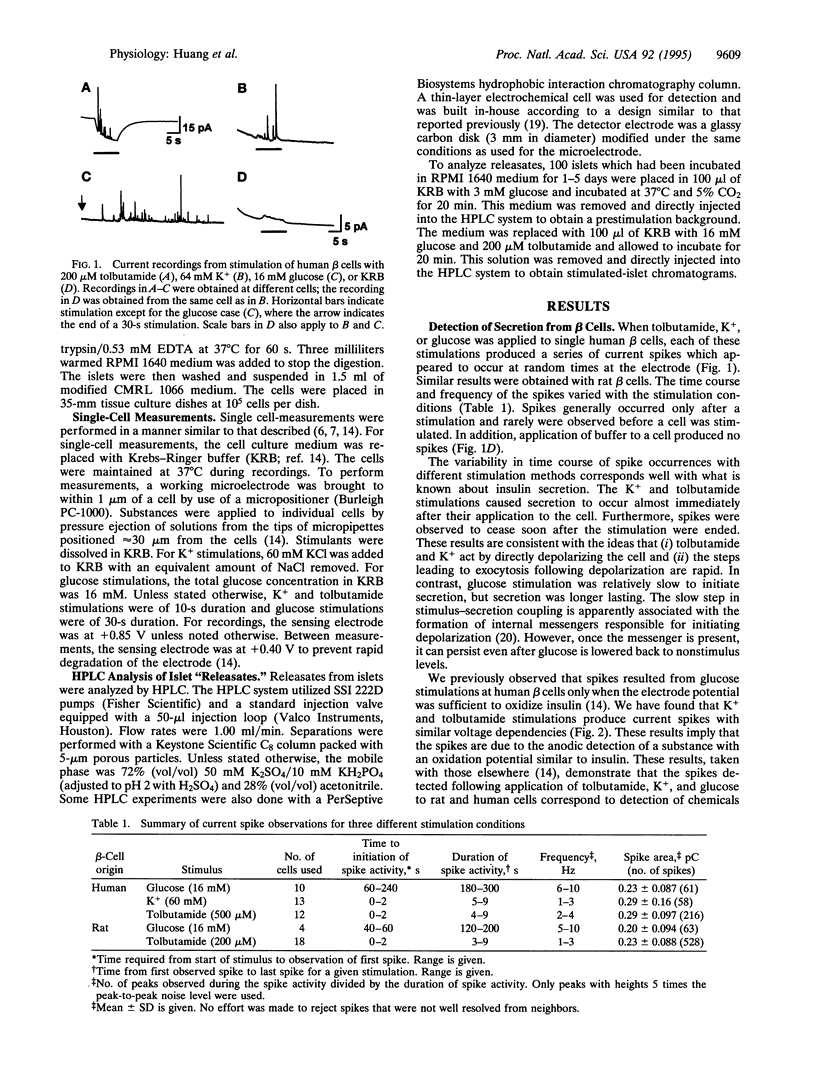
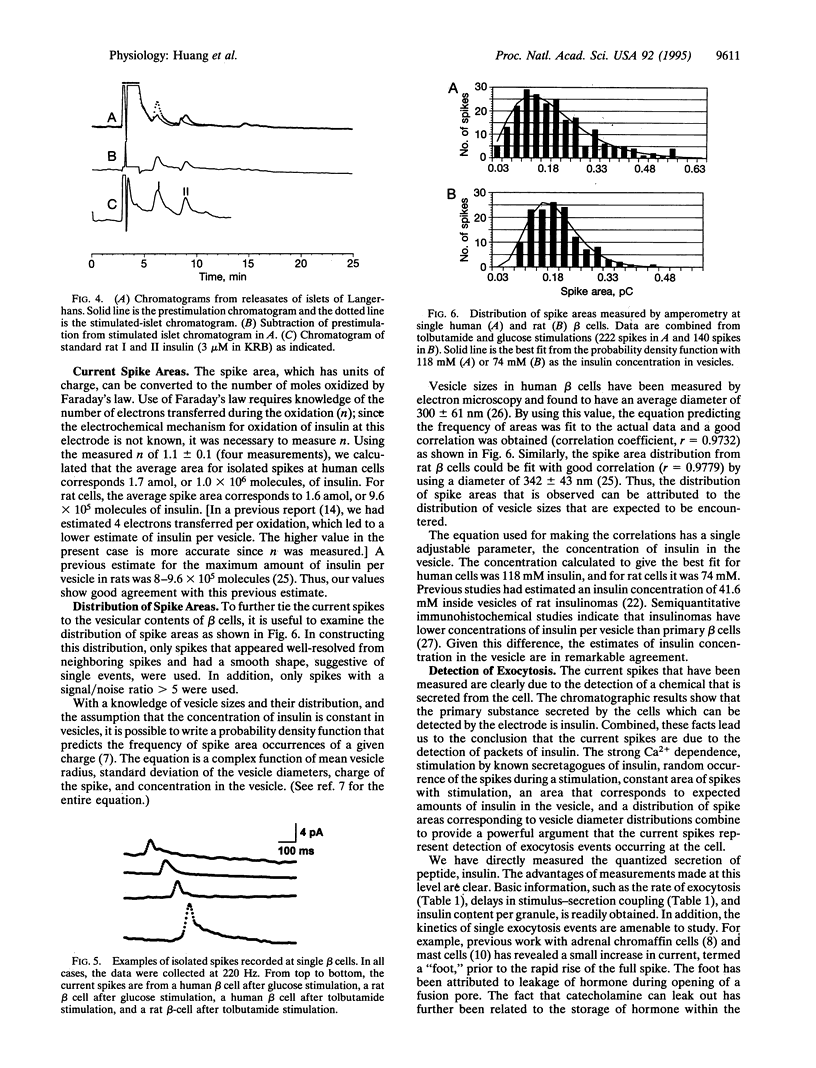
Abstract
Amperometry at a carbon fiber microelectrode modified with a composite of ruthenium oxide and cyanoruthenate was used to monitor chemical secretions of single pancreatic beta cells from rats and humans. When the insulin secretagogues glucose, tolbutamide, and K+ were applied to the cell, a series of randomly occurring current spikes was observed. The current spikes were shown to be due to the detection of chemical substances secreted from the cell. Chromatography showed that the primary secreted substance detected by the electrode was insulin. The current spikes were strongly dependent on external Ca2+, had an average area that was independent of the stimulation method, and had an area distribution which corresponded to the distribution of vesicle sizes in beta cells. It was concluded that the spikes were due to the detection of concentration pulses of insulin secreted by exocytosis.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alvarez de Toledo G., Fernández-Chacón R., Fernández J. M. Release of secretory products during transient vesicle fusion. Nature. 1993 Jun 10;363(6429):554–558. doi: 10.1038/363554a0. [DOI] [PubMed] [Google Scholar]
- Ammälä C., Eliasson L., Bokvist K., Larsson O., Ashcroft F. M., Rorsman P. Exocytosis elicited by action potentials and voltage-clamp calcium currents in individual mouse pancreatic B-cells. J Physiol. 1993 Dec;472:665–688. doi: 10.1113/jphysiol.1993.sp019966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen T. K., Luo G., Ewing A. G. Amperometric monitoring of stimulated catecholamine release from rat pheochromocytoma (PC12) cells at the zeptomole level. Anal Chem. 1994 Oct 1;66(19):3031–3035. doi: 10.1021/ac00091a007. [DOI] [PubMed] [Google Scholar]
- Chow R. H., von Rüden L., Neher E. Delay in vesicle fusion revealed by electrochemical monitoring of single secretory events in adrenal chromaffin cells. Nature. 1992 Mar 5;356(6364):60–63. doi: 10.1038/356060a0. [DOI] [PubMed] [Google Scholar]
- Cox J. A., Gray T. J. Flow injection amperometric determination of insulin based upon its oxidation at a modified electrode. Anal Chem. 1989 Nov 1;61(21):2462–2464. doi: 10.1021/ac00196a027. [DOI] [PubMed] [Google Scholar]
- Fehmann H. C., Weber V., Göke R., Göke B., Arnold R. Cosecretion of amylin and insulin from isolated rat pancreas. FEBS Lett. 1990 Mar 26;262(2):279–281. doi: 10.1016/0014-5793(90)80210-a. [DOI] [PubMed] [Google Scholar]
- Gillis K. D., Misler S. Single cell assay of exocytosis from pancreatic islet B cells. Pflugers Arch. 1992 Jan;420(1):121–123. doi: 10.1007/BF00378654. [DOI] [PubMed] [Google Scholar]
- Hutton J. C., Penn E. J., Peshavaria M. Low-molecular-weight constituents of isolated insulin-secretory granules. Bivalent cations, adenine nucleotides and inorganic phosphate. Biochem J. 1983 Feb 15;210(2):297–305. doi: 10.1042/bj2100297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jankowski J. A., Schroeder T. J., Ciolkowski E. L., Wightman R. M. Temporal characteristics of quantal secretion of catecholamines from adrenal medullary cells. J Biol Chem. 1993 Jul 15;268(20):14694–14700. [PubMed] [Google Scholar]
- Kalina M., Grimelius L., Cedermark B., Hammel I. Insulin and C-peptide co-localization in the beta granules of normal human pancreas and insulinomas. A quantitative immunocytochemical approach. Virchows Arch A Pathol Anat Histopathol. 1989;416(1):19–23. doi: 10.1007/BF01606466. [DOI] [PubMed] [Google Scholar]
- Kennedy R. T., Huang L., Atkinson M. A., Dush P. Amperometric monitoring of chemical secretions from individual pancreatic beta-cells. Anal Chem. 1993 Jul 15;65(14):1882–1887. doi: 10.1021/ac00062a012. [DOI] [PubMed] [Google Scholar]
- Larsson L. I., Sundler F., Håkanson R. Pancreatic polypeptide - a postulated new hormone: identification of its cellular storage site by light and electron microscopic immunocytochemistry. Diabetologia. 1976 Jul;12(3):211–226. doi: 10.1007/BF00422088. [DOI] [PubMed] [Google Scholar]
- Leszczyszyn D. J., Jankowski J. A., Viveros O. H., Diliberto E. J., Jr, Near J. A., Wightman R. M. Nicotinic receptor-mediated catecholamine secretion from individual chromaffin cells. Chemical evidence for exocytosis. J Biol Chem. 1990 Sep 5;265(25):14736–14737. [PubMed] [Google Scholar]
- Matthews E. K., McKay D. B., O'Connor M. D., Borowitz J. L. Biochemical and biophysical characterization of insulin granules isolated from rat pancreatic islets by an iso-osmotic gradient. Biochim Biophys Acta. 1982 Mar 15;715(1):80–89. doi: 10.1016/0304-4165(82)90052-6. [DOI] [PubMed] [Google Scholar]
- Misler S., Barnett D. W., Gillis K. D., Pressel D. M. Electrophysiology of stimulus-secretion coupling in human beta-cells. Diabetes. 1992 Oct;41(10):1221–1228. doi: 10.2337/diab.41.10.1221. [DOI] [PubMed] [Google Scholar]
- Orci L. Macro- and micro-domains in the endocrine pancreas. Diabetes. 1982 Jun;31(6 Pt 1):538–565. doi: 10.2337/diab.31.6.538. [DOI] [PubMed] [Google Scholar]
- Orci L., Vassalli J. D., Perrelet A. The insulin factory. Sci Am. 1988 Sep;259(3):85–94. doi: 10.1038/scientificamerican0988-85. [DOI] [PubMed] [Google Scholar]
- Rajan A. S., Aguilar-Bryan L., Nelson D. A., Yaney G. C., Hsu W. H., Kunze D. L., Boyd A. E., 3rd Ion channels and insulin secretion. Diabetes Care. 1990 Mar;13(3):340–363. doi: 10.2337/diacare.13.3.340. [DOI] [PubMed] [Google Scholar]
- Roe M. W., Lancaster M. E., Mertz R. J., Worley J. F., 3rd, Dukes I. D. Voltage-dependent intracellular calcium release from mouse islets stimulated by glucose. J Biol Chem. 1993 May 15;268(14):9953–9956. [PubMed] [Google Scholar]
- Wightman R. M., Jankowski J. A., Kennedy R. T., Kawagoe K. T., Schroeder T. J., Leszczyszyn D. J., Near J. A., Diliberto E. J., Jr, Viveros O. H. Temporally resolved catecholamine spikes correspond to single vesicle release from individual chromaffin cells. Proc Natl Acad Sci U S A. 1991 Dec 1;88(23):10754–10758. doi: 10.1073/pnas.88.23.10754. [DOI] [PMC free article] [PubMed] [Google Scholar]


