Abstract
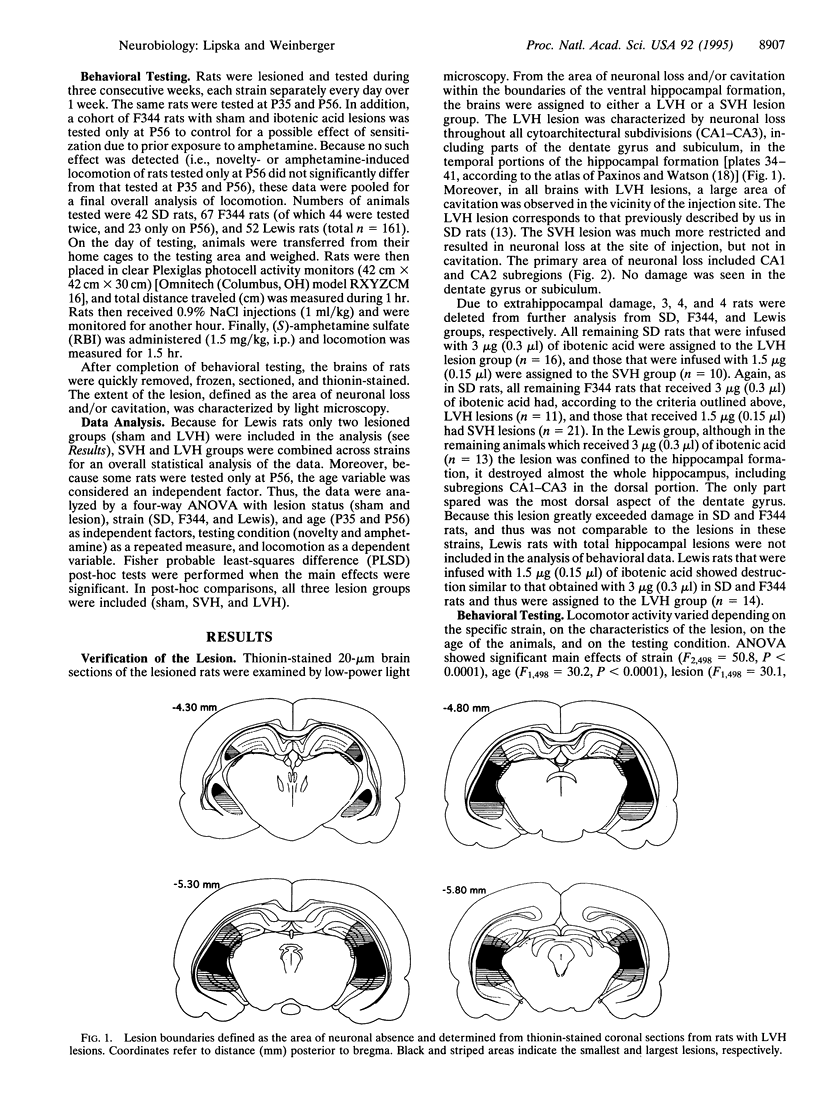
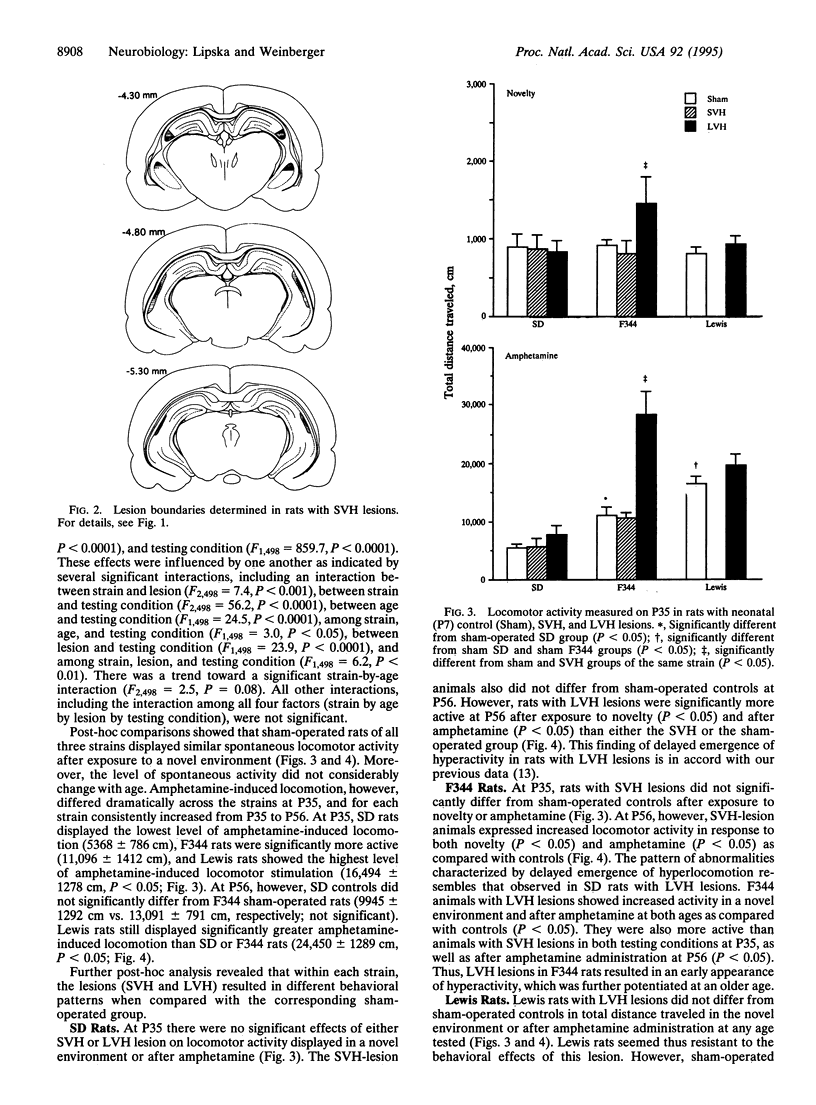
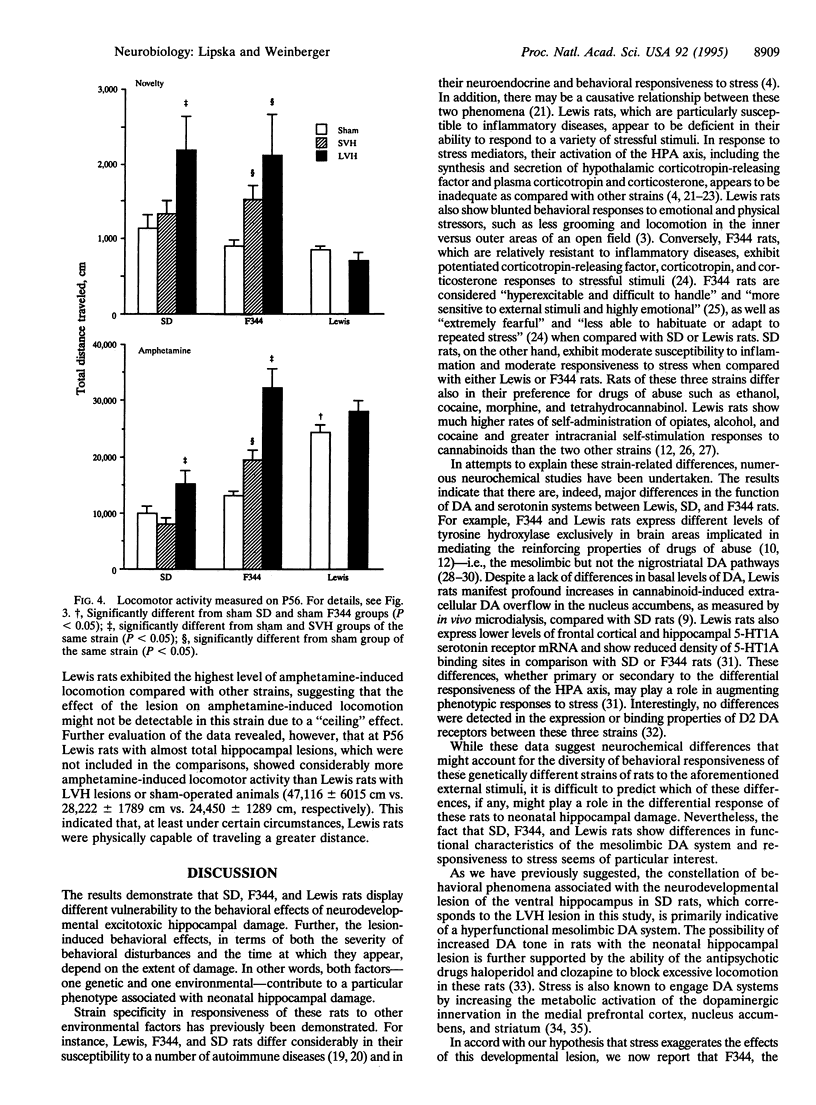
We explored how two independent variables, one genetic (i.e., specific rat strains) and another environmental (i.e., a developmental excitotoxic hippocampal lesion), contribute to phenotypic variation. Sprague-Dawley (SD), Fischer 344 (F344), and Lewis rats underwent two grades of neonatal excitotoxic damage: small and large ventral hippocampal (SVH and LVH) lesions. Locomotion was tested before puberty [postnatal day 35 (P35)] and after puberty (P56) following exposure to a novel environment or administration of amphetamine. The behavioral effects were strain- and lesion-specific. As shown previously, SD rats with LVH lesions displayed enhanced spontaneous and amphetamine-induced locomotion as compared with controls at P56, but not at P35. SVH lesions in SD rats had no effect at any age. In F344 rats with LVH lesions, enhanced spontaneous and amphetamine-induced locomotion appeared early (P35) and was exaggerated at P56. SVH lesions in F344 rats resulted in a pattern of effects analogous to LVH lesions in SD rats--i.e., postpubertal onset of hyperlocomotion (P56). In Lewis rats, LVH lesions had no significant effect on novelty- or amphetamine-induced locomotion at any age. These data show that the degree of genetic predisposition and the extent of early induced hippocampal defect contribute to the particular pattern of behavioral outcome. These results may have implications for modeling interactions of genetic and environmental factors involved in schizophrenia, a disorder characterized by phenotypic heterogeneity, genetic predisposition, a developmental hippocampal abnormality, and vulnerability to environmental stress.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abercrombie E. D., Keefe K. A., DiFrischia D. S., Zigmond M. J. Differential effect of stress on in vivo dopamine release in striatum, nucleus accumbens, and medial frontal cortex. J Neurochem. 1989 May;52(5):1655–1658. doi: 10.1111/j.1471-4159.1989.tb09224.x. [DOI] [PubMed] [Google Scholar]
- Aksentijevich S., Whitfield H. J., Jr, Young W. S., 3rd, Wilder R. L., Chrousos G. P., Gold P. W., Sternberg E. M. Arthritis-susceptible Lewis rats fail to emerge from the stress hyporesponsive period. Brain Res Dev Brain Res. 1992 Jan 17;65(1):115–118. doi: 10.1016/0165-3806(92)90014-n. [DOI] [PubMed] [Google Scholar]
- Beitner-Johnson D., Guitart X., Nestler E. J. Dopaminergic brain reward regions of Lewis and Fischer rats display different levels of tyrosine hydroxylase and other morphine- and cocaine-regulated phosphoproteins. Brain Res. 1991 Oct 4;561(1):147–150. doi: 10.1016/0006-8993(91)90759-o. [DOI] [PubMed] [Google Scholar]
- Beitner-Johnson D., Guitart X., Nestler E. J. Glial fibrillary acidic protein and the mesolimbic dopamine system: regulation by chronic morphine and Lewis-Fischer strain differences in the rat ventral tegmental area. J Neurochem. 1993 Nov;61(5):1766–1773. doi: 10.1111/j.1471-4159.1993.tb09814.x. [DOI] [PubMed] [Google Scholar]
- Berrettini W. H., Ferraro T. N., Alexander R. C., Buchberg A. M., Vogel W. H. Quantitative trait loci mapping of three loci controlling morphine preference using inbred mouse strains. Nat Genet. 1994 May;7(1):54–58. doi: 10.1038/ng0594-54. [DOI] [PubMed] [Google Scholar]
- Bozarth M. A. Neural basis of psychomotor stimulant and opiate reward: evidence suggesting the involvement of a common dopaminergic system. Behav Brain Res. 1986 Nov;22(2):107–116. doi: 10.1016/0166-4328(86)90032-x. [DOI] [PubMed] [Google Scholar]
- Burnet P. W., Mefford I. N., Smith C. C., Gold P. W., Sternberg E. M. Hippocampal 8-[3H]hydroxy-2-(di-n-propylamino) tetralin binding site densities, serotonin receptor (5-HT1A) messenger ribonucleic acid abundance, and serotonin levels parallel the activity of the hypothalamopituitary-adrenal axis in rat. J Neurochem. 1992 Sep;59(3):1062–1070. doi: 10.1111/j.1471-4159.1992.tb08348.x. [DOI] [PubMed] [Google Scholar]
- Calogero A. E., Sternberg E. M., Bagdy G., Smith C., Bernardini R., Aksentijevich S., Wilder R. L., Gold P. W., Chrousos G. P. Neurotransmitter-induced hypothalamic-pituitary-adrenal axis responsiveness is defective in inflammatory disease-susceptible Lewis rats: in vivo and in vitro studies suggesting globally defective hypothalamic secretion of corticotropin-releasing hormone. Neuroendocrinology. 1992 May;55(5):600–608. doi: 10.1159/000126173. [DOI] [PubMed] [Google Scholar]
- Chen J. P., Paredes W., Lowinson J. H., Gardner E. L. Strain-specific facilitation of dopamine efflux by delta 9-tetrahydrocannabinol in the nucleus accumbens of rat: an in vivo microdialysis study. Neurosci Lett. 1991 Aug 5;129(1):136–180. doi: 10.1016/0304-3940(91)90739-g. [DOI] [PubMed] [Google Scholar]
- Deutch A. Y., Clark W. A., Roth R. H. Prefrontal cortical dopamine depletion enhances the responsiveness of mesolimbic dopamine neurons to stress. Brain Res. 1990 Jun 25;521(1-2):311–315. doi: 10.1016/0006-8993(90)91557-w. [DOI] [PubMed] [Google Scholar]
- Dhabhar F. S., McEwen B. S., Spencer R. L. Stress response, adrenal steroid receptor levels and corticosteroid-binding globulin levels--a comparison between Sprague-Dawley, Fischer 344 and Lewis rats. Brain Res. 1993 Jul 9;616(1-2):89–98. doi: 10.1016/0006-8993(93)90196-t. [DOI] [PubMed] [Google Scholar]
- Fibiger H. C. Drugs and reinforcement mechanisms: a critical review of the catecholamine theory. Annu Rev Pharmacol Toxicol. 1978;18:37–56. doi: 10.1146/annurev.pa.18.040178.000345. [DOI] [PubMed] [Google Scholar]
- Gardner E. L., Lowinson J. H. Marijuana's interaction with brain reward systems: update 1991. Pharmacol Biochem Behav. 1991 Nov;40(3):571–580. doi: 10.1016/0091-3057(91)90365-9. [DOI] [PubMed] [Google Scholar]
- George F. R., Goldberg S. R. Genetic approaches to the analysis of addiction processes. Trends Pharmacol Sci. 1989 Feb;10(2):78–83. doi: 10.1016/0165-6147(89)90083-7. [DOI] [PubMed] [Google Scholar]
- Glowa J. R., Geyer M. A., Gold P. W., Sternberg E. M. Differential startle amplitude and corticosterone response in rats. Neuroendocrinology. 1992 Nov;56(5):719–723. doi: 10.1159/000126298. [DOI] [PubMed] [Google Scholar]
- Griffin A. C., Whitacre C. C. Sex and strain differences in the circadian rhythm fluctuation of endocrine and immune function in the rat: implications for rodent models of autoimmune disease. J Neuroimmunol. 1991 Dec;35(1-3):53–64. doi: 10.1016/0165-5728(91)90161-y. [DOI] [PubMed] [Google Scholar]
- Griffiths M. M., DeWitt C. W. Genetic control of collagen-induced arthritis in rats: the immune response to type II collagen among susceptible and resistant strains and evidence for multiple gene control. J Immunol. 1984 Jun;132(6):2830–2836. [PubMed] [Google Scholar]
- Guitart X., Beitner-Johnson D., Marby D. W., Kosten T. A., Nestler E. J. Fischer and Lewis rat strains differ in basal levels of neurofilament proteins and their regulation by chronic morphine in the mesolimbic dopamine system. Synapse. 1992 Nov;12(3):242–253. doi: 10.1002/syn.890120310. [DOI] [PubMed] [Google Scholar]
- Kelly P. H., Seviour P. W., Iversen S. D. Amphetamine and apomorphine responses in the rat following 6-OHDA lesions of the nucleus accumbens septi and corpus striatum. Brain Res. 1975 Sep 5;94(3):507–522. doi: 10.1016/0006-8993(75)90233-4. [DOI] [PubMed] [Google Scholar]
- Koob G. F., Bloom F. E. Cellular and molecular mechanisms of drug dependence. Science. 1988 Nov 4;242(4879):715–723. doi: 10.1126/science.2903550. [DOI] [PubMed] [Google Scholar]
- Kosten T. A., Miserendino M. J., Chi S., Nestler E. J. Fischer and Lewis rat strains show differential cocaine effects in conditioned place preference and behavioral sensitization but not in locomotor activity or conditioned taste aversion. J Pharmacol Exp Ther. 1994 Apr;269(1):137–144. [PubMed] [Google Scholar]
- Lander E. S., Schork N. J. Genetic dissection of complex traits. Science. 1994 Sep 30;265(5181):2037–2048. doi: 10.1126/science.8091226. [DOI] [PubMed] [Google Scholar]
- Lipska B. K., Jaskiw G. E., Weinberger D. R. Postpubertal emergence of hyperresponsiveness to stress and to amphetamine after neonatal excitotoxic hippocampal damage: a potential animal model of schizophrenia. Neuropsychopharmacology. 1993 Aug;9(1):67–75. doi: 10.1038/npp.1993.44. [DOI] [PubMed] [Google Scholar]
- Lipska B. K., Weinberger D. R. Delayed effects of neonatal hippocampal damage on haloperidol-induced catalepsy and apomorphine-induced stereotypic behaviors in the rat. Brain Res Dev Brain Res. 1993 Oct 15;75(2):213–222. doi: 10.1016/0165-3806(93)90026-7. [DOI] [PubMed] [Google Scholar]
- Lipska B. K., Weinberger D. R. Subchronic treatment with haloperidol and clozapine in rats with neonatal excitotoxic hippocampal damage. Neuropsychopharmacology. 1994 May;10(3):199–205. doi: 10.1038/npp.1994.22. [DOI] [PubMed] [Google Scholar]
- Luedtke R. R., Artymyshyn R. P., Monks B. R., Molinoff P. B. Comparison of the expression, transcription and genomic organization of D2 dopamine receptors in outbred and inbred strains of rat. Brain Res. 1992 Jul 3;584(1-2):45–54. doi: 10.1016/0006-8993(92)90876-b. [DOI] [PubMed] [Google Scholar]
- Rosecrans J. A., Robinson S. E., Johnson J. H., Mokler D. J., Hong J. S. Neuroendocrine, biogenic amine and behavioral responsiveness to a repeated foot-shock-induced analgesia (FSIA) stressor in Sprague-Dawley (CD) and Fischer-344 (CDF) rats. Brain Res. 1986 Sep 10;382(1):71–80. doi: 10.1016/0006-8993(86)90112-5. [DOI] [PubMed] [Google Scholar]
- Rosecrans J. A., Schechter M. D. Brain 5-hydroxytryptamine correlates of behavior in rats: strain and sex variability. Physiol Behav. 1972 Mar;8(3):503–510. doi: 10.1016/0031-9384(72)90336-8. [DOI] [PubMed] [Google Scholar]
- Smith T., Hewson A. K., Quarrie L., Leonard J. P., Cuzner M. L. Hypothalamic PGE2 and cAMP production and adrenocortical activation following intraperitoneal endotoxin injection: in vivo microdialysis studies in Lewis and Fischer rats. Neuroendocrinology. 1994 Apr;59(4):396–405. doi: 10.1159/000126683. [DOI] [PubMed] [Google Scholar]
- Sternberg E. M., Glowa J. R., Smith M. A., Calogero A. E., Listwak S. J., Aksentijevich S., Chrousos G. P., Wilder R. L., Gold P. W. Corticotropin releasing hormone related behavioral and neuroendocrine responses to stress in Lewis and Fischer rats. Brain Res. 1992 Jan 20;570(1-2):54–60. doi: 10.1016/0006-8993(92)90563-o. [DOI] [PubMed] [Google Scholar]
- Sternberg E. M., Hill J. M., Chrousos G. P., Kamilaris T., Listwak S. J., Gold P. W., Wilder R. L. Inflammatory mediator-induced hypothalamic-pituitary-adrenal axis activation is defective in streptococcal cell wall arthritis-susceptible Lewis rats. Proc Natl Acad Sci U S A. 1989 Apr;86(7):2374–2378. doi: 10.1073/pnas.86.7.2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sternberg E. M., Young W. S., 3rd, Bernardini R., Calogero A. E., Chrousos G. P., Gold P. W., Wilder R. L. A central nervous system defect in biosynthesis of corticotropin-releasing hormone is associated with susceptibility to streptococcal cell wall-induced arthritis in Lewis rats. Proc Natl Acad Sci U S A. 1989 Jun;86(12):4771–4775. doi: 10.1073/pnas.86.12.4771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki T., George F. R., Meisch R. A. Differential establishment and maintenance of oral ethanol reinforced behavior in Lewis and Fischer 344 inbred rat strains. J Pharmacol Exp Ther. 1988 Apr;245(1):164–170. [PubMed] [Google Scholar]
- Suzuki T., Lu M. S., Motegi H., Yoshii T., Misawa M. Genetic differences in the development of physical dependence upon diazepam in Lewis and Fischer 344 inbred rat strains. Pharmacol Biochem Behav. 1992 Oct;43(2):387–393. doi: 10.1016/0091-3057(92)90167-e. [DOI] [PubMed] [Google Scholar]
- Weinberger D. R., Lipska B. K. Cortical maldevelopment, anti-psychotic drugs, and schizophrenia: a search for common ground. Schizophr Res. 1995 Aug 1;16(2):87–110. doi: 10.1016/0920-9964(95)00013-c. [DOI] [PubMed] [Google Scholar]


