Abstract
The macrophage migration inhibition test was applied to the study of delayed hypersensitivity in mice vaccinated intravenously with oil-treated cell walls of Mycobacterium bovis BCG. Migration inhibition of peritoneal exudate cells from sensitized mice was demonstrated directly upon incubation of the cells with purified protein derivative, but indicator cells such as normal peritoneal cells had to be included to demonstrate migration and migration inhibition with sensitized lung cells. Inhibition of migration induced by mouse cells was greatest 3 to 4 weeks after sensitization but was still considerable after 11 weeks. The migration inhibitory factor (MIF) was not detected in cells freshly isolated from sensitized mice but was released into the supernatant fluid when cells were incubated with purified protein derivative for 24 hr at 37 C in a tissue culture system. Production of MIF was inhibited by actinomycin D and puromycin. MIF was nondialyzable, resistant to heating at 56 C for 1 hr, and of a lower molecular weight than mouse gamma globulin. All data indicated that migration inhibition induced by cells from cell wall-vaccinated mice was very similar to that caused by guinea pig lymphocytes.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- AL ASKARI S., DAVID J. R., LAWRENCE H. S., THOMAS L. IN VITRO STUDIES ON HOMOGRAFT SENSITIVITY. Nature. 1965 Feb 27;205:916–917. doi: 10.1038/205916a0. [DOI] [PubMed] [Google Scholar]
- Amos H. E., Gurner B. W., Olds R. J., Coombs R. R. Passive sensitization of tissue cells. II. Ability of cytophilic antibody to render the migration of guinea pig peritoneal exudate cells inhibitable by antigen. Int Arch Allergy Appl Immunol. 1967;32(5):496–505. [PubMed] [Google Scholar]
- Anacker R. L., Barclay W. R., Brehmer W., Goode G., List R. H., Ribi E., Tarmina D. F. Effectiveness of cell walls of Mycobacterium bovis strain BCG administered by various routes and in different adjuvants in protecting mice against airborne infection with Mycobacterium tuberculosis strain H37Rv. Am Rev Respir Dis. 1969 Feb;99(2):242–248. doi: 10.1164/arrd.1969.99.2.242. [DOI] [PubMed] [Google Scholar]
- Anacker R. L., Ribi E., Tarmina D. F., Fadness L., Mann R. E. Relationship of footpad sensitivity to purified protein derivatives and resistance to airborne infection with Mycobacterium tuberculosis of mice vaccinated with mycobacterial cell walls. J Bacteriol. 1969 Oct;100(1):51–57. doi: 10.1128/jb.100.1.51-57.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BOYDEN S. V. The adsorption of proteins on erythrocytes treated with tannic acid and subsequent hemagglutination by antiprotein sera. J Exp Med. 1951 Feb;93(2):107–120. doi: 10.1084/jem.93.2.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barclay W. R., Anacker R., Brehmer W., Ribi E. Effects of oil-treated mycobacterial cell walls on the organs of mice. J Bacteriol. 1967 Nov;94(5):1736–1745. doi: 10.1128/jb.94.5.1736-1745.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartfeld H., Atoynatan T., Kelly R. Delayed hypersensitivity studies with a modified capillary migration culture system. Transplantation. 1969 Apr;7(4):242–245. doi: 10.1097/00007890-196904000-00002. [DOI] [PubMed] [Google Scholar]
- Bennett B., Bloom B. R. Reactions in vivo and in vitro produced by a soluble substance associated with delayed-type hypersensitivity. Proc Natl Acad Sci U S A. 1968 Mar;59(3):756–762. doi: 10.1073/pnas.59.3.756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloom B. R., Bennett B. Mechanism of a reaction in vitro associated with delayed-type hypersensitivity. Science. 1966 Jul 1;153(3731):80–82. doi: 10.1126/science.153.3731.80. [DOI] [PubMed] [Google Scholar]
- Brehmer W., Anacker R. L., Ribi E. Immunogenicity of cell walls from various mycobacteria against airborne tuberculosis in mice. J Bacteriol. 1968 Jun;95(6):2000–2004. doi: 10.1128/jb.95.6.2000-2004.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins F. M., Mackaness G. B. Delayed hypersensitivity and arthus reactivity in relation to host resistance in salmonella-infected mice. J Immunol. 1968 Nov;101(5):830–845. [PubMed] [Google Scholar]
- DAVID J. R., AL-ASKARI S., LAWRENCE H. S., THOMAS L. DELAYED HYPERSENSITIVITY IN VITRO. I. THE SPECIFICITY OF INHIBITION OF CELL MIGRATION BY ANTIGENS. J Immunol. 1964 Aug;93:264–273. [PubMed] [Google Scholar]
- DAVID J. R., LAWRENCE H. S., THOMAL L. DELAYED HYPERSENSITIVITY IN VITRO. II. EFFECT OF SENSITIVE CELLS ON NORMAL CELLS IN THE PRESENCE OF ANTIGEN. J Immunol. 1964 Aug;93:274–278. [PubMed] [Google Scholar]
- David J. R. Cell-associated hypersensitivity studied in vitro. Cancer Res. 1968 Jul;28(7):1387–1391. [PubMed] [Google Scholar]
- David J. R. Delayed hypersensitivity in vitro: its mediation by cell-free substances formed by lymphoid cell-antigen interaction. Proc Natl Acad Sci U S A. 1966 Jul;56(1):72–77. doi: 10.1073/pnas.56.1.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David J. R. Suppression of delayed hypersensitivity in vitro by inhibition of protein synthesis. J Exp Med. 1965 Dec 1;122(6):1125–1134. doi: 10.1084/jem.122.6.1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferraresi R. W., Dedrick C. T., Raffel S., Goihman-Yahr M. Studies of the macrophage inhibition test. I. Comparison of the skin and cell migration reactions during the course of development of delayed hypersensitivity. J Immunol. 1969 Apr;102(4):852–858. [PubMed] [Google Scholar]
- GEORGE M., VAUGHAN J. H. In vitro cell migration as a model for delayed hypersensitivity. Proc Soc Exp Biol Med. 1962 Nov;111:514–521. doi: 10.3181/00379727-111-27841. [DOI] [PubMed] [Google Scholar]
- Heise E. R., Han S., Weiser R. S. In vitro studies on the mechanism of macrophage migration inhibition in tuberculin sensitivity. J Immunol. 1968 Nov;101(5):1004–1015. [PubMed] [Google Scholar]
- Holm G., Perlmann P. Cytotoxic potential of stimulated human lymphocytes. J Exp Med. 1967 Apr 1;125(4):721–736. doi: 10.1084/jem.125.4.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MACKANESS G. B. Cellular resistance to infection. J Exp Med. 1962 Sep 1;116:381–406. doi: 10.1084/jem.116.3.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MACKANESS G. B. THE IMMUNOLOGICAL BASIS OF ACQUIRED CELLULAR RESISTANCE. J Exp Med. 1964 Jul 1;120:105–120. doi: 10.1084/jem.120.1.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackaness G. B. The influence of immunologically committed lymphoid cells on macrophage activity in vivo. J Exp Med. 1969 May 1;129(5):973–992. doi: 10.1084/jem.129.5.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribi E., Anacker R. L., Brehmer W., Goode G., Larson C. L., List R. H., Milner K. C., Wicht W. C. Factors influencing protection against experimental tuberculosis in mice by heat-stable cell wall vaccines. J Bacteriol. 1966 Oct;92(4):869–879. doi: 10.1128/jb.92.4.869-879.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCHEIDEGGER J. J. Une micro-méthode de l'immuno-electrophorèse. Int Arch Allergy Appl Immunol. 1955;7(2):103–110. [PubMed] [Google Scholar]
- Ward P. A., Remold H. G., David J. R. Leukotactic factor produced by sensitized lymphocytes. Science. 1969 Mar 7;163(3871):1079–1081. doi: 10.1126/science.163.3871.1079. [DOI] [PubMed] [Google Scholar]
- YOUMANS G. P., YOUMANS A. S. AN ACUTE PULMONARY GRANULOMATOUS RESPONSE IN MICE PRODUCED BY MYCOBACTERIAL CELLS AND ITS RELATION TO INCREASED RESISTANCE AND INCREASED SUSCEPTIBILITY TO EXPERIMENTAL TUBERCULOUS INFECTION. J Infect Dis. 1964 Apr;114:135–151. doi: 10.1093/infdis/114.2.135. [DOI] [PubMed] [Google Scholar]
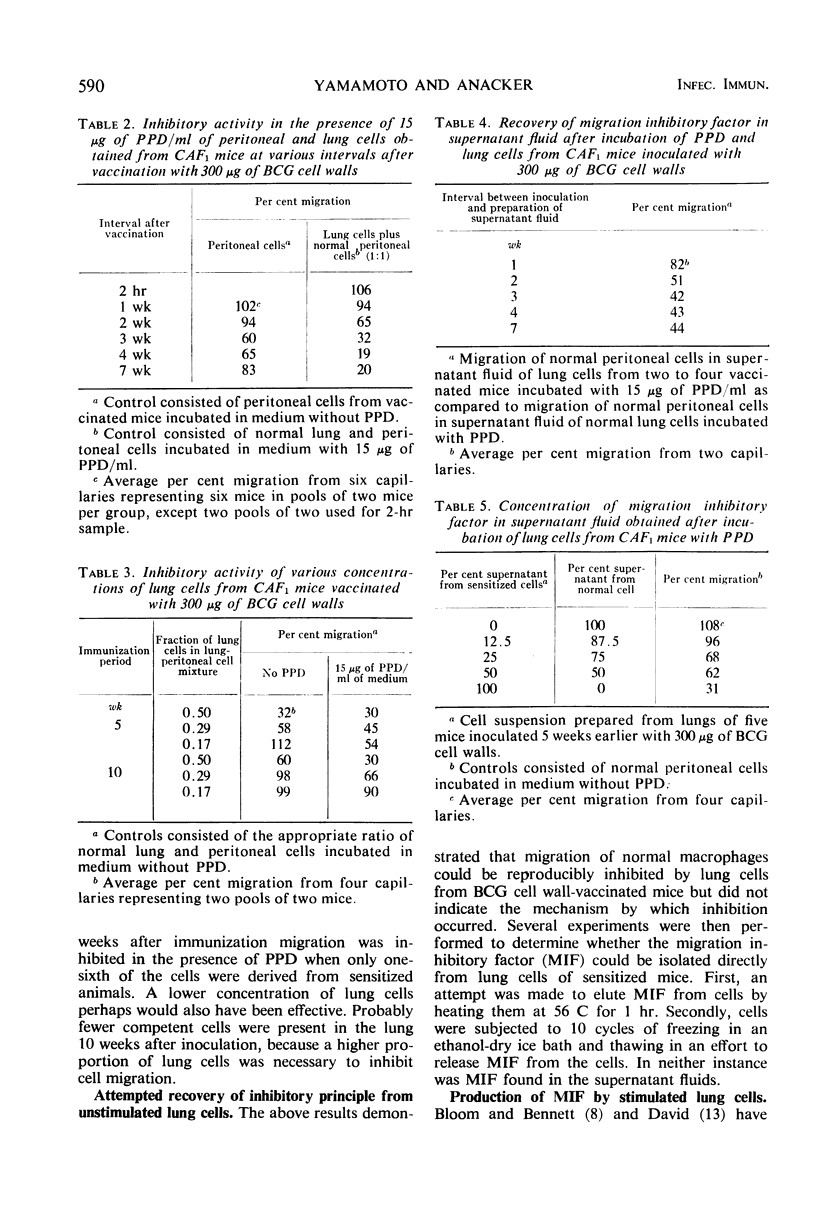
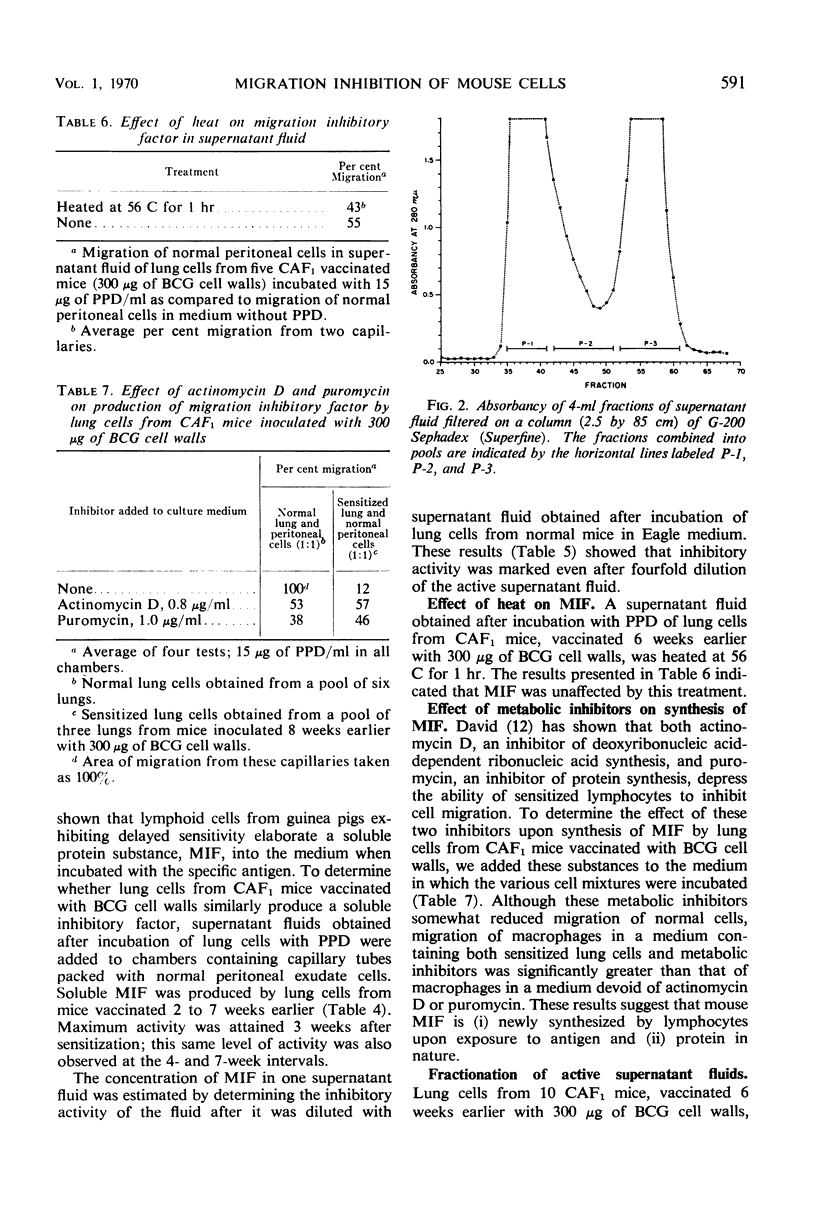
- Yamamoto K., Anacker R. L., Ribi E. Macrophage Migration Inhibition Studies with Cells from Mice Vaccinated with Cell Walls of Mycobacterium bovis BCG: Relationship Between Inhibitory Activity of Lung Cells and Resistance to Airborne Challenge with Mycobacterium tuberculosis H37Rv. Infect Immun. 1970 Jun;1(6):595–599. doi: 10.1128/iai.1.6.595-599.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youmans G. P., Youmans A. S. Allergenicity of mycobacterial ribosomal and ribonucleic acid preparations in mice and guinea pigs. J Bacteriol. 1969 Jan;97(1):134–139. doi: 10.1128/jb.97.1.134-139.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]