05 were selected for further analysis. Default settings in STEM [60] were used and. Standalone InterProScan [35] with default settings was used to find conserved domains and peptide fingerprints for apoplastic secretome protein classification [61]. TargetP was used for secretory signal peptide identification [36]. MapMan analysis was performed to explore gene pathways based on sequence identity to Arabidopsis genes [61].
Electronic supplementary material
Additional file 1: Table S1: Clone-specific peptide identifications. ORFs were predicted based on RNA-seq data in Desirée, Sarpo Mira and SW93-1015. (PDF 9 KB)
Additional file 4: Table S3: Gene expression during Phytophthora infection. Microarray data was normalized and analyzed with the Limma R-package. Expression ratios and corresponding adjusted p-values are given for all microarray probes with matching potato transcripts. (ZIP 11 MB)
Additional file 3: Table S2: List of enriched Gene Ontology terms across the time course. Enrichment was done with GOEast using default settings and a cut-off of p < 0.1. (TXT 118 KB)
Additional file 2: Figure S1: Total peptide intensity for proteases and peptidases. a, Sum of total intensities of 49 proteins in the apoplast identified as glucanases and glucosidases (4 replicates) b, Sum of total intensities of 78 proteins in the apoplast identified as proteases and carboxypeptidases (4 replicates). Error bars indicated ± SD of the mean. Figure S2. Functional analysis of gene expression by MapMan. Bins for genes related to hormones (a) and transcription (b) for Desirée, Sarpo Mira and SW-1015 at 6, 24 or 72 hpi with an adjusted p-value of <0.05 are shown. (PDF 1 MB)
Additional file 5: Table S4: Protein identifiers for resistance candidates identified in STEM profiles. Excel file with identifiers correspond to profiles presented in Figure 6b-d. (PDF 55 KB)
Additional file 6: Table S5: Peptide data during Phytophthora infection. Raw mass spectrometer peptide intensities. (TXT 4 MB)
Additional file 7: Table S6: LC-MS file names. Raw data was have been deposited to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the PRIDE partner repository with the dataset identifier PXD000435 and DOI 10.6019/PXD000435. (PDF 32 KB)
Additional file 8: Table S7: Gene ontology terms for the potato genome and custom-made Agilent expression array. The file (Extended_GOterms_JHI_Solanum_tuberosum_60kv1_expression_array.txt) is formatted for use in GOEast [57]. (TXT 4 MB)
Acknowledgements
We thank Mia Mogren, Jenny Morris and Karin Hansson for excellent technical assistance, and Estelle Proux-Wera for assisting with bioinformatics. We thank the MapMan team for the mapping file based on the Solanum tuberosum genome. The Swedish foundation for Strategic research, The Crafoord foundation, The Swedish Government and The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning are thanked for financial support.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
AA and EAlex carried out the molecular studies, participated in the bioinformatic analysis and drafted the manuscript. ML and PH participated in the molecular analysis. MS, SR and FL participated in the bioinformatic analysis. EA conceived the study, and participated in its design and coordination and drafted the manuscript. All authors read and approved the final manuscript.
Contributor Information
Ashfaq Ali, Email: ashfaq.ali@med.lu.se.
Erik Alexandersson, Email: Erik.Alexandersson@slu.se.
Marianne Sandin, Email: Marianne.Sandin@immun.lth.se.
Svante Resjö, Email: svante.resjo@slu.se.
Marit Lenman, Email: Marit.Lenman@slu.se.
Pete Hedley, Email: Pete.Hedley@hutton.ac.uk.
Fredrik Levander, Email: Fredrik.Levander@immun.lth.se.
Erik Andreasson, Email: Erik.Andreasson@slu.se.
References
- 1.Haverkort A, Struik P, Visser R, Jacobsen E. Applied biotechnology to combat late blight in potato caused by Phytophthora infestans. Potato Res. 2009;52(3):249–264. doi: 10.1007/s11540-009-9136-3. [DOI] [Google Scholar]
- 2.Widmark AK, Andersson B, Cassel‒Lundhagen A, Sandström M, Yuen J. Phytophthora infestans in a single field in southwest Sweden early in spring: symptoms, spatial distribution and genotypic variation. Plant Pathol. 2007;56(4):573–579. doi: 10.1111/j.1365-3059.2007.01618.x. [DOI] [Google Scholar]
- 3.Judelson HS, Blanco FA. The spores of Phytophthora: weapons of the plant destroyer. Nat Rev Microbiol. 2005;3(1):47–58. doi: 10.1038/nrmicro1064. [DOI] [PubMed] [Google Scholar]
- 4.Toth IK, Birch PRJ. Rotting softly and stealthily. Curr Opin Plant Biol. 2005;8(4):424–429. doi: 10.1016/j.pbi.2005.04.001. [DOI] [PubMed] [Google Scholar]
- 5.Jones JD, Dangl JL. The plant immune system. Nature. 2006;444(7117):323–329. doi: 10.1038/nature05286. [DOI] [PubMed] [Google Scholar]
- 6.Haas BJ, Zody MC, Jiang RHY, Handsaker RE, Cano LM, Grabherr M, Kodira CD, Raffaele S, Torto-Alalibo T, Bozkurt TO, Ah-Fong AM, Alvarado L, Anderson VL, Armstrong MR, Avrova A, Baxter L, Beynon J, Boevink PC, Bollmann SR, Bos JI, Bulone V, Cai G, Cakir C, Carrington JC, Chawner M, Conti L, Costanzo S, Ewan R, Fahlgren N, Fischbach MA, et al. Genome sequence and analysis of the Irish potato famine pathogen Phytophthora infestans. Nature. 2009;461(7262):393–398. doi: 10.1038/nature08358. [DOI] [PubMed] [Google Scholar]
- 7.Kamoun S. A catalogue of the effector secretome of plant pathogenic oomycetes. Annu Rev Phytopathol. 2006;44:41–60. doi: 10.1146/annurev.phyto.44.070505.143436. [DOI] [PubMed] [Google Scholar]
- 8.Hok S, Attard A, Keller H. Getting the most from the host: how pathogens force plants to cooperate in disease. Mol Plant Microbe Interact. 2010;23(10):1253–1259. doi: 10.1094/MPMI-04-10-0103. [DOI] [PubMed] [Google Scholar]
- 9.Tian M, Huitema E, Da Cunha L, Torto-Alalibo T, Kamoun S. A Kazal-like extracellular serine protease inhibitor from Phytophthora infestans targets the tomato pathogenesis-related protease P69B. J Biol Chem. 2004;279(25):26370–26377. doi: 10.1074/jbc.M400941200. [DOI] [PubMed] [Google Scholar]
- 10.Tian M, Benedetti B, Kamoun S. A Second Kazal-like protease inhibitor from Phytophthora infestans inhibits and interacts with the apoplastic pathogenesis-related protease P69B of tomato. Plant Physiol. 2005;138(3):1785–1793. doi: 10.1104/pp.105.061226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tian M, Win J, Song J, van der Hoorn R, van der Knaap E, Kamoun S. A Phytophthora infestans cystatin-like protein targets a novel tomato papain-like apoplastic protease. Plant Physiol. 2007;143(1):364–377. doi: 10.1104/pp.106.090050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Song J, Win J, Tian M, Schornack S, Kaschani F, Ilyas M, van der Hoorn RA, Kamoun S. Apoplastic effectors secreted by two unrelated eukaryotic plant pathogens target the tomato defense protease Rcr3. Proc Natl Acad Sci U S A. 2009;106(5):1654–1659. doi: 10.1073/pnas.0809201106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van der Hoorn RAL, Kaschani F, Shabab M, Bozkurt T, Shindo T, Schornack S, Gu C, Ilyas M, Win J, Kamoun S. An effector-targeted Protease contributes to defense against Phytophthora infestans and is under diversifying selection in natural hosts. Plant Physiol. 2010;154(4):1794–1804. doi: 10.1104/pp.110.158030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bozkurt TO, Schornack S, Win J, Shindo T, Ilyas M, Oliva R, Cano LM, Jones AM, Huitema E, van der Hoorn RA. Phytophthora infestans effector AVRblb2 prevents secretion of a plant immune protease at the haustorial interface. Proc Natl Acad Sci. 2011;108(51):20832–20837. doi: 10.1073/pnas.1112708109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jones RW. Multiple copies of genes encoding XEGIPs are Harbored in an 85-kB Region of the Potato Genome. Plant Mol Biol Rep. 2012;30(4):1040–1046. doi: 10.1007/s11105-012-0416-8. [DOI] [Google Scholar]
- 16.Dangl JL, Jones JDG. Plant pathogens and integrated defence responses to infection. Nature. 2001;411(6839):826–833. doi: 10.1038/35081161. [DOI] [PubMed] [Google Scholar]
- 17.Chen LG, Zhang LP, Yu DQ. Wounding-Induced WRKY8 Is Involved in Basal Defense in Arabidopsis. Mol Plant Microbe In. 2010;23(5):558–565. doi: 10.1094/MPMI-23-5-0558. [DOI] [PubMed] [Google Scholar]
- 18.AbuQamar S, Luo HL, Laluk K, Mickelbart MV, Mengiste T. Crosstalk between biotic and abiotic stress responses in tomato is mediated by the AIM1 transcription factor. Plant J. 2009;58(2):347–360. doi: 10.1111/j.1365-313X.2008.03783.x. [DOI] [PubMed] [Google Scholar]
- 19.Ishihama N, Yamada R, Yoshioka M, Katou S, Yoshioka H. Phosphorylation of the Nicotiana benthamiana WRKY8 transcription factor by MAPK functions in the defense response. Plant Cell. 2011;23(3):1153–1170. doi: 10.1105/tpc.110.081794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Halim VA, Altmann S, Ellinger D, Eschen-Lippold L, Miersch O, Scheel D, Rosahl S. PAMP-induced defense responses in potato require both salicylic acid and jasmonic acid. Plant J. 2009;57(2):230–242. doi: 10.1111/j.1365-313X.2008.03688.x. [DOI] [PubMed] [Google Scholar]
- 21.Huibers RP, de Jong M, Dekter RW, Van den Ackerveken G. Disease-specific expression of host genes during downy mildew infection of Arabidopsis. Mol Plant Microbe Interact. 2009;22(9):1104–1115. doi: 10.1094/MPMI-22-9-1104. [DOI] [PubMed] [Google Scholar]
- 22.Wang XJ, Liu W, Chen XM, Tang CL, Dong YL, Ma JB, Huang XL, Wei GR, Han QM, Huang LL, Kang Z. Differential gene expression in incompatible interaction between wheat and stripe rust fungus revealed by cDNA- AFLP and comparison to compatible interaction. BMC Plant Biol. 2010;10:9. doi: 10.1186/1471-2229-10-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fu XZ, Gong XQ, Zhang YX, Wang Y, Liu JH. Different transcriptional response to Xanthomonas citri subsp. citri between Kumquat and sweet orange with contrasting canker tolerance. PLoS One. 2012;7(7):e41790. doi: 10.1371/journal.pone.0041790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang BL, Liu J, Tian ZD, Song BT, Xie CH. Monitoring the expression patterns of potato genes associated with quantitative resistance to late blight during Phytophthora infestans infection using cDNA microarrays. Plant Sci. 2005;169(6):1155–1167. doi: 10.1016/j.plantsci.2005.07.020. [DOI] [Google Scholar]
- 25.Lindqvist-Kreuze H, Carbajulca D, Gonzalez-Escobedo G, Perez W, Bonierbale M. Comparison of transcript profiles in late blight-challenged Solanum cajamarquense and B3C1 potato clones. Mol Plant Pathol. 2010;11(4):513–530. doi: 10.1111/j.1364-3703.2010.00622.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Restrepo S, Myers KL, del Pozo O, Martin GB, Hart AL, Buell CR, Fry WE, Smart CD. Gene profiling of a compatible interaction between Phytophthora infestans and Solanum tuberosum suggests a role for carbonic anhydrase. Mol Plant Microbe Interact. 2005;18(9):913–922. doi: 10.1094/MPMI-18-0913. [DOI] [PubMed] [Google Scholar]
- 27.Gyetvai G, Sonderkaer M, Gobel U, Basekow R, Ballvora A, Imhoff M, Kersten B, Nielsen KL, Gebhardt C. The transcriptome of compatible and incompatible interactions of potato (Solanum tuberosum) with Phytophthora infestans revealed by DeepSAGE analysis. PLoS One. 2012;7(2):e31526. doi: 10.1371/journal.pone.0031526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Agrawal GK, Jwa NS, Lebrun MH, Job D, Rakwal R. Plant secretome: unlocking secrets of the secreted proteins. Proteomics. 2010;10(4):799–827. doi: 10.1002/pmic.200900514. [DOI] [PubMed] [Google Scholar]
- 29.Alexandersson E, Ali A, Resjo S, Andreasson E. Plant secretome proteomics. Front Plant Sci. 2013;4:9. doi: 10.3389/fpls.2013.00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim ST, Kang YH, Wang Y, Wu J, Park ZY, Rakwal R, Agrawal GK, Lee SY, Kang KY. Secretome analysis of differentially induced proteins in rice suspension-cultured cells triggered by rice blast fungus and elicitor. Proteomics. 2009;9(5):1302–1313. doi: 10.1002/pmic.200800589. [DOI] [PubMed] [Google Scholar]
- 31.Oh IS, Park AR, Bae MS, Kwon SJ, Kim YS, Lee JE, Kang NY, Lee S, Cheong H, Park OK. Secretome analysis reveals an Arabidopsis lipase involved in defense against Alternaria brassicicola. Plant Cell. 2005;17(10):2832–2847. doi: 10.1105/tpc.105.034819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xu X, Pan S, Cheng S, Zhang B, Mu D, Ni P, Zhang G, Yang S, Li R, Wang J, Orjeda G, Guzman F, Torres M, Lozano R, Ponce O, Martinez D, De la Cruz G, Chakrabarti SK, Patil VU, Skryabin KG, Kuznetsov BB, Ravin NV, Kolganova TV, Beletsky AV, Mardanov AV, Di Genova A, Bolser DM, Martin DM, Li G, Yang Y, et al. Genome sequence and analysis of the tuber crop potato. Nature. 2011;475(7355):189–195. doi: 10.1038/nature10158. [DOI] [PubMed] [Google Scholar]
- 33.Brurberg MB, Elameen A, Le VH, Naerstad R, Hermansen A, Lehtinen A, Hannukkala A, Nielsen B, Hansen J, Andersson B, Yuen J. Genetic analysis of Phytophthora infestans populations in the Nordic European countries reveals high genetic variability. Fungal Biol-Uk. 2011;115(4–5):335–342. doi: 10.1016/j.funbio.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 34.Ali A, Moushib LI, Lenman M, Levander F, Olsson K, Carlson-Nilson U, Zoteyeva N, Liljeroth E, Andreasson E. Paranoid potato phytophthora-resistant genotype shows constitutively activated defense. Plant Signal Behav. 2012;7(3):400–408. doi: 10.4161/psb.19149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Quevillon E, Silventoinen V, Pillai S, Harte N, Mulder N, Apweiler R, Lopez R. InterProScan: protein domains identifier. Nucleic Acids Res. 2005;33(Web Server issue):W116–W120. doi: 10.1093/nar/gki442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Emanuelsson O, Brunak S, von Heijne G, Nielsen H. Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc. 2007;2(4):953–971. doi: 10.1038/nprot.2007.131. [DOI] [PubMed] [Google Scholar]
- 37.Dombrecht B, Xue GP, Sprague SJ, Kirkegaard JA, Ross JJ, Reid JB, Fitt GP, Sewelam N, Schenk PM, Manners JM, Kazan K. MYC2 differentially modulates diverse jasmonate-dependent functions in Arabidopsis. Plant Cell. 2007;19(7):2225–2245. doi: 10.1105/tpc.106.048017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gawehns F, Cornelissen BJ, Takken FL. The potential of effector‒target genes in breeding for plant innate immunity. J Microbial Biotechnol. 2013;6(3):223–229. doi: 10.1111/1751-7915.12023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schwanhausser B, Busse D, Li N, Dittmar G, Schuchhardt J, Wolf J, Chen W, Selbach M. Global quantification of mammalian gene expression control. Nature. 2011;473(7347):337–342. doi: 10.1038/nature10098. [DOI] [PubMed] [Google Scholar]
- 40.Ding Y, Wang J, Stierhof YD, Robinson DG, Jiang L. Unconventional protein secretion. Trends Plant Sci. 2012;17(10):606–615. doi: 10.1016/j.tplants.2012.06.004. [DOI] [PubMed] [Google Scholar]
- 41.Kaffarnik FA, Jones AM, Rathjen JP, Peck SC. Effector proteins of the bacterial pathogen Pseudomonas syringae alter the extracellular proteome of the host plant, Arabidopsis thaliana. Mol Cell Proteomics. 2009;8(1):145–156. doi: 10.1074/mcp.M800043-MCP200. [DOI] [PubMed] [Google Scholar]
- 42.La Camera S, L’Haridon F, Astier J, Zander M, Abou-Mansour E, Page G, Thurow C, Wendehenne D, Gatz C, Metraux JP, Lamotte O. The glutaredoxin ATGRXS13 is required to facilitate Botrytis cinerea infection of Arabidopsis thaliana plants. Plant J. 2011;68(3):507–519. doi: 10.1111/j.1365-313X.2011.04706.x. [DOI] [PubMed] [Google Scholar]
- 43.Ndamukong I, Al Abdallat A, Thurow C, Fode B, Zander M, Weigel R, Gatz C. SA-inducible Arabidopsis glutaredoxin interacts with TGA factors and suppresses JA-responsive PDF1.2 transcription. Plant J. 2007;50(1):128–139. doi: 10.1111/j.1365-313X.2007.03039.x. [DOI] [PubMed] [Google Scholar]
- 44.Vogelmann K, Drechsel G, Bergler J, Subert C, Philippar K, Soll J, Engelmann JC, Engelsdorf T, Voll LM, Hoth S. Early senescence and cell death in Arabidopsis saul1 mutants involves the PAD4-dependent salicylic acid pathway. Plant Physiol. 2012;159(4):1477–1487. doi: 10.1104/pp.112.196220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rietman H, Bijsterbosch G, Cano LM, Lee HR, Vossen JH, Jacobsen E, Visser RG, Kamoun S, Vleeshouwers VG. Qualitative and quantitative late blight resistance in the potato cultivar Sarpo Mira is determined by the perception of five distinct RXLR effectors. Mol Plant Microbe Interact. 2012;25(7):910–919. doi: 10.1094/MPMI-01-12-0010-R. [DOI] [PubMed] [Google Scholar]
- 46.Lehtinen A, Andersson B, Le VH, Naerstad R, Rastas M, Ketoja E, Hannukkala AO, Hermansen A, Nielsen BJ, Hansen JG, Yuen J. Aggressiveness of Phytophthora infestans on detached potato leaflets in four Nordic countries. Plant Pathol. 2009;58(4):690–702. doi: 10.1111/j.1365-3059.2009.02038.x. [DOI] [Google Scholar]
- 47.Sandin M, Ali A, Hansson K, Mansson O, Andreasson E, Resjo S, Levander F. An adaptive alignment algorithm for quality-controlled label-free LC-MS. Mol Cell Proteomics. 2013;12(5):1407–1420. doi: 10.1074/mcp.O112.021907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Martens L, Chambers M, Sturm M, Kessner D, Levander F, Shofstahl J, Tang WH, Rompp A, Neumann S, Pizarro AD, Montecchi-Palazzi L, Tasman N, Colemen M, Reisinger F, Souda P, Hermjakob H, Binz PA, Deutsch EW. mzML--a community standard for mass spectrometry data. Mol Cell Proteomics. 2011;10(1):R110–R000133. doi: 10.1074/mcp.R110.000133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kessner D, Chambers M, Burke R, Agus D, Mallick P. ProteoWizard: open source software for rapid proteomics tools development. Bioinformatics. 2008;24(21):2534–2536. doi: 10.1093/bioinformatics/btn323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hakkinen J, Vincic G, Mansson O, Warell K, Levander F. The proteios software environment: an extensible multiuser platform for management and analysis of proteomics data. J Proteome Res. 2009;8(6):3037–3043. doi: 10.1021/pr900189c. [DOI] [PubMed] [Google Scholar]
- 51.Levander F, Krogh M, Warell K, Garden P, James P, Hakkinen J. Automated reporting from gel-based proteomics experiments using the open source Proteios database application. Proteomics. 2007;7(5):668–674. doi: 10.1002/pmic.200600814. [DOI] [PubMed] [Google Scholar]
- 52.Bellew M, Coram M, Fitzgibbon M, Igra M, Randolph T, Wang P, May D, Eng J, Fang R, Lin C, Chen J, Goodlett D, Whiteaker J, Paulovich A, McIntosh M. A suite of algorithms for the comprehensive analysis of complex protein mixtures using high-resolution LC-MS. Bioinformatics. 2006;22(15):1902–1909. doi: 10.1093/bioinformatics/btl276. [DOI] [PubMed] [Google Scholar]
- 53.Sandin M, Teleman J, Malmström J, Levander F. Data processing methods and quality control strategies for label-free LC-MS protein quantification. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics. 2013;1884(1 Pt A):29–41. doi: 10.1016/j.bbapap.2013.03.026. [DOI] [PubMed] [Google Scholar]
- 54.Wettenhall JM, Smyth GK. limmaGUI: a graphical user interface for linear modeling of microarray data. Bioinformatics. 2004;20(18):3705–3706. doi: 10.1093/bioinformatics/bth449. [DOI] [PubMed] [Google Scholar]
- 55.Bengtsson T, Weighill D, Proux-Wéra E, Levander F, Resjö S, Burra DD, Moushib LI, Hedley PE, Liljeroth E, Jacobson D, Alexandersson E, Andreasson E. Proteomics and transcriptomics of the BABA-induced resistance response in potato using a novel functional annotation approach. BMC Genom. 2014;15:315. doi: 10.1186/1471-2164-15-315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zheng Q, Wang XJ. GOEAST: a web-based software toolkit for Gene Ontology enrichment analysis. Nucleic Acids Res. 2008;36(Web Server issue):W358–W363. doi: 10.1093/nar/gkn276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Karpievitch YV, Taverner T, Adkins JN, Callister SJ, Anderson GA, Smith RD, Dabney AR. Normalization of peak intensities in bottom-up MS-based proteomics using singular value decomposition. Bioinformatics. 2009;25(19):2573–2580. doi: 10.1093/bioinformatics/btp426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Karpievitch Y, Stanley J, Taverner T, Huang J, Adkins JN, Ansong C, Heffron F, Metz TO, Qian WJ, Yoon H, Smith RD, Dabney AR. A statistical framework for protein quantitation in bottom-up MS-based proteomics. Bioinformatics. 2009;25(16):2028–2034. doi: 10.1093/bioinformatics/btp362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Taverner T, Karpievitch YV, Polpitiya AD, Brown JN, Dabney AR, Anderson GA, Smith RD. DanteR: an extensible R-based tool for quantitative analysis of -omics data. Bioinformatics. 2012;28(18):2404–2406. doi: 10.1093/bioinformatics/bts449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ernst J, Bar-Joseph Z. STEM: a tool for the analysis of short time series gene expression data. BMC Bioinformatics. 2006;7:191. doi: 10.1186/1471-2105-7-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Thimm O, Blasing O, Gibon Y, Nagel A, Meyer S, Kruger P, Selbig J, Muller LA, Rhee SY, Stitt M. MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J. 2004;37(6):914–939. doi: 10.1111/j.1365-313X.2004.02016.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1: Clone-specific peptide identifications. ORFs were predicted based on RNA-seq data in Desirée, Sarpo Mira and SW93-1015. (PDF 9 KB)
Additional file 4: Table S3: Gene expression during Phytophthora infection. Microarray data was normalized and analyzed with the Limma R-package. Expression ratios and corresponding adjusted p-values are given for all microarray probes with matching potato transcripts. (ZIP 11 MB)
Additional file 3: Table S2: List of enriched Gene Ontology terms across the time course. Enrichment was done with GOEast using default settings and a cut-off of p < 0.1. (TXT 118 KB)
Additional file 2: Figure S1: Total peptide intensity for proteases and peptidases. a, Sum of total intensities of 49 proteins in the apoplast identified as glucanases and glucosidases (4 replicates) b, Sum of total intensities of 78 proteins in the apoplast identified as proteases and carboxypeptidases (4 replicates). Error bars indicated ± SD of the mean. Figure S2. Functional analysis of gene expression by MapMan. Bins for genes related to hormones (a) and transcription (b) for Desirée, Sarpo Mira and SW-1015 at 6, 24 or 72 hpi with an adjusted p-value of <0.05 are shown. (PDF 1 MB)
Additional file 5: Table S4: Protein identifiers for resistance candidates identified in STEM profiles. Excel file with identifiers correspond to profiles presented in Figure 6b-d. (PDF 55 KB)
Additional file 6: Table S5: Peptide data during Phytophthora infection. Raw mass spectrometer peptide intensities. (TXT 4 MB)
Additional file 7: Table S6: LC-MS file names. Raw data was have been deposited to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the PRIDE partner repository with the dataset identifier PXD000435 and DOI 10.6019/PXD000435. (PDF 32 KB)
Additional file 8: Table S7: Gene ontology terms for the potato genome and custom-made Agilent expression array. The file (Extended_GOterms_JHI_Solanum_tuberosum_60kv1_expression_array.txt) is formatted for use in GOEast [57]. (TXT 4 MB)


