Abstract
Combined treatment with allogeneic small lymphocytes or T-depleted small lymphocytes plus a blocking antibody to CD40 ligand (CD40L) permitted indefinite pancreatic islet allograft survival in 37 of 40 recipients that differed from islet donors at major and minor histocompatibility loci. The effect of the allogeneic small lymphocytes was donor antigen-specific. Neither treatment alone was as effective as combined treatment, although anti-CD40L by itself allowed indefinite islet allograft survival in 40% of recipients. Our interpretation is that small lymphocytes expressing donor antigens in the absence of appropriate costimulatory signals are tolerogenic for alloreactive host cells. Anti-CD40L antibody may prevent host T cells from inducing costimulatory signals in donor lymphocytes or islet grafts.
Full text
PDF
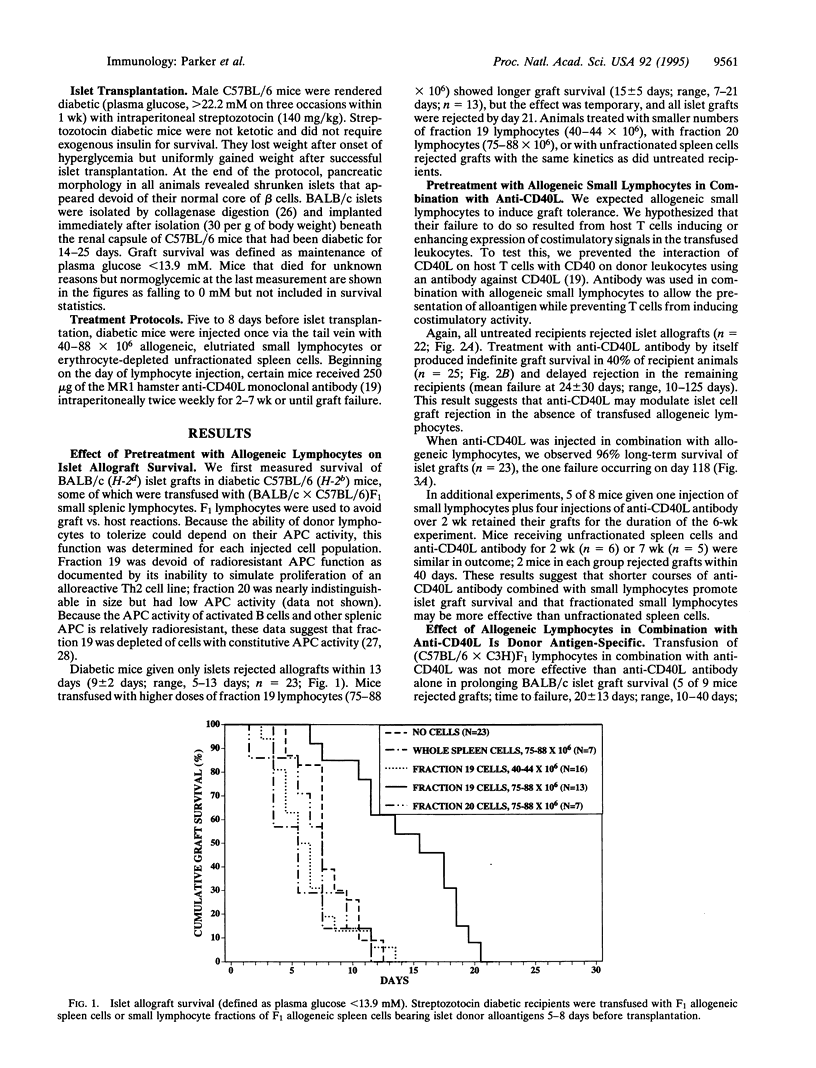
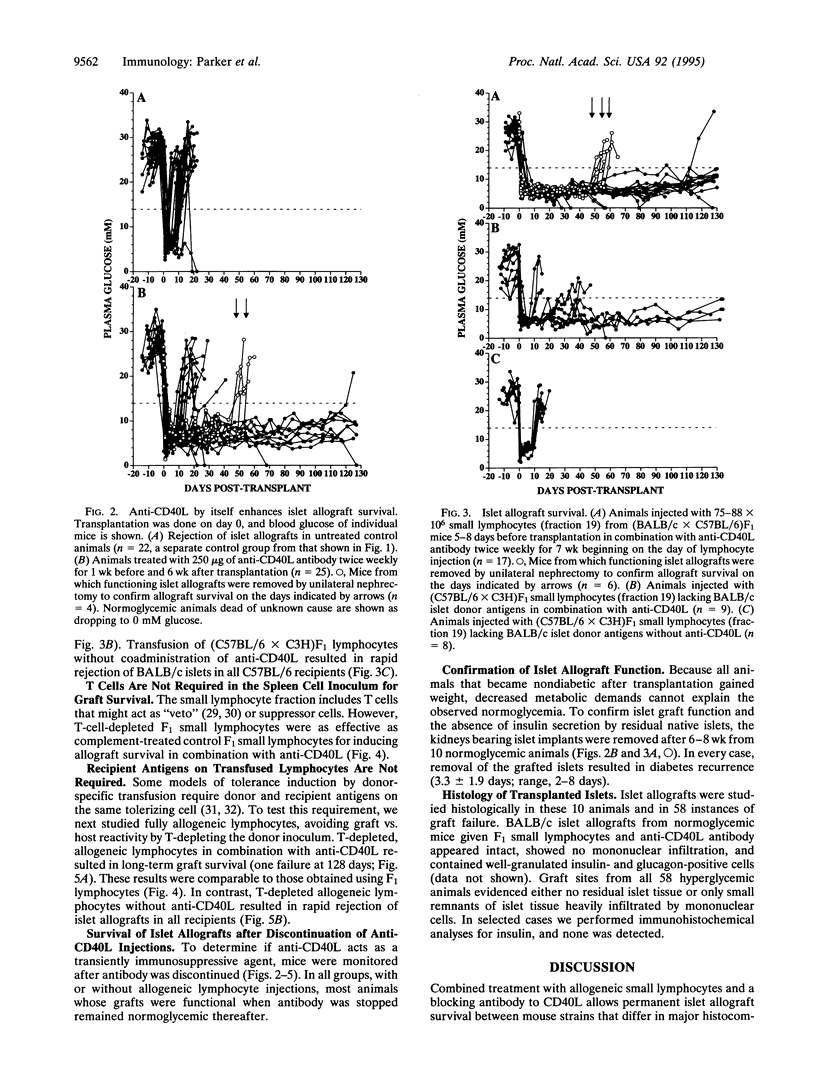
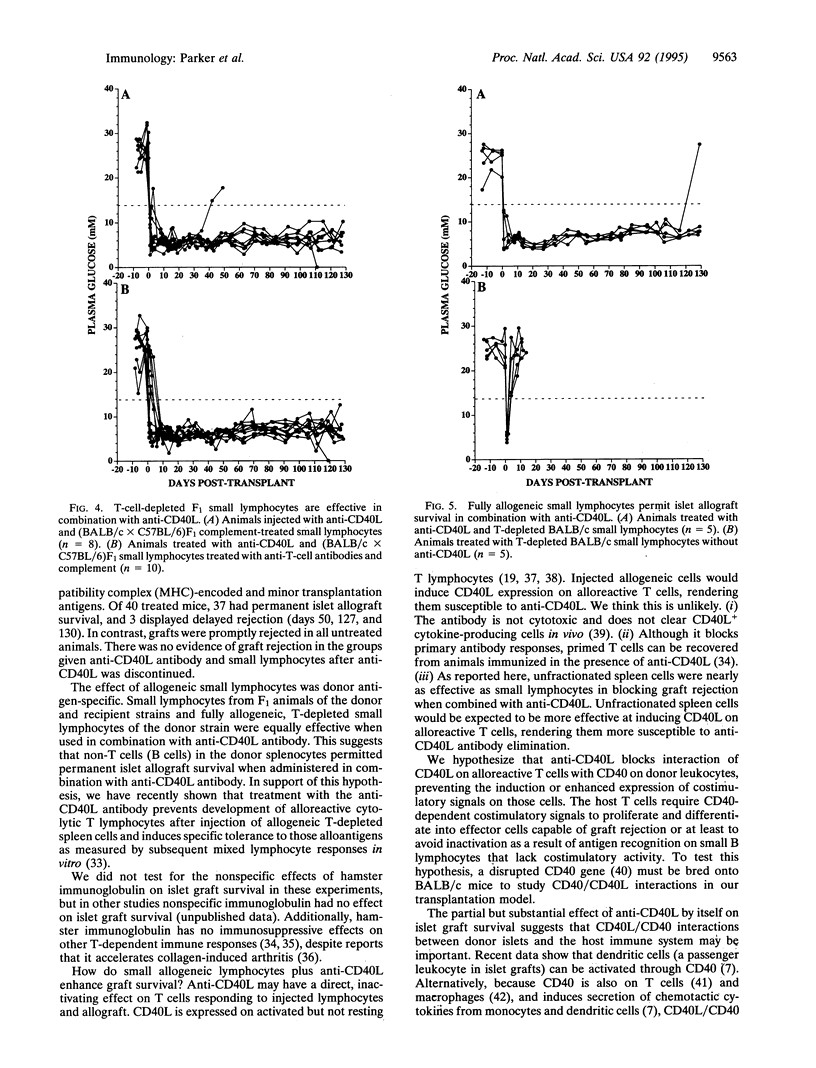

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alderson M. R., Armitage R. J., Tough T. W., Strockbine L., Fanslow W. C., Spriggs M. K. CD40 expression by human monocytes: regulation by cytokines and activation of monocytes by the ligand for CD40. J Exp Med. 1993 Aug 1;178(2):669–674. doi: 10.1084/jem.178.2.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armitage R. J., Fanslow W. C., Strockbine L., Sato T. A., Clifford K. N., Macduff B. M., Anderson D. M., Gimpel S. D., Davis-Smith T., Maliszewski C. R. Molecular and biological characterization of a murine ligand for CD40. Nature. 1992 May 7;357(6373):80–82. doi: 10.1038/357080a0. [DOI] [PubMed] [Google Scholar]
- Ashwell J. D., DeFranco A. L., Paul W. E., Schwartz R. H. Antigen presentation by resting B cells. Radiosensitivity of the antigen-presentation function and two distinct pathways of T cell activation. J Exp Med. 1984 Mar 1;159(3):881–905. doi: 10.1084/jem.159.3.881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunet J. F., Denizot F., Luciani M. F., Roux-Dosseto M., Suzan M., Mattei M. G., Golstein P. A new member of the immunoglobulin superfamily--CTLA-4. Nature. 1987 Jul 16;328(6127):267–270. doi: 10.1038/328267a0. [DOI] [PubMed] [Google Scholar]
- Castle B. E., Kishimoto K., Stearns C., Brown M. L., Kehry M. R. Regulation of expression of the ligand for CD40 on T helper lymphocytes. J Immunol. 1993 Aug 15;151(4):1777–1788. [PubMed] [Google Scholar]
- Caux C., Massacrier C., Vanbervliet B., Dubois B., Van Kooten C., Durand I., Banchereau J. Activation of human dendritic cells through CD40 cross-linking. J Exp Med. 1994 Oct 1;180(4):1263–1272. doi: 10.1084/jem.180.4.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C., Nabavi N. In vitro induction of T cell anergy by blocking B7 and early T cell costimulatory molecule ETC-1/B7-2. Immunity. 1994 May;1(2):147–154. doi: 10.1016/1074-7613(94)90108-2. [DOI] [PubMed] [Google Scholar]
- Corry D. B., Reiner S. L., Linsley P. S., Locksley R. M. Differential effects of blockade of CD28-B7 on the development of Th1 or Th2 effector cells in experimental leishmaniasis. J Immunol. 1994 Nov 1;153(9):4142–4148. [PubMed] [Google Scholar]
- Damle N. K., Klussman K., Leytze G., Linsley P. S. Proliferation of human T lymphocytes induced with superantigens is not dependent on costimulation by the CD28 counter-receptor B7. J Immunol. 1993 Feb 1;150(3):726–735. [PubMed] [Google Scholar]
- Durie F. H., Fava R. A., Foy T. M., Aruffo A., Ledbetter J. A., Noelle R. J. Prevention of collagen-induced arthritis with an antibody to gp39, the ligand for CD40. Science. 1993 Sep 3;261(5126):1328–1330. doi: 10.1126/science.7689748. [DOI] [PubMed] [Google Scholar]
- Fanslow W. C., Clifford K. N., Seaman M., Alderson M. R., Spriggs M. K., Armitage R. J., Ramsdell F. Recombinant CD40 ligand exerts potent biologic effects on T cells. J Immunol. 1994 May 1;152(9):4262–4269. [PubMed] [Google Scholar]
- Fink P. J., Shimonkevitz R. P., Bevan M. J. Veto cells. Annu Rev Immunol. 1988;6:115–137. doi: 10.1146/annurev.iy.06.040188.000555. [DOI] [PubMed] [Google Scholar]
- Foy T. M., Laman J. D., Ledbetter J. A., Aruffo A., Claassen E., Noelle R. J. gp39-CD40 interactions are essential for germinal center formation and the development of B cell memory. J Exp Med. 1994 Jul 1;180(1):157–163. doi: 10.1084/jem.180.1.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foy T. M., Shepherd D. M., Durie F. H., Aruffo A., Ledbetter J. A., Noelle R. J. In vivo CD40-gp39 interactions are essential for thymus-dependent humoral immunity. II. Prolonged suppression of the humoral immune response by an antibody to the ligand for CD40, gp39. J Exp Med. 1993 Nov 1;178(5):1567–1575. doi: 10.1084/jem.178.5.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedman A. S., Freeman G. J., Rhynhart K., Nadler L. M. Selective induction of B7/BB-1 on interferon-gamma stimulated monocytes: a potential mechanism for amplification of T cell activation through the CD28 pathway. Cell Immunol. 1991 Oct 15;137(2):429–437. doi: 10.1016/0008-8749(91)90091-o. [DOI] [PubMed] [Google Scholar]
- Fuchs E. J., Matzinger P. B cells turn off virgin but not memory T cells. Science. 1992 Nov 13;258(5085):1156–1159. doi: 10.1126/science.1439825. [DOI] [PubMed] [Google Scholar]
- Gosselin E. J., Tony H. P., Parker D. C. Characterization of antigen processing and presentation by resting B lymphocytes. J Immunol. 1988 Mar 1;140(5):1408–1413. [PubMed] [Google Scholar]
- Gottlieb P. A., Berrios J. P., Mariani G., Handler E. S., Greiner D., Mordes J. P., Rossini A. A. Autoimmune destruction of islets transplanted into RT6-depleted diabetes-resistant BB/Wor rats. Diabetes. 1990 May;39(5):643–645. doi: 10.2337/diab.39.5.643. [DOI] [PubMed] [Google Scholar]
- Hori S., Sato S., Kitagawa S., Azuma T., Kokudo S., Hamaoka T., Fujiwara H. Tolerance induction of allo-class II H-2 antigen-reactive L3T4+ helper T cells and prolonged survival of the corresponding class II H-2-disparate skin graft. J Immunol. 1989 Sep 1;143(5):1447–1452. [PubMed] [Google Scholar]
- Inaba K., Witmer-Pack M., Inaba M., Hathcock K. S., Sakuta H., Azuma M., Yagita H., Okumura K., Linsley P. S., Ikehara S. The tissue distribution of the B7-2 costimulator in mice: abundant expression on dendritic cells in situ and during maturation in vitro. J Exp Med. 1994 Nov 1;180(5):1849–1860. doi: 10.1084/jem.180.5.1849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jameson S. C., Bevan M. J. T cell receptor antagonists and partial agonists. Immunity. 1995 Jan;2(1):1–11. doi: 10.1016/1074-7613(95)90074-8. [DOI] [PubMed] [Google Scholar]
- Janeway C. A., Jr Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb Symp Quant Biol. 1989;54(Pt 1):1–13. doi: 10.1101/sqb.1989.054.01.003. [DOI] [PubMed] [Google Scholar]
- June C. H., Bluestone J. A., Nadler L. M., Thompson C. B. The B7 and CD28 receptor families. Immunol Today. 1994 Jul;15(7):321–331. doi: 10.1016/0167-5699(94)90080-9. [DOI] [PubMed] [Google Scholar]
- Kakiuchi T., Chesnut R. W., Grey H. M. B cells as antigen-presenting cells: the requirement for B cell activation. J Immunol. 1983 Jul;131(1):109–114. [PubMed] [Google Scholar]
- Kawabe T., Naka T., Yoshida K., Tanaka T., Fujiwara H., Suematsu S., Yoshida N., Kishimoto T., Kikutani H. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity. 1994 Jun;1(3):167–178. doi: 10.1016/1074-7613(94)90095-7. [DOI] [PubMed] [Google Scholar]
- Kennedy M. K., Mohler K. M., Shanebeck K. D., Baum P. R., Picha K. S., Otten-Evans C. A., Janeway C. A., Jr, Grabstein K. H. Induction of B cell costimulatory function by recombinant murine CD40 ligand. Eur J Immunol. 1994 Jan;24(1):116–123. doi: 10.1002/eji.1830240118. [DOI] [PubMed] [Google Scholar]
- Lafferty K. J., Prowse S. J., Simeonovic C. J., Warren H. S. Immunobiology of tissue transplantation: a return to the passenger leukocyte concept. Annu Rev Immunol. 1983;1:143–173. doi: 10.1146/annurev.iy.01.040183.001043. [DOI] [PubMed] [Google Scholar]
- Lenschow D. J., Zeng Y., Thistlethwaite J. R., Montag A., Brady W., Gibson M. G., Linsley P. S., Bluestone J. A. Long-term survival of xenogeneic pancreatic islet grafts induced by CTLA4lg. Science. 1992 Aug 7;257(5071):789–792. doi: 10.1126/science.1323143. [DOI] [PubMed] [Google Scholar]
- Lin H., Bolling S. F., Linsley P. S., Wei R. Q., Gordon D., Thompson C. B., Turka L. A. Long-term acceptance of major histocompatibility complex mismatched cardiac allografts induced by CTLA4Ig plus donor-specific transfusion. J Exp Med. 1993 Nov 1;178(5):1801–1806. doi: 10.1084/jem.178.5.1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombardi G., Sidhu S., Daly M., Batchelor J. R., Makgoba W., Lechler R. I. Are primary alloresponses truly primary? Int Immunol. 1990;2(1):9–13. doi: 10.1093/intimm/2.1.9. [DOI] [PubMed] [Google Scholar]
- Nickoloff B. J., Turka L. A. Immunological functions of non-professional antigen-presenting cells: new insights from studies of T-cell interactions with keratinocytes. Immunol Today. 1994 Oct;15(10):464–469. doi: 10.1016/0167-5699(94)90190-2. [DOI] [PubMed] [Google Scholar]
- Noelle R. J., Roy M., Shepherd D. M., Stamenkovic I., Ledbetter J. A., Aruffo A. A 39-kDa protein on activated helper T cells binds CD40 and transduces the signal for cognate activation of B cells. Proc Natl Acad Sci U S A. 1992 Jul 15;89(14):6550–6554. doi: 10.1073/pnas.89.14.6550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker D. C. T cell-dependent B cell activation. Annu Rev Immunol. 1993;11:331–360. doi: 10.1146/annurev.iy.11.040193.001555. [DOI] [PubMed] [Google Scholar]
- Rammensee H. G., Kroschewski R., Frangoulis B. Clonal anergy induced in mature V beta 6+ T lymphocytes on immunizing Mls-1b mice with Mls-1a expressing cells. Nature. 1989 Jun 15;339(6225):541–544. doi: 10.1038/339541a0. [DOI] [PubMed] [Google Scholar]
- Ranheim E. A., Kipps T. J. Activated T cells induce expression of B7/BB1 on normal or leukemic B cells through a CD40-dependent signal. J Exp Med. 1993 Apr 1;177(4):925–935. doi: 10.1084/jem.177.4.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocha B., Tanchot C., Von Boehmer H. Clonal anergy blocks in vivo growth of mature T cells and can be reversed in the absence of antigen. J Exp Med. 1993 May 1;177(5):1517–1521. doi: 10.1084/jem.177.5.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy M., Waldschmidt T., Aruffo A., Ledbetter J. A., Noelle R. J. The regulation of the expression of gp39, the CD40 ligand, on normal and cloned CD4+ T cells. J Immunol. 1993 Sep 1;151(5):2497–2510. [PubMed] [Google Scholar]
- Schwartz R. H. Costimulation of T lymphocytes: the role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell. 1992 Dec 24;71(7):1065–1068. doi: 10.1016/s0092-8674(05)80055-8. [DOI] [PubMed] [Google Scholar]
- Sheng-Tanner X., Miller R. G. Correlation between lymphocyte-induced donor-specific tolerance and donor cell recirculation. J Exp Med. 1992 Aug 1;176(2):407–413. doi: 10.1084/jem.176.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tony H. P., Parker D. C. Major histocompatibility complex-restricted, polyclonal B cell responses resulting from helper T cell recognition of antiimmunoglobulin presented by small B lymphocytes. J Exp Med. 1985 Jan 1;161(1):223–241. doi: 10.1084/jem.161.1.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Eertwegh A. J., Noelle R. J., Roy M., Shepherd D. M., Aruffo A., Ledbetter J. A., Boersma W. J., Claassen E. In vivo CD40-gp39 interactions are essential for thymus-dependent humoral immunity. I. In vivo expression of CD40 ligand, cytokines, and antibody production delineates sites of cognate T-B cell interactions. J Exp Med. 1993 Nov 1;178(5):1555–1565. doi: 10.1084/jem.178.5.1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenberghe P., Delabie J., de Boer M., De Wolf-Peeters C., Ceuppens J. L. In situ expression of B7/BB1 on antigen-presenting cells and activated B cells: an immunohistochemical study. Int Immunol. 1993 Mar;5(3):317–321. doi: 10.1093/intimm/5.3.317. [DOI] [PubMed] [Google Scholar]
- Whalen B. J., Tony H. P., Parker D. C. Characterization of the effector mechanism of help for antigen-presenting and bystander resting B cell growth mediated by Ia-restricted Th2 helper T cell lines. J Immunol. 1988 Oct 1;141(7):2230–2239. [PubMed] [Google Scholar]
- van Rood J. J., Claas F. H. The influence of allogeneic cells on the human T and B cell repertoire. Science. 1990 Jun 15;248(4961):1388–1393. doi: 10.1126/science.1972596. [DOI] [PubMed] [Google Scholar]
- van Twuyver E., Kast W. M., Mooijaart R. J., Melief C. J., De Waal L. P. Induction of transplantation tolerance by intravenous injection of allogeneic lymphocytes across an H-2 class II mismatch. Different mechanisms operate in tolerization across an H-2 class I vs. H-2 class II disparity. Eur J Immunol. 1990 Feb;20(2):441–444. doi: 10.1002/eji.1830200232. [DOI] [PubMed] [Google Scholar]


