Abstract
The neurovascular unit—comprised of glia, pericytes, neurons and cerebrovasculature—is a dynamic interface that ensures physiological central nervous system (CNS) functioning. In disease dynamic remodeling of the neurovascular interface triggers a cascade of responses that determine the extent of CNS degeneration and repair. The dynamics of these processes can be adequately captured by imaging in vivo, which allows the study of cellular responses to environmental stimuli and cell-cell interactions in the living brain in real time. This perspective focuses on intravital imaging studies of the neurovascular unit in stroke, multiple sclerosis (MS) and Alzheimer disease (AD) models and discusses their potential for identifying novel therapeutic targets.
Keywords: blood-brain barrier, neuroinflammation, neurodegeneration, dendritic spines, microglia
Introduction
The study of discreet processes underlying CNS function as they occur in real time in the living brain is a goal that has long fascinated neuroscientists. For many decades, the skull was a seemingly impenetrable vault, a black box preventing researchers from looking through it in the living mammal. The advent of two-photon laser scanning microscopy (TPLSM) enabled neuroscientists to record ongoing cellular processes in real time.1 The first applications of intravital imaging to study the CNS set the stage for an innovative field within neuroscience research.2–5 In vivo imaging in the CNS became possible when the first transgenic mice expressing fluorescent proteins in different cell types of the brain were generated.6 In recent years, the field has experienced an impressive growth by the introduction of novel imaging technologies, new surgical and technical methods and a wealth of sophisticated experimental approaches that have immensely expanded our insight on nervous system function in health and disease.7–9 The advantage of in vivo imaging is following the highly dynamic, time-dependent processes that are characteristic of CNS function in real time. The neurovascular unit, composed of cerebral blood vessels, glial cells and neurons, is at the center of precisely regulated and dynamic neuronal and vascular activities.10 The features of this dynamic interface of complex, multiple cell-cell interactions render it a vulnerable target in CNS pathology,10,11 and an ideal site for in vivo imaging studies.
In this perspective, we will focus on intravital imaging using TPLSM as an indispensable experimental tool for studying real time alterations in the functioning of the neurovascular unit. We will discuss why and how the results of in vivo imaging of the neurovascular unit could provide critical information regarding the pathogenesis of several CNS diseases, including stroke, MS and AD, and how this approach could reveal novel and much needed therapeutic avenues.
Studying the Neurovascular Unit
The neurovascular unit initially served as a conceptual framework to describe the relationship between neuronal activity and cerebral blood flow (CBF), a mechanism known as functional hyperaemia.10 Elevated neuronal activity is accompanied by increased blood supply, which accommodates higher neuronal metabolic demands and provides a means for the efflux of metabolic end products. The discovery that astrocytes, microglia, and recently also pericytes control vital functional processes in this tightly coupled, neuronal-CBF relationship expanded the perception of the neurovascular unit into one of a highly regulated apparatus functioning at a multicellular level.10
Imaging the elements of the neurovascular unit
The study of the neurovascular unit by histological means has identified the main cellular components and provided some crucial structural clarity into its functions.11 Real-time in vivo imaging is required to capture the time-dependent activities and dynamic cell-cell interactions that take place within the complex cerebral network. It is the merit of such elegant and sophisticated studies that, for example, intricate functional contacts between dendritic spines—the major site of excitatory synaptic inputs in the brain—and astrocytic endfeet have been revealed, showing that timely spine dynamics are dependent on interactions with astrocytes.12 In addition, the idea that intercellular signaling within the brain is solely a neuronal privilege lost credence following in vivo imaging studies revealing communication between astrocytes in the form of Ca2+ waves that parallel neuronal activity.8,13 While the belief of astrocytes acting as a mere glue substance without functional roles was challenged as early as the end of the 19th century,14 only recently in vivo imaging studies have provided strong support for the active role of astrocytes in neuronal health, stability and transmission.
Some of the earliest in vivo imaging studies in the mouse cortex provided the first visual demonstrations of how complex and to this day unresolved processes as memory formation and learning could be taking place at the cellular level in the mammalian neocortex by following the same dendritic branches in the cortex over periods of time ranging from minutes to months.15,16 These studies identified the stochastic nature of new spine formation and revealed that some spines remain stable for prolonged periods of time while others exhibit a high turnover rate.15,16 By reimaging the exact same spines over time, neuronal activity was identified as a major regulator of spine turnover that can be modified by peripheral sensory stimulation of the corresponding input organs.17 Similarly, in vivo imaging of microglial behavior in the intact cortex revealed that the resident immune cells of the CNS are far from “resting” at steady-state, but rather are continuously surveying the physiological brain parenchyma.18,19 In vivo imaging studies also detailed the nature of the physical interactions between microglial processes and dendritic spines in the brain of mice where both neurons and microglia were fluorescently labeled. These studies suggested that microglial process-dynamics might play a central role in regulating the fate of dendritic spines in the adult as well as the developing brain.20–22
Diseases of the Neurovascular Unit
As much as the tightly regulated functions of the neurovascular unit lie in the strictly regulated interplay between its cellular constituents, its vulnerability shares the very same origins. Failure of even one component can affect the system as a whole and can lead to overall malfunctioning. Stroke, MS and AD are neurological disorders that strongly impact the integrity of the neurovascular unit, whereas accumulating evidence suggests that neurovascular breakdown is an early pathological step, possibly crucial for disease initiation and progression.10,23 In light of these findings, there is an urgent need for tools that enable the study of the neurovascular unit in the context of disease, which could lead to the discovery of robust therapies and early disease biomarkers.24 This section focuses on the impact of intravital imaging on deciphering the role of the neurovascular unit in stroke, MS and AD.
Stroke
Because cerebrovascular injury initiates cerebral impairment within a confined, chosen area of the brain, in vivo stroke models provide valuable tools for understanding the consequences of vascular disruption on cerebral functioning. Mouse and rat models of middle cerebral artery occlusion (MCAO) are based on the generation of an ischemic core causing neural cell loss as a result of mechanical vessel blockage, thereby mimicking the pathophysiological state as seen in stroke patients. In vivo imaging is a well-established method to measure subtle as well as large changes in CBF both in physiological and pathophysiological conditions.25 Intravascular administration of fluorescently labeled dyes allows for visualization of blood vessels and red blood cells, the latter appearing as dark cellular shapes within a bright plasma background. This is the most commonly used approach to quantify CBF (Table 1), whereby the number of red blood cells passing through a given length of vessel within a specified time window functions as a measure of blood flow.25 Altered CBF is a key phenomenon in MCAO which is partly the result of collapse of the lumenal space of microvessels followed by reorganization of the microvasculature in the stroke-affected area.26 Another study employing in vivo imaging in combination with a fluorescent probe that detects activated factor XIII, revealed thrombotic events correlating with fibrin deposition within 1 to 24 h after MCAO, indicating early and time-dependent compromise of the blood-brain barrier (BBB) in stroke.27 These studies exemplify how MCAO combined with intravital imaging has provided valuable understanding of the consequences of stroke on the cerebrovasculature and beyond.
Table 1.
In vivo imaging tools for the study of the neurovascular unit
| Diseases | ||||
|---|---|---|---|---|
| Cell type | Imaging tool | Stroke | EAE | AD |
| Astrocytes | Sulforhodamine-101 dye25 Ca2+ sensitive dyes13,34 GFAPGFP mouse73 |
Ischemia-induced astrocytic Ca2+ elevation13,34 Spreading depression increases astrocytic Ca2+ 34 |
Astrocytic hypertrophy in the spinal cord74 | Increased astrocytic [Ca2+] affects cerebrovascular functioning13 |
| Microglia | CX3CR1GFP mouse75 | Microglial involvement in axonal degeneration46 | Microglial CX3CR1 regulates Aβ phagocytosis65 | |
| Pericytes | Fluorescent dextran-conjugated dyes76 αSMA-RFPcherry mouse77 |
Modulation of capillary blood flow by pericytes32 | Pericyte-facilitated blood-to-brain migration of neutrophils78 | Modulation of capillary blood flow by pericytes76 |
| Vasculature | Fluorescent dextran dyes76 Tie2GFP mouse79 |
Extravasation of fibrinogen over the compromised BBB 1–24 h after MCAO27 | T cell dynamics at the cerebrovasculature45,47 | Instability of vascular tone13 |
| Neurons | Thy1YFP mouse80 | Ischemia-induced dendrite remodelling and spine loss36,40,41 | Focal axonal lesions46 | Aβ-mediated dendrite and spine loss2,15 |
Reporter mice and fluorescent dyes have been developed for specific detection of components of the neurovascular unit. Examples of key in vivo imaging applications for the study of astrocytes, microglia, pericytes, vasculature and neurons in neurodegenerative and neuroimmune diseases are shown.
MCAO usually affects a very extensive area of the brain similar to a major artery occlusion in humans. However, there are also other, much more confined and probably more common incidents that result from the infarction of smaller diameter vessels or capillaries in humans. Using an in vivo stroke model that closely recapitulates such micro-occlusions, Lam and colleagues28 combined the infusion of fluorescently-labeled micro-emboli with in vivo two-photon microscopy to induce and study the dynamics of microvascular remodelling following stroke. This study revealed another highly unanticipated function within the neurovascular unit, whereby reorganization of the endothelial cells of the vascular wall resulted in extravasation of the emboli and restoration of the blood flow.28 Furthermore, the extravasated emboli were taken up by neighboring microglia,28 demonstrating a unique self-repair mechanism of the CNS that involves interactions between two cellular components working together at the neurovascular unit to restore blood circulation and prevent neuronal damage.
In vivo imaging of the pathological cascade downstream of the initial vascular insult has substantiated and clarified cellular events that occur in stroke, and importantly, are consistent with results of earlier histological approaches. Elegant electron microscopy work by Tagami and colleagues29 revealed stroke-induced degeneration of the enigmatic pericytes. In vivo imaging studies showed that stroke alters pericyte morphology, causing weakening of the BBB and entry of peripheral immune components including inflammatory T cells in the brain.30,31 Unexpectedly, in vivo imaging showed that pericytes can contribute to stroke pathogenesis by intensifying cerebral ischemia via extensive microvascular constriction, leading to far-reaching and lasting reductions in CBF.32
Similar to pericytes, astrocytes and recently also microglia were shown to negatively affect CBF following stroke.33 This unexpected contributing role for astrocytes in stroke pathology occurs via a mechanism called spreading depression.34 Spreading depression is characterized by a temporary inhibition of neuronal activity following neurological insult, such as stroke and traumatic brain injury. This inhibition spreads across parts of the cerebral cortex and originates from increased propagation of Ca2+ waves within astrocytes and neurons. The in vivo imaging studies of Chuquet and colleagues34 brought to light a synergy that exists between astrocytic and neuronal Ca2+ waves that leads to the detrimental halting of capillary CBF and results in ischemia. Astrocytes mediate signals between neurons and the cerebral vasculature to guarantee that elevated neuronal activity is paralleled by increased CBF, which facilitates the increased demand in oxygen supply. A disturbance in this tightly regulated mechanism can thus have dramatic effects on cerebral functioning.
The chronic occurrence of intracellular Ca2+ waves in neurons, as imaged in spreading depression, is not only detrimental for proper CBF, but affects neuronal integrity as well. Neuronal glutamate release is controlled by the intracellular Ca2+ concentration [Ca2+], and excessive increases in the intracellular Ca2+ pool causes neurons to release pathologic levels of glutamate, leading to neuronal swelling and breakdown.35 Intravital imaging has provided further convincing evidence that stroke-induced ischemia alters dendrite morphology and affects dendritic spine dynamics within a time frame ranging from minutes to hours after the onset of CBF blockage.36–38 These imaging studies also yielded clarity into the mechanisms of neuronal plasticity in the recovery period after a stroke insult.36 Such knowledge is essential for the advancement of stroke therapies, as they reveal the temporal response of the neurovascular unit and the time points at which therapeutic interventions might have a protective effect. With no adequate drug therapies available, the application of intravital imaging to the study of stroke pathogenesis could help to inform treatment strategies for this debilitating neurological disorder.
Multiple Sclerosis
The origins of MS, a devastating and often disabling neuroinflammatory disease, remain unknown. Some of the most prominent pathophysiological findings identified mostly in brain biopsies of MS patients are the formation of lesions in the white matter, characterized by extensive loss of myelin, axonal damage and inflammation, involving both resident and infiltrating immune cells in the CNS.39 In addition to T cells, microglia,40 peripheral monocytes41 and the blood protein fibrinogen42,43 contribute to the onset and progression of neuroinflammation. Although microglia and the disruption of the BBB are considered early events in MS pathology,44 the sequence of events that lead to neuroinflammatory disease remains poorly understood. Repetitive in vivo imaging using two-photon microscopy in the animal model of MS—experimental autoimmune encephalomyelitis (EAE)—is uniquely positioned to answer these questions, since it allows the study of disease in the same animal over time. Indeed, in vivo imaging studies of T cell migration in EAE demonstrated formerly unknown interactions between autoimmune effector T cells and the cerebrovasculature. Such research, for example, revealed that T cells, after their attachment to the endothelium, explore the lumenal vessel wall in a bidirectional manner for multiple days before entering the CNS.45 These imaging experiments have provided novel understanding of the migratory paths of T cells into the CNS and have expanded our understanding regarding how the physiological separation of the peripheral blood stream and the CNS is breached in neuroinflammation.
Intravital imaging has also increased our understanding of axonal pathology in neuroinflammation. Indeed, with the aid of in vivo two-photon imaging, Siffrin et al.44 found an intriguing direct contact between myelin-specific Th17 and neuronal cells that was associated with extensive axonal damage. T cells induced changes in neuronal Ca2+ levels, suggesting that they participate in early stages of axonal damage. Intravital imaging was crucial in identifying “focal axonal degeneration” as a novel feature of immune-mediated axonal damage.46 Nikic and colleagues,46 using in vivo two-photon microscopy showed that axonal abnormalities start with focal swellings, and while some axons degenerate, in others swellings are reversible resulting in spontaneous recovery. Focal axonal degeneration was also observed in samples from human MS lesions.46 Although these pioneering studies have identified novel mechanisms for T cells and axons, the contribution of glial cells and BBB leakage in the dynamic remodelling of the neurovascular interface in neuroinflammatory disease remains elusive. Technological advances of stable imaging in the spinal cord will further facilitate studies of repetitive imaging of multiple cell types within myelinated areas in anatomical areas accessible by two-photon microscopy.47–49 The availability of reporter transgenic mice and specific dyes for glia, T cells and BBB leakage (Table 1), makes possible the simultaneous labeling of different components of the neurovascular unit for the study of neuroinflammatory disease.
Alzheimer Disease (AD)
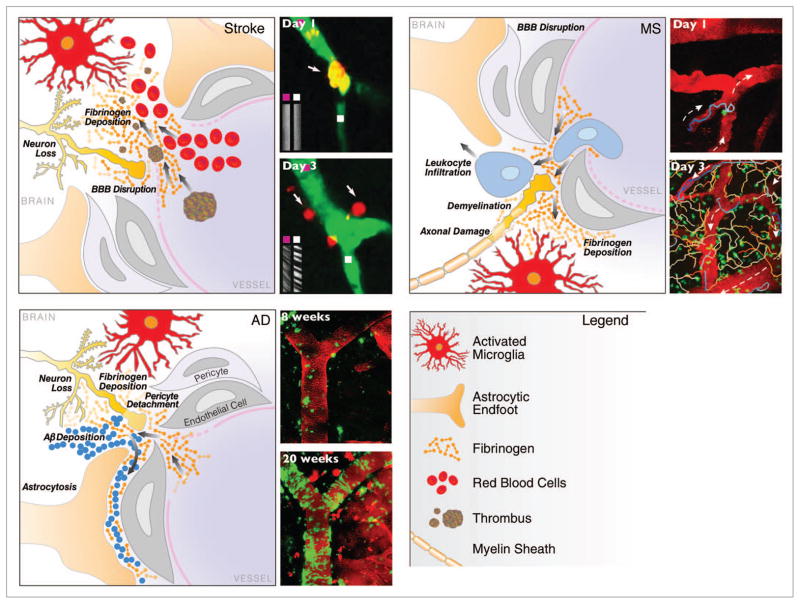
Although AD has been traditionally considered a mere neuronal disease, cutting-edge studies have countered this view by identifying that breakdown of the neurovascular unit is central to the onset and progression of AD (Fig. 1).10 One of the classic views in AD research is described by the amyloid hypothesis, according to which the pathological accumulation of amyloid β (Aβ)—a cellular cleavage product of the amyloid precursor protein APP—leads to the gradual formation of Aβ plaques over time,10 and thus creating a toxic environment that causes neurodegeneration in the AD brain. Recently, the validity of the amyloid hypothesis has been strongly questioned and whether Aβ is indeed an instigator or an epiphenomenon in AD pathology remains a subject of active debate.50 Nevertheless, the deposition of Aβ plaques occurs both in the brain parenchyma and on cerebral vessels, with the latter leading to cerebral amyloid angiopathy and causing a plethora of vessel pathologies including BBB breakdown, vascular stenoses and decreased CBF.23 Aβ can therefore be regarded as one of the key instigators of neurovascular unit impairment in AD (Fig. 1). Excellent fluorescent Aβ dyes have been developed that make use of the characteristic fibrillar and β-sheet structure of Aβ within plaques and show highly specific binding. These dyes have allowed the application of intravital imaging to studies aimed at understanding the kinetics of Aβ deposition and evaluate the efficacy of anti-Aβ therapies in vivo.51–53 While in vivo imaging studies in AD animal models agree on the dynamics of individual plaque growth, different results have been reported on the growth kinetics of fibrillar amyloid deposits, ranging from within a day,54 to gradually over weeks55 to months.56 Deposition kinetics of vascular Aβ have been shown to be strongly affected by the interaction of Aβ with the blood protein fibrinogen.57 This interaction leads to both a decrease in lysis of fibrin clots and impaired vascular Aβ clearance inducing cerebral amyloid angiopathy, vascular Aβ deposits affecting the physiology of the cerebrovasculature.57 The reciprocal character of the Aβ-fibrinogen interaction in AD was further supported by the results of in vivo imaging in a transgenic AD mouse model that demonstrated decreased fibrinolysis with blood clot formation paralleled by impaired CBF and cognitive functioning.57 Such studies are demonstrative of the close relationship that exists between neuronal health and an intact, properly functioning neurovascular unit.
Figure 1.
In vivo imaging of the neurovascular unit in stroke, MS and AD. Schematic representation of the dynamic alterations at the neurovascular unit in different neurologic diseases. Despite different origins, neuropathology and clinical disease progression, there are striking similarities in the cellular processes underlying breakdown of the neurovascular unit. Leakage of the BBB with entry of blood proteins, activation of microglia, astrocyte endfeet retraction, pericyte detachment, neuronal and axonal changes are key elements of cerebrovascular dysfunction. Repetitive in vivo imaging shows extravasation of micro-emboli causing alterations in CBF and restructuring of the microvasculature in stroke,28 migratory paths of T cells after their attachment to the leptomeningeal vessels in a mouse model of MS,45 and gradual progression of cerebral amyloid angiopathy over weeks as captured by time-lapse in vivo imaging in a transgenic mouse model of AD.54 Images reproduced with permission.
A more careful evaluation of the relationship between amyloid plaque growth and the degree of neuritic dystrophy in their proximity seems to confirm a causal relationship between the two, however it would appear that the kinetics of amyloid deposition is a more critical determinant of neurotoxicity, possibly more so than the extent of the plaque growth itself.58 Microglial activation influences Aβ plaque deposition by causing chronic cerebral inflammation, neuron loss and BBB breakdown. An example is the unexpected discovery that the microglial CX3CR1 chemokine receptor directly affects neuronal viability in mouse models of AD.59 Another recent study by Liu and colleagues60 showed that microglia control brain Aβ concentrations and hence the degree of Aβ plaque formation in AD mouse models, opening the road for a potential new drug target in microglia. In vivo imaging studies in transgenic mouse lines with microglia-specific fluorescent protein expression (Table 1) have thus elucidated how microglial activation contributes to AD-mediated cerebral inflammation. In addition to microglia, astrocyte activation is also a hallmark of AD pathology, especially around Aβ plaques, as first discovered by traditional histological approaches.10 However, it had not been clearly established whether their activation is linked to detrimental effects on surrounding tissue. In some studies, astrogliosis was found to be indicative of Aβ plaque clearance.61 Yet, Aβ-induced astrogliosis is also known to significantly impair astrocytic functions vital to neuronal integrity and CBF—including glucose uptake and lactate release—by triggering the retraction of astrocyte endfeet from Aβ-affected blood vessels, thereby disturbing neurovascular coupling.23 To date, in vivo imaging of astrocytes in AD models is focused mainly on understanding the function of these cells in the vicinity of Aβ plaques. These studies are based on imaging the intra- and intercellular astrocytic Ca2+ waves key to neuron-astrocyte communication, and have provided knowledge of the astrocytic activation state as exemplified by increased intracellular [Ca2+].8,13 The in vivo imaging techniques that are now available (Table 1) enables a much-needed and better understanding of the fate and role of astrocytes in AD.
The extensive failure of glial cell and vascular function within the neurovascular unit is increasingly regarded as an initiating or at least largely contributing factor in AD-related neurodegeneration. Cognitive decline as a result of neurodegeneration is one of the symptomatic characteristics of AD. The availability of transgenic mouse lines with neuron-specific fluorescent protein expression (Table 1) has allowed the study of the relationship between neuronal morphology/function and cognitive performance in vivo. Chronic loss of dendritic spines—the functional postsynaptic units critical for plasticity and ultimately cognitive function—and dendrites in the vicinity of Aβ plaques has been one of the first discoveries revealed by in vivo imaging.62,63
These studies also demonstrated that a gradient exists in the degree of Aβ plaque toxicity on dendritic spine dynamics, whereby neuronal dendrites in close proximity to Aβ plaques (< 15 μm distance) display significantly less spine density than ones farther away.64 Others have found similar results,65 although the toxicity-to-distance ratio can differ significantly between the various transgenic AD mouse models. Besides dendritic spine loss, observations of neurite retraction and abnormal neurite morphology, including swelling and fragmentation in relation to Aβ plaque growth over time, have also come to light with intravital imaging.63,65 In vivo imaging is not only a powerful tool to study morphologic changes over time, but can also assess functional activity within the CNS. Combining structural and functional in vivo imaging, Busche and colleagues66 discovered that neurons in close proximity to Aβ plaques showed significantly increased activity. These findings offer crucial clues to help explain the phenomenon of altered neuronal firing patterns that can disturb neuronal function in AD.
Concluding Remarks
Research in neuroimmune and neurodegenerative disorders, including stroke, MS and AD has advanced tremendously our understanding of the mechanisms underlying disease pathology, onset and progression, as well as revealing potential drug targets. Unfortunately, the number of breakthroughs in actual drug discovery to treat these devastating diseases does not parallel the number of mechanistic findings of disease pathogenesis, perhaps due to the inherently costly and time-consuming features of CNS drug discovery. Intravital imaging has the potential to enhance translational efforts as it reveals novel and unanticipated features of disease pathogenesis and is instrumental in identifying the early events that trigger CNS disease.
Stroke and AD research in particular was traditionally approached from a mere neuron-centered point of view.67 Given that these diseases cause dramatic neuron loss and decreased neuronal connectivity such focus is understandable. However, a more unifying view is emerging in which neurons are no longer acting alone, but instead act together with their counterparts within the dynamic environment of the neurovascular unit.10 This view has evolved in large part thanks to the numerous intravital imaging studies discussed in this perspective, and will likely inform future CNS drug discovery efforts, as exemplified by the increasing use of in vivo imaging methods in drug discovery.68 The potential of in vivo imaging to bridge the void between the outcomes of basic research and their translation to the clinic is also demonstrated by the increasing use of clinical imaging techniques in basic in vivo research and vice versa. Functional magnetic resonance imaging (fMRI) and positron-emission tomography (PET) studies of CBF and metabolic changes as a result of CNS pathology are examples of this interchangeable, translational character of clinical and in vivo imaging bringing to light formerly unknown similarities in (mal)functioning of brain regions between humans and rodents.69
Stroke, MS and AD are characterized by unique molecular players, distinct disease onset and progression patterns, symptoms, and survival rates. Despite differences in mechanisms of disease pathogenesis, in vivo imaging studies in animal models have revealed compelling common alterations of the neurovascular unit in neuroimmune and neurodegenerative diseases (Fig. 1). In essence, the multifactorial character of these disorders and the substantial prevalence of co-morbidities could be explained by the similarities of neurovascular unit dysfunction. Furthering our knowledge and understanding of these intriguing parallels by employing a powerful tool like in vivo imaging could not only provide essential insight in these debilitating diseases, but perhaps also lead to the discovery of new targets for disease-modifying therapeutics or, ideally, cures. Given the contribution of the neurovascular unit to brain injury,70 future in vivo imaging studies could thus also be instrumental for the understanding of the mechanisms at play in traumatic brain injury.
Since the neurovascular unit is composed of a multitude of cellular processes, its study and understanding require a multiform research approach. Although intravital imaging is a powerful tool for the study of the neurovascular unit, there remain significant technical challenges. Intravital imaging can be beset by sub-optimal signal-to-noise ratio due to poor resolution as a consequence of light scattering within the imaged tissue.7 Moreover, brain movements caused by breathing patterns and beating of the heart can greatly disturb the quality of imaging, especially during continuous (time-lapse) imaging of small structures such as dendritic spines, microglial protrusions and astrocytic end-feet.9 Furthermore, while intravital imaging sheds exceptional light on structural and cellular changes, the molecular pathways and mechanisms underlying those alterations cannot be revealed using this technique. However, combining the power of in vivo imaging with other research tools can bring forth far-reaching knowledge of the neurovascular unit in CNS disease. The emerging advances in molecular imaging and the development of molecular probes, which can bind with high specificity to their target27,51,71,72 bear great promise toward achieving enhanced understanding within the field.
Acknowledgments
We thank Anna Lisa Lucido for editorial assistance. M.M. was supported by a Swiss National Science Foundation Postdoctoral Fellowship, D.D. by the Nancy Davis Young Investigator Award and K.A. by the Dana Foundation in Brain and Immunoimaging and NIH/NINDS grants NS051470, NS052189 and NS066361.
Abbreviations
- CNS
central nervous system
- MS
multiple sclerosis
- AD
Alzheimer disease
- TPLSM
two-photon laser scanning microscopy
- CBF
cerebral blood flow
- MCAO
middle cerebral artery occlusion
- BBB
blood-brain barrier
- EAE
experimental autoimmune encephalomyelitis
- Aβ
amyloid β
- fMRI
functional magnetic resonance imaging
- PET
positron-emission tomography
Footnotes
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
References
- 1.Denk W, Strickler JH, Webb WW. Two-photon laser scanning fluorescence microscopy. Science. 1990;248:73–6. doi: 10.1126/science.2321027. http://dx.doi.org/10.1126/science.2321027. [DOI] [PubMed] [Google Scholar]
- 2.Gan WB, Grutzendler J, Wong WT, Wong RO, Lichtman JW. Multicolor “DiOlistic” labeling of the nervous system using lipophilic dye combinations. Neuron. 2000;27:219–25. doi: 10.1016/s0896-6273(00)00031-3. http://dx.doi.org/10.1016/S0896-6273(00)00031-3. [DOI] [PubMed] [Google Scholar]
- 3.Helmchen F, Svoboda K, Denk W, Tank DW. In vivo dendritic calcium dynamics in deep-layer cortical pyramidal neurons. Nat Neurosci. 1999;2:989–96. doi: 10.1038/14788. http://dx.doi.org/10.1038/14788. [DOI] [PubMed] [Google Scholar]
- 4.Lendvai B, Stern EA, Chen B, Svoboda K. Experience-dependent plasticity of dendritic spines in the developing rat barrel cortex in vivo. Nature. 2000;404:876–81. doi: 10.1038/35009107. http://dx.doi.org/10.1038/35009107. [DOI] [PubMed] [Google Scholar]
- 5.Svoboda K, Helmchen F, Denk W, Tank DW. Spread of dendritic excitation in layer 2/3 pyramidal neurons in rat barrel cortex in vivo. Nat Neurosci. 1999;2:65–73. doi: 10.1038/4569. http://dx.doi.org/10.1038/4569. [DOI] [PubMed] [Google Scholar]
- 6.Misgeld T, Kerschensteiner M. In vivo imaging of the diseased nervous system. Nat Rev Neurosci. 2006;7:449–63. doi: 10.1038/nrn1905. http://dx.doi.org/10.1038/nrn1905. [DOI] [PubMed] [Google Scholar]
- 7.Hillman EM. Optical brain imaging in vivo: techniques and applications from animal to man. J Biomed Opt. 2007;12:051402. doi: 10.1117/1.2789693. http://dx.doi.org/10.1117/1.2789693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nimmerjahn A, Kirchhoff F, Kerr JN, Helmchen F. Sulforhodamine 101 as a specific marker of astroglia in the neocortex in vivo. Nat Methods. 2004;1:31–7. doi: 10.1038/nmeth706. http://dx.doi.org/10.1038/nmeth706. [DOI] [PubMed] [Google Scholar]
- 9.Yoder EJ. In Vivo Microscopy of the Mouse Brain Using Multiphoton Laser Scanning Techniques. Proc Soc Photo Opt Instrum Eng. 2002;4620:14–29. doi: 10.1117/12.470705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zlokovic BV. Neurodegeneration and the neurovascular unit. Nat Med. 2010;16:1370–1. doi: 10.1038/nm1210-1370. http://dx.doi.org/10.1038/nm1210-1370. [DOI] [PubMed] [Google Scholar]
- 11.Baeten KM, Akassoglou K. Extracellular matrix and matrix receptors in blood-brain barrier formation and stroke. Dev Neurobiol. 2011;71:1018–39. doi: 10.1002/dneu.20954. http://dx.doi.org/10.1002/dneu.20954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barker AJ, Ullian EM. Astrocytes and synaptic plasticity. Neuroscientist. 2010;16:40–50. doi: 10.1177/1073858409339215. http://dx.doi.org/10.1177/1073858409339215. [DOI] [PubMed] [Google Scholar]
- 13.Takano T, Han X, Deane R, Zlokovic B, Nedergaard M. Two-photon imaging of astrocytic Ca2+ signaling and the microvasculature in experimental mice models of Alzheimer’s disease. Ann N Y Acad Sci. 2007;1097:40–50. doi: 10.1196/annals.1379.004. http://dx.doi.org/10.1196/annals.1379.004. [DOI] [PubMed] [Google Scholar]
- 14.Andriezen WL. The neuroglia elements in the human brain. Br Med J. 1893;2:227–30. doi: 10.1136/bmj.2.1700.227. http://dx.doi.org/10.1136/bmj.2.1700.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Grutzendler J, Kasthuri N, Gan WB. Long-term dendritic spine stability in the adult cortex. Nature. 2002;420:812–6. doi: 10.1038/nature01276. http://dx.doi.org/10.1038/nature01276. [DOI] [PubMed] [Google Scholar]
- 16.Trachtenberg JT, Chen BE, Knott GW, Feng G, Sanes JR, Welker E, et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature. 2002;420:788–94. doi: 10.1038/nature01273. http://dx.doi.org/10.1038/nature01273. [DOI] [PubMed] [Google Scholar]
- 17.Bhatt DH, Zhang S, Gan WB. Dendritic spine dynamics. Annu Rev Physiol. 2009;71:261–82. doi: 10.1146/annurev.physiol.010908.163140. http://dx.doi.org/10.1146/annurev.physiol.010908.163140. [DOI] [PubMed] [Google Scholar]
- 18.Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, Jung S, et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci. 2005;8:752–8. doi: 10.1038/nn1472. http://dx.doi.org/10.1038/nn1472. [DOI] [PubMed] [Google Scholar]
- 19.Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science. 2005;308:1314–8. doi: 10.1126/science.1110647. http://dx.doi.org/10.1126/science.1110647. [DOI] [PubMed] [Google Scholar]
- 20.Schafer DP, Lehrman EK, Kautzman AG, Koyama R, Mardinly AR, Yamasaki R, et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron. 2012;74:691–705. doi: 10.1016/j.neuron.2012.03.026. http://dx.doi.org/10.1016/j.neuron.2012.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tremblay ME, Lowery RL, Majewska AK. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 2010;8:e1000527. doi: 10.1371/journal.pbio.1000527. http://dx.doi.org/10.1371/journal.pbio.1000527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wake H, Moorhouse AJ, Jinno S, Kohsaka S, Nabekura J. Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J Neurosci. 2009;29:3974–80. doi: 10.1523/JNEUROSCI.4363-08.2009. http://dx.doi.org/10.1523/JNEUROSCI.4363-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Merlini M, Meyer EP, Ulmann-Schuler A, Nitsch RM. Vascular β-amyloid and early astrocyte alterations impair cerebrovascular function and cerebral metabolism in transgenic arcAβ mice. Acta Neuropathol. 2011;122:293–311. doi: 10.1007/s00401-011-0834-y. http://dx.doi.org/10.1007/s00401-011-0834-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stanimirovic DB, Friedman A. Pathophysiology of the neurovascular unit: disease cause or consequence? J Cereb Blood Flow Metab. 2012;32:1207–21. doi: 10.1038/jcbfm.2012.25. http://dx.doi.org/10.1038/jcbfm.2012.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Helmchen F, Kleinfeld D. Chapter 10. In vivo measurements of blood flow and glial cell function with two-photon laser-scanning microscopy. Methods Enzymol. 2008;444:231–54. doi: 10.1016/S0076-6879(08)02810-3. http://dx.doi.org/10.1016/S0076-6879(08)02810-3. [DOI] [PubMed] [Google Scholar]
- 26.Masamoto K, Tomita Y, Toriumi H, Aoki I, Unekawa M, Takuwa H, et al. Repeated longitudinal in vivo imaging of neuroglio-vascular unit at the peripheral boundary of ischemia in mouse cerebral cortex. Neuroscience. 2012;212:190–200. doi: 10.1016/j.neuroscience.2012.03.034. http://dx.doi.org/10.1016/j.neuroscience.2012.03.034. [DOI] [PubMed] [Google Scholar]
- 27.Zhang ZG, Zhang L, Ding G, Jiang Q, Zhang RL, Zhang X, et al. A model of mini-embolic stroke offers measurements of the neurovascular unit response in the living mouse. Stroke. 2005;36:2701–4. doi: 10.1161/01.STR.0000190007.18897.e3. http://dx.doi.org/10.1161/01.STR.0000190007.18897.e3. [DOI] [PubMed] [Google Scholar]
- 28.Lam CK, Yoo T, Hiner B, Liu Z, Grutzendler J. Embolus extravasation is an alternative mechanism for cerebral microvascular recanalization. Nature. 2010;465:478–82. doi: 10.1038/nature09001. http://dx.doi.org/10.1038/nature09001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tagami M, Nara Y, Kubota A, Fujino H, Yamori Y. Ultrastructural changes in cerebral pericytes and astrocytes of stroke-prone spontaneously hypertensive rats. Stroke. 1990;21:1064–71. doi: 10.1161/01.str.21.7.1064. http://dx.doi.org/10.1161/01.STR.21.7.1064. [DOI] [PubMed] [Google Scholar]
- 30.Bell RD, Winkler EA, Sagare AP, Singh I, LaRue B, Deane R, et al. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron. 2010;68:409–27. doi: 10.1016/j.neuron.2010.09.043. http://dx.doi.org/10.1016/j.neuron.2010.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fumagalli S, Coles JA, Ejlerskov P, Ortolano F, Bushell TJ, Brewer JM, et al. In vivo real-time multiphoton imaging of T lymphocytes in the mouse brain after experimental stroke. Stroke. 2011;42:1429–36. doi: 10.1161/STROKEAHA.110.603704. http://dx.doi.org/10.1161/STROKEAHA.110.603704. [DOI] [PubMed] [Google Scholar]
- 32.Fernández-Klett F, Offenhauser N, Dirnagl U, Priller J, Lindauer U. Pericytes in capillaries are contractile in vivo, but arterioles mediate functional hyperemia in the mouse brain. Proc Natl Acad Sci U S A. 2010;107:22290–5. doi: 10.1073/pnas.1011321108. http://dx.doi.org/10.1073/pnas.1011321108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Masuda T, Croom D, Hida H, Kirov SA. Capillary blood flow around microglial somata determines dynamics of microglial processes in ischemic conditions. Glia. 2011;59:1744–53. doi: 10.1002/glia.21220. http://dx.doi.org/10.1002/glia.21220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chuquet J, Hollender L, Nimchinsky EA. High-resolution in vivo imaging of the neurovascular unit during spreading depression. J Neurosci. 2007;27:4036–44. doi: 10.1523/JNEUROSCI.0721-07.2007. http://dx.doi.org/10.1523/JNEUROSCI.0721-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tymianski M. Emerging mechanisms of disrupted cellular signaling in brain ischemia. Nat Neurosci. 2011;14:1369–73. doi: 10.1038/nn.2951. http://dx.doi.org/10.1038/nn.2951. [DOI] [PubMed] [Google Scholar]
- 36.Brown CE, Li P, Boyd JD, Delaney KR, Murphy TH. Extensive turnover of dendritic spines and vascular remodeling in cortical tissues recovering from stroke. J Neurosci. 2007;27:4101–9. doi: 10.1523/JNEUROSCI.4295-06.2007. http://dx.doi.org/10.1523/JNEUROSCI.4295-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Johnston DG, Denizet M, Mostany R, Portera-Cailliau C. Chronic In Vivo Imaging Shows No Evidence of Dendritic Plasticity or Functional Remapping in the Contralesional Cortex after Stroke. Cereb Cortex. 2012 doi: 10.1093/cercor/bhs092. In press; http://dx.doi.org/10.1093/cercor/bhs092. [DOI] [PMC free article] [PubMed]
- 38.Murphy TH, Li P, Betts K, Liu R. Two-photon imaging of stroke onset in vivo reveals that NMDA-receptor independent ischemic depolarization is the major cause of rapid reversible damage to dendrites and spines. J Neurosci. 2008;28:1756–72. doi: 10.1523/JNEUROSCI.5128-07.2008. http://dx.doi.org/10.1523/JNEUROSCI.5128-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lassmann H. Axonal and neuronal pathology in multiple sclerosis: what have we learnt from animal models. Exp Neurol. 2010;225:2–8. doi: 10.1016/j.expneurol.2009.10.009. http://dx.doi.org/10.1016/j.expneurol.2009.10.009. [DOI] [PubMed] [Google Scholar]
- 40.Heppner FL, Greter M, Marino D, Falsig J, Raivich G, Hövelmeyer N, et al. Experimental autoimmune encephalomyelitis repressed by microglial paralysis. Nat Med. 2005;11:146–52. doi: 10.1038/nm1177. http://dx.doi.org/10.1038/nm1177. [DOI] [PubMed] [Google Scholar]
- 41.Ajami B, Bennett JL, Krieger C, McNagny KM, Rossi FM. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci. 2011;14:1142–9. doi: 10.1038/nn.2887. http://dx.doi.org/10.1038/nn.2887. [DOI] [PubMed] [Google Scholar]
- 42.Adams RA, Bauer J, Flick MJ, Sikorski SL, Nuriel T, Lassmann H, et al. The fibrin-derived gamma377-395 peptide inhibits microglia activation and suppresses relapsing paralysis in central nervous system autoimmune disease. J Exp Med. 2007;204:571–82. doi: 10.1084/jem.20061931. http://dx.doi.org/10.1084/jem.20061931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Adams RA, Schachtrup C, Davalos D, Tsigelny I, Akassoglou K. Fibrinogen signal transduction as a mediator and therapeutic target in inflammation: lessons from multiple sclerosis. Curr Med Chem. 2007;14:2925–36. doi: 10.2174/092986707782360015. http://dx.doi.org/10.2174/092986707782360015. [DOI] [PubMed] [Google Scholar]
- 44.Siffrin V, Radbruch H, Glumm R, Niesner R, Paterka M, Herz J, et al. In vivo imaging of partially reversible th17 cell-induced neuronal dysfunction in the course of encephalomyelitis. Immunity. 2010;33:424–36. doi: 10.1016/j.immuni.2010.08.018. http://dx.doi.org/10.1016/j.immuni.2010.08.018. [DOI] [PubMed] [Google Scholar]
- 45.Bartholomäus I, Kawakami N, Odoardi F, Schläger C, Miljkovic D, Ellwart JW, et al. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature. 2009;462:94–8. doi: 10.1038/nature08478. http://dx.doi.org/10.1038/nature08478. [DOI] [PubMed] [Google Scholar]
- 46.Nikic I, Merkler D, Sorbara C, Brinkoetter M, Kreutzfeldt M, Bareyre FM, et al. A reversible form of axon damage in experimental autoimmune encephalomyelitis and multiple sclerosis. Nat Med. 2011;17:495–9. doi: 10.1038/nm.2324. http://dx.doi.org/10.1038/nm.2324. [DOI] [PubMed] [Google Scholar]
- 47.Davalos D, Lee JK, Smith WB, Brinkman B, Ellisman MH, Zheng B, et al. Stable in vivo imaging of densely populated glia, axons and blood vessels in the mouse spinal cord using two-photon microscopy. J Neurosci Methods. 2008;169:1–7. doi: 10.1016/j.jneumeth.2007.11.011. http://dx.doi.org/10.1016/j.jneumeth.2007.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Davalos D, Akassoglou K. In vivo imaging of the mouse spinal cord using two-photon microscopy. J Vis Exp. 2012;59:e2760. doi: 10.3791/2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kim JV, Jiang N, Tadokoro CE, Liu L, Ransohoff RM, Lafaille JJ, et al. Two-photon laser scanning microscopy imaging of intact spinal cord and cerebral cortex reveals requirement for CXCR6 and neuro-inflammation in immune cell infiltration of cortical injury sites. J Immunol Methods. 2010;352:89–100. doi: 10.1016/j.jim.2009.09.007. http://dx.doi.org/10.1016/j.jim.2009.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pimplikar SW. Reassessing the amyloid cascade hypothesis of Alzheimer’s disease. Int J Biochem Cell Biol. 2009;41:1261–8. doi: 10.1016/j.biocel.2008.12.015. http://dx.doi.org/10.1016/j.biocel.2008.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bacskai BJ, Kajdasz ST, Christie RH, Carter C, Games D, Seubert P, et al. Imaging of amyloid-beta deposits in brains of living mice permits direct observation of clearance of plaques with immunotherapy. Nat Med. 2001;7:369–72. doi: 10.1038/85525. http://dx.doi.org/10.1038/85525. [DOI] [PubMed] [Google Scholar]
- 52.Burgold S, Bittner T, Dorostkar MM, Kieser D, Fuhrmann M, Mitteregger G, et al. In vivo multiphoton imaging reveals gradual growth of newborn amyloid plaques over weeks. Acta Neuropathol. 2011;121:327–35. doi: 10.1007/s00401-010-0787-6. http://dx.doi.org/10.1007/s00401-010-0787-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Skoch J, Hickey GA, Kajdasz ST, Hyman BT, Bacskai BJ. In vivo imaging of amyloid-beta deposits in mouse brain with multiphoton microscopy. Methods Mol Biol. 2005;299:349–63. doi: 10.1385/1-59259-874-9:349. [DOI] [PubMed] [Google Scholar]
- 54.Dong J, Revilla-Sanchez R, Moss S, Haydon PG. Multiphoton in vivo imaging of amyloid in animal models of Alzheimer’s disease. Neuropharmacology. 2010;59:268–75. doi: 10.1016/j.neuropharm.2010.04.007. http://dx.doi.org/10.1016/j.neuropharm.2010.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yan P, Bero AW, Cirrito JR, Xiao Q, Hu X, Wang Y, et al. Characterizing the appearance and growth of amyloid plaques in APP/PS1 mice. J Neurosci. 2009;29:10706–14. doi: 10.1523/JNEUROSCI.2637-09.2009. http://dx.doi.org/10.1523/JNEUROSCI.2637-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hefendehl JK, Wegenast-Braun BM, Liebig C, Eicke D, Milford D, Calhoun ME, et al. Long-term in vivo imaging of β-amyloid plaque appearance and growth in a mouse model of cerebral β-amyloidosis. J Neurosci. 2011;31:624–9. doi: 10.1523/JNEUROSCI.5147-10.2011. http://dx.doi.org/10.1523/JNEUROSCI.5147-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cortes-Canteli M, Paul J, Norris EH, Bronstein R, Ahn HJ, Zamolodchikov D, et al. Fibrinogen and beta-amyloid association alters thrombosis and fibrinolysis: a possible contributing factor to Alzheimer’s disease. Neuron. 2010;66:695–709. doi: 10.1016/j.neuron.2010.05.014. http://dx.doi.org/10.1016/j.neuron.2010.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Condello C, Schain A, Grutzendler J. Multicolor timestamp reveals the dynamics and toxicity of amyloid deposition. Sci Rep. 2011;1:19. doi: 10.1038/srep00019. http://dx.doi.org/10.1038/srep00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fuhrmann M, Bittner T, Jung CK, Burgold S, Page RM, Mitteregger G, et al. Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer’s disease. Nat Neurosci. 2010;13:411–3. doi: 10.1038/nn.2511. http://dx.doi.org/10.1038/nn.2511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu Z, Condello C, Schain A, Harb R, Grutzendler J. CX3CR1 in microglia regulates brain amyloid deposition through selective protofibrillar amyloid-β phagocytosis. J Neurosci. 2010;30:17091–101. doi: 10.1523/JNEUROSCI.4403-10.2010. http://dx.doi.org/10.1523/JNEUROSCI.4403-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wyss-Coray T, Loike JD, Brionne TC, Lu E, Anankov R, Yan F, et al. Adult mouse astrocytes degrade amyloid-beta in vitro and in situ. Nat Med. 2003;9:453–7. doi: 10.1038/nm838. http://dx.doi.org/10.1038/nm838. [DOI] [PubMed] [Google Scholar]
- 62.Spires-Jones TL, Mielke ML, Rozkalne A, Meyer-Luehmann M, de Calignon A, Bacskai BJ, et al. Passive immunotherapy rapidly increases structural plasticity in a mouse model of Alzheimer disease. Neurobiol Dis. 2009;33:213–20. doi: 10.1016/j.nbd.2008.10.011. http://dx.doi.org/10.1016/j.nbd.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tsai J, Grutzendler J, Duff K, Gan WB. Fibrillar amyloid deposition leads to local synaptic abnormalities and breakage of neuronal branches. Nat Neurosci. 2004;7:1181–3. doi: 10.1038/nn1335. http://dx.doi.org/10.1038/nn1335. [DOI] [PubMed] [Google Scholar]
- 64.Spires TL, Meyer-Luehmann M, Stern EA, McLean PJ, Skoch J, Nguyen PT, et al. Dendritic spine abnormalities in amyloid precursor protein transgenic mice demonstrated by gene transfer and intravital multiphoton microscopy. J Neurosci. 2005;25:7278–87. doi: 10.1523/JNEUROSCI.1879-05.2005. http://dx.doi.org/10.1523/JNEUROSCI.1879-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bittner T, Fuhrmann M, Burgold S, Ochs SM, Hoffmann N, Mitteregger G, et al. Multiple events lead to dendritic spine loss in triple transgenic Alzheimer’s disease mice. PLoS One. 2010;5:e15477. doi: 10.1371/journal.pone.0015477. http://dx.doi.org/10.1371/journal.pone.0015477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Busche MA, Eichhoff G, Adelsberger H, Abramowski D, Wiederhold KH, Haass C, et al. Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer’s disease. Science. 2008;321:1686–9. doi: 10.1126/science.1162844. http://dx.doi.org/10.1126/science.1162844. [DOI] [PubMed] [Google Scholar]
- 67.de la Torre JC. Is Alzheimer’s disease a neurodegenerative or a vascular disorder? Data, dogma, and dialectics. Lancet Neurol. 2004;3:184–90. doi: 10.1016/S1474-4422(04)00683-0. http://dx.doi.org/10.1016/S1474-4422(04)00683-0. [DOI] [PubMed] [Google Scholar]
- 68.Bullen A. Microscopic imaging techniques for drug discovery. Nat Rev Drug Discov. 2008;7:54–67. doi: 10.1038/nrd2446. http://dx.doi.org/10.1038/nrd2446. [DOI] [PubMed] [Google Scholar]
- 69.Borsook D, Becerra L, Hargreaves R. A role for fMRI in optimizing CNS drug development. Nat Rev Drug Discov. 2006;5:411–24. doi: 10.1038/nrd2027. http://dx.doi.org/10.1038/nrd2027. [DOI] [PubMed] [Google Scholar]
- 70.Sashindranath M, Sales E, Daglas M, Freeman R, Samson AL, Cops EJ, et al. The tissue-type plasminogen activator-plasminogen activator inhibitor 1 complex promotes neurovascular injury in brain trauma: evidence from mice and humans. Brain. 2012;135:3251–64. doi: 10.1093/brain/aws178. http://dx.doi.org/10.1093/brain/aws178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lee DK, Nahrendorf M, Schellingerhout D, Kim DE. Will molecular optical imaging have clinically important roles in stroke management, and how? J Clin Neurol. 2010;6:10–8. doi: 10.3988/jcn.2010.6.1.10. http://dx.doi.org/10.3988/jcn.2010.6.1.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chen B, Friedman B, Whitney MA, Winkle JA, Lei IF, Olson ES, et al. Thrombin activity associated with neuronal damage during acute focal ischemia. J Neurosci. 2012;32:7622–31. doi: 10.1523/JNEUROSCI.0369-12.2012. http://dx.doi.org/10.1523/JNEUROSCI.0369-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nolte C, Matyash M, Pivneva T, Schipke CG, Ohlemeyer C, Hanisch UK, et al. GFAP promoter-controlled EGFP-expressing transgenic mice: a tool to visualize astrocytes and astrogliosis in living brain tissue. Glia. 2001;33:72–86. http://dx.doi.org/10.1002/1098-1136(20010101)33:1<72::AID-GLIA1007>3.0.CO;2-A. [PubMed] [Google Scholar]
- 74.Guo F, Maeda Y, Ma J, Delgado M, Sohn J, Miers L, et al. Macroglial plasticity and the origins of reactive astroglia in experimental autoimmune encephalomyelitis. J Neurosci. 2011;31:11914–28. doi: 10.1523/JNEUROSCI.1759-11.2011. http://dx.doi.org/10.1523/JNEUROSCI.1759-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jung S, Aliberti J, Graemmel P, Sunshine MJ, Kreutzberg GW, Sher A, et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol Cell Biol. 2000;20:4106–14. doi: 10.1128/mcb.20.11.4106-4114.2000. http://dx.doi.org/10.1128/MCB.20.11.4106-4114.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hirase H, Creso J, Singleton M, Barthó P, Buzsáki G. Two-photon imaging of brain pericytes in vivo using dextran-conjugated dyes. Glia. 2004;46:95–100. doi: 10.1002/glia.10295. http://dx.doi.org/10.1002/glia.10295. [DOI] [PubMed] [Google Scholar]
- 77.Yokota T, Kawakami Y, Nagai Y, Ma JX, Tsai JY, Kincade PW, et al. Bone marrow lacks a transplantable progenitor for smooth muscle type alpha-actin-expressing cells. Stem Cells. 2006;24:13–22. doi: 10.1634/stemcells.2004-0346. http://dx.doi.org/10.1634/stemcells.2004-0346. [DOI] [PubMed] [Google Scholar]
- 78.Proebstl D, Voisin MB, Woodfin A, Whiteford J, D’Acquisto F, Jones GE, et al. Pericytes support neutrophil subendothelial cell crawling and breaching of venular walls in vivo. J Exp Med. 2012;209:1219–34. doi: 10.1084/jem.20111622. http://dx.doi.org/10.1084/jem.20111622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Motoike T, Loughna S, Perens E, Roman BL, Liao W, Chau TC, et al. Universal GFP reporter for the study of vascular development. Genesis. 2000;28:75–81. doi: 10.1002/1526-968x(200010)28:2<75::aid-gene50>3.0.co;2-s. http://dx.doi.org/10.1002/1526-968X(200010)28:2<75::AID-GENE50>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 80.Feng G, Mellor RH, Bernstein M, Keller-Peck C, Nguyen QT, Wallace M, et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron. 2000;28:41–51. doi: 10.1016/s0896-6273(00)00084-2. http://dx.doi.org/10.1016/S0896-6273(00)00084-2. [DOI] [PubMed] [Google Scholar]