Abstract
Previous studies have reported declines in high density lipoprotein (HDL) cholesterol 1–2 years after pregnancy. In 1986–1996, the authors prospectively examined the association between childbearing and changes in fasting plasma lipids (low density lipoprotein, HDL, and total cholesterol; triglycerides) among 1,952 US women (980 Black, 972 White) in the Coronary Artery Risk Development in Young Adults study. Repeated-measures multiple linear regression was used to examine lipid changes over three time intervals (baseline to years 5, 7, and 10) in time-dependent follow-up groups: P0 (0 pregnancies), P1 (≥1 miscarriages/abortions), B1 (1 birth), and B2 (≥2 births). Means stratified by race and baseline parity (nulliparous or parous) were fully adjusted for study center, time, height, baseline diet, and other baseline and time-dependent covariates (age, smoking, education, weight, waist circumference, alcohol intake, oral contraceptive use, physical activity, short pregnancies). For both races, fully adjusted HDL cholesterol declines of −3 to −4 mg/dl were associated with a first birth versus no pregnancies during follow-up (p < 0.001). Higher-order births were not associated with greater declines in HDL cholesterol (B2 similar to B1, no association among women parous at baseline). In Whites, total and low density lipoprotein cholesterol declines were associated with follow-up births. HDL cholesterol declines of −3 to −4 mg/dl after a first birth persisted during the 10 years of follow-up independent of weight, central adiposity, and selected behavior changes.
Keywords: ethnic groups, lipids, lipoproteins, HDL cholesterol, parity, pregnancy
Evidence for an association between parity and the risk of cardiovascular disease in women has been conflicting (1–8) partly because of residual confounding from socioeconomic status and age that influence childbearing patterns as well as disease risk. Secondly, measures of preexisting risk factors for cardiovascular disease (obesity, hypertension, and hyperlipidemia) and longitudinal changes in risk factors during the childbearing years have not been collected prospectively to differentiate pregnancy-specific effects from those associated with aging, secular trends, and changes in behaviors.
During healthy pregnancy, the progressive rise in blood insulin and gestational hormones (progesterone, 17β-estradiol, human placental lactogen) is accompanied by elevations in blood lipids, insulin resistance, and weight gain (9–14). Blood lipids peak by 31–36 weeks of gestation, and percentage increases above early pregnancy levels range from 43 to 53 for total cholesterol, 36 to 40 for low density lipoprotein (LDL) cholesterol, 0 to 25 for high density lipoprotein (HDL) cholesterol (13–20), and 150 to 230 for triglycerides (12, 15, 17, 20). During the puerperium, triglycerides decrease rapidly (12–14, 20) while total cholesterol and LDL cholesterol (12, 13, 21) remain above and HDL cholesterol (18, 20) below preconception or early pregnancy levels for several months to 1 year postpartum.
Large cross-sectional studies of both pre- and postmenopausal women (2, 8, 22, 23) have reported inverse correlations between parity and HDL cholesterol or lower HDL cholesterol levels at a threshold of high parity (e.g., five or more births vs. four or fewer) (24, 25). Few studies have examined longitudinal changes in blood lipids from preconception to 6 or more months postpartum or have controlled for the effect of aging and secular trends on lipids by comparison to nonparous women. A study of 22 pregnant Dutch women found that HDL cholesterol levels at 1 year postpartum were lower than at preconception (18). In White women, HDL cholesterol declines were −1 mg/dl per birth in 8 years (26) and −2.4 mg/dl in 3 years for one or more pregnancies versus none, adjusted for other covariates (27). The Coronary Artery Risk Development in Young Adults (CARDIA) study previously reported a 2-year decline in HDL cholesterol associated with a first birth but not subsequent births (28), independent of changes in weight, waist-hip ratio, and behavioral covariates. In CARDIA study women, excess weight gain was associated with a first birth (29, 30).
Previous studies provided conflicting evidence for a first versus higher-order birth effect, had insufficient power to assess differences by race, estimated short-term lipid changes associated with change in parity, and did not control for central adiposity, except waist-hip ratio in the CARDIA study. The present study prospectively examined the association of childbearing with long-term plasma lipid changes during 10 years to determine whether the association varies by race (Blacks and Whites) or parity at baseline and is mediated primarily by gains in weight and waist circumference or by changes in alcohol intake, cigarette smoking, oral contraceptive use, and physical activity associated with childbearing.
Materials And Methods
Study population
The CARDIA study is a multicenter, longitudinal, observational study designed to describe the development of risk factors for coronary heart disease in young Blacks and Whites, both men and women. Details of the study design, methodology, and cohort characteristics have been reported elsewhere (31–33). The study population was recruited from four geographic areas of the United States: Birmingham, Alabama; Chicago, Illinois; Minneapolis, Minnesota; and Oakland, California. In 1985–1986, baseline data were collected on 5,115 subjects (2,787 women) aged 18–30 years, of whom 52 percent were Black and 48 percent were White. Baseline and follow-up examinations in years 2, 5, 7, and 10 included a variety of physiologic and self-reported measures that have been described elsewhere (31, 33, 34). Overall retention rates for the cohort were 91, 86, 81, and 79 percent of the surviving cohort at years 2, 5, 7, and 10, respectively (34).
Sample selection criteria
Of 2,787 women enrolled at baseline, there were 2,192 who had attended one or more follow-up examinations, and their pregnancies and births occurred at least 12 months before the year 10 examination. Women were excluded if, at baseline, they had had a hysterectomy or removal of both ovaries (n = 20), were currently pregnant (n = 3) or breastfeeding (n = 40), or reported a pregnancy 3 months prior to the examination (n = 46). Also excluded were those who were nonfasting (<8 hours) prior to venipuncture at baseline (n = 85), had taken lipid-lowering medications (n = 15), had plasma triglyceride levels of more than 400 mg/dl at any examination (n = 8), or were missing covariables at all examinations (n = 18). At baseline, none of the women had taken lipid-lowering medications. At follow-up examinations, lipid measurements for women pregnant or lactating or nonfasting were excluded from the analysis. The analytic sample included 1,952 women (980 Blacks, 972 Whites), 70 percent of the original cohort. Fasting plasma lipid measurements were available at all four examinations and at three of four examinations for 61.0 percent (n = 1,190) and 28.7 percent (n = 561) of the sample, respectively. Institutional review boards at each participating study center approved the study.
Data collection methods
The methodology used to recruit subjects and collect data for the CARDIA study has been described in detail previously (31–33). Participants were asked to fast for 12 hours prior to each examination, and they reported the number of hours since their last intake of food or beverages prior to the blood draw. Blood samples were drawn in the morning after an overnight fast by using a Vacutainer tube (Becton, Dickinson and Company, Franklin Lakes, New Jersey) containing ethylenediaminetetraacetic acid (EDTA) (35). Procedures followed to collect and store plasma samples, as well as laboratory quality control procedures and the methodology used to determine concentrations of HDL cholesterol, LDL cholesterol, total cholesterol, and triglycerides, have been reported in detail elsewhere (35).
Measurements of weight, height, and waist circumference were obtained at each examination according to standardized protocol described previously (36). Body mass index (BMI) was computed as weight in kilograms divided by squared height in meters (overweight: BMI ≥25).
At each examination, sociodemographic and behavioral characteristics (medication use, alcohol intake (ml/day), cigarette smoking, education, marital status, employment status, oral contraceptive use, and physical activity) were measured by self- and interviewer-administered questionnaires. Categorical variables were defined as smoking (never, past, or current), years of education (≤12, 13–15, or ≥16), marital status (never married, widowed, divorced or separated, or married), employment outside the home (none, part-time, or full-time), and oral contraceptive use (never, past, or current). At baseline, dieting to lose weight was categorized as never, past, or current.
The CARDIA Physical Activity History was used to calculate a physical activity score at each examination (37). The CARDIA Dietary History was used to assess dietary intake as percentage of kilocalories from total fat, saturated fat, protein, and carbohydrate at baseline (38). At the year 10 examination, oral contraceptive use in months was self-reported since age at menarche.
Reproductive history was obtained by self-report of number of pregnancies and births (live- or stillborn). Baseline parity groups were defined as nulliparous (zero livebirths) and parous (one or more livebirths). At follow-up examinations, participants were asked whether they were currently pregnant or breastfeeding and the number of times they had been pregnant, including the number of abortions, miscarriages, and stillbirths since the previous examination; duration of gestation; and dates of delivery. Pregnancies ending in miscarriages, in abortions, and/or after less than 20 weeks of gestation were counted as “short” pregnancies. Pregnancies of more than 20 weeks of gestation were counted as births and were classified as zero, one, and two or more.
Variables
Repeated-outcome measures
Changes in lipid profiles (total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides) were computed for three time intervals, each starting from baseline (0–5, 0–7, and 0–10 years), by subtracting the baseline from each follow-up measurement.
Time-dependent follow-up groups
Follow-up pregnancy and birth groups were constructed as time-dependent variables based on number of pregnancies and births during the three follow-up intervals. Women were assigned to one of four groups for each interval, defined as follows: P0 (zero pregnancies, nongravid), P1 (one or more miscarriages or abortions, short pregnancies), B1 (one birth), and B2 (two or more births). For the P0 group, no pregnancies occurred during the current or any prior interval. The P1 group had one or more short pregnancies but no births during current or prior intervals. The B1 group had only one birth and any number of short pregnancies during current or prior intervals. B2 included women each of whom had two or more births during the current interval; one or more birth(s) and any number of short pregnancies may have occurred in a prior interval(s). Group assignments continued into subsequent intervals unless new pregnancies and/or births had occurred since the last examination. For example, a woman reporting a miscarriage between years 0 and 5 and one birth and any number of additional miscarriages between years 5 and 7 would be classified as P1 for the year 0–5 interval and as B1 for the year 0–7 interval. If no further births were reported, classification as B1 would remain for the subsequent interval, year 0–10.
The analysis included time-dependent covariables (age, number of short pregnancies, changes in weight, waist circumference, months of oral contraceptive use, physical activity, and alcohol intake constructed by subtracting the baseline from the follow-up measurement) and categorical variables (smoking, marital, employment, educational status) for each time interval.
Statistical methods
Preliminary analyses involved a description of baseline characteristics stratified by baseline parity and race. Multiple linear regression methods (analysis of variance) were used to assess baseline differences in plasma lipids (HDL cholesterol, LDL cholesterol, total cholesterol, and triglycerides), age, height, weight, BMI, waist circumference, and dietary intake among follow-up pregnancy and birth groups (P0, P1, B1, B2). Chi-square tests were used to assess associations with baseline demographic and behavioral categorical variables. Two-sided tests were used to obtain p values (significance <0.05). The Kruskal-Wallis one-way test was used to assess differences in alcohol intake and physical activity due to skewedness in the distributions.
Plasma lipid measurements were assembled from years 0, 5, 7, and 10 along with fixed variables, race and study center, and time-dependent follow-up pregnancy and birth groups. Repeated-measures linear regression methods were used (PROC MIXED 8.2; SAS Institute, Inc., Cary, North Carolina) to assess the association of follow-up pregnancy and birth groups with plasma lipid changes over the three intervals. Two-way interaction terms were examined simultaneously (race, baseline parity, and age) to assess heterogeneity in the associations of lipid changes with follow-up groups by including appropriate cross-product terms in the models. Based on these analyses, models were stratified by baseline parity; adjusted means for HDL cholesterol, total cholesterol, LDL cholesterol, and triglyceride changes were contrasted among follow-up groups separately for each race. Both baseline and time-dependent covariables included in models as confounders were selected on the basis of their association with outcome measures independent of association with follow-up groups. Model fit was assessed by comparing Akaike Information Criterion statistics.
Minimally adjusted models included study center, time, baseline covariates (weight, height, waist circumference, dieting to lose weight, percentage of kilocalories from carbohydrate and saturated fat, alcohol intake, smoking, education, physical activity, oral contraceptive use) and time-dependent age and number of short pregnancies. Fully adjusted models additionally included time-dependent covariates (education, smoking, changes in body weight, waist circumference, alcohol intake, physical activity score, months of oral contraceptive use).
The overall significance of differences in lipid changes among time-dependent follow-up pregnancy and birth groups was tested within race and baseline parity-specific models. Differences in mean changes in HDL cholesterol, LDL cholesterol, total cholesterol, and triglycerides among follow-up groups were estimated and tested by pair-wise comparisons of the P1, B1, and B2 groups with the referent group, P0 (nongravid), and by comparing B1 with B2. We assumed that the covariance structure was compound symmetry in the repeated-measures regression models; analyses based on the assumption of an unstructured variance-covariance matrix yielded similar results.
Results
Of 1,952 women (980 Black, 972 White) for whom fasting plasma lipid measurements were avaliable, 506 (51.6 percent) Blacks and 800 (82.3 percent) Whites were nulliparous at baseline. For Blacks and Whites, respectively, follow-up pregnancy and birth groups included 445 and 482 women with no pregnancies, 119 and 112 women with one or more short pregnancies and no births (P1), 278 and 209 women with one birth (B1), and 136 and 169 women with two or more births (B2) during follow-up.
Baseline characteristics of the P0, P1, B1, and B2 groups at the end of follow-up are shown by race and baseline parity in tables 1 and 2. For those women nulliparous at baseline, baseline characteristics that differed among follow-up groups included age, oral contraceptive use, dieting to lose weight, and plasma total cholesterol for Blacks; and plasma LDL cholesterol and total cholesterol, education, smoking, oral contraceptive use, and weight for Whites. For those women parous at baseline, follow-up groups differed by baseline age, oral contraceptive use, dieting to lose weight, and plasma triglycerides for Blacks; and age, education, weight, BMI, overweight status, marital status (data not shown), and waist circumference for Whites. Baseline dietary intake did not differ among follow-up groups except for percentage of calories from saturated fat, carbohydrate, and protein among parous Blacks; and borderline differences in percentage of calories from total fat among nulliparous and parous Blacks.
Table 1. Baseline characteristics of follow-up pregnancy and birth groups of women nulliparous at baseline stratified by race, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Baseline characteristic | Follow-up pregnancy and birth group (end of follow-up) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| Blacks nulliparaus at baseline (n = 506) | Whites nulliparaus at baseline (n = 800) | |||||||||
|
|
|
|||||||||
| P0: no pregnancies (n = 224) | P1: one or more short pregnancies (n = 65) | B1: one birth (n = 135) | B2: two or more births (n = 82) | p value | P0: no pregnancies (n = 403) | P1: one or more short pregnancies (n = 89) | B1: one birth (n = 160) | B2: two or more births (n = 148) | p value | |
| Mean (standard deviation) | ||||||||||
| Age (years) | 23.6 (3.7) | 22.7 (3.4) | 22.7 (3.6) | 22.0 (3.1) | 0.002 | 25.1 (3.6) | 24.8 (3.4) | 25.4 (3.3) | 25.8 (3.1) | 0.10 |
| Weight (kg) | 68.7 (18.2) | 67.0 (18.9) | 66.0 (16.7) | 66.2 (13.6) | 0.46 | 63.1 (13.0) | 62.9 (12.0) | 61.4 (11.0) | 60.4 (9.6) | 0.08 |
| Height (cm) | 163.6 (6.8) | 163.3 (6.8) | 164.0 (8.8) | 162.9 (6.5) | 0.37 | 165.3 (6.7) | 165.9 (6.0) | 164.6 (5.7) | 165.3 (6.2) | 0.42 |
| Body mass index (kg/m2) | 25.7 (6.2) | 25.2 (6.7) | 24.5 (5.6) | 25.0 (5.1) | 0.47 | 23.1 (4.4) | 22.9 (4.1) | 22.7 (3.7) | 22.2 (3.3) | 0.10 |
| Waist circumference (cm) | 75.9 (13.8) | 73.3 (12.9) | 73.0 (10.3) | 74.1 (10.3) | 0.13 | 71.5 (9.2) | 71.1 (8.9) | 70.6 (7.8) | 69.6 (7.3) | 0.11 |
| Plasma levels | ||||||||||
| Triglycerides (mg/dl) | 60.5 (28.9) | 54.2 (21.8) | 59.4 (27.9) | 57.5 (24.5) | 0.41 | 67.8 (33.6) | 63.4 (27.5) | 70.7 (35.0) | 64.6 (30.9) | 0.25 |
| HDL* cholesterol (mg/dl) | 56.7 (13.1) | 58.5 (14.1) | 55.3 (12.0) | 55.1 (11.5) | 0.28 | 57.5 (12.2) | 57.0 (13.6) | 58.3 (13.1) | 57.2 (13.1) | 0.85 |
| LDL* cholesterol (mg/dl) | 113.8 (32.4) | 109.3 (28.3) | 106.1 (28.9) | 110.3 (37.1) | 0.17 | 104.2 (27.2) | 98.0 (24.8) | 108.3 (29.4) | 106.3 (30.5) | 0.04 |
| Total cholesterol (mg/dl) | 182.6 (34.6) | 178.6 (30.6) | 173.2 (31.0) | 176.9 (35.9) | 0.07 | 175.3 (29.2) | 167.6 (24.7) | 180.7 (32.0) | 176.4 (33.0) | 0.01 |
| Dietary intake (% kcal) | ||||||||||
| Total fat | 36.7 (5.6) | 38.5 (4.9) | 37.5 (5.5) | 38.0 (5.3) | 0.07 | 35.8 (6.1) | 35.8 (6.6) | 36.6 (6.9) | 36.1 (5.9) | 0.52 |
| Saturated fat | 6.0 (1.2) | 6.3 (1.2) | 6.1 (1.2) | 6.4 (1.3) | 0.16 | 6.0 (1.3) | 6.0 (1.4) | 6.1 (1.4) | 6.1 (1.4) | 0.91 |
| Carbohydrate | 49.0 (6.8) | 46.8 (6.7) | 47.9 (7.0) | 48.1 (7.0) | 0.11 | 47.6 (7.1) | 47.0 (7.4) | 46.3 (7.7) | 46.7 (7.4) | 0.27 |
| Protein | 13.8 (2.2) | 14.2 (2.1) | 14.2 (2.6) | 13.7 (2.5) | 0.35 | 15.4 (2.9) | 15.4 (3.0) | 15.4 (3.0) | 15.6 (2.6) | 0.90 |
| Physical activity score† | 236.5 (315.0) | 262.0 (269.0) | 231.0 (284.0) | 265.0 (336.0) | 0.20 | 404.0 (340.0) | 354.0 (378.0) | 362.5 (396.0) | 387.5 (284.0) | 0.76 |
| Alcohol intake† (ml/day) | 0 (5.0) | 2.4 (5.2) | 0 (5.2) | 0 (4.9) | 0.30 | 4.8 (12.3) | 4.9 (12.3) | 4.9 (14.3) | 7.2 (14.3) | 0.11 |
| No. (%) | ||||||||||
| Overweight (body mass index ≥25) | 93 (41.5) | 26 (40.0) | 45 (33.3) | 34 (41.5) | 0.45 | 87 (21.6) | 17 (19.1) | 33 (20.6) | 19 (12.8) | 0.14 |
| Education | 0.20 | <0.001 | ||||||||
| High school or less | 78 (34.8) | 20 (30.8) | 61 (45.2) | 31 (37.8) | 73 (18.1) | 23 (25.8) | 36 (22.5) | 16 (10.8) | ||
| Some college | 96 (42.9) | 35 (53.8) | 54 (40.0) | 37 (45.1) | 131 (32.5) | 29 (32.6) | 47 (29.4) | 29 (19.6) | ||
| College graduate | 50 (22.3) | 10 (15.4) | 20 (14.8) | 14 (17.1) | 199 (49.4) | 37 (41.6) | 77 (48.1) | 103 (69.6) | ||
| Smoking | 0.77 | 0.01 | ||||||||
| Never | 162 (72.3) | 44 (67.7) | 97 (71.9) | 56 (68.3) | 244 (60.6) | 37 (41.6) | 91 (56.9) | 98 (66.2) | ||
| Past | 13 (5.8) | 5 (7.7) | 4 (3.0) | 4 (4.9) | 71 (17.6) | 22 (24.7) | 34 (21.3) | 25 (16.9) | ||
| Current | 49 (21.9) | 16 (24.6) | 34 (25.1) | 22 (26.8) | 88 (21.8) | 30 (33.7) | 35 (21.9) | 25 (16.9) | ||
| Dieting to lose weight | <0.001 | 0.31 | ||||||||
| Never | 108 (48.2) | 43 (66.2) | 96 (71.1) | 54 (65.9) | 157 (39.0) | 35 (39.3) | 55 (34.4) | 47 (31.8) | ||
| Past | 99 (44.2) | 16 (24.6) | 35 (25.9) | 20 (24.4) | 197 (48.8) | 49 (55.1) | 82 (51.2) | 80 (54.0) | ||
| Current | 17 (7.6) | 6 (9.2) | 4 (3.0) | 8 (9.7) | 49 (12.2) | 5 (5.6) | 23 (14.4) | 21 (14.2) | ||
| Oral contraceptive use | 0.02 | <0.001 | ||||||||
| Never | 80 (35.7) | 16 (24.6) | 35 (25.9) | 12 (14.6) | 153 (38.0) | 19 (21.4) | 29 (18.1) | 31 (21.0) | ||
| Past | 71 (31.7) | 26 (40.0) | 52 (38.5) | 33 (40.2) | 149 (37.0) | 51 (57.3) | 66 (41.3) | 56 (37.8) | ||
| Current | 73 (32.6) | 23 (35.4) | 48 (35.6) | 37 (45.1) | 101 (25.0) | 19 (21.4) | 65 (40.6) | 61 (41.2) | ||
HDL, high density lipoprotein; LDL, low density lipoprotein.
Values expressed as median (interquartile range); Kruskal-Walllis test: nonparametric one-way tests to assess differences among groups.
Table 2. Baseline characteristics of follow-up pregnancy and birth groups of women parous at baseline stratified by race, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Baseline characteristic | Follow-up pregnancy and birth group (end of follow-up) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| Blacks parous at baseline (n = 474) | Whites parous at baseline (n = 172) | |||||||||
|
|
|
|||||||||
| P0: no pregnancies (n = 221) |
P1: one or more short pregnancies (n = 54) |
B1: one birth (n = 144) |
B2: two or more births (n = 55) |
p value | P0: no pregnancies (n = 79) |
P1: one or more short pregnancies (n = 23) |
B1: one birth (n = 49) |
B2: two or more births (n = 21) |
p value | |
| Mean (standard deviation) | ||||||||||
| Age (years) | 27.0 (3.1) | 25.9 (3.3) | 25.2 (3.6) | 23.5 (3.7) | 0.001 | 27.9 (2.3) | 26.0 (3.8) | 26.2 (3.2) | 25.7 (3.1) | <0.001 |
| Weight (kg) | 71.6 (18.5) | 68.7 (19.0) | 68.2 (17.1) | 69.3 (21.3) | 0.35 | 68.4 (15.7) | 65.5 (10.5) | 62.1 (15.1) | 57.7 (6.8) | 0.008 |
| Height (cm) | 163.4 (6.5) | 163.3 (6.4) | 163.8 (6.8) | 163.6 (6.6) | 0.95 | 163.6 (6.9) | 166.0 (5.5) | 163.7 (6.0) | 165.1 (5.3) | 0.36 |
| Body mass index (kg/m2) | 26.9 (7.0) | 25.9 (7.4) | 25.4 (6.0) | 25.9 (7.2) | 0.22 | 25.6 (5.6) | 23.9 (4.4) | 23.2 (5.4) | 21.3 (2.9) | 0.003 |
| Waist circumference (cm) | 79.2 (12.7) | 78.0 (14.5) | 76.0 (11.9) | 77.1 (15.1) | 0.14 | 78.6 (11.7) | 73.8 (8.1) | 72.2 (10.3) | 69.2 (5.2) | <0.001 |
| Plasma levels | ||||||||||
| Triglycerides (mg/dl) | 64.9 (30.3) | 63.4 (27.8) | 61.2 (22.7) | 75.5 (34.3) | 0.02 | 78.1 (43.3) | 81.9 (50.4) | 71.5 (31.7) | 60.0 (26.4) | 0.21 |
| HDL* cholesterol (mg/dl) | 54.0 (12.4) | 55.5 (12.6) | 56.1 (12.3) | 53.7 (13.8) | 0.38 | 48.9 (11.2) | 50.0 (12.7) | 51.9 (12.4) | 50.4 (8.6) | 0.57 |
| LDL* cholesterol (mg/dl) | 111.0 (31.8) | 107.8 (31.3) | 109.5 (24.8) | 111.2 (31.5) | 0.89 | 109.0 (30.8) | 100.0 (23.8) | 107.9 (26.5) | 102.9 (26.5) | 0.52 |
| Total cholesterol (mg/dl) | 177.9 (32.7) | 175.9 (33.1) | 177.9 (28.4) | 180.0 (33.2) | 0.93 | 173.5 (30.4) | 166.3 (29.1) | 174.0 (27.6) | 165.2 (25.1) | 0.48 |
| Dietary intake (% kcal) | ||||||||||
| Total fat | 36.5 (6.4) | 38.0 (5.4) | 37.7 (5.6) | 38.5 (6.2) | 0.06 | 36.7 (6.6) | 36.6 (6.1) | 36.8 (5.1) | 38.2 (5.5) | 0.77 |
| Saturated fat | 6.0 (1.3) | 6.2 (1.2) | 6.3 (1.3) | 6.5 (1.3) | 0.03 | 6.3 (1.5) | 6.6 (1.3) | 6.4 (1.4) | 6.6 (1.3) | 0.80 |
| Carbohydrate | 49.6 (7.8) | 47.3 (6.9) | 47.6 (7.4) | 46.4 (8.4) | 0.007 | 48.1 (8.6) | 47.0 (7.7) | 47.0 (6.6) | 45.9 (4.4) | 0.61 |
| Protein | 13.4 (2.7) | 14.0 (2.3) | 14.4 (2.7) | 14.4 (3.1) | 0.003 | 14.9 (2.9) | 14.6 (2.9) | 15.9 (3.3) | 15.0 (2.2) | 0.26 |
| Physical activity score† | 216.0 (288.0) | 181.0 (292.0) | 225.5 (299.0) | 195.0 (360.0) | 0.66 | 288.0 (334.0) | 294.0 (255.0) | 298.0 (255.0) | 252.0 (198.0) | 0.83 |
| Alcohol intake† (ml/day) | 0 (8.2) | 1.2 (7.6) | 0 (4.8) | 0 (7.2) | 0.32 | 0 (4.9) | 0 (15.2) | 2.4 (7.6) | 2.4 (11.9) | 0.22 |
| No. (%) | ||||||||||
| Overweight (body mass index >25) | 116 (52.5) | 25 (46.3) | 61 (42.4) | 23 (41.8) | 0.21 | 38 (48.1) | 7 (30.4) | 10 (20.4) | 1 (4.8) | <0.001 |
| Education | 0.14 | 0.002 | ||||||||
| High school or less | 134 (60.6) | 27 (50.0) | 69 (47.9) | 34 (61.8) | 47 (59.5) | 13 (56.5) | 22 (44.9) | 8 (38.1) | ||
| Some college | 72 (32.6) | 21 (38.9) | 58 (40.3) | 14 (25.5) | 25 (31.7) | 8 (34.8) | 15 (30.6) | 3 (14.3) | ||
| College graduate | 15 (6.8) | 6 (11.1) | 17 (11.8) | 7 (12.7) | 7 (8.8) | 2 (8.7) | 12 (24.5) | 10 (47.6) | ||
| Smoking | 0.25 | 0.42 | ||||||||
| Never | 109 (49.3) | 26 (48.2) | 79 (54.9) | 31 (56.4) | 36 (45.6) | 6 (26.1) | 27 (55.1) | 10 (47.6) | ||
| Past | 26 (11.8) | 4 (7.4) | 20 (13.9) | 2 (3.6) | 16 (20.2) | 5 (21.7) | 9 (18.4) | 4 (19.1) | ||
| Current | 86 (38.9) | 24 (44.4) | 45 (31.3) | 22 (40.0) | 27 (34.2) | 12 (52.2) | 13 (26.5) | 7 (33.3) | ||
| Dieting to lose weight | 0.05 | 0.12 | ||||||||
| Never | 136 (61.5) | 36 (66.7) | 88 (61.1) | 44 (80.0) | 27 (34.2) | 12 (52.2) | 23 (46.9) | 10 (47.6) | ||
| Past | 70 (31.7) | 12 (22.2) | 38 (26.4) | 7 (12.7) | 42 (53.2) | 6 (26.1) | 19 (38.8) | 11 (52.4) | ||
| Current | 15 (6.8) | 6 (11.1) | 18 (12.5) | 4 (7.3) | 10 (12.7) | 5 (21.7) | 7 (14.3) | 0 (0.0) | ||
| Oral contraceptive use | <0.001 | 0.11 | ||||||||
| Never | 37 (16.7) | 7 (13.0) | 12 (8.3) | 3 (5.5) | 10 (12.7) | 1 (4.3) | 7 (14.3) | 7 (33.3) | ||
| Past | 138 (62.4) | 26 (48.1) | 67 (46.5) | 23 (41.8) | 55 (69.6) | 18 (78.3) | 29 (59.2) | 10 (47.6) | ||
| Current | 46 (20.8) | 21 (38.9) | 65 (45.2) | 29 (52.7) | 14 (17.7) | 4(17.4) | 13 (26.5) | 4 (19.1) | ||
HDL, high density lipoprotein; LDL, low density lipoprotein.
Values expressed as median (interquartile range); Kruskal-Wallis test: nonparametric one-way tests to assess differences among groups.
Evidence for a strong two-way interaction by baseline parity was found within the association of time-dependent follow-up pregnancy and birth groups with HDL cholesterol change (p < 0.001) but not with LDL cholesterol, total cholesterol, or triglyceride changes. The two-way interaction term for race in the association for HDL cholesterol was not significant (p = 0.10). Interactions by age, BMI, and race were not significant for HDL cholesterol or other lipids.
For women nulliparous at baseline (tables 3 and 4), greater declines in HDL cholesterol were associated with the B1 and B2 groups compared with the P0 group during follow-up in minimally adjusted models (p < 0.001); differences of −4 to −5 mg/dl in HDL cholesterol changes (figures 1 and 2) were similar for Blacks and Whites. HDL cholesterol declines were also greater for the P1 compared with the P0 group (p < 0.05) for Blacks nullliparous at baseline. In minimally adjusted models, smaller LDL cholesterol declines and larger triglyceride increases were associated with short pregnancies and one or more births compared with no pregnancies (table 3) in Blacks nulliparous at baseline, and greater total cholesterol increases were associated with short pregnancies compared with none (table 4) in Whites nulliparous at baseline.
Table 3. Mean changes in plasma lipoprotein cholesterol and triglycerides by follow-up pregnancy and birth groups of Black women nulliparous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Lipid profile | Time-dependent follow-up pregnancy and birth group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| P0: no pregnancies (referent) (n = 224) |
P1: one or more short
pregnancies (n = 65) |
B1: one birth (n = 135) |
B2: two or more births (n = 82) |
Global p value | |||||
|
|
|
|
|
||||||
| Mean | 95% CI† | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| ΔTotal cholesterol (mg/dl) | |||||||||
| Minimally adjusted‡ | −7.1 | − 9.7, −4.5 | −2.3 | −7.4, 2.8 | −5.0 | −8.2, −1.8 | −7.7 | −12.6, −2.8 | 0.28 |
| Fully adjusted§ | −6.4 | − 9.0, −3.8 | −1.4 | − 6.6, 3.8 | −6.5 | −9.8, −3.2 | −9.3 | −14.4, −4.2 | 0.17 |
| ΔHDL† cholesterol (mg/dl) | |||||||||
| Minimally adjusted | 0.5 | − 0.7, 1.6 | −3.2** | −5.4, −1.0 | −3.3*** | −4.7, −1.9 | −4.9*** | −7.0, −2.8 | <0.001 |
| Fully adjusted | −0.1 | − 1.1, 1.0 | −2.4 | −4.5, −0.3 | −2.9*** | −4.2, −1.6 | −4.1*** | −6.2, −2.1 | 0.003 |
| ΔLDL† cholesterol (mg/dl) | |||||||||
| Minimally adjusted | −8.1 | − 10.5, −5.8 | −1.6* | −6.2, 3.1 | −3.8* | −6.8, −0.9 | −5.4 | −9.8, −0.9 | 0.04 |
| Fully adjusted | −7.1 | − 9.4, −4.7 | − 1.3 | − 6.0, −3.3 | −5.4 | −8.4, −2.5 | −7.4 | −12.0, −2.8 | 0.14 |
| ΔTriglycerides (mg/dl) | |||||||||
| Minimally adjusted | 0.8 | − 2.4, 4.0 | 9.8* | 3.2, 16.3 | 7.7** | 3.7, 11.8 | 10.4* | 4.1, 16.7 | 0.02 |
| Fully adjusted | 2.2 | − 0.9, 5.2 | 8.7 | 2.2, 15.1 | 6.1 | 2.0, 10.1 | 9.0 | 2.7, 15.3 | 0.17 |
p < 0.05;
p < 0.01;
p < 0.001; pairwise comparisons to referent group.
CI, confidence interval; HDL, high density lipoprotein; LDL, low density lipoprotein.
Minimally adjusted models included study center, time, baseline characteristics (body weight, height, waist circumference, education, smoking status, dieting to lose weight, oral contraceptive use, dietary carbohydrates and saturated fat, alcohol intake, and physical activity score), and time-dependent age and number of short pregnancies.
Fully adjusted models additionally included time-dependent covariates (smoking status, months of oral contraceptive use, education, and changes in body weight, waist circumference, physical activity score, and alcohol intake).
Table 4. Mean changes in plasma lipoprotein cholesterol and triglycerides by follow-up pregnancy and birth groups of White women nulliparous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Lipid profile | Time-dependent follow-up pregnancy and birth group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| P0: no pregnancies (referent) (n = 403) | P1: one or more short pregnancies (n = 89) | B1: one birth (n = 160) | B2: two or more births (n = 148) | Global p value | |||||
|
|
|
|
|
||||||
| Mean | 95% CI† | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| ΔTotal cholesterol (mg/dl) | |||||||||
| Minimally adjusted‡ | −1.6 | −3.6, 0.3 | 4.7* | −0.4, 9.8 | −3.6 | −6.5, −0.7 | −4.1 | −7.6, −0.6 | 0.02 |
| Fully adjusted§ | −0.6 | −2.5, 1.2 | 4.1 | −1.0, 9.2 | −6.3** | −9.4, −3.2 | −5.1* | −8.7, −1.4 | <0.001 |
| ΔHDL† cholesterol (mg/dl) | |||||||||
| Minimally adjusted | 0.9 | 0.1, 1.8 | 0.1 | −2.0, 2.3 | −2.7*** | −4.0, −1.5 | −3.8*** | −5.3, −2.3 | <0.001 |
| Fully adjusted | 0.6 | −0.1, 1.4 | −0.3 | −2.4, 1.8 | −2.2*** | −3.5, −1.0 | −2.7*** | −4.3, −1.2 | <0.001 |
| ΔLDL† cholesterol (mg/dl) | |||||||||
| Minimally adjusted | −3.8 | −5.5, −2.0 | 2.2 | −2.4, 6.7 | −2.8 | −5.4, −0.2 | −2.5 | −5.7, 0.6 | 0.15 |
| Fully adjusted | −2.7 | −4.3, −1.1 | 1.9 | −2.5, 6.4 | −5.6 | −8.3, −3.0 | −4.4 | −7.6, −1.3 | 0.02 |
| ΔTriglycerides (mg/dl) | |||||||||
| Minimally adjusted | 4.2 | 1.4, 7.0 | 8.7 | 0.9, 16.4 | 5.9 | 1.4, 10.3 | 7.6 | 2.3, 12.9 | 0.63 |
| Fully adjusted | 5.0 | 2.3, 7.6 | 9.2 | 1.6, 16.8 | 4.1 | −0.5, 8.7 | 7.1 | 1.7, 12.6 | 0.61 |
p < 0.05;
p < 0.01;
p < 0.001; pairwise comparisons to referent group.
CI, confidence interval; HDL, high density lipoprotein; LDL, low density lipoprotein.
Minimally adjusted models included study center, time, baseline characteristics (body weight, height, waist circumference, education, smoking status, dieting to lose weight, oral contraceptive use, dietary carbohydrates and saturated fat, alcohol intake, and physical activity score), and time-dependent age and number of short pregnancies.
Fully adjusted models additionally included time-dependent covariates (smoking status, months of oral contraceptive use, education, and changes in body weight, waist circumference, physical activity score, and alcohol intake).
Figure 1.
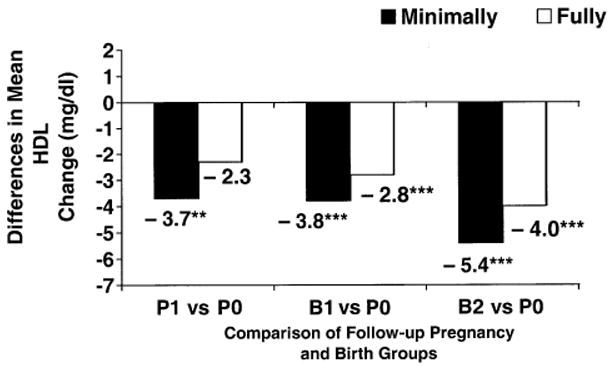
Differences in mean high density lipoprotein (HDL) cholesterol changes (mg/dl) between follow-up short pregnancy and birth groups (P1 (one or more short pregnancies), B1 (one birth), and B2 (two or more births)) vs. the no pregnancies group (P0) from minimally and fully adjusted models for Black women nulliparous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996. ** p < 0.01; *** p < 0.001.
Figure 2.
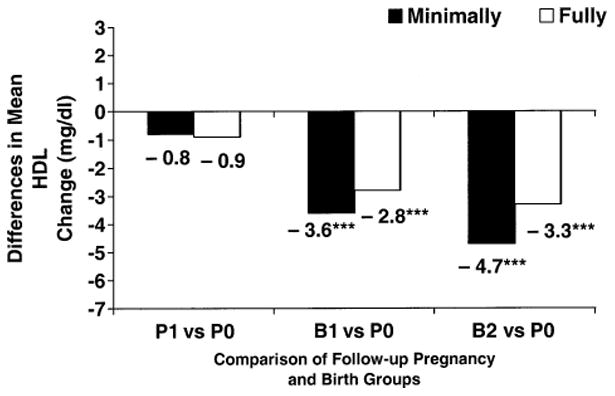
Differences in mean high density lipoprotein (HDL) cholesterol changes (mg/dl) between follow-up short pregnancy and birth groups (P1 (one or more short pregnancies), B1 (one birth), and B2 (two or more births)) vs. the no pregnancies group (P0) from minimally and fully adjusted models for White women nulliparous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996. *** p < 0.001.
In fully adjusted models for women nulliparous at baseline, differences in HDL cholesterol declines for the B1 and B2 groups compared with the P0 group were attenuated to −2.8 to −4.0 mg/dl for Blacks and −2.8 to −3.3 mg/dl for Whites, but they remained significantly greater (figures 1 and 2). The B1 and B2 groups did not differ from one another. Fully adjusted HDL cholesterol declines for the P1 group compared with the P0 group were no longer significant for Blacks. HDL cholesterol declines associated with follow-up groups did not vary by level of weight gain or increased waist girth during follow-up (two-way interaction p > 0.10). In fully adjusted models for Whites nulliparous at baseline, total cholesterol declines were greater for the B1 and B2 groups (differences of −4.5 to −5.7 mg/dl) compared with the P0 group (p < 0.01), and LDL cholesterol declines for the B1 group (difference of −2.9 mg/dl) compared with the P0 group were somewhat greater (p = 0.053). For Blacks nulliparous at baseline, fully adjusted LDL cholesterol and triglyceride changes did not differ among follow-up groups.
For women parous at baseline, no association of HDL cholesterol or other lipid changes with follow-up groups was found for either race, except for somewhat greater triglyceride increases among Whites (tables 5 and 6) in minimally adjusted models. Fully adjusted plasma lipid changes were not associated with follow-up groups for either race except for wide variability among follow-up groups in triglyceride changes among Whites parous at baseline due to influential data points and small sample size (table 6). In fully adjusted models, short pregnancies were not associated with any differences in blood lipid profiles regardless of baseline parity or race.
Table 5. Mean changes in plasma lipoprotein cholesterol and triglycerides by follow-up pregnancy and birth groups of Black women parous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Lipid profile | Time-dependent follow-up pregnancy and birth group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| P0: no pregnancies (referent) (n = 221) | P1: one or more short pregnancies (n = 54) | B1: one birth (n = 144) | B2: two or more births (n = 55) | Global p value | |||||
|
|
|
|
|
||||||
| Mean | 95% CI† | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| ΔTotal cholesterol (mg/dl) | |||||||||
| Minimally adjusted† | −1.0 | −3.7, 1.7 | −0.7 | −6.6, 5.2 | −2.0 | −5.2, 1.1 | −7.2 | −12.9, −1.4 | 0.27 |
| Fully adjusted‡ | −1.2 | −3.9, 1.5 | 0.4 | −5.6, 6.3 | −3.3 | −6.6, 0.0 | −8.4 | −14.2, −2.6 | 0.12 |
| ΔHDL* cholesterol (mg/dl) | |||||||||
| Minimally adjusted | 1.0 | −0.2, 2.3 | 0.3 | −2.4, 3.0 | −1.1 | −2.6, 0.3 | −0.6 | −3.3, 2.0 | 0.15 |
| Fully adjusted | 1.0 | −0.2, 2.2 | 0.3 | −2.4, 2.9 | −0.9 | −2.4, 0.5 | −0.2 | −2.8, 2.3 | 0.25 |
| ΔLDL* cholesterol (mg/dl) | |||||||||
| Minimally adjusted | −3.3 | −5.9, −0.8 | −3.2 | −8.8, 2.4 | −2.9 | −5.9, 0.2 | −8.6 | −14.1, −3.1 | 0.26 |
| Fully adjusted | −3.5 | −6.0, −0.9 | −2.0 | −7.5, 3.6 | −3.9 | −7.0, −0.9 | −10.1 | −15.6, −4.7 | 0.12 |
| ΔTriglycerides (mg/dl) | |||||||||
| Minimally adjusted | 4.7 | 0.8, 8.7 | 5.6 | −3.6, 14.8 | 7.8 | 3.0, 12.6 | 8.0 | −0.8, 16.8 | 0.78 |
| Fully adjusted | 4.5 | 0.7, 8.3 | 5.7 | −3.0, 14.5 | 5.8 | 1.1, 10.5 | 7.1 | −1.3, 15.5 | 0.95 |
CI, confidence interval; HDL, high density lipoprotein; LDL, low density lipoprotein.
Minimally adjusted models included study center, time, baseline characteristics (body weight, height, waist circumference, education, smoking status, dieting to lose weight, oral contraceptive use, dietary carbohydrates and saturated fat, alcohol intake, and physical activity score), and time-dependent age and number of short pregnancies.
Fully adjusted models additionally included time-dependent covariates (smoking status, months of oral contraceptive use, education, and changes in body weight, waist circumference, physical activity score, and alcohol intake).
Table 6. Mean changes in plasma lipoprotein cholesterol and triglycerides by follow-up pregnancy and birth groups of White women parous at baseline, the Coronary Artery Risk Development in Young Adults Study, 1985–1996.
| Lipid profile | Time-dependent follow-up pregnancy and birth group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| P0: no pregnancies (referent) (n = 79) | P1: one or more short pregnancies (n = 23) | B1: one birth (n = 49) | B2: two or more births (n = 21) | Global p value | |||||
|
|
|
|
|
||||||
| Mean | 95% CI* | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| ΔTotal cholesterol (mg/dl) | |||||||||
| Minimally adjusted† | 6.8 | 1.3, 12.2 | 8.1 | −2.5, 18.7 | −1.0 | −7.4, 5.5 | −1.2 | −11.7, 9.2 | 0.26 |
| Fully adjusted‡ | 5.9 | 0.5, 11.3 | 10.8 | 0.1, 21.5 | −1.3 | −7.8, 5.1 | 0.3 | −10.3, 10.9 | 0.19 |
| ΔHDL* cholesterol (mg/dl) | |||||||||
| Minimally adjusted | 1.2 | −0.7, 3.1 | 4.7 | 1.0, 8.5 | 1.8 | −0.5, 4.0 | 2.0 | −1.6, 5.7 | 0.46 |
| Fully adjusted | 1.3 | −0.3, 3.0 | 3.7 | 0.3, 7.0 | 1.4 | −0.6, 3.4 | 1.4 | −2.0, 4.7 | 0.65 |
| ΔLDL* cholesterol (mg/dl) | |||||||||
| Minimally adjusted | 1.9 | −3.1, 6.9 | 0.7 | −8.8, 10.2 | −3.4 | −9.2, 2.5 | −2.2 | −11.6, 7.2 | 0.61 |
| Fully adjusted | 1.1 | −3.7, 5.9 | 3.6 | −5.8, 13.0 | −3.4 | −9.0, 2.3 | −0.4 | −9.7, 8.9 | 0.50 |
| ΔTriglycerides (mg/dl) | |||||||||
| Minimally adjusted | 15.2 | 7.6, 22.8 | 8.1 | −7.4, 23.7 | 0.1 | −9.1, 9.2 | −6.8 | −22.2, 8.6 | 0.09 |
| Fully adjusted | 14.2 | 7.4, 20.9 | 12.2 | −2.2, 26.6 | 0.5 | −7.7, 8.8 | −6.0 | −20.4, 8.3 | 0.06 |
CI, confidence interval; HDL, high density lipoprotein; LDL, low density lipoprotein.
Minimally adjusted models included study center, time, baseline characteristics (body weight, height, waist circumference, education, smoking status, dieting to lose weight, oral contraceptive use, dietary carbohydrates and saturated fat, alcohol intake, and physical activity score), and time-dependent age and number of short pregnancies.
Fully adjusted models additionally included time-dependent covariates (smoking status, months of oral contraceptive use, education, and changes in body weight, waist circumference, physical activity score, and alcohol intake).
Discussion
To our knowledge, this is the largest prospective study of Black women and White women to examine long-term changes in plasma lipids associated with childbearing in reference to nongravidity. For both races, HDL cholesterol declines of −4 to −5 mg/dl associated with a first birth versus no pregnancies were attenuated to −3 to −4 mg/dl after accounting for changes in behavioral covariates, body weight, and waist circumference. For women nulliparous at baseline, additional HDL cholesterol declines (−0.5 mg/dl for Whites and −1.2 mg/dl for Blacks) with two or more follow-up births were not significantly different from those for one birth. Similarly, HDL cholesterol changes were not associated with higher-order births among women already parous at baseline. These findings indicate that HDL cholesterol declines associated with a first birth persist over several years, are similar for Black women and White women, and are not mediated primarily by weight gain, increased central adiposity, or behavioral changes such as smoking cessation, alcohol intake, oral contraceptive use, and physical activity associated with childbearing. Low HDL cholesterol (<40 mg/dl) is a strong independent predictor of coronary heart disease (39, 40). Each 1-mg/dl increase in HDL cholesterol is estimated to decrease coronary heart disease risk by 2–3 percent (41), and a 10 percent decline in HDL cholesterol over 12 years equates to a 10 percent increase in coronary heart disease risk (42). The 6 percent HDL cholesterol decline in 10 years among childbearing women may potentially increase their coronary heart disease risk by 6–12 percent.
Among Blacks, HDL cholesterol declines associated with short pregnancies were nonsignificant after we accounted for behavior changes. This lack of an association of short pregnancies with HDL cholesterol declines is consistent with the dominant hyperlipidemia and insulin resistance observed in the second half of pregnancy. To our knowledge, short pregnancies have not been evaluated prospectively, and studies have combined this group with the nonparous comparison group.
Our findings are consistent with a previous CARDIA analysis showing that a first birth was associated with a 2-year decrease in HDL cholesterol (−2.6 (standard error, 1.3) mg/dl) compared with an increase (2.5 (standard error, 0.4) mg/dl) for women who had never given birth (28). This net 5.1-mg/dl lower HDL cholesterol level after a first birth in the 2-year CARDIA study is somewhat higher than our estimate of a 3–4-mg/dl lower HDL cholesterol level over 10 years. However, the estimates differ because of stable HDL cholesterol levels rather than increases over time among nongravid women in the current analysis. A 3-year prospective study estimated that HDL cholesterol decreased by −2.43 mg/dl in women with one or more pregnancies independent of a change in BMI, alcohol consumption, physical activity, employment, smoking, and oral contraceptive use, but dietary intake was not reported (27).
One prospective study and several cross-sectional studies reported an inverse correlation with parity rather than a first-birth effect. In the Framingham Offspring Study (26), 8-year declines in both total (−2.7 mg/dl) and HDL (−1.05 mg/dl) cholesterol per birth for White women were independent of smoking, change in physical activity, alcohol intake, BMI, and oral contraceptive use. However, parity (total number of births) was examined instead of change in parity during follow-up. Several large, population-based, cross-sectional studies reported negative correlations between gravidity or parity and HDL cholesterol levels in both pre- and postmenopausal women, or they found that lower HDL cholesterol levels were associated with a high parity threshold (2, 22–25). For example, in the Rancho Bernardo Study of women aged 50–89 years, HDL cholesterol levels were −4.9 mg/dl lower in those with five or more pregnancies versus four or fewer pregnancies independent of weight change since age 18 years (24). In the Hispanic Health and Nutrition Examination Survey (25), six to eight pregnancies versus none was associated with −3.7 to −3.9 mg/dl lower HDL cholesterol levels in Mexican-American and Cuban-American women. An explanation for the association of high parity instead of a first birth is that cross-sectional studies are unable to assess the temporality of pregnancy and changes in HDL cholesterol or the influence of socioeconomic status on childbearing patterns, weight gain, dietary habits and obesity, or changes in other factors that may influence risk factors for heart disease in women.
Prospective studies have found that parity is not associated with changes in LDL cholesterol and triglycerides (26, 28), but total cholesterol declines associated with parity have been reported in Whites (26). In our study, declines of −5 mg/dl in total cholesterol and −2 to −3 mg/dl in LDL cholesterol were associated with primiparity in Whites. Although total cholesterol declines mirrored HDL cholesterol declines in both races, total cholesterol and LDL cholesterol declines were similar for nongravid and primiparous groups of Black women. The CARDIA study previously reported favorable 10-year secular trends in total and LDL cholesterol for Black women and unfavorable trends in HDL cholesterol and BMI for both races (43). The major contributor to the downward secular trends in total and LDL cholesterol was consumption of a less atherogenic diet (35). Our 10-year analysis reflects these same trends, but greater HDL cholesterol declines after a first birth persist independent of these secular trends among nongravid CARDIA study women. No differences in total and LDL cholesterol declines among childbearing groups for Blacks may be related to potential confounding from changes in dietary intake or other factors during follow-up that were not measured in this study. However, the estimates were adjusted for baseline dietary intake.
Less favorable lipid profiles have been associated with oral contraceptive use by both Blacks and Whites; oral contraceptive use is associated with higher LDL cholesterol (26) and lower HDL cholesterol (44) levels, with the progestin dose correlating with decreasing HDL cholesterol levels (45). In the CARDIA study, current oral contraceptive use declined in Blacks and Whites from 34.8 percent and 28.8 percent at baseline to 12.0 percent and 17.3 percent, respectively, by year 10. Oral contraceptive use acted as a weak confounder of the association between childbearing and lipid changes and was inversely associated with HDL cholesterol change. Other behavioral characteristics including physical activity, smoking, and alcohol intake contributed to confounding of HDL cholesterol declines since these behaviors tend to decrease during and often remain lower after pregnancy. Weight gain had the greatest adverse influence on HDL cholesterol declines associated with a first birth consistent with excess weight gain of 1–6 kg among CARDIA study primiparas (29, 30).
In addition, ours is the first prospective study known to examine lipid profile changes associated with childbearing in Black women and White women separately. On the basis of our findings of no significant interaction of follow-up groups by race, declines in HDL cholesterol associated with a first birth were similar for Whites and Blacks. No association between childbearing and changes in other plasma lipids was found among Blacks nulliparous at baseline; however, among Whites, total cholesterol declines were strongly associated with a first birth but LDL cholesterol declines were weakly associated overall. Thus, a first birth appears to exert adverse effects on lipid profiles principally because of the declines in HDL cholesterol.
Our study is the largest known population-based sample with the longest follow-up period to include repeated measurements of plasma lipids before and after each pregnancy and to account for secular trends and aging by comparison with nongravid women. In addition, we controlled for a number of potential confounders of the association between childbearing and plasma lipid changes, including changes in weight, central adiposity, and behavioral characteristics. Limitations of this study include the variable time interval from delivery to the next examination, lack of data on dietary changes, and lack of information on whether women who were never pregnant had experienced infertility due to hormonal disorders such as polycystic ovary syndrome or other factors that could affect lipids. Despite our large sample size, some groups were relatively small, limiting the power to detect differences.
Our findings support the hypothesis that pregnancy exerts persistent adverse effects on HDL cholesterol that are not primarily mediated by weight gain, central adiposity, or selected behaviors. The effect is not cumulative with additional births but occurs primarily after a first birth. Mechanisms to explain this association include endocrine changes due to a first birth that may induce long-term alterations in lipid and glucose metabolism not mediated by excess fat deposition. For example, lower concentrations of serum dehydroepiandrosterone sulfate (DHEAS) and dehydroepiandrosterone (DHEA) have been associated with a first fullterm pregnancy rather than increasing parity (46, 47). Lower basal serum prolactin levels have been found in primiparas than nulliparas (48, 49) independent of age, and lower levels in parous women were unrelated to number of pregnancies (48). Hormonal changes of pregnancy that promote adipose tissue gain, retention, and/or redistribution resulting in insulin resistance may influence blood lipid metabolism, but little is known about these associations postpartum. In our study, weight gain, increased central adiposity, and behavioral attributes were partly responsible for the long-term HDL cholesterol declines associated with a first birth, but the majority of the decline is not explained by these factors. A more complex interplay of changes in endocrine milieu, insulin resistance, or adipose tissue depots other than visceral fat may alter maternal fuel metabolism and homeostasis to adversely affect HDL cholesterol concentrations after a first birth, but, to our knowledge, these measures have not been examined in epidemiologic studies. Genetic factors, changes in fat distribution, and behavioral practices (e.g., lactation) that may offset the negative effects of a first birth on lipid profiles should be examined.
Acknowledgments
Supported by contracts N01-HC-48047, N01-HC-48048, N01-HC-48049, N01-HC-48050, and N01-HC-95095 from the National Heart, Lung, and Blood Institute and by Career Development Award grant 1 K01 DK59944-01A1 from the National Institute of Diabetes and Digestive and Kidney Diseases.
Abbreviations
- BMI
body mass index
- CARDIA
Coronary Artery Risk Development in Young Adults
- HDL
high density lipoprotein
- LDL
low density lipoprotein
References
- 1.Ness RB, Schotland HM, Flegal KM, et al. Reproductive history and coronary heart disease risk in women. Epidemiol Rev. 1994;16:298–314. doi: 10.1093/oxfordjournals.epirev.a036155. [DOI] [PubMed] [Google Scholar]
- 2.Humphries KH, Westendorp IC, Bots ML, et al. Parity and carotid artery atherosclerosis in elderly women: The Rotterdam Study. Stroke. 2001;32:2259–64. doi: 10.1161/hs1001.097224. [DOI] [PubMed] [Google Scholar]
- 3.Green A, Beral V, Moser K. Mortality in women in relation to their childbearing history. BMJ. 1988;297:391–5. doi: 10.1136/bmj.297.6645.391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Colditz GA, Willett WC, Stampfer MJ, et al. A prospective study of age at menarche, parity, age at first birth, and coronary heart disease in women. Am J Epidemiol. 1987;126:861–70. doi: 10.1093/oxfordjournals.aje.a114723. [DOI] [PubMed] [Google Scholar]
- 5.Rosenberg L, Palmer JR, Sowmya Rao R, et al. Risk factors for coronary heart disease in African American women. Am J Epidemiol. 1999;150:904–9. doi: 10.1093/oxfordjournals.aje.a010098. [DOI] [PubMed] [Google Scholar]
- 6.Steenland K, Lally C, Thun M. Parity and coronary heart disease among women in the American Cancer Society CPS II population. Epidemiology. 1996;7:641–3. doi: 10.1097/00001648-199611000-00014. [DOI] [PubMed] [Google Scholar]
- 7.Cooper GS, Ephross SA, Weinberg CR, et al. Menstrual and reproductive risk factors for ischemic heart disease. Epidemiology. 1999;10:255–9. [PubMed] [Google Scholar]
- 8.Lawlor DA, Emberson JR, Ebrahim S, et al. Is the association between parity and coronary heart disease due to biological effects of pregnancy or adverse lifestyle risk factors associated with child-rearing? Findings from the British Women's Heart and Health Study and the British Regional Heart Study. Circulation. 2003;107:1254–8. doi: 10.1161/01.cir.0000053441.43495.1a. [DOI] [PubMed] [Google Scholar]
- 9.Tulchinsky D, Hobel CJ, Yeager E, et al. Plasma estrone, estradiol, estriol, progesterone, and 17 hydroxyprogesterone in human pregnancy. I. Normal pregnancy. Am J Obstet Gynecol. 1972;112:1095–100. doi: 10.1016/0002-9378(72)90185-8. [DOI] [PubMed] [Google Scholar]
- 10.Desoye G, Schweditsch MO, Pfeiffer KP, et al. Correlation of hormones with lipid and lipoprotein levels during normal pregnancy and postpartum. J Clin Endocrinol Metab. 1987;64:704–12. doi: 10.1210/jcem-64-4-704. [DOI] [PubMed] [Google Scholar]
- 11.Ledoux F, Genest J, Nowaczynski W, et al. Plasma progesterone and aldosterone in pregnancy. Can Med Assoc J. 1975;112:943–7. [PMC free article] [PubMed] [Google Scholar]
- 12.Potter JA, Nestel PJ. The hyperlipidemia of pregnancy in normal and complicated pregnancies. Am J Obstet Gynecol. 1979;133:165–70. doi: 10.1016/0002-9378(79)90469-1. [DOI] [PubMed] [Google Scholar]
- 13.Jimenez DM, Pocovi M, Ramon-Cajal J, et al. Longitudinal study of plasma lipids and lipoprotein cholesterol in normal pregnancy and puerperium. Gynecol Obstet Invest. 1988;25:158–64. doi: 10.1159/000293765. [DOI] [PubMed] [Google Scholar]
- 14.Qureshi IA, Xi XR, Limbu YR, et al. Hyperlipidaemia during normal pregnancy, parturition and lactation. Ann Acad Med Singapore. 1999;28:217–21. [PubMed] [Google Scholar]
- 15.Piechota W, Staszewski A. Reference ranges of lipids and lipoproteins in pregnancy. Eur J Obstet Gynecol Reprod Biol. 1992;45:27–35. doi: 10.1016/0028-2243(92)90190-a. [DOI] [PubMed] [Google Scholar]
- 16.Miller VT. Dyslipoproteinemia in women. Endocrinol Metab Clin North Am. 1990;19:381–8. [PubMed] [Google Scholar]
- 17.Martin U, Davies C, Hayavi S, et al. Is normal pregnancy atherogenic? Clin Sci (Lond) 1999;96:421–5. doi: 10.1042/cs0960421. [DOI] [PubMed] [Google Scholar]
- 18.van Stiphout WA, Hofman A, de Bruijn AM. Serum lipids in young women before, during, and after pregnancy. Am J Epidemiol. 1987;126:922–8. doi: 10.1093/oxfordjournals.aje.a114729. [DOI] [PubMed] [Google Scholar]
- 19.Choi JW, Pai SH. Serum lipid concentrations change with serum alkaline phosphatase activity during pregnancy. Ann Clin Lab Sci. 2000;30:422–8. [PubMed] [Google Scholar]
- 20.Darmady JM, Postle AD. Lipid metabolism in pregnancy. Br J Obstet Gynecol. 1982;89:211–15. doi: 10.1111/j.1471-0528.1982.tb03616.x. [DOI] [PubMed] [Google Scholar]
- 21.Knopp RH, Bergelin RO, Wahl PW, et al. Population-based lipoprotein lipid reference values for pregnant women compared to nonpregnant women classified by sex hormone usage. Am J Obstet Gynecol. 1982;143:626–37. doi: 10.1016/0002-9378(82)90107-7. [DOI] [PubMed] [Google Scholar]
- 22.Flegal KM, Ness RB, Kramer RA. Parity and high density lipoprotein (HDL cholesterol) cholesterol levels in white women from the Second National Health and Nutrition Examination Survey (NHANES II) Am J Epidemiol. 1990;132:766. Abstract. [Google Scholar]
- 23.Cowan LD, Go OT, Howard BV, et al. Parity, postmenopausal estrogen use, and cardiovascular disease risk factors in American Indian women: The Strong Heart Study. J Womens Health. 1997;6:441–9. doi: 10.1089/jwh.1997.6.441. [DOI] [PubMed] [Google Scholar]
- 24.Kritz-Silverstein D, Barrett-Connor E, Wingard KL. The relationship between multiparity and lipoprotein levels in older women. J Clin Epidemiol. 1992;45:761–7. doi: 10.1016/0895-4356(92)90053-p. [DOI] [PubMed] [Google Scholar]
- 25.Ness RB, Cosmatos I, Flegal KM. Gravidity and serum lipids among Hispanic women in the Hispanic Health and Nutrition Examination Survey. J Womens Health. 1995;4:149–59. [Google Scholar]
- 26.Hubert HB, Eaker ED, Garrison RJ, et al. Life-style correlates of risk factor change in young adults: an eight-year study of coronary heart disease risk factors in the Framingham offspring. Am J Epidemiol. 1987;125:812–31. doi: 10.1093/oxfordjournals.aje.a114598. [DOI] [PubMed] [Google Scholar]
- 27.Haertel U, Heiss G, Filipiak B, et al. Cross-sectional and longitudinal associations between high density lipoprotein cholesterol and women's employment. Am J Epidemiol. 1992;135:68–78. doi: 10.1093/oxfordjournals.aje.a116203. [DOI] [PubMed] [Google Scholar]
- 28.Lewis CE, Funkhouser E, Raczynski JM, et al. Adverse effect of pregnancy on high density lipoprotein (HDL) cholesterol in young adult women: The CARDIA Study. Am J Epidemiol. 1996;144:247–54. doi: 10.1093/oxfordjournals.aje.a008919. [DOI] [PubMed] [Google Scholar]
- 29.Smith DE, Lewis CE, Caveny JL, et al. Longitudinal changes in adiposity associated with pregnancy. JAMA. 1994;271:1747–51. [PubMed] [Google Scholar]
- 30.Gunderson EP, Murtaugh M, Lewis CE, et al. Excess gains in weight and waist circumference associated with childbearing: the Coronary Artery Risk Development in Young Adults Study (CARDIA) Int J Obes. doi: 10.1038/sj.ijo.0802551. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cutter GR, Burke GL, Dyer AR, et al. Cardiovascular risk factors in young adults. The CARDIA baseline monograph. Control Clin Trials. 1991;2:1S–77S. doi: 10.1016/0197-2456(91)90002-4. [DOI] [PubMed] [Google Scholar]
- 32.Friedman GD, Cutter GR, Donahue RP, et al. CARDIA: study design, recruitment, and some characteristics of the examined subjects. J Clin Epidemiol. 1988;41:1105–16. doi: 10.1016/0895-4356(88)90080-7. [DOI] [PubMed] [Google Scholar]
- 33.Hughes GH, Cutter G, Donahue R, et al. Recruitment in the Coronary Artery Disease Risk Development in Young Adults (Cardia) Study. Control Clin Trials. 1987;8(4 suppl):68S–73S. doi: 10.1016/0197-2456(87)90008-0. [DOI] [PubMed] [Google Scholar]
- 34.Lewis CE, Jacobs DR, Jr, McCreath H, et al. Weight gain continues in the 1990s: 10-year trends in weight and overweight from the CARDIA study. Am J Epidemiol. 2000;151:1172–81. doi: 10.1093/oxfordjournals.aje.a010167. [DOI] [PubMed] [Google Scholar]
- 35.Bild DE, Jacobs DR, Liu K, et al. Seven-year trends in plasma low-density-lipoprotein-cholesterol in young adults: the CAR-DIA Study. Ann Epidemiol. 1996;6:235–45. doi: 10.1016/1047-2797(96)00005-1. [DOI] [PubMed] [Google Scholar]
- 36.Lewis CE, Smith DE, Wallace DD, et al. Seven-year trends in body weight and associations with lifestyle and behavioral characteristics in black and white young adults in CARDIA. Am J Public Health. 1997;87:635–42. doi: 10.2105/ajph.87.4.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Anderssen N, Jacobs DR, Jr, Sidney S, et al. Change and secular trends in physical activity patterns in young adults: a seven-year longitudinal follow-up in the Coronary Artery Risk Development in Young Adults Study (CARDIA) Am J Epidemiol. 1996;143:351–62. doi: 10.1093/oxfordjournals.aje.a008749. [DOI] [PubMed] [Google Scholar]
- 38.MacDonald A, Van Horn L, Slattery M, et al. The CARDIA dietary history. J Am Diet Assoc. 1991;91:1104–12. [PubMed] [Google Scholar]
- 39.Gordon T, Castelli WP, Hjortland MC, et al. High density lipoprotein as a protective factor against coronary heart disease. The Framingham Study. Am J Med. 1977;62:707–14. doi: 10.1016/0002-9343(77)90874-9. [DOI] [PubMed] [Google Scholar]
- 40.Kannel WB. High density lipoproteins: epidemiologic profile and risks of coronary disease. Am J Cardiol. 1983;52:9b–72b. doi: 10.1016/0002-9149(83)90649-5. [DOI] [PubMed] [Google Scholar]
- 41.Gotto AM. High-density lipoprotein cholesterol and triglycerides as therapeutic targets for preventing and treating coronary artery disease. Am Heart J. 2002;144:S33–42. doi: 10.1067/mhj.2002.130301. [DOI] [PubMed] [Google Scholar]
- 42.Wilson PW, Anderson KM, Castelli WP. Twelve-year incidence of coronary heart disease in middle-aged adults during the era of hypertensive therapy: The Framingham Offspring Study. Am J Med. 1991;90:11–16. doi: 10.1016/0002-9343(91)90500-w. [DOI] [PubMed] [Google Scholar]
- 43.Lewis CE, Wagenknecht L, Jacobs DR, et al. A balance of opposing forces: ten-year CHD risk changes in young adults. results from the CARDIA study. CVD Prev. 1998;1:271–81. [Google Scholar]
- 44.Greenlund KJ, Webber LS, Srinivasan S, et al. Associations of oral contraceptive use with serum lipids and lipoproteins in young women: the Bogalusa Heart Study. Ann Epidemiol. 1997;7:561–7. doi: 10.1016/s1047-2797(97)00119-1. [DOI] [PubMed] [Google Scholar]
- 45.Knopp RH, Walden CE, Wahl PW, et al. Effects of oral contraceptives on lipoprotein triglyceride and cholesterol: relationships to estrogen and progestin potency. Am J Obstet Gynecol. 1982;142:725–31. doi: 10.1016/s0002-9378(16)32478-4. [DOI] [PubMed] [Google Scholar]
- 46.Key TJA, Pike MC, Wang DY, et al. Long term effects of a first pregnancy on serum concentrations of dehydroepiandrosterone sulfate and dehydroepiandrosterone. J Clin Endocrinol Metab. 1990;70:1651–3. doi: 10.1210/jcem-70-6-1651. [DOI] [PubMed] [Google Scholar]
- 47.Musey VC, Collins DC, Brogan DR, et al. Long term effects of a first pregnancy on the hormonal environment: estrogens and androgens. J Clin Endocrinol Metab. 1987;64:111–18. doi: 10.1210/jcem-64-1-111. [DOI] [PubMed] [Google Scholar]
- 48.Musey VC, Collins DC, Brogan DR, et al. Long term effects of a first pregnancy on secretion of prolactin. N Engl J Med. 1987;316:229–34. doi: 10.1056/NEJM198701293160501. [DOI] [PubMed] [Google Scholar]
- 49.Parra A, Alarcon J, Gavino F, et al. Age-related changes in the metoclopramide-induced prolactin release in nulliparous women. Fertil Steril. 1993;60:34–9. doi: 10.1016/s0015-0282(16)56032-4. [DOI] [PubMed] [Google Scholar]


