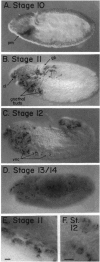
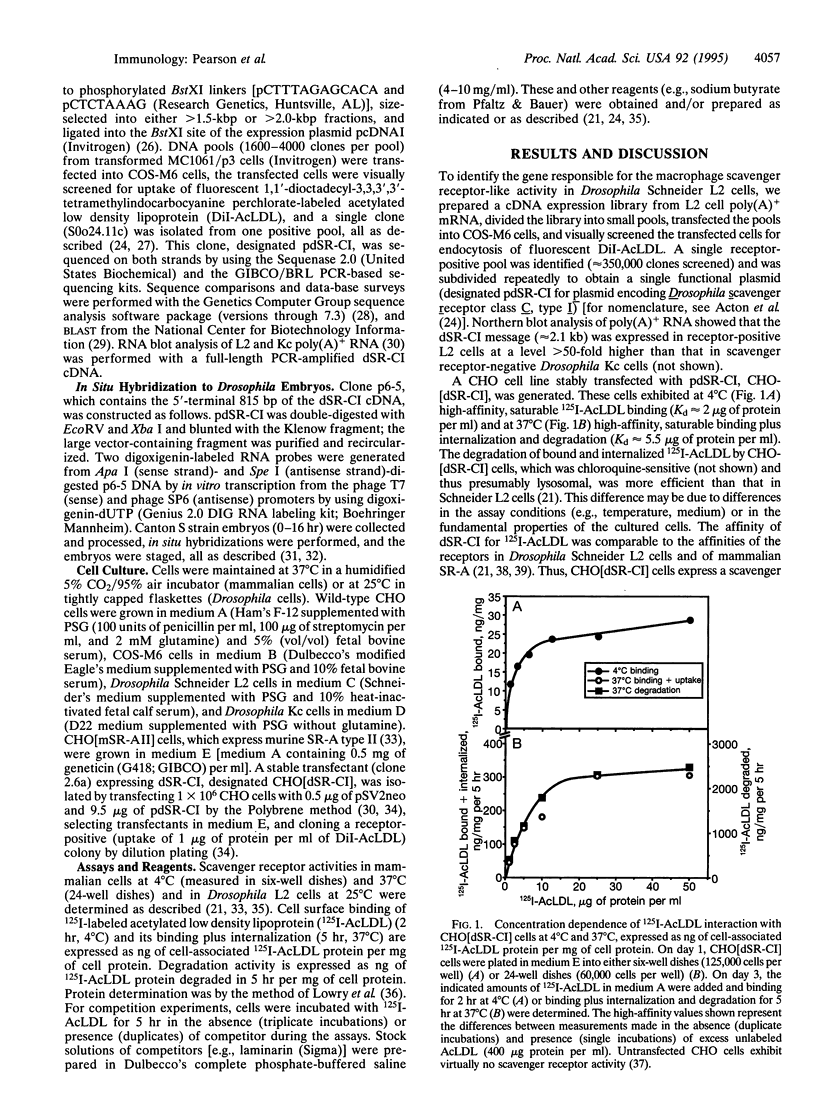
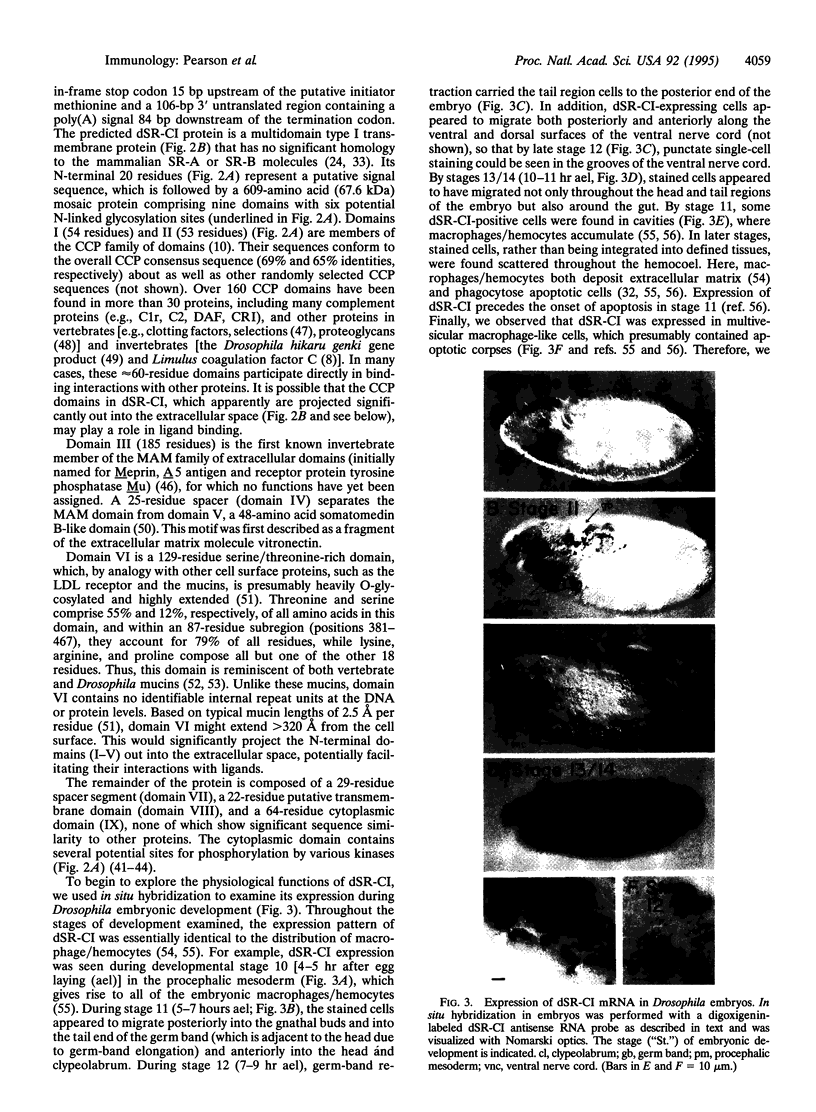
Abstract
Mammalian class A macrophage-specific scavenger receptors (SR-A) exhibit unusually broad binding specificity for a wide variety of polyanionic ligands. The properties of these receptors suggest that they may be involved in atherosclerosis and host defense. We have previously observed a similar receptor activity in Drosophila melanogaster embryonic macrophages and in the Drosophila macrophage-like Schneider L2 cell line. Expression cloning was used to isolate from L2 cells a cDNA that encodes a third class (class C) of scavenger receptor, Drosophila SR-CI (dSR-CI). dSR-CI expression was restricted to macrophages/hemocytes during embryonic development. When expressed in mammalian cells, dSR-CI exhibited high affinity and saturable binding of 125I-labeled acetylated low density lipoprotein and mediated its chloroquine-dependent, presumably lysosomal, degradation. Although the broad polyanionic ligand-binding specificity of dSR-CI was similar to that of SR-A, their predicted protein sequences are not similar. dSR-CI is a 609-residue type I integral membrane protein containing several well-known sequence motifs, including two complement control protein (CCP) domains and somatomedin B, MAM, and mucin-like domains. Macrophage scavenger receptors apparently mediate important, well-conserved functions and may be pattern-recognition receptors that arose early in the evolution of host-defense mechanisms. Genetic and physiologic analysis of dSR-CI function in Drosophila should provide further insights into the roles played by scavenger receptors in host defense and development.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrams J. M., Lux A., Steller H., Krieger M. Macrophages in Drosophila embryos and L2 cells exhibit scavenger receptor-mediated endocytosis. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10375–10379. doi: 10.1073/pnas.89.21.10375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abrams J. M., White K., Fessler L. I., Steller H. Programmed cell death during Drosophila embryogenesis. Development. 1993 Jan;117(1):29–43. doi: 10.1242/dev.117.1.29. [DOI] [PubMed] [Google Scholar]
- Acton S. L., Scherer P. E., Lodish H. F., Krieger M. Expression cloning of SR-BI, a CD36-related class B scavenger receptor. J Biol Chem. 1994 Aug 19;269(33):21003–21009. [PubMed] [Google Scholar]
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Aruffo A., Seed B. Molecular cloning of a CD28 cDNA by a high-efficiency COS cell expression system. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8573–8577. doi: 10.1073/pnas.84.23.8573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashkenas J., Penman M., Vasile E., Acton S., Freeman M., Krieger M. Structures and high and low affinity ligand binding properties of murine type I and type II macrophage scavenger receptors. J Lipid Res. 1993 Jun;34(6):983–1000. [PubMed] [Google Scholar]
- Ashkenas J., Penman M., Vasile E., Acton S., Freeman M., Krieger M. Structures and high and low affinity ligand binding properties of murine type I and type II macrophage scavenger receptors. J Lipid Res. 1993 Jun;34(6):983–1000. [PubMed] [Google Scholar]
- Baughman G., Lesley J., Trotter J., Hyman R., Bourgeois S. Tcl-30, a new T cell-specific gene expressed in immature glucocorticoid-sensitive thymocytes. J Immunol. 1992 Sep 1;149(5):1488–1496. [PubMed] [Google Scholar]
- Beckmann G., Bork P. An adhesive domain detected in functionally diverse receptors. Trends Biochem Sci. 1993 Feb;18(2):40–41. doi: 10.1016/0968-0004(93)90049-s. [DOI] [PubMed] [Google Scholar]
- Bevilacqua M. P., Nelson R. M. Selectins. J Clin Invest. 1993 Feb;91(2):379–387. doi: 10.1172/JCI116210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown M. S., Goldstein J. L. Lipoprotein metabolism in the macrophage: implications for cholesterol deposition in atherosclerosis. Annu Rev Biochem. 1983;52:223–261. doi: 10.1146/annurev.bi.52.070183.001255. [DOI] [PubMed] [Google Scholar]
- Cociancich S., Bulet P., Hetru C., Hoffmann J. A. The inducible antibacterial peptides of insects. Parasitol Today. 1994 Apr;10(4):132–139. doi: 10.1016/0169-4758(94)90260-7. [DOI] [PubMed] [Google Scholar]
- Cociancich S., Bulet P., Hetru C., Hoffmann J. A. The inducible antibacterial peptides of insects. Parasitol Today. 1994 Apr;10(4):132–139. doi: 10.1016/0169-4758(94)90260-7. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doi T., Higashino K., Kurihara Y., Wada Y., Miyazaki T., Nakamura H., Uesugi S., Imanishi T., Kawabe Y., Itakura H. Charged collagen structure mediates the recognition of negatively charged macromolecules by macrophage scavenger receptors. J Biol Chem. 1993 Jan 25;268(3):2126–2133. [PubMed] [Google Scholar]
- Dunne D. W., Resnick D., Greenberg J., Krieger M., Joiner K. A. The type I macrophage scavenger receptor binds to gram-positive bacteria and recognizes lipoteichoic acid. Proc Natl Acad Sci U S A. 1994 Mar 1;91(5):1863–1867. doi: 10.1073/pnas.91.5.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezekowitz R. A., Williams D. J., Koziel H., Armstrong M. Y., Warner A., Richards F. F., Rose R. M. Uptake of Pneumocystis carinii mediated by the macrophage mannose receptor. Nature. 1991 May 9;351(6322):155–158. doi: 10.1038/351155a0. [DOI] [PubMed] [Google Scholar]
- Ezekowitz R. A., Williams D. J., Koziel H., Armstrong M. Y., Warner A., Richards F. F., Rose R. M. Uptake of Pneumocystis carinii mediated by the macrophage mannose receptor. Nature. 1991 May 9;351(6322):155–158. doi: 10.1038/351155a0. [DOI] [PubMed] [Google Scholar]
- Feramisco J. R., Glass D. B., Krebs E. G. Optimal spatial requirements for the location of basic residues in peptide substrates for the cyclic AMP-dependent protein kinase. J Biol Chem. 1980 May 10;255(9):4240–4245. [PubMed] [Google Scholar]
- Fraser I., Hughes D., Gordon S. Divalent cation-independent macrophage adhesion inhibited by monoclonal antibody to murine scavenger receptor. Nature. 1993 Jul 22;364(6435):343–346. doi: 10.1038/364343a0. [DOI] [PubMed] [Google Scholar]
- Freeman M., Ekkel Y., Rohrer L., Penman M., Freedman N. J., Chisolm G. M., Krieger M. Expression of type I and type II bovine scavenger receptors in Chinese hamster ovary cells: lipid droplet accumulation and nonreciprocal cross competition by acetylated and oxidized low density lipoprotein. Proc Natl Acad Sci U S A. 1991 Jun 1;88(11):4931–4935. doi: 10.1073/pnas.88.11.4931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freeman M., Ekkel Y., Rohrer L., Penman M., Freedman N. J., Chisolm G. M., Krieger M. Expression of type I and type II bovine scavenger receptors in Chinese hamster ovary cells: lipid droplet accumulation and nonreciprocal cross competition by acetylated and oxidized low density lipoprotein. Proc Natl Acad Sci U S A. 1991 Jun 1;88(11):4931–4935. doi: 10.1073/pnas.88.11.4931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glass D. B., Smith S. B. Phosphorylation by cyclic GMP-dependent protein kinase of a synthetic peptide corresponding to the autophosphorylation site in the enzyme. J Biol Chem. 1983 Dec 25;258(24):14797–14803. [PubMed] [Google Scholar]
- Hampton R. Y., Golenbock D. T., Penman M., Krieger M., Raetz C. R. Recognition and plasma clearance of endotoxin by scavenger receptors. Nature. 1991 Jul 25;352(6333):342–344. doi: 10.1038/352342a0. [DOI] [PubMed] [Google Scholar]
- Hoshino M., Matsuzaki F., Nabeshima Y., Hama C. hikaru genki, a CNS-specific gene identified by abnormal locomotion in Drosophila, encodes a novel type of protein. Neuron. 1993 Mar;10(3):395–407. doi: 10.1016/0896-6273(93)90329-p. [DOI] [PubMed] [Google Scholar]
- Hoshino M., Matsuzaki F., Nabeshima Y., Hama C. hikaru genki, a CNS-specific gene identified by abnormal locomotion in Drosophila, encodes a novel type of protein. Neuron. 1993 Mar;10(3):395–407. doi: 10.1016/0896-6273(93)90329-p. [DOI] [PubMed] [Google Scholar]
- Hultmark D. Immune reactions in Drosophila and other insects: a model for innate immunity. Trends Genet. 1993 May;9(5):178–183. doi: 10.1016/0168-9525(93)90165-e. [DOI] [PubMed] [Google Scholar]
- Iwanaga S., Miyata T., Tokunaga F., Muta T. Molecular mechanism of hemolymph clotting system in Limulus. Thromb Res. 1992 Oct 1;68(1):1–32. doi: 10.1016/0049-3848(92)90124-s. [DOI] [PubMed] [Google Scholar]
- Iwanaga S., Miyata T., Tokunaga F., Muta T. Molecular mechanism of hemolymph clotting system in Limulus. Thromb Res. 1992 Oct 1;68(1):1–32. doi: 10.1016/0049-3848(92)90124-s. [DOI] [PubMed] [Google Scholar]
- Janeway C. A., Jr The immune system evolved to discriminate infectious nonself from noninfectious self. Immunol Today. 1992 Jan;13(1):11–16. doi: 10.1016/0167-5699(92)90198-G. [DOI] [PubMed] [Google Scholar]
- Jentoft N. Why are proteins O-glycosylated? Trends Biochem Sci. 1990 Aug;15(8):291–294. doi: 10.1016/0968-0004(90)90014-3. [DOI] [PubMed] [Google Scholar]
- Jomori T., Natori S. Function of the lipopolysaccharide-binding protein of Periplaneta americana as an opsonin. FEBS Lett. 1992 Jan 27;296(3):283–286. doi: 10.1016/0014-5793(92)80305-z. [DOI] [PubMed] [Google Scholar]
- Kodama T., Freeman M., Rohrer L., Zabrecky J., Matsudaira P., Krieger M. Type I macrophage scavenger receptor contains alpha-helical and collagen-like coiled coils. Nature. 1990 Feb 8;343(6258):531–535. doi: 10.1038/343531a0. [DOI] [PubMed] [Google Scholar]
- Krieger M., Acton S., Ashkenas J., Pearson A., Penman M., Resnick D. Molecular flypaper, host defense, and atherosclerosis. Structure, binding properties, and functions of macrophage scavenger receptors. J Biol Chem. 1993 Mar 5;268(7):4569–4572. [PubMed] [Google Scholar]
- Krieger M., Herz J. Structures and functions of multiligand lipoprotein receptors: macrophage scavenger receptors and LDL receptor-related protein (LRP). Annu Rev Biochem. 1994;63:601–637. doi: 10.1146/annurev.bi.63.070194.003125. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Libermann T. A., Friesel R., Jaye M., Lyall R. M., Westermark B., Drohan W., Schmidt A., Maciag T., Schlessinger J. An angiogenic growth factor is expressed in human glioma cells. EMBO J. 1987 Jun;6(6):1627–1632. doi: 10.1002/j.1460-2075.1987.tb02410.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin H. Y., Kaji E. H., Winkel G. K., Ives H. E., Lodish H. F. Cloning and functional expression of a vascular smooth muscle endothelin 1 receptor. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3185–3189. doi: 10.1073/pnas.88.8.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin H. Y., Kaji E. H., Winkel G. K., Ives H. E., Lodish H. F. Cloning and functional expression of a vascular smooth muscle endothelin 1 receptor. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3185–3189. doi: 10.1073/pnas.88.8.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin C. H., Mayeda C. A., Meyerowitz E. M. Evolution and expression of the Sgs-3 glue gene of Drosophila. J Mol Biol. 1988 May 20;201(2):273–287. doi: 10.1016/0022-2836(88)90138-6. [DOI] [PubMed] [Google Scholar]
- Nelson R. E., Fessler L. I., Takagi Y., Blumberg B., Keene D. R., Olson P. F., Parker C. G., Fessler J. H. Peroxidasin: a novel enzyme-matrix protein of Drosophila development. EMBO J. 1994 Aug 1;13(15):3438–3447. doi: 10.1002/j.1460-2075.1994.tb06649.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson A. M., Rich A., Krieger M. Polynucleotide binding to macrophage scavenger receptors depends on the formation of base-quartet-stabilized four-stranded helices. J Biol Chem. 1993 Feb 15;268(5):3546–3554. [PubMed] [Google Scholar]
- Penman M., Lux A., Freedman N. J., Rohrer L., Ekkel Y., McKinstry H., Resnick D., Krieger M. The type I and type II bovine scavenger receptors expressed in Chinese hamster ovary cells are trimeric proteins with collagenous triple helical domains comprising noncovalently associated monomers and Cys83-disulfide-linked dimers. J Biol Chem. 1991 Dec 15;266(35):23985–23993. [PubMed] [Google Scholar]
- Penman M., Lux A., Freedman N. J., Rohrer L., Ekkel Y., McKinstry H., Resnick D., Krieger M. The type I and type II bovine scavenger receptors expressed in Chinese hamster ovary cells are trimeric proteins with collagenous triple helical domains comprising noncovalently associated monomers and Cys83-disulfide-linked dimers. J Biol Chem. 1991 Dec 15;266(35):23985–23993. [PubMed] [Google Scholar]
- Perkins S. J., Haris P. I., Sim R. B., Chapman D. A study of the structure of human complement component factor H by Fourier transform infrared spectroscopy and secondary structure averaging methods. Biochemistry. 1988 May 31;27(11):4004–4012. doi: 10.1021/bi00411a017. [DOI] [PubMed] [Google Scholar]
- Perkins S. J., Haris P. I., Sim R. B., Chapman D. A study of the structure of human complement component factor H by Fourier transform infrared spectroscopy and secondary structure averaging methods. Biochemistry. 1988 May 31;27(11):4004–4012. doi: 10.1021/bi00411a017. [DOI] [PubMed] [Google Scholar]
- Pinna L. A. Casein kinase 2: an 'eminence grise' in cellular regulation? Biochim Biophys Acta. 1990 Sep 24;1054(3):267–284. doi: 10.1016/0167-4889(90)90098-x. [DOI] [PubMed] [Google Scholar]
- Reid K. B., Day A. J. Structure-function relationships of the complement components. Immunol Today. 1989 Jun;10(6):177–180. doi: 10.1016/0167-5699(89)90317-4. [DOI] [PubMed] [Google Scholar]
- Rohrer L., Freeman M., Kodama T., Penman M., Krieger M. Coiled-coil fibrous domains mediate ligand binding by macrophage scavenger receptor type II. Nature. 1990 Feb 8;343(6258):570–572. doi: 10.1038/343570a0. [DOI] [PubMed] [Google Scholar]
- Rohrer L., Freeman M., Kodama T., Penman M., Krieger M. Coiled-coil fibrous domains mediate ligand binding by macrophage scavenger receptor type II. Nature. 1990 Feb 8;343(6258):570–572. doi: 10.1038/343570a0. [DOI] [PubMed] [Google Scholar]
- Samakovlis C., Kimbrell D. A., Kylsten P., Engström A., Hultmark D. The immune response in Drosophila: pattern of cecropin expression and biological activity. EMBO J. 1990 Sep;9(9):2969–2976. doi: 10.1002/j.1460-2075.1990.tb07489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambrano G. R., Parthasarathy S., Steinberg D. Recognition of oxidatively damaged erythrocytes by a macrophage receptor with specificity for oxidized low density lipoprotein. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):3265–3269. doi: 10.1073/pnas.91.8.3265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg D., Parthasarathy S., Carew T. E., Khoo J. C., Witztum J. L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. 1989 Apr 6;320(14):915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- Steinberg D., Parthasarathy S., Carew T. E., Khoo J. C., Witztum J. L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. 1989 Apr 6;320(14):915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- Tepass U., Fessler L. I., Aziz A., Hartenstein V. Embryonic origin of hemocytes and their relationship to cell death in Drosophila. Development. 1994 Jul;120(7):1829–1837. doi: 10.1242/dev.120.7.1829. [DOI] [PubMed] [Google Scholar]
- Toribara N. W., Roberton A. M., Ho S. B., Kuo W. L., Gum E., Hicks J. W., Gum J. R., Jr, Byrd J. C., Siddiki B., Kim Y. S. Human gastric mucin. Identification of a unique species by expression cloning. J Biol Chem. 1993 Mar 15;268(8):5879–5885. [PubMed] [Google Scholar]
- Toribara N. W., Roberton A. M., Ho S. B., Kuo W. L., Gum E., Hicks J. W., Gum J. R., Jr, Byrd J. C., Siddiki B., Kim Y. S. Human gastric mucin. Identification of a unique species by expression cloning. J Biol Chem. 1993 Mar 15;268(8):5879–5885. [PubMed] [Google Scholar]
- White K., Grether M. E., Abrams J. M., Young L., Farrell K., Steller H. Genetic control of programmed cell death in Drosophila. Science. 1994 Apr 29;264(5159):677–683. doi: 10.1126/science.8171319. [DOI] [PubMed] [Google Scholar]
- Woodgett J. R., Gould K. L., Hunter T. Substrate specificity of protein kinase C. Use of synthetic peptides corresponding to physiological sites as probes for substrate recognition requirements. Eur J Biochem. 1986 Nov 17;161(1):177–184. doi: 10.1111/j.1432-1033.1986.tb10139.x. [DOI] [PubMed] [Google Scholar]
- Yamada H., Watanabe K., Shimonaka M., Yamaguchi Y. Molecular cloning of brevican, a novel brain proteoglycan of the aggrecan/versican family. J Biol Chem. 1994 Apr 1;269(13):10119–10126. [PubMed] [Google Scholar]
- Yamada H., Watanabe K., Shimonaka M., Yamaguchi Y. Molecular cloning of brevican, a novel brain proteoglycan of the aggrecan/versican family. J Biol Chem. 1994 Apr 1;269(13):10119–10126. [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]