Abstract
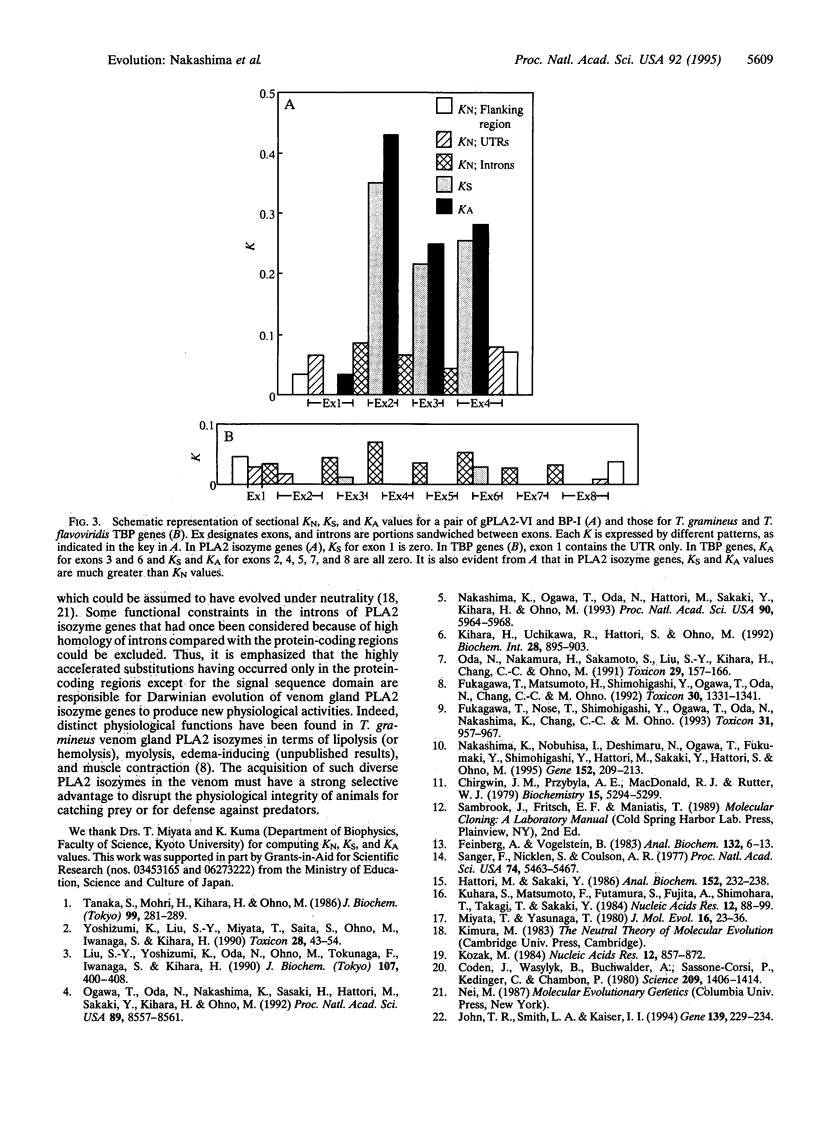
The nucleotide sequences of four genes encoding Trimeresurus gramineus (green habu snake, crotalinae) venom gland phospholipase A2 (PLA2; phosphatidylcholine 2-acylhydrolase, EC 3.1.1.4) isozymes were compared internally and externally with those of six genes encoding Trimeresurus flavoviridis (habu snake, crotalinae) venom gland PLA2 isozymes. The numbers of nucleotide substitutions per site (KN) for the noncoding regions including introns were one-third to one-eighth of the numbers of nucleotide substitutions per synonymous site (KS) for the protein-coding regions of exons, indicating that the noncoding regions are much more conserved than the protein-coding regions. The KN values for the introns were found to be nearly equivalent to those of introns of T. gramineus and T. flavoviridis TATA box-binding protein genes, which are assumed to be a general (nonvenomous) gene. Thus, it is evident that the introns of venom gland PLA2 isozyme genes have evolved at a similar rate to those of nonvenomous genes. The numbers of nucleotide substitutions per nonsynonymous site (KA) were close to or larger than the KS values for the protein-coding regions in venom gland PLA2 isozyme genes. All of the data combined reveal that Darwinian-type accelerated evolution has universally occurred only in the protein-coding regions of crotalinae snake venom PLA2 isozyme genes.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Corden J., Wasylyk B., Buchwalder A., Sassone-Corsi P., Kedinger C., Chambon P. Promoter sequences of eukaryotic protein-coding genes. Science. 1980 Sep 19;209(4463):1406–1414. doi: 10.1126/science.6251548. [DOI] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Fukagawa T., Matsumoto H., Shimohigashi Y., Ogawa T., Oda N., Chang C. C., Ohno M. Sequence determination and characterization of a phospholipase A2 isozyme from Trimeresurus gramineus (green habu snake) venom. Toxicon. 1992 Nov;30(11):1331–1341. doi: 10.1016/0041-0101(92)90510-c. [DOI] [PubMed] [Google Scholar]
- Fukagawa T., Nose T., Shimohigashi Y., Ogawa T., Oda N., Nakashima K., Chang C. C., Ohno M. Purification, sequencing and characterization of single amino acid-substituted phospholipase A2 isozymes from Trimeresurus gramineus (green habu snake) venom. Toxicon. 1993 Aug;31(8):957–967. doi: 10.1016/0041-0101(93)90255-h. [DOI] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- John T. R., Smith L. A., Kaiser I. I. Genomic sequences encoding the acidic and basic subunits of Mojave toxin: unusually high sequence identity of non-coding regions. Gene. 1994 Feb 25;139(2):229–234. doi: 10.1016/0378-1119(94)90761-7. [DOI] [PubMed] [Google Scholar]
- Kihara H., Uchikawa R., Hattori S., Ohno M. Myotoxicity and physiological effects of three Trimeresurus flavoviridis phospholipases A2. Biochem Int. 1992 Dec;28(5):895–903. [PubMed] [Google Scholar]
- Kozak M. Compilation and analysis of sequences upstream from the translational start site in eukaryotic mRNAs. Nucleic Acids Res. 1984 Jan 25;12(2):857–872. doi: 10.1093/nar/12.2.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhara S., Matsuo F., Futamura S., Fujita A., Shinohara T., Takagi T., Sakaki Y. GENAS: a database system for nucleic acid sequence analysis. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):89–99. doi: 10.1093/nar/12.1part1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S. Y., Yoshizumi K., Oda N., Ohno M., Tokunaga F., Iwanaga S., Kihara H. Purification and amino acid sequence of basic protein II, a lysine-49-phospholipase A2 with low activity, from Trimeresurus flavoviridis venom. J Biochem. 1990 Mar;107(3):400–408. doi: 10.1093/oxfordjournals.jbchem.a123057. [DOI] [PubMed] [Google Scholar]
- Miyata T., Yasunaga T. Molecular evolution of mRNA: a method for estimating evolutionary rates of synonymous and amino acid substitutions from homologous nucleotide sequences and its application. J Mol Evol. 1980 Sep;16(1):23–36. doi: 10.1007/BF01732067. [DOI] [PubMed] [Google Scholar]
- Nakashima K., Nobuhisa I., Deshimaru M., Ogawa T., Shimohigashi Y., Fukumaki Y., Hattori M., Sakaki Y., Hattori S., Ohno M. Structures of genes encoding TATA box-binding proteins from Trimeresurus gramineus and T. flavoviridis snakes. Gene. 1995 Jan 23;152(2):209–213. doi: 10.1016/0378-1119(94)00681-h. [DOI] [PubMed] [Google Scholar]
- Nakashima K., Ogawa T., Oda N., Hattori M., Sakaki Y., Kihara H., Ohno M. Accelerated evolution of Trimeresurus flavoviridis venom gland phospholipase A2 isozymes. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):5964–5968. doi: 10.1073/pnas.90.13.5964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oda N., Nakamura H., Sakamoto S., Liu S. Y., Kihara H., Chang C. C., Ohno M. Amino acid sequence of a phospholipase A2 from the venom of Trimeresurus gramineus (green habu snake). Toxicon. 1991;29(2):157–166. doi: 10.1016/0041-0101(91)90100-6. [DOI] [PubMed] [Google Scholar]
- Ogawa T., Oda N., Nakashima K., Sasaki H., Hattori M., Sakaki Y., Kihara H., Ohno M. Unusually high conservation of untranslated sequences in cDNAs for Trimeresurus flavoviridis phospholipase A2 isozymes. Proc Natl Acad Sci U S A. 1992 Sep 15;89(18):8557–8561. doi: 10.1073/pnas.89.18.8557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka S., Mohri N., Kihara H., Ohno M. Amino acid sequence of Trimeresurus flavoviridis phospholipase A2. J Biochem. 1986 Jan;99(1):281–289. doi: 10.1093/oxfordjournals.jbchem.a135471. [DOI] [PubMed] [Google Scholar]
- Yoshizumi K., Liu S. Y., Miyata T., Saita S., Ohno M., Iwanaga S., Kihara H. Purification and amino acid sequence of basic protein I, a lysine-49-phospholipase A2 with low activity, from the venom of Trimeresurus flavoviridis (Habu snake). Toxicon. 1990;28(1):43–54. doi: 10.1016/0041-0101(90)90005-r. [DOI] [PubMed] [Google Scholar]


