Abstract
Mice injected intravenously with a commercially available extract of Coxiella burnetii prepared for use as the antigen in the complement fixation diagnostic test for Q fever were subsequently resistant to infection with Babesia microti, Babesia rodhaini, and Plasmodium vinckei petteri. The parasites appeared to die inside circulating erythrocytes. Protection was unaffected by exposing the pretreated mice to 900 rads on the day before they were infected. To explain these findings, it is postulated that pretreatment with Coxiella extract protects by potentiating the interferon-inducing capacity of the challenge dose of protozoa, which perhaps leads to enhanced of natural killer cells. Tumor necrosis factor also warrants investigation.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ABERNATHY R. S., BRADLEY G. M., SPINK W. W. Increased susceptibility of mice with brucellosis to bacterial endotoxins. J Immunol. 1958 Oct;81(4):271–275. [PubMed] [Google Scholar]
- Brocklesby D. W., Harradine D. L. The effect of an interferon inducer on experimental mouse piroplasmosis (Babesia rodhaini infection). Res Vet Sci. 1973 May;14(3):397–398. [PubMed] [Google Scholar]
- Carswell E. A., Old L. J., Kassel R. L., Green S., Fiore N., Williamson B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3666–3670. doi: 10.1073/pnas.72.9.3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
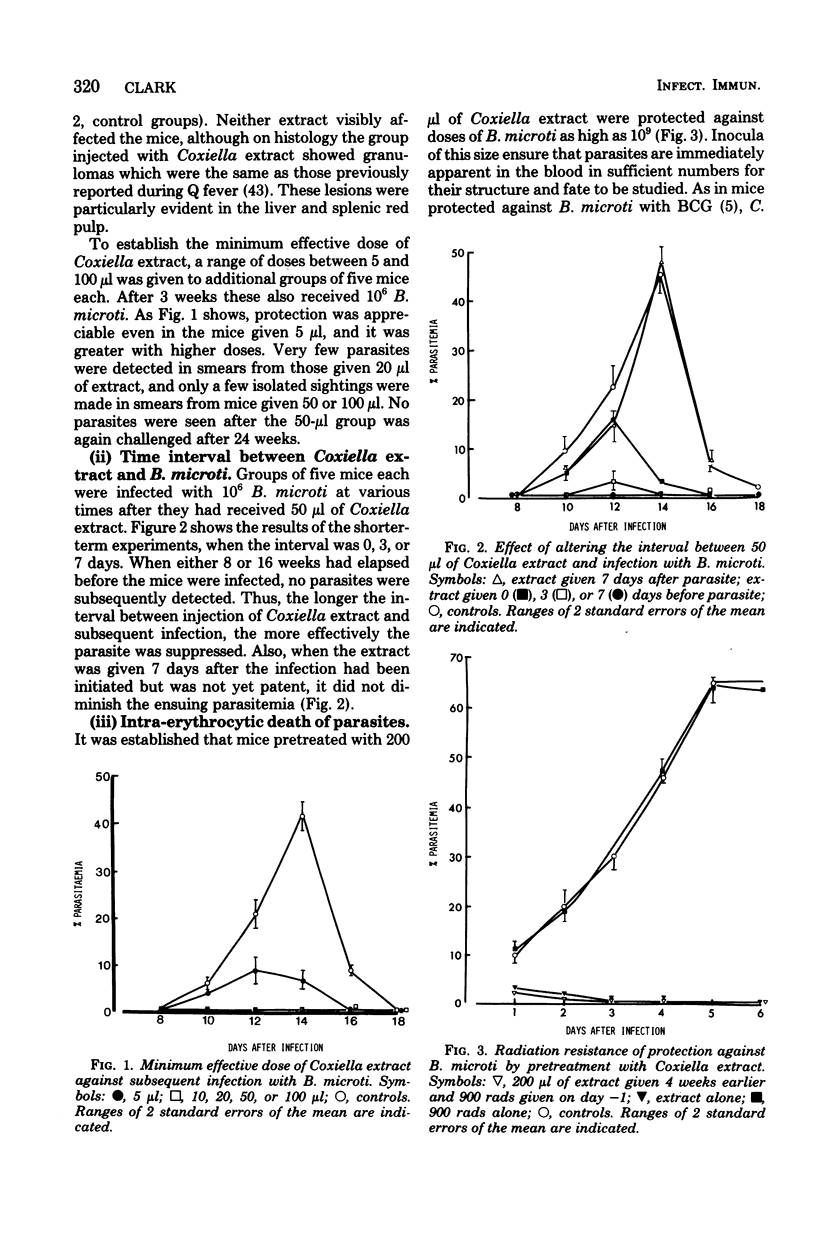
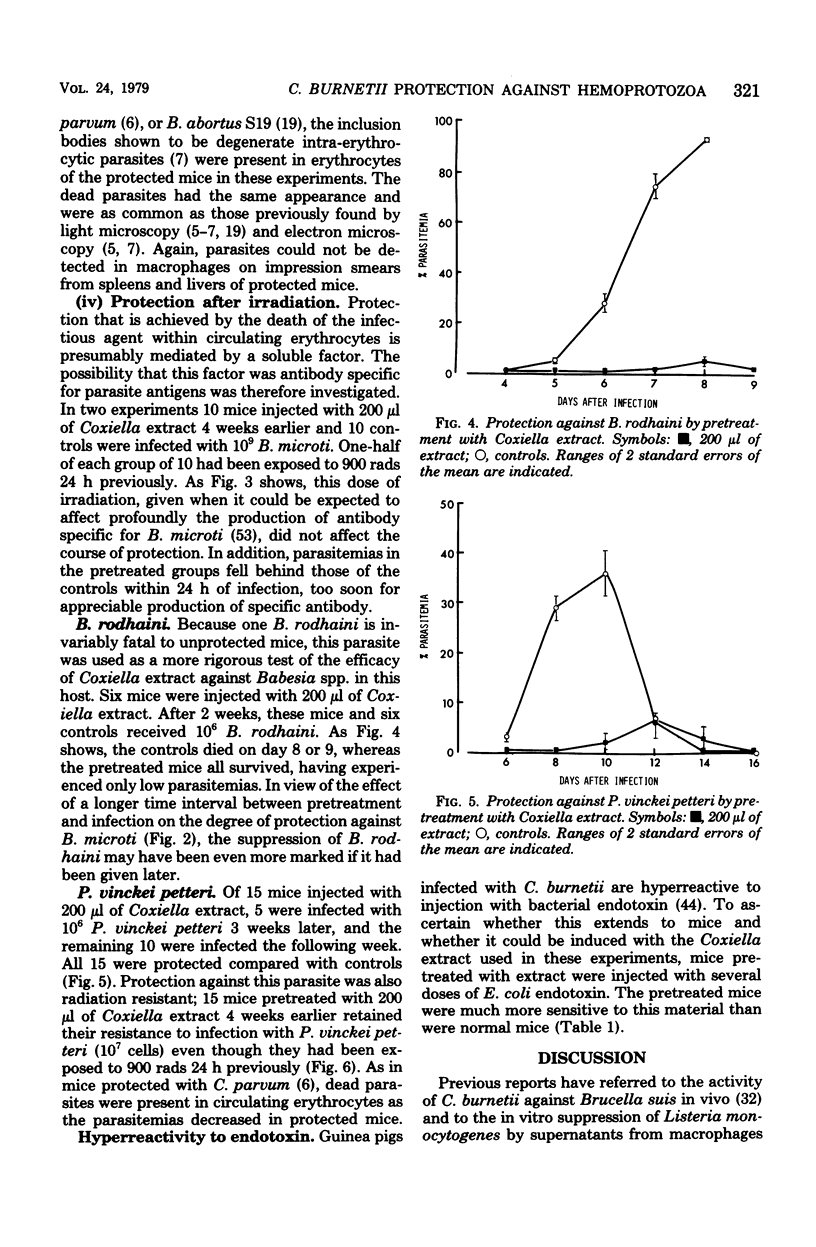
- Clark I. A., Allison A. C., Cox F. E. Protection of mice against Babesia and Plasmodium with BCG. Nature. 1976 Jan 29;259(5541):309–311. doi: 10.1038/259309a0. [DOI] [PubMed] [Google Scholar]
- Clark I. A., Cox F. E., Allison A. C. Protection of mice against Babesia spp. and Plasmodium spp. with killed Corynebacterium parvum. Parasitology. 1977 Feb;74(1):9–18. doi: 10.1017/s003118200004748x. [DOI] [PubMed] [Google Scholar]
- Clark I. A., Richmond J. E., Wills E. J., Allison A. C. Intra-erythrocytic death of the parasite in mice recovering from infection with Babesia microti. Parasitology. 1977 Oct;75(2):189–196. doi: 10.1017/s0031182000062338. [DOI] [PubMed] [Google Scholar]
- Clark I. A., Wills E. J., Richmond J. E., Allison A. C. Suppression of babesiosis in BCG-infected mice and its correlation with tumor inhibition. Infect Immun. 1977 Aug;17(2):430–438. doi: 10.1128/iai.17.2.430-438.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diggs C. L., Osler A. G. Humoral immunity in rodent malaria. II. Inhibition of parasitemia by serum antibody. J Immunol. 1969 Feb;102(2):298–305. [PubMed] [Google Scholar]
- ELBERG S. S., SCHNEIDER P., FONG J. Cross-immunity between Brucella melitensis and Mycobacterium tuberculosis; intracellular behavior of Brucella melitensis in monocytes from vaccinated animals. J Exp Med. 1957 Oct 1;106(4):545–554. doi: 10.1084/jem.106.4.545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frenkel J. K., Lunde M. N. Effects of corticosteroids on antibody and immunity in Besnoitia infection of hamsters. J Infect Dis. 1966 Oct;116(4):414–424. doi: 10.1093/infdis/116.4.414. [DOI] [PubMed] [Google Scholar]
- GLEDHILL A. W., NIVEN J. S. The toxicity of some bacterial filtrates for mice pre-infected with eperythrozoon coccoides. Br J Exp Pathol. 1957 Jun;38(3):284–290. [PMC free article] [PubMed] [Google Scholar]
- Gidlund M., Orn A., Wigzell H., Senik A., Gresser I. Enhanced NK cell activity in mice injected with interferon and interferon inducers. Nature. 1978 Jun 29;273(5665):759–761. doi: 10.1038/273759a0. [DOI] [PubMed] [Google Scholar]
- Glasgow L. A., Odugbemi T., Dwyer P., Ritterson A. L. Eperythrozoon coccoides. I. Effect on the interferon response in mice. Infect Immun. 1971 Oct;4(4):425–430. doi: 10.1128/iai.4.4.425-430.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halpern B. N., Biozzi G., Stiffel C., Mouton D. Inhibition of tumour growth by administration of killed corynebacterium parvum. Nature. 1966 Nov 19;212(5064):853–854. doi: 10.1038/212853a0. [DOI] [PubMed] [Google Scholar]
- Hamburger J., Kreier J. P. Plasmodium berghei: use of free blood stage parasites to demonstrate protective humoral activity in the serum of recovered rats. Exp Parasitol. 1976 Oct;40(2):158–169. doi: 10.1016/0014-4894(76)90078-3. [DOI] [PubMed] [Google Scholar]
- Herod E., Clark I. A., Allison A. C. Protection of mice against haemoprotozoan Babesia microti with Brucella abortus strain 19. Clin Exp Immunol. 1978 Mar;31(3):518–523. [PMC free article] [PubMed] [Google Scholar]
- Hochman P. S., Cudkowicz G., Dausset J. Decline of natural killer cell activity in sublethally irradiated mice. J Natl Cancer Inst. 1978 Jul;61(1):265–268. doi: 10.1093/jnci/61.1.265. [DOI] [PubMed] [Google Scholar]
- Huang K. Y., Schultz W. W., Gordon F. B. Interferon induced by Plasmodium berghei. Science. 1968 Oct 4;162(3849):123–124. doi: 10.1126/science.162.3849.123. [DOI] [PubMed] [Google Scholar]
- Ito Y., Nagata I., Kunii A. Mechanism of endotoxin-type interferon production in mice. Virology. 1973 Apr;52(2):439–446. doi: 10.1016/0042-6822(73)90339-5. [DOI] [PubMed] [Google Scholar]
- Jahiel R. I., Nussenzweig R. S., Vilcek J., Vanderberg J. Protective effect of interferon inducers on Plasmodium berghei malaria. Am J Trop Med Hyg. 1969 Nov;18(6):823–835. doi: 10.4269/ajtmh.1969.18.823. [DOI] [PubMed] [Google Scholar]
- Jahiel R. I., Vilcek J., Nussenzweig R., Vanderberg J. Interferon inducers protect mice against plasmodium berghei malaria. Science. 1968 Aug 23;161(3843):802–804. doi: 10.1126/science.161.3843.802. [DOI] [PubMed] [Google Scholar]
- Kazár J. Interferon-like inhibitor in mouse sera induced by rickettsiae. Acta Virol. 1966 May;10(3):277–277. [PubMed] [Google Scholar]
- Kelly M. T. Activation of guinea pig macrophages by Q fever rickettsiae. Cell Immunol. 1977 Jan;28(1):198–205. doi: 10.1016/s0008-8749(77)80020-8. [DOI] [PubMed] [Google Scholar]
- Ketteridge D. S. Lipopolysaccharide from Trypanosoma cruzi. Trans R Soc Trop Med Hyg. 1978;72(1):101–102. doi: 10.1016/0035-9203(78)90310-3. [DOI] [PubMed] [Google Scholar]
- Kiessling R., Klein E., Wigzell H. "Natural" killer cells in the mouse. I. Cytotoxic cells with specificity for mouse Moloney leukemia cells. Specificity and distribution according to genotype. Eur J Immunol. 1975 Feb;5(2):112–117. doi: 10.1002/eji.1830050208. [DOI] [PubMed] [Google Scholar]
- Kirchner H., Hirt H. M., Becker H., Munk K. Production of an antiviral factor by murine spleen cells after treatment with Corynebacterium parvum. Cell Immunol. 1977 Jun 1;31(1):172–176. doi: 10.1016/0008-8749(77)90016-8. [DOI] [PubMed] [Google Scholar]
- Lindahl-Magnusson P., Leary P., Gresser I. Interferon inhibits DNA synthesis induced in mouse lymphocyte suspensions by phytohaemagglutinin or by allogeneic cells. Nat New Biol. 1972 May 24;237(73):120–121. doi: 10.1038/newbio237120a0. [DOI] [PubMed] [Google Scholar]
- MIKA L. A., GOODLOW R. J., VICTOR J., BRAUN W. Studies on mixed infections. I. Brucellosis and Q fever. Proc Soc Exp Biol Med. 1954 Dec;87(3):500–507. doi: 10.3181/00379727-87-21424. [DOI] [PubMed] [Google Scholar]
- NYKA W. Enhancement of resistance to tuberculosis in mice experimentally infected with brucella abortus. Am Rev Tuberc. 1956 Feb;73(2):251–265. doi: 10.1164/artpd.1956.73.2.251. [DOI] [PubMed] [Google Scholar]
- Nagano Y., Mizunoe K., Maehara N., Kumazawa Y. Induction of virus-inhibiting factor or interferon by cell fractions of Mycobacterium tuberculosis. Jpn J Microbiol. 1971 Nov;15(6):542–544. doi: 10.1111/j.1348-0421.1971.tb00617.x. [DOI] [PubMed] [Google Scholar]
- OLD L. J., CLARKE D. A., BENACERRAF B. Effect of Bacillus Calmette-Guerin infection on transplanted tumours in the mouse. Nature. 1959 Jul 25;184(Suppl 5):291–292. doi: 10.1038/184291a0. [DOI] [PubMed] [Google Scholar]
- Oehler J. R., Herberman R. B. Natural cell-mediated cytotoxicity in rats. III. Effects of immunopharmacologic treatments on natural reactivity and on reactivity augmented by polyinosinic-polycytidylic acid. Int J Cancer. 1978 Feb 15;21(2):221–229. doi: 10.1002/ijc.2910210214. [DOI] [PubMed] [Google Scholar]
- Oehler J. R., Lindsay L. R., Nunn M. E., Herberman R. B. Natural cell-mediated cytotoxicity in rats. I. Tissue and strain distribution, and demonstration of a membrance receptor for the Fc portion of IgG. Int J Cancer. 1978 Feb 15;21(2):204–209. doi: 10.1002/ijc.2910210212. [DOI] [PubMed] [Google Scholar]
- Oehler J. R., Lindsay L. R., Nunn M. E., Holden H. T., Herberman R. B. Natural cell-mediated cytotoxicity in rats. II. In vivo augmentation of NK-cell activity. Int J Cancer. 1978 Feb 15;21(2):210–220. doi: 10.1002/ijc.2910210213. [DOI] [PubMed] [Google Scholar]
- Orsay C. P., Heath J. E. Hyperthermia in rabbits from injection of malaria infected erythrocytes in the pre-optic area. Nature. 1973 Nov 16;246(5429):162–163. doi: 10.1038/246162a0. [DOI] [PubMed] [Google Scholar]
- Ortiz-Ortiz L., Gonzalez-Mendoza A., Lamoyi E. A vaccination procedure against Trypanosoma cruzi infection in mice by nonspecific immunization. J Immunol. 1975 Apr;114(4):1424–1425. [PubMed] [Google Scholar]
- PICCHI J., NELSON A. R., WALLER E. E., RAZAVI M., CLIZER E. E. Q fever associated with granulomatous hepatitis. Ann Intern Med. 1960 Nov;53:1065–1074. doi: 10.7326/0003-4819-53-5-1065. [DOI] [PubMed] [Google Scholar]
- PIRSCH J. B., MIKA L. A., VAN DER MAATEN M. J. Hyperreactivity of Coxiella burnetii infected guinea pigs to subsequent injections of bacterial endotoxins. Proc Soc Exp Biol Med. 1957 Nov;96(2):376–380. doi: 10.3181/00379727-96-23482. [DOI] [PubMed] [Google Scholar]
- Peter H. H., Eife R. F., Kalden J. R. Spontaneous cytotoxicity (SCMC) of normal human lymphocytes against a human melanoma cell line: a phenomenon due to a lymphotoxin-like mediator. J Immunol. 1976 Feb;116(2):342–348. [PubMed] [Google Scholar]
- Phillips R. S., Jones V. E. Immunity to Plasmodium berghei in rats: maximum levels of protective antibody activity are associated with eradication of the infection. Parasitology. 1972 Feb;64(1):117–127. doi: 10.1017/s0031182000044693. [DOI] [PubMed] [Google Scholar]
- Roberts J. A., Tracey-Patte P. Adoptive transfer of immunity to Plasmodium berghei. J Protozool. 1969 Nov;16(4):728–730. doi: 10.1111/j.1550-7408.1969.tb02334.x. [DOI] [PubMed] [Google Scholar]
- Rytel M. W., Marsden P. D. Induction of an interferon-like inhibitor by Trypanosoma cruzi infection in mice. Am J Trop Med Hyg. 1970 Nov;19(6):929–931. doi: 10.4269/ajtmh.1970.19.929. [DOI] [PubMed] [Google Scholar]
- STOKER M. G. Variation in complement-fixing activity of Rickettsia burneti during egg adaptation. J Hyg (Lond) 1953 Sep;51(3):311–321. doi: 10.1017/s0022172400015746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SUTER E., ULLMAN G. E., HOFFMAN R. G. Sensitivity of mice to endotoxin after vaccination with BCG (Bacillus Calmette-Guérin). Proc Soc Exp Biol Med. 1958 Oct;99(1):167–169. doi: 10.3181/00379727-99-24282. [DOI] [PubMed] [Google Scholar]
- Smith T. J., Wagner R. R. Rabbit macrophage interferons. I. Conditions for biosynthesis by virus-infected and uninfected cells. J Exp Med. 1967 Apr 1;125(4):559–577. doi: 10.1084/jem.125.4.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprent J. Circulating T and B lymphocytes of the mouse. I. Migratory properties. Cell Immunol. 1973 Apr;7(1):10–39. doi: 10.1016/0008-8749(73)90180-9. [DOI] [PubMed] [Google Scholar]
- Stahl W., Matsubayashi H., Akao S. Experimental toxoplasmosis: effects of suppression of the immune response of mice by cortisone and splenectomy. Keio J Med. 1966 Mar;15(1):1–10. doi: 10.2302/kjm.15.1. [DOI] [PubMed] [Google Scholar]
- Stechschulte D. J. Cell-mediated immunity in rats infected with Plasmodium berghei. Mil Med. 1969 Sep;134(10):1147–1152. [PubMed] [Google Scholar]
- TALIAFERRO W. H., TALIAFERRO L. G., JANSSEN E. F. The localization of x-ray injury to the initial phases of antibody response. J Infect Dis. 1952 Sep-Oct;91(2):105–124. doi: 10.1093/infdis/91.2.105. [DOI] [PubMed] [Google Scholar]
- Veskova T. K., Chimishkyan K. L., Sver-Moldavsky G. J. Effect of Brucella abortus infection (vaccine strain 19BA) on Rauscher leukemia virus and L1210 leukemia in mice. J Natl Cancer Inst. 1974 May;52(5):1651–1653. doi: 10.1093/jnci/52.5.1651. [DOI] [PubMed] [Google Scholar]
- Wolf R. E. Effects of antilymphocyte serum and splenectomy on resistance to Babesia microti infection in hamsters. Clin Immunol Immunopathol. 1974 Apr;2(3):381–394. doi: 10.1016/0090-1229(74)90056-7. [DOI] [PubMed] [Google Scholar]
- Wolfe S. A., Tracey D. E., Henney C. S. Introduction of "natural" killer' cells by BCG. Nature. 1976 Aug 12;262(5569):584–586. doi: 10.1038/262584a0. [DOI] [PubMed] [Google Scholar]
- YOUNGNER J. S., STINEBRING W. R. INTERFERON REDUCTION IN CHICKENS INJECTED WITH BRUCELLA ABORTUS. Science. 1964 May 22;144(3621):1022–1023. doi: 10.1126/science.144.3621.1022. [DOI] [PubMed] [Google Scholar]
- Young A. S., Cox F. E. The effect of betamethasone on Babesia microti and B. rodhaini infections in rodents. Parasitology. 1971 Dec;63(3):447–453. doi: 10.1017/s003118200007997x. [DOI] [PubMed] [Google Scholar]
- Youngner J. S., Stinebring W. R. Interferon appearance stimulated by endotoxin, bacteria, or viruses in mice pre-treated with Escherichia coli endotoxin or infected with Mycobacterium tuberculosis. Nature. 1965 Oct 30;208(5009):456–458. doi: 10.1038/208456a0. [DOI] [PubMed] [Google Scholar]


