Abstract
Clinical management of endometriosis is limited by the complex relationship between symptom severity, heterogeneous surgical presentations, and variability in clinical outcomes. As a complement to visual classification schemes, molecular profiles of disease activity may improve risk stratification to better inform treatment decisions and identify novel approaches to targeted treatment. Here, we employ a network analysis of information flow within and between inflammatory cells to discern consensus behaviors characterizing patient sub-populations. Unsupervised multivariate analysis of cytokine profiles quantified by multiplex immunoassays identified a subset of patients with a shared “consensus signature” of thirteen elevated cytokines that was associated with common clinical features, but was not observed among patient subpopulations defined by morphologic presentation alone. Enrichment analysis of consensus markers reinforced the primacy of peritoneal macrophage infiltration and activation, which was demonstrably elevated in ex vivo cultures. Although familiar targets of the NFκB family emerged among over-represented transcriptional binding sites for consensus markers, our analysis provides evidence for a previously unrecognized contribution from c-Jun, c-Fos, and AP-1 effectors of mitogen associated kinase signaling. Their crucial involvement in propagation of macrophage-driven inflammatory networks was confirmed via targeted inhibition of upstream kinases. Collectively, these analyses provide in vivo validation of a clinically relevant inflammatory network that may serve as an objective measure for guiding treatment decisions for endometriosis management, and in the future may provide a mechanistic endpoint for assessing efficacy of novel agents aimed at curtailing inflammatory mechanisms that drive disease progression.
Introduction
Endometriosis remains a widely prevalent gynepathology frequently associated with infertility and chronic pelvic pain affecting 6-10% of women of reproductive age (1). Multiple origins of ectopic endometrial glands and stroma have been postulated, but surgical evaluation of peritoneal lesion distribution in a majority of patients is consistent with implantation following retrograde menstruation (2,3). Emerging evidence suggests that molecular alterations disrupting normal hormone responsiveness may attenuate progesterone-directed differentiation in the secretory endometrium of affected women, conferring displaced tissue fragments with an ability to resist apoptosis, evade peritoneal immune surveillance, invade mesothelial surfaces, and rapidly acquire a vascular supply (4). Progressive accumulation of invasive implants is associated with a local sterile inflammatory response that further supports the growth of new and existing lesions, and contributes to molecular and ultrastructural dysregulation within the eutopic endometrium (5, 6).
Despite enormous efforts to dissect the well-documented clinical heterogeneity in endometriosis presentations, there remains a clear need to define objective measures of disease activity that are causally related to the severity of patient symptoms and variability in treatment responses, i.e. fertility preservation, pain reduction, and disease recurrence (7, 8). The most widely adopted pathological classification framework developed by the American Society for Reproductive Medicine (ASRM) utilizes a weighted scoring index to summarize the visual appearance, distribution, depth of invasion, and extent of endometriotic foci and adhesions observed at the time of diagnosis. However, the absence of a dose-response relationship between visual staging and symptomatic pain, infertility or post-operative outcomes has led many investigators to conclude that cell and molecular features may be more discriminating of complex symptomatology and poor clinical response than anatomic characteristics alone (9, 10, 11, 12). In contrast, the peritoneal inflammatory milieu has long been recognized as a relevant dimension of disease activity, as localized accumulation of immune cells and their effectors are thought to directly mediate or exacerbate the pathologic effects of ectopic lesions (13). Numerous cellular, protein, and metabolite markers of peritoneal inflammation have been associated with endometriosis in significant proportions of patients, and efforts are underway to incorporate these into noninvasive diagnostic tests (14, 15). Nonetheless, their relevance to disease management beyond empirical diagnosis remains unclear, due in part to a continued reliance upon morphologic sub-classification of patient populations (e.g. via ASRM staging criteria) to improve statistical discovery of novel associations.
An alternative approach to identify pathogenic mechanisms in the absence of unambiguous criteria for disease stratification is to exploit comprehensive multivariate measurements of molecular markers to empirically identify recurrent patterns conserved among women with heterogeneous clinical assessments of pain, infertility and recurrence. This multivariate paradigm is emerging as informative in other inflammatory disorders and is an attractive framework for building a robust molecular taxonomy of endometriosis to complement traditional clinical parameters. In contrast to strictly supervised univariate comparisons, unsupervised multivariate techniques use molecular co-variation as empirical evidence for biological co-regulation, and thereby improve the detection of coherent “signatures” of underlying physiological mechanisms, particularly when available class memberships are of a probabilistic nature or subject to observational error. In other words, when several molecules change in a coordinated manner in the same patient, and similar patterns emerge among multiple patients (even among patients who are otherwise clinically heterogeneous), this suggests they are part of a common cellular communication network that can be decoded using bioinformatics and subsequently validated with targeted interventions.
An attendant advantage to molecular stratification is an explicit emphasis towards informed therapeutic interventions that directly target disease-specific pathways. As first-line medical suppression of ovarian hormones ameliorates pain in a fraction of endometriosis patients and has no benefit on fertility outcomes, there is substantial interest to identify alternative targets that function independently of the endocrine axis and circumvent the morbid side effects that frequently limit long-term use of hormone modulators (3). Although empirical inhibition of canonical inflammatory mediators such as TNF-α, COX-2, and PPAR-γ reduces lesion burden in rodent and non-human primate models of endometriosis, results from observational and pre-clinical trials in humans modulating these mediators have been mixed (16, 17). Furthermore, the inability to accurately assess pain, infertility, and spontaneous recurrence in animal studies continues to confound their relevance to mitigating symptoms in women. Therefore, systematically prioritizing the role of canonical and novel inflammatory targets on the basis of human studies where direct relationships to clinical measures are unambiguously established is a prerequisite to developing novel and efficacious therapies.
In this study, we investigated the hypothesis that molecular profiles of peritoneal inflammation associated with endometriosis contain informative measures of symptom severity, and therefore provide a biological rationale for prioritizing specific therapeutic interventions. We therefore characterized native and cell secreted levels of fifty inflammatory cytokines, chemokines, and growth factors within peritoneal aspirates collected from women seeking surgical treatment for a variety of endometriosis-related symptoms and anatomic presentations, and subsequently asked whether patients within molecular sub-populations exhibited specific characteristics of pain and infertility. While a number of individual protein markers demonstrated significant variations with respect to conventional ASRM staging criteria, unsupervised stratification according to molecular profiles alone revealed a broader signature of pelvic inflammation that was strongly associated with invasive rectovaginal and ovarian lesions in conjunction with reduced patient gravidity. Biological enrichment analysis confirmed the primary contribution from recruited and activated macrophages, which actively secreted a majority of signature cytokines. In-depth investigation of global co-expression patterns similarly reinforced the role of over-represented transcriptional targets of macrophage-driven inflammation, and confirmed the predicted inhibitory potential of small molecule inhibitors interfering with c-Jun and NFκB regulation of cytokine production. Together, our integrated analysis provides a holistic framework for relating heterogeneous clinical presentations to molecular signatures, and potential new avenues of therapeutic intervention for a subpopulation of endometriosis patients.
Results
Cytokine Profiling of Peritoneal Aspirates
Demographic and clinical variables for the seventy-seven premenopausal women presenting with pain, infertility, and/or suspected endometriosis recruited for this study are summarized in Table I. Disease-free controls (n=20) were predominantly women seeking treatment for symptomatic leiomyomata and limited to patients not receiving any form of progestin-based therapy within three months of surgery. Patients with laparoscopically confirmed endometriosis were classified as ‘untreated’ (n=41) or ‘treated’ (n=16) according to a history of concurrent progestin-based hormonal therapy within three months of surgery. In addition to age, BMI, and cycle length, untreated cases and controls were selectively enrolled to obtain near equal proportions of follicular and luteal phase samples. The frequency of prior surgical interventions unrelated to endometriosis was likewise equivalent in all three groups.
Table I.
Patient and Peritoneal Aspirate Characteristics
| Characteristics | Controls | Endometriosis | ||
|---|---|---|---|---|
| Untreated | Treated | P-value | ||
| Number | 20 | 41 | 16 | - |
| Age1 | 41 (20-53) | 39 (16-51) | 34 (21-53) | 0.0510 |
| BMI1 | 25 (20-35) | 26 (19-41) | 28 (21-45) | 0.5329 |
| Gynecologic History | ||||
| Cycle length, days2 | 28 (21-30) | 29 (27-35) | - | 0.4253 |
| Cycle phase3 | 0.5856 | |||
| Follicular | 9 (45%) | 23 (56%) | - | |
| Luteal | 11 (55%) | 18 (44%) | - | |
| Unrelated abdominal surgeries3 | 8 (40%) | 15 (37%) | 6 (38%) | 0.9671 |
| Concurrent leiomyoma3 | 18 (90%) | 19 (46%) | 3 (19%) | <0.0001 |
| Reproductive History | ||||
| Gravidity4 | 1.8 (0-5) | 1.1 (0-4) | 0.9 (0-2) | <0.0001 |
| Parity4 | 1.0 (0-3) | 0.7 (0-4) | 0.5 (0-2) | 0.0002 |
| Infertility3 | 1 (5%) | 12 (32%) | 2 (15%) | 0.0374 |
| Pain Symptoms3 | ||||
| Dysmenorrhea | 4 (20%) | 27 (66%) | 10 (63%) | 0.0024 |
| Dyspareunia | 4 (20%) | 18 (50%) | 8 (57%) | 0.0454 |
| Pelvic pain | 5 (25%) | 22 (54%) | 4 (25%) | 0.0379 |
| Bowel | 5 (25%) | 16 (39%) | 6 (38%) | 0.5450 |
| Urinary | 5 (25%) | 9 (22%) | 2 (13%) | 0.6323 |
| Distribution 3 | ||||
| Peritoneum | - | 34 (83%) | 11 (69%) | 0.2865 |
| Ovaries | - | 15 (37%) | 3 (19%) | 0.2250 |
| Posterior cul-de-sac | - | 7 (17%) | 1 (6%) | 0.4195 |
| Adhesions | - | 15 (37%) | 3 (19%) | 0.2250 |
| ASRM Classification 3 | 0.5493 | |||
| Minimal/Mild | - | 23 (56%) | 11 (69%) | |
| Moderate/Severe | - | 18 (44%) | 5 (31%) | |
| Recurrence 3 | - | 20 (49%) | 8 (50%) | 1.0000 |
| Peritoneal Aspirates 1 | ||||
| Volume, mL | 7.9 (0.5-20) | 9.8 (0.1-35) | 5.4 (0.5-15) | 0.1353 |
| Total Protein, μg/mL | 55 (46-70) | 57 (38-81) | 56 (49-64) | 0.6689 |
| Leukocytes, 106/mL | 0.59 (0.15-1.6) | 1.0 (0.09-2.8) | 1.5 (0.44-4.2) | 0.0175 |
Data are mean (range) or proportion (percent).
Significance of non-equivalence by omnibus ANOVA.
Significance of inequality by unpaired t-test.
Significance of non-equivalent proportions by Fisher's exact test.
Significance of non-equivalent distributions by multinomial logistic regression.
Consistent with previous epidemiological findings a diagnosis of endometriosis was associated with significantly reduced gravidity and parity, as well as an increased frequency of concurrently diagnosed infertility (18). Severe dysmenorrhea or dyspareunia were reported in approximately two-thirds of enrolled cases independent of treatment status, whereas non-cyclic pelvic pain was more frequent among untreated women. In contrast, infertility was reported in a single control patient, and only one-quarter of control patients reported one or more severe pain symptoms. The physical distribution of lesions, revised ASRM staging scores, and reoperation rate were comparable between untreated and treated endometriosis cases. Although peritoneal fluid volume and total protein concentrations were similar in all patient groups, total leukocyte concentrations were significantly elevated in both untreated and treated endometriosis patients, in agreement with localized immune cell recruitment described in previous reports (19).
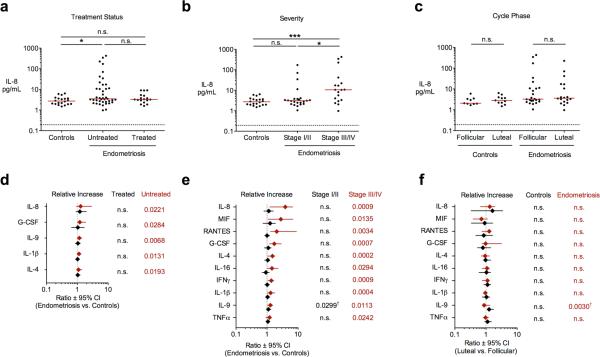
Soluble levels of fifty inflammatory cytokines, chemokines, and growth factors within corresponding peritoneal aspirates were determined via multiplex immunoassay as described in Materials and Methods. All assayed proteins were detected above background levels with the exception of IL-1-α, TNFβ, and IL-17, whose variation below ~1 pg/mL could not be quantified and were therefore excluded from subsequent analyses (Fig. S2). After correcting for multiple comparisons (FDR<0.05, Wilcoxon rank-sum test), ten of the remaining forty-seven cytokines exhibited statistically significant associations with endometriosis in univariate pair-wise contrasts between patient groups defined by treatment status or ASRM staging (Fig. 1; Tables S1-S2). Interleukin-8 displayed a pattern prototypical for elevated cytokines, with highly variable levels among untreated women, and intermediate levels among patients receiving progestin-based therapy. Stratification of untreated patient samples according to ASRM staging confirmed a statistical trend towards greater IL-8 abundance in cases classified as moderate/severe, but increased levels were invariant with respect to cycle phase. G-CSF, IL-9, IL-1β, and IL-4 were similarly elevated among untreated women, while significant increases in MIF, RANTES, IL-16, IFNγ, and TNFα were limited to patients with Stage III/IV disease. Among these, no cytokines demonstrated statistical elevation among Stage I/II samples or cycle-dependent variation after correcting for multiple comparisons (Table S3). Additional sub-group analyses revealed no significant associations between patient subpopulations defined according to symptom recurrence (primary versus secondary surgical intervention), lesion distribution (peritoneal, ovarian, or deep disease), and primary indication (pain versus infertility) (Tables S4-S6). A majority of our findings confirm associations reported in independent cross-sectional studies utilizing morphological staging of disease severity, with the frequency of replication roughly corresponding to the observed effect size (Table S7). Collectively, these profiles underscore the relationship between patient morbidity, local leukocyte accumulation, and variable elevation of secreted inflammatory factors associated with pelvic endometriosis.
Figure 1. Differentially regulated peritoneal cytokines according to clinical diagnosis.
Interleukin-8 concentrations among control and endometriosis patients grouped according to (A) hormonal treatment status, (B) ASRM staging, and (C) menstrual cycle phase. Median concentrations (solid lines), limits of detection (dashed lines), and unadjusted significance levels are indicated. (D-F) Forest plots summarizing significant cytokine associations (FDR<0.05, Wilcoxon rank-sum test) between comparison groups according to treatment status, severity, and cycle phase as in (A-C). Ratio between population medians (symbols) and 95% confidence intervals (bars) are shown for each analyte together with unadjusted P-values from corresponding pair wise comparisons. n.s., not significant; * P<0.05; ** P<0.01; *** P<0.001; † Not significant after correction for multiple comparisons.
Unsupervised Multivariate Analysis of Endometriosis Patient Cytokine Profiles
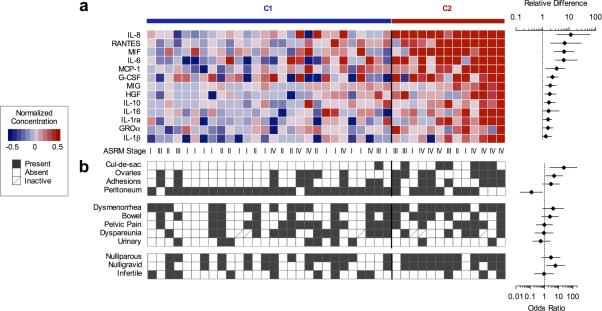
In order to circumvent the limitations associated with the visual staging of endometriosis and simultaneously focus our analysis on those cytokine markers exhibiting the greatest evidence for co-regulated expression, we pursued a fully unsupervised approach that leveraged the multivariate structure of aspirate profiles across our diverse patient population. To this end, we first applied a consensus-based strategy to naïvely stratify normalized cytokine profiles from untreated endometriosis patients into self-similar subpopulations across an ensemble of unsupervised solutions derived from non-negative matrix factorization (NMF) (20). NMF identified an optimal subdivision of two patient sub-populations over higher levels of stratification, which was statistically significant not only with respect to marker permutation, but also remained optimal across reduced data sets constructed by sequentially eliminating makers on the basis of total variation (Fig S3). Approximately two-thirds (n=28) of endometriosis samples were assigned to a class indistinguishable from control samples (C1), with the second class (C2) comprising samples exhibiting four or more markedly elevated markers. Those cytokines differentially expressed (FDR<0.05, Wilcoxon rank-sum test) between C1 and C2 subpopulations included thirteen analytes (IL-8, RANTES, MIF, IL-6, MCP-1, G-CSF, MIG, HGF, IL-10, IL-16, IL-1ra, GROα, and IL-1β), exhibiting strong co-variation across all 41 samples. (Fig. 2a). This collection of markers – hereafter referred to as a “consensus” cytokine signature – thus represent robust molecular features defining the spectrum of inflammatory activation in a substantial fraction of endometriosis patients in this study. Among those thirteen “consensus” cytokines identified in multivariate analysis, six cytokines were also statistically related with patient ASRM scores in univariate tests of association (c.f. Fig. 1e and Table S2). Those six cytokines jointly declared significant in both analyses simultaneously fulfill the requirements for strong co-variation as well as correlation with morphologic presentation. Conversely, incomplete correspondence between the remaining univariate and multivariate associations reflects divergence in the underlying data, with four univariate associations (IL-4, IL-9, IFNγ, and TNFα) exhibiting marginal covariation with the regulatory axis captured by the consensus signature.
Figure 2. Unsupervised classification of molecular subpopulations among untreated endometriosis patients.
(A) Individual patient profiles optimally stratify into two subpopulations by non-negative matrix factorization of all 47 target cytokines. Thirteen differentially expressed cytokines between patient classes C1 and C2 (FDR<0.05, Wilcoxon rank-sum test) are shown with forest plots indicating mean difference ± 95% confidence intervals. (B) Univariate associations between molecular subpopulations and clinical metrics are indicated with corresponding odds ratios ± 95% confidence intervals (Fisher's exact test).
Subsequent comparison of patient characteristics on the basis of molecular class assignment revealed negligible differences in age, BMI, cycle phase, cycle length, or symptomatic disease recurrence between endometriosis subgroups (data not shown). However, median and total variation in aspirate volume was markedly reduced among the elevated cytokine patient subgroup (11.5 mL, 3.5-18.8 mL IQR vs. 3.0 mL, 0.7-6.8 mL IQR; P=0.0091 Wilcoxon rank sum test) despite equivalent concentrations of total protein and leukocytes (Fig. S4). As normal peritoneal fluid exudation by the ovaries and serous membranes is compromised in patients with substantial lesion burden (21), elevated concentrations in these low volume samples may reflect a reduced capacity for homeostatic cytokine resorption and clearance afforded by a larger fluid interface with surrounding tissues. Consistent with this interpretation, elevated cytokines and reduced fluid volume were more frequently associated with lesions affecting the rectovaginal cul-de-sac (OR=23.14, P=0.0022, Fisher's exact test) and ovaries (OR=4.8, P=0.0376, Fisher's exact test). A similar degree of association with pelvic adhesions did not reach significance; however, elevated cytokines were less likely to be found among women presenting exclusively with peritoneal lesions versus those with multifocal implants (OR=0.1231, P=0.0239, Fisher's exact test).
Although symptomatic pain was reported more frequently among endometriosis patients versus controls, a proportional association between elevated consensus markers and severe pain did not reach significance, regardless of site or context (Fig. 2b). These data reinforce previous findings demonstrating partial correspondence between elevated cytokine levels and severity of pain (22, 23). Increased consensus cytokine concentrations were however, observed more frequently among nulligravid women (OR = 6.0, P = 0.0203, Fisher's exact test), and were more discriminating of patient gravidity than ASRM staging alone (Moderate/Severe vs. Minimal/Mild OR = 2.444, P = 0.2146, Fisher's exact test). This decrease could not be explained by a prior diagnosis of infertility, as the proportion of clinically-diagnosed infertile patients was equivalent in both subpopulations. Parity was likewise equivalent between patient groups, suggesting that endometriosis associated inflammation may markedly impair successful conception, but has a more limited influence on successful gestation.
Enrichment Analysis of Consensus Cytokine Markers
Having established a consensus signature of inflammatory factors distinguishing severe presentations of endometriosis associated with reduced fertility, we next investigated cellular mediators of this signature via systematic enrichment analysis. Although comprehensive secretomic profiles for representative peritoneal cell populations are currently unavailable, we reasoned that variations in cytokine transcript abundance might sufficiently capture relative propensities for protein production and secretion (24). To this end, we mined the Immune Response In Silico (IRIS) transcriptional compendia (25) using Gene Set Enrichment Analysis (GSEA) to identify canonical leukocyte phenotypes enriched for expression of consensus cytokines or their cognate receptors as relevant secreting and responding populations, respectively. In contrast to similar compendia of resting immune cell populations (e.g. ImmGen, DMAP) or cell lines established from hematological malignancies (e.g. Lymphochip), the IRIS compendia specifically incorporates profiles from diverse leukocyte populations under both resting and stimulated experimental conditions, where stimulants include a diverse assortment of model antigens, polarizing cytokines, and growth factors with canonical roles in both acute and chronic inflammation. This breadth of inflammatory phenotypes thereby strengthens our ability to make accurate inferences regarding lineage-specific contributions in endometriosis-associated inflammation.
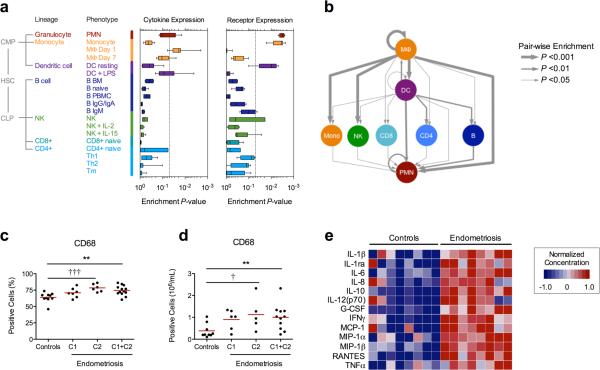
As anticipated, differentiated myeloid lineages (i.e. macrophages, dendritic cells, and neutrophils) demonstrated the greatest positive enrichment for consensus cytokine transcripts, while undifferentiated monocytes, dendritic cells, and neutrophils represented dominant responding populations (Fig. 3a). Extending this approach to identify specific routes of intercellular communication in which consensus cytokine-receptor interactions were overrepresented, we computed pair-wise enrichment scores between each major lineage in the IRIS compendia to examine the overall topology of the cytokine signaling network (Fig. 3b). While this later analysis excludes contributions from peritoneal tissues or ectopic lesions, the inferred hierarchical structure reinforces a model of macrophage activation as the locus of cytokine production and cell recruitment among the peritoneal immune cell populations.
Figure 3. Enrichment analysis of the consensus cytokine signature emphasizes a macrophage-directed inflammatory phenotype.
(A) Significance of lineage-specific enrichment scores for consensus cytokine expression obtained by GSEA among Immune Response in silico (IRIS) transcriptional profiles. (B) Hierarchy of enriched intercellular cytokine-receptor interactions. Flow cytometric confirmation of increased (C) absolute and (D) relative CD68+ macrophage abundance among endometriosis patient populations. Increasing CD68+ counts among patient subpopulations C1 and C2 are shown for comparison. **P<0.01 Wilcoxon rank-sum test; †P<0.05, †††P<0.001, linear trend test. (E) Normalized concentration of secreted cytokine in media conditioned from isolated peritoneal macrophages. Shown are 13 differentially secreted cytokines (FDR<0.05 Wilcoxon rank-sum test, two-fold increase) from eight control and eight untreated donors.
To explicitly confirm that activated macrophages were a primary source of consensus cytokines in our study population, we subsequently quantified the concentration, proportion, and secretion status of CD68+ cells in a subset of aspirate samples from control and endometriosis patients (Fig. 3c-e). In addition to an overall increase in macrophage concentration and proportion among all patients, an increasing trend in both measures was observed when samples were grouped according to C1/C2 membership (linear trend test, P = 0.0148, and P = 0.0007, respectively) indicating further concordance between macrophage accumulation and increased cytokine abundance in peritoneal fluid. When cultured ex vivo, macrophages isolated from endometriosis patients spontaneously produced a marked abundance of thirteen cytokines relative to controls (Wilcoxon rank sum test P<0.05; >2-fold increase) (Figs. 3e, S5). These included eight of the thirteen consensus markers (IL-1β, IL-1ra, IL-6, IL-8, IL-10, G-CSF, MCP-1, RANTES), demonstrating that locally recruited macrophages are a significant if not primary source of elevated cytokines observed within native peritoneal fluid.
Genome-wide Inference of a Macrophage Co-expression Network in Endometriosis
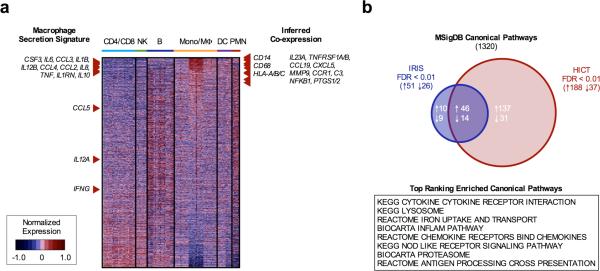
While reference compendia comprised of exogenously stimulated leukocyte populations such as IRIS provide a limited approximation to peritoneal immune cell activation in vivo, we hypothesized that global co-expression patterns therein might be sufficiently conserved to accurately infer the relevance of genes beyond those fifty cytokine markers assayed in peritoneal fluid and conditioned media specimens. To further explore candidate co-expression partners relevant to the macrophage inflammatory response in endometriosis, we reordered transcripts in the IRIS compendia according to their median correlation distance to the thirteen cytokines differentially secreted by isolated macrophages in vitro (Fig. 4a). As anticipated, the macrophage secretion signature collectively ranked very high within this ordering, along with canonical macrophage surface markers such as CD14, CD68, and HLA-A/B/C. Moreover, numerous gene products previously associated with endometriosis in independent studies were observed within the top ten percent of genes in the co-expression profile, including IL-23 (26), soluble TNF receptors I & II (27), MIP-3β/CCL19 (28), ENA-78/CXCL5 (29), MMP-9 (30), CCR1 (31), C3 (32), NFκB (33), and Cox-1/2 (34). To verify that these inferences were not unique to the expression structure present in IRIS, we examined a second co-expression profile derived from the Human Immune Cell Transcriptome (HICT) compendia (35), which demonstrated broad correspondence with IRIS profile rankings (P < 10−9, Spearman correlation) and comparable enrichment of the independently validated inflammatory markers noted above among the top ten percent of proximal co-expression partners.
Figure 4. Inferred transcriptional co-expression profiles and pathway enrichment of peritoneal macrophage secretion signature.
(A) Transcripts from the IRIS compendia rank-ordered according to median correlation distance to cytokines differentially secreted by isolated peritoneal macrophages (left). Inferred co-expression of macrophage surface markers and reported disease markers among the top 10% of proximal transcripts (right). (B) Conservation of highly enriched Canonical Pathway gene sets between IRIS and HICT co-expression profiles (FDR<0.01) following gene set enrichment analysis. The top eight positively enriched gene sets among the shared subset are listed.
Extending this approach to pathway-level analysis, we next applied GSEA to discern biological processes most strongly enriched among the inferred IRIS and HICT co-expression profiles. From 1320 annotated gene sets represented in the Canonical Pathways collection of MSigDB v4.0, we identified 60 up- and down-regulated gene sets coordinately enriched in both profiles (FDR<0.01) (Fig. 4b). In addition to nominal annotations corresponding to cytokine/receptor gene families, the top-scoring members of this subset included well-known processes associated with classical macrophage activation (e.g. nod-like receptor signaling, antigen cross-presentation) and scavenger function (e.g. proteosome, lysosome, iron uptake pathways). Thus, reconstructing the co-expression network most closely related to the macrophage secretory signature not only implicates cellular processes relevant to endometriosis, but simultaneously prioritizes their constituent gene products for further investigation as potential therapeutic targets.
Validation of Predicted Transcriptional Regulators in Activated Peritoneal Macrophages
Provided the ability to associate the observed macrophage secretory signature with defined molecular and cellular processes, we narrowed our analysis to exclusively consider individual transcriptional regulatory factors that contribute to the sustained production of inflammatory cytokines represented in the macrophage secretion signature. We first examined over-representation of transcriptional regulatory binding motifs among the measured cytokine signature, and subsequently confirmed positive enrichment among inferred IRIS and HICT profiles. While we expect the former analysis to have modest statistical power given the limited number of protein measurements, the use of the genome-wide profiles provides a more robust assessment of over-representation under the assumption that changes in transcript levels primarily reflect the activity of specific regulators. From 244 annotated binding sequences within the promoter regions of the fifty assayed cytokines, 62 binding motifs corresponding to ten transcription factors - C/EBPb, RelA, CREB-1, NFκB:RelA, ATF-2, NFκB, c-Fos:c-Jun, c-Ets-1, AP-1, and STAT1 - were significantly over-represented (P<0.05, Fisher's exact test) among the thirteen differentially secreted cytokines (Table S6; cf. Fig 3e). Gene sets sharing the specific binding motifs for RelA, NFκB, c-Ets-1, and AP-1 were likewise enriched among the inferred IRIS and HICT co-expression profiles (Table II), adding additional evidence to their role in mediating the observed secretory signature.
Table II.
Overrepresentation of Transcriptional Binding Sites Among Macrophage Secreted Cytokines
| Transcription Factor | All Motifs1 | Consensus Sequence | Co-Expression2 | |
|---|---|---|---|---|
| IRIS | HICT | |||
| RelA-p65 | 0.0026 | V$NFKAPPAB65_01 | 0.0021 | <0.0001 |
| V$NFKAPPAB_01 | <0.0001 | <0.0001 | ||
| V$NFKB_Q6_01 | <0.0001 | <0.0001 | ||
| NF-kappaB1-p50 | 0.0198 | V$NFKB_Q6 | <0.0001 | <0.0001 |
| V$NFKB_C | 0.0022 | <0.0001 | ||
| c-Ets-1 | 0.0286 | V$ETS_Q4 | 0.0035 | 0.0131 |
| AP-1 | 0.0417 | V$AP1_01 | <0.0001 | <0.0001 |
| V$AP1_Q4_01 | <0.0001 | <0.0001 | ||
Fisher's exact test P-value for all motifs distinguishing differentially secreted cytokines.
Gene Set Enrichment Analysis P-value for individual motifs positively enriched among IRIS and HICT co-expression profiles.
Although the implied role for RelA and NFκB activity in macrophage cytokine secretion was largely unsurprising, the similar level of evidence supporting overrepresentation of binding motifs involving c-Jun (i.e. AP-1 and ATF-2) was less anticipated and suggested a novel avenue to limit the secretory activity of peritoneal macrophages. To directly test the predicted efficacy in inhibiting transcriptional activity of c-Jun and its AP-1 binding partner c-Fos, we evaluated the aggregate inhibition of the macrophage secretion signature in the presence of small molecule inhibitors targeting JNK, MEK1, and IKK (Fig. 5 a-b). We simultaneously evaluated the inhibitory effects upon four unrelated targets, whose downstream transcriptional regulators demonstrated partial (p38 and Jak) or negligible (Src, PKC, PI3K) enrichment in our analysis of promoter binding motifs.
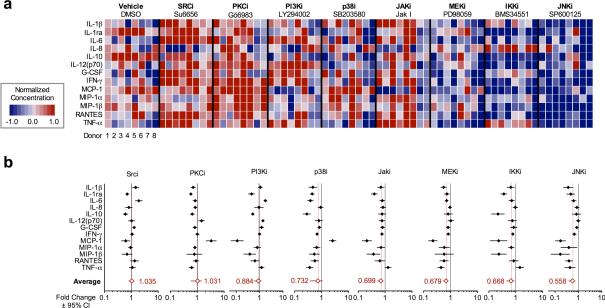
Figure 5. Targeted inhibition of enriched transcriptional regulators maximally inhibits macrophage cytokine secretion.
(A) Secretion profiles of isolated peritoneal macrophages from eight endometriosis donors following targeted inhibition of select transcriptional regulators. (B) Individual and aggregate inhibitory profiles (relative to vehicle controls) across eight donor samples in (A). Symbols indicate mean difference ± 95% confidence intervals in cytokine concentrations relative to vehicle controls.
Surprisingly, JNK and MEK1 inhibition mediated a comprehensive reduction in cytokine secretion on par with targeting IKK, (56% and 68% vs. 67% average secretion relative to vehicle, respectively) with no activating effects. In contrast, inhibitors targeting Jak1 and p38 demonstrated variably reduced secretion of some, but not all thirteen signature cytokines, and in the case of p38 markedly potentiated the production of MCP-1. Inhibitors targeting Src, PCK, and PI3K likewise mediated a combination of inhibiting and potentiating secretion responses, with the aggregate effect proving modest to negligible (88-104% average secretion). Thus, the predicted role for transcriptional regulation of macrophage cytokine secretion extends beyond processes contributing to canonical NFκB signaling, highlighting a novel route to blunting the inflammatory response for therapeutic effect.
Discussion
Improving clinical stratification of women with surgically documented endometriosis on the basis of disease mechanisms remains a major hurdle to understanding and treating this heterogeneous disorder. Basic and clinical research efforts to develop evidence-based recommendations to improve clinical management of endometriosis presently confront simultaneous challenges associated with (i) sampling representative patient populations with divergent manifestations of endometriosis, (ii) characterization of patient specimens with partially or non-standardized anatomic, pathological, and biochemical methods, and (iii) logistical considerations precluding prospective longitudinal studies to chronicle the natural course of disease. While daunting, these challenges are not unique to endometriosis, and hence there is enormous potential to translate insights from other clinical investigations into the strengths and limits of molecular stratification as a complement to conventional clinical parameters.
The absolute degree to which common pathogenic mechanisms are shared among women with endometriosis - and therefore justify targeted therapeutic intervention in well-defined patient subpopulations - is currently unknown. Beyond the dynamic changes that accompany implantation, growth, and fibrosis of individual lesions, a significant proportion of patients harbor lesions of varying maturity, indicating that the local and systemic consequences of lesion progression reflect a mosaic of pathologic processes. Results from genome-wide and focused association studies of recurrent somatic variants further support the hypothesis that endometriosis is polygenic in nature and likely results from a combination of genetic susceptibility and environmental interactions (36, 37, 38). Thus, as in other complex inflammatory disorders such as asthma, inflammatory bowel disease, and rheumatoid arthritis, substantial intra- and interindividual heterogeneity poses a severe challenge to the prognostic utility of low-dimensional indices summarizing clinicopathological features. These considerations strongly motivate empirical data-driven studies to identify multivariate features that provide optimal discrimination and robust generalization among larger populations. The rapid expansion of genomic annotations derived from complementary experimental profiles likewise facilitates a rich interpretation of multivariate signatures via enrichment and integrated network inference methodologies (39, 40). Together, tackling the intrinsic heterogeneity of complex disease processes at a cell and molecular level offers a tractable framework for building principled hypotheses and avoiding ad hoc biological interpretations.
In an effort to address these challenges, we identified a conserved set of inflammatory cytokine markers that maximally differentiated nascent and complex presentations of endometriosis, the latter associated with reduced patient fertility. In contrast to previous studies relying exclusively on morphologic criteria, this “consensus signature” of peritoneal inflammation derives from an unsupervised multivariate stratification of patient specimens according to shared molecular patterns of dysregulation within the peritoneal compartment. We subsequently applied an integrative enrichment analysis leveraging annotated transcriptional profiles to identify cellular mediators that were responsible for this observed signature, and validated activated macrophage populations as a dominant source of these secreted factors. Finally, reconstruction of a proximal co-expression network around macrophage secreted cytokines implicated a well-defined subset of transcriptional binding sequences and associated regulatory factors (including c-Jun and NFκB), whose predicted activity was directly confirmed via pharmacologic inhibition. While these results warrant further investigation, they provide additional proof of principle for the utility of cytokine profiling as a means to broadly interrogate the immune cell-mediated response to complex pathologies, including endometriosis. Pending further validation among independent patient populations, the specific signatures described herein may also have direct clinical application for improving diagnostic classification and prospectively identifying women with a high likelihood to benefit from post-operative immune modulation in the course of pain remediation or fertility preservation (8).
By adopting an unsupervised multivariate analysis, we explicitly accommodated the strong pattern of covariation between cytokines observed across endometriosis patients. Parallel enrichment and co-expression analyses using the univariate cytokine signature distinguishing ASRM III/IV patients from controls (c.f. Fig. 1b) failed to predict significant macrophage enrichment using GSEA, despite significant trends towards increased macrophage prevalence and abundance in advanced stage patients (Fig. S7). Furthermore, inferred co-expression profiles derived from the ASRM III/IV associations were markedly dissimilar to those derived using the consensus signature, and resulted in far less coherent enrichment of related cellular pathways (Fig. S8). Collectively, these disjointed inferences highlight the distinctive advantage in deriving a molecular signature in a multivariate fashion which retains co-varying markers that likely represent a common underlying regulatory program.
A critical finding from our unsupervised analysis is that marked variation in peritoneal fluid cytokines concentrations is attributable to a combination of reduced peritoneal fluid production and retention, together with increased macrophage abundance. Whether local accumulation of macrophages and their inflammatory markers represents a cause or consequence of reduced fluid levels is unclear, but our preliminary findings that both occur in subsets of patients with minimal or advanced disease suggests an underlying phenomenon that may directly correspond to the severity of pain and infertiltiy symptoms. A related finding with implications for diagnostic applications was the statistically significant, but modest absolute deviation in a number of canonical inflammatory markers between patient subpopulations, i.e. TNF-α, IL-1β, IFN-β. Although central in importance to immune cell function and differentiation, the limited overall variation between patient populations suggests that such tightly regulated markers may prove sub-optimal for empirical patient classification alone. More strategic choices for molecular stratification will include downstream effectors such as chemokines (e.g. IL-8, RANTES, and MCP-1) that exhibit substantial dynamic range within the peritoneal cavity.
We were unable to identify additional associations distinguishing patient subpopulations stratified according to duration of symptoms (initial versus secondary treatment), lesion distribution (peritoneal, ovarian, or deep disease), or surgical indication (pain versus infertility). While this may be a consequence of small or unbalanced samples across distinct disease sub-groups, the absence of additional molecular sub-populations may also reflect the high degree of overlapping disease characteristics that were present in a majority of cases. Future observational studies designed to selectively characterize patient sub-populations with truly homogeneous disease characteristics will be of great value to address whether such sub-populations represent wholly distinct pathological entities.
To circumvent ad hoc biological interpretation of the observed molecular associations with endometriosis, we performed systematic enrichment analyses that leverage annotated genome-scale compendia to integrate the relative degrees of evidence from individual protein markers. Although this methodology relies upon transcriptional variation as a proxy for protein level expression, the general framework may serve as a useful tool for protein-based profiling studies until proteomic compendia become available. We extended this approach to examine transcriptional co-expression patterns among two representative collections of resting and activated immune populations to infer (i) probable members of the inflammatory milieu not characterized in our immunoassay panel and (ii) candidate regulators of the inflammatory signature as potential avenues for therapeutic intervention. One insight from this analysis that has not been previously appreciated – JNK/c-Jun transcriptional activity – was validated as a potent regulatory element contributing to peritoneal macrophage cytokine production. Intriguingly, this insight resonates with recent animal studies demonstrating the potent efficacy of JNK inhibition of cytokine production in a murine xenograft model of endometriosis that accompanied a marked reduction in lesion burden (41, 42). We have also linked JNK-mediated signaling as an integral compensatory pathway regulating proteolytic cascades governing human endometriotic cell motility (40). Future studies will examine this pathway in greater detail to clarify the mechanistic origins of upstream activation, and evaluate complementary inhibitory agents as a therapeutic strategy to suppress peritoneal inflammation.
The origins of macrophage-driven peritoneal inflammation in endometriosis have been variably ascribed to autoantibody deposition (43), secretory products of ectopic endometrial implants (44), iron overload (33), scavenger receptor ligation (45), bacterial endotoxin (46), and altered sensitivity to ovarian steroids (4). A plausible hypothesis is that in the context of endometriosis, a multiplicity of signals collectively contributes to a distinct macrophage activation phenotype. Intriguingly, the observed secretion signature in our study exhibits characteristics of both prototypical classes of macrophage polarization, i.e. marked IL-1β and IL-10 secretion (47). Such redundancy in upstream activation signals emphasizes the need to target regulatory nodes that control a majority of downstream effectors with minimal compensatory modes to activation. Enrichment analysis of peritoneal aspirates did not support a distinct polarization phenotype among CD4+ lymphocyte sub-populations (cf. Fig. 3a). Conflicting evidence for Th1, Th2, and Th17 polarization in endometriosis have been reported (Table S7), but a clear consensus as to whether the unambiguous activity of innate leukocyte populations induces strongly polarized lymphocytes has yet to emerge.
Although our results implicate a clear role of macrophage-directed inflammation in both the progressive and debilitating characteristics of endometriosis, the generality of our findings may be limited by selection biases towards certain demographic and/or clinical characteristics that were unique to our patient population. In particular, these findings may be restricted to similar cohorts of predominantly older women pursuing treatment for established symptomatic endometriosis. Nonetheless, our data support the theory that pathogenic signatures of disease activity are present at diagnosis in and that molecular profiling has the potential to improve outcome prediction across diverse endometriosis populations. Future studies from independent patient populations to replicate the consensus signature described herein or to define other signatures by similar analysis of different anatomical locations of disease impact may ultimately reveal a set of informative biological measurements that improve the efficacy of disease management.
Materials and Methods
Study Design
Sample size requirements were estimated by meta-analysis of twenty published reports describing significant molecular alterations in the peritoneal fluid of women surgically diagnosed with endometriosis (Supplementary Materials and Methods; Fig. S1). Peritoneal fluid was obtained from seventy-seven women undergoing laparoscopic surgery for non-malignant gynecologic indications (endometriosis, adenomyosis, leiomyomata, menorrhagia, primary infertility, pelvic pain, hysterectomy, and/or elective sterilization). All participants provided informed consent in accordance with a protocol approved by the Partners Human Research Committee and the Massachusetts Institute of Technology Committee on the Use of Humans as Experimental Subjects. Study enrollment was limited to pre-menopausal women with regular cycles (26-35 days) and excluded patients with an irregular or ambiguous cycle history. In addition to electronic medical records documenting each patient's original consultation, pre-operative visit, pathology report, and post-operative visit, all clinical data was documented using a standardized questionnaire. Abstracted clinical data were examined prior to laboratory analysis to establish each patient's eligibility and exclude patients with any concomitant indications of inflammatory disease (e.g. autoimmune disorders, malignancy, appendicitis, etc.). Inclusion of patients who reported use of progestin-based hormonal therapy within three months of surgery was limited to those with a concurrent diagnosis of endometriosis, in order to evaluate the role of hormone suppression on inflammatory markers. Severity of endometriosis was visually scored according to the revised criteria of the American Society for Reproductive Medicine (ASRM) (48) and classified as Minimal/Mild (Stage I/II) or Moderate/Severe (Stage III/IV) disease. Endometriosis was absent in twenty patients, whose samples served as disease-free controls. In all cases, atypical lesions were evaluated by histopathology to confirm the presence of endometriotic glands and stroma. All specimens were classified as luteal (day 1-14) or follicular (day 15-28) according to the donor's menstrual history. Severity of pain was characterized as Absent/Mild or Moderate/Severe.
Sample Collection and Processing
Peritoneal fluid was aspirated from the rectovaginal cul-de-sac immediately following trocar insertion and prior to peritoneal lavage or surgical manipulation. Specimens were placed immediately on ice, the volume and color recorded, and 50 μL of primary aspirate aliquoted for cytological characterization. The remaining volume was clarified within 15 min. of collection by centrifugation for 10 min. at 1000 rcf. Clarified aspirates were transported to the laboratory on wet ice, aliquoted, and stored at −80°C until batch analysis. Peritoneal cell pellets were resuspended in phenol red free DMEM/F12 supplemented with 10% v/v dextran-charcoal stripped FBS, penicillin/streptomycin/amphotercin and 10% v/v DMSO prior to cryopreservation. Leukocyte and erythrocyte content of the primary aspirate were determined by hemocytometer count. Total protein concentration was determined by bicinchoninic acid assay (Thermo Scientific Pierce, Rockford, IL) to identify and exclude saline-contaminated samples.
Multiplex Cytokine Immunoassays
Determination of fifty peritoneal cytokines, chemokines, and growth factors was performed using a Group I 27-plex, Group II 21-plex, and ICAM-1 and VCAM-1 singleplex immunoassay panels according to manufacturer recommendations (Bio-Rad Laboratories, Hercules, CA). Prepared arrays were read using a Bio-Plex 200 system and data collected using Bio-Plex Manager Software. For VCAM-1 and ICAM-1, peritoneal aspirates were diluted 50-fold to bring the final analyte concentration within the linear detection range of the assay. All Group I and II analytes were evaluated in neat, undiluted samples and assays performed in parallel (unmixed) to avoid antibody cross-reactivity between Groups. Assay performance characteristics for each analyte are summarized in Table S9. Median fluorescent intensity values were converted to absolute concentrations via calibration to a nine-point standard series.
Flow Cytometry and Cell Culture
Macrophage prevalence in peritoneal aspirates was assessed by flow cytometric quantitation of CD45+/CD68+ leukocytes. For cytokine secretion assays, cryopreserved peritoneal leukocytes were rejuvenated in complete media to recover mature adherent macrophages. Following two hours of selective adhesion, non-adherent lymphocyte populations were removed by repeated washing, and fresh 1% serum-reduced media replaced for a twenty-four hour incubation period. Cytometric evaluation confirmed 90-95% CD68+ cells among cultures prepared by selective adherence (Table S10). Adherent CD68+ macrophages represented >85% of the total CD68+ pool present in peritoneal aspirates from both controls and endometriosis patients, and were shown to be the principal leukocyte population responsible for production of differentially secreted cytokines identified in culture media (Fig. S5). To ensure that macrophages isolated from a select subset of samples were representative of the diverse peritoneal cytokine profiles while avoiding potential bias towards samples with low or high aspirate concentrations, cases for in vitro macrophage experiments (Figs. 3 and 5) were sampled from the 41 donors with equivalent proportional representation from the corresponding C1/C2 aspirate profiles (i.e. a 5:3 ratio of C1:C2). For inhibitor experiments, vehicle (DMSO) or the indicated inhibitors were added to conditioning media in replicate cultures to obtain a final concentration listed in Table S11.
Data Processing and Statistical Analysis
Patient demographics and sample characteristics were evaluated by omnibus ANOVA, unpaired t-test, Fisher's exact test, or multinomial logistic regression as indicated in Table I. Pair-wise comparison of cytokine levels between populations stratified according to treatment status, ASRM staging, and cycle phase were evaluated by Wilcoxon rank sum tests, and 95% confidence intervals on test statistics constructed via bootstrap. Significant associations were declared for cytokine associations achieving a two-sided P-value less than 0.05 following a Benjamini-Hochberg multiple testing correction. For multivariate analysis and heatmap visualization, cytokine concentrations were log-transformed and median centered. Unsupervised classification of cytokine profiles into consensus clusters was carried out using non-negative matrix factorization (NMF) as described (49). Class assignment stability was quantified by the cophenetic correlation coefficient for the co-ocurruence matrix following 1000 randomly initialized factorizations. Reduced data sets evaluated by NMF were generated by sequentially eliminating cytokine markers exhibiting the smallest total variation. A multiple testing threshold (FDR<0.05) for individual Wilcoxon rank-sum tests was similarly applied to determine cytokines discriminating NMF subpopulations C1 and C2. All statistical analysis was performed with Matlab Statistics and Bioinformatics toolboxes.
Gene Set Enrichment and Co-expression Analysis
Gene Set Enrichment Analysis (GSEA) was performed as described (50) using the Immune Response in silico (IRIS; GES22886; Ref. 25) compendia to assess enrichment for cytokine production across resting and activate immune cell lineages. To accommodate incomplete measurement of all known secreted gene products, statistical significance of phenotype enrichment scores was computed using a null distribution of enrichment scores corresponding to randomly generated cytokine signatures of identical size from the list of forty-seven assayed proteins. Pair-wise enrichment for coordinated cytokine ligand and receptor expression between lineages was similarly calculated using a two-dimensional enrichment statistic to identify overrepresented intercellular interactions. Directed edges between lineages were constructed for scores attaining significance (P<0.05), and nodes arranged hierarchically in order of decreasing out-degree. For co-expression analyses, gene transcripts represented in the IRIS and the Human Immune Cell Transcriptome (HICT; GSE3982; Ref. 35) compendia were rank-ordered by the median Pearson correlation to signature cytokines of interest. Re-ordered gene lists served as a basis for standard GSEA utilizing the Canonical Pathways and Transcription Factor Target collections from the Molecular Signatures Database release 4.0 (51). Statistical over-representation of predicted transcription factor binding sites obtained from TRANSFAC Match (52) was determined by Fisher's exact test. Additional details describing data preprocessing and enrichment analyses are provided in Supplementary Materials and Methods.
Supplementary Material
One sentence summary.
The molecular correlates of heterogeneous endometriosis symptoms strongly support a role for innate inflammatory responses converging upon c-Jun and NFκB signaling cascades.
Acknowledgements
The authors gratefully acknowledge the study participants and surgical staff who assisted in the collection of specimens. We are indebted to David Clarke, Brian Joughin, Justin Pritchard, and Melody Morris for their helpful comments and suggestions during preparation of the manuscript.
Funding: This work received funding support from an anonymous foundation, the John D. and Catherine T. MacArthur Foundation Fellowship, the NIH (R01 EB10246), and the Institute for Collaborative Biotechnologies (W911NF-09-0001) from the U.S. Army Research Office. The content of the information does not necessarily reflect the position or the policy of the funding agencies, government, and no official endorsement should be inferred.
Symbols, abbreviations, and acronyms
- ASRM
American Society for Reproductive Medicine
- GSEA
Gene Set Enrichment Analysis
- HICT
Human Immune Cell Transcriptome
- IQR
Interquartile Range
- IRIS
Immune Response in Silico
- NMF
Non-negative Matrix Factorization
Footnotes
Author Contributions: M.T.B., L.G.G., K.B.I, and D.A.L conceived the project; N.P.D., E.A.P, and M.TB. coordinated patient recruitment, collected, processed and banked biological specimens; K.B.I and S.N.M. clinically evaluated patients and performed surgeries; M.T.B performed experiments and statistical/network analyses. M.T.B., L.G.G. and K.B.I. wrote the manuscript which was read and approved by all co-authors.
Competing Interests: The authors declare no competing interests.
References
- 1.Eskenazi B, Warner ML. Epidemiology of endometriosis. Obstetrics and gynecology clinics of North America. 1997;24:235–258. doi: 10.1016/s0889-8545(05)70302-8. [DOI] [PubMed] [Google Scholar]
- 2.Sampson JA. Metastatic or embolic endometriosis, due to the menstrual dissemination of endometrial tissue into the venous circulation. The American journal of pathology. 1927;3:93. [PMC free article] [PubMed] [Google Scholar]
- 3.Giudice LC. Endometriosis. New England Journal of Medicine. 2010;362:2389–2398. doi: 10.1056/NEJMcp1000274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Herington JL, Bruner-Tran KL, Lucas JA, Osteen KG. Immune interactions in endometriosis. Expert review of clinical immunology. 2011;7:611–626. doi: 10.1586/eci.11.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bulun SE. Endometriosis. New England Journal of Medicine. 2009;360:268–79. doi: 10.1056/NEJMra0804690. [DOI] [PubMed] [Google Scholar]
- 6.Braundmeier AG, Fazleabas AT. The non-human primate model of endometriosis: research and implications for fecundity. Molecular Human Reproduction. 2009;15:577–586. doi: 10.1093/molehr/gap057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rogers PA, D'Hooghe TM, Fazleabas AT, Gargett CE, Giudice LC, Montgomery GW, Rombauts L, Salamonsen L, Zondervan KT. Priorities for endometriosis research: recommendations from an international consensus workshop. Reproductive Sciences. 2009;16:335–346. doi: 10.1177/1933719108330568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Palmer SS, Barnhart KT. Biomarkers in reproductive medicine: the promise, and can it be fulfilled?. Fertility and sterility. 2012;6:81–92. doi: 10.1016/j.fertnstert.2012.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vercellini P, Fedele L, Aimi G, De Giorgi O, Consonni D, Crosignani PG. Reproductive performance, pain recurrence and disease relapse after conservative surgical treatment for endometriosis: the predictive value of the current classification system. Human reproduction. 2006;21:2679–2685. doi: 10.1093/humrep/del230. [DOI] [PubMed] [Google Scholar]
- 10.Vercellini P, Fedele L, Aimi G, Pietropaolo G, Consonni D, Crosignani PG. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Human reproduction. 2007;22:266–271. doi: 10.1093/humrep/del339. [DOI] [PubMed] [Google Scholar]
- 11.Roberts CP, Rock JA. The current staging system for endometriosis: does it help?. Obstetrics and gynecology clinics of North America. 2003;30:115–132. doi: 10.1016/s0889-8545(02)00056-6. [DOI] [PubMed] [Google Scholar]
- 12.Adamson GD. Endometriosis classification: an update. Current Opinion in Obstetrics and Gynecology. 2011;23:213–220. doi: 10.1097/GCO.0b013e328348a3ba. [DOI] [PubMed] [Google Scholar]
- 13.Lebovic DI, Mueller MD, Taylor RN. Immunobiology of endometriosis. Fertility and sterility. 2001;75:1–10. doi: 10.1016/s0015-0282(00)01630-7. [DOI] [PubMed] [Google Scholar]
- 14.Seeber B, Sammel MD, Fan X, Gerton GL, Shaunik A, Chittams J, Barnhart KT. Proteomic analysis of serum yields six candidate proteins that are differentially regulated in a subset of women with endometriosis. Fertility and sterility. 2010;93:2137–2144. doi: 10.1016/j.fertnstert.2008.12.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vodolazkaia A, El-Aalamat Y, Popovic D, Mihalyi A, Bossuyt X, Kyama CM, Fassbender A, Bokor A, Schols D, Huskens D, Meuleman C, Peeraer K, Tomasetti C, Gevaert O, Waelkens E, Kasran A, De Moor B, D'Hooghe TM. Evaluation of a panel of 28 biomarkers for the non-invasive diagnosis of endometriosis. Human reproduction. 2012;27:2698–2711. doi: 10.1093/humrep/des234. [DOI] [PubMed] [Google Scholar]
- 16.Kyama CM, Mihalyi A, Simsa P, Mwenda JM, Tomassetti C, Meuleman C, D'Hooghe TM. Non-steroidal targets in the diagnosis and treatment of endometriosis. Current medicinal chemistry. 2008;15:1006–1017. doi: 10.2174/092986708784049595. [DOI] [PubMed] [Google Scholar]
- 17.Soares SR, Martínez-Varea A, Hidalgo-Mora JJ, Pellicer A. Pharmacologic therapies in endometriosis: a systematic review. Fertility and sterility. 2012;98:529–555. doi: 10.1016/j.fertnstert.2012.07.1120. [DOI] [PubMed] [Google Scholar]
- 18.Missmer SA, Hankinson SE, Spiegelman D, Barbieri RL, Malpeis S, Willett WC, Hunter DJ. Reproductive history and endometriosis among premenopausal women. Obstetrics & Gynecology. 2004;104:965–974. doi: 10.1097/01.AOG.0000142714.54857.f8. [DOI] [PubMed] [Google Scholar]
- 19.Olive DL, Weinberg JB, Haney AF. Peritoneal macrophages and infertility: the association between cell number and pelvic pathology. Fertility and sterility. 1985;44:772–777. doi: 10.1016/s0015-0282(16)49036-9. [DOI] [PubMed] [Google Scholar]
- 20.Brunet JP, Tamayo P, Golub TR, Mesirov JP. Metagenes and molecular pattern discovery using matrix factorization. Proceedings of the National Academy of Sciences. 2004;101.12:4164–4169. doi: 10.1073/pnas.0308531101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koninckx PR, Renaer M, Brosens IA. Origin of peritoneal fluid in women: an ovarian exudation product. BJOG: An International Journal of Obstetrics & Gynaecology. 1980;87:177–183. doi: 10.1111/j.1471-0528.1980.tb04514.x. [DOI] [PubMed] [Google Scholar]
- 22.Overton C, Fernandez-Shaw S, Hicks B, Barlow D, Starkey P. Peritoneal fluid cytokines and the relationship with endometriosis and pain. Human reproduction. 1996;11:380–386. doi: 10.1093/humrep/11.2.380. [DOI] [PubMed] [Google Scholar]
- 23.Gazvani MR, Christmas S, Quenby S, Kirwan J, Johnson PM, Kingsland CR. Peritoneal fluid concentrations of interleukin-8 in women with endometriosis: relationship to stage of disease. Human Reproduction. 1998;13:1957–1961. doi: 10.1093/humrep/13.7.1957. [DOI] [PubMed] [Google Scholar]
- 24.Meissner F, Scheltema RA, Mollenkopf HJ, Mann M. Direct Proteomic Quantification of the Secretome of Activated Immune Cells. Science. 2013;340:475–478. doi: 10.1126/science.1232578. [DOI] [PubMed] [Google Scholar]
- 25.Abbas AR, Baldwin D, Ma Y, Ouyang W, Gurney A, Martin F, Fong S, van Lookeren Campagne M, Godowski P, Williams PM, Chan AC, Clark HF. Immune response in silico (IRIS): immune-specific genes identified from a compendium of microarray expression data. Genes and immunity. 2005;6:319–331. doi: 10.1038/sj.gene.6364173. [DOI] [PubMed] [Google Scholar]
- 26.Andreoli CG, Genro VK, Souza CA, Michelon T, Bilibio JP, Scheffel C, Cunha-Filho JS. T helper (Th) 1, Th2, and Th17 interleukin pathways in infertile patients with minimal/mild endometriosis. Fertility and sterility. 2011;95:2477–2480. doi: 10.1016/j.fertnstert.2011.02.019. [DOI] [PubMed] [Google Scholar]
- 27.Koga K, Osuga Y, Tsutsumi O, Okagaki R, Momoeda M, Yano T, Fujiwara T, Takai Y, Kugu K, Morita Y, Taketani Y. Increased concentrations of soluble tumour necrosis factor receptor (sTNFR) I and II in peritoneal fluid from women with endometriosis. Molecular human reproduction. 2000;6:929–933. doi: 10.1093/molehr/6.10.929. [DOI] [PubMed] [Google Scholar]
- 28.Laudański P, Szamatowicz J, Oniszczuk M. Profiling of peritoneal fluid of women with endometriosis by chemokine protein array. Adv Med Sci. 2006;51:148–52. [PubMed] [Google Scholar]
- 29.Suzumori N, Katano K, Suzumori K. Peritoneal fluid concentrations of epithelial neutrophil-activating peptide-78 correlate with the severity of endometriosis. Fertility and sterility. 2004;81:305–308. doi: 10.1016/j.fertnstert.2003.08.011. [DOI] [PubMed] [Google Scholar]
- 30.Szamatowicz J, Laudański P, Tomaszewska I. Matrix metalloproteinase-9 and tissue inhibitor of matrix metalloproteinase-1: a possible role in the pathogenesis of endometriosis. Human Reproduction. 2002;17:284–288. doi: 10.1093/humrep/17.2.284. [DOI] [PubMed] [Google Scholar]
- 31.Wieser F, Dogan D, Klingel K, Diedrich K, Taylor RN, Hornung D. Expression and regulation of CCR1 in peritoneal macrophages from women with and without endometriosis. Fertility and sterility. 2005;83:1878–1881. doi: 10.1016/j.fertnstert.2004.12.034. [DOI] [PubMed] [Google Scholar]
- 32.Kabut J, Kondera-Anasz Z, Sikora J, Mielczarek-Palacz A. Levels of complement components iC3b, C3c, C4, and SC5b-9 in peritoneal fluid and serum of infertile women with endometriosis. Fertility and sterility. 2007;88:1298–1303. doi: 10.1016/j.fertnstert.2006.12.061. [DOI] [PubMed] [Google Scholar]
- 33.Lousse JC, Van Langendonckt A, González-Ramos R, Defrère S, Renkin E, Donnez J. Increased activation of nuclear factor-kappa B (NF-κB) in isolated peritoneal macrophages of patients with endometriosis. Fertility and sterility. 2008;90:217–220. doi: 10.1016/j.fertnstert.2007.06.015. [DOI] [PubMed] [Google Scholar]
- 34.Wu MH, Sun HS, Lin CC, Hsiao KY, Chuang PC, Pan HA, Tsai SJ. Distinct mechanisms regulate cyclooxygenase-1 and-2 in peritoneal macrophages of women with and without endometriosis. Molecular human reproduction. 2002;8:1103–1110. doi: 10.1093/molehr/8.12.1103. [DOI] [PubMed] [Google Scholar]
- 35.Jeffrey KL, Brummer T, Rolph MS, Liu SM, Callejas NA, Grumont RJ, Gillieron C, Mackay F, Grey S, Camps M, Rommel C, Gerondakis SD, Mackay CR. Positive regulation of immune cell function and inflammatory responses by phosphatase PAC-1. Nature immunology. 2006;7:274–283. doi: 10.1038/ni1310. [DOI] [PubMed] [Google Scholar]
- 36.Painter JN, Anderson CA, Nyholt DR, Macgregor S, Lin J, Lee SH, Lambert A, Zhao ZZ, Roseman F, Guo Q, Gordon SD, Wallace L, Henders AK, Visscher PM, Kraft P, Martin NG, Morris AP, Treloar SA, Kennedy SH, Missmer SA, Montgomery GW, Zondervan KT. Genome-wide association study identifies a locus at 7p15. 2 associated with endometriosis. Nature genetics. 2010;43:51–54. doi: 10.1038/ng.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Grechukhina O, Petracco R, Popkhadze S, Massasa E, Paranjape T, Chan E, Flores I, Weidhaas JB, Taylor HS. A polymorphism in a let-7 microRNA binding site of KRAS in women with endometriosis. EMBO molecular medicine. 2012;4:206–217. doi: 10.1002/emmm.201100200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nyholt DR, Low SK, Anderson CA, Painter JN, Uno S, Morris AP, MacGregor S, Gordon SD, Henders AK, Martin NG, Attia J, Holliday EG, McEvoy M, Scott RJ, Kennedy SH, Treloar SA, Missmer SA, Adachi S, Tanaka K, Nakamura Y, Zondervan KT, Zembutsu H, Montgomery GW. Genome-wide association meta-analysis identifies new endometriosis risk loci. Nature genetics. 2012;44:1355–1359. doi: 10.1038/ng.2445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Huang SSC, Clarke DC, Gosline SJ, Labadorf A, Chouinard CR, Gordon W, Lauffenburger DA, Fraenkel E. Linking proteomic and transcriptional data through the interactome and epigenome reveals a map of oncogene-induced signaling. PLoS computational biology. 2013;9:e1002887. doi: 10.1371/journal.pcbi.1002887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Miller MA, Meyer AS, Beste MT, Lasisi Z, Reddy S, Jeng KW, Chen CH, Han J, Isaacson KB, Griffith LG, Lauffenburger DA. ADAM-10 and-17 regulate endometriotic cell migration via concerted ligand and receptor shedding feedback on kinase signaling. Proceedings of the National Academy of Sciences. 2013;110:E2074–E2083. doi: 10.1073/pnas.1222387110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Altan M, Bruner-Tran K, Osteen K, Palmer S. JNK inhibitor AS-01 causes regression of endometriosis and suppression of inflammatory cytokines and tissue remodeling enzymes. Biology of Reproduction. 2008;SI:168–169. [Google Scholar]
- 42.Nataraja S, Kagan D, Starzinski-Powitz A, Tos EG, Cirillo R, Ferrandi C, Gotteland JP, Osteen KG, Bruner-Tran K, Palmer S. Interruption of inflammatory pathways with JNK inhibitors induces regression of human endometriotic lesions and restores progesterone receptor sensitivity in endometrium and lesions. Biology of Reproduction. 2008;SI:113. [Google Scholar]
- 43.Mathur S, Peress MR, Williamson HO, Youmans CD, Maney SA, Garvin AJ, Rust PF, Fudenberg HH. Autoimmunity to endometrium and ovary in endometriosis. Clinical and experimental immunology. 1982;50:259–266. [PMC free article] [PubMed] [Google Scholar]
- 44.Isaacson KB, Coutifaris C, Garcia CR, Lyttle CR. Production and secretion of complement component 3 by endometriotic tissue. Journal of Clinical Endocrinology & Metabolism. 1989;69:1003–1009. doi: 10.1210/jcem-69-5-1003. [DOI] [PubMed] [Google Scholar]
- 45.Bacci M, Capobianco A, Monno A, Cottone L, Di Puppo F, Camisa B, Mariani M, Brignole C, Pnzoni M, Ferrari S, Panina-Bordignon P, Manfredi AA, Rovere-Querini P. Macrophages are alternatively activated in patients with endometriosis and required for growth and vascularization of lesions in a mouse model of disease. The American journal of pathology. 2009;175:547–556. doi: 10.2353/ajpath.2009.081011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Khan KN, Kitajima M, Hiraki K, Yamaguchi N, Katamine S, Matsuyama T, Nakashima M, Fujishita A, Ishimaru T, Masuzaki H. Escherichia coli contamination of menstrual blood and effect of bacterial endotoxin on endometriosis. Fertility and sterility. 2010;94:2860–2863. doi: 10.1016/j.fertnstert.2010.04.053. [DOI] [PubMed] [Google Scholar]
- 47.Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends in immunology. 2004;25:677–686. doi: 10.1016/j.it.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 48.Schenken RS, Guzick DS. Revised endometriosis classification: 1996. Fertility and sterility. 1997;67.5:815–816. doi: 10.1016/s0015-0282(97)81390-8. [DOI] [PubMed] [Google Scholar]
- 49.Brunet JP, Tamayo P, Golub TR, Mesirov JP. Metagenes and molecular pattern discovery using matrix factorization. Proceedings of the National Academy of Sciences. 2004;101:4164–4169. doi: 10.1073/pnas.0308531101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liberzon A, Subramanian A, Pinchback R, Thorvaldsdóttir H, Tamayo P, Mesirov JP. Molecular signatures database (MSigDB) 3.0. Bioinformatics. 2011;27:1739–1740. doi: 10.1093/bioinformatics/btr260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kel AE, Gößling E, Reuter I, Cheremushkin E, Kel-Margoulis OV, Wingender E. MATCHTM: a tool for searching transcription factor binding sites in DNA sequences. Nucleic acids research. 2003;31:3576–3579. doi: 10.1093/nar/gkg585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ni S, Vingron M. R2KS: A Novel Measure for Comparing Gene Expression Based on Ranked Gene Lists. Journal of Computational Biology. 2012;19:766–775. doi: 10.1089/cmb.2012.0026. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.