Abstract
There has been a significant increase over the past few decades in the number of reproductive age women who are either overweight or obese. Overweight and obese women are at increased risk for having decreased insulin sensitivity as compared with lean or average weight women. The combination of obesity and decreased insulin sensitivity increases the long term risk of these individuals developing the metabolic syndrome and associated problems of diabetes, hypertension, hyperlipidemia and cardiovascular disorders. Because of the metabolic alterations during normal pregnancy, particularly the 60% decrease in insulin sensitivity, overweight and obese women are at increased risk of metabolic dysregulation in pregnancy, i.e. gestational diabetes, preeclampsia, and fetal overgrowth. Hence pregnancy can be considered a metabolic stress test for the future risk of the metabolic syndrome. In this chapter we will review the underlying pathophysiology related to these disorders. More importantly, an understanding of these risks provides an opportunity for prevention. For example, a planned pregnancy offers an opportunity to address weight control prior to conception. At the very least, by avoiding excessive weight gain during pregnancy, this may prevent excessive weight retention post partum. Finally based on the concept of in utero programming, these lifestyle measures may not only have short and long term benefits for the women but also for her offspring as well.
Obesity
There has been a significant increase in obesity in the last 40 years not only in developed areas of the world but also in developing countries (World Health Organization 2000). In the United States approximately ~ 62% of the female population > 20 years of age are overweight and 33% are obese, based on WHO body mass index (BMI, kg/m2) criteria (Ogden et al. 2006). Furthermore, 6.9% of the female population have a BMI >40, or Class III obesity (Ogden et al. 2006). In Asian populations, because of differences in body composition, the criteria for risk of metabolic dysfunction such as diabetes and cardiovascular disease are different than in a Caucasian population. A BMI of 23–27.5 is designated as increased risk and a BMI greater than 27.5 as high risk for metabolic dysfunction (WHO Expert Consultation 2004). Approximately 11% of US children are obese based on the Center for Health and Disease criteria (CDC), i.e. weight for height greater than 2 standard deviations for age and gender (Ogden et al. 2008). Last, there has been reported a significant increase in the mean term birth weight in various North American and European populations (Ananth & Wen 2002; Surkan et al. 2004).
Although BMI is a commonly used clinical criterion for obesity, in pregnancy BMI is a less robust estimate of adiposity. This is because on the physiological changes in pregnancy, specifically the significant increases in total body water. There is a decrease in the hydration constant of fat free mass from 76% in non pregnant individuals to approximately 72% in late gestation (Catalano et al. 1995). The correlations of BMI with measures of body composition decrease with advancing gestation; from R2=0.86 pregravid to R2=0.54 in the third trimester (Sewell et al. 2007). Similarly in neonates the ponderal index [weight [g] × 100/length [cm]) is a frequently used measure of obesity at birth. However, as with BMI, the ponderal index accounts for only a fraction (15%) of the variance in adiposity in the neonate (Catalano et al. 1992).
Insulin Resistance
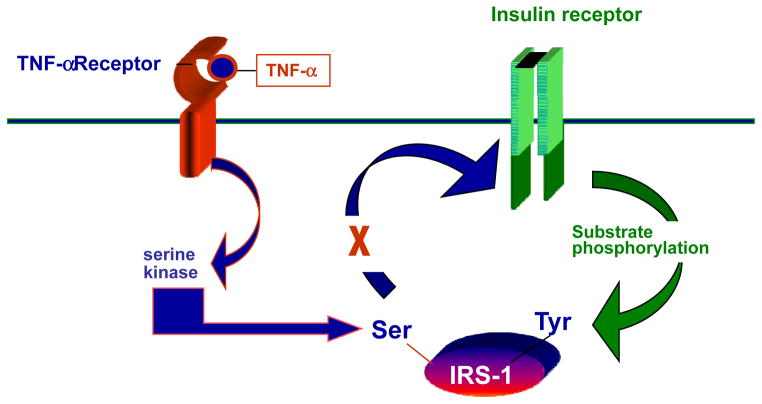
Decreased insulin sensitivity or increased insulin resistance is defined as the decreased biologic response of a nutrient to a given concentration of insulin at the target tissue; for e.g. liver, muscle or adipose tissue. Obesity is the most common risk factor related to decreased insulin sensitivity. The resistance to insulin can be characterized as pre receptor (insulin antibodies), receptor (decreased number of receptors on the cell surface) or post receptor (defects in the intracellular insulin signaling pathway). In pregnancy the decreased insulin sensitivity is best characterized as a post receptor defect resulting in the decreased ability of insulin to bring about GLUT 4 mobilization from the interior of the cell to the cell surface. Although human placental lactogen (HPL) has often been cited as the cause of the decreased insulin sensitivity of pregnancy, because of its production from the placenta and increasing concentrations with advancing gestation (Ryan & Enns 1988), more recently the role of cytokines and elevated lipid concentrations in pregnancy have been correlated with the longitudinal changes in insulin sensitivity in non-pregnant (Hotamisligil et al. 1994) as well as pregnant women (Xiang et al. 1999; Kirwan et al. 2002). A potential mechanism for TNF alpha affecting the post receptor insulin signaling cascade in skeletal muscle resulting in decreased insulin sensitivity is shown in Figure 1.
Figure 1.
Proposed mechanism by which TNF-alpha may decrease insulin sensitivity. TNF-alpha activates a pathway that increases sphingomyelinases and ceramides and may interfere with insulin receptor 1 (IRS-1) autophosphorylation.
Insulin sensitivity in vivo during pregnancy can be estimated using a variety of techniques. The gold standard, however, is the hyperinsulinemic euglycemic clamp techniques as described by DeFronzo et al (DeFronzo et al. 1985). In this technique insulin is infused as a primed constant infusion in order to attain a steady state insulin concentration. A variable glucose infusion is then based on frequent glucose sampling in order to maintain a constant euglycemic glucose concentration, i.e. the glucose is clamped at a steady state level. Stable isotopes of glucose, lipids and amino acids can be incorporated with the clamp technique to measure specific turnover rates of individual nutrients and tissues relative to the specific action of insulin; for example, gluconeogenesis or lipolysis. Because of the complexity, expense and time involved in the hyperinsulinemic-euglycemic clamp, this methodology is primarily used in research studies when accuracy and reproducibility of the measures are required. The minimal model developed by Bergman et al (Bergman et al. 1987), estimates insulin sensitivity using an intravenous glucose tolerance test (IVGTT). The minimal model may be less useful in late pregnancy because of the decreased insulin sensitivity in late gestation. The minimal model has increased variability when estimating insulin sensitivity in late gestation in comparison with the euglycemic clamp. More common clinical methods include measures of fasting glucose and insulin (HOMA and QUICKI, (Matthews et al. 1985; Katz et al. 2000) as well as models using the oral glucose tolerance test (OGTT), for e.g. as described by Matsuda and DeFronzo (Matsuda & DeFronzo 1999). All these methodologies, however, have limitations. During pregnancy there is non-insulin mediated glucose disposal from the mother to the fetus through placental facilitated diffusion of glucose. Therefore all estimates of insulin sensitivity in pregnancy are overestimates of maternal insulin sensitivity, particularly in late gestation.
Maternal Metabolic Complications of Obesity
Women who are obese and therefore more likely have decreased insulin sensitivity are at increased risk for many adverse pregnancy outcomes. Similar to what is observed in the non pregnant population, the constellation of conditions mimics the metabolic syndrome (World Health Organization 1999). The metabolic syndrome of pregnancy includes an increased risk of hypertensive, metabolic disturbances of nutrient metabolism and inflammation. Although these pregnancy related conditions are most likely to clinically resolve once the woman is delivered, these individuals still have the subclinical underlying metabolic disorder and are at increased risk for the metabolic syndrome in later life, particularly if there is increased post partum weight gain (Villamor & Cnattingius 2006).
Hypertensive Disease
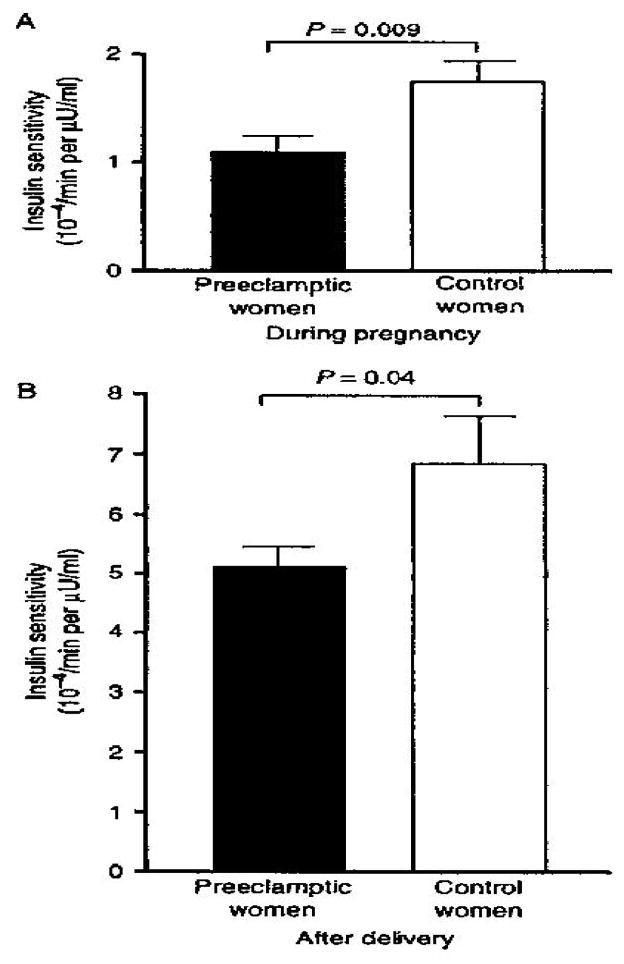
Obese women have a 10–15% increased risk for preeclampsia (Barton & Sibai 2008). The decreased insulin sensitivity is manifest as early as the first trimester of pregnancy. Wolf et al reported that sex hormone binding globulin (Wolf et al. 2002) as a marker of decreased insulin sensitivity was significantly decreased in nulliparous women during the first trimester that eventually went on to develop preeclampsia as compared with a control group. Furthermore when stratifying the women based on BMI, overweight women had lower SHBG concentrations as compared with lean women and within each stratum of BMI (Wolf et al. 2002). Similarly Mazaki-Tovi et al using measures of serum adiponectin, a cytokine associated with increased insulin sensitivity, reported that in women with mild preeclampsia, there were decreased concentrations of high molecular weight (HMW) adiponectin, the most active form of adiponectin, in comparison with normotensive pregnant women. In contrast to decreasing concentrations of circulating adiponectin with advancing gestation, preeclampsia was not associated with decreased adiponectin in overweight and obese women (Mazaki-Tovi et al. 2009). Using the minimal model technique, Kaaja et al, showed that in late gestation women developing preeclampsia have decreased insulin sensitivity as compared with a control group matched for BMI and other potential confounding variables (Figure 2a). Consistent with the decreased insulin sensitivity, the women developing preeclampsia also had elevated free fatty acid, triglycerides and C- peptide concentrations. Although there was the expected improvement in insulin sensitivity post partum, the women developing preeclampsia had decreased insulin sensitivity (Figure 2b) that persisted for at least 3 months (Kaaja et al. 1999). Taken as a whole these studies are consistent with the concept of obesity and insulin resistance as putative factors related to the development of preeclampsia. Of interest is the decreased insulin sensitivity which is present in early in gestation, prior to the clinical manifestations of the disorder, and persists post partum; thereby providing a link between the pregnancy related hypertensive disorders of pregnancy and later manifestations of the metabolic syndrome. Much as with the mechanism associated with decreased insulin sensitivity and essential hypertension, the underlying mechanisms related to the development of preeclampsia is not very well characterized (Solomon & Seely 2001).
Figure 2.
Insulin sensitivity in intravenous glucose tolerance assessed by minimal model analysis in women with preeclampsia and normotensive control women (A) during pregnancy and (B) 12 weeks after delivery. Data are mean ± SE (Kaaja et al. 1999)
Glucose Intolerance/Gestational Diabetes
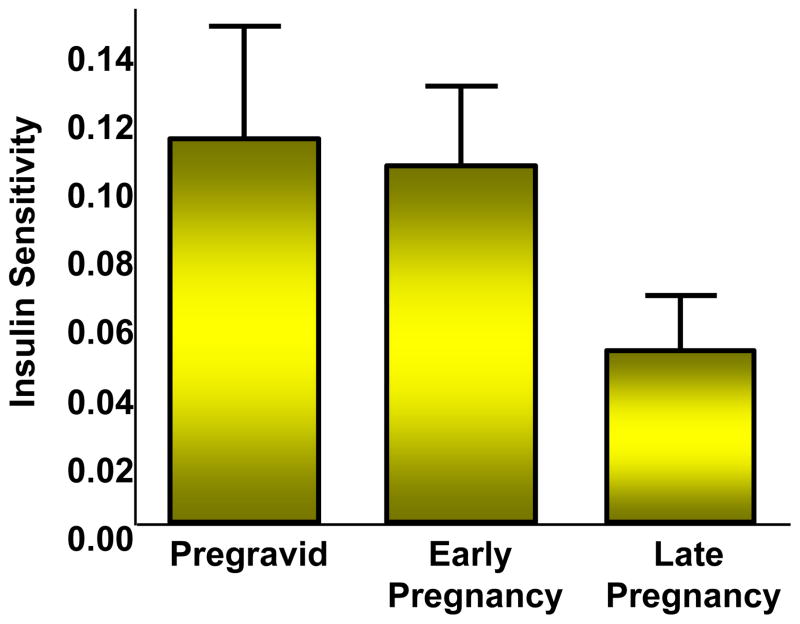
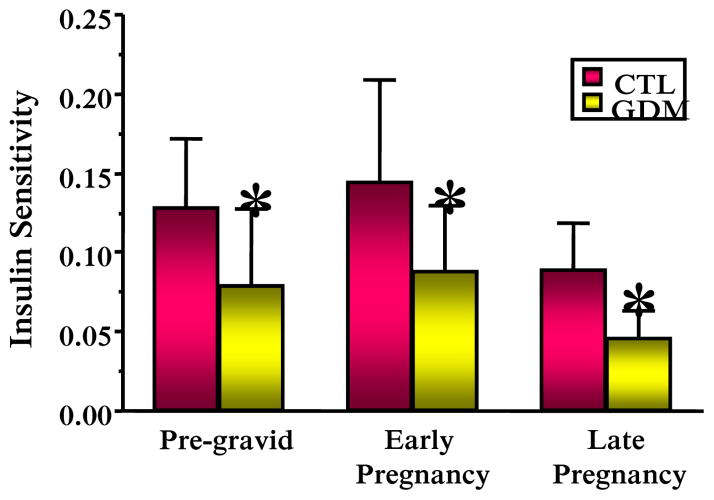
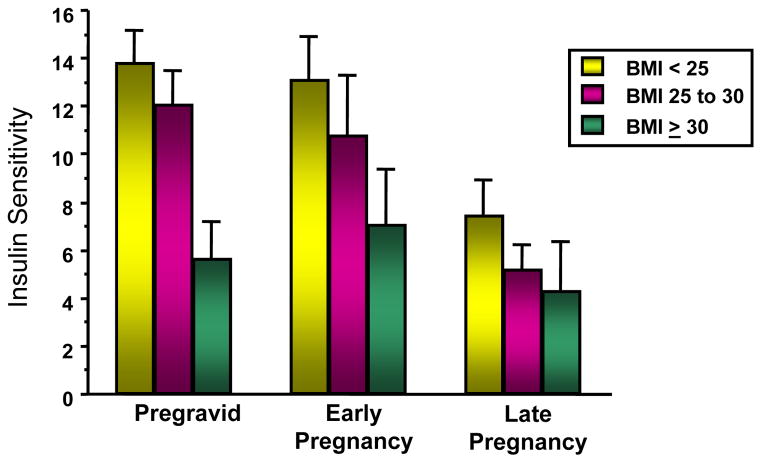
Obesity is the strongest risk factor for the development of type 2 diabetes in the non pregnant individual and for the development of glucose intolerance during gestation, i.e. gestational diabetes (GDM). The underlying metabolic defects related to the development of both these disorders are decreased insulin sensitivity coupled with an inadequate insulin response (Buchanan & Xiang 2005). In prospective longitudinal studies our group has shown that women with normal glucose tolerance have a 50–60 percent decrease in insulin sensitivity during the course of gestation (Figure 3), (Catalano et al. 1991). When compared with weight matched controls, women with normal glucose tolerance prior to conception and go on to develop GDM during pregnancy, have significant decreased insulin sensitivity both before and during pregnancy (Figure 4), (Catalano et al. 1999). However, both groups have a similar percent decrease in insulin sensitivity. Similar as to what was observed in normal glucose tolerant women and those developing GDM, overweight and obese women have decreased insulin sensitivity as compared with lean or average weight women, although both groups have a similar 50% decrease over the period of gestation (Figure 5), (Catalano & Ehrenberg 2006). Hence, just as there has been increase in the number of individuals classified as having either impaired fasting glucose (33.8%) or impaired glucose tolerance (15.4%), (Center for Disease Control and Prevention 2007), the number of women with GDM presently at 5–10% of the population may significantly increase based on the recommendations of the International Diabetes in Pregnancy Study group (Consensus panel: International Association of Diabetes and Pregnancy Study Groups (IADPSG) 2010). These data are also consistent with the underlying pathophysiology of GDM resulting in a 50–60% increase in type 2 diabetes in 10 years after the diagnosis of GDM (Kim et al. 2002).
Figure 3.
The longitudinal changes in insulin sensitivity in normal glucose tolerant women as estimated by the hypoinsulinemic-euglycemic clamp. Early pregnancy, 12–14 weeks and late pregnancy 34–36 weeks, longitudinal changes over time, p=0.0001. (Adapted from Catalano et al. 1991)
Figure 4.
The longitudinal change in insulin sensitivity in normal glucose tolerant women (CTL) and women with gestational diabetes (GDM). Early pregnancy 12–14 weeks and late pregnancy 34–36 weeks. Changes over time; p=0.0001, changes between groups; p=0.03. (Adapted from Catalano et al. 1999)
Figure 5.
The longitudinal changes in insulin sensitivity in average (BMI<25), overweight (BMI 25–30) and obese (BMI>30) women over time. Early pregnancy 12–14 weeks and late pregnancy 34–36 weeks longitudinal changes over time; p=0.0004. (Adapted from Catalano & Ehrenberg 2006)
Hyperlipidemia
The decrease in insulin sensitivity in pregnancy is not limited only to glucose metabolism but is also is observed in relation to lipid metabolism as well (Catalano et al. 2002). There is a two to threefold increase in basal triglyceride and cholesterol concentrations with advancing gestation (Knopp et al. 1982). The increases are more pronounced in the GDM as compared with the normal glucose tolerant pregnant woman (Catalano et al. 2002). The increase in free fatty acid concentration is related to the decreased ability of insulin to suppress lipolysis in late gestation. Freinkel coined the term the “accelerated starvation of pregnancy” to describe the increasing risk of ketosis observed in pregnant women (Freinkel 1980). The increased free fatty acids are then useful for providing an energy source for maternal needs in late gestation when maternal energy requirements are greatest. The increase in free fatty acids may also play a role in excessive of fetal growth. Although the increased glucose concentrations in pregnancy observed in obese and GDM women are a stimulus for fetal insulin production necessary, the nutrient substrates for fetal growth in particular adiposity are less well described. Recent studies by Shaefer-Graf (Schaefer-Graf et al. 2008) and DiCianni (DiCianni et al. 2005) have reported a significant correlation between maternal triglyceride concentration in late pregnancy and fetal growth/adiposity. Consistent with these clinical observations, Radaelli et al have reported that enzymes related to intermediary lipid metabolism in contrast to glucose are increased in placenta of obese GDM women (Radaelli et al. 2009). These data support the concept that the elevated lipid concentration in obese pregnant women is also related to the increase in fetal growth and adiposity.
Body Composition
The assessment of fetal growth has been variously classified over time. Initially any small baby was classified as “preemie”, whereas large babies were classified as “macrosomic”. In the latter part of the last century it became more common to classify fetal growth or birth weight as a function of gestational age. Birth weights for gestational age were categorized as percentiles with the small being arbitrarily defined as less than the 10th percentile (small for gestational age or SGA) and large as greater than the 90th percentile (large for gestational age or LGA).
A further refinement of growth includes the estimation of body composition, i.e. the description of lean and fat mass. In contrast to most mammals at birth, the human has a large proportion of birth weight as fat mass; approximately 10–12%. In contrast most murine models have only 1–2% body fat, whereas non human primates have only 3–5 percent body fat (Widdowson 1950; Ausman et al. 1982; Russo et al. 1980). Based on studies by Sparks, lean body mass has a stronger correlation with genetic factors, while fat mass may relate more to the maternal environment (Sparks 1984). Therefore the use of body composition is important in assessing fetal growth relative to the in utero environment.
It has long been recognized that infants of GDM and obese women weigh more at the birth than either women with weight matched normal glucose tolerance or normal weight women, respectively. Based on studies by our group and others, the infant of the GDM weighs more at birth because of an increase in fat mass and not lean body mass (Figure 6), (Catalano et al. 2003). Similarly the offspring of the obese women weighs more at birth because of increased fat mass and not lean mass (Figure 7), (Sewell et al. 2006). The strongest correlate with neonatal fat mass in women with GDM is decreased maternal pregravid insulin sensitivity (Catalano et al. 1995). In obese women, maternal pregravid BMI, as a surrogate for insulin resistance, has the strongest correlation with neonatal adiposity (Catalano & Ehrenberg 2006). The increased adiposity at birth is also a risk factor for childhood obesity and long term metabolic dysfunction (Catalano et al. 2009).
Figure 6.
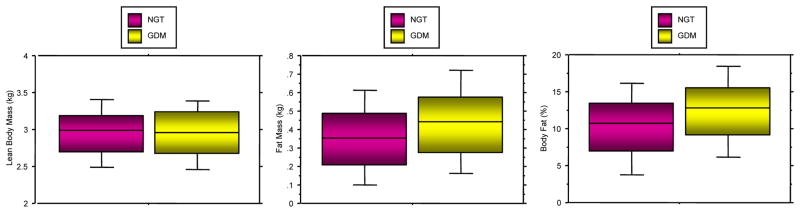
- Lean body mass (LBM); p=0.74
- Fat mass (FM); p=0.0002
- Percent body fat (%BF); p=0.0001
(Adapted from Catalano et al. 2003)
Figure 7.
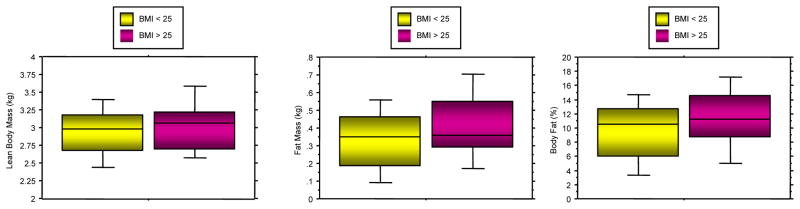
- Lean body mass (LBM); p=0.22
- Fat mass (FM); p=0.0008
- Percent body fat (%BF); p=0.006
(Adapted from Sewell et al. 2006)
Summary
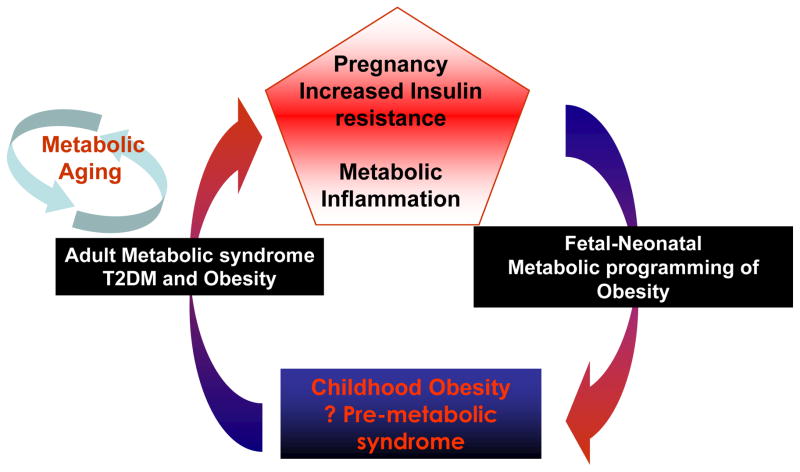
Maternal obesity and underlying insulin resistance are significant short and long term risk factors for both the mother and her fetus. For the obese women with subclinical decreased insulin sensitivity, pregnancy represents a metabolic stress test for those disorders in pregnancy which are the harbingers of the metabolic syndrome in later life, for e.g. GDM and preeclampsia. As a consequence, in the offspring of obese women are at increased risk of indicated preterm delivery and associates neonatal morbidity. These children are also at risk for childhood metabolic dysfunction, which creates the potential of a vicious cycle of obesity and insulin resistance begetting obesity and metabolic dysfunction (Figure 8) (Catalano 2003). Hence, lifestyle intervention of diet, exercise and weight control, ideally before and at the very least during gestation, offers the potential of benefit for both short and long term benefit for the mother and her child.
Figure 8.
Proposed model of fetal programming of offspring of women with abnormal metabolic environment (increased insulin resistance) (Adapted from Catalano 2003)
Acknowledgments
The study was supported by NIH HD 22965.
References
- Ananth CV, Wen SW. Trends in fetal growth among singleton gestations in the United States and Canada. Seminars in Perinatology. 2002;26:260–267. doi: 10.1053/sper.2002.34772. [DOI] [PubMed] [Google Scholar]
- Ausman LM, Powell EM, Mercado DL, Samonds KW, el Lozy M, Gallina DL. Growth and developmental body composition of the cebus monkey (Cebus Albifrons) American Journal Perinatology. 1982;3:211–217. doi: 10.1002/ajp.1350030119. [DOI] [PubMed] [Google Scholar]
- Barton JR, Sibai BM. Prediction and prevention of recurrent preeclampsia. Obstetrics and Gynecology. 2008;112:359–372. doi: 10.1097/AOG.0b013e3181801d56. [DOI] [PubMed] [Google Scholar]
- Bergman RN, Prager R, Volund A, Ofefsky JM. Equivalence of the insulin sensitivity index in man derived by the minimal model method and the euglycemic glucose clamp. Journal of Clinical Investigation. 1987;79:790–800. doi: 10.1172/JCI112886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan T, Xiang AH. Gestational diabetes mellitus. Journal of Clinical Investigations. 2005;115:485–491. doi: 10.1172/JCI24531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catalano PM. Obesity and pregnancy – the propagation of a viscous cycle? Journal of Clinical Endocrinology and Metabolism. 2003;88:3505–3506. doi: 10.1210/jc.2003-031046. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Ehrenberg HM. The short and long term implications of maternal obesity on the mother and her offspring. British Journal of Obstetrics & Gynecology. 2006;113:1126–1133. doi: 10.1111/j.1471-0528.2006.00989.x. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Tyzbir ED, Roman NM, Samini SB, Sims EA. Longitudinal changes in insulin release and insulin resistance in non-obese pregnant women. American Journal of Obstetrics Gynecology. 1991;165:1667–1672. doi: 10.1016/0002-9378(91)90012-g. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Tyzbir ED, Allen SR, McBean JH, McAuliffe TL. Evaluation of fetal growth by estimation of neonatal body composition. Obstetrics and Gynecology. 1992;79:46–50. [PubMed] [Google Scholar]
- Catalano PM, Drago NM, Amini SB. Maternal carbohydrate metabolism and its relationship to fetal growth and body composition. American Journal of Obstetrics and Gynecology. 1995;72:1464–1470. doi: 10.1016/0002-9378(95)90479-4. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Wong WW, Drago NM, Amini SB. Estimating body composition in late gestation: A new hydration constant for body density and total body water. American Journal of Physiology, endocrinology and Metabolism. 1995b;268:E153–E158. doi: 10.1152/ajpendo.1995.268.1.E153. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Huston L, Amini SB, kalhan SC. Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes mellitus. American Journal of Obstetrics and Gynecology. 1999;180:903–916. doi: 10.1016/s0002-9378(99)70662-9. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Nizielski SE, Shao J, Preston L, Qiao L, Friedman JE. Downregulated IRS-1 and PPARγ in obese women with gestational diabetes: Relationship to free fatty acids during pregnancy. American Journal of Physiology Endocrine and Metabolism. 2002;282:E522–E533. doi: 10.1152/ajpendo.00124.2001. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Thomas A, Huston-Presley L, Amini SB. Increased fetal adiposity: A very sensitive marker of abnormal in-utero development. American Journal of Obstetrics and Gynecology. 2003;189:1698–1704. doi: 10.1016/s0002-9378(03)00828-7. [DOI] [PubMed] [Google Scholar]
- Catalano PM, Farrell K, Thomas A, Huston-Presley L, Mencin P, de Mouzon SH, Amini SB. Perinatal risk factors for childhood obesity and metabolic dysregulation. American Journal of Clinical Nutrition. 2009;90:1303–1313. doi: 10.3945/ajcn.2008.27416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Center for Disease Control and Prevention. 2007 National Diabetes Fact Sheet 2007. Atlanta, GA: US Department of Health and Human Services Centers for Disease Control and Prevention; 2008. [Google Scholar]
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel . International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33:676–682. doi: 10.2337/dc09-1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeFronzo RA, Gunnasson R, Bjorkman O, Olsson M, Wahren J. Effects of insulin on peripheral and splanclinic glucose metabolism in noninsulin-dependent (type II) diabetes mellitus. Journal of Clinical Investigation. 1985;76:149–55. doi: 10.1172/JCI111938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Cianni G, Miccoli R, Volpe L, lencioni C, Ghio A, Giovannitti MG, Cuccuru I, Pellegrini G, Chatzianagnostou K, Boldrini A, et al. Maternal triglyceride levels and newborn weight in pregnant women with normal glucose tolerance. Diabetic Medicine. 2005;22:21–25. doi: 10.1111/j.1464-5491.2004.01336.x. [DOI] [PubMed] [Google Scholar]
- Freinkel N. Banting Lecture of 1980: Of Pregnancy and Progeny. Diabetes. 1980;29:1023–1035. doi: 10.2337/diab.29.12.1023. [DOI] [PubMed] [Google Scholar]
- Hotamisligil GS, Murray DL, Choy LN, Spiegelman BM. Tumor necrosis factor alpha inhibits signaling from the insulin receptor. PNAS. 1994;91:4854–4858. doi: 10.1073/pnas.91.11.4854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaaja R, Laivuori H, Laaksa M, Tikkanen MJ, Yikorkala O. Evidence of a state of increased insulin resistance in preeclampsia. Metabolism. 1999;48:892–896. doi: 10.1016/s0026-0495(99)90225-1. [DOI] [PubMed] [Google Scholar]
- Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, Quon MJ. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. Journal of Clinical Endocrinology and Metabolism. 2000;85:2402–2410. doi: 10.1210/jcem.85.7.6661. [DOI] [PubMed] [Google Scholar]
- Kim C, Newton KM, Knopp RH. Gestational diabetes and the incidence of type 2 diabetes. Diabetes Care. 2002;25:1862–1868. doi: 10.2337/diacare.25.10.1862. [DOI] [PubMed] [Google Scholar]
- Kirwan JP, Hauguel-de Mouzon S, Lepercq J, Challier JC, Huston-Presley L, Friedman JE, Kalhan SC, Catalano PM. TNFα is a predictor of insulin resistance in human pregnancy. Diabetes. 2002;51:2207–2213. doi: 10.2337/diabetes.51.7.2207. [DOI] [PubMed] [Google Scholar]
- Knopp RH, Chapman M, Bergelin RO, Wahl PW, Warth MR, Irvine S. Relationship of lipoprotein lipids to mild fasting hyperglycemia and diabetes in pregnancy. Diabetes Care. 1980;3:416–420. doi: 10.2337/diacare.3.3.416. [DOI] [PubMed] [Google Scholar]
- Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22:1462–1470. doi: 10.2337/diacare.22.9.1462. [DOI] [PubMed] [Google Scholar]
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- Mazaki-Tovi S, Romero R, Vaisbuch E, Kusanovic JP, Erez O, Gotsch F, Chaiworapongsa T, Than NG, Kim SK, Nhan-Chang C-L, et al. Maternal serum adiponectin mulimers in preeclampsia. Journal of Perinatal Medicine. 2009;37:349–363. doi: 10.1515/JPM.2009.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogden CC, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. Journal of the American medical Association. 2006;295:1549–1555. doi: 10.1001/jama.295.13.1549. [DOI] [PubMed] [Google Scholar]
- Ogden CL, Caroll MD, Flegal KM. High body mass index for age among US children and adolescents 2003–2006. Journal of the American medical Association. 2008;299:2401–2405. doi: 10.1001/jama.299.20.2401. [DOI] [PubMed] [Google Scholar]
- Radaelli T, Lepercq J, Varastehpour A, Basu S, Catalano PM, Hauguel-de Mouzon S. Differential regulation of genes for fetoplacental lipid pathways in pregnancy with gestational and type 1 diabetes mellitus. American Journal Obstetrics Gynecology. 2009:201–209. e1–209, e10. doi: 10.1016/j.ajog.2009.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo AR, Ausman LM, Gallina DL, Hegsted DM. Developmental body composition of the squirrel monkey (Saimiri sciureus) Growth. 1980;44:271–286. [PubMed] [Google Scholar]
- Ryan EA, Enns L. Role of gestational hormones in the induction of insulin resistance. Journal of Clinical Endocrinology and Metabolism. 1988;67:341–347. doi: 10.1210/jcem-67-2-341. [DOI] [PubMed] [Google Scholar]
- Schaefer-Graf UM, Graf K, Kulbacka I, Kjos SL, Dudenhausen J, Vetter K, Herrera E. Maternal lipids as strong determinants of fetal environment and growth in pregnancies with gestational diabetes mellitus. Diabetes Care. 2008;31:1858–1863. doi: 10.2337/dc08-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sewell MF, Huston-Presley L, Super DM, Catalano P. Increased neonatal fat mass, not lean body mass, is associated with maternal obesity. American Journal of Obstetrics and Gynecology. 2006;195:1100–1103. doi: 10.1016/j.ajog.2006.06.014. [DOI] [PubMed] [Google Scholar]
- Sewell MF, Huston-Presley L, Amini S, Catalano PM. Body mass index, a true indicator of body fat in obese gravidas. Journal of Reproductive Medicine. 2007;52:907–911. [PubMed] [Google Scholar]
- Solomon CG, Seely EW. Hypertension in pregnancy A manifestation of the insulin resistance syndrome? Hypertension. 2001;37:232–239. doi: 10.1161/01.hyp.37.2.232. [DOI] [PubMed] [Google Scholar]
- Sparks J. Human intrauterine growth and nutrient accretion. Seminars in Perinatology. 1984;18:74–93. [PubMed] [Google Scholar]
- Surkan PJ, Hsieh CC, Johansson AL, Dickman PW, Cnattinguis S. Reasons for increasing trends in large for gestational age births. Obstetrics and Gynecology. 2004;104:720–726. doi: 10.1097/01.AOG.0000141442.59573.cd. [DOI] [PubMed] [Google Scholar]
- Villamor E, Cnattingius S. Interpregnancy weight change and risk of adverse pregnancy outcomes: A population-based study. Lancet. 2006;368:1164–1170. doi: 10.1016/S0140-6736(06)69473-7. [DOI] [PubMed] [Google Scholar]
- WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–163. doi: 10.1016/S0140-6736(03)15268-3. [DOI] [PubMed] [Google Scholar]
- Widdowson EM. Chemical composition of newly born mammals. Nature. 1950;66:626–628. doi: 10.1038/166626a0. [DOI] [PubMed] [Google Scholar]
- Wolf M, Sandler L, Munoz K, Hsu K, Ecker JL, Thadhani R. First trimester insulin resistance and subsequent preeclampsia: A prospective study. Journal Clinical Endocrinology and Metabolism. 2002;87:1563–1568. doi: 10.1210/jcem.87.4.8405. [DOI] [PubMed] [Google Scholar]
- World Health Organization. Definition, diagnosis and classification of diabetes mellitus and its complications: Report of a WHO Consultation. Part 1: Diagnosis and classification of diabetes mellitus. Geneva, Switzerland: World Health Organization; 1999. [Google Scholar]
- World Health Organization. World Health Organization Technical Report Series. Vol. 894. Geneva: World Health Organisation; 2000. Obesity: Preventing and managing a global epidemic; pp. 6–13. chapter 2. [PubMed] [Google Scholar]
- Xiang AH, Peters RH, Trigo E, Kjos SL, Lee WP, Buchanan TA. Multiple metabolic defects during late pregnancy in women at high risk for type 2 diabetes. Diabetes. 1999;48:848–854. doi: 10.2337/diabetes.48.4.848. [DOI] [PubMed] [Google Scholar]