ABSTRACT
CD4+ T-cell responses are crucial for effective antibody and CD8+ T-cell induction following virus infection. However, virus-specific CD4+ T cells can be preferential targets for human immunodeficiency virus (HIV) infection. HIV-specific CD4+ T-cell induction by vaccination may thus result in enhancement of virus replication following infection. In the present study, we show that vaccine-elicited CD4+ T cells expressing CD107a are relatively resistant to depletion in a macaque AIDS model. Comparison of virus-specific CD107a, macrophage inflammatory protein-1β, gamma interferon, tumor necrosis factor alpha, and interleukin-2 responses in CD4+ T cells of vaccinated macaques prechallenge and 1 week postchallenge showed a significant reduction in the CD107a− but not the CD107a+ subset after virus exposure. Those vaccinees that failed to control viremia showed a more marked reduction and exhibited significantly higher viral loads at week 1 than unvaccinated animals. Our results indicate that vaccine-induced CD107a− CD4+ T cells are depleted following virus infection, suggesting a rationale for avoiding virus-specific CD107a− CD4+ T-cell induction in HIV vaccine design.
IMPORTANCE Induction of effective antibody and/or CD8+ T-cell responses is a principal vaccine strategy against human immunodeficiency virus (HIV) infection. CD4+ T-cell responses are crucial for effective antibody and CD8+ T-cell induction. However, virus-specific CD4+ T cells can be preferential targets for HIV infection. Here, we show that vaccine-induced virus-specific CD107a− CD4+ T cells are largely depleted following infection in a macaque AIDS model. While CD4+ T-cell responses are important in viral control, our results indicate that virus-specific CD107a− CD4+ T-cell induction by vaccination may not lead to efficient CD4+ T-cell responses following infection but rather be detrimental and accelerate viral replication in the acute phase. This suggests that HIV vaccine design should avoid virus-specific CD107a− CD4+ T-cell induction. Conversely, this study found that vaccine-induced CD107a+ CD4+ T cells are relatively resistant to depletion following virus challenge, implying that induction of these cells may be an alternative approach toward HIV control.
INTRODUCTION
Virus-specific CD8+ T-cell responses play a central role in the control of human immunodeficiency virus (HIV) replication (1 – 6). CD8+ T cells, via their T-cell receptor, specifically recognize viral epitopes bound to human leukocyte antigen (HLA) class I molecules on the surface of virus-infected cells. Previous studies on HIV-infected individuals have shown an association of several HLA genotypes with delayed AIDS progression, implying possible HIV control by effective CD8+ T-cell responses (7 – 10). Current vaccine trials in macaque AIDS models with simian immunodeficiency virus (SIV) infection have shown that induction of effective CD8+ T-cell responses can result in reduction of postchallenge viral loads (11 – 16). Furthermore, cumulative studies have shown protection of SIV challenge by passive immunization with neutralizing antibody in macaques, suggesting the possibility of HIV protection by vaccine-induced effective antibodies (17 – 19).
Virus-specific CD4+ T-cell responses are crucial for induction of effective CD8+ T-cell and antibody responses (20 – 28). CD4+ T cells, however, are targets for HIV, which can be an obstacle to potent virus-specific CD4+ T-cell responses following HIV infection (29 – 31). Because HIV preferentially infects HIV-specific CD4+ T cells, induction of HIV-specific memory CD4+ T cells by vaccination may increase the target cell pool for HIV infection and thus enhance viral replication (32).
Our previous trial of a prophylactic vaccine regimen of a DNA prime and a boost with a Sendai virus (SeV) vector expressing SIV Gag (SeV-Gag) showed control of an SIV challenge in some vaccinated rhesus macaques (11). Vaccine-induced Gag-specific CD8+ T cells were shown to be responsible for this SIV control (33, 34). However, the effect of SIV-specific CD4+ T-cell induction by vaccination on postchallenge virus replication remains unclear. Virus-specific CD4+ T cells can be divided into multiple subsets producing a variety of cytokines following viral antigen stimulation (35, 36). In the present study, we examined changes in multiple subsets of vaccine-induced CD4+ T cells following SIV infection in a macaque AIDS model. Comparison of SIV-specific CD4+ T-cell profiles pre- and postchallenge indicated that vaccine-elicited CD4+ T cells expressing CD107a are relatively resistant to depletion whereas virus-specific CD107a− CD4+ T cells are largely depleted in the postchallenge acute phase of infection. These results imply that induction of the latter CD4+ T-cell subset by vaccination may result in enhanced HIV replication after virus exposure.
MATERIALS AND METHODS
Samples.
The present study used frozen peripheral blood mononuclear cell (PBMC) samples derived from 18 vaccinated and 21 unvaccinated Burmese rhesus macaques (Macaca mulatta) for analysis of SIV-specific CD4+ T-cell responses. Our previous SIVmac239 challenge experiments using these animals (34, 37 – 40) were conducted at the Tsukuba Primate Research Center, National Institute of Biomedical Innovation (NIBP), and the Institute for Virus Research, Kyoto University (IVRKU), with the help of the Corporation for Production and Research of Laboratory Primates. This study was approved by the Committee on the Ethics of Animal Experiments of NIBP and IVRKU under the guidelines for animal experiments at NIBP, IVRKU, and the National Institute of Infectious Diseases, which is in accordance with the Guidelines for Proper Conduct of Animal Experiments established by the Science Council of Japan (http://www.scj.go.jp/ja/info/kohyo/pdf/kohyo-20-k16-2e.pdf).
Vaccinated animals received a DNA prime and an SeV-Gag boost. The DNA used for the vaccination, CMV-SHIVdEN DNA (11), was constructed from an env- and nef-deleted simian-human immunodeficiency virus (SHIV) molecular clone DNA (SIVGP1) and has the genes encoding SIVmac239 Gag, Pol, Vif, Vpx, and a part of Vpr and HIV Tat and Rev. Animals received 5 mg of CMV-SHIVdEN DNA intramuscularly. Six weeks after the DNA prime, animals received a single boost intranasally with 1 × 108 cell infectious units (CIU) of replication-competent SeV-Gag (macaques R02-003, R02-012, R02-005, and R02-001) or 6 × 109 CIU of replication-defective F-deleted SeV-Gag (n = 14) (11, 41). There were no differences observed for CD4+ T-cell markers between animals receiving replication-competent boosts and those receiving replication-defective boosts. Vaccinated (3 months postboost) and unvaccinated animals were intravenously challenged with 1,000 50% tissue culture infective doses (TCID50) of SIVmac239 (42). In our previous study (34, 38), the geometric mean of viral loads at 6 months was approximately 2.5 × 105 copies/ml and the “M − 2 × SD” value (where M is the mean and SD is the standard deviation) of log-transformed viral loads was 3.2 (corresponding to 1.6 × 103 copies/ml) in unvaccinated animals possessing major histocompatibility complex class I (MHC-I) haplotype 90-120-Ie, which exhibit a typical course of SIV infection in Burmese rhesus macaques. Animals whose viral load at 6 months was less than 1.6 × 103 copies/ml were considered SIV controllers. The 21 unvaccinated animals included 17 with persistent viremia and 4 with undetectable or marginal levels of set-point plasma viral loads (see Fig. S1 in the supplemental material).
Analysis of SIV-specific CD4+ T-cell responses.
We examined SIV-specific induction of CD107a, macrophage inflammatory protein-1β (MIP-1β), gamma interferon (IFN-γ), tumor necrosis factor alpha (TNF-α), and interleukin-2 (IL-2) in CD4+ T cells as described previously (38, 43, 44). In brief, 5 × 105 PBMCs were prestimulated with 5 μg/ml immobilized anti-human CD28 (BD) and 5 μg/ml immobilized anti-human CD49d (Biolegend) in 96-well U-bottom plates at 37°C for 12 h, followed by coculture at 37°C for 6 h in the presence of Alexa Fluor 647-conjugated anti-human CD107a (Biolegend) with 1 × 105 autologous herpesvirus papio-immortalized B-lymphoblastoid cell lines (B-LCLs) infected with vesicular stomatitis virus G protein (VSV-G)-pseudotyped SIVGP1 for SIV-specific stimulation or mock B-LCLs for nonspecific stimulation. Monensin (final concentration, 0.7 μg/ml; BD) and brefeldin A (final concentration, 10 μg/ml; Sigma-Aldrich) were added to the culture 1 h after the start of coculture. The pseudotyped virus was obtained by cotransfection of 293T cells with a vesicular stomatitis virus G protein expression plasmid and an SIVGP1 DNA. SIV Gag capsid p27-positive cells detected by immunostaining were 5 to 10% of B-LCLs infected with VSV-G-pseudotyped SIVGP1. Immunostaining was performed using the Fix & Perm fixation and permeabilization kit (Invitrogen) and the following monoclonal antibodies: APC-Cy7-conjugated anti-nonhuman primate CD3 (BD), phycoerythrin (PE)-Texas Red-conjugated anti-human CD4 (Invitrogen), Alexa Fluor 700-conjugated anti-human CD8 (BD), PE-Cy7-conjugated anti-human IFN-γ (eBioscience), Pacific Blue-conjugated anti-human TNF-α (Biolegend), peridinin chlorophyll protein (PerCP)-Cy5.5-conjugated anti-human IL-2 (Biolegend), and PE-conjugated anti-human MIP-1β (BD). Dead cells were stained using the Live/Dead Fixable Dead Cell stain kit (Invitrogen).
Flow cytometric analysis was performed using FlowJo. Each subset positive for the marker of interest was determined in the dot plot gated by CD4+ T cells as shown in Fig. S2 in the supplemental material. The frequency of each subset of SIV-specific CD4+ T cells was calculated by subtracting the frequency after nonspecific stimulation from that after SIV-specific stimulation. As negative controls, we examined SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies in naive PBMCs derived from vaccinated (preprime; n = 13) and unvaccinated (prechallenge; n = 16) animals. The “M + 2 × SD” values of these negative controls, 0.031%, 0.034%, 0.028%, 0.017%, and 0.010%, were considered cutoff values for SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies, respectively. SIV-specific CD4+ T-cell frequencies less than 0.01% are shown as 0.01% in the figures, while statistical analyses were performed by using data in which values below the cutoff were set as zero. SIV-specific CD107a− CD4+ T-cell frequencies, shown in Fig. 1C, were calculated as the sum of the frequencies of CD107a− MIP-1β+, IFN-γ+, TNF-α+, or IL-2+ CD4+ T cells determined by Boolean gating. In our previous analyses (41, 45), SIV-specific IFN-γ+ CD4+ T-cell frequencies peaked 1 week after SeV-Gag boost and were largely reduced 1 week after the peak, followed by only a gradual, <2-fold decrease for a few months until challenge. In this study, SIV-specific stimulation was performed by coculture with the E/T (effector [PBMCs]/target [B-LCLs infected with VSV-G-pseudotyped SIVGP1]) ratio of 5:1, while stimulation with the E/T ratio of 2.5:1 was confirmed to induce similar levels of responses, implying that the E/T ratio of 5:1 is sufficient for the stimulation.
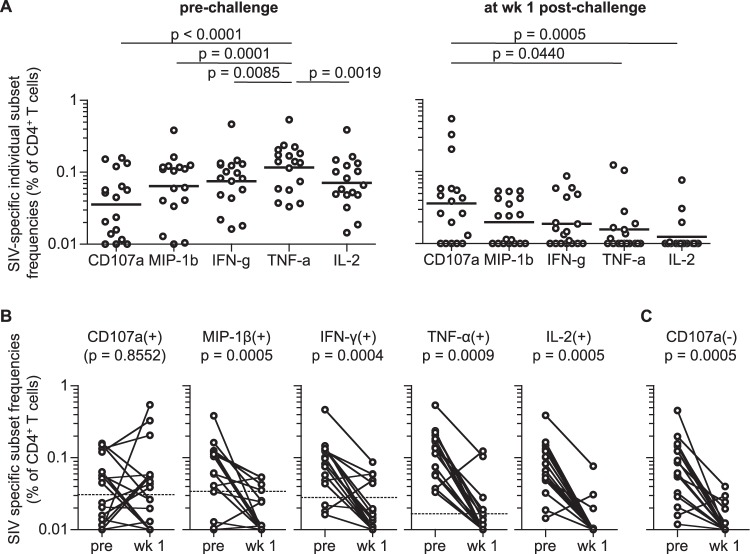
FIG 1.
SIV-specific CD4+ T-cell responses before and after SIV challenge in vaccinated macaques. (A) SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ frequencies in total CD4+ T cells at 1 or 2 months prechallenge (left panel [n = 17; samples of macaque R01-008 prechallenge were unavailable]) and 1 week postchallenge (right panel [n = 18]). TNF-α+ frequencies were significantly higher than those of any of the other four markers prechallenge, whereas CD107a+ frequencies were significantly higher than those of TNF-α+ and IL-2+ at week 1 postchallenge (Friedman's test and Wilcoxon signed-rank test). (B) Comparison of SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies prechallenge (pre) and at week 1 (wk 1) in vaccinated animals (n = 17). Cutoff values are indicated by dotted lines (see Materials and Methods). No significant change in SIV-specific CD107a+ CD4+ T-cell frequencies was observed, whereas other subset frequencies were significantly reduced following challenge (MIP-1β+, P = 0.0005; IFN-γ+, P = 0.0004; TNF-α+, P = 0.0009; IL-2+, P = 0.0005 by Wilcoxon signed-rank test). (C) Comparison of SIV-specific CD107a− CD4+ T-cell frequencies prechallenge and 1 week postchallenge in vaccinated macaques (n = 17). The prechallenge frequencies were significantly higher than those at week 1 (P = 0.0005 by Wilcoxon signed-rank test).
Statistical analysis.
Differences in two sets of measurements were examined by the Wilcoxon signed-rank test or the Mann-Whitney U test. Multiple comparisons of measurements were performed by Friedman's test and Wilcoxon signed-rank test with Bonferroni's multiple-comparison procedure or the Kruskal-Wallis test and Mann-Whitney U test with Bonferroni's multiple-comparison procedure. Correlation between T-cell frequencies and viral loads was analyzed by the Spearman's test. We set significance levels of all statistical tests at P values of <0.05.
RESULTS
SIV-specific CD4+ T-cell responses pre- and postchallenge in vaccinated macaques.
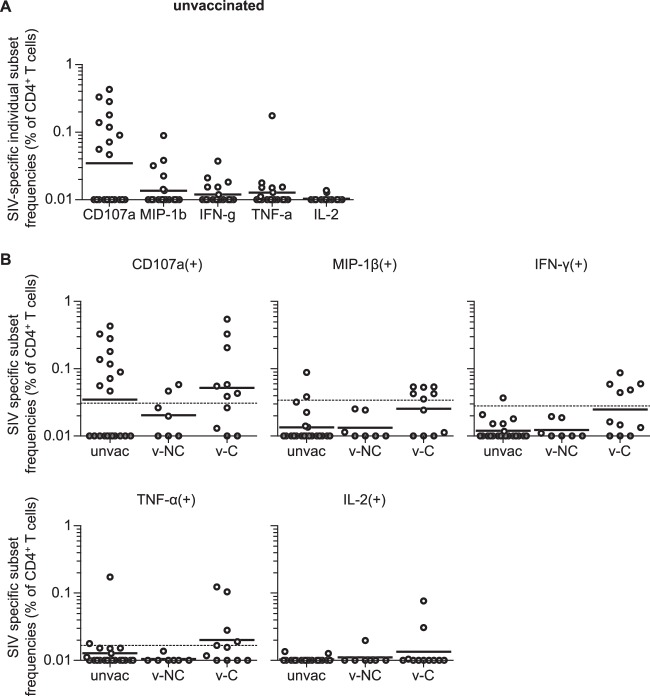
In the present study, we analyzed SIV-specific T-cell responses using frozen PBMC samples derived from 18 vaccinated and 21 unvaccinated Burmese rhesus macaques (see Fig. S1 in the supplemental material). These animals had been used in our previous SIVmac239 challenge experiments (34, 37 – 40). Vaccinated animals received a DNA prime and an SeV-Gag boost, followed by an SIVmac239 challenge at 3 months postboost. Eleven vaccinated animals, referred to as vaccinated controllers (v-C), showed undetectable or marginal levels of set-point plasma viral loads, whereas the remaining seven, referred to as vaccinated noncontrollers (v-NC), failed to control SIV replication (see Fig. S1 in the supplemental material).
We examined SIV-specific CD4+ T-cell responses by measurement of five markers, CD107a, MIP-1β, IFN-γ, TNF-α, and IL-2, after SIV-specific stimulation (35, 36, 38, 44). We used an env- and nef-deleted SHIV molecular clone DNA, SIVGP1, to measure the frequencies of T cells responding to SIVGP1-transduced cells (referred to as SIV-specific T cells) (11, 33). The DNA used for the prime and SIVGP1 both encode SIVmac239 Gag, Pol, Vif, Vpx, and a part of Vpr (see Materials and Methods). A representative gating schema for the flow cytometric analysis is shown in Fig. S2 in the supplemental material.
We first examined SIV-specific individual marker frequencies in total CD4+ T cells 1 or 2 months before and 1 week after SIVmac239 challenge in vaccinated macaques (Fig. 1A). Multiple comparisons among the five markers prechallenge revealed that SIV-specific TNF-α+ CD4+ T-cell frequencies were the highest while CD107a+ frequencies were the lowest. In contrast, SIV-specific CD4+ T cells postchallenge showed a different hierarchy of individual marker frequencies, with the highest being CD107a+ and IL-2+ the lowest.
We then compared pre- and postchallenge SIV-specific CD107a, MIP-1β, IFN-γ, TNF-α, and IL-2 responses in CD4+ T cells (Fig. 1B). Remarkably, frequencies of SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ subsets were significantly reduced following challenge (P = 0.0005, P = 0.0004, P = 0.0009, and P = 0.0005, respectively), but no significant reduction was observed in SIV-specific CD107a+ CD4+ T-cell frequencies. SIV-specific TNF-α+/IL-2+ CD4+ T-cell frequencies were above the cutoff values (see Materials and Methods) in all vaccinated animals at prechallenge but in only 4/17 at week 1 postchallenge. SIV-specific MIP-1β+/IFN-γ+ CD4+ T-cell frequencies were above the cutoff in 13/17 and 14/17 animals, respectively, prechallenge but in only 5/17 postchallenge. SIV-specific CD107a− CD4+ T-cell frequencies (CD107a− populations in SIV-specific MIP-1β+, IFN-γ+, TNF-α+, or IL-2+ CD4+ T cells) were significantly reduced (P = 0.0005) (Fig. 1C). In contrast, SIV-specific CD107a+ CD4+ T-cell responses were above the cutoff in nine vaccinees prechallenge and in nine postchallenge. These results indicate that SIV-specific CD4+ T cells producing MIP-1β, IFN-γ, TNF-α, and/or IL-2 are efficiently elicited by the DNA-prime/SeV-Gag-boost vaccination but are depleted in the acute phase postchallenge, whereas vaccine-elicited SIV-specific CD4+ T cells expressing CD107a are resistant to depletion following SIV infection.
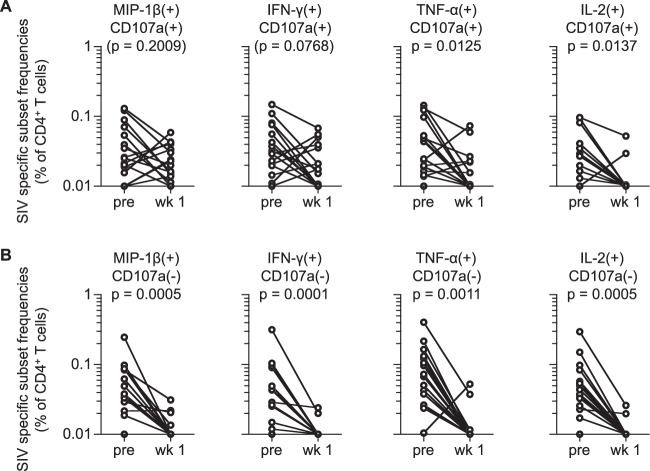
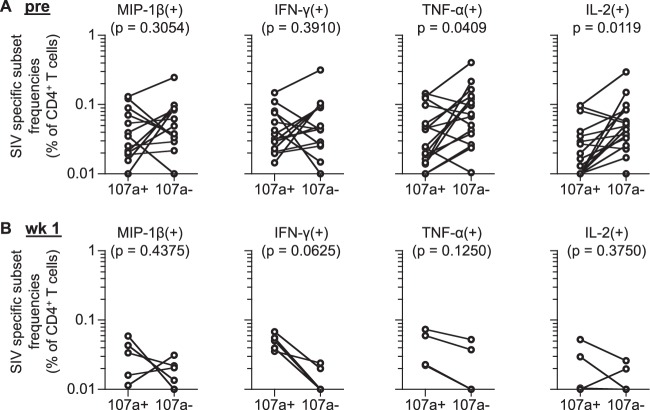
We further examined whether vaccine-elicited SIV-specific CD4+ T cells producing MIP-1β+, IFN-γ+, TNF-α+, or IL-2+ together with CD107a are resistant to depletion postchallenge. SIV-specific CD107a+ TNF-α+ and CD107a+ IL-2+ CD4+ T-cell frequencies were significantly reduced following SIV challenge (P = 0.0125 and P = 0.0137, respectively), whereas no significant reduction was observed in SIV-specific CD107a+ MIP-1β+ or CD107a+ IFN-γ+ CD4+ T-cell subset (Fig. 2A). SIV-specific CD107a− MIP-1β+, CD107a− IFN-γ+, CD107a− TNF-α+, and CD107a− IL-2+ CD4+ T-cell frequencies showed more profound and significant reductions following challenge (P = 0.0005, P = 0.0001, P = 0.0011, and P = 0.0005, respectively) (Fig. 2B). Comparison of CD107a+ and CD107a− populations in SIV-specific TNF-α+ and IL-2+ CD4+ T cells revealed that the latter (CD107a−) subset was higher at prechallenge (Fig. 3A) whereas the former (CD107a+) subset was predominant mostly in those that remained above the cutoff values at week 1 postchallenge (Fig. 3B). These results imply that vaccine-elicited CD4+ T cells producing these markers together with CD107a are relatively resistant to depletion following SIV challenge.
FIG 2.
SIV-specific CD107a+ and CD107a− CD4+ T-cell responses pre- and postchallenge in vaccinated macaques. (A) Comparison of SIV-specific MIP-1β+ CD107a+, IFN-γ+ CD107a+, TNF-α+ CD107a+, and IL-2+ CD107a+ CD4+ T-cell frequencies prechallenge and at week 1 postchallenge. No significant change in SIV-specific MIP-1β+ or IFN-γ+ CD107a+ CD4+ T-cell frequencies was observed, whereas frequencies of the other two subsets were significantly reduced following challenge (TNF-α+, P = 0.0125; IL-2+, P = 0.0137 by Wilcoxon signed-rank test). (B) Comparison of SIV-specific MIP-1β+ CD107a−, IFN-γ+ CD107a−, TNF-α+ CD107a−, and IL-2+ CD107a− CD4+ T-cell frequencies prechallenge and at week 1 postchallenge. All these frequencies were significantly reduced following challenge (MIP-1β+, P = 0.0005; IFN-γ+, P = 0.0001; TNF-α+, P = 0.0011; IL-2+, P = 0.0005 by Wilcoxon signed-rank test).
FIG 3.
Comparison of SIV-specific CD107a+ and CD107a− CD4+ T-cell responses in vaccinated macaques. (A) Comparison of frequencies of prechallenge SIV-specific CD4+ T-cell subsets inducing individual markers with (107a+) and without CD107a (107a−). Data for animals having SIV-specific MIP-1β+ (n = 13), IFN-γ+ (n = 14), TNF-α+ (n = 17), and IL-2+ (n = 17) CD4+ T-cell frequencies above individual cutoff values are shown. (B) Comparison of frequencies of postchallenge SIV-specific CD4+ T-cell subsets inducing individual markers with (107a+) and without CD107a (107a−). Data for animals having SIV-specific MIP-1β+ (n = 5), IFN-γ+ (n = 5), TNF-α+ (n = 4), and IL-2+ (n = 4) CD4+ T-cell frequencies above individual cutoff values are shown.
SIV-specific CD4+ T-cell responses pre- and postchallenge in vaccinated noncontrollers and controllers.
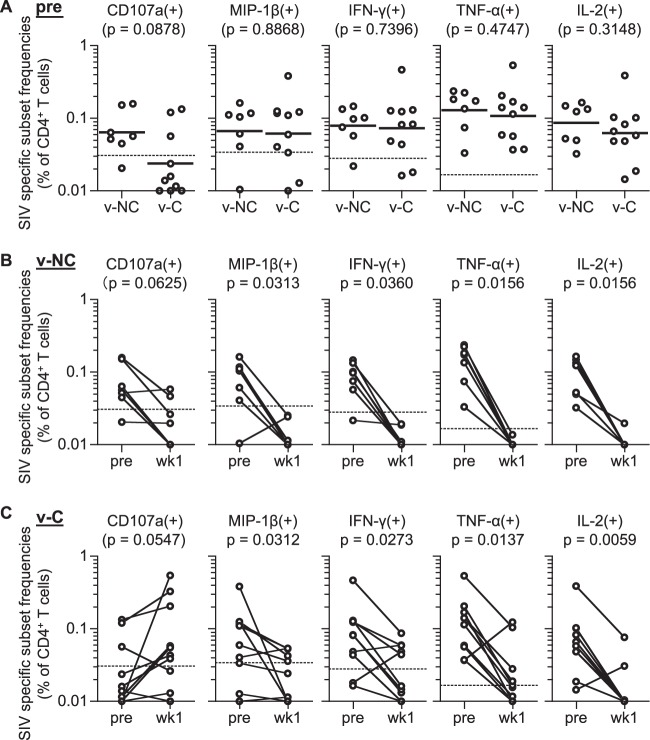
Next, we compared SIV-specific CD4+ T-cell responses in vaccinated noncontrollers (v-NC) and controllers (v-C). No significant difference was observed in SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, or IL-2+ CD4+ T-cell frequencies between these two groups before SIV challenge (Fig. 4A), indicating that prechallenge SIV-specific CD4+ T-cell responses are not the major determinant for SIV control in these vaccinated animals.
FIG 4.
SIV-specific CD4+ T-cell responses pre- and postchallenge in vaccinated noncontrollers (v-NC) and controllers (v-C). (A) Comparison of SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies prechallenge in v-NC (n = 7) and v-C (n = 10; samples of macaque R01-008 prechallenge were unavailable]). No significant difference was detected between the two groups for any of the 5 markers. (B) Comparison of SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies prechallenge and at week 1 postchallenge in v-NC. SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies were significantly reduced following challenge (MIP-1β+, P = 0.0313; IFN-γ+, P = 0.0360; TNF-α+, P = 0.0156; IL-2+, P = 0.0156 by Wilcoxon signed-rank test). (C) Comparison of SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies prechallenge and at week 1 postchallenge in v-C. SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T cells were significantly reduced following challenge (MIP-1β+, P = 0.0312; IFN-γ+, P = 0.0273; TNF-α+, P = 0.0137; IL-2+, P = 0.0059 by Wilcoxon signed-rank test).
In vaccinated noncontrollers, SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T cells were significantly reduced following SIV challenge, while reduction in SIV-specific CD107a+ CD4+ T cells was not significant (Fig. 4B). SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies at 1 week postchallenge were below the cutoff values in almost all noncontrollers, and even the CD107a+ subsets were below the cutoff in five of the seven. In contrast, SIV-specific CD107a+ CD4+ T-cell frequencies were not reduced but rather increased following challenge in vaccinated controllers; 7 of the 10 showed an increase in SIV-specific CD107a+ CD4+ T-cell responses (Fig. 4C). MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ subsets postchallenge were above the cutoff in 5/10, 5/10, 4/10, and 3/10, respectively, although significant reductions in these subset frequencies were observed. Thus, reductions in vaccine-elicited SIV-specific CD4+ T cells following SIV challenge were prominent in noncontrollers but not in controllers.
Comparison of SIV-specific CD4+ T-cell responses postchallenge in unvaccinated animals, vaccinated noncontrollers, and vaccinated controllers.
We then examined SIV-specific individual marker responses in CD4+ T cells at week 1 postinfection in unvaccinated macaques (Fig. 5A). Unvaccinated animals showed a higher frequency of SIV-specific CD107a+ CD4+ T cells than other markers, as seen in vaccinees at week 1 postchallenge (Fig. 1A), implying that the CD107a+ subset in unvaccinated animals may also be relatively resistant to depletion in the acute phase of SIV infection.
FIG 5.
SIV-specific CD4+ T-cell responses at week 1 postchallenge in unvaccinated and vaccinated macaques. (A) SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ frequencies in CD4+ T cells in unvaccinated macaques (n = 21). No significant difference was indicated by multiple comparisons (Friedman's test and Wilcoxon signed-rank test). (B) SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T-cell frequencies in unvaccinated animals (unvac; n = 21), vaccinated noncontrollers (v-NC; n = 7), and vaccinated controllers (v-C; n = 11).
Next, we compared SIV-specific CD4+ T-cell responses at 1 week postchallenge in unvaccinated animals, vaccinated noncontrollers, and vaccinated controllers (Fig. 5B). No significant difference in SIV-specific CD107a+ CD4+ T-cell responses was observed among these groups, but there was a trend for a lower frequency of this subset in vaccinated noncontrollers. SIV-specific CD107a+ CD4+ T-cell frequencies were above the cutoff values in 10 of 21 unvaccinated animals and 7 of 11 vaccinated controllers but only in 2 of 7 vaccinated noncontrollers. SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T cells were below the cutoff in almost all vaccinated noncontrollers. Thus, SIV-specific CD4+ T-cell depletion occurred primarily following SIV challenge in vaccinated noncontrollers.
Comparison of plasma viral loads in the acute phase in unvaccinated, vaccinated noncontroller, and controller groups.
Finally, we compared plasma viral loads in the acute phase in unvaccinated, vaccinated noncontroller, and controller groups. Interestingly, vaccinated noncontrollers showed significantly higher viral loads at week 1 than unvaccinated as well as vaccinated controllers (Fig. 6A). Even compared to the unvaccinated noncontrollers, vaccinated noncontrollers had significantly higher viral loads at week 1 (Fig. 6B). Unvaccinated but not vaccinated animals showed a significant increase in viral loads from week 1 to week 2 postchallenge (Fig. 6C), indicating that viral loads peaked earlier in vaccinated macaques. At week 2, unvaccinated animals had viral loads that were at levels similar to those of vaccinated noncontrollers but significantly higher than those of vaccinated controllers (Fig. 6A). These results suggest a higher acceleration of viral replication in the acute phase following SIV infection in vaccinated noncontrollers than in unvaccinated animals.
FIG 6.
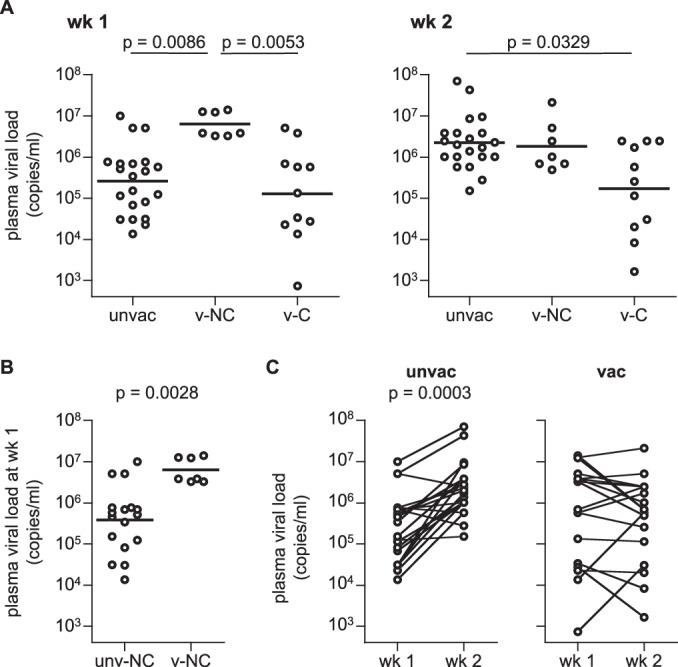
Plasma viral loads at weeks 1 and 2 after SIVmac239 challenge. (A) Comparison of viral loads in unvaccinated animals (unvac), vaccinated noncontrollers (v-NC), and vaccinated controllers (v-C) at weeks 1 (left panel) and 2 (right panel). Multiple comparisons (Kruskal-Wallis test and Mann-Whitney U test) indicated significantly higher viral loads at week 1 in v-NC than unvac and v-C (P = 0.0086 and P = 0.0053, respectively) and significantly lower viral loads at week 2 in v-C than unvac (P = 0.0329). (B) Comparison of viral loads at week 1 between unvaccinated noncontrollers (unv-NC; n = 17) and vaccinated noncontrollers (v-NC; n = 7). The load for the latter set was significantly higher than for the former (P = 0.0028 by Mann-Whitney U test). (C) Comparison of viral loads between weeks 1 and 2 in unvaccinated (left panel) and vaccinated (right panel) animals. Unvaccinated animals showed significantly higher viral loads at week 2 than week 1 (P = 0.0003 by Wilcoxon signed-rank test).
DISCUSSION
Virus-specific CD4+ T-cell responses are crucial for induction of effective antibody and CD8+ T-cell responses against virus infection. Current vaccine strategies include induction of neutralizing antibody and/or CD8+ T-cell responses, which are accompanied by CD4+ T-cell induction. Vaccine-induced CD4+ T cells, however, can be the preferential targets for HIV/SIV infection. In the present study, we found that vaccine-elicited SIV-specific CD107a− CD4+ T cells are depleted in the acute phase of infection after SIV challenge. In contrast, our results indicate that SIV-specific CD4+ T cells expressing CD107a are relatively resistant to depletion following infection.
HIV is known to preferentially infect HIV-specific CD4+ T cells (32). Our results present the basis of this preference. However, the mechanism of relative resistance of the CD107a+ population in HIV/SIV-specific CD4+ T cells to depletion following infection remains undetermined. Analysis using PBMCs found no significant difference in CCR5+ frequencies among SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T cells (see Fig. S3 in the supplemental material). CD107a+ subset frequencies were the lowest among the five markers after vaccination (Fig. 1A), and if this subset's responses were also lower following infection, it may contribute to lower sensitivity to depletion. It is difficult, however, to examine in vitro SIV infection and T-cell responses under the conditions exactly reflecting what occurs in vivo. It is also difficult to determine the possibility of changes in SIV-specific CD4+ T-cell function following infection.
It has been reported that virus-specific CD107a expression in CD4+ T cells is associated with cytotoxic CD4+ T-cell function via cytotoxic granules (46 – 49), which may confer resistance. Virus-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ rather than CD107a+ CD4+ T cells are believed to be important for helper function (27, 36). In particular, IFN-γ is an important marker for TH1 cells. However, our results indicate that vaccine-induced CD4+ T cells producing MIP-1β, IFN-γ, TNF-α, or IL-2 are largely depleted following SIV challenge. SIV-specific TNF-α+ and IL-2+ populations decreased postchallenge even in CD107a+ CD4+ T cells, suggesting that these TNF-α and IL-2 responses may confer higher sensitivity to depletion on CD4+ T cells. Nevertheless, the reduction of the CD107a+ population postchallenge was less prominent than that of CD107a− in SIV-specific TNF-α+ and IL-2+ CD4+ T cells. Furthermore, the CD107a+ population of SIV-specific MIP-1β+ or IFN-γ+ CD4+ T cells showed no significant reduction postchallenge. These results imply that the CD107a+ subset of vaccine-elicited CD4+ T cells with helper function may be relatively resistant to depletion following HIV/SIV infection.
Our previous studies (33, 34, 39) showed that vaccine-induced Gag-specific CD8+ T-cell responses are responsible for the control of SIV replication in the vaccinated controllers used in the present study. No significant difference in prechallenge SIV-specific CD4+ T-cell responses was observed between vaccinated controllers and noncontrollers, supporting a notion that vaccine-induced CD4+ T-cell responses are not the determinant for SIV control in these animals. There was no correlation between prechallenge SIV-specific CD107a+, MIP-1β+, IFN-γ+, TNF-α+, or IL-2+ CD4+ T-cell frequencies and viral loads at week 1. However, the noncontrollers showed a larger reduction in SIV-specific CD4+ T cells following SIV challenge and higher plasma viral loads at week 1 than the controllers. Even the CD107a+ as well as IFN-γ+ subset frequencies at week 1 were inversely correlated with viral loads at week 1 postchallenge in vaccinated animals (Fig. 7). These results imply that the reduction of vaccine-induced SIV-specific CD4+ T cells reflects killing of these cells by SIV within 1 week postchallenge. Vaccine-induced CD4+ T cells would be subjected to the killing without effectors such as CD8+ T cells, which protect these cells following infection.
FIG 7.
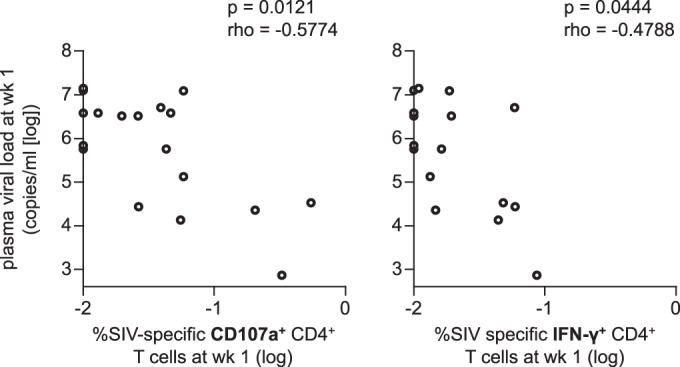
Correlation analysis between SIV-specific CD4+ T-cell frequencies at week 1 and plasma viral loads at week 1. SIV-specific CD107a+ (left panel) and IFN-γ+ (right panel) CD4+ T-cell frequencies were inversely correlated with the viral loads (P = 0.0121, rho = −0.5774 for the left panel and P = 0.0444, rho = −0.4788 for the right panel by Spearman's test).
SIV-specific MIP-1β+, IFN-γ+, TNF-α+, and IL-2+ CD4+ T cells were mostly depleted at week 1 in vaccinated noncontrollers. We found that viral loads peaked earlier in vaccinated than in unvaccinated animals. Furthermore, vaccinated noncontrollers that showed depletion of vaccine-elicited CD4+ T cells had significantly higher viral loads at week 1 than unvaccinated animals. While virus-specific CD4+ T-cell responses are important in viral control (50 – 52), our results suggest that induction of virus-specific CD4+ T cells, especially CD107a− cells, by vaccination may not lead to efficient CD4+ T-cell responses following infection but rather enhance or accelerate viral replication in the early acute phase after HIV/SIV exposure. It is speculated that vaccinated controllers elicited highly effective CD8+ T-cell responses, which could overwhelm this enhanced viral replication. Without this enhancement, however, such highly potent effectors may not be required for HIV/SIV control. Thus, it would be reasonable to develop a vaccine to induce effective responses without inducing HIV-specific memory CD107a− CD4+ T cells. Indeed, our previous study suggested that vaccine induction of epitope-specific CD8+ T cells with the help of SeV-specific but not SIV-specific CD4+ T cells can result in effective CD8+ T-cell responses against SIV infection in the acute phase postchallenge (53). Alternatively, induction of HIV-specific CD107a+ CD4+ T cells may be a promising HIV vaccine approach, although the strategy for induction of these cells remains unknown (27, 54).
In summary, this study found that vaccine-elicited SIV-specific CD4+ T cells expressing CD107a are relatively resistant to depletion following infection in a macaque AIDS model. In contrast, our analysis revealed massive depletion of SIV-specific CD107a− CD4+ T cells following SIV exposure. These results suggest a rationale for vaccine design to elicit effective antibody or CD8+ T-cell responses without induction of HIV-specific CD107a− CD4+ T cells toward HIV control.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by a grant-in-aid from the Ministry of Education, Culture, Sports, Science, and Technology in Japan and grants-in-aid from the Ministry of Health, Labor, and Welfare in Japan.
We thank F. Ono, K. Oto, K. Hanari, S. Okabayashi, H. Akari, Y. Yasutomi, T. Miura, and Y. Koyanagi for their assistance in animal experiments. We also thank M. de Souza for his comments on the manuscript.
Footnotes
Published ahead of print 1 October 2014
Supplemental material for this article may be found at http://dx.doi.org/10.1128/JVI.02032-14.
REFERENCES
- 1.Koup RA, Safrit JT, Cao Y, Andrews CA, McLeod G, Borkowsky W, Farthing C, Ho DD. 1994. Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J. Virol. 68:4650–4655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Borrow P, Lewicki H, Hahn BH, Shaw GM, Oldstone MB. 1994. Virus-specific CD8+ cytotoxic T-lymphocyte activity associated with control of viremia in primary human immunodeficiency virus type 1 infection. J. Virol. 68:6103–6110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Matano T, Shibata R, Siemon C, Connors M, Lane HC, Martin MA. 1998. Administration of an anti-CD8 monoclonal antibody interferes with the clearance of chimeric simian/human immunodeficiency virus during primary infections of rhesus macaques. J. Virol. 72:164–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jin X, Bauer DE, Tuttleton SE, Lewin S, Gettie A, Blanchard J, Irwin CE, Safrit JT, Mittler J, Weinberger L, Kostrikis LG, Zhang L, Perelson AS, Ho DD. 1999. Dramatic rise in plasma viremia after CD8(+) T cell depletion in simian immunodeficiency virus-infected macaques. J. Exp. Med. 189:991–998. 10.1084/jem.189.6.991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schmitz JE, Kuroda MJ, Santra S, Sasseville VG, Simon MA, Lifton MA, Racz P, Tenner-Racz K, Dalesandro M, Scallon BJ, Ghrayeb J, Forman MA, Montefiori DC, Rieber EP, Letvin NL, Reimann KA. 1999. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science 283:857–860. 10.1126/science.283.5403.857. [DOI] [PubMed] [Google Scholar]
- 6.Goulder PJR, Watkins DI. 2004. HIV and SIV CTL escape: implications for vaccine design. Nat. Rev. Immunol. 4:630–640. 10.1038/nri1417. [DOI] [PubMed] [Google Scholar]
- 7.Migueles SA, Sabbaghian MS, Shupert WL, Bettinotti MP, Marincola FM, Martino L, Hallahan CW, Selig SM, Schwartz D, Sullivan J, Connors M. 2000. HLA B*5701 is highly associated with restriction of virus replication in a subgroup of HIV-infected long term nonprogressors. Proc. Natl. Acad. Sci. U. S. A. 97:2709–2714. 10.1073/pnas.050567397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Altfeld M, Addo MM, Rosenberg ES, Hecht FM, Lee PK, Vogel M, Yu XG, Draenert R, Johnston MN, Strick D, Allen TM, Feeney ME, Kahn JO, Sekaly RP, Levy JA, Rockstroh JK, Goulder PJ, Walker BD. 2003. Influence of HLA-B57 on clinical presentation and viral control during acute HIV-1 infection. AIDS 17:2581–2591. 10.1097/00002030-200312050-00005. [DOI] [PubMed] [Google Scholar]
- 9.Kiepiela P, Leslie AJ, Honeyborne I, Ramduth D, Thobakgale C, Chetty S, Rathnavalu P, Moore C, Pfafferott KJ, Hilton L, Zimbwa P, Moore S, Allen T, Brander C, Addo MM, Altfeld M, James I, Mallal S, Bunce M, Barber LD, Szinger J, Day C, Klenerman P, Mullins J, Korber B, Coovadia HM, Walker BD, Goulder PJ. 2004. Dominant influence of HLA-B in mediating the potential co-evolution of HIV and HLA. Nature 432:769–775. 10.1038/nature03113. [DOI] [PubMed] [Google Scholar]
- 10.Goulder PJR, Watkins DI. 2008. Impact of MHC class I diversity on immune control of immunodeficiency virus replication. Nat. Rev. Immunol. 8:619–630. 10.1038/nri2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matano T, Kobayashi M, Igarashi H, Takeda A, Nakamura H, Kano M, Sugimoto C, Mori K, Iida A, Hirata T, Hasegawa M, Yuasa T, Miyazawa M, Takahashi Y, Yasunami M, Kimura A, O'Connor DH, Watkins DI, Nagai Y. 2004. Cytotoxic T lymphocyte-based control of simian immunodeficiency virus replication in a preclinical AIDS vaccine trial. J. Exp. Med. 199:1709–1718. 10.1084/jem.20040432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Letvin NL, Mascola JR, Sun Y, Gorgone DA, Buzby AP, Xu L, Yang ZY, Chakrabarti B, Rao SS, Schmitz JE, Montefiori DC, Barker BR, Bookstein FL, Nabel GJ. 2006. Preserved CD4+ central memory T cells and survival in vaccinated SIV-challenged monkeys. Science 312:1530–1533. 10.1126/science.1124226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wilson NA, Reed J, Napoe GS, Piaskowski S, Szymanski A, Furlott J, Gonzalez EJ, Yant LJ, Maness NJ, May GE, Soma T, Reynolds MR, Rakasz E, Rudersdorf R, McDermott AB, O'Connor DH, Friedrich TC, Allison DB, Patki A, Picker LJ, Burton DR, Lin J, Huang L, Patel D, Heindecker G, Fan J, Citron M, Horton M, Wang F, Liang X, Shiver JW, Casimiro DR, Watkins DI. 2006. Vaccine-induced cellular immune responses reduce plasma viral concentrations after repeated low-dose challenge with pathogenic simian immunodeficiency virus SIVmac239. J. Virol. 80:5875–5885. 10.1128/JVI.00171-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu J, O'Brien KL, Lynch DM, Simmons NL, La Porte A, Riggs AM, Abbink P, Coffey RT, Grandpre LE, Seaman MS, Landucci G, Forthal DN, Montefiori DC, Carville A, Mansfield KG, Havenga MJ, Pau MG, Goudsmit J, Barouch DH. 2009. Immune control of an SIV challenge by a T-cell-based vaccine in rhesus monkeys. Nature 457:87–91. 10.1038/nature07469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hansen SG, Ford JC, Lewis MS, Ventura AB, Hughes CM, Coyne-Johnson L, Whizin N, Oswald K, Shoemaker R, Swanson T, Legasse AW, Chiuchiolo MJ, Parks CL, Axthelm MK, Nelson JA, Jarvis MA, Piatak M, Jr, Lifson JD, Picker LJ. 2011. Profound early control of highly pathogenic SIV by an effector memory T-cell vaccine. Nature 473:523–527. 10.1038/nature10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mudd PA, Martins MA, Ericsen AJ, Tully DC, Power KA, Bean AT, Piaskowski SM, Duan L, Seese A, Gladden AD, Weisgrau KL, Furlott JR, Kim YI, Veloso de Santana MG, Rakasz E, Capuano S, III, Wilson NA, Bonaldo MC, Galler R, Allison DB, Piatak M, Jr, Haase AT, Lifson JD, Allen TM, Watkins DI. 2012. Vaccine-induced CD8+ T cells control AIDS virus replication. Nature 491:129–133. 10.1038/nature11443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shibata R, Igarashi T, Haigwood N, Buckler-White A, Ogert R, Ross W, Willey R, Cho MW, Martin MA. 1999. Neutralizing antibody directed against the HIV-1 envelope glycoprotein can completely block HIV-1/SIV chimeric virus infections of macaque monkeys. Nat. Med. 5:204–210. 10.1038/5568. [DOI] [PubMed] [Google Scholar]
- 18.Mascola JR, Stiegler G, VanCott TC, Katinger H, Carpenter CB, Hanson CE, Beary H, Hayes D, Frankel SS, Birx DL, Lewis MG. 2000. Protection of macaques against vaginal transmission of a pathogenic HIV-1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 6:207–210. 10.1038/72318. [DOI] [PubMed] [Google Scholar]
- 19.Kwong PD, Mascola JR. 2012. Human antibodies that neutralize HIV-1: identification, structures, and B cell ontogenies. Immunity 37:412–425. 10.1016/j.immuni.2012.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Matloubian M, Concepcion RJ, Ahmed R. 1994. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J. Virol. 68:8056–8063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Janssen EM, Lemmens EE, Wolfe T, Christen U, von Herrath MG, Schoenberger SP. 2003. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421:852–856. 10.1038/nature01441. [DOI] [PubMed] [Google Scholar]
- 22.Shedlock DJ, Shen H. 2003. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300:337–339. 10.1126/science.1082305. [DOI] [PubMed] [Google Scholar]
- 23.Sun JC, Bevan MJ. 2003. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300:339–342. 10.1126/science.1083317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Elsaesser H, Sauer K, Brooks DG. 2009. IL-21 is required to control chronic viral infection. Science 324:1569–1572. 10.1126/science.1174182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yi JS, Du M, Zajac AJ. 2009. A vital role for interleukin-21 in the control of a chronic viral infection. Science 324:1572–1576. 10.1126/science.1175194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Frohlich A, Kisielow J, Schmitz I, Freigang S, Shamshiev AT, Weber J, Marsland BJ, Oxenius A, Kopf M. 2009. IL-21R on T cells is critical for sustained functionality and control of chronic viral infection. Science 324:1576–1580. 10.1126/science.1172815. [DOI] [PubMed] [Google Scholar]
- 27.Swain SL, McKinstry KK, Strutt TM. 2012. Expanding roles for CD4+ T cells in immunity to viruses. Nat. Rev. Immunol. 12:136–148. 10.1038/nri3152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Streeck H, D'Souza MP, Littman DR, Crotty S. 2013. Harnessing CD4(+) T cell responses in HIV vaccine development. Nat. Med. 19:143–149. 10.1038/nm.3054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Veazey RS, Tham IC, Mansfield KG, DeMaria M, Forand AE, Shvetz DE, Chalifoux LV, Sehgal PK, Lackner AA. 2000. Identifying the target cell in primary simian immunodeficiency virus (SIV) infection: highly activated memory CD4(+) T cells are rapidly eliminated in early SIV infection in vivo. J. Virol. 74:57–64. 10.1128/JVI.74.1.57-64.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mattapallil JJ, Douek DC, Hill B, Nishimura Y, Martin MA, Roederer M. 2005. Massive infection and loss of memory CD4+ T cells in multiple tissues during acute SIV infection. Nature 434:1093–1097. 10.1038/nature03501. [DOI] [PubMed] [Google Scholar]
- 31.Li Q, Duan L, Estes JD, Ma ZM, Rourke T, Wang Y, Reilly C, Carlis J, Miller CJ, Haase AT. 2005. Peak SIV replication in resting memory CD4+ T cells depletes gut lamina propria CD4+ T cells. Nature 434:1148–1152. 10.1038/nature03513. [DOI] [PubMed] [Google Scholar]
- 32.Douek DC, Brenchley JM, Betts MR, Ambrozak DR, Hill BJ, Okamoto Y, Casazza JP, Kuruppu J, Kunstman K, Wolinsky S, Grossman Z, Dybul M, Oxenius A, Price DA, Connors M, Koup RA. 2002. HIV preferentially infects HIV-specific CD4+ T cells. Nature 417:95–98. 10.1038/417095a. [DOI] [PubMed] [Google Scholar]
- 33.Kawada M, Tsukamoto T, Yamamoto H, Iwamoto N, Kurihara K, Takeda A, Moriya C, Takeuchi H, Akari H, Matano T. 2008. Gag-specific cytotoxic T-lymphocyte-based control of primary simian immunodeficiency virus replication in a vaccine trial. J. Virol. 82:10199–10206. 10.1128/JVI.01103-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Iwamoto N, Takahashi N, Seki S, Nomura T, Yamamoto H, Inoue M, Shu T, Naruse TK, Kimura A, Matano T. 2014. Control of SIV replication by vaccine-induced Gag- and Vif-specific CD8+ T cells. J. Virol. 88:425–433. 10.1128/JVI.02634-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Casazza JP, Betts MR, Price DA, Precopio ML, Ruff LE, Brenchley JM, Hill BJ, Roederer M, Douek DC, Koup RA. 2006. Acquisition of direct antiviral effector functions by CMV-specific CD4+ T lymphocytes with cellular maturation. J. Exp. Med. 203:2865–2877. 10.1084/jem.20052246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Seder RA, Darrah PA, Roederer M. 2008. T-cell quality in memory and protection: implications for vaccine design. Nat. Rev. Immunol. 8:247–258. 10.1038/nri2274. [DOI] [PubMed] [Google Scholar]
- 37.Kawada M, Tsukamoto T, Yamamoto H, Takeda A, Igarashi H, Watkins DI, Matano T. 2007. Long-term control of simian immunodeficiency virus replication with central memory CD4+ T-cell preservation after non-sterile protection by a cytotoxic T lymphocyte-based vaccine. J. Virol. 81:5202–5211. 10.1128/JVI.02881-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nomura T, Yamamoto H, Shiino T, Takahashi N, Nakane T, Iwamoto N, Ishii H, Tsukamoto T, Kawada M, Matsuoka S, Takeda A, Terahara K, Tsunetsugu-Yokota Y, Iwata-Yoshikawa N, Hasegawa H, Sata T, Naruse TK, Kimura A, Matano T. 2012. Association of major histocompatibility complex class I haplotypes with disease progression after simian immunodeficiency virus challenge in Burmese rhesus macaques. J. Virol. 86:6481–6490. 10.1128/JVI.07077-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Takahashi N, Nomura T, Takahara Y, Yamamoto H, Shiino T, Takeda A, Inoue M, Iida A, Hara H, Shu T, Hasegawa M, Sakawaki H, Miura T, Igarashi T, Koyanagi Y, Naruse TK, Kimura A, Matano T. 2013. A novel protective MHC-I haplotype not associated with dominant Gag-specific CD8+ T-cell responses in SIVmac239 infection of Burmese rhesus macaques. PLoS One 8:e54300. 10.1371/journal.pone.0054300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nakane T, Nomura T, Shi S, Nakamura M, Naruse TK, Kimura A, Matano T, Yamamoto H. 2013. Limited impact of passive non-neutralizing antibody immunization in acute SIV infection on viremia control in rhesus macaques. PLoS One 8:e73453. 10.1371/journal.pone.0073453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Takeda A, Igarashi H, Nakamura H, Kano M, Iida A, Hirata T, Hasegawa M, Nagai Y, Matano T. 2003. Protective efficacy of an AIDS vaccine, a single DNA priming followed by a single booster with a recombinant replication-defective Sendai virus vector, in a macaque AIDS model. J. Virol. 77:9710–9715. 10.1128/JVI.77.17.9710-9715.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kestler HW, III, Ringler DJ, Mori K, Panicali DL, Sehgal PK, Daniel MD, Desrosiers RC. 1991. Importance of the nef gene for maintenance of high virus loads and for development of AIDS. Cell 65:651–662. 10.1016/0092-8674(91)90097-I. [DOI] [PubMed] [Google Scholar]
- 43.Lamoreaux L, Roederer M, Koup R. 2006. Intracellular cytokine optimization and standard operating procedure. Nat. Protoc. 1:1507–1516. 10.1038/nprot.2006.268. [DOI] [PubMed] [Google Scholar]
- 44.Yamamoto T, Iwamoto N, Yamamoto H, Tsukamoto T, Kuwano T, Takeda A, Kawada M, Tsunetsugu-Yokota Y, Matano T. 2009. Polyfunctional CD4+ T-cell induction in neutralizing antibody-triggered control of simian immunodeficiency virus infection. J. Virol. 83:5514–5524. 10.1128/JVI.00145-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Matano T, Kano M, Nakamura H, Takeda A, Nagai Y. 2001. Rapid appearance of secondary immune responses and protection from acute CD4 depletion after a highly pathogenic immunodeficiency virus challenge in macaques vaccinated with a DNA prime/Sendai virus vector boost regimen. J. Virol. 75:11891–11896. 10.1128/JVI.75.23.11891-11896.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jellison ER, Kim SK, Welsh RM. 2005. MHC class II-restricted killing in vivo during viral infection. J. Immunol. 174:614–618. 10.4049/jimmunol.174.2.614. [DOI] [PubMed] [Google Scholar]
- 47.Zheng N, Fujiwara M, Ueno T, Oka S, Takiguchi M. 2009. Strong ability of Nef-specific CD4+ cytotoxic T cells to suppress human immunodeficiency virus type 1 (HIV-1) replication in HIV-1-infected CD4+ T cells and macrophages. J. Virol. 83:7668–7677. 10.1128/JVI.00513-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Soghoian DZ, Streeck H. 2010. Cytolytic CD4+ T cells in viral immunity. Expert Rev. Vaccines 9:1453–1463. 10.1586/erv.10.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Soghoian DZ, Jessen H, Flanders M, Sierra-Davidson K, Cutler S, Pertel T, Ranasinghe S, Lindqvist M, Davis I, Lane K, Rychert J, Rosenberg ES, Piechocka-Trocha A, Brass AL, Brenchley JM, Walker BD, Streeck H. 2012. HIV-specific cytolytic CD4 T cell responses during acute HIV infection predict disease outcome. Sci. Transl. Med. 4:123ra125. 10.1126/scitranslmed.3003165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rosenberg ES, Billingsley JM, Caliendo AM, Boswell SL, Sax PE, Kalams SA, Walker BD. 1997. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278:1447–1450. 10.1126/science.278.5342.1447. [DOI] [PubMed] [Google Scholar]
- 51.Ranasinghe S, Cutler S, Davis I, Lu R, Soghoian DZ, Qi Y, Sidney J, Kranias G, Flanders MD, Lindqvist M, Kuhl B, Alter G, Deeks SG, Walker BD, Gao X, Sette A, Carrington M, Streeck H. 2013. Association of HLA-DRB1-restricted CD4+ T cell responses with HIV immune control. Nat. Med. 19:930–933. 10.1038/nm.3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Schieffer M, Jessen H, Oster A, Pissani F, Soghoian DZ, Lu R, Jessen A, Zedlack C, Schultz B, Davis I, Ranasinghe S, Rosenberg ES, Alter G, Schumann R, Streeck H. 2014. Induction of Gag-specific CD4 T cell responses during acute HIV infection is associated with improved viral control. J. Virol. 88:7357–7366. 10.1128/JVI.00728-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tsukamoto T, Takeda A, Yamamoto T, Yamamoto H, Kawada M, Matano T. 2009. Impact of cytotoxic-T-lymphocyte memory induction without virus-specific CD4+ T-cell help on control of a simian immunodeficiency virus challenge in rhesus macaques. J. Virol. 83:9339–9346. 10.1128/JVI.01120-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hou S, Doherty PC, Zijlstra M, Jaenisch R, Katz JM. 1992. Delayed clearance of Sendai virus in mice lacking class I MHC-restricted CD8+ T cells. J. Immunol. 149:1319–1325. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.