Abstract
Brown adipose tissue (BAT) is specialized to dissipate chemical energy in the form of heat as a defense against cold and excessive feeding. Interest in the field of BAT biology has exploded in the past few years because of the therapeutic potential of BAT to counteract obesity and obesity-related diseases, including insulin resistance. Much progress has been made, particularly in the areas of BAT physiology in adult humans, developmental lineages of brown adipose cell fate, and hormonal control of BAT thermogenesis. As we enter into a new era of brown fat biology, the next challenge will be to develop strategies for activating BAT thermogenesis in adult humans to increase whole-body energy expenditure. This article reviews the recent major advances in this field and discusses emerging questions.
Keywords: obesity, metabolic syndrome, thermogenesis, brown adipose tissue, brown fat, beige cell, UCP1, interorgan networks
INTRODUCTION
Brown adipose tissue (BAT) evolved to generate heat and to protect animals from hypothermia in mammals. This process, termed nonshivering thermogenesis, is particularly important during hibernation and for small animals and infants who have greater demands on thermogenesis due to a large surface-to-volume ratio. Recent advances in brown fat biology suggest that BAT plays a pivotal role in controlling energy homeostasis and thus may offer new therapeutics with which to treat human obesity.
Obesity develops when food intake chronically exceeds total energy expenditure (1). All antiobesity medications currently approved by the FDA act to repress energy intake, either by suppressing appetite or by inhibiting intestinal fat absorption (e.g., Orlistat). However, given that these drugs often elicit serious side effects, including depression, oily bowel movements, and steatorrhea, alternative strategies for treating obesity are needed. Because BAT has a remarkable capacity to dissipate energy and produce heat via brown fat–specific uncoupling protein 1 (UCP1) in mitochondria, targeting BAT-mediated thermogenesis may offer a viable alternative approach to increase energy expenditure.
Over the past few years, several major advancements have been made in the field of brown fat biology. First, clinical observations in oncology using 18fluoro-labeled 2-deoxyglucose positron emission tomography (18FDG-PET) scanning in combination with computed tomography (CT) led to the unexpected discoveries that distinct and active BAT deposits exist in adult humans. The amount of BAT correlates inversely with body mass index (BMI) and adiposity, raising the possibility that variations in the amount or thermogenic function of BAT may contribute to the propensity for weight gain (2–7). These findings also suggest strongly that molecular pathways controlling BAT thermogenesis are evolutionarily conserved in mammals. Second, studies in rodent models have led to a better understanding of developmental lineages in brown adipocytes. The current evidence indicates that rodents and humans possess two types of UCP1-positive thermogenic adipocytes arising from developmentally distinct lineages: (a) classical brown adipocytes and (b) so-called beige or brite cells that reside sporadically within white adipose tissue (WAT). Third, deconvoluting the developmental pathways of brown fat has resulted in the definition of several key transcriptional regulators and signaling molecules. Researchers have been able to manipulate these factors to generate new brown adipocytes in vivo. Lastly, in addition to the primary control of BAT thermogenesis by the sympathetic nervous system, cross talk between BAT and other peripheral tissues, such as skeletal muscle, liver, and immune cells, is now recognized and suggests that secondary BAT-mediated physiological networks also control systemic energy homeostasis and glucose homeostasis.
As we enter into a new and exciting era in brown fat biology, the next challenge will certainly be aimed at activating BAT thermogenesis in adult humans to increase whole-body energy expenditure. Meanwhile, much will be learned from efforts focused on elucidating the fundamental mechanisms underlying brown fat development and function. Identifying specific and independent regulatory circuits in addition to known pathways such as the β-adrenoceptor (β-AR) signaling pathway that control BAT development and function would enable us to synergistically increase energy expenditure. Although other review articles have discussed BAT thermogenesis (8–14), this review adds to these by focusing on exciting new advances in this field and by outlining new challenges and emerging questions for the field.
UCP1-MEDIATED ADAPTIVE THERMOGENESIS IN BAT
Structural Insights into UCP1-Dependent BAT Thermogenesis
Despite high mitochondria content and high cellular respiration rates, brown adipocytes have a remarkably low capacity for ATP synthesis. Most cells lack UCPs and produce ATP through ATP synthase, utilizing the proton gradient across the mitochondrial inner membrane. In contrast, brown adipocytes express substantially low levels of ATP synthase (15) and instead utilize UCP1, which diminishes the proton gradient by uncoupling cellular respiration and mitochondrial ATP synthesis, to stimulate thermogenesis. Although other UCP members, including UCP2 and UCP3, share structural homology with UCP1, they do not contribute to adaptive thermogenesis in vivo (16). Hence, UCP1 is considered to be the sole thermogenin responsible for adaptive nonshivering thermogenesis.
UCP1 expression levels do not necessarily reflect its thermogenic activity in brown adipocytes. In the resting state, UCP1’s activity is constitutively repressed by purine di- and triphosphate nucleotides. Purine nucleotides, primarily ATP, bind on the cytosolic side of UCP1 and prevent proton transport (17). Free fatty acids (FFAs), in contrast, are well-known activators of UCP1 activity. The Kirichok group recently employed whole-cell patch-clamp techniques and found that UCP1 is a fatty acid anion/H+ symporter (18). This group found that, although UCP1 has no constitutive activity due to ATP inhibition, long-chain fatty acids (LCFAs) bind to the cytoplasmic side of UCP1 and override inhibition of UCP1 activity by purine nucleotides. As illustrated in Figure 1a, one LCFA molecule binds directly to the UCP1 protein, serving as a UCP1 substrate to transport one H+ per transport cycle. Intriguingly, the transported FFA anion requires a long, hydrophobic tail, as the affinity of the alkyl anion for the substrate-binding site of UCP1 dramatically decreases as the tail length decreases. Although LCFA anions compete with ATP for binding to UCP1, LCFA and ATP are unlikely to competitively bind the same exact surface of UCP1 due to their drastically different structures. Although structural models, such as the allosteric change model (8), have been proposed to account for the stimulatory effects of LCFAs on UCP1 in brown fat mitochondria and for their quick reversal of ATP’s inhibitory effects on UCP1, further studies including crystal structure analyses are needed to define the precise mechanisms underlying this phenomenon.
Figure 1.
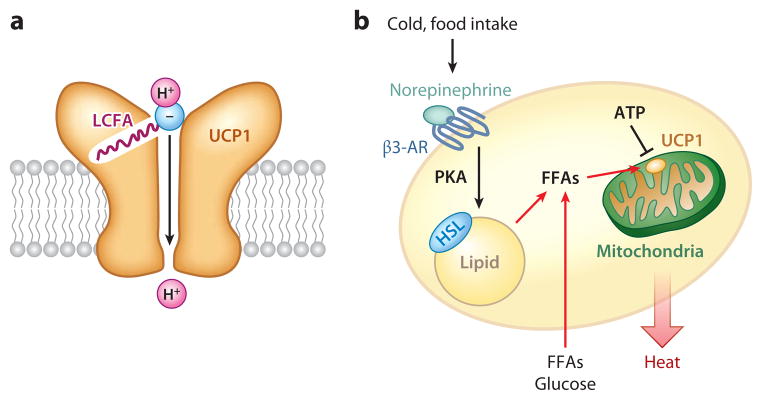
Uncoupling protein 1 (UCP1)-dependent thermogenesis in brown adipocytes. (a) Hypothetical model of UCP1-dependent proton uncoupling in brown fat mitochondria. A long-chain fatty acid (LCFA) molecule directly binds to the UCP1 protein, serving as a UCP1 substrate to transport one H+ per transport cycle. The transported free-fatty-acid (FFA) anion needs a long, hydrophobic tail. (b) ATP inhibits the activity of UCP1 in the resting state. In response to cold stimuli or excess food intake, norepinephrine acts on the β3-adrenoceptor (β3-AR), leading to the activation of cAMP-dependent protein kinase (PKA) and the phosphorylation of hormone-sensitive lipase (HSL). FFAs are generated by cAMP-induced lipolysis or taken up from the circulation and utilized as UCP1 substrates for H+ transport and as substrates of β-oxidation in brown fat mitochondria (indicated by red arrows).
FFA-mediated control of UCP1 activity makes perfect sense from a physiological standpoint, as FFAs are end products of cold stimuli or excess food intake. In response to these two physiological stimuli, norepinephrine release by sympathetic nerves acts on adrenergic receptors (primarily the β3-adrenoceptor) in BAT and activates adenylyl cyclase to increase intracellular cAMP levels, which then triggers activation of cAMP-dependent protein kinase (PKA). In turn, PKA phosphorylates hormone-sensitive lipase and lipid droplet–binding proteins such as perilipins, leading to the hydrolysis of triglycerides in BAT lipid droplets (Figure 1b). FFAs are either generated by cAMP-induced lipolysis or taken up from the circulation and subsequently utilized as substrates of β-oxidation in brown adipocytes and as UCP1 substrates for H+ transport. To activate UCP1, as measured by UCP1-dependent current, FFA levels must exceed ATP by nearly 100-fold, as shown by Fedorenko et al. (18), implying that UCP1 activity is suppressed under normal physiological states.
Although transgenic expression of UCP1 keeps brown fat mitochondria in an active uncoupled state in vivo, excessive UCP1 expression can be cytotoxic to adipocytes and causes atrophy of BAT (19). Indeed, efforts in the 1930s to use ubiquitous chemical uncouplers such as 2,4-dinitrophenol as antiobesity drugs were not successful (20). With these newer data, future efforts to unravel the structural determinants by which UCP1 activity is controlled by ATP, FFAs, and other metabolites may provide strategies for developing a new class of antiobesity drugs.
Metabolic Significance of UCP1: Temperature Matters
More than 30 years ago, investigators suggested that BAT plays a pivotal role not only in cold-induced adaptive thermogenesis but also in diet-induced thermogenesis in which energy expenditure increases in response to certain diets to presumably protect animals from obesity (21). Later studies using genetic mouse models clearly demonstrated that defects in BAT thermogenesis lead to obesity. The first genetic evidence supporting a causal relationship between defects in BAT function and the development of obesity came from a study showing that transgenic mice expressing diphtheria toxin driven by the UCP1 gene promoter display obesity and insulin resistance without hyperphagia (22). Moreover, UCP1-null mice exhibit an obese phenotype when kept in thermoneutral conditions (23). Initially, UCP1-null mice were not reported to be obese but did exhibit impaired tolerance to cold temperature when housed in ambient temperatures (20–24°C) (24, 25). Later, the Cannon & Nedergaard group (23) found that shivering thermogenesis is activated in skeletal muscle of UCP1-null mice under ambient temperature; this group concluded that UCP1-null mice consume substantial amounts of energy to maintain their body temperature by shivering thermogenesis. As such, an obese phenotype due to the lack of UCP1-dependent, diet-induced thermogenesis is revealed only in UCP1-null mice at thermoneutrality (29–30°C) or at temperatures that do not require muscle shivering. This result validated the importance of BAT thermogenesis in preventing obesity and underscored the importance of cage temperature when metabolic phenotypes in rodent models are assessed.
What are the proper experiments needed to fairly assess BAT thermogenesis and to avoid confounding variables that might lead to paradoxical metabolic outcomes in animal models? On the basis of numerous studies in the field, other factors need to be considered, namely, metabolic changes in nonshivering thermogenesis and in shivering thermogenesis and the animal’s insulation (e.g., skin, fur, or body surface-to-volume ratio). A thorough assessment should include (a) metabolic analyses of animals at thermoneutrality (29–30°C for mice) to measure basal metabolic rates; (b) analyses of changes in whole-body oxygen consumption rate in response to norepinephrine or a β3-adrenoceptor agonist (e.g., CL316243), preferably at thermoneutrality; and (c) parallel testing of experimental conditions or compounds in UCP1-null mice. With regard to human studies, similar temperature-dependent control of BAT thermogenesis can clearly be observed in adult humans (2, 4–6). Indeed, PET scanning of healthy adult humans detects active BAT depots, often only after cold exposure. Given that humans normally now live at thermoneutrality, the lack of PET positivity does not necessarily imply a lack of BAT deposits.
Potential Caveats in UCP1 Studies
Numerous studies using animal models have attempted to attribute an alteration in energy expenditure to underlying changes in UCP1 transcript levels in adipose tissue (BAT and WAT). Although UCP1 is certainly a major determinant of BAT thermogenic activity, this approach is oversimplified because BAT thermogenesis is also affected by a host of other factors, including defects in oxidative phosphorylation, fatty acid uptake and metabolism, and mitochondrial biogenesis. Importantly, UCP1 mRNA levels do not always reflect the protein levels and activity of UCP1 (26). For example, UCP1 transcripts are highly inducible following treatment with cAMP (2–4 h) or peroxisome proliferator–activated receptor γ (PPARγ) ligands; these high levels quickly return to basal levels when stimuli are removed. In contrast, these same two stimuli induce a slow but sustainable increase in UCP1 protein levels that persists for several days. The half-life of UCP1 mRNA is approximately 2.7 h, whereas that of the UCP1 protein is estimated to be 5–7 days in vivo (27, 28). Hence, the biological relevance of changing UCP1 transcript levels in BAT function should be evaluated with other parameters such as respiratory capacity, uncoupling capacity, and heat generation.
DEVELOPMENTAL LINEAGES OF THERMOGENIC ADIPOCYTES
The Two Types of Thermogenic Adipocytes Have Distinct Developmental Origins
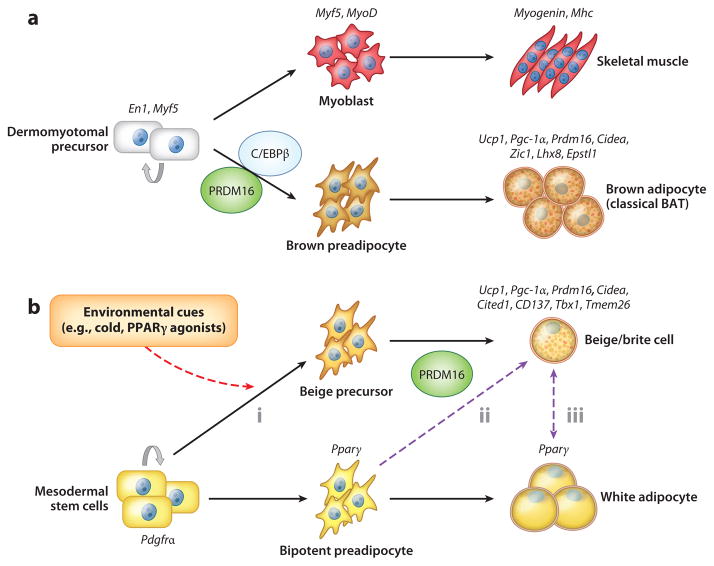
The current evidence indicates that two types of UCP1-positive thermogenic adipocytes, classical brown adipocytes and beige/brite cells, arise from distinct developmental lineages. Classical brown adipocytes are found mainly in interscapular and perirenal BAT deposits and develop during the prenatal stages (Figure 2a). Genetic fate-mapping experiments indicate that brown adipocytes in the interscapular region and skeletal muscle arise from cells that express Myf5, a gene previously assumed to be present almost exclusively in committed skeletal muscle precursors (29). Engrailed-1 (En1)-expressing cells in the central dermomyotome give rise to BAT, skeletal muscle, and dermis (30). These findings were further explored in a lineage-tracing study in which embryonic myoblasts were traced by using a myogenic marker gene, Pax7, in a time-inducible manner (31). On the basis of this study, the divergence of myoblasts and BAT progenitors occurs between embryonic days 9.5 and 11.5 in mice. Interestingly, the transcriptional profile of brown but not white adipocyte precursors resembles that of skeletal muscle cells (32). Similarly, the mitochondrial proteomic signature of BAT is highly related to that of skeletal muscle, but not to that of WAT (33).
Figure 2.
Hierarchical developmental relationships in brown and white adipocytes. The two types of thermogenic adipocytes (classical brown adipocytes and beige/brite cells) have separate developmental origins. (a) BAT and skeletal muscle originate from precursors in the dermomyotome that express Engrailed-1 (En1) and Myf5. Brown adipose fate in the somite is determined by transcriptional regulators, including PRDM16 (PRD1-BF-1-RIZ1 homologous domain–containing protein 16) and C/EBPβ(CCAAT/enhancer-binding protein-β), during embryonic development. (b) Beige/brite cells are not descended from Myf5-expressing cells. Adipocyte precursors that express PDGFα(36) differentiate into beige/brite cells mainly in subcutaneous white adipose tissue in response to several environmental cues, including chronic cold exposure, exercise, and peroxisome proliferator–activated receptor γ(PPARγ) agonists (red arrow), through the action of PRDM16. These cells appear to be derived from (i ) defined beige precursors, (ii ) directed differentiation from bipotent preadipocytes, or (iii ) transdifferentiation from mature white adipocytes. The dashed purple arrows depict hypothetical relationships that need further investigation.
As we discuss below, several key transcriptional regulators control the brown adipose–versus–skeletal muscle cell fate switch. Depletion or genetic ablation of such regulators, including PRDM16 (PRD1-BF-1-RIZ1 homologous domain–containing protein 16) and C/EBPβ (CCAAT/enhancer-binding protein-β), disrupt the determination of cell fate between brown adipocytes and myocytes from a common cellular precursor compartment. Indeed, presumptive brown adipocytes lacking PRDM16 or C/EBPβ display a muscle-like phenotype with elevated expression of skeletal muscle–selective markers such as Myogenin and Mhc (29, 34). In contrast, myogenin-deficient mice completely lack differentiated skeletal muscle but have an expanded BAT depot in the interscapular region (35). Together, these findings are consistent with the hypothesis that embryonic brown adipocytes share a direct common upstream precursor with skeletal muscle cells.
Beige/brite cells, the second type of UCP1-positive thermogenic adipocytes, are found sporadically in the subcutaneous WAT of adult animals that have been exposed to chronic cold, β-adrenergic agonists, PPARγ agonists, or endurance exercise (Figure 2b). This subset of inducible thermogenic adipocytes possesses many biochemical and morphological characteristics of classical brown adipocytes, including the presence of multilocular lipid droplets, enriched mitochondria, and UCP1 expression (11). However, these inducible thermogenic adipocytes arise from a Myf5-negative cell lineage and therefore have origins distinct from those of classical brown adipocytes. A recent study showed that Pdgfrα-positive progenitors in abdominal WAT give rise to UCP1-positive adipocytes in response to a β3-adrenoceptor agonist in vivo (36). Approximately 62% of adipocytes in inguinal WAT also arise from Myf5-positive cells (37), indicating high heterogeneity of adipogenic precursors in subcutaneous WAT. Because of such high heterogeneity, taking a random sampling in subcutaneous WAT (often of inguinal WAT) rather than harvesting the entire WAT could easily create large individual variations in the numbers of beige/brite cells.
Spiegelman’s group (38) recently isolated clonal populations of beige cells by immortalization of stromal vascular (SV) fractions from mouse subcutaneous WAT. The molecular signature of these beige cells is quite distinct from that of white adipocytes, suggesting that a defined set of WAT precursors gives rise to beige cells in response to cold. However, nearly all adipocytes within subcutaneous WAT can become UCP1-positive cells when mice are exposed to cold or treated with a β3-adrenoceptor agonist for a prolonged period (11). Furthermore, all preadipocytes isolated from the SV fraction of subcutaneous WAT express brown/beige cell markers, including UCP1, when cells, even at the postmitotic stages, are chronically treated with synthetic PPARγ agonists (39). Hence, cellular plasticity between white and beige adipocytes may exist at the precursor stage (Figure 2b).
The two types of thermogenic adipocytes are also distinct at gene expression levels. Although beige/brite cells and classical brown adipocytes share a number of BAT-specific or enriched genes, such as Ucp1, Pgc1a, Cidea, and Prdm16, these cell types also express unique markers that presumably reflect their developmental origins (38–43). For example, beige cells do not express myocyte-enriched genes such as Zic1, Lhx8, and Epstl1 but express beige-enriched genes such as Cited1, Tmem26, CD137, and Tbx1. Importantly, human BAT isolated from multiple body locations, including the supraclavicular and retroperitoneal regions, abundantly express markers of beige cells, indicating that these subpopulations of human BAT are molecularly similar to beige cells (38, 43). Recently, other researchers have found that the molecular signature of human BAT obtained from the interscapular region in infants or from the neck region in adults resembles that of classical brown adipocytes (44–46). Taken together, these findings indicate that humans also possess both beige/brite cells and classical brown adipocytes.
Cellular Plasticity of Brown Versus White Adipocytes: Does Transdifferentiation Occur?
Transdifferentiation is a process in which differentiated somatic cells transform into another cell type without undergoing a precursor cell stage or an intermediate pluripotent state. This cellular process is different from cell reprogramming, which involves the generation of embryonic stem cells or cells in a multipotent or a pluripotent state (47). Several papers indicate that transdifferentiation from mature white adipocytes into brown adipocytes may occur in response to chronic cold exposure. When mice are chronically exposed to a cold environment, UCP1-positive adipocytes at an intermediate state of white and brown fat (termed paucilocular adipocytes) are observed in WAT. These cells also contain mixed populations of mitochondria with classic brown fat and white fat mitochondria, suggesting intermediate steps in the process of transdifferentiation of mature white adipocytes into brown adipocytes (48). Another line of studies supporting white-to-brown-fat transdifferentiation is that newly formed beige/brite cells in subcutaneous WAT following adrenergic stimulation are mostly negative for BrdU or Ki67, suggesting that the formation of beige/brite cells does not require a mitotic proliferation of precursors (36, 49, 50). Furthermore, a recent lineage-tracing study showed that cold-induced beige/brite cells are converted into unilocular white adipocytes after 5 weeks of warm adaptation (51). These phenomena could also be explained by dedifferentiation of mature white adipocytes into bipotent or multipotent precursors with subsequent differentiation into beige/brite cells. In the future, single-cell resolution and live imaging should help to distinguish whether the transdifferentiation model or the dedifferentiation model is correct.
Physiological Roles of Brown Adipocytes Versus Beige/Brite Cells in Energy Metabolism
Among the major current research areas in beige/brite cells are defining their biological significance in energy homeostasis and testing their potential as targets for antiobesity therapies. Because the total amount of UCP1 protein found in beige/brite cells per animal is low, approximately 10% of those found in classical brown adipocytes, the relative contribution of beige/brite cells to overall UCP1-dependent thermogenic capacity may be marginal (26). However, three lines of evidence indicate that beige/brite cells significantly contribute to whole-body energy expenditure and propensity for weight gain, at least in rodents.
First, Kozak’s group (52–56) found a strong association between so-called browning propensity in WAT and susceptibility to diet-induced obesity. By generating recombinant inbred strains from A/J mice and C57BL/6J mice, this group found that mouse strains with a high propensity for browning (i.e., beige/brite cell formation) in retroperitoneal WAT by β3-adrenoceptor agonists are more resistant to diet-induced obesity. Importantly, UCP1 levels in interscapular BAT are indistinguishable among these strains. Furthermore, quantitative-trait-locus analyses of these strains identified several gene loci associated with Ucp1 transcription levels in WAT, but not in BAT, suggesting that there are discrete regulatory mechanisms by which to control the Ucp1 gene between WAT and BAT (55).
Second, several mouse models with an increased number of beige/brite cells in WAT are protected from diet-induced obesity (57–59). For example, transgenic expression of PRDM16 driven by the fat-specific Fabp4 gene promoter stimulates beige/brite formation in subcutaneous WAT without substantially changing UCP1 levels in classical BAT. Fabp4-PRDM16 transgenic mice also displayed increased energy expenditure, limited weight gain, and improved glucose tolerance under a high-fat diet, albeit with the caveat that ectopic expression of the transgene in nonfat tissues, such as macrophages, may partially contribute to these observed changes (58). Similarly, inhibition of retinaldehyde dehydrogenase (Aldh), which encodes a limiting enzyme that converts retinaldehyde to retinoic acid, preferentially induced beige/brite cell development in WAT without affecting UCP1 expression in classical BAT. Administration of Aldh1a1 antisense oligonucleotides in obese mice induced browning of white fat and limited diet-induced obesity (59).
Lastly, induction of beige/brite cells contributes to nonshivering adaptive thermogenesis in the absence of classical brown adipocytes (60). Myf5-derived cell-specific ablation of the type 1A bone morphogenetic protein receptor (Bmpr1A) causes a severe loss of classical brown adipocytes, which in turn activates beige/brite cell formation in WAT due to increased sympathetic input. Interestingly, these conditional Bmpr1A knockout mice exhibited thermogenic defects under short-time cold exposure (48 h) but maintained body temperature after prolonged cold exposure (11 days), indicating that beige/brite cells can compensate for the loss of classical brown adipocytes. Although no Cre lines currently exist for specifically targeting beige/brite cells, additional loss-of-function studies aimed at depleting beige/brite cells should help distinguish the unique in vivo functions of beige/brite cells from those of brown adipocytes.
CONTROL OF THERMOGENIC ADIPOCYTE DEVELOPMENT AND FUNCTION
Transcriptional Regulators of Brown and Beige/Brite Cell Development
Transcriptional cascades that control the process of adipocyte differentiation are well conserved between brown and white adipocytes. PPARγ and the C/EBPs are the major transcription factors that control adipocyte differentiation (61). Indeed, genetic ablation of PPARγ completely disrupts adipocyte differentiation in both white fat and brown fat. C/EBPα is required only for the formation of white fat, but not for brown fat, suggesting a possible role for other C/EBP family members in brown fat development. C/EBPβ is more highly expressed in brown adipocytes than in white adipocytes and plays an important role in regulating the thermogenic gene program in brown adipocytes (34, 62, 63). C/EBPβ and -δ, as well as other transcription factors, also participate in the transcriptional cascade of adipogenesis by regulating PPARγ gene expression (64). Intriguingly, brown adipocyte differentiation requires PPARγ, but ectopic expression of PPARγ in fibroblasts or mesenchymal cells induces only white adipocyte differentiation, indicating that additional factors are required to induce brown adipocyte differentiation (12). A number of positive or negative transcriptional regulators of brown adipocyte and beige/brite cell development were recently identified; these regulators are summarized below.
PGC-1α and its modulators
PPARγ coactivator-1α (PGC-1α) was originally identified from brown fat cells as a cold-inducible transcriptional coactivator of PPARγ (65). PGC-1α is a critical regulator of mitochondrial biogenesis and oxidative metabolism in many cell types, including brown fat and skeletal muscle. Ectopic expression of PGC-1α in white adipocytes induces the expression of mitochondrial genes and thermogenic genes (65, 66). Consistent with the results, deletion of PGC-1α reduces the capacity for cold-induced thermogenesis (CIT) in vivo and the response to cAMP signaling in cultured brown fat cells (67, 68). However, loss of PGC-1α does not affect brown adipocyte differentiation (67–69), indicating that PGC-1α is dispensable for brown adipose cell fate determination.
Several transcriptional regulators control brown fat development and thermogenic function by modulating either gene expression or activity of PGC-1α. For example, RIP140 is a corepressor of many nuclear receptors and coregulators, including PGC-1α (70). Loss of RIP140 induces the formation of beige/brite cells in WAT (71). Similarly, SRC2/TIF2/GRIP1, a member of the steroid receptor coactivator (SRC) family, represses PGC-1α transcriptional activity. SRC2 deletion leads to increases in adaptive thermogenesis and energy expenditure in vivo (72). Retinoblastoma (Rb) protein and p107, another member of the Rb pocket protein family, also negatively regulate PGC-1α gene expression. Adipocytes derived from Rb-deficient fibroblasts have high mitochondrial content and elevated expression of UCP1, PGC-1α, and mitochondrial genes (73). In addition, Rb deletion results in the expansion of interscapular BAT in vivo (74). p107-deficient mice also exhibit increased amounts of beige/brite cells in WAT with high UCP1 and PGC-1α expression (75). Lastly, twist-1 is a negative regulator of PGC-1α function in brown fat. Indeed, heterozygous twist-1+/− mice exhibit an induction of brown fat–selective genes, whereas transgenic mice overexpressing twist-1 repress these genes in a PGC-1α-dependent fashion (76).
PRDM16
PRDM16 is a 140-kDa zinc-finger protein that is highly expressed in BAT (77). Ectopic expression of PRDM16 in white fat precursors or in myoblasts induces a brown fat gene program, including mitochondrial biogenesis, increased cellular respiration, and expression of brown fat–selective genes. Mechanistically, PRDM16 increases the transcriptional activities of PGC-1α, PPARγ, and C/EBPs through direct interactions. In addition, PRDM16 forms a transcriptional repressor complex with C-terminal binding proteins and represses white fat–selective gene expression (78). These results suggest that PRDM16 is a coregulatory protein that functions as a bidirectional molecular switch in brown fat development through multiple protein-protein interactions. Ectopic expression of PRDM16 and C/EBPβ is sufficient to convert nonadipogenic fibroblasts, including skin fibroblasts from mice and humans, to fully functional brown fat in vivo (34). Transplantation of fibroblasts expressing these two factors into mice gives rise to an ectopic fat pad that displays the morphological and biochemical characteristics of brown fat. As with endogenous brown fat, this synthetic brown fat tissue is a sink for glucose uptake, as determined by 18FDG-PET scans.
Recent studies have identified several upstream regulators of PRDM16 or C/EBPβ that control brown fat development. For example, early B cell factor 2 (EBF2) activates PRDM16 transcription and induces a brown fat gene program in myoblasts and in white adipocytes (79). EHMT1 (euchromatic histone-lysine N-methyltransferase 1) forms a transcriptional complex with PRDM16 and determines brown adipose cell fate by activating PRDM16 transcription (80). Plac8 is an upstream activator of C/EBPβ transcription and induces brown fat differentiation (81). In contrast, TLE3 is a white fat–selective cofactor that antagonizes the function of PRDM16 and suppresses brown fat differentiation and thermogenesis (82). In addition to these transcriptional regulators, several microRNAs such as miR-133, miR-193b, and miR-365 target PRDM16 and negatively regulate brown fat development (83–85). Furthermore, miR-196a activates C/EBPβ expression and induces beige/brite cell differentiation by directly repressing HoxC8, a negative regulator of C/EBPβ (86).
Lastly, the protein stability of PRDM16 is highly controlled by PPARγ agonists. Synthetic PPARγ agonists such as thiazolidinedione induce beige/brite cell differentiation in WAT (41, 87–92). Ohno et al. (39) found that PPARγ agonists such as rosiglitazone induce white-to-brown-fat conversion by extending the half-life of the PRDM16 protein. These studies suggest that the PRDM16-C/EBPβ-PPARγ pathway plays a central role in brown and beige/brite cell development.
Forkhead box C2
Forkhead box C2 (FoxC2) is a member of the forkhead/winged helix transcription factor family that is expressed exclusively in the adipose tissues of humans and mice. Transgenic expression of FoxC2 in WAT induces the formation of beige/brite cells, with increased mitochondria and elevated expression of thermogenic genes, including UCP1 and PGC-1α (57). This browning effect by FoxC2 is due to the elevated expression of the RIa subunit of PKA, which sensitizes cells to cAMP signaling through the β-adrenergic pathway. Importantly, FoxC2 transgenic mice gain less weight under a high-fat diet and are protected from obesity-associated disorders such as insulin resistance and hypertriglyceridemia (57).
Signaling Pathways That Control Brown and Beige/Brite Cell Development
Given that BAT functions as a defense against hypothermia and obesity, brown and beige/brite cell development is highly regulated by changes in temperature and nutrition demands. Indeed, β-AR signaling is a dominant activator pathway not only for BAT thermogenesis but also for the development of brown adipocytes and beige/brite cells. Additionally, previously unappreciated signaling pathways have recently been reported, as summarized below.
β-Adrenoceptor signaling
Norepinephrine released from the sympathetic nerve terminals binds to β-ARs and increases intercellular cAMP levels, leading to the phosphorylation of PKA and subsequently to p38MAPK activation. Phosphorylation of p38MAPK indirectly triggers expression of UCP1 and PGC-1α by directly kinasing several transcriptional regulators such as ATF2 and PGC-1α (93). Among the three subtypes of β-ARs (β1-, β2-, and β3-ARs), β1-AR is considered important for proliferation of classical brown adipocyte precursors in response to norepinephrine, whereas β3-AR plays the major role in thermogenic function of mature brown adipocytes (94, 95). However, only β3-AR signaling appears to be crucial for beige/brite cell development. Indeed, in β3-AR knockout mice, cold-induced beige/brite cell development is severely impaired, whereas classical brown adipocyte development remains unchanged. Furthermore, β3-AR agonists, but not β1-AR agonists, selectively induce the formation of beige/brite cells in WAT (48, 96). The distinct functions of β1-AR and β3-AR in human beige/brite cell development remain unknown.
Nitric oxide signaling
Nitric oxide (NO) is a short-lived gaseous signaling molecule that is synthesized by endothelial cells and other cell types. Guanosine 3′5′-monophosphate (cGMP) is produced by NO-sensitive guanylyl cyclase and activates cGMP-dependent protein kinase (PKG). cGMP treatment in brown adipocytes induces UCP1 expression and mitochondrial biogenesis in a PKG-dependent manner (97). Mechanistically, the cGMP-PKG signaling pathway activates the phosphatidylinositol 3-kinase type I (PI3K)-Akt cascade by inhibiting RhoA and Rho-associated kinase. In addition, cGMP signaling induces beige/brite cell development in WAT (98). The physiological role of NO signaling in BAT thermogenesis and development remains unknown.
Transient receptor potential vanilloid signaling
Transient receptor potential vanilloid (TRPV) is a family of transient receptor potential ion channels. TRPV1 is activated by a number of environmental cues such as heat greater than 43°C and the pungent compounds in chili peppers. Interestingly, nonpungent capsaicin analogs (capsinoids) activate gastrointestinal TRPV1 and induce BAT thermogenesis in humans (99) and in rodents (100, 101). In contrast, TRPV4 expressed in adipocytes negatively regulates a BAT-selective thermogenic program in a cell-autonomous manner. Inhibition of TRPV4 signaling in white adipocytes by a TRPV4 antagonist or by shRNA-mediated knockdown activates UCP1 and PGC-1α expression and cellular respiration through the ERK1/2 protein kinases (102).
PI3K signaling (via PTEN)
Phosphatase and tensin homolog (PTEN) counteracts the activity of PI3K, a major kinase mediator of insulin, insulin-like growth factors, and other growth factors. Ortega-Molina et al. (103) showed that PTEN positively regulates a BAT-selective thermogenic program by blocking the PI3K pathway. Intriguingly, mouse embryonic fibroblasts (MEFs) over-expressing PTEN are efficiently reprogrammed to functional brown adipocytes by PRDM16 and C/EBPβ. Importantly, pharmacological PI3K inhibitors increase BAT thermogenesis and whole-body energy expenditure.
Endogenous Hormones That Control Brown and Beige/Brite Cell Development
Endogenous proteins and polypeptides that induce brown and beige/brite cell development have recently attracted much attention because they may lead to a new therapeutic intervention for obesity. Important aspects to consider are (a) sources of signal (i.e., organs or cells from which such signaling entities are derived) and (b) specificity of action (i.e., distribution and regulation of corresponding receptors). The subsections below describe recently identified endogenous molecules that positively or negatively control brown and beige/brite cells.
Transforming growth factor family members
BMPs belong to the transforming growth factor (TGF)-β superfamily. BMP7 treatment of fibroblast cultures or adipogenic precursors induces brown adipogenic regulators such as PRDM16 (42, 104). Conversely, BMP7-deficient embryos possess reduced amounts of BAT and lack UCP1 expression (104). Another BMP family member, BMP4, also directs mesenchymal cells to an adipocyte lineage and activates beige/brite cell differentiation in the subcutaneous WAT of mice expressing BMP4 (105). BMP8b acts on mature brown adipocytes as well as in the hypothalamus to positively regulate BAT thermogenesis but does not affect differentiation or commitment of brown adipocytes (106).
In contrast, several TGF-β superfamily members, including GDF-8 (myostatin), TGF-β1, and activins, negatively regulate brown fat differentiation and BAT thermogenesis. Genetic deletion of Smad3, a major mediator of TGF-β signaling, enhances the formation of beige/brite cells in WAT and increases whole-body energy expenditure in mice (107). Importantly, inhibition of TGF-β signaling, by administering either a neutralizing anti-TGF-β antibody or a soluble form of activin receptor type IIB fused to an immunoglobulin Fc domain, activates BAT thermogenesis and protects animals from diet-induced obesity and insulin resistance (107–109).
Fibroblast growth factors
In contrast to most fibroblast growth factors (FGFs) that act in an autocrine or a paracrine fashion, FGF-19 (FGF-15 in the mouse), FGF-21, and FGF-23 are endocrine forms of FGFs. Endocrine FGFs bind to cell surface coreceptors α-Klotho and/or β-Klotho (110). Transgenic expression of FGF-19 in mice increases metabolic rate and reduces fat mass, in part through activating BAT thermogenesis (111, 112). In neonatal mice, circulating FGF-21 secreted from the liver is acutely elevated at birth in response to PPARα and activates BAT thermogenesis (113). FGF-21 also induces beige/brite cell differentiation in WAT by enhancing adipose tissue PGC-1α protein levels (114). Although chronically elevated circulating FGF-21 causes bone loss in mice (115), adipose-derived FGF-21 remains local and does not contribute to circulating levels. Thus, inducing FGF-21 in adipose tissues may constitute an effective treatment for obesity and insulin resistance, without affecting bone mass.
Irisin
Irisin is a newly identified myokine that is cleaved from a membrane protein, fibronectin type III domain–containing 5 (FNDC5) (116). Endurance exercise or PGC-1α overexpression stimulates FNDC5 expression in skeletal muscle and increases circulating levels of Irisin. Irisin treatment in primary adipocytes or adenoviral delivery of FNDC5 in the mouse liver induces a thermogenic gene program in WAT and protects animals from diet-induced obesity. In addition, Irisin fused with the Fc fragment of human IgG in CD137+ preadipocyte populations stimulates beige cell differentiation (38). Although PPARα has been suggested to be a downstream mediator of Irisin actions, identification of the cognate Irisin receptor and its signaling cascades requires further investigation.
Cardiac natriuretic peptides
Atrial natriuretic peptide (ANP) and brain NP (BNP) are released from the heart and are important endocrine regulators of fluid and hemodynamic homeostasis. The actions of NPs are mediated through NP receptor A (NPRA), whereas another type of NP clearance receptor, NPRC, also binds ANP and BNP to remove them from circulation. Collins’s group (117) showed that cold exposure induces circulating levels of NPs and NPRA expression in adipose tissues. Administration of BNP in mice or in vitro treatment of NPs in human adipocytes activates the BAT-selective thermogenic gene program, mitochondria biogenesis, and uncoupling respiration in a p38MAPK-dependent fashion. Furthermore, WAT from NPRC-deficient mice contains a significantly higher number of beige/brite cells. Because higher circulating levels of NPs are associated with heart failure and cardiac cachexia, it becomes important to define the therapeutic window of these peptides that will increase energy expenditure without adversely affecting the heart or other tissues.
Prostaglandins
Cyclooxygenase (COX) is a rate-limiting enzyme in prostaglandin (PG) synthesis. Chronic cold exposure induces COX2 gene expression and enhances the release of PG, PGE2, and PGI2 in WAT. Transgenic expression of COX2 in WAT or PGI2 treatment in adipocyte precursors induces the expression of BAT-selective genes such as Ucp1 and Cidea. Conversely, COX2 gene ablation or pharmacological inhibition of COX activity impairs beige/brite cell formation in WAT, which indicates that COX-dependent production of PG is an important step for cold-induced beige/brite cell formation (118, 119).
Interorgan Networks That Control BAT Development and Thermogenesis
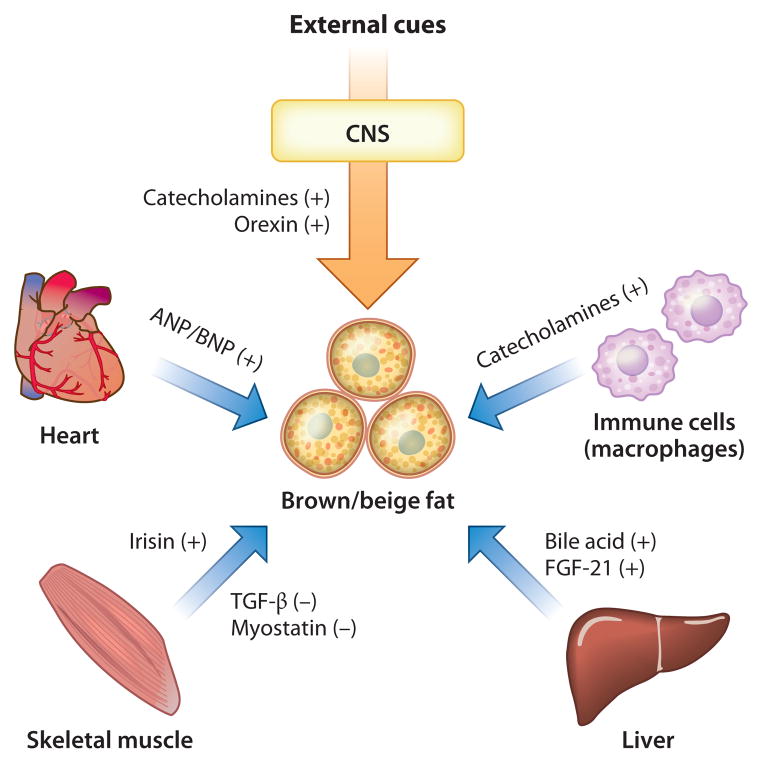
As we discuss above, a number of signaling entities are derived from central and peripheral tissues and contribute to the regulation of brown fat development and thermogenesis. Figure 3 illustrates the recently appreciated BAT-mediated interorgan networks. The central nervous system produces catecholamines and orexin (120), both powerful activators of brown fat development and thermogenic function. Activated macrophages residing in adipose tissues also secrete catecholamines, and these cells regulate adaptive thermogenesis (121). The skeletal muscle secretes both positive and negative regulators of beige/brite cell development such as Irisin (116) and TGF-β (107), respectively. From the heart, NPs induce BAT thermogenesis and browning of white fat (117). From the liver, bile acid (122) and FGF-21 (113, 114) are important mediators. Furthermore, a recent study identified a new BAT-liver connection in which obesity-induced expression of hepatic glucokinase represses BAT thermogenesis by modulating sympathetic nerve activity via the afferent vagus nerve originating in the liver (123).
Figure 3.
BAT-mediated interorgan networks. Several endocrine factors regulate BAT development and thermogenesis and mediate interorgan communication with central and peripheral tissues. Abbreviations: ANP, atrial natriuretic peptide; BNP, brain natriuretic peptide; CNS, central nervous system; FGF, fibroblast growth factor; TGF, transforming growth factor.
Just as other tissues affect brown fat development and thermogenesis, brown fat in turn contributes to glucose and lipid homeostasis in peripheral tissues. Intriguingly, ectopic expression of UCP1 in epididymal WAT improves both leptin and insulin sensitivities in mice. Blocking afferent nerve signals from intra-abdominal WAT by surgical and pharmacological methods completely blunts the improvement in leptin but not insulin sensitivity (124). Moreover, transplantation of BAT into the interabdominal regions of age- and sex-matched mice significantly improves whole-body insulin sensitivity in mice. BAT transplantation increases insulin-stimulated glucose uptake in endogenous BAT, WAT, and the heart, in part through BAT-derived interleukin 6 (125). In the future, and as this research area progresses, other BAT-mediated interorgan networks utilizing new signaling pathways are likely to emerge.
SIGNIFICANCE OF BAT IN HUMANS
Cold-Activated BAT Detected by 18FDG-PET/CT in Adult Humans
BAT depots are abundant in human newborns, but BAT rapidly decreases in size during childhood. For several decades, the existence of BAT in the general adult population was questioned and thought to be restricted to patients with pheochromocytomas (neuroendocrine tumors of the adrenal medulla) and to workers exposed to chronic cold (8, 126). Therefore, BAT was believed to play a negligible role in adult humans. However, this notion was challenged by a clinical study using 18FDG-PET imaging and that reported symmetrical 18FDG uptake in adipose tissue of the shoulder and thoracic spine regions in the absence of any tumor (2). The prevalent 18FDG uptake in supraclavicular area (USA) fat is increased significantly at lower temperature and is reduced by pretreatment with β-adrenergic blockers, collectively suggesting that USA fat reflects metabolically active BAT. Prospective 18FDG-PET/CT studies in healthy volunteers confirm the presence of active BAT depots in adult humans (3–6). Indeed, when subjects were exposed to mild cold (19°C) with light clothing for 2 h, some participants showed substantial 18FDG uptake into adipose tissue in the supraclavicular and paraspinal regions, whereas when subjects were kept at a warm temperature (27°C), no detectable uptake was observed. Histological examinations further confirmed the presence of UCP-1-positive multilocular adipocytes in these regions.
Cold-activated BAT detected by 18FDG-PET/CT in human adults is localized mainly to the supraclavicular and paravertebral regions. In some cases, weaker 18FDG uptake by BAT was also found in the cervical, axillary, epicardial, mediastinal, and perirenal regions. BAT is abundant in the interscapular region in infants and children but disappears rapidly in adolescence and is completely absent after the third decade of life. The striking differences in localization of human BAT between infants and adults suggest that molecular differences exist between these stage-specific BAT depots. In fact, gene expression profiles indicate that interscapular BAT from the neck regions of infants resembles that of classical brown adipocytes (44–46), whereas other BAT depots in adults appears to be composed mainly of beige cells (38, 43).
The mass of human BAT estimated from 18FDG-PET/CT images is largely variable, fluctuating from 0.02 to 288 g in adults (Table 1) (3, 4, 127–129). However, this range may be either an underestimation, because 18FDG-PET/CT detects only metabolically active BAT depots and does not detect BAT in the resting state, or an overestimation, given that human BAT depots contain a mixture of multilocular brown adipocytes interspersed with a greater volume of unilocular white adipocytes.
Table 1.
Prevalence and estimated mass of human BAT
| Reference | Number of patients or subjects | Age (years) | Prevalence (%) | Influencing factors | Mass (g) | BAT-dependent CIT (kcal/day)a |
|---|---|---|---|---|---|---|
| Retrospective studies | ||||||
| 130 | 905 | 58 (1–93) | 7 | Sex, outdoor temperature, plasma glucose | ||
| 3 | 1,972 | 59 | 5 | Age, sex, adiposity, outdoor temperature | Male, 11.6 (0.5–42.0); female, 12.3 (1.1–170.0) | |
| 131 | 3,614 | 52 (13–88) | 5 | Sex, season | ||
| 127 | 3,604 | 48 (11–82) | 5 | Age, sex, adiposity | 25.2 (0.02–287.9) | |
| 132 | 2,934 | 36 (18–87) | 9 | Age, sex, adiposity, blood glucose | ||
| 7 | 4,842 | 58 (2–94) | 7 | Age, sex, adiposity, outdoor temperature | ||
| 133 | 71 | 15 (6–20) | 42 | Muscle mass | ||
| Prospective studies | ||||||
| 4 | 24 | 24 (18–32) | 95 | Adiposity | Lean, 117; obese, 69.3 | |
| 6 | 56 | 37 (23–65) | 33 | Age, adiposity, outdoor temperature | ||
| 128 | 27 | 40 | 70 | 34 (9–90) | 286 | |
| 134 | 162 | 32 (20–73) | 59 | Age, adiposity | ||
| 135 | 13 | 23 (20–27) | 46 | 368 | ||
| 129 | 25 | 30 | 36 | Lean body mass, sex | 59.1 (32–85) | 275 |
Brown adipose tissue (BAT)-dependent energy expenditure is calculated as the difference in cold-induced thermogenesis (CIT) between BAT-positive and BAT-negative subjects.
Prevalence and Activity of BAT: Effects of Ambient Temperature, Season, and Aging
Various physiological and environmental factors influence the prevalence and activity of human BAT as assessed by 18FDG-PET/CT (Table 1). Retrospective studies estimate that the prevalence of BAT in the population is less than 10% (3, 7, 127, 130–132), except in the case of children (133), whereas prospective studies in healthy volunteers suggests that this value may increase to 30% (4, 6, 128, 129, 134, 135). Such a wide discrepancy may reflect 18FDG-PET/CT scanning carried out at different temperatures: In prospective studies, 18FDG-PET/CT was performed after acute cold exposure at 16–19°C for 1–2 h, whereas retrospective studies were performed mostly at ambient room temperatures (22–26°C). Acute cold exposure presumably increases the intensity of the 18FDG-PET signal in BAT, yielding a high prevalence of BAT detection. Indeed, no BAT signals were detected at 27–28°C, even in subjects who showed high BAT activity after cold exposure (4, 6).
Seasonal variations in outdoor temperatures also significantly influence the prevalence of BAT detection by 18FDG-PET/CT scanning. As predicted, both the prevalence and absolute levels of BAT in human adults are higher during the colder winter temperatures and are lower during the summer (Table 1). The seasonal variation of BAT activity within subjects (6) suggests that human BAT is a dynamic tissue and is responsive to environmental stimuli. Consistent with this notion, repeated cold exposure over 6 weeks resulted in the induction of BAT in subjects who previously had undetectable BAT levels before the cold exposure (136). This finding is also consistent with reports that adult human BAT consists largely of inducible beige/brite cells, as this type of BAT is induced during chronic cold exposure, as discussed above (38, 43).
In addition to temperature effects, the prevalence and activity of BAT are substantially modulated with age (Table 1). A study of 162 healthy participants aged 20–73 years revealed that the prevalence of cold-activated BAT reached 50% in young subjects (in their twenties) but dropped precipitously to less than 10% in older subjects (>50 years of age) (134). Most retrospective studies for adults also report an inverse correlation of BAT with age (3, 127, 132). Studies in children and adolescents show that BAT is present in more than 40% of subjects, with the highest BAT activity found in young teenagers between 13 and 15 years of age (133). Collectively, these studies imply that the prevalence and activity of BAT decrease as we age.
A number of cross-sectional studies also show that the prevalence and activity of BAT are inversely correlated with BMI, body fat, and visceral fat (Table 1). Conversely, a longitudinal study found that lower body weights were associated with more active BAT (132). Intriguingly, this inverse relation between body weight and BAT appears to be relevant only in older subjects. In adults (in their forties), visceral fat mass was twice as high in subjects with undetectable BAT (BAT negative) as in subjects with detectable BAT (BAT positive). In younger subjects, all adiposity-related parameters were comparable, regardless of whether the subjects were BAT negative or BAT positive (134). A retrospective study also reports an age-dependent correlation between BAT mass and BMI (127). These studies suggest that the implied protective role of BAT against body fat accumulation is age dependent.
Effects of Endocrine and Genetic Factors on Human BAT Activity
Medical conditions and pharmacological treatments that affect the β-adrenergic system appear to have profound effects on human BAT. As mentioned above, an intense 18FDG uptake in BAT is observed in patients with a catecholamine-secreting tumor (pheochromocytoma); uptake disappears after surgical removal of the tumor (137). A significant and positive correlation between plasma norepinephrine level and BAT activity has also been noted (138). In addition, 18FDG uptake in BAT is significantly attenuated by pretreatment with the β-adrenergic blocker propranolol (2), whereas 18FDG uptake in BAT is increased after injection of the sympathomimetic agent ephedrine (139). These results collectively indicate that the stimulatory effect of the β-adrenergic system on BAT activity is conserved in adult humans.
Several retrospective studies showed that females exhibit higher BAT prevalence than do males (Table 1). Although it is presumed that sex hormones are involved in controlling BAT activity, as no sexual dimorphism in BAT is observed prior to puberty (133), no apparent sex differences in BAT activity are observed after acute cold exposure (Table 1). A stimulatory role of thyroid hormones is also suggested by a case report describing increased 18FDG uptake after T4 treatment in a thyroidectomized patient (140).
In addition to hormonal factors, single-nucleotide polymorphisms (SNPs) affect BAT activity in humans. For example, the G allele of the UCP1 gene at −3826 A/G is associated with lower promoter activity and lower expression levels of UCP1 in intraperitoneal adipose tissue (141, 142). SNPs within the UCP1 gene (−3826 A/G) and the β3-AR gene (64 Trp/Arg) act synergistically to lower BAT prevalence and activity (143). This finding is in agreement with reports that these two particular polymorphisms are associated with lower resting energy expenditure and with decreased cold-induced or postprandial thermogenesis (144, 145).
Contribution of BAT to the Regulation of Whole-Body Energy Expenditure
The presence of cold-activated BAT implies a significant contribution of BAT to adaptive thermogenesis, particularly to CIT in humans. In fact, CIT is significantly higher in BAT-positive subjects than in BAT-negative subjects, although basal energy expenditure under warm conditions is comparable between the two groups. CIT and 18FDG uptake in BAT are also positively correlated (135). As neither shivering nor a change in 18FDG uptake is observed in skeletal muscle during acute cold exposure, these findings suggest that BAT significantly contributes to whole-body energy expenditure in adult humans. In addition, BAT may also contribute to diet-induced thermogenesis, as energy expenditure after oral ingestion of food is significantly higher in BAT-positive subjects than in BAT-negative subjects, particularly during the initial period (S. Aita, T. Yoneshiro & M. Saito, unpublished observations). Hence, BAT is most likely a major site for adaptive thermogenesis in adult humans.
BAT as a Therapeutic Target for Reducing Body Fat
On the basis of the difference in CIT between BAT-positive and BAT-negative subjects, BAT-dependent energy expenditure is estimated at approximately 200–400 kcal/day under cold conditions (Table 1); in warm conditions this value would be much lower. Regardless, small differences of just 10 kcal/day can lead to substantial differences in body fat in the long term. For example, a difference of 10 kcal/day is equivalent to 1.1 g of body fat per day and 4 kg (~10 lb) of body fat per 10 years, which matches well with the gradual accumulation of body fat during aging. Indeed, BAT-negative subjects in their forties possess approximately 6 kg more body fat than do BAT-positive subjects (134).
Hence it is tempting to consider BAT as a therapeutic target for combating human obesity. Importantly, recent studies clearly indicate that adult human BAT can be recruited by external cues such as chronic cold exposure. When nonobese BAT-negative subjects were repeatedly exposed to cold for 6 weeks, BAT recruitment was observed in association with an increase in CIT (136). Furthermore, TRPV1 agonists found in food ingredients such as capsaicin and its nonpungent analogs (capsinoids) recruit new BAT in adult humans (99, 136), indicating that such food ingredients could be eventually exploited as antiobesity agents.
PERSPECTIVES AND NEW CHALLENGES
Contrary to conventional dogma, it is now appreciated that significant amounts of active BAT depots are present in adult humans and are likely involved in energy homeostasis and regulation of adiposity. Thus, targeting components that contribute to BAT activity may provide new therapeutic strategies for combating human obesity and related metabolic disorders. However, better tools are needed in assessing BAT levels and function in humans. For example, the use of 18FDG-PET/CT is limited because of its high cost and radiation exposure. Although this technique provides information regarding the glucose uptake capacity in BAT, it fails to accurately measure BAT mass per se or thermogenic activity of BAT. Magnetic resonance imaging may circumvent some of these issues in the evaluation of BAT mass and its metabolic activity but also carries a high cost (146). Another important area of research will be to understand the regulatory mechanisms of age-related reduction in BAT mass in humans. Such insights would be a prerequisite for developing feasible and efficient methods to recruit and activate BAT in obese adults as a possible therapeutic modality.
SUMMARY POINTS.
External cues at posttranscriptional levels tightly regulate the thermogenic activity of UCP1. Direct binding of long-chain free fatty acids to the UCP1 protein is required for the uncoupling capacity of UCP1.
Rodents and humans possess two types of thermogenic adipocytes: classical brown adipocytes and beige/brite cells that originate from distinct developmental lineages.
The PRDM16-C/EBPβ-PPARγ pathway plays a central role in the fate determination and differentiation of brown adipocytes and beige/brite cells.
Brown fat development and function are regulated by several hormonal factors derived from central and peripheral tissues, such as liver, skeletal muscle, heart, and immune cells.
The prevalence and activity of adult human BAT are greatly influenced by ambient temperature, season, and aging and are inversely correlated with body fat content.
Adult human BAT can be recruited by chronic cold exposure and TRPV1 agonists. Therefore, BAT is a promising therapeutic antiobesity target.
Acknowledgments
We thank Louis Sharp and Dylan Lowe for their critical reading of the manuscript. This work is funded by NIH grants (DK097441 and DK087853) to S.K. and by the Japan Society for the Promotion of Science grants (15081201 and 22590227) to M.S. We also acknowledge support from PRESTO by the Japan Science Technology Agency and from the Pew Charitable Trusts to S.K.
Glossary
- BAT
brown adipose tissue
- UCP1
uncoupling protein 1
- 18FDG-PET
18fluoro-labeled 2-deoxyglucose positron emission tomography
- CT
computed tomography
- BMI
body mass index
- WAT
white adipose tissue
- β-AR
β-adrenoceptor
- FFAs
free fatty acids
- LCFA
long-chain fatty acid
- PPARγ
peroxisome proliferator–activated receptor γ
- PRDM16
PRD1-BF-1-RIZ1 homologous domain–containing protein 16
- C/EBPβ
CCAAT/enhancer-binding protein-β
- CIT
cold-induced thermogenesis
- TRPV
transient receptor potential vanilloid
Footnotes
DISCLOSURE STATEMENT
The authors are not aware of any affiliations, memberships, funding, or financial holdings that might be perceived as affecting the objectivity of this review.
Contributor Information
Shingo Kajimura, Email: skajimura@diabetes.ucsf.edu.
Masayuki Saito, Email: saito@tenshi.ac.jp.
LITERATURE CITED
- 1.Spiegelman BM, Flier JS. Obesity and the regulation of energy balance. Cell. 2001;104:531–43. doi: 10.1016/s0092-8674(01)00240-9. [DOI] [PubMed] [Google Scholar]
- 2.Nedergaard J, Bengtsson T, Cannon B. Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocr Metab. 2007;293:E444–52. doi: 10.1152/ajpendo.00691.2006. [DOI] [PubMed] [Google Scholar]
- 3.Cypess AM, Lehman S, Williams G, Tal I, Rodman D, et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med. 2009;360:1509–17. doi: 10.1056/NEJMoa0810780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, et al. Cold-activated brown adipose tissue in healthy men. N Engl J Med. 2009;360:1500–8. doi: 10.1056/NEJMoa0808718. [DOI] [PubMed] [Google Scholar]
- 5.Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, et al. Functional brown adipose tissue in healthy adults. N Engl J Med. 2009;360:1518–25. doi: 10.1056/NEJMoa0808949. [DOI] [PubMed] [Google Scholar]
- 6.Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T, et al. High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes. 2009;58:1526–31. doi: 10.2337/db09-0530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ouellet V, Routhier-Labadie A, Bellemare W, Lakhal-Chaieb L, Turcotte E, et al. Outdoor temperature, age, sex, body mass index, and diabetic status determine the prevalence, mass, and glucose-uptake activity of 18F-FDG-detected BAT in humans. J Clin Endocrinol Metab. 2012;96:192–99. doi: 10.1210/jc.2010-0989. [DOI] [PubMed] [Google Scholar]
- 8.Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84:277–359. doi: 10.1152/physrev.00015.2003. [DOI] [PubMed] [Google Scholar]
- 9.Nedergaard J, Cannon B. The changed metabolic world with human brown adipose tissue: therapeutic visions. Cell Metab. 2010;11:268–72. doi: 10.1016/j.cmet.2010.03.007. [DOI] [PubMed] [Google Scholar]
- 10.Enerback S. Human brown adipose tissue. Cell Metab. 2010;11:248–52. doi: 10.1016/j.cmet.2010.03.008. [DOI] [PubMed] [Google Scholar]
- 11.Frontini A, Cinti S. Distribution and development of brown adipocytes in the murine and human adipose organ. Cell Metab. 2010;11:253–56. doi: 10.1016/j.cmet.2010.03.004. [DOI] [PubMed] [Google Scholar]
- 12.Kajimura S, Seale P, Spiegelman BM. Transcriptional control of brown fat development. Cell Metab. 2010;11:257–62. doi: 10.1016/j.cmet.2010.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tseng YH, Cypess AM, Kahn CR. Cellular bioenergetics as a target for obesity therapy. Nat Rev Drug Discov. 2010;9:465–82. doi: 10.1038/nrd3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu J, Cohen P, Spiegelman BM. Adaptive thermogenesis in adipocytes: Is beige the new brown? Genes Dev. 2013;27:234–50. doi: 10.1101/gad.211649.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kramarova TV, Shabalina IG, Andersson U, Westerberg R, Carlberg I, et al. Mitochondrial ATP synthase levels in brown adipose tissue are governed by the c-Fo subunit P1 isoform. FASEB J. 2008;22:55–63. doi: 10.1096/fj.07-8581com. [DOI] [PubMed] [Google Scholar]
- 16.Golozoubova V, Hohtola E, Matthias A, Jacobsson A, Cannon B, Nedergaard J. Only UCP1 can mediate adaptive nonshivering thermogenesis in the cold. FASEB J. 2001;15:2048–50. doi: 10.1096/fj.00-0536fje. [DOI] [PubMed] [Google Scholar]
- 17.Nicholls DG. Hamster brown-adipose-tissue mitochondria: the chloride permeability of the inner membrane under respiring conditions, the influence of purine nucleotides. Eur J Biochem. 1974;49:585–93. doi: 10.1111/j.1432-1033.1974.tb03862.x. [DOI] [PubMed] [Google Scholar]
- 18.Fedorenko A, Lishko PV, Kirichok Y. Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell. 2012;151:400–13. doi: 10.1016/j.cell.2012.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stefl B, Janovska A, Hodny Z, Rossmeisl M, Horakova M, et al. Brown fat is essential for cold-induced thermogenesis but not for obesity resistance in aP2-Ucp mice. Am J Physiol Endocr Metab. 1998;274:E527–33. doi: 10.1152/ajpendo.1998.274.3.E527. [DOI] [PubMed] [Google Scholar]
- 20.Grundlingh J, Dargan PI, El-Zanfaly M, Wood DM. 2,4-Dinitrophenol (DNP): a weight loss agent with significant acute toxicity and risk of death. J Med Toxicol. 2011;7:205–12. doi: 10.1007/s13181-011-0162-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rothwell NJ, Stock MJ. A role for brown adipose tissue in diet-induced thermogenesis. Nature. 1979;281:31–35. doi: 10.1038/281031a0. [DOI] [PubMed] [Google Scholar]
- 22.Lowell BB, S-Susulic V, Hamann A, Lawitts JA, Himms-Hagen J, et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature. 1993;366:740–42. doi: 10.1038/366740a0. [DOI] [PubMed] [Google Scholar]
- 23.Feldmann HM, Golozoubova V, Cannon B, Nedergaard J. UCP1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Cell Metab. 2009;9:203–9. doi: 10.1016/j.cmet.2008.12.014. [DOI] [PubMed] [Google Scholar]
- 24.Enerback S, Jacobsson A, Simpson EM, Guerra C, Yamashita H, et al. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature. 1997;387:90–94. doi: 10.1038/387090a0. [DOI] [PubMed] [Google Scholar]
- 25.Liu X, Rossmeisl M, McClaine J, Riachi M, Harper ME, Kozak LP. Paradoxical resistance to diet-induced obesity in UCP1-deficient mice. J Clin Investig. 2003;111:399–407. doi: 10.1172/JCI15737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nedergaard J, Cannon B. UCP1 mRNA does not produce heat. Biochim Biophys Acta. 2013;1831:943–49. doi: 10.1016/j.bbalip.2013.01.009. [DOI] [PubMed] [Google Scholar]
- 27.Pico C, Herron D, Palou A, Jacobsson A, Cannon B, Nedergaard J. Stabilization of the mRNA for the uncoupling protein thermogenin by transcriptional/translational blockade and by noradrenaline in brown adipocytes differentiated in culture: a degradation factor induced by cessation of stimulation? Biochem J. 1994;302(Pt. 1):81–86. doi: 10.1042/bj3020081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Puigserver P, Herron D, Gianotti M, Palou A, Cannon B, Nedergaard J. Induction and degradation of the uncoupling protein thermogenin in brown adipocytes in vitro and in vivo. Evidence for a rapidly degradable pool. Biochem J. 1992;284(Pt. 2):393–98. doi: 10.1042/bj2840393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Seale P, Bjork B, Yang W, Kajimura S, Chin S, et al. PRDM16 controls a brown fat/skeletal muscle switch. Nature. 2008;454:961–67. doi: 10.1038/nature07182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Atit R, Sgaier SK, Mohamed OA, Taketo MM, Dufort D, et al. β-Catenin activation is necessary and sufficient to specify the dorsal dermal fate in the mouse. Dev Biol. 2006;296:164–76. doi: 10.1016/j.ydbio.2006.04.449. [DOI] [PubMed] [Google Scholar]
- 31.Lepper C, Fan CM. Inducible lineage tracing of Pax7-descendant cells reveals embryonic origin of adult satellite cells. Genesis. 2010;48:424–36. doi: 10.1002/dvg.20630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Timmons JA, Wennmalm K, Larsson O, Walden TB, Lassmann T, et al. Myogenic gene expression signature establishes that brown and white adipocytes originate from distinct cell lineages. Proc Natl Acad Sci USA. 2007;104:4401–6. doi: 10.1073/pnas.0610615104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Forner F, Kumar C, Luber CA, Fromme T, Klingenspor M, Mann M. Proteome differences between brown and white fat mitochondria reveal specialized metabolic functions. Cell Metab. 2009;10:324–35. doi: 10.1016/j.cmet.2009.08.014. [DOI] [PubMed] [Google Scholar]
- 34.Kajimura S, Seale P, Kubota K, Lunsford E, Frangioni JV, et al. Initiation of myoblast to brown fat switch by a PRDM16-C/EBP-β transcriptional complex. Nature. 2009;460:1154–58. doi: 10.1038/nature08262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hasty P, Bradley A, Morris JH, Edmondson DG, Venuti JM, et al. Muscle deficiency and neonatal death in mice with a targeted mutation in the myogenin gene. Nature. 1993;364:501–6. doi: 10.1038/364501a0. [DOI] [PubMed] [Google Scholar]
- 36.Lee YH, Petkova AP, Mottillo EP, Granneman JG. In vivo identification of bipotential adipocyte progenitors recruited by β3-adrenoceptor activation and high-fat feeding. Cell Metab. 2012;15:480–91. doi: 10.1016/j.cmet.2012.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sanchez-Gurmaches J, Hung CM, Sparks CA, Tang Y, Li H, Guertin DA. PTEN loss in the Myf5 lineage redistributes body fat and reveals subsets of white adipocytes that arise from Myf5 precursors. Cell Metab. 2012;16:348–62. doi: 10.1016/j.cmet.2012.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wu J, Bostrom P, Sparks LM, Ye L, Choi JH, et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell. 2012;150:366–76. doi: 10.1016/j.cell.2012.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ohno H, Shinoda K, Spiegelman BM, Kajimura S. PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab. 2012;15:395–404. doi: 10.1016/j.cmet.2012.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Walden TB, Hansen IR, Timmons JA, Cannon B, Nedergaard J. Recruited versus nonrecruited molecular signatures of brown, “brite,” and white adipose tissues. Am J Physiol Endocr Metab. 2012;302:E19–31. doi: 10.1152/ajpendo.00249.2011. [DOI] [PubMed] [Google Scholar]
- 41.Petrovic N, Walden TB, Shabalina IG, Timmons JA, Cannon B, Nedergaard J. Chronic per-oxisome proliferator–activated receptor γ (PPARγ) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J Biol Chem. 2009;285:7153–64. doi: 10.1074/jbc.M109.053942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schulz TJ, Huang TL, Tran TT, Zhang H, Townsend KL, et al. Identification of inducible brown adipocyte progenitors residing in skeletal muscle and white fat. Proc Natl Acad Sci USA. 2011;108:143–48. doi: 10.1073/pnas.1010929108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sharp LZ, Shinoda K, Ohno H, Scheel DW, Tomoda E, et al. Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS ONE. 2012;7:e49452. doi: 10.1371/journal.pone.0049452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cypess AM, White AP, Vernochet C, Schulz TJ, Xue R, et al. Anatomical localization, gene expression profiling and functional characterization of adult human neck brown fat. Nat Med. 2013;19:635–39. doi: 10.1038/nm.3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lidell ME, Betz MJ, Leinhard OD, Heglind M, Elander L, et al. Evidence for two types of brown adipose tissue in humans. Nat Med. 2013;19:631–34. doi: 10.1038/nm.3017. [DOI] [PubMed] [Google Scholar]
- 46.Jespersen NZ, Larsen TJ, Peijs L, Daugaard S, Homøe P, et al. A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans. Cell Metab. 2013;17:798–805. doi: 10.1016/j.cmet.2013.04.011. [DOI] [PubMed] [Google Scholar]
- 47.Graf T. Historical origins of transdifferentiation and reprogramming. Cell Stem Cell. 2011;9:504–16. doi: 10.1016/j.stem.2011.11.012. [DOI] [PubMed] [Google Scholar]
- 48.Barbatelli G, Murano I, Madsen L, Hao Q, Jimenez M, et al. The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol Endocr Metab. 2010;298:E1244–53. doi: 10.1152/ajpendo.00600.2009. [DOI] [PubMed] [Google Scholar]
- 49.Himms-Hagen J, Melnyk A, Zingaretti MC, Ceresi E, Barbatelli G, Cinti S. Multilocular fat cells in WAT of CL-316243-treated rats derive directly from white adipocytes. Am J Physiol Cell Physiol. 2000;279:C670–81. doi: 10.1152/ajpcell.2000.279.3.C670. [DOI] [PubMed] [Google Scholar]
- 50.Frontini A, Vitali A, Perugini J, Murano I, Romiti C, et al. White-to-brown transdifferentiation of omental adipocytes in patients affected by pheochromocytoma. Biochim Biophys Acta. 2013;1831:950–59. doi: 10.1016/j.bbalip.2013.02.005. [DOI] [PubMed] [Google Scholar]
- 51.Rosenwald M, Perdikari A, Rulicke T, Wolfrum C. Bi-directional interconversion of brite and white adipocytes. Nat Cell Biol. 2013;15:659–67. doi: 10.1038/ncb2740. [DOI] [PubMed] [Google Scholar]
- 52.Guerra C, Koza RA, Yamashita H, Walsh K, Kozak LP. Emergence of brown adipocytes in white fat in mice is under genetic control. Effects on body weight and adiposity. J Clin Investig. 1998;102:412–20. doi: 10.1172/JCI3155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Coulter AA, Bearden CM, Liu X, Koza RA, Kozak LP. Dietary fat interacts with QTLs controlling induction of Pgc-1α and Ucp1 during conversion of white to brown fat. Physiol Genomics. 2003;14:139–47. doi: 10.1152/physiolgenomics.00057.2003. [DOI] [PubMed] [Google Scholar]
- 54.Koza RA, Hohmann SM, Guerra C, Rossmeisl M, Kozak LP. Synergistic gene interactions control the induction of the mitochondrial uncoupling protein (Ucp1) gene in white fat tissue. J Biol Chem. 2000;275:34486–92. doi: 10.1074/jbc.M002136200. [DOI] [PubMed] [Google Scholar]
- 55.Xue B, Coulter A, Rim JS, Koza RA, Kozak LP. Transcriptional synergy and the regulation of Ucp1 during brown adipocyte induction in white fat depots. Mol Cell Biol. 2005;25:8311–22. doi: 10.1128/MCB.25.18.8311-8322.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xue B, Rim JS, Hogan JC, Coulter AA, Koza RA, Kozak LP. Genetic variability affects the development of brown adipocytes in white fat but not in interscapular brown fat. J Lipid Res. 2007;48:41–51. doi: 10.1194/jlr.M600287-JLR200. [DOI] [PubMed] [Google Scholar]
- 57.Cederberg A, Gronning LM, Ahren B, Tasken K, Carlsson P, Enerback S. FOXC2 is a winged helix gene that counteracts obesity, hypertriglyceridemia, and diet-induced insulin resistance. Cell. 2001;106:563–73. doi: 10.1016/s0092-8674(01)00474-3. [DOI] [PubMed] [Google Scholar]
- 58.Seale P, Conroe HM, Estall J, Kajimura S, Frontini A, et al. Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Investig. 2011;121:96–105. doi: 10.1172/JCI44271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kiefer FW, Vernochet C, O’Brien P, Spoerl S, Brown JD, et al. Retinaldehyde dehydrogenase 1 regulates a thermogenic program in white adipose tissue. Nat Med. 2012;18:918–25. doi: 10.1038/nm.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Schulz TJ, Huang P, Huang TL, Xue R, McDougall LE, et al. Brown-fat paucity due to impaired BMP signalling induces compensatory browning of white fat. Nature. 2013;495:379–83. doi: 10.1038/nature11943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Farmer SR. Transcriptional control of adipocyte formation. Cell Metab. 2006;4:263–73. doi: 10.1016/j.cmet.2006.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Carmona MC, Hondares E, Rodriguez de la Concepcion ML, Rodriguez-Sureda V, Peinado-Onsurbe J, et al. Defective thermoregulation, impaired lipid metabolism, but preserved adrenergic induction of gene expression in brown fat of mice lacking C/EBPβ. Biochem J. 2005;389:47–56. doi: 10.1042/BJ20050009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Karamanlidis G, Karamitri A, Docherty K, Hazlerigg DG, Lomax MA. C/EBPβ reprograms white 3T3-L1 preadipocytes to a brown adipocyte pattern of gene expression. J Biol Chem. 2007;282:24660–69. doi: 10.1074/jbc.M703101200. [DOI] [PubMed] [Google Scholar]
- 64.Tanaka T, Yoshida N, Kishimoto T, Akira S. Defective adipocyte differentiation in mice lacking the C/EBPβ and/or C/EBPζ gene. EMBO J. 1997;16:7432–43. doi: 10.1093/emboj/16.24.7432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell. 1998;92:829–39. doi: 10.1016/s0092-8674(00)81410-5. [DOI] [PubMed] [Google Scholar]
- 66.Tiraby C, Tavernier G, Lefort C, Larrouy D, Bouillaud F, et al. Acquirement of brown fat cell features by human white adipocytes. J Biol Chem. 2003;278:33370–76. doi: 10.1074/jbc.M305235200. [DOI] [PubMed] [Google Scholar]
- 67.Lin J, Wu PH, Tarr PT, Lindenberg KS, St-Pierre J, et al. Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1α null mice. Cell. 2004;119:121–35. doi: 10.1016/j.cell.2004.09.013. [DOI] [PubMed] [Google Scholar]
- 68.Uldry M, Yang W, St-Pierre J, Lin J, Seale P, Spiegelman BM. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 2006;3:333–41. doi: 10.1016/j.cmet.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 69.Kleiner S, Mepani RJ, Laznik D, Ye L, Jurczak MJ, et al. Development of insulin resistance in mice lacking PGC-1α in adipose tissues. Proc Natl Acad Sci USA. 2012;109:9635–40. doi: 10.1073/pnas.1207287109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hallberg M, Morganstein DL, Kiskinis E, Shah K, Kralli A, et al. A functional interaction between RIP140 and PGC-1α regulates the expression of the lipid droplet protein CIDEA. Mol Cell Biol. 2008;28:6785–95. doi: 10.1128/MCB.00504-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Leonardsson G, Steel JH, Christian M, Pocock V, Milligan S, et al. Nuclear receptor corepressor RIP140 regulates fat accumulation. Proc Natl Acad Sci USA. 2004;101:8437–42. doi: 10.1073/pnas.0401013101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Picard F, Gehin M, Annicotte J, Rocchi S, Champy MF, et al. SRC-1 and TIF2 control energy balance between white and brown adipose tissues. Cell. 2002;111:931–41. doi: 10.1016/s0092-8674(02)01169-8. [DOI] [PubMed] [Google Scholar]
- 73.Hansen JB, Jorgensen C, Petersen RK, Hallenborg P, De Matteis R, et al. Retinoblastoma protein functions as a molecular switch determining white versus brown adipocyte differentiation. Proc Natl Acad Sci USA. 2004;101:4112–17. doi: 10.1073/pnas.0301964101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Calo E, Quintero-Estades JA, Danielian PS, Nedelcu S, Berman SD, Lees JA. Rb regulates fate choice and lineage commitment in vivo. Nature. 2010;466:1110–14. doi: 10.1038/nature09264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Scime A, Grenier G, Huh MS, Gillespie MA, Bevilacqua L, et al. Rb and p107 regulate preadipocyte differentiation into white versus brown fat through repression of PGC-1α. Cell Metab. 2005;2:283–95. doi: 10.1016/j.cmet.2005.10.002. [DOI] [PubMed] [Google Scholar]
- 76.Pan D, Fujimoto M, Lopes A, Wang YX. Twist-1 is a PPARζ-inducible, negative-feedback regulator of PGC-1α in brown fat metabolism. Cell. 2009;137:73–86. doi: 10.1016/j.cell.2009.01.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Seale P, Kajimura S, Yang W, Chin S, Rohas LM, et al. Transcriptional control of brown fat determination by PRDM16. Cell Metab. 2007;6:38–54. doi: 10.1016/j.cmet.2007.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kajimura S, Seale P, Tomaru T, Erdjument-Bromage H, Cooper MP, et al. Regulation of the brown and white fat gene programs through a PRDM16/CtBP transcriptional complex. Genes Dev. 2008;22:1397–409. doi: 10.1101/gad.1666108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rajakumari S, Wu J, Ishibashi J, Lim HW, Giang AH, et al. EBF2 determines and maintains brown adipocyte identity. Cell Metab. 2013;17:562–74. doi: 10.1016/j.cmet.2013.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ohno H, Shinoda K, Ohyama K, Sharp LZ, Kajimura S. EHMT1 methyltransferase controls brown adipose cell fate and adaptive thermogenesis through the PRDM16 complex. Nature. 2013;504:163–67. doi: 10.1038/nature12652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Jimenez-Preitner M, Berney X, Uldry M, Vitali A, Cinti S, et al. Plac8 is an inducer of C/EBPβ required for brown fat differentiation, thermoregulation, and control of body weight. Cell Metab. 2011;14:658–70. doi: 10.1016/j.cmet.2011.08.008. [DOI] [PubMed] [Google Scholar]
- 82.Villanueva CJ, Vergnes L, Wang J, Drew BG, Hong C, et al. Adipose subtype-selective recruitment of TLE3 or Prdm16 by PPARγ specifies lipid storage versus thermogenic gene programs. Cell Metab. 2013;17:423–35. doi: 10.1016/j.cmet.2013.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sun L, Xie H, Mori MA, Alexander R, Yuan B, et al. Mir193b–365 is essential for brown fat differentiation. Nat Cell Biol. 2011;13:958–65. doi: 10.1038/ncb2286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Trajkovski M, Ahmed K, Esau CC, Stoffel M. MyomiR-133 regulates brown fat differentiation through Prdm16. Nat Cell Biol. 2012;14:1330–35. doi: 10.1038/ncb2612. [DOI] [PubMed] [Google Scholar]
- 85.Yin H, Pasut A, Soleimani VD, Bentzinger CF, Antoun G, et al. MicroRNA-133 controls brown adipose determination in skeletal muscle satellite cells by targeting Prdm16. Cell Metab. 2013;17:210–24. doi: 10.1016/j.cmet.2013.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mori M, Nakagami H, Rodriguez-Araujo G, Nimura K, Kaneda Y. Essential role for miR-196a in brown adipogenesis of white fat progenitor cells. PLoS Biol. 2012;10:e1001314. doi: 10.1371/journal.pbio.1001314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rong JX, Qiu Y, Hansen MK, Zhu L, Zhang V, et al. Adipose mitochondrial biogenesis is suppressed in db/db and high-fat diet–fed mice and improved by rosiglitazone. Diabetes. 2007;56:1751–60. doi: 10.2337/db06-1135. [DOI] [PubMed] [Google Scholar]
- 88.Wilson-Fritch L, Nicoloro S, Chouinard M, Lazar MA, Chui PC, et al. Mitochondrial remodeling in adipose tissue associated with obesity and treatment with rosiglitazone. J Clin Investig. 2004;114:1281–89. doi: 10.1172/JCI21752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sell H, Berger JP, Samson P, Castriota G, Lalonde J, et al. Peroxisome proliferator–activated receptor γ agonism increases the capacity for sympathetically mediated thermogenesis in lean and ob/ob mice. Endocrinology. 2004;145:3925–34. doi: 10.1210/en.2004-0321. [DOI] [PubMed] [Google Scholar]
- 90.Fukui Y, Masui S, Osada S, Umesono K, Motojima K. A new thiazolidinedione, NC-2100, which is a weak PPAR-γ activator, exhibits potent antidiabetic effects and induces uncoupling protein 1 in white adipose tissue of KKAy obese mice. Diabetes. 2000;49:759–67. doi: 10.2337/diabetes.49.5.759. [DOI] [PubMed] [Google Scholar]
- 91.Vernochet C, Peres SB, Davis KE, McDonald ME, Qiang L, et al. C/EBPα and the corepressors CtBP1 and CtBP2 regulate repression of select visceral white adipose genes during induction of the brown phenotype in white adipocytes by peroxisome proliferator–activated receptor γ agonists. Mol Cell Biol. 2009;29:4714–28. doi: 10.1128/MCB.01899-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tai TA, Jennermann C, Brown KK, Oliver BB, MacGinnitie MA, et al. Activation of the nuclear receptor peroxisome proliferator–activated receptor γ promotes brown adipocyte differentiation. J Biol Chem. 1996;271:29909–14. doi: 10.1074/jbc.271.47.29909. [DOI] [PubMed] [Google Scholar]
- 93.Cao W, Daniel KW, Robidoux J, Puigserver P, Medvedev AV, et al. p38 mitogen-activated protein kinase is the central regulator of cyclic AMP–dependent transcription of the brown fat uncoupling protein 1 gene. Mol Cell Biol. 2004;24:3057–67. doi: 10.1128/MCB.24.7.3057-3067.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bronnikov G, Houstek J, Nedergaard J. β-Adrenergic, cAMP-mediated stimulation of proliferation of brown fat cells in primary culture. Mediation via β1 but not via β3 adrenoceptors. J Biol Chem. 1992;267:2006–13. [PubMed] [Google Scholar]
- 95.Bronnikov G, Bengtsson T, Kramarova L, Golozoubova V, Cannon B, Nedergaard J. β1 to β3 switch in control of cyclic adenosine monophosphate during brown adipocyte development explains distinct β-adrenoceptor subtype mediation of proliferation and differentiation. Endocrinology. 1999;140:4185–97. doi: 10.1210/endo.140.9.6972. [DOI] [PubMed] [Google Scholar]
- 96.Jimenez M, Barbatelli G, Allevi R, Cinti S, Seydoux J, et al. β3-Adrenoceptor knockout in C57BL/6J mice depresses the occurrence of brown adipocytes in white fat. Eur J Biochem. 2003;270:699–705. doi: 10.1046/j.1432-1033.2003.03422.x. [DOI] [PubMed] [Google Scholar]
- 97.Haas B, Mayer P, Jennissen K, Scholz D, Berriel Diaz M, et al. Protein kinase G controls brown fat cell differentiation and mitochondrial biogenesis. Sci Signal. 2009;2:ra78. doi: 10.1126/scisignal.2000511. [DOI] [PubMed] [Google Scholar]
- 98.Mitschke MM, Hoffmann LS, Gnad T, Scholz D, Kruithoff K, et al. Increased cGMP promotes healthy expansion and browning of white adipose tissue. FASEB J. 2013;27:1621–30. doi: 10.1096/fj.12-221580. [DOI] [PubMed] [Google Scholar]
- 99.Saito M, Yoneshiro T. Capsinoids and related food ingredients activating brown fat thermogenesis and reducing body fat in humans. Curr Opin Lipidol. 2013;24:71–77. doi: 10.1097/MOL.0b013e32835a4f40. [DOI] [PubMed] [Google Scholar]
- 100.Masuda Y, Haramizu S, Oki K, Ohnuki K, Watanabe T, et al. Upregulation of uncoupling proteins by oral administration of capsiate, a nonpungent capsaicin analog. J Appl Physiol. 2003;95:2408–15. doi: 10.1152/japplphysiol.00828.2002. [DOI] [PubMed] [Google Scholar]
- 101.Kawabata F, Inoue N, Masamoto Y, Matsumura S, Kimura W, et al. Non-pungent capsaicin analogs (capsinoids) increase metabolic rate and enhance thermogenesis via gastrointestinal TRPV1 in mice. Biosci Biotechnol Biochem. 2009;73:2690–97. doi: 10.1271/bbb.90555. [DOI] [PubMed] [Google Scholar]
- 102.Ye L, Kleiner S, Wu J, Sah R, Gupta RK, et al. TRPV4 is a regulator of adipose oxidative metabolism, inflammation, and energy homeostasis. Cell. 2012;151:96–110. doi: 10.1016/j.cell.2012.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ortega-Molina A, Efeyan A, Lopez-Guadamillas E, Munoz-Martin M, Gomez-Lopez G, et al. Pten positively regulates brown adipose function, energy expenditure, and longevity. Cell Metab. 2012;15:382–94. doi: 10.1016/j.cmet.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 104.Tseng YH, Kokkotou E, Schulz TJ, Huang TL, Winnay JN, et al. New role of bone morphogenetic protein 7 in brown adipogenesis and energy expenditure. Nature. 2008;454:1000–4. doi: 10.1038/nature07221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Qian SW, Tang Y, Li X, Liu Y, Zhang YY, et al. BMP4-mediated brown fat–like changes in white adipose tissue alter glucose and energy homeostasis. Proc Natl Acad Sci USA. 2013;110:E798–807. doi: 10.1073/pnas.1215236110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Whittle AJ, Carobbio S, Martins L, Slawik M, Hondares E, et al. BMP8B increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell. 2012;149:871–85. doi: 10.1016/j.cell.2012.02.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yadav H, Quijano C, Kamaraju AK, Gavrilova O, Malek R, et al. Protection from obesity and diabetes by blockade of TGF-β/Smad3 signaling. Cell Metab. 2011;14:67–79. doi: 10.1016/j.cmet.2011.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Koncarevic A, Kajimura S, Cornwall-Brady M, Andreucci A, Pullen A, et al. A novel therapeutic approach to treating obesity through modulation of TGFβ signaling. Endocrinology. 2012;153:3133–46. doi: 10.1210/en.2012-1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Fournier B, Murray B, Gutzwiller S, Marcaletti S, Marcellin D, et al. Blockade of the activin receptor IIB activates functional brown adipogenesis and thermogenesis by inducing mitochondrial oxidative metabolism. Mol Cell Biol. 2012;32:2871–79. doi: 10.1128/MCB.06575-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Potthoff MJ, Kliewer SA, Mangelsdorf DJ. Endocrine fibroblast growth factors 15/19 and 21: from feast to famine. Genes Dev. 2012;26:312–24. doi: 10.1101/gad.184788.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Tomlinson E, Fu L, John L, Hultgren B, Huang X, et al. Transgenic mice expressing human fibroblast growth factor-19 display increased metabolic rate and decreased adiposity. Endocrinology. 2002;143:1741–47. doi: 10.1210/endo.143.5.8850. [DOI] [PubMed] [Google Scholar]
- 112.Fu L, John LM, Adams SH, Yu XX, Tomlinson E, et al. Fibroblast growth factor 19 increases metabolic rate and reverses dietary and leptin-deficient diabetes. Endocrinology. 2004;145:2594–603. doi: 10.1210/en.2003-1671. [DOI] [PubMed] [Google Scholar]
- 113.Hondares E, Rosell M, Gonzalez FJ, Giralt M, Iglesias R, Villarroya F. Hepatic FGF21 expression is induced at birth via PPARα in response to milk intake and contributes to thermogenic activation of neonatal brown fat. Cell Metab. 2010;11:206–12. doi: 10.1016/j.cmet.2010.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Fisher FM, Kleiner S, Douris N, Fox EC, Mepani RJ, et al. FGF21 regulates PGC-1α and browning of white adipose tissues in adaptive thermogenesis. Genes Dev. 2012;26:271–81. doi: 10.1101/gad.177857.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Wei W, Dutchak PA, Wang X, Ding X, Wang X, et al. Fibroblast growth factor 21 promotes bone loss by potentiating the effects of peroxisome proliferator–activated receptor γ. Proc Natl Acad Sci USA. 2012;109:3143–48. doi: 10.1073/pnas.1200797109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Bostrom P, Wu J, Jedrychowski MP, Korde A, Ye L, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–68. doi: 10.1038/nature10777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Bordicchia M, Liu D, Amri EZ, Ailhaud G, Dessi-Fulgheri P, et al. Cardiac natriuretic peptides act via p38 MAPK to induce the brown fat thermogenic program in mouse and human adipocytes. J Clin Investig. 2012;122:1022–36. doi: 10.1172/JCI59701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Madsen L, Pedersen LM, Lillefosse HH, Fjaere E, Bronstad I, et al. UCP1 induction during recruitment of brown adipocytes in white adipose tissue is dependent on cyclooxygenase activity. PLoS ONE. 2010;5:e11391. doi: 10.1371/journal.pone.0011391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Vegiopoulos A, Müller-Decker K, Strzoda D, Schmitt I, Chichelnitskiy E, et al. Cyclooxygenase-2 controls energy homeostasis in mice by de novo recruitment of brown adipocytes. Science. 2010;328:1158–61. doi: 10.1126/science.1186034. [DOI] [PubMed] [Google Scholar]
- 120.Sellayah D, Bharaj P, Sikder D. Orexin is required for brown adipose tissue development, differentiation, and function. Cell Metab. 2011;14:478–90. doi: 10.1016/j.cmet.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 121.Nguyen KD, Qiu Y, Cui X, Goh YP, Mwangi J, et al. Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis. Nature. 2011;480:104–8. doi: 10.1038/nature10653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Watanabe M, Houten SM, Mataki C, Christoffolete MA, Kim BW, et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature. 2006;439:484–89. doi: 10.1038/nature04330. [DOI] [PubMed] [Google Scholar]
- 123.Tsukita S, Yamada T, Uno K, Takahashi K, Kaneko K, et al. Hepatic glucokinase modulates obesity predisposition by regulating BAT thermogenesis via neural signals. Cell Metab. 2012;16:825–32. doi: 10.1016/j.cmet.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 124.Yamada T, Katagiri H, Ishigaki Y, Ogihara T, Imai J, et al. Signals from intra-abdominal fat modulate insulin and leptin sensitivity through different mechanisms: neuronal involvement in food-intake regulation. Cell Metab. 2006;3:223–29. doi: 10.1016/j.cmet.2006.02.001. [DOI] [PubMed] [Google Scholar]
- 125.Stanford KI, Middelbeek RJ, Townsend KL, An D, Nygaard EB, et al. Brown adipose tissue regulates glucose homeostasis and insulin sensitivity. J Clin Investig. 2012;123:215–23. doi: 10.1172/JCI62308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Lean ME. Brown adipose tissue in humans. Proc Nutr Soc. 1989;48:243–56. doi: 10.1079/pns19890036. [DOI] [PubMed] [Google Scholar]
- 127.Pfannenberg C, Werner MK, Ripkens S, Stef I, Deckert A, et al. Impact of age on the relationships of brown adipose tissue with sex and adiposity in humans. Diabetes. 2010;59:1789–93. doi: 10.2337/db10-0004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Orava J, Nuutila P, Lidell ME, Oikonen V, Noponen T, et al. Different metabolic responses of human brown adipose tissue to activation by cold and insulin. Cell Metab. 2011;14:272–79. doi: 10.1016/j.cmet.2011.06.012. [DOI] [PubMed] [Google Scholar]
- 129.Muzik O, Mangner TJ, Leonard WR, Kumar A, Janisse J, Granneman JG. 15O PET measurement of blood flow and oxygen consumption in cold-activated human brown fat. J Nucl Med. 2013;54:523–31. doi: 10.2967/jnumed.112.111336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Cohade C, Osman M, Pannu HK, Wahl RL. Uptake in supraclavicular area fat (“USA-Fat”): description on 18F-FDG PET/CT. J Nucl Med. 2003;44:170–76. [PubMed] [Google Scholar]
- 131.Au-Yong IT, Thorn N, Ganatra R, Perkins AC, Symonds ME. Brown adipose tissue and seasonal variation in humans. Diabetes. 2009;58:2583–87. doi: 10.2337/db09-0833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lee P, Greenfield JR, Ho KK, Fulham MJ. A critical appraisal of the prevalence and metabolic significance of brown adipose tissue in adult humans. Am J Physiol Endocr Metab. 2010;299:E601–6. doi: 10.1152/ajpendo.00298.2010. [DOI] [PubMed] [Google Scholar]
- 133.Gilsanz V, Chung SA, Jackson H, Dorey FJ, Hu HH. Functional brown adipose tissue is related to muscle volume in children and adolescents. J Pediatr. 2011;158:722–26. doi: 10.1016/j.jpeds.2010.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Yoneshiro T, Aita S, Matsushita M, Okamatsu-Ogura Y, Kameya T, et al. Age-related decrease in cold-activated brown adipose tissue and accumulation of body fat in healthy humans. Obesity. 2011;19:1755–60. doi: 10.1038/oby.2011.125. [DOI] [PubMed] [Google Scholar]
- 135.Yoneshiro T, Aita S, Matsushita M, Kameya T, Nakada K, et al. Brown adipose tissue, whole-body energy expenditure, and thermogenesis in healthy adult men. Obesity. 2011;19:13–16. doi: 10.1038/oby.2010.105. [DOI] [PubMed] [Google Scholar]
- 136.Yoneshiro T, Aita S, Matsushita M, Kayahara T, Kameya T, et al. Recruited brown adipose tissue as an anti-obesity agent in humans. J Clin Investig. 2013;123:3404–8. doi: 10.1172/JCI67803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kuji I, Imabayashi E, Minagawa A, Matsuda H, Miyauchi T. Brown adipose tissue demonstrating intense FDG uptake in a patient with mediastinal pheochromocytoma. Ann Nucl Med. 2008;22:231–35. doi: 10.1007/s12149-007-0096-x. [DOI] [PubMed] [Google Scholar]
- 138.Wang Q, Zhang M, Ning G, Gu W, Su T, et al. Brown adipose tissue in humans is activated by elevated plasma catecholamines levels and is inversely related to central obesity. PLoS ONE. 2011;6:e21006. doi: 10.1371/journal.pone.0021006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Carey AL, Formosa MF, Van Every B, Bertovic D, Eikelis N, et al. Ephedrine activates brown adipose tissue in lean but not obese humans. Diabetologia. 2013;56:147–55. doi: 10.1007/s00125-012-2748-1. [DOI] [PubMed] [Google Scholar]
- 140.Skarulis MC, Celi FS, Mueller E, Zemskova M, Malek R, et al. Thyroid hormone induced brown adipose tissue and amelioration of diabetes in a patient with extreme insulin resistance. J Clin Endocrinol Metab. 2010;95:256–62. doi: 10.1210/jc.2009-0543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Esterbauer H, Oberkofler H, Liu YM, Breban D, Hell E, et al. Uncoupling protein-1 mRNA expression in obese human subjects: the role of sequence variations at the uncoupling protein-1 gene locus. J Lipid Res. 1998;39:834–44. [PubMed] [Google Scholar]
- 142.Rose G, Crocco P, D’Aquila P, Montesanto A, Bellizzi D, Passarino G. Two variants located in the upstream enhancer region of human UCP1 gene affect gene expression and are correlated with human longevity. Exp Gerontol. 2011;46:897–904. doi: 10.1016/j.exger.2011.07.011. [DOI] [PubMed] [Google Scholar]
- 143.Yoneshiro T, Ogawa T, Okamoto N, Matsushita M, Aita S, et al. Impact of UCP1 and β3AR gene polymorphisms on age-related changes in brown adipose tissue and adiposity in humans. Int J Obes. 2013;37:993–98. doi: 10.1038/ijo.2012.161. [DOI] [PubMed] [Google Scholar]
- 144.Nagai N, Sakane N, Tsuzaki K, Moritani T. UCP1 genetic polymorphism (−3826 A/G) diminishes resting energy expenditure and thermoregulatory sympathetic nervous system activity in young females. Int J Obes. 2011;35:1050–55. doi: 10.1038/ijo.2010.261. [DOI] [PubMed] [Google Scholar]
- 145.Nagai N, Sakane N, Ueno LM, Hamada T, Moritani T. The −3826 A→G variant of the uncoupling protein-1 gene diminishes postprandial thermogenesis after a high fat meal in healthy boys. J Clin Endocrinol Metab. 2003;88:5661–67. doi: 10.1210/jc.2003-030672. [DOI] [PubMed] [Google Scholar]
- 146.Hu HH, Perkins TG, Chia JM, Gilsanz V. Characterization of human brown adipose tissue by chemical-shift water-fat MRI. Am J Roentgenol. 2013;200:177–83. doi: 10.2214/AJR.12.8996. [DOI] [PMC free article] [PubMed] [Google Scholar]