Abstract
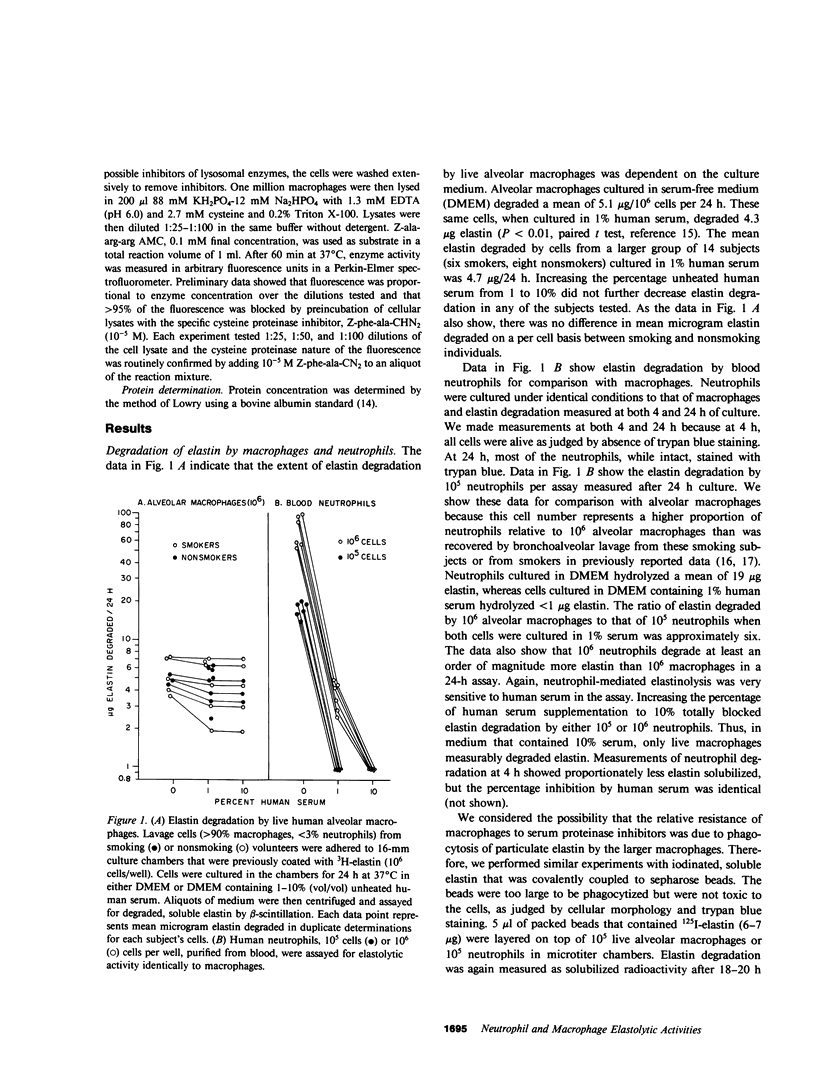
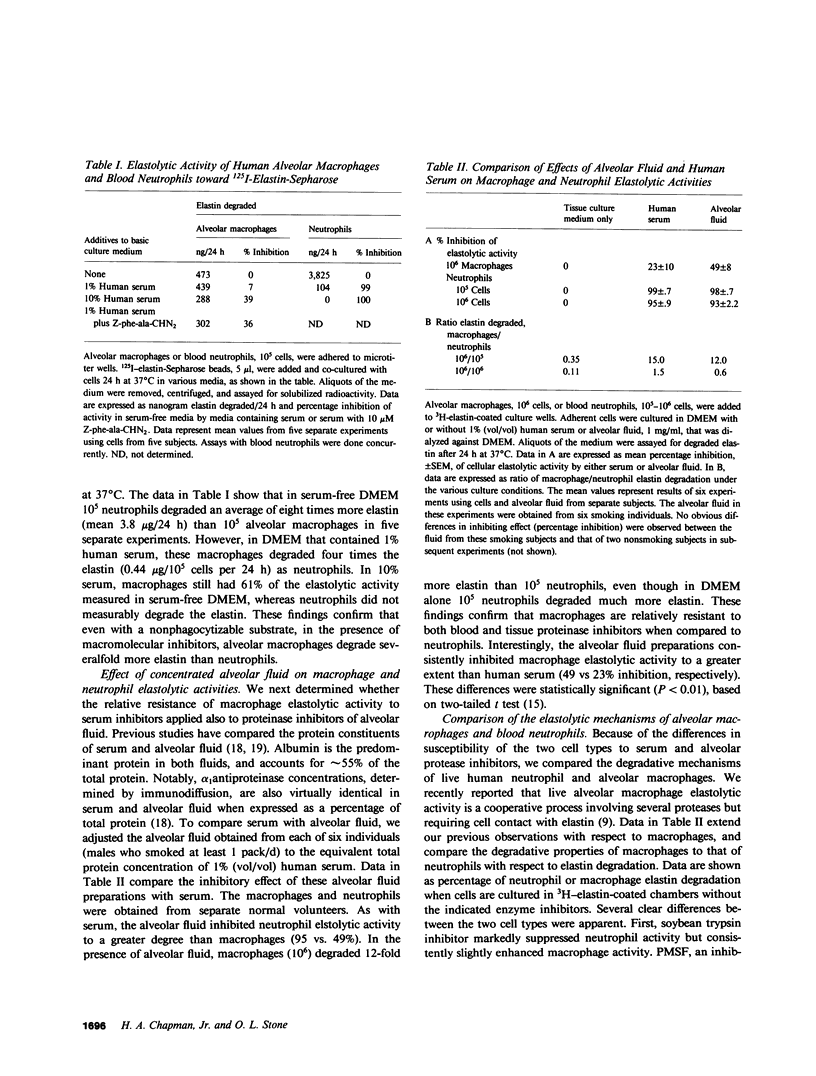
Elastin is an extracellular matrix protein critical to the normal structure and function of human lung. Recently reported data indicate that live human alveolar macrophages can degrade purified elastin in vitro. In this study, we directly compared the elastolytic activity of alveolar macrophages with that of human neutrophils. In the absence of proteinase inhibitors, human neutrophils degrade much more elastin than do human alveolar macrophages. However, macrophages cultured in 10% human serum and in contact with purified 3H-elastin degraded 4.7 micrograms elastin/10(6) cells per 24 h, as compared to less than 1 microgram/10(6) cells/24 h for neutrophils. We observed a similar pattern when the two cells were cultured in human alveolar fluid. We determined that the relative resistance of macrophage elastolytic activity to serum or alveolar proteinase inhibitors was not simply due to phagocytosis of substrate by the larger macrophages. Live macrophages as well as neutrophils degrade 125I-elastin coupled to noningestible sepharose beads. Again in serum-free media, neutrophils degraded eight-fold more elastin than macrophages but only macrophages degraded sepharose-coupled elastin in the presence of 10% serum. Because of these findings, we compared the enzymatic mechanisms of elastin breakdown by macrophages with that of neutrophils. Macrophage elastolytic activity is largely (65-80%) due to a cysteine proteinase(s), at least part of which is Cathepsin B. Approximately half of the cysteine proteinase activity appeared to be expressed at or near the cell surface. These experiments defined two enzymatically distinct pathways of elastin breakdown by human inflammatory cells: the classic, neutrophil derived soluble elastase(s) that is sensitive to serum and alveolar proteinase inhibitors, and a macrophage-mediated pathway that is largely cell associated and relatively resistant to inhibitors. The function of the two pathways depends on the relative excess or deficiency of soluble inhibitors. At inflammatory sites rich in proteinase inhibitors, tissue macrophages may degrade more extracellular matrix elastin than neutrophils. In smokers without antiproteinase deficiency, pulmonary macrophages, which are known to be increased in number, may be the more important cause of elastin breakdown and emphysema.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barrett A. J., Kirschke H. Cathepsin B, Cathepsin H, and cathepsin L. Methods Enzymol. 1981;80(Pt 100):535–561. doi: 10.1016/s0076-6879(81)80043-2. [DOI] [PubMed] [Google Scholar]
- Baugh R. J., Travis J. Human leukocyte granule elastase: rapid isolation and characterization. Biochemistry. 1976 Feb 24;15(4):836–841. doi: 10.1021/bi00649a017. [DOI] [PubMed] [Google Scholar]
- Bell D. Y., Haseman J. A., Spock A., McLennan G., Hook G. E. Plasma proteins of the bronchoalveolar surface of the lungs of smokers and nonsmokers. Am Rev Respir Dis. 1981 Jul;124(1):72–79. doi: 10.1164/arrd.1981.124.1.72. [DOI] [PubMed] [Google Scholar]
- Boudier C., Holle C., Bieth J. G. Stimulation of the elastolytic activity of leukocyte elastase by leukocyte cathepsin G. J Biol Chem. 1981 Oct 25;256(20):10256–10258. [PubMed] [Google Scholar]
- Burnett D., Crocker J., Stockley R. A. Cathepsin B-like cysteine proteinase activity in sputum and immunohistologic identification of cathepsin B in alveolar macrophages. Am Rev Respir Dis. 1983 Nov;128(5):915–919. doi: 10.1164/arrd.1983.128.5.915. [DOI] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L., Vavrin Z. Degradation of fibrin and elastin by intact human alveolar macrophages in vitro. Characterization of a plasminogen activator and its role in matrix degradation. J Clin Invest. 1984 Mar;73(3):806–815. doi: 10.1172/JCI111275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark R. A., Stone P. J., El Hag A., Calore J. D., Franzblau C. Myeloperoxidase-catalyzed inactivation of alpha 1-protease inhibitor by human neutrophils. J Biol Chem. 1981 Apr 10;256(7):3348–3353. [PubMed] [Google Scholar]
- Cogen R. B., Taubman S. B., Tuddenham E. G. Detachment of endothelial cells by polymorphonuclear leukocytes in vitro: potentiation by antibody coating and prevention by inflammatory exudate. J Periodontol. 1981 Nov;52(11):668–672. doi: 10.1902/jop.1981.52.11.668. [DOI] [PubMed] [Google Scholar]
- Eriksson S. Studies in alpha 1-antitrypsin deficiency. Acta Med Scand Suppl. 1965;432:1–85. [PubMed] [Google Scholar]
- Etherington D. J. Proteinases in connective tissue breakdown. Ciba Found Symp. 1979;(75):87–103. doi: 10.1002/9780470720585.ch6. [DOI] [PubMed] [Google Scholar]
- GROSS P., BABYAK M. A., TOLKER E., KASCHAK M. ENZYMATICALLY PRODUCED PULMONARY EMPHYSEMA; A PRELIMINARY REPORT. J Occup Med. 1964 Dec;6:481–484. [PubMed] [Google Scholar]
- Gadek J. E., Fells G. A., Zimmerman R. L., Rennard S. I., Crystal R. G. Antielastases of the human alveolar structures. Implications for the protease-antiprotease theory of emphysema. J Clin Invest. 1981 Oct;68(4):889–898. doi: 10.1172/JCI110344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Noriega A., Grubb J. H., Talkad V., Sly W. S. Chloroquine inhibits lysosomal enzyme pinocytosis and enhances lysosomal enzyme secretion by impairing receptor recycling. J Cell Biol. 1980 Jun;85(3):839–852. doi: 10.1083/jcb.85.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green G. D., Shaw E. Peptidyl diazomethyl ketones are specific inactivators of thiol proteinases. J Biol Chem. 1981 Feb 25;256(4):1923–1928. [PubMed] [Google Scholar]
- Hadler N. M., Spitznagel J. K., Quinet R. J. Lysosomal enzymes in inflammatory synovial effusions. J Immunol. 1979 Aug;123(2):572–577. [PubMed] [Google Scholar]
- Harlan J. M., Killen P. D., Harker L. A., Striker G. E., Wright D. G. Neutrophil-mediated endothelial injury in vitro mechanisms of cell detachment. J Clin Invest. 1981 Dec;68(6):1394–1403. doi: 10.1172/JCI110390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinman L. M., Stevens C. A., Matthay R. A., Gee J. B. Elastase and lysozyme activities in human alveolar macrophages. Effects of cigarette smoking. Am Rev Respir Dis. 1980 Feb;121(2):263–271. doi: 10.1164/arrd.1980.121.2.263. [DOI] [PubMed] [Google Scholar]
- Hunninghake G. W., Crystal R. G. Cigarette smoking and lung destruction. Accumulation of neutrophils in the lungs of cigarette smokers. Am Rev Respir Dis. 1983 Nov;128(5):833–838. doi: 10.1164/arrd.1983.128.5.833. [DOI] [PubMed] [Google Scholar]
- Johnson D., Travis J. The oxidative inactivation of human alpha-1-proteinase inhibitor. Further evidence for methionine at the reactive center. J Biol Chem. 1979 May 25;254(10):4022–4026. [PubMed] [Google Scholar]
- Kaplan P. D., Kuhn C., Pierce J. A. The induction of emphysema with elastase. I. The evolution of the lesion and the influence of serum. J Lab Clin Med. 1973 Sep;82(3):349–356. [PubMed] [Google Scholar]
- Kuhn C., Yu S. Y., Chraplyvy M., Linder H. E., Senior R. M. The induction of emphysema with elastase. II. Changes in connective tissue. Lab Invest. 1976 Apr;34(4):372–380. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lesser M., Chang J. C., Orlowski J., Kilburn K. H., Orlowski M. Cathepsin B and prolyl endopeptidase activity in rat peritoneal and alveolar macrophages. Stimulation of peritoneal macrophages by saline lavage. J Lab Clin Med. 1983 Feb;101(2):327–334. [PubMed] [Google Scholar]
- McConahey P. J., Dixon F. J. Radioiodination of proteins by the use of the chloramine-T method. Methods Enzymol. 1980;70(A):210–213. doi: 10.1016/s0076-6879(80)70050-2. [DOI] [PubMed] [Google Scholar]
- Merrill W. W., Goodenberger D., Strober W., Matthay R. A., Naegel G. P., Reynolds H. Y. Free secretory component and other proteins in human lung lavage. Am Rev Respir Dis. 1980 Jul;122(1):156–161. doi: 10.1164/arrd.1980.122.1.156. [DOI] [PubMed] [Google Scholar]
- Niewoehner D. E., Kleinerman J., Rice D. B. Pathologic changes in the peripheral airways of young cigarette smokers. N Engl J Med. 1974 Oct 10;291(15):755–758. doi: 10.1056/NEJM197410102911503. [DOI] [PubMed] [Google Scholar]
- Powers J. C., Gupton B. F., Harley A. D., Nishino N., Whitley R. J. Specificity of porcine pancreatic elastase, human leukocyte elastase and cathepsin G. Inhibition with peptide chloromethyl ketones. Biochim Biophys Acta. 1977 Nov 23;485(1):156–166. doi: 10.1016/0005-2744(77)90203-0. [DOI] [PubMed] [Google Scholar]
- Riches D. W., Morris C. J., Stanworth D. R. Induction of selective acid hydrolase release from mouse macrophages during exposure to chloroquine and quinine. Biochem Pharmacol. 1981 Mar 15;30(6):629–634. doi: 10.1016/0006-2952(81)90136-2. [DOI] [PubMed] [Google Scholar]
- Rifkin D. B., Crowe R. M. A sensitive assay for elastase employing radioactive elastin coupled to sepharose. Anal Biochem. 1977 May 1;79(1-2):268–275. doi: 10.1016/0003-2697(77)90402-x. [DOI] [PubMed] [Google Scholar]
- Sandhaus R. A., McCarthy K. M., Musson R. A., Henson P. M. Elastolytic proteinases of the human macrophage. Chest. 1983 May;83(5 Suppl):60S–62S. doi: 10.1378/chest.83.5_supplement.60s. [DOI] [PubMed] [Google Scholar]
- Showell H. J., Naccache P. H., Sha'afi R. I., Becker E. L. The effects of extracellular K+, Na+ and Ca++ on lysosomal enzyme secretion from polymorphonuclear leukocytes. J Immunol. 1977 Sep;119(3):804–811. [PubMed] [Google Scholar]
- Snider G. L., Sherter C. B., Koo K. W., Karlinsky J. B., Hayes J. A., Franzblau C. Respiratory mechanics in hamsters following treatment with endotracrael elastase or collagenase. J Appl Physiol Respir Environ Exerc Physiol. 1977 Feb;42(2):206–215. doi: 10.1152/jappl.1977.42.2.206. [DOI] [PubMed] [Google Scholar]
- Stone P. J., Calore J. D., McGowan S. E., Bernardo J., Snider G. L., Franzblau C. Functional alpha 1-protease inhibitor in the lower respiratory tract of cigarette smokers is not decreased. Science. 1983 Sep 16;221(4616):1187–1189. doi: 10.1126/science.6612333. [DOI] [PubMed] [Google Scholar]
- Vaes G. On the mechanisms of bone resorption. The action of parathyroid hormone on the excretion and synthesis of lysosomal enzymes and on the extracellular release of acid by bone cells. J Cell Biol. 1968 Dec;39(3):676–697. doi: 10.1083/jcb.39.3.676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WRIGHT R. R. Elastic tissue of normal and emphysematous lungs. A tridimensional histologic study. Am J Pathol. 1961 Sep;39:355–367. [PMC free article] [PubMed] [Google Scholar]
- Werb Z., Bainton D. F., Jones P. A. Degradation of connective tissue matrices by macrophages. III. Morphological and biochemical studies on extracellular, pericellular, and intracellular events in matrix proteolysis by macrophages in culture. J Exp Med. 1980 Dec 1;152(6):1537–1553. doi: 10.1084/jem.152.6.1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White R., Lin H. S., Kuhn C., 3rd Elastase secretion by peritoneal exudative and alveolar macrophages. J Exp Med. 1977 Sep 1;146(3):802–808. doi: 10.1084/jem.146.3.802. [DOI] [PMC free article] [PubMed] [Google Scholar]



